Clinical Benefit of Tamsulosin and the Hexanic Extract of Serenoa Repens, in Combination or as Monotherapy, in Patients with Moderate/Severe LUTS-BPH: A Subset Analysis of the QUALIPROST Study
Abstract
1. Introduction
2. Experimental Section
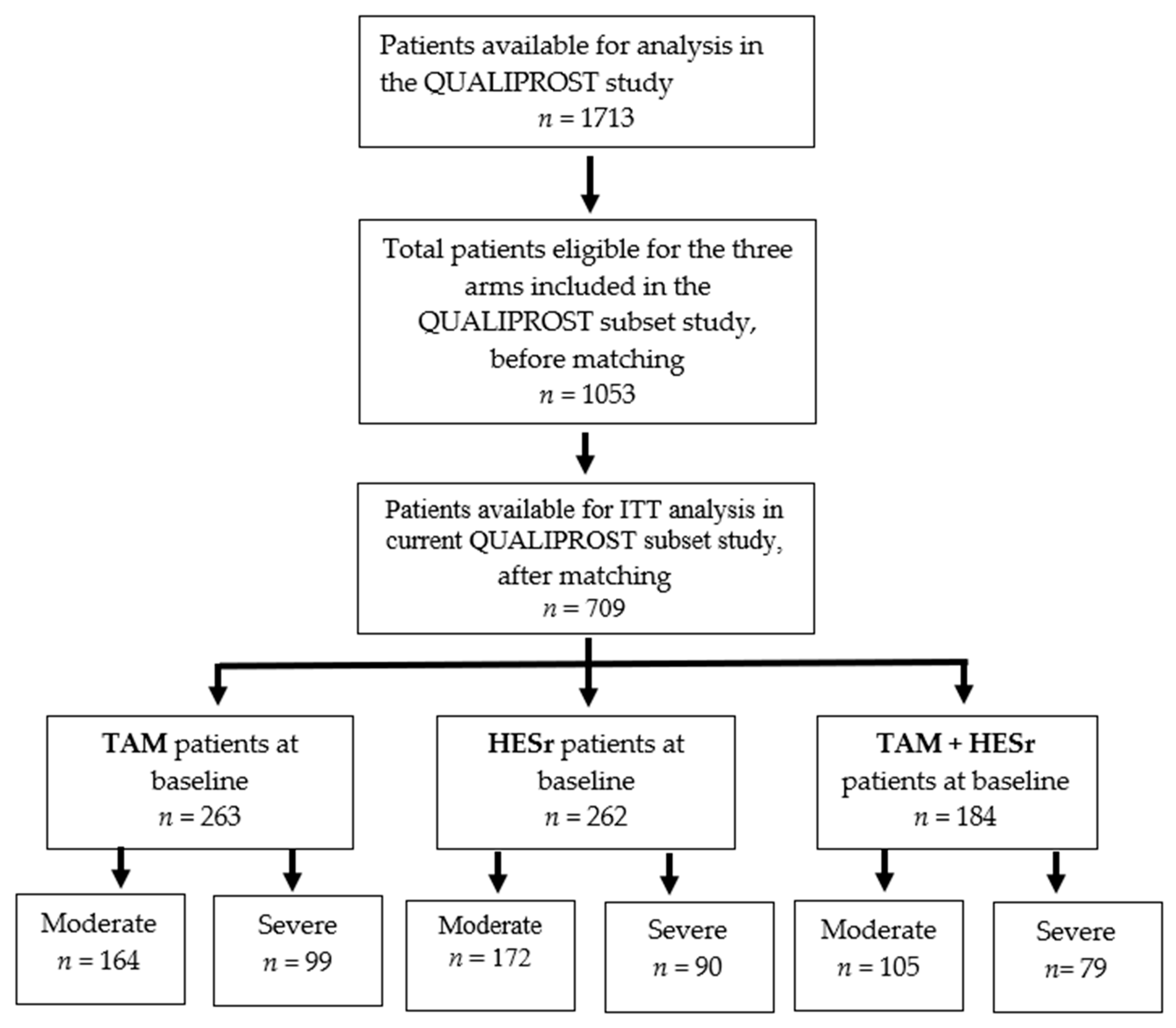
2.1. Patients and Study Design
2.2. Study Variables
2.3. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Ethical Approval
Appendix A
- A Coruña: Dr. L. Busto Castañón; Dr. J.E. Duarte Novo; Dr. M. Montes Couceiro; Dr. A. Rodríguez Alonso; Alicante: Dr. J.A. Canovas Ivorra; Dr. C. López López; Dr. L. Prieto Chaparro; Almería: Dr. F. Gómez Berjón; Dr. J. Hortelano Parras; Asturias: Dr. A. M. Huescar; Dr. P.L. Muntañola Armora; Badajoz: Dr. E. Godoy Rubio; Dr. J. Mariño del Real; Barcelona: Dr. J. Armona Mani; Dr. F.J. Blasco Casares; Dr. S. Bucar Terrades; Dr. Ll. Cortadellas Angel; Dr. J. Fernández Zuazu; Dr. J.M. Malet Carreras; Dr. S. Mando Dakkar; Dr. J.M. Prats Puig; Dr. M. Puyol Pallás; Dr. J.A. Romero Martín; Dr. J. Sáenz de Cabezón Martí; Dr. J. Sánchez Macías; Dr. C. Vargas Blasco; Dr. J.M. Vayreda Martija; Bilbao: Dr. J.A. Gallego Sánchez; Dr. N. Prieto Ugidos; Cádiz: Dr. J.M. Arroyo Maestre; Dr. S. Garrido Insua; Dr. A. Gutiérrez de Pablo; Dr. M. Marmol Ruíz; Dr. F. Reyes Martínez; Castellón: Dr. J. Beltrán Persiva; Dr. L. Monzonis Rey; Dr. J. Palacios Castañeda; Ciudad Real: Dr. N. Jiménez López-Lucendo; Dr. D. Rodríguez Leal; Córdoba: Dr. J.C. Regueiro López; Granada: Dr. M. Cabezas Zamora; Guipúzcoa: Dr. G. Garmendia Olaizola; Dr. J.A. Rodríguez Andrés; Jaén: Dr. J. Jiménez Verdejo; Dr. E.J. Zarate Rodríguez; León: Dr. S.C. Gómez Cisneros; Dr. M. Lozano Rebollo; Lleida: Dr. J. Cortada Robert; Dr. L.M. Flavian Domenech; Logroño: Dr. A. Fernández Fernández; Lugo: Dr. F.J. Neira Pampin; Madrid: Dr. A. Abdallah Merhi; Dr. F. Arias Funes; Dr. I.T. Castillón Vela; Dr. J.M. Duarte Ojeda; Dr. M.J. García-Matres Cortés; Dr. J.F. Hermina Gutierrez; Dr. J.J. López-Tello García; Dr. C. Martín García; Dr. B. M.El-Awadeh; Dr. J.D. Rendón Sánchez; Dr. R. Rodríguez-Patron Rodríguez; Dr. J.C. Ruíz de la Roja; Dr. J. Vallejo Herrador; Dr. D. Vázquez Alba; Málaga: Dr. A. Bonilla Maldonado; Dr. J.M. Fernández Montero; Dr. A. Galacho Bech; Dr. C. Marchal Escalona; Dr. P. Rodero García; Dr. G. Sanz; Dr. J.D. Vázquez Cervilla; Murcia: Dr. G. Server Pastor; Pontevedra: Dr. D. Jamardo González; Dr. A. Selas Pérez; Salamanca: Dr. F. Díaz Alférez; Dr. M.F. Lorenzo Gómez; Santander: Dr. J.A. Portillo Martín; Sevilla: Dr. F.J. Giráldez Puig; Dr. M.A. Gutiérrez González; Dr. J.I. Huesa Martínez; Dr. Y. Ismail Tomaizeh; Dr. J. Leal Arenas; Dr. J. Martín Calero; Dr. J.L. Moyano Calvo; Dr. A. Ortiz Gámiz; Dr. J.M. Poyato Galán; Dr. J.A. Valero Puerta; Dr. E. Vilches Cocovi; Tarragona: Dr. P.L. Álvarez de la Red; Dr. J. Benagues Pamies; Dr. M. Prados Saavedra; Tenerife: Dr. T. Concepción Masip; Dr. C. Gómez de Segura Melcon; Dr. E. González de Chaves; Dr. J. Postius Robert; Toledo: Dr. F. Álvarez Fernández; Dr. A. Iglesias Justo; Dr. A. Melchor Galán; Valencia: Dr. J.E. Blasco Alfonso; Dr. M.A. Bonillo García; Dr. A. Collado Serra; Dr. J.A. García Cebrián; Dr. L.García Reboll; Dr. Y. Pallás Costa; Dr. M.J. Sánchez Sanchiz; Dr. J. Santamaría Meseguer; Zaragoza: Dr. A. García de Jalón Martínez; Dr. J.M. Gil Fabra; Dr. F. Monzón Alebesque; Dr. A. Ucar Terren.
References
- Gravas, S.; Cornu, J.N.; Gacci, M.; Gratzke, C.; Herrmann, T.R.W.; Mamoulakis, C.; Rieken, M.; Speakman, M.J.; Tikkinen, K.A.O. Management of Non-Neurogenic Male Lower Urinary Tract Symptoms (LUTS), Incl. Benign Prostatic Obstruction (BPO); EAU Guidelines Office: Arnhem, The Netherlands, 2019; Available online: https://uroweb.org/guideline/treatment-of-non-neurogenic-male-luts/ (accessed on 19 June 2020).
- Fitzpatrick, J.M. The natural history of benign prostatic hyperplasia. BJU Int. 2006, 97, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Speakman, M.; Kirby, R.; Doyle, S.; Ioannou, C. Burden of male lower urinary tract symptoms (LUTS) suggestive of benign prostatic hyperplasia (BPH)—focus on the UK. BJU Int. 2015, 115, 508–519. [Google Scholar] [CrossRef] [PubMed]
- Vuichoud, C.; Loughlin, K.R. Benign prostatic hyperplasia: Epidemiology, economics and evaluation. Can. J. Urol. 2015, 22 (Suppl. 1), 1–6. [Google Scholar] [PubMed]
- Gacci, M.; Ficarra, V.; Sebastianelli, A.; Corona, G.; Serni, S.; Shariat, S.F.; Maggi, M.; Zattoni, F.; Carini, M.; Novara, G. Impact of medical treatments for male lower urinary tract symptoms due to benign prostatic hyperplasia on ejaculatory function: A systematic review and meta-analysis. J. Sex. Med. 2014, 11, 1554–1566. [Google Scholar] [CrossRef] [PubMed]
- Roehrborn, C.G.; Siami, P.; Barkin, J.; Damião, R.; Major-Walker, K.; Nandy, I.; Morrill, B.B.; Gagnier, R.P.; Montorsi, F.; CombAT Study Group. The effects of combination therapy with dutasteride and tamsulosin on clinical outcomes in men with symptomatic benign prostatic hyperplasia: 4-year results from the CombAT study. Eur. Urol. 2010, 57, 123–131. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Assessment Report on Serenoa Repens (W. Bartram) Small, Fructus; Final; European Medicines Agency: Amsterdam, The Netherlands, 2015; Available online: https://www.ema.europa.eu/en/documents/herbal-report/final-assessment-report-serenoa-repens-w-bartram-small-fructus_en.pdf (accessed on 19 June 2020).
- Sirab, N.; Robert, G.; Fasolo, V.; Descazeaud, A.; Vacherot, F.; Taille, A.D.; Terry, S. Lipidosterolic extract of Serenoa repens modulates the expression of inflammation related-genes in benign prostatic hyperplasia epithelial and stromal cells. Int. J. Mol. Sci. 2013, 14, 14301–14320. [Google Scholar] [CrossRef] [PubMed]
- Latil, A.; Libon, C.; Templier, M.; Junquero, D.; Lantoine-Adam, F.; Nguyen, T. Hexanic lipidosterolic extract of Serenoa repens inhibits the expression of two key inflammatory mediators, MCP-1/CCL2 and VCAM-1, in vitro. BJU Int. 2012, 110, E301–E307. [Google Scholar] [CrossRef]
- Bayne, C.W.; Donnelly, F.; Ross, M.; Habib, F.K. Serenoa repens (Permixon): A 5alpha-reductase types I and II inhibitor-new evidence in a coculture model of BPH. Prostate 1999, 40, 232–241. [Google Scholar] [CrossRef]
- Di Silverio, F.; Monti, S.; Sciarra, A.; Varasano, P.A.; Martini, C.; Lanzara, S.; D’Eramo, G.; di Nicola, S.; Toscano, V. Effects of long-term treatment with Serenoa repens (Permixon) on the concentrations and regional distribution of androgens and epidermal growth factor in benign prostatic hyperplasia. Prostate 1998, 37, 77–83. [Google Scholar] [CrossRef]
- Scaglione, F.; Lucini, V.; Pannacci, M.; Dugnani, S.; Leone, C. Comparison of the potency of 10 different brands of Serenoa repens extracts. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 569–574. [Google Scholar]
- Habib, F.K.; Wyllie, M.G. Not all brands are created equal: A comparison of selected components of different brands of Serenoa repens extract. Prostate Cancer Prostatic Dis. 2004, 7, 195–200. [Google Scholar] [CrossRef]
- Scaglione, F.; Lucini, V.; Pannacci, M.; Dugnani, S.; Leone, C. Comparison of the potency of different brands of Serenoa repens extract on 5alpha-reductase types I and II in prostatic co-cultured epithelial and fibroblast cells. Pharmacology 2008, 82, 270–275. [Google Scholar] [CrossRef]
- Bent, S.; Kane, C.; Shinohara, K.; Neuhaus, J.; Hudes, E.S.; Goldberg, H.; Avins, A.L. Saw palmetto for benign prostatic hyperplasia. N. Engl. J. Med. 2006, 354, 557–566. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, R.; Tacklind, J.W.; Rutks, I.; Wilt, T.J. Serenoa repens monotherapy for benign prostatic hyperplasia (BPH): An updated Cochrane systematic review. BJU Int. 2012, 109, 1756–1761. [Google Scholar] [CrossRef] [PubMed]
- Alcaraz, A.; Rodriguez, J.C.; Unda-Urzaiz, M.; Medina-Lopez, R.; Ruiz-Cerdá, J.L.; Rodríguez-Rubio, F.; García-Rojo, D.; Brenes-Bermúdez, F.J.; Cózar-Olmo, J.M.; Baena-González, V.; et al. Quality of life in patients with lower urinary tract symptoms associated with BPH: Change over time in real-life practice according to treatment–the QUALIPROST study. Int. Urol. Nephrol. 2016, 48, 645–656. [Google Scholar] [CrossRef] [PubMed]
- Vela-Navarrete, R.; Alcaraz, A.; Rodríguez-Antolín, A.; López, B.M.; Fernández-Gómez, J.M.; Angulo, J.C.; Diaz, D.C.; Romero-Otero, J.; Brenes, F.J.; Carballido, J.; et al. Efficacy and safety of a hexanic extract of Serenoa repens (Permixon®) for the treatment of lower urinary tract symptoms associated with benign prostatic hyperplasia (LUTS/BPH): Systematic review and meta-analysis of randomized controlled trials and observational studies. BJU Int. 2018, 122, 1049–1065. [Google Scholar] [PubMed]
- Novara, G.; Giannarini, G.; Alcaraz, A.; Cózar-Olmo, J.M.; Descazeaud, A.; Montorsi, F.; Ficarra, V. Efficacy and Safety of Hexanic Lipidosterolic Extract of Serenoa Repens (Permixon) in the Treatment of Lower Urinary Tract Symptoms Due to Benign Prostatic Hyperplasia: Systematic Review and Meta-analysis of Randomized Controlled Trials. Eur. Urol. Focus 2016, 2, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Gravas, S.; Cornu, J.N.; Drake, M.J.; Gacci, M.; Gratzke, C.; Herrmann, T.R.W.; Tikkine, K. EAU Guidelines on management of non-neurogenic male LUTS. In Proceedings of the EAU Annual Congress Amsterdam 2020, Arnhem, The Netherlands, 2020; ISBN 978-94-92671-07-3. Available online: https://uroweb.org/guideline/treatment-of-non-neurogenic-male-luts/#5_2 (accessed on 19 June 2020).
- Fourcade, R.O.; Lacoin, F.; Rouprêt, M.; Slama, A.; Le Fur, C.; Michel, E.; Sitbon, A.; Cotté, F.E. Outcomes and general health-related quality of life among patients medically treated in general daily practice for lower urinary tract symptoms due to benign prostatic hyperplasia. World J. Urol. 2012, 30, 419–426. [Google Scholar] [CrossRef]
- Ryu, Y.W.; Lim, S.W.; Kim, J.H.; Ahn, S.H.; Choi, J.D. Comparison of tamsulosin plus Serenoa repens with tamsulosin in the treatment of benign prostatic hyperplasia in Korean men: 1-year randomized open label study. Urol. Int. 2015, 94, 187–193. [Google Scholar] [CrossRef]
- Boeri, L.; Capogrosso, P.; Ventimiglia, E.; Cazzaniga, W.; Pederzoli, F.; Moretti, D.; Dehò, F.; Montanari, E.; Montorsi, F.; Salonia, A. Clinically Meaningful Improvements in LUTS/BPH Severity in Men Treated with Silodosin Plus Hexanic Extract of Serenoa repens or Silodosin Alone. Sci. Rep. 2017, 7, 15179. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. STROBE Initiative. Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. BMJ 2007, 335, 806–808. [Google Scholar] [CrossRef]
- Barry, M.J.; Fowler, F.J., Jr.; O’Leary, M.P.; Bruskewitz, R.C.; Holtgrewe, H.L.; Mebust, W.K. Measuring disease-specific health status in men with benign prostatic hyperplasia. Measurement Committee of The American Urological Association. Med. Care 1995, 33, AS145–AS155. [Google Scholar]
- Haynes, R.B.; Sackett, D.L.; Gibson, E.S.; Taylor, D.W.; Hackett, B.C.; Roberts, R.S.; Johnson, A.L. Improvement of medication compliance in uncontrolled hypertension. Lancet 1976, 1, 1265–1268. [Google Scholar] [CrossRef]
- Barry, M.J.; Williford, W.O.; Chang, Y.; Machi, M.; Jones, K.M.; Walker-Corkery, E.; Lepor, H. Benign prostatic hyperplasia specific health status measures in clinical research: How much change in the American Urological Association symptom index and the benign prostatic hyperplasia impact index is perceptible to patients? J. Urol. 1995, 154, 1770–1774. [Google Scholar] [CrossRef]
- Ficarra, V.; Rossanese, M.; Zazzara, M.; Giannarini, G.; Abbinante, M.; Bartoletti, R.; Mirone, V.; Scaglione, F. The role of inflammation in lower urinary tract symptoms (LUTS) due to benign prostatic hyperplasia (BPH) and its potential impact on medical therapy. Curr. Urol. Rep. 2014, 15, 463. [Google Scholar] [CrossRef]
- Michel, M.C. The forefront for novel therapeutic agents based on the pathophysiology of lower urinary tract dysfunction: Alpha-blockers in the treatment of male voiding dysfunction—how do they work and why do they differ in tolerability? J. Pharmacol. Sci. 2010, 112, 151–157. [Google Scholar] [CrossRef]
- Debruyne, F.; Koch, G.; Boyle, P.; da Silva, F.C.; Gillenwater, J.G.; Hamdy, F.C.; Perrin, P.; Teillac, P.; Vela-Navarrete, R.; Raynaud, J.P. Comparison of a phytotherapeutic agent (Permixon) with an alpha-blocker (Tamsulosin) in the treatment of benign prostatic hyperplasia: A 1-year randomized international study. Eur. Urol. 2002, 41, 497–506. [Google Scholar] [CrossRef]
- Debruyne, F.; Boyle, P.; Calais da Silva, F.; Gillenwater, J.G.; Hamdy, F.C.; Perrin, P.; Teillac, P.; Vela-Navarrete, R.; Raynaud, J.P.; Schulman, C.C. Evaluation of the clinical benefit of Permixon and tamsulosin in severe BPH patients—PERMAL study subset analysis. Eur. Urol. 2004, 45, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Chung, B.H.; Roehrborn, C.G.; Siami, P.; Major-Walker, K.; Morrill, B.B.; Wilson, T.H.; Montorsi, F. Efficacy and safety of dutasteride, tamsulosin and their combination in a subpopulation of the CombAT study: 2-year results in Asian men with moderate-to-severe BPH. Prostate Cancer Prostatic Dis. 2009, 12, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Lepor, H.; Williford, W.O.; Barry, M.J.; Brawer, M.K.; Dixon, C.M.; Gormley, G.; Haakenson, C.; Machi, M.; Narayan, P.; Padley, R.J. The efficacy of terazosin, finasteride, or both in benign prostatic hyperplasia. Veterans Affairs Cooperative Studies Benign Prostatic Hyperplasia Study Group. N. Engl. J. Med. 1996, 335, 533–539. [Google Scholar] [CrossRef]
- McConnell, J.D.; Roehrborn, C.G.; Bautista, O.M.; Andriole, G.L., Jr.; Dixon, C.M.; Kusek, J.W.; Lepor, H.; McVary, K.T.; Nyberg, L.M., Jr.; Clarke, H.S.; et al. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N. Engl. J. Med. 2003, 349, 2387–2398. [Google Scholar] [CrossRef]
- Barkin, J.; Roehrborn, C.G.; Siami, P.; Haillot, O.; Morrill, B.; Black, L.; Montorsi, F.; CombAT Study Group. Effect of dutasteride, tamsulosin and the combination on patient-reported quality of life and treatment satisfaction in men with moderate-to-severe benign prostatic hyperplasia: 2-year data from the CombAT trial. BJU Int. 2009, 103, 919–926. [Google Scholar] [CrossRef] [PubMed]
- Rosen, R.C.; Roehrborn, C.G.; Manyak, M.J.; Palacios-Moreno, J.M.; Wilson, T.H.; Lulic, Z.; Giuliano, F. Evaluation of the impact of dutasteride/tamsulosin combination therapy on libido in sexually active men with lower urinary tract symptoms (LUTS) secondary to benign prostatic hyperplasia (BPH): A post hoc analysis of a prospective randomised placebo-controlled study. Int. J. Clin. Pract. 2019, 73, 1–9. [Google Scholar] [PubMed]
- Cindolo, L.; Pirozzi, L.; Fanizza, C.; Romero, M.; Tubaro, A.; Autorino, R.; de Nunzio, C.; Schips, L. Drug adherence and clinical outcomes for patients under pharmacological therapy for lower urinary tract symptoms related to benign prostatic hyperplasia: Population-based cohort study. Eur. Urol. 2015, 68, 418–425. [Google Scholar] [CrossRef]
- Chou, C.H.; Lin, C.L.; Lin, M.C.; Sung, F.C.; Kao, C.H. 5α-Reductase inhibitors increase acute coronary syndrome risk in patients with benign prostate hyperplasia. J. Endocrinol. Investig. 2015, 38, 799–805. [Google Scholar] [CrossRef]
- Serati, M.; Andersson, K.E.; Dmochowski, R.; Agrò, E.F.; Heesakkers, J.; Iacovelli, V.; Novara, G.; Khullar, V.; Chapple, C. Systematic Review of Combination Drug Therapy for Non-neurogenic Lower Urinary Tract Symptoms. Eur. Urol. 2019, 75, 129–168. [Google Scholar] [CrossRef]
- De Nunzio, C.; Salonia, A.; Gacci, M.; Ficarra, V. Inflammation is a target of medical treatment for lower urinary tract symptoms associated with benign prostatic hyperplasia. World J. Urol. 2020. [Google Scholar] [CrossRef] [PubMed]
| Variable | TAM | HESr | TAM + HESr | p Value | |||
|---|---|---|---|---|---|---|---|
| N * | Mean (SD) | N * | Mean (SD) | N * | Mean (SD) | ||
| Age (years) | 238 | 65.4 (8.0) | 223 | 64.6 (8.9) | 161 | 65.1 (8.0) | 0.589 |
| BMI (Kg/m2) | 233 | 26.9 (3.1) | 221 | 26.8 (3.0) | 160 | 27.1 (2.9) | 0.514 |
| IPSS total | 263 | 18.7 (4.5) | 262 | 18.6 (4.8) | 184 | 19.5 (4.9) | 0.150 |
| BII | 263 | 7.8 (2.1) | 262 | 7.9 (1.9) | 184 | 8.2 (2.1) | 0.102 |
| IPSS storage subscore | 263 | 7.9 (2.1) | 262 | 8.1 (2.2) | 184 | 8.4 (2.2) | 0.105 |
| IPSS voiding subscore | 263 | 10.8 (3.1) | 262 | 10.5 (3.1) | 184 | 11.1 (3.2) | 0.209 |
| IPSS item 8 (QoL) | 263 | 3.9 (1.0) | 262 | 3.8 (1.1) | 184 | 4.0 (1.0) | 0.372 |
| Time since diagnosis (years) | 237 | 1.1 (2.6) | 222 | 1.2 (2.7) | 159 | 1.4 (3.2) | 0.607 |
| Qmax (ml/s) | 104 | 12.1 (3.7) | 117 | 13.2 (4.1) | 118 | 12.8 (3.1) | 0.105 |
| PSA (ng/ml) | 240 | 2.5 (1.3) | 242 | 2.3 (1.2) | 176 | 2.4 (1.4) | 0.409 |
| Prostate volume (cm3) | 220 | 52.3 (18.3) | 230 | 51.1 (18.0) | 169 | 54.8 (16.4) | 0.106 |
| TAM (n = 263) | HESr (n = 262) | TAM + HESr (n = 184) | p Value | |
|---|---|---|---|---|
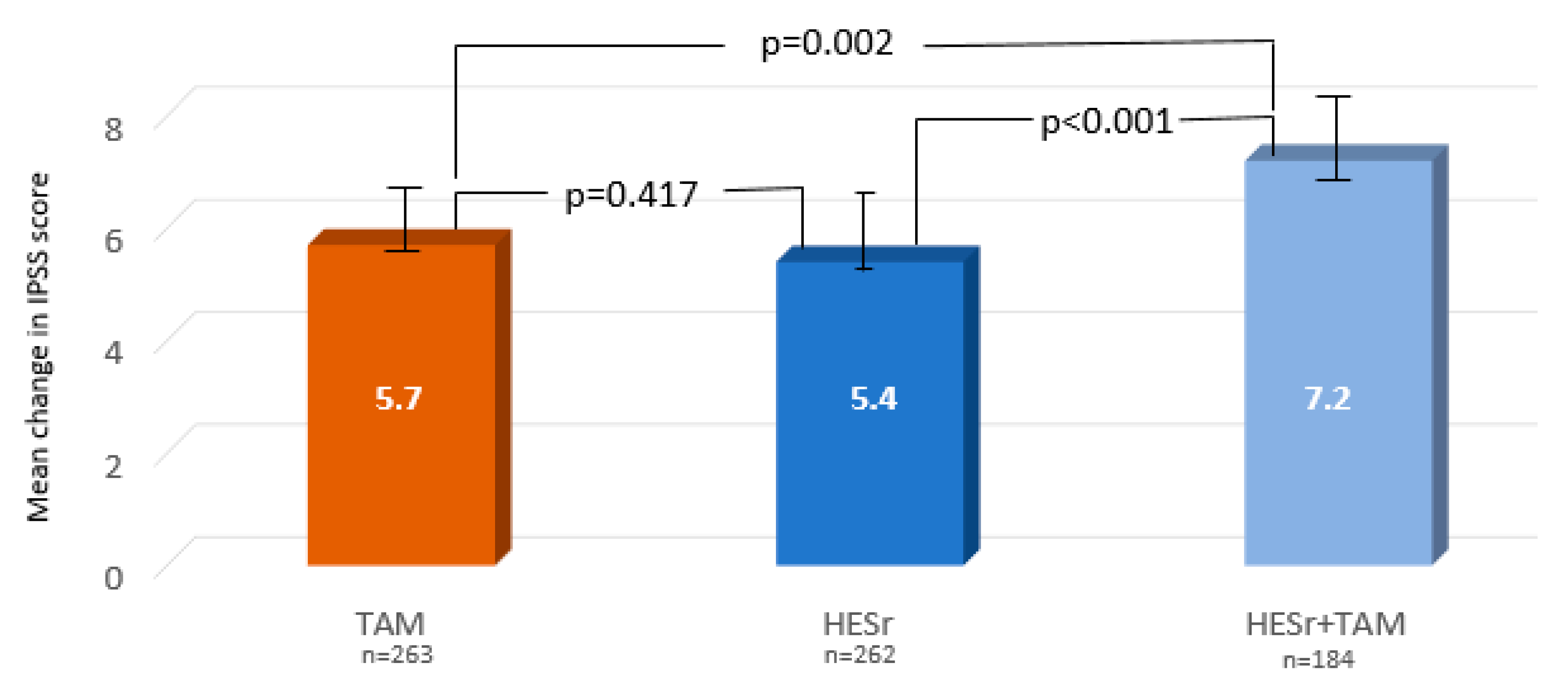
| IPSS total * | 5.7 (4.3) | 5.4 (4.6) | 7.2 (5.0) | <0.001 |
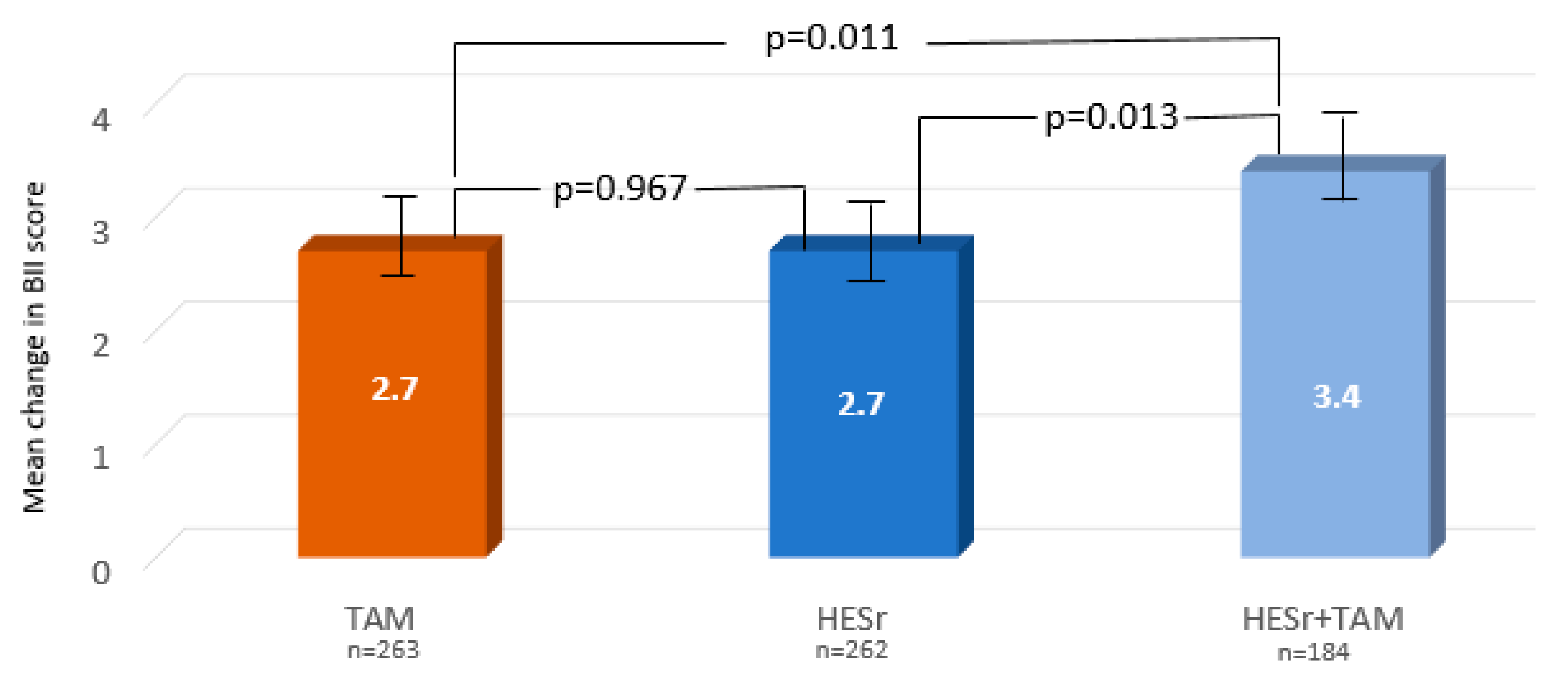
| BII total * | 2.7 (2.4) | 2.7 (2.5) | 3.4 (2.5) | 0.020 |
| IPSS storage subscore * | 2.4 (2.1) | 2.4 (2.2) | 3.2 (2.3) | <0.001 |
| IPSS voiding subscore * | 3.3 (2.8) | 3.0 (3.0) | 3.9 (3.3) | 0.014 |
| IPSS item 8 (QoL) * | 1.3 (1.2) | 1.3 (1.3) | 1.8 (1.2) | <0.001 |
| TAM (n = 99) | HESr (n = 90) | TAM + HESr (n = 79) | p Value | |
|---|---|---|---|---|
| IPSS total * | 8.0 (5.0) | 7.8 (5.1) | 10.1 (5.1) | 0.006 |
| BII total * | 2.9 (2.7) | 2.9 (2.7) | 3.9 (2.4) | 0.021 |
| IPSS storage subscore * | 3.3 (2.3) | 3.2 (2.4) | 4.3 (2.4) | 0.005 |
| IPSS voiding subscore * | 4.7 (3.2) | 4.6 (3.2) | 5.8 (3.2) | 0.026 |
| IPSS item 8 (QoL) * | 1.6 (1.3) | 1.4 (1.4) | 2.2 (1.0) | <0.001 |
| TAM (n = 263) | HESr (n = 262) | TAM + HESr (n = 184) | p Value | |
|---|---|---|---|---|
| Any adverse effect | 35 (13.3%) | 5 (1.9%) | 22 (12.0%) | <0.001 |
| Ejaculation disturbances * | 30 (11.4%) | 2 (0.8%) | 15 (8.2%) | <0.001 |
| Erectile dysfunction | 3 (1.1%) | 0 (0.0%) | 5 (2.7%) | 0.021 |
| Hypotension | 7 (2.7%) | 0 (0.0%) | 5 (2.7%) | 0.011 |
| Reduced libido | 3 (1.1%) | 0 (0.0%) | 1 (0.5%) | 0.198 |
| Dizziness | 0 (0.0%) | 2 (0.8%) | 2 (1.1%) | 0.253 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alcaraz, A.; Rodríguez-Antolín, A.; Carballido-Rodríguez, J.; Castro-Díaz, D.; Esteban-Fuertes, M.; Cózar-Olmo, J.M.; Ficarra, V.; Medina-López, R.; Fernández-Gómez, J.M.; Angulo, J.C.; et al. Clinical Benefit of Tamsulosin and the Hexanic Extract of Serenoa Repens, in Combination or as Monotherapy, in Patients with Moderate/Severe LUTS-BPH: A Subset Analysis of the QUALIPROST Study. J. Clin. Med. 2020, 9, 2909. https://doi.org/10.3390/jcm9092909
Alcaraz A, Rodríguez-Antolín A, Carballido-Rodríguez J, Castro-Díaz D, Esteban-Fuertes M, Cózar-Olmo JM, Ficarra V, Medina-López R, Fernández-Gómez JM, Angulo JC, et al. Clinical Benefit of Tamsulosin and the Hexanic Extract of Serenoa Repens, in Combination or as Monotherapy, in Patients with Moderate/Severe LUTS-BPH: A Subset Analysis of the QUALIPROST Study. Journal of Clinical Medicine. 2020; 9(9):2909. https://doi.org/10.3390/jcm9092909
Chicago/Turabian StyleAlcaraz, Antonio, Alfredo Rodríguez-Antolín, Joaquín Carballido-Rodríguez, David Castro-Díaz, Manuel Esteban-Fuertes, José M. Cózar-Olmo, Vincenzo Ficarra, Rafael Medina-López, Jesús M. Fernández-Gómez, Javier C. Angulo, and et al. 2020. "Clinical Benefit of Tamsulosin and the Hexanic Extract of Serenoa Repens, in Combination or as Monotherapy, in Patients with Moderate/Severe LUTS-BPH: A Subset Analysis of the QUALIPROST Study" Journal of Clinical Medicine 9, no. 9: 2909. https://doi.org/10.3390/jcm9092909
APA StyleAlcaraz, A., Rodríguez-Antolín, A., Carballido-Rodríguez, J., Castro-Díaz, D., Esteban-Fuertes, M., Cózar-Olmo, J. M., Ficarra, V., Medina-López, R., Fernández-Gómez, J. M., Angulo, J. C., Medina-Polo, J., Brenes-Bermúdez, F. J., Molero-García, J. M., Fernández-Pro-Ledesma, A., Manasanch, J., & on behalf of The QUALIPROST Study Group. (2020). Clinical Benefit of Tamsulosin and the Hexanic Extract of Serenoa Repens, in Combination or as Monotherapy, in Patients with Moderate/Severe LUTS-BPH: A Subset Analysis of the QUALIPROST Study. Journal of Clinical Medicine, 9(9), 2909. https://doi.org/10.3390/jcm9092909






