Photo and Plasma Activation of Dental Implant Titanium Surfaces. A Systematic Review with Meta-Analysis of Pre-Clinical Studies
Abstract
1. Introduction
2. Material and Methods
- Population: Healthy animals with at least one titanium dental implant.
- Intervention: Any surface activation with UV or non-thermal plasma.
- Comparison: Any type of “non-activated” titanium dental implant.
- Outcomes: Bone-to-implant contact (BIC), implant stability quotient (ISQ) or removal torque.
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Selection of Studies
2.4. Data Extraction
2.5. Quality Assessment
2.6. Statistical Analysis
3. Results
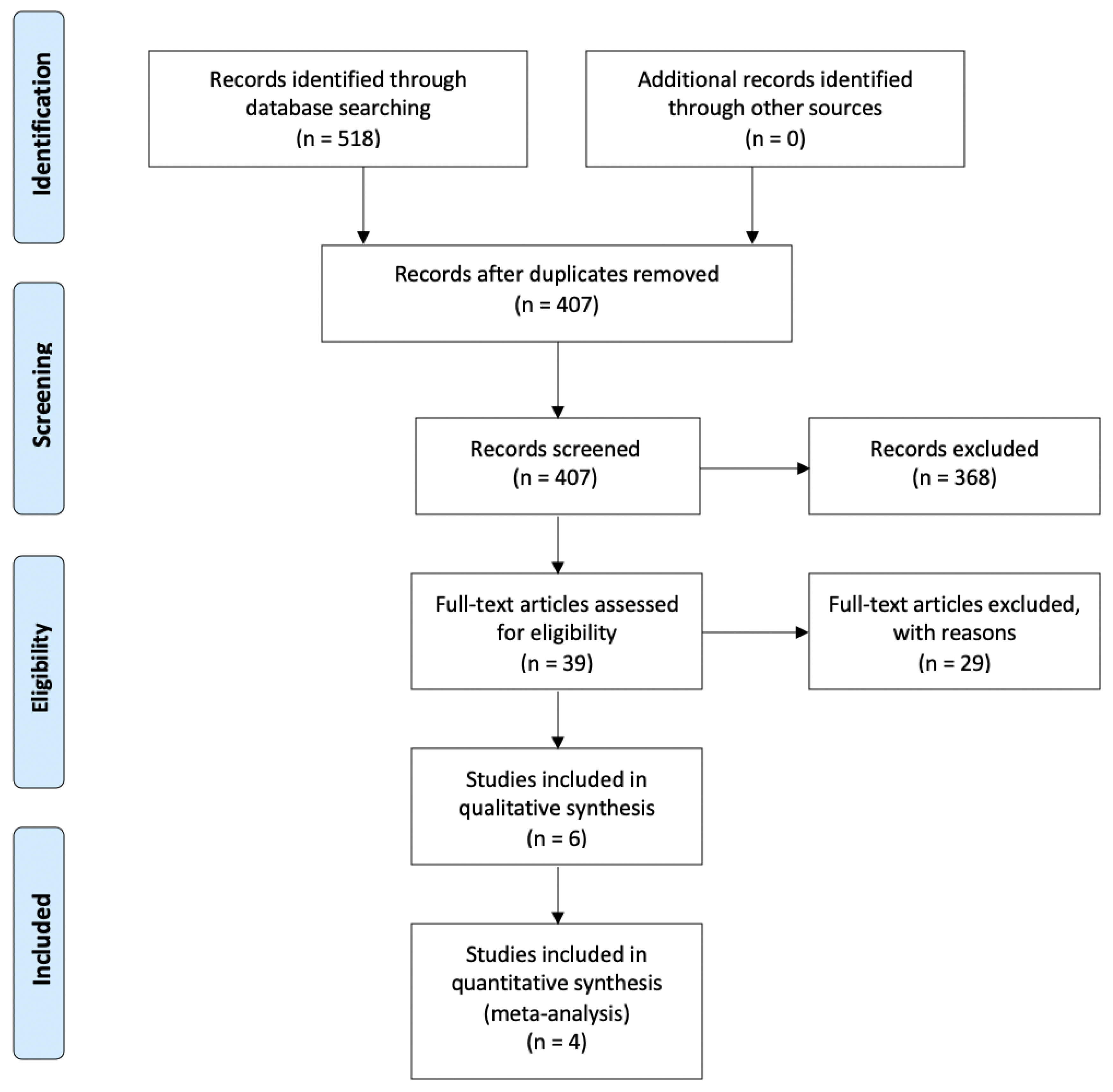
3.1. Bibliographic Search and Study Selection
3.2. Description of Included Studies
3.3. Excluded Studies
3.4. Quality Assessment
3.5. Qualitative Assessment of Outcomes
3.6. Quantitative Assessment of Outcomes
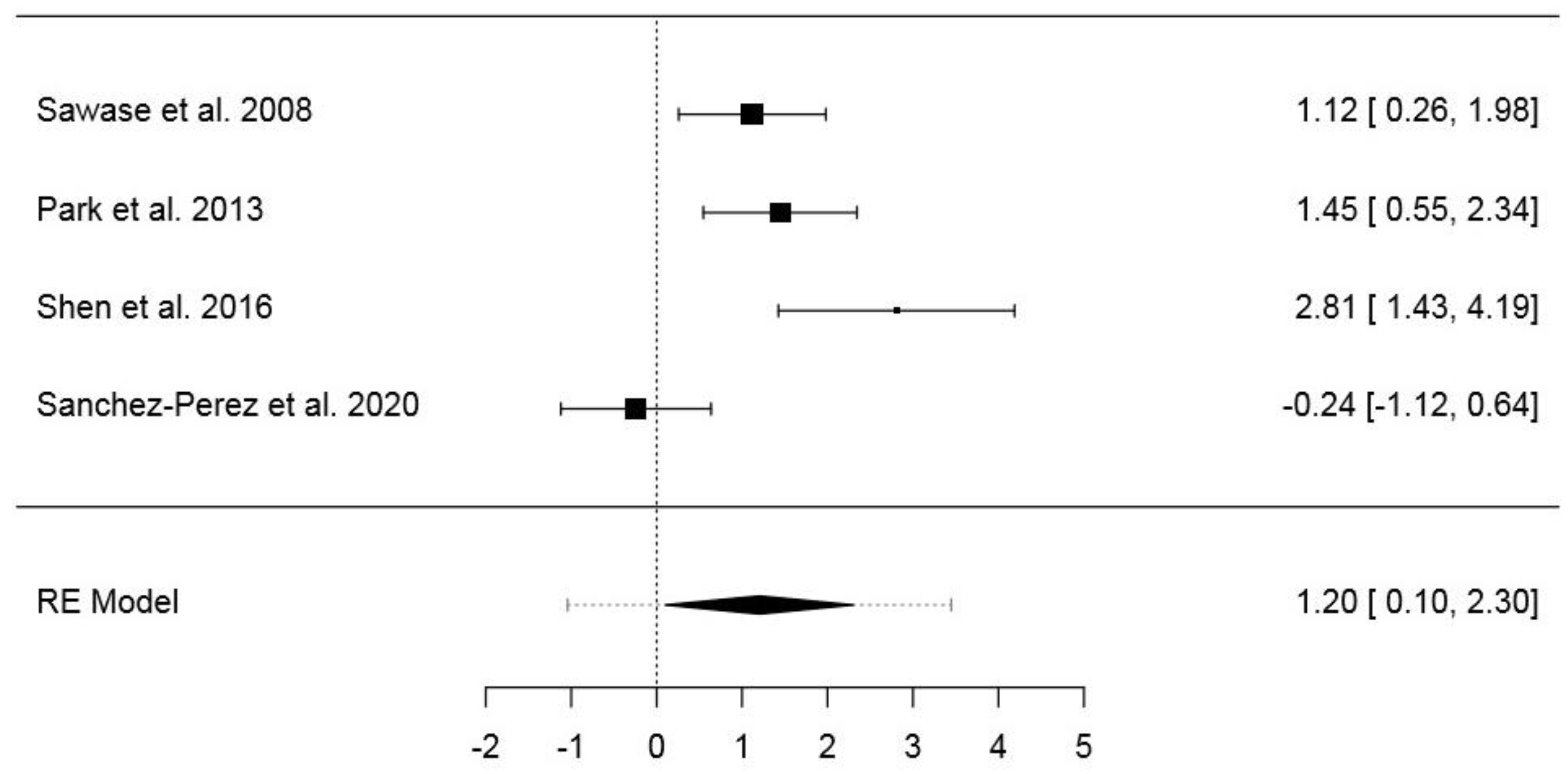
3.6.1. First Dataset
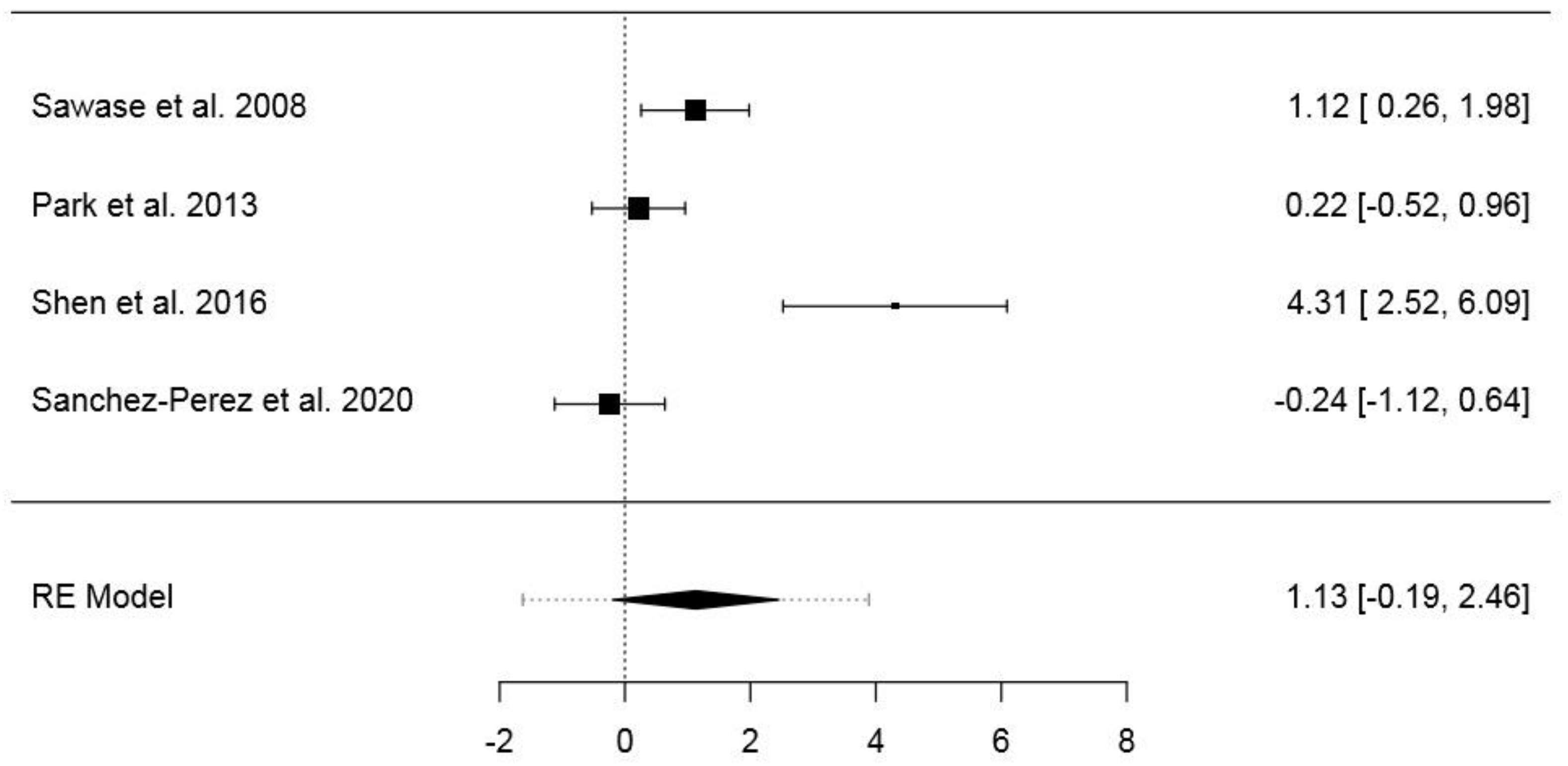
3.6.2. Second Dataset
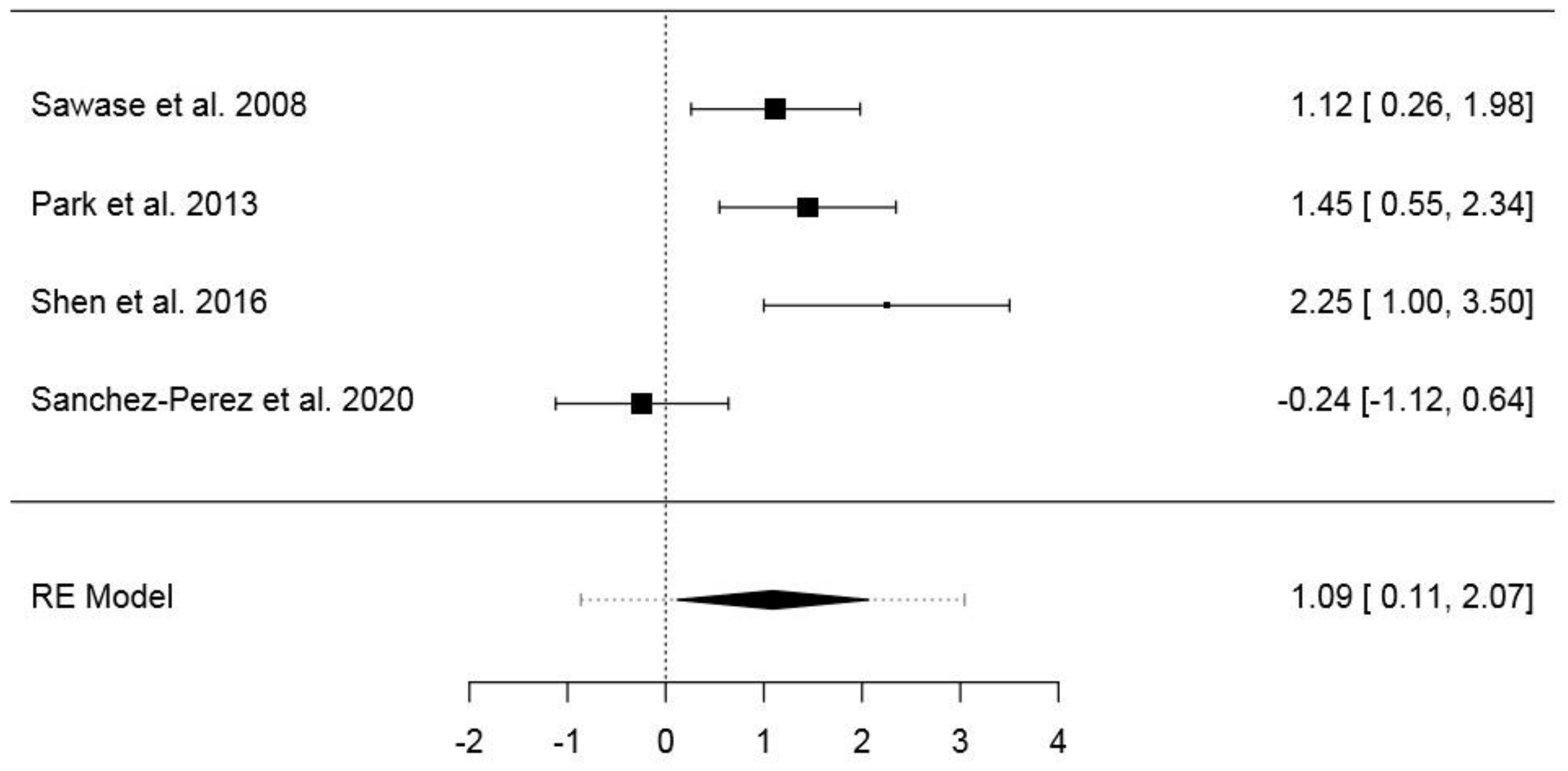
3.6.3. Third Dataset
3.6.4. Fourth Dataset
4. Discussion
Summary of the Results and Possible Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Article Excluded | Reason for Exclusion |
|---|---|
| Shon et al., 2013 [30] | Specimens material (zirconia implants) |
| Naujok et al., 2019 [31] | Sample size |
| Aita H et al., 2009 [32] | Outcome |
| Suzuki T et al., 2009 [13] | Outcome |
| Ueno T et al., 2010 [33] | Outcome |
| Ueno T et al., 2010 [34] | Outcome |
| Minamikaw et al., 2014 [35] | Outcome |
| Hirota M et al., 2016 [36] | Sample size |
| Ishijima M et al., 2016 [37] | Outcome |
| Soltanzadeh P et al., 2017 [38] | Outcome |
| Sugita Y et al., 2014 [39] | Outcome |
| Taniyama et al., 2020 [40] | Outcome |
| Jimbo R et al., 2011 [41] | Specimens |
| Hayashi M et al., 2014 [42] | Specimens |
| Yamazaki M et al., 2015 [43] | Outcome |
| Kim H.S et al., 2017 [44] | Farmacological treatment (alendronate) |
| Lee J.B et al., 2019 [45] | Sample size |
| Miki T et al., 2019 [46] | Specimens |
| Hirakawa et al., 2013 [47] | Surface treatment |
| Pyo et al., 2013 [48] | Sample size |
| Ishii et al., 2016 [49] | Outcome |
| Mehl et al. 2018 [50] | Sample size |
| Funato et al., 2013 [51] | Case series |
| Suzuki et al., 2013 [52] | Cross-sectional retrospective analysis |
| Funato et al., 2013 [53] | Retrospective analysis |
| Kitajima et al., 2016 [54] | Cross-sectional retrospective analysis |
| Hirota et al., 2016 [55] | Complex cases |
| Hirota et al., 2018 [56] | Outcome |
| Puisys et al., 2018 [57] | Specimens |
References
- Moraschini, V.; Poubel, L.A.; Ferreira, V.F.; Edos, B.S. Evaluation of survival and success rates of dental implants reported in longitudinal studies with a follow-up period of at least 10 years: A systematic review. Int. J. Oral Maxillofac. Surg. 2015, 44, 377–388. [Google Scholar] [CrossRef] [PubMed]
- Zita Gomes, R.; de Vasconcelos, M.R.; Lopes Guerra, I.M.; de Almeida, R.A.B.; de Campos Felino, A.C. Implant stability in the posterior maxilla: A controlled clinical trial. Biomed. Res. Int. 2017, 2017, 6825213. [Google Scholar] [CrossRef] [PubMed]
- Albrektsson, T.; Jemt, T.; Molne, J.; Tengvall, P.; Wennerberg, A. On inflammation-immunological balance theory—A critical apprehension of disease concepts around implants: Mucositis and marginal bone loss may represent normal conditions and not necessarily a state of disease. Clin. Implant Dent. Relat. Res. 2019, 21, 183–189. [Google Scholar] [CrossRef]
- Albrektsson, T.; Chrcanovic, B.; Molne, J.; Wennerberg, A. Foreign body reactions, marginal bone loss and allergies in relation to titanium implants. Eur. J. Oral Implantol. 2018, 11, S37–S46. [Google Scholar] [PubMed]
- Jemat, A.; Ghazali, M.J.; Razali, M.; Otsuka, Y. Surface modifications and their effects on titanium dental implants. BioMed Res. Int. 2015, 2015, 791725. [Google Scholar] [CrossRef] [PubMed]
- Conserva, E.; Menini, M.; Ravera, G.; Pera, P. The role of surface implant treatments on the biological behavior of SaOS-2 osteoblast-like cells. An in vitro comparative study. Clin. Oral Implants Res. 2013, 24, 880–889. [Google Scholar] [CrossRef]
- Conserva, E.; Lanuti, A.; Menini, M. Cell behavior related to implant surfaces with different microstructure and chemical composition: An in vitro analysis. Int. J. Oral Maxillofac. Implants 2010, 25, 1099–1107. [Google Scholar]
- Menini, M.; Dellepiane, E.; Chvartszaid, D.; Baldi, D.; Schiavetti, I.; Pera, P. Influence of different surface Characteristics on Peri-implant tissue behavior: A six-year prospective report. Int. J. Prosthodont. 2015, 28, 389–395. [Google Scholar] [CrossRef]
- Alharbi, H.M.; Babay, N.; Alzoman, H.; Basudan, S.; Anil, S.; Jansen, J.A. Bone morphology changes around two types of bone-level implants installed in fresh extraction sockets—A histomorphometric study in beagle dogs. Clin. Oral Implants Res. 2015, 26, 1106–1112. [Google Scholar] [CrossRef]
- Canullo, L.; Menini, M.; Santori, G.; Rakic, M.; Sculean, A.; Pesce, P. Titanium abutment surface modifications and peri-implant tissue behavior: A systematic review and meta-analysis. Clin. Oral Investig. 2020, 24, 1113–1124. [Google Scholar] [CrossRef]
- Pesce, P.; Menini, M.; Tommasato, G.; Patini, R.; Canullo, L. Influence of modified titanium abutment surface on peri-implant soft tissue behaviour: A systematic review of histological findings. Int. J. Oral Implantol. 2019, 12, 419–429. [Google Scholar]
- Mariscal-Munoz, E.; Costa, C.A.; Tavares, H.S.; Bianchi, J.; Hebling, J.; Machado, J.P.; Lerner, U.H.; Souza, P.P. Osteoblast differentiation is enhanced by a nano-to-microhybrid titanium surface created by Yb: YAG laser irradiation. Clin. Oral Investig. 2016, 20, 503–551. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Hori, N.; Att, W.; Kubo, K.; Iwasa, F.; Ueno, T.; Maeda, H.; Ogawa, T. Ultraviolet treatment overcomes time-related degrading bioactivity of titanium. Tissue Eng. A 2009, 15, 3679–3688. [Google Scholar] [CrossRef] [PubMed]
- Att, W.; Hori, N.; Takeuchi, M.; Ouyang, J.; Yang, Y.; Anpo, M.; Ogawa, T. Time-dependent degradation of titanium osteoconductivity: An implication of biological aging of implant materials. Biomaterials 2009, 30, 5352–5536. [Google Scholar] [CrossRef]
- Henningsen, A.; Smeets, R.; Hartjen, P.; Heinrich, O.; Heuberger, R.; Heiland, M.; Precht, C.; Cacaci, C. Photofunctionalization and non-thermal plasma activation of titanium surfaces. Clin. Oral Investig. 2018, 22, 1045–1054. [Google Scholar] [CrossRef]
- Canullo, L.; Genova, T.; Tallarico, M.; Gautier, G.; Mussano, F.; Botticelli, D. Plasma of argon affects the earliest biological response of different implant surfaces: An in vitro comparative study. J. Dent. Res. 2016, 95, 566–573. [Google Scholar] [CrossRef]
- Canullo, L.; Genova, T.; Gross Trujillo, E.; Pradies, G.; Petrillo, S.; Muzzi, M.; Carossa, S.; Mussano, F. Fibroblast interaction with different abutment surfaces: In vitro study. Int. J. Mol. Sci. 2020, 21, 1919. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef]
- Shen, J.; Liu, J.; Chen, X.; Wang, X.; He, F.; Wang, H. The In Vivo Bone Response of Ultraviolet-Irradiated Titanium Implants Modified with Spontaneously Formed Nanostructures: An Experimental Study in Rabbits. Int. J. Oral Maxillofac. Implants 2016, 31, 776–784. [Google Scholar] [CrossRef]
- Coelho, P.G.; Giro, G.; Teixeira, H.S.; Marin, C.; Witek, L.; Thompson, V.P.; Tovar, N.; Silva, N.R. Argon-based atmospheric pressure plasma enhances early bone response to rough titanium surfaces. J. Biomed. Mater. Res. A 2012, 100, 1901–1906. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, H.S.; Marin, C.; Witek, L.; Freitas Jr, A.; Silva, N.R.; Lilin, T.; Tovar, N.; Janal, M.N.; Coelho, P.G. Assessment of a chair-side argon-based non-thermal plasma treatment on the surface characteristics and integration of dental implants with textured surfaces. J. Mech. Behav. Biomed. Mater. 2012, 9, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Giro, G.; Tovar, N.; Witek, L.; Marin, C.; Silva, N.R.; Bonfante, E.A.; Coelho, P.G. Osseointegration assessment of chairside argon-based non thermal plasma-treated Ca-P coated dental implants. J. Biomed. Mater. Res. A 2013, 101, 98–103. [Google Scholar] [CrossRef]
- Canullo, L.; Tallarico, M.; Botticelli, D.; Alccayhuaman, K.A.A.; Martins Neto, E.C.; Xavier, S.P. Hard and soft tissue changes around implants activated using plasma of argon: A histomorphometric study in dog. Clin. Oral Implants Res. 2018, 29, 389–395. [Google Scholar] [CrossRef] [PubMed]
- Hung, Y.W.; Chen, H.L.; Lee, L.T.; Tung, K.C.; Bau, D.T.; Wong, Y.K. Effects of non-thermal plasma on sandblasted titanium dental implants in beagle dogs. J. Chin. Med Assoc. 2018, 81, 920–925. [Google Scholar] [CrossRef] [PubMed]
- Sawase, T.; Jimbo, R.; Baba, K.; Shibata, Y.; Ikeda, T.; Atsuta, M. Photo-induced hydrophilicity enhances initial cell behavior and early bone apposition. Clin. Oral Implants Res. 2008, 19, 491–496. [Google Scholar] [CrossRef]
- Park, K.H.; Koak, J.Y.; Kim, S.K.; Han, C.H.; Heo, S.J. The effect of ultraviolet-C irradiation via a bactericidal ultraviolet sterilizer on an anodized titanium implant: A study in rabbits. Int. J. Oral Maxillofac. Implants 2013, 28, 57–66. [Google Scholar] [CrossRef][Green Version]
- Yamauchi, R.; Itabashi, T.; Wada, K.; Tanaka, T.; Kumagai, G.; Ishibashi, Y. Photofunctionalised Ti6Al4V implants enhance early phase osseointegration. Bone Jt. Res. 2017, 6, 331–336. [Google Scholar] [CrossRef]
- Sanchez-Perez, A.; Cachazo-Jiménez, C.; Sánchez-Matás, C.; Martín-de-Llano, J.J.; Davis, S.; Carda-Batalla, C. Effects of Ultraviolet Photoactivation on Osseointegration of Commercial Pure Titanium Dental Implant After 8 Weeks in a Rabbit Model. J. Oral Implantol. 2020, 46, 101–107. [Google Scholar] [CrossRef]
- Shon, W.J.; Chung, S.H.; Kim, H.K.; Han, G.J.; Cho, B.H.; Park, Y.S. Peri-implant bone formation of non-thermal atmospheric pressure plasma-treated zirconia implants with different surface roughness in rabbit tibiae. Clin. Oral Implants Res. 2014, 25, 573–579. [Google Scholar] [CrossRef]
- Naujokat, H.; Harder, S.; Schulz, L.Y.; Wiltfang, J.; Flörke, C.; Açil, Y. Surface conditioning with cold argon plasma and its effect on the osseointegration of dental implants in miniature pigs. J. Craniomaxillofac. Surg. 2019, 47, 484–490. [Google Scholar] [CrossRef] [PubMed]
- Aita, H.; Hori, N.; Takeuchi, M.; Suzuki, T.; Yamada, M.; Anpo, M.; Ogawa, T. The effect of ultraviolet functionalization of titanium on integration with bone. Biomaterials 2009, 30, 1015–1025. [Google Scholar] [CrossRef] [PubMed]
- Ueno, T.; Yamada, M.; Hori, N.; Suzuki, T.; Ogawa, T. Effect of ultraviolet photoactivation of titanium on osseointegration in a rat model. Int. J. Oral Maxillofac. Implants 2010, 25, 287–294. [Google Scholar] [PubMed]
- Ueno, T.; Yamada, M.; Suzuki, T.; Minamikawa, H.; Sato, N.; Hori, N.; Takeuchi, K.; Hattori, M.; Ogawa, T. Enhancement of bone-titanium integration profile with UV-photofunctionalized titanium in a gap healing model. Biomaterials 2010, 31, 1546–1557. [Google Scholar] [CrossRef]
- Minamikawa, H.; Ikeda, T.; Att, W.; Hagiwara, Y.; Hirota, M.; Tabuchi, M.; Aita, H.; Park, W.; Ogawa, T. Photofunctionalization increases the bioactivity and osteoconductivity of the titanium alloy Ti6Al4V. J. Biomed. Mater. Res. A 2014, 102, 3618–3630. [Google Scholar] [CrossRef]
- Hirota, M.; Tanaka, M.; Ishijima, M.; Iwasaki, C.; Park, W.; Ogawa, T. Effect of photofunctionalization on Ti6Al4V screw stability placed in segmental bone defects in rat femurs. J. Oral Maxillofac. Surg. 2016, 74, 861.e1–861.e16. [Google Scholar] [CrossRef]
- Ishijima, M.; Ghassemi, A.; Soltanzadeh, P.; Tanaka, M.; Nakhaei, K.; Park, W.; Hirota, M.; Tsukimura, N.; Ogawa, T. Effect of UV Photofunctionalization on Osseointegration in Aged Rats. Implant Dent. 2016, 25, 744–750. [Google Scholar] [CrossRef]
- Soltanzadeh, P.; Ghassemi, A.; Ishijima, M.; Tanaka, M.; Park, W.; Iwasaki, C.; Hirota, M.; Ogawa, T. Success rate and strength of osseointegration of immediately loaded UV-photofunctionalized implants in a rat model. J. Prosthet. Dent. 2017, 118, 357–362. [Google Scholar] [CrossRef]
- Sugita, Y.; Honda, Y.; Kato, I.; Kubo, K.; Maeda, H.; Ogawa, T. Role of photofunctionalization in mitigating impaired osseointegration associated with type 2 diabetes in rats. Int. J. Oral Maxillofac. Implants 2014, 29, 1293–1300. [Google Scholar] [CrossRef]
- Taniyama, T.; Saruta, J.; Mohammadzadeh Rezaei, N.; Nakhaei, K.; Ghassemi, A.; Hirota, M.; Okubo, T.; Ikeda, T.; Sugita, Y.; Hasegawa, M.; et al. UV-Photofunctionalization of Titanium Promotes Mechanical Anchorage in A Rat Osteoporosis Model. Int. J. Mol. Sci. 2020, 21, 1235. [Google Scholar] [CrossRef]
- Jimbo, R.; Ono, D.; Hirakawa, Y.; Odatsu, T.; Tanaka, T.; Sawase, T. Accelerated photo-induced hydrophilicity promotes osseointegration: An animal study. Clin. Implant Dent. Relat. Res. 2011, 13, 79–85. [Google Scholar] [CrossRef]
- Hayashi, M.; Jimbo, R.; Xue, Y.; Mustafa, K.; Andersson, M.; Wennerberg, A. Photocatalytically induced hydrophilicity influences bone remodelling at longer healing periods: A rabbit study. Clin. Oral Implants Res. 2014, 25, 749–754. [Google Scholar] [CrossRef]
- Yamazaki, M.; Yamada, M.; Ishizaki, K.; Sakurai, K. Ultraviolet-C irradiation to titanium implants increases peri-implant bone formation without impeding mineralization in a rabbit femur model. Acta Odontol. Scand. 2015, 73, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Lee, J.I.; Yang, S.S.; Kim, B.S.; Kim, B.C.; Lee, J. The effect of alendronate soaking and ultraviolet treatment on bone-implant interface. Clin. Oral Implants Res. 2017, 28, 1164–1172. [Google Scholar] [CrossRef]
- Lee, J.B.; Jo, Y.H.; Choi, J.Y.; Seol, Y.J.; Lee, Y.M.; Ku, Y.; Rhyu, I.C.; Yeo, I.S.L. The effect of ultraviolet photofunctionalization on a titanium dental implant with machined surface: An in vitro and in vivo study. Materials 2019, 12, 2078. [Google Scholar] [CrossRef] [PubMed]
- Miki, T.; Matsuno, T.; Hashimoto, Y.; Miyake, A.; Satomi, T. In vitro and in vivo evaluation of titanium surface modification for biological aging by electrolytic reducing ionic water. Appl. Sci. 2019, 9, 713. [Google Scholar] [CrossRef]
- Hirakawa, Y.; Jimbo, R.; Shibata, Y.; Watanabe, I.; Wennerberg, A.; Sawase, T. Accelerated bone formation on photo-induced hydrophilic titanium implants: An experimental study in the dog mandible. Clin. Oral Implants Res. 2013, 24, 139–144. [Google Scholar] [CrossRef]
- Pyo, S.W.; Park, Y.B.; Moon, H.S.; Lee, J.H.; Ogawa, T. Photofunctionalization enhances bone-implant contact, dynamics of interfacial osteogenesis, marginal bone seal, and removal torque value of implants: A dog jawbone study. Implant Dent. 2013, 22, 666–675. [Google Scholar] [CrossRef]
- Ishii, K.; Matsuo, M.; Hoshi, N.; Takahashi, S.S.; Kawamata, R.; Kimoto, K. Effect of ultraviolet irradiation of the implant surface on progression of Periimplantitis—A pilot study in dogs. Implant Dent. 2016, 25, 47–53. [Google Scholar] [CrossRef]
- Mehl, C.; Kern, M.; Neumann, F.; Bähr, T.; Wiltfang, J.; Gassling, V. Effect of ultraviolet photofunctionalization of dental titanium implants on osseointegration. J. Zhejiang Univ. Sci. B 2018, 19, 525–534. [Google Scholar] [CrossRef]
- Funato, A.; Ogawa, T. Photofunctionalized dental implants: A case series in compromised bone. Int. J. Oral Maxillofac. Implants 2013, 28, 1589–1601. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Kobayashi, H.; Ogawa, T. Implant stability change and osseointegration speed of immediately loaded photofunctionalized implants. Implant Dent. 2013, 22, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Funato, A.; Yamada, M.; Ogawa, T. Success rate, healing time, and implant stability of photofunctionalized dental implants. Int. J. Oral Maxillofac. Implants. 2013, 28, 1261–1271. [Google Scholar] [CrossRef]
- Kitajima, H.; Ogawa, T. The use of photofunctionalized implants for low or extremely low primary stability cases. Int. J. Oral Maxillofac. Implants 2016, 31, 439–447. [Google Scholar] [CrossRef]
- Hirota, M.; Ozawa, T.; Iwai, T.; Ogawa, T.; Tohnai, I. Implant stability development of photofunctionalized implants placed in regular and complex cases: A case-control study. Int. J. Oral Maxillofac. Implants 2016, 31, 676–686. [Google Scholar] [CrossRef]
- Hirota, M.; Ozawa, T.; Iwai, T.; Ogawa, T.; Tohnai, I. Effect of Photofunctionalization on Early Implant Failure. Int. J. Oral Maxillofac. Implants 2018, 33, 1098–1102. [Google Scholar] [CrossRef] [PubMed]
- Puisys, A.; Schlee, M.; Linkevicius, T.; Petrakakis, P.; Tjaden, A. Photo-activated implants: A triple-blinded, split-mouth, randomized controlled clinical trial on the resistance to removal torque at various healing intervals. Clin. Oral Investig. 2020, 24, 1789–1799. [Google Scholar] [CrossRef]
- Annunziata, M.; Canullo, L.; Donnarumma, G.; Caputo, P.; Nastri, L.; Guida, L. Bacterial inactivation/sterilization by argon plasma treatment on contaminated titanium implant surfaces: In vitro study. Med. Oral Patol. Oral Cir. Bucal 2016, 1, e118–e121. [Google Scholar] [CrossRef]
- Canullo, L.; Genova, T.; Pesce, P.; Nakajima, Y.; Yonezawa, D.; Mussano, F. Surface bio-functionalization using plasma of Argon could alter microbiologicaland topographic surface analysis of dental implants? Ann. Anat. 2020, 230, 151489. [Google Scholar] [CrossRef]
- Genova, T.; Pesce, P.; Mussano, F.; Tanaka, K.; Canullo, L. The influence of bone-graft biofunctionalization with plasma of argon on bacterial contamination. J. Biomed. Mater. Res. Part. A 2019, 107, 67–70. [Google Scholar] [CrossRef]
- Canullo, L.; Genova, T.; Rakic, M.; Sculean, A.; Miron, R.; Muzzi, M.; Carossa, S.; Mussano, F. Effects of argon plasma treatment on the osteoconductivity of bone grafting materials. Clin. Oral Investig. 2020, 24, 2611–2623. [Google Scholar] [CrossRef] [PubMed]
- Hirota, A.; Yamada, Y.; Canullo, L.; Xavier, S.P.; Baba, S. Bioactivation of bovine bone matrix using argon plasma: An experimental study for sinus augmentation in rabbits. Int. J. Oral Maxillofac. Implants 2020, 35, 731–738. [Google Scholar] [CrossRef] [PubMed]
- Makary, C.; Rebaudi, A.; Sammartino, G.; Naaman, N. Implant primary stability determined by resonance frequency analysis: Correlation with insertion torque, histologic bone volume, and torsional stability at 6 weeks. Implant Dent. 2012, 21, 474–480. [Google Scholar] [CrossRef] [PubMed]

| Study Title | Reference | Year | No of Animals | Specimens | Surface Treatment | Plasma Argon Treatment | Follow-Up | Outcome | Results |
|---|---|---|---|---|---|---|---|---|---|
| Argon-based atmospheric pressure plasma enhances early bone response to rough titanium surfaces | Coelho PG et al. [21] | 2012 | Six dogs (adult beagle) | Two Ti-6Al-4V implants each side. The different implant surfaces (Ti or Ti-Plasma) were alternately placed from proximal to distal at distances of 1 cm from each other along the central region of the bone. | Alumina- blasted/acid-etched | Atmospheric pressure plasma (CaP-plasma) treatment with Ar gas for a period of 60 s per quadrant with a KinPenTM device | 1 and 3 weeks | BIC (%) BAFO (bone area fraction occupacy) | No significant difference was found for BIC and BAFO between surfaces at 1 week. At 3 weeks in vivo, bone formation in close contact to the implant surface (BIC) was strongly observed in the Ti-plasma group, where an increase of over 300% was observed when compared to the control (p < 0.001). No significant differences were observed in BAFO (p > 0.14), although an improvement of 30% was observed for the Ti-plasma group |
| Assessment of a chair-side argon-based non-thermal plasma treatment on the surface characteristics and integration of dental implants with textured surfaces | Teixeira H et al. [22] | 2012 | Six dogs (adult beagle) | Three root-form endosseous grade IV titanium alloy implants placed into each limb. Test: 20 sand 60 s plasma-treated implants; control: untreated implants. | Alumina-blasted and acid-etched surface | Twenty or sixty seconds of non-thermal plasma per quadrant applied with a KinPenTM device | 2 and 4 weeks | Removal torque (Ncm) | Torque value at 2 weeks: control: 35; plasma: 20 s—43; plasma: 60 s—55. Torque value at 4 weeks: control: 43; plasma: 20 s—67; plasma 60 s—72. |
| Osseointegration assessment of chairside argon-based non thermal plasma-treated Ca-P coated dental implant | Giro G et al. [23] | 2013 | Six dogs (adult beagle) | Two Ti-6Al-4V implants each side. Different implant surfaces were alternately placed from proximal to distal at distances of 1 cm from each other along the central region of the bone, and the start surface site (CaP or CaP-Plasma) was alternated between animals. The implant distribution resulted in an equal number of implants for 1 and 3 weeks. | Calcium-phosphate (CaP) | Ar gas at atmospheric pressure for a period of 20 s per quadrant with a KinPenTM device | 1 and 3 weeks | BIC (%) BAFO (bone area graction occupacy) | No significant difference was found for BIC and BAFO between surfaces at 1 week. At 3 weeks, BIC and BAFO were strongly observed in the CaP-plasma group. The morphologic findings for both 1 and 3 weeks were supported by the morphometric results at the 3-week period, as CaP-plasma BIC increased by more than 100% and an improvement of 82% was found for BAFO when compared to the CaP group. |
| Hard and soft tissue changes around implants activated using plasma of argon: a histomorphometric study in dog | Canullo L et al. [24] | 2018 | Eight dogs (adult beagle) | For each hemi-mandible, four implants with a ZirTi surface were used; two implants were treated with argon plasma (test), while the other two implants were left untreated (control). | ZirTi surface | Treated for 12 min at room temperature with plasma of argon in a plasma reactor (Diener electronic) | 1 and 2 months | BIC (%) old bone total amount of mineralized bone | One month of healing: new bone in close contact with the implant surface: treated (60.1% ± 15.6%; 95% CI 56.5%–78.0%); untreated (57.2% ± 13.1%; 95% CI 49.3%–67.5%) (p = 0.400). Old bone: treated (4.4% ± 3.0%; 95% CI. 2%–5.4%); untreated (3.4% ± 3.1%; 95% CI. 6%–4.9%) (p = 726). Total amount of mineralized bone: treated (95% CI 59.5%–82.3%); untreated (95% CI 53.3%–73.5%) (p = 0.208). Two months of healing of new bone: treated sites: 72.5% ± 12.4% (95% CI 69.6%–86.8%); untreated: 64.7% ± 17.3% (95% CI 59.4%–83.3% (p = 0.012). Old bone: treated sites: 3.1% ± 1.7% (95% CI 1.8%–4.2%); untreated sites: 3.8% ± 1.9% (95% CI 3.2%–5.8%) (p = 0.270). Total amount of mineralized bone: treated: 75.6% ± 13.0% (95% CI 73.3%–91.3%); untreated 68.4% ± 16.8% (95% CI 64.2%–87.6%). |
| Effects of non thermal plasma on sandblasted titanium dental implants in beagle dogs | Hung YW et al. [25] | 2018 | Nine dogs (adult beagle) | Four implants in each dog; control group: one implant withot non-thermal plasma was inserted into each jaw; test group: one implant treated with non-thermal plasma was inserted into each jaw. | Sandblasting and etching | Non-thermal plasma apparatus (Line through ISO 9001) generates plasma in a dielectric barrier. Each implant receive 60s of plasma spray | 4,8 and 12 weeks | ISQ Value | ISQ values: Control group: Initial: 68.04 ± 3.37 4 weeks: 66.53 ± 7.40 8 weeks: 69.20 ± 2.55 12 weeks: 74.20 ± 2.68 Plasma group: Initial: 67.36 ± 0.52 4 weeks: 70.17 ± 0.76 8 weeks: 71.50 ± 1.41 12 weeks: 77.00 ± 5.87 |
| Study Title | Reference | Year | No of Animals | Specimens | Surface Treatment | UV Treatment | Follow-Up | Outcome | Results |
|---|---|---|---|---|---|---|---|---|---|
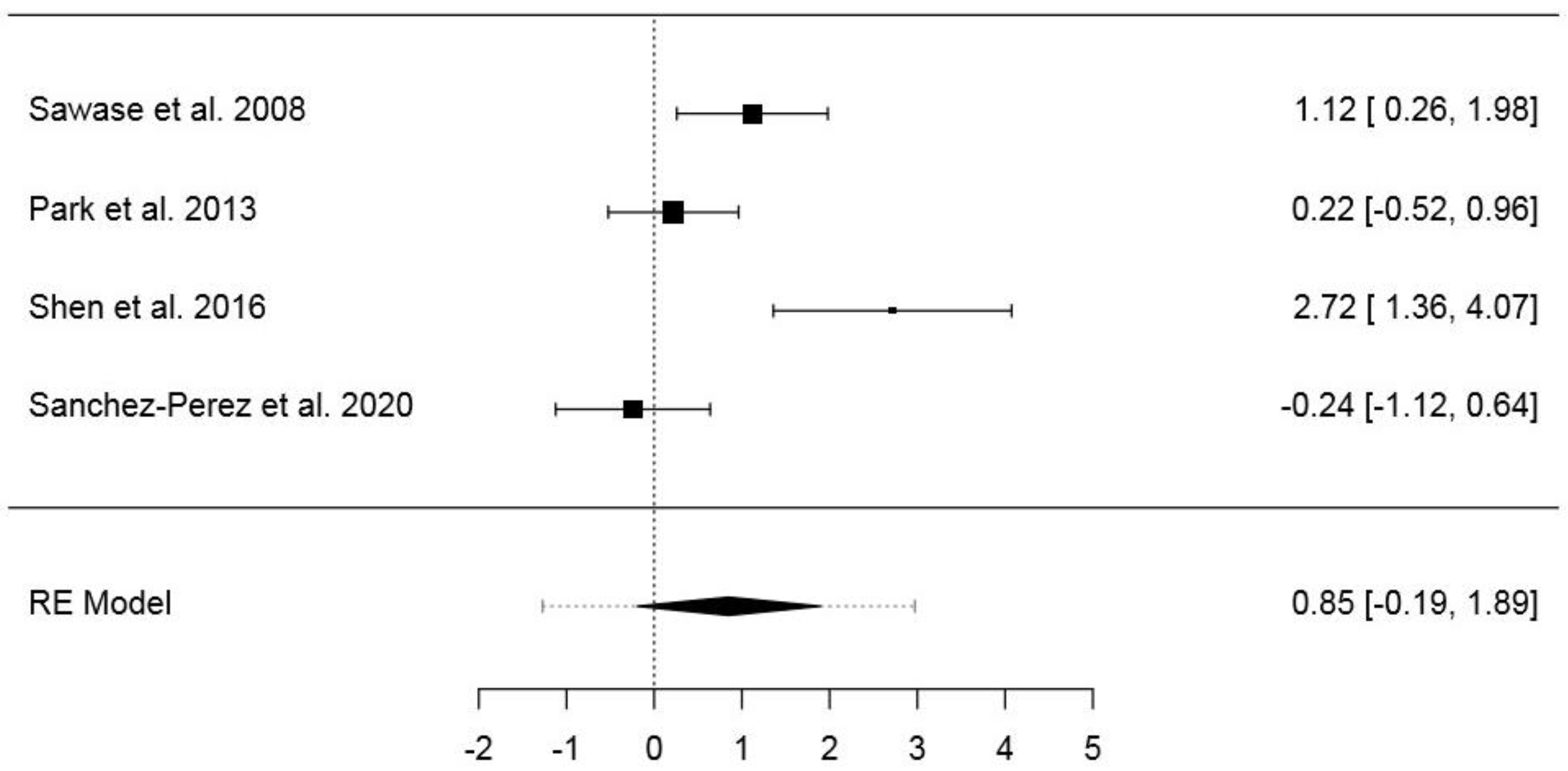
| Photo-induced hydrophilicity enhances initial cell behavior and early bone apposition. | Sawase, T et al. [26] | 2008 | Six rabbits (tibia) | One implant each side of the tibia; cpTi screw implants (Nobel Biocare RP Mark III fixtures; Nobel Biocare AB, Göteborg, Sweden). | Post-annealed from the titanium implant; tetraisoproxide plasma by the plasma source; ion implantation | UV irradiation for 24 h | 2 weeks | BIC (%) | BIC untreated: 17.97%; BIC UV: treated 28.2%. |
| The effect of ultraviolet C irradiation via a bactericidal ultraviolet sterilizer on an anodized titanium implant. A study in rabbits | Park K.H et al. [27] | 2013 | Fourteen rabbits (tibia) | Twenty-five titanium discs and 56 screw tipe implants. Each rabbits received four control or test implants (UV treated). | Anodized implants | UV irradiation via a 15W lamp for 24 h | 4 and 12 weeks | BIC (%) | Four-week mean value: control group (12): 42.92%; test group (12): 55.11%. Twelve-week value: control group (14): 55.81%; test group (14): 57.78%. |
| The in vivo bone response of ultraviolet-irradiated titanium implants modified with spontaneusly formed nanostructures | Shen J et al. [20] | 2016 | Forty rabbits (femur and tibia) | A total of 160 screw-shaped implants divided in 5 groups: (1) SLA new (2) SLA old (3) modified SLA (4) UV SLA (5) UV modified SLA. | Sandblasted and acid-etched | UV irradiation via a 15W bactericidial lamp for 24 h | 3 and 6 weeks | BIC (%) RT (removal torque) | BIC mean value at 3 weeks: group (1): 40.05% group (2): 30.2% group (3): 35.3% group (4): 59.6% group (5): 61.8% BIC mean value at 6 weeks: group (1): 41.6% group (2): 31.3% group (3): 39.3% group (4): 69.5% group (5): 72.0% Torque removal mean at value 3 weeks: group (1): 42 group (2): 30 group (3): 39 group (4): 70 group (5): 90 Toque removal mean value at 6 weeks: group (1): 70 group (2): 42 group (3): 60 group (4): 82 group (5): 105 |
| Photofunctionalised Ti6Al4V implants enhance early phase osseointegration. | Yamauchi, R et al. [28] | 2017 | Five rats (femur) | One implant each side; implant made from pure Ti and Ti6Al4V (B. Braun Aesculap Japan Co., Ltd. Tokyo, Japan). | Specimens: pure Ti and Ti6Al4V with average surface roughness values of 0.66 and 0.34 μm, respectively | Exposure to UV irradiation for 15 min using a photo device (TheraBeam Affinity; Ushio Inc., Tokyo, Japan) at an intensity of 3 mW/cm2 | 2 and 4 weeks | BIC value (BV/TV %) | Pure Ti value: Untreated 2-week value: 39.8%; treated 2-week value: 56.8%. Untreated 4-week value: 61.6%; treated 4-week value: 80.7%. Ti6Al4V value: Untreated 2-week value: 44.4%; treated 2-week value: 65.0%; untreated 4-week value: 58.6%; treated 4-week value: 76.3%. |
| Effects of ultraviolet Photoactivation on osseointegration of commercial pure titanium dental implant after 8 weeks in a rabbit model | Sanchez-Perez A et al. [29] | 2020 | Five rabbits | Twenty commercial implants. | Group 1: as received; group 2: UV treated | A 6W UVC source for 15 min (Analizer VL 6c) | 8 weeks | BIC (%) | BIC mean value: Control group: 26.835%; test group 24.225%. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pesce, P.; Menini, M.; Santori, G.; Giovanni, E.D.; Bagnasco, F.; Canullo, L. Photo and Plasma Activation of Dental Implant Titanium Surfaces. A Systematic Review with Meta-Analysis of Pre-Clinical Studies. J. Clin. Med. 2020, 9, 2817. https://doi.org/10.3390/jcm9092817
Pesce P, Menini M, Santori G, Giovanni ED, Bagnasco F, Canullo L. Photo and Plasma Activation of Dental Implant Titanium Surfaces. A Systematic Review with Meta-Analysis of Pre-Clinical Studies. Journal of Clinical Medicine. 2020; 9(9):2817. https://doi.org/10.3390/jcm9092817
Chicago/Turabian StylePesce, Paolo, Maria Menini, Gregorio Santori, Emanuele De Giovanni, Francesco Bagnasco, and Luigi Canullo. 2020. "Photo and Plasma Activation of Dental Implant Titanium Surfaces. A Systematic Review with Meta-Analysis of Pre-Clinical Studies" Journal of Clinical Medicine 9, no. 9: 2817. https://doi.org/10.3390/jcm9092817
APA StylePesce, P., Menini, M., Santori, G., Giovanni, E. D., Bagnasco, F., & Canullo, L. (2020). Photo and Plasma Activation of Dental Implant Titanium Surfaces. A Systematic Review with Meta-Analysis of Pre-Clinical Studies. Journal of Clinical Medicine, 9(9), 2817. https://doi.org/10.3390/jcm9092817








