Effects of Exercise on Cognitive Performance in Children and Adolescents with ADHD: Potential Mechanisms and Evidence-based Recommendations
Abstract
1. ADHD, Cognitive Functions and the Ameliorating Effects of Exercise
1.1. ADHD is Associated with Lower Performance in Cognitive Tests
1.2. Exercise Benefits Performance in Neurocognitive Tests
1.2.1. Acute Exercise Affects Performance in Tests of Cognitive Functions
1.2.2. Association between Fitness or Physical Activity Level and Cognitive Functions
1.2.3. Long-Term Exercise Improves Performance in Cognitive Tests
1.2.4. Meta-Analyses Suggest Beneficial Effects of Exercise on Cognitive Functions in Children with ADHD
2. The Neurophysiology of ADHD and how Exercise may Exert Beneficial Effects
2.1. Brain Structure Abnormalities Associated with Pediatric ADHD and the Effects of Exercise
2.1.1. ADHD Is Associated with Structural Cortical, Cerebellar and Subcortical Abnormalities
2.1.2. Exercise Leads to Structural Changes in the CNS
2.2. Abnormalities Associated with Neuronal Network Activity in Pediatric ADHD and the Effects of Exercise
2.2.1. ADHD is Associated with Altered Activity in Networks across the Brain
2.2.2. Exercise Leads to Changes in Network Activity
2.3. ADHD, Exercise and Biochemical Changes in the Brain
2.3.1. ADHD is Associated with Abnormal Monoaminergic Signaling
Dopamine and Reward-Related Processing
Control of Attention and Monoaminergic Signaling Systems
2.3.2. Exercise Changes Monoaminergic Signaling
2.3.3. ADHD is Associated with Abnormal BDNF Signaling
2.3.4. Exercise Changes BDNF Signaling
3. Exploring the Parameter-Space of Exercise Characteristics: Effects of Exercise on Cognitive Performance in Children and Adolescents with ADHD
3.1. Effects of Acute vs. Long-Term Exercise on Cognitive Functions
3.2. Effects of Exercise on Performance in Specific Cognitive Domains
3.3. Exercise Intensity, Duration and Volume as Potential Moderators of Effects on Cognitive Functions
3.4. Type of Exercise—Modality—as Potential Moderator of Effects on Cognitive Functions
3.5. Recommendations
3.5.1. Science and Evidence-Based Recommendations
3.5.2. Directions for Future Research
Reporting Exercise Characteristics
Study Design
Outcome Measures
4. Conclusions and Perspectives
Author Contributions
Conflicts of Interest
Appendix A
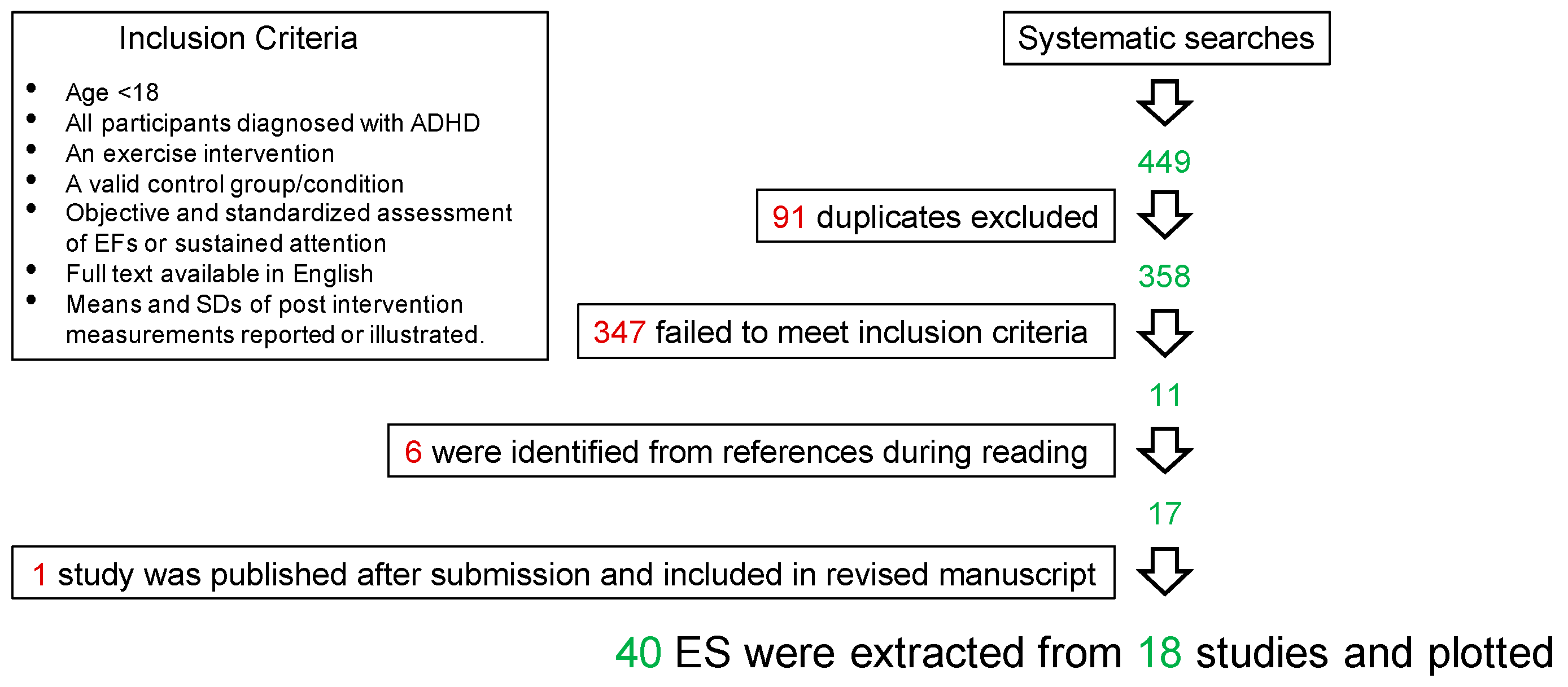
Systematic Search and Extraction of Effect Sizes from Previous Studies
and for Web of Science:(“exercise” [Title] OR “physical activity” [Title] OR “fitness” [Title] OR “physical exercise” [Title] OR “acute exercise” [Title] OR “chronic exercise” [Title] OR “aerobic” [Title] OR “resistance” [Title] OR “anaerobic” [Title] OR ”coordinative” OR “training”) AND (“attention deficit hyperactivity disorder” [Title] OR “ADHD” [Title] OR “attention deficit disorder” [Title] OR “hyperkin*” [Title] OR attention-deficit/hyperactivity disorder [Title]) AND (“child*” [Title] OR “young” [Title] OR “adolescent” [Title] OR “teenagers” [Title] OR “student*” [Title]) AND (“executive function” [Title] OR “executive functions” [Title] OR “inhibition” [Title] OR “interference” [Title] OR “cognitive control” [Title] OR “inhibitory control” [Title] OR “flexibility” [Title] OR “working memory” [Title] OR “switching” [Title] OR “shifting” [Title] OR “sustained attention” [Title] OR “attention” [Title])
TI = (“exercise” OR “physical activity” OR “fitness” OR “physical exercise” OR “acute exercise” OR “chronic exercise” OR “aerobic” OR “resistance” OR “anaerobic” OR ”coordinative” OR “training”) AND TI = (“attention deficit hyperactivity disorder” OR “ADHD” OR “attention deficit disorder” OR “hyperkin*” OR attention-deficit/hyperactivity disorder) AND TI = (“child*” OR “young” OR “adolescent” OR “teenagers” OR “student*”) AND TI = (“executive function” OR “executive functions” OR “inhibition” OR “interference” OR “cognitive control” OR “inhibitory control” OR “flexibility” OR “working memory” OR “switching” OR “shifting” OR “sustained attention” OR “attention”).
| Study [#ref] | Type of Task | Included Measures | Cognitive Domain | Not Included | Extraction Method |
|---|---|---|---|---|---|
| Benzing et al., 2018 [77] | Modified Flanker Task, Modified Colour Span Backwards Task | Congruent RT, Inhibitory (incongruent) RT, Global Switch RT cost, Correct responses | PS, INH, CF, WM | Congruent accuracy, incongruent accuracy, switch accuracy, Global switch accuracy cost | Mean ± SD reported |
| Chang et al., 2012 [81] | Stroop, Wisconsin Card Sorting Task | Colour-word, perseverative errors | INH, CF | Word, Colour, total correct, perseverative responses, Non-Perseverative errors, Conceptual level, responses, categories completed, | Mean ± SD reported |
| Chuang et al., 2015 [75] | Go/No-Go | RT, Commission Error rate | PS, INH | Hit rate, Omission Error rate | Mean ± SD reported |
| Gawrilow et al., 2016 [74] | Modified Go/No-Go | Successful No-Go | INH | Overall errors on go-trials | Mean ± SD reported |
| Hung et al., 2016 [82] | Task-Switching Paradigm | Global Switch RT pure, Global Switch RT cost, Local Switch RT cost | PS, WM, CF | Global switch RT Mixed, Local Non Switch RT, Local Switch RT, Global Switch Accuracy Mixed, Local Switch RT Global Switch accuracy Pure, Local Non- Switch accuracy, local switch Accuracy, Global Switch Accuracy Cost, Local Switch Accuracy cost | Mean ± SD reported |
| Ludyga et al., 2018 [83] | Alternate Use | Fluency, Flexibility | CF, CF | Originality, Elaboration | Mean ± SD reported |
| Ludyga et al., 2017 [76] | Modified Flanker Task | RT congruent, RT incongruent | PS, INH | NA | Mean ± SD reported |
| Medina et al., 2010 [363] | Connor’s Continuous Performance Test II | Commission errors, Hit RT, Hit RT block Change | INH, PS, SA | Omission errors, Hit RT Standard Error, Variability, Detectability, Response style, Hit RT ISI change, Hit Standard Error interstimulus change, Perseverations, Hit standard error Block | Mean ± SD reported |
| Study [#ref] | Type of Task | Included Measures | Cognitive Domain | Not Included | Extraction Method |
|---|---|---|---|---|---|
| Benzing & Schmidt 2019 [398] | Simon Task, modified Flanker Task, Colour Span Backwards task | Switch trials RT, correct Responses | CF, WM | RT across trials | Mean ± SD reported |
| Chang et al., 2014 [115] | Go/No-Go | Accuracy no-go trials | INH | Accuracy go trials | Mean ± SD from figures. |
| Choi et al., 2015 [114] | Wisconsin Card Sorting Test | Perseverative Errors | CF | NA | Mean ± SD reported |
| Kang et al., 2011 [49] | Trail Making Test part b | Time to completion | CF | Digit Symbol Task | Mean ± SD from figures |
| Lee et al., 2017 [357] | Stroop | Interference | INH | Colour-Word | Mean ± SD reported |
| Memarmoghaddam et al., 2016 [112] | Stroop, Go/No-Go | Consistent RT, Interference, No-Go true number, True RT | PS, INH, INH, PS | Consistent and inconcistent error number, consistent and inconcistent no reponses, consistent and inconcistent true number, inconsistent RT, Go and no-go true number, Go and no-go error number, error RT | Mean ± SD reported |
| Pan et al., 2015 [111] | Stroop, Wisconsin Card Sorting Test | Colour-word, Perseverative errors | INH, CF | Total correct, perseverative responses, non-perseverative errors, conceptual levels, Responses, categories completed | Mean ± SD reported |
| Pan et al., 2016 [110] | Stroop | Colour word | INH | NA | Mean ± SD reported |
| Verret et al., 2012 [109] | Sky Search | Score pondering, walk/don’t walk pondering | SA, INH | Time targeted pondering, attention pondering, sky search DT pondering | Mean ± SD reported |
| Ziereis & Jansen 2015 [360] | Digit Span, Letter-Number Sequencing | Digit span Index score, Letter-number sequencing index score | WM, WM | Backwards digit span, forward digit span | Mean ± SD from figures |
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®); American Psychiatric Pub: Washington, DC, USA, 2013. [Google Scholar]
- Colvin, M.K.; Stern, T.A. Diagnosis, evaluation, and treatment of attention-deficit/hyperactivity disorder. J. Clin. Psychiatry 2015, 76, e1148. [Google Scholar] [CrossRef]
- Polanczyk, G.; De Lima, M.S.; Horta, B.L.; Biederman, J.; Rohde, L.A. The worldwide prevalence of ADHD: A systematic review and metaregression analysis. Am. J. Psychiatry 2007, 164, 942–948. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Sanders, S.; Doust, J.; Beller, E.; Glasziou, P. Prevalence of attention-deficit/hyperactivity disorder: A systematic review and meta-analysis. Pediatrics 2015, 135, e994–e1001. [Google Scholar] [CrossRef] [PubMed]
- Fayyad, J.; Sampson, N.A.; Hwang, I.; Adamowski, T.; Aguilar-Gaxiola, S.; Al-Hamzawi, A.; Andrade, L.H.; Borges, G.; de Girolamo, G.; Florescu, S. The descriptive epidemiology of DSM-IV Adult ADHD in the world health organization world mental health surveys. Adhd Atten. Deficit Hyperact. Disord. 2017, 9, 47–65. [Google Scholar] [CrossRef]
- Coelho, L.; Chaves, E.; Vasconcelos, S.; Fonteles, M.; De Sousa, F.; Viana, G. Attention Deficit Hyperactivity Disorder (ADHD) in children: Neurobiological aspects, diagnosis and therapeutic approach. Acta Med. Port. 2010, 23, 689–696. [Google Scholar] [PubMed]
- Barkley, R.A.; Fischer, M. Hyperactive Child Syndrome and Estimated Life Expectancy at Young Adult Follow-Up: The Role of ADHD Persistence and Other Potential Predictors. J. Atten. Disord. 2018. [Google Scholar] [CrossRef] [PubMed]
- Rydén, E.; Thase, M.E.; Stråht, D.; Åberg-Wistedt, A.; Bejerot, S.; Landén, M. A history of childhood attention-deficit hyperactivity disorder (ADHD) impacts clinical outcome in adult bipolar patients regardless of current ADHD. Acta Psychiatr. Scand. 2009, 120, 239–246. [Google Scholar] [CrossRef] [PubMed]
- Biederman, J.; Wilens, T.; Mick, E.; Milberger, S.; Spencer, T.J.; Faraone, S.V. Psychoactive substance use disorders in adults with attention deficit hyperactivity disorder (ADHD): Effects of ADHD and psychiatric comorbidity. Am. J. Psychiatry 1995, 152, 1652–1658. [Google Scholar] [PubMed]
- Peasgood, T.; Bhardwaj, A.; Biggs, K.; Brazier, J.E.; Coghill, D.; Cooper, C.L.; Daley, D.; De Silva, C.; Harpin, V.; Hodgkins, P. The impact of ADHD on the health and well-being of ADHD children and their siblings. Eur. Child Adolesc. Psychiatry 2016, 25, 1217–1231. [Google Scholar] [CrossRef]
- Childress, A.C.; Sallee, F.R. Attention-deficit/hyperactivity disorder with inadequate response to stimulants: Approaches to management. CNS Drugs 2014, 28, 121–129. [Google Scholar] [CrossRef]
- Shyu, Y.-C.; Yuan, S.-S.; Lee, S.-Y.; Yang, C.-J.; Yang, K.-C.; Lee, T.-L.; Wang, L.-J. Attention-deficit/hyperactivity disorder, methylphenidate use and the risk of developing schizophrenia spectrum disorders: A nationwide population-based study in Taiwan. Schizophr. Res. 2015, 168, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Raga, J.; Knecht, C.; Szerman, N.; Martinez, M.I. Risk of serious cardiovascular problems with medications for attention-deficit hyperactivity disorder. CNS Drugs 2013, 27, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Fusar-Poli, P.; Rubia, K.; Rossi, G.; Sartori, G.; Balottin, U. Striatal Dopamine Transporter Alterations in ADHD: Pathophysiology or Adaptation to Psychostimulants? A Meta-Anal. Am. J. Psychiatry 2012, 169, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.-J.; Volkow, N.D.; Wigal, T.; Kollins, S.H.; Newcorn, J.H.; Telang, F.; Logan, J.; Jayne, M.; Wong, C.T.; Han, H.; et al. Long-Term Stimulant Treatment Affects Brain Dopamine Transporter Level in Patients with Attention Deficit Hyperactive Disorder. PLoS ONE 2013, 8, e63023. [Google Scholar] [CrossRef]
- Scherer, E.B.S.; da Cunha, M.J.; Matté, C.; Schmitz, F.; Netto, C.A.; Wyse, A.T. Methylphenidate affects memory, brain-derived neurotrophic factor immunocontent and brain acetylcholinesterase activity in the rat. Neurobiol. Learn. Mem. 2010, 94, 247–253. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc-Duchin, D.; Taukulis, H.K. Chronic oral methylphenidate administration to periadolescent rats yields prolonged impairment of memory for objects. Neurobiol. Learn. Mem. 2007, 88, 312–320. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc-Duchin, D.; Taukulis, H.K. Chronic oral methylphenidate induces post-treatment impairment in recognition and spatial memory in adult rats. Neurobiol. Learn. Mem. 2009, 91, 218–225. [Google Scholar] [CrossRef]
- Taylor, M.J.; Martin, J.; Lu, Y.; Brikell, I.; Lundström, S.; Larsson, H.; Lichtenstein, P. Association of genetic risk factors for psychiatric disorders and traits of these disorders in a swedish population twin sample. JAMA Psychiatry 2018. [Google Scholar] [CrossRef]
- Levy, F.; Hay, D.A.; McSTEPHEN, M.; Wood, C.; Waldman, I. Attention-deficit hyperactivity disorder: A category or a continuum? Genetic analysis of a large-scale twin study. J. Am. Acad. Child Adolesc. Psychiatry 1997, 36, 737–744. [Google Scholar] [CrossRef]
- Henrik, L.; Henrik, A.; Maria, R.; Zheng, C.; Paul, L. Childhood attention-deficit hyperactivity disorder as an extreme of a continuous trait: A quantitative genetic study of 8,500 twin pairs. J. Child Psychol. Psychiatry 2012, 53, 73–80. [Google Scholar]
- Lubke, G.H.; Hudziak, J.J.; Derks, E.M.; van Bijsterveldt, T.C.E.M.; Boomsma, D.I. Maternal Ratings of Attention Problems in ADHD: Evidence for the Existence of a Continuum. J. Am. Acad. Child Adolesc. Psychiatry 2009, 48, 1085–1093. [Google Scholar] [CrossRef]
- Middeldorp, C.M.; Hammerschlag, A.R.; Ouwens, K.G.; Groen-Blokhuis, M.M.; Pourcain, B.S.; Greven, C.U.; Pappa, I.; Tiesler, C.M.; Ang, W.; Nolte, I.M. A genome-wide association meta-analysis of attention-deficit/hyperactivity disorder symptoms in population-based pediatric cohorts. J. Am. Acad. Child Adolesc. Psychiatry 2016, 55, 896–905.e896. [Google Scholar] [CrossRef] [PubMed]
- Hillman, C.H.; Erickson, K.I.; Kramer, A.F. Be smart, exercise your heart: Exercise effects on brain and cognition. Nat. Rev. Neurosci. 2008, 9, 58. [Google Scholar] [CrossRef] [PubMed]
- Penedo, F.J.; Dahn, J.R. Exercise and well-being: A review of mental and physical health benefits associated with physical activity. Curr. Opin. Psychiatry 2005, 18, 189–193. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A. Executive functions. Annu. Rev. Psychol. 2013, 64, 135–168. [Google Scholar] [CrossRef] [PubMed]
- Coghill, D.R.; Seth, S.; Matthews, K. A comprehensive assessment of memory, delay aversion, timing, inhibition, decision making and variability in attention deficit hyperactivity disorder: Advancing beyond the three-pathway models. Psychol. Med. 2013, 44, 1989–2001. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, M.J.; Faraone, S.V.; Leon, T.L.; Biederman, J.; Spencer, T.J.; Adler, L.A. The Relationship Between Executive Function Deficits and DSM-5-Defined ADHD Symptoms. J. Atten. Disord. 2018. [Google Scholar] [CrossRef] [PubMed]
- Willcutt, E.G.; Doyle, A.E.; Nigg, J.T.; Faraone, S.V.; Pennington, B.F. Validity of the Executive Function Theory of Attention-Deficit/Hyperactivity Disorder: A Meta-Analytic Review. Biol. Psychiatry 2005, 57, 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Miyake, A.; Friedman, N.P.; Emerson, M.J.; Witzki, A.H.; Howerter, A.; Wager, T.D. The unity and diversity of executive functions and their contributions to complex “frontal lobe” tasks: A latent variable analysis. Cogn. Psychol. 2000, 41, 49–100. [Google Scholar] [CrossRef]
- Sonuga-Barke, E.J.S. Psychological heterogeneity in AD/HD—A dual pathway model of behaviour and cognition. Behav. Brain Res. 2002, 130, 29–36. [Google Scholar] [CrossRef]
- Wasserman, L.D. The Sensitivity and Specificity of Neuropsychological Tests in the Diagnosis of Attention Deficit Hyperactivity Disorder AU—Wasserman, Theodore. Appl. Neuropsychol. Child 2012, 1, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Nigg, J.T.; Willcutt, E.G.; Doyle, A.E.; Sonuga-Barke, E.J.S. Causal Heterogeneity in Attention-Deficit/Hyperactivity Disorder: Do We Need Neuropsychologically Impaired Subtypes? Biol. Psychiatry 2005, 57, 1224–1230. [Google Scholar] [CrossRef] [PubMed]
- Snyder, H.R.; Miyake, A.; Hankin, B.L. Advancing understanding of executive function impairments and psychopathology: Bridging the gap between clinical and cognitive approaches. Front. Psychol. 2015, 6, 328. [Google Scholar] [CrossRef] [PubMed]
- Kofler, M.J.; Rapport, M.D.; Sarver, D.E.; Raiker, J.S.; Orban, S.A.; Friedman, L.M.; Kolomeyer, E.G. Reaction time variability in ADHD: A meta-analytic review of 319 studies. Clin. Psychol. Rev. 2013, 33, 795–811. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, K.; Verhaeghen, P. ADHD and Stroop interference from age 9 to age 41 years: A meta-analysis of developmental effects. Psychol. Med. 2008, 38, 1607–1616. [Google Scholar] [CrossRef] [PubMed]
- Metin, B.; Roeyers, H.; Wiersema, J.R.; van der Meere, J.; Sonuga-Barke, E. A Meta-Analytic Study of Event Rate Effects on Go/No-Go Performance in Attention-Deficit/Hyperactivity Disorder. Biol. Psychiatry 2012, 72, 990–996. [Google Scholar] [CrossRef] [PubMed]
- Lijffijt, M.; Kenemans, J.L.; Verbaten, M.N.; van Engeland, H. A meta-analytic review of stopping performance in attention-deficit/hyperactivity disorder: Deficient inhibitory motor control? J. Abnorm. Psychol. 2005, 114, 216. [Google Scholar] [CrossRef] [PubMed]
- Pauli-Pott, U.; Becker, K. Neuropsychological basic deficits in preschoolers at risk for ADHD: A meta-analysis. Clin. Psychol. Rev. 2011, 31, 626–637. [Google Scholar] [CrossRef] [PubMed]
- Wells, E.L.; Kofler, M.J.; Soto, E.F.; Schaefer, H.S.; Sarver, D.E. Assessing working memory in children with ADHD: Minor administration and scoring changes may improve digit span backward’s construct validity. Res. Dev. Disabil. 2018, 72, 166–178. [Google Scholar] [CrossRef]
- Kofler, M.J.; Irwin, L.N.; Soto, E.F.; Groves, N.B.; Harmon, S.L.; Sarver, D.E. Executive Functioning Heterogeneity in Pediatric ADHD. J. Abnorm. Child Psychol. 2018, 1–14. [Google Scholar] [CrossRef]
- Hervey, A.S.; Epstein, J.N.; Curry, J.F.; Tonev, S.; Eugene Arnold, L.; Keith Conners, C.; Hinshaw, S.P.; Swanson, J.M.; Hechtman, L. Reaction time distribution analysis of neuropsychological performance in an ADHD sample. Child Neuropsychol. 2006, 12, 125–140. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, F.X.; Sonuga-Barke, E.J.; Scheres, A.; Di Martino, A.; Hyde, C.; Walters, J.R. Varieties of attention-deficit/hyperactivity disorder-related intra-individual variability. Biol. Psychiatry 2005, 57, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Klein, C.; Wendling, K.; Huettner, P.; Ruder, H.; Peper, M. Intra-subject variability in attention-deficit hyperactivity disorder. Biol. Psychiatry 2006, 60, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Sonuga-Barke, E.J.; Castellanos, F.X. Spontaneous attentional fluctuations in impaired states and pathological conditions: A neurobiological hypothesis. Neurosci. Biobehav. Rev. 2007, 31, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Alderson, R.M.; Rapport, M.D.; Kofler, M.J. Attention-Deficit/Hyperactivity Disorder and Behavioral Inhibition: A Meta-Analytic Review of the Stop-signal Paradigm. J. Abnorm. Child Psychol. 2007, 35, 745–758. [Google Scholar] [CrossRef] [PubMed]
- Martinussen, R.; Hayden, J.; Hogg-Johnson, S.; Tannock, R. A Meta-Analysis of Working Memory Impairments in Children With Attention-Deficit/Hyperactivity Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Patros, C.H.G.; Alderson, R.M.; Kasper, L.J.; Tarle, S.J.; Lea, S.E.; Hudec, K.L. Choice-impulsivity in children and adolescents with attention-deficit/hyperactivity disorder (ADHD): A meta-analytic review. Clin. Psychol. Rev. 2016, 43, 162–174. [Google Scholar] [CrossRef]
- Kang, K.; Choi, J.; Kang, S.; Han, D. Sports therapy for attention, cognitions and sociality. Int. J. Sports Med. 2011, 32, 953–959. [Google Scholar] [CrossRef]
- Anderson, P. Assessment and Development of Executive Function (EF) During Childhood. Child Neuropsychol. 2002, 8, 71–82. [Google Scholar] [CrossRef]
- Rubia, K. Neuro-anatomic evidence for the maturational delay hypothesis of ADHD. Proc. Natl. Acad. Sci. USA 2007, 104, 19663–19664. [Google Scholar] [CrossRef]
- Berger, I.; Slobodin, O.; Aboud, M.; Melamed, J.; Cassuto, H. Maturational delay in ADHD: Evidence from CPT. Front. Hum. Neurosci. 2013, 7, 691. [Google Scholar] [CrossRef] [PubMed]
- Shaw, P.; Eckstrand, K.; Sharp, W.; Blumenthal, J.; Lerch, J.; Greenstein, D.; Clasen, L.; Evans, A.; Giedd, J.; Rapoport, J. Attention-deficit/hyperactivity disorder is characterized by a delay in cortical maturation. Proc. Natl. Acad. Sci. USA 2007, 104, 19649–19654. [Google Scholar] [CrossRef] [PubMed]
- Colomer, C.; Berenguer, C.; Roselló, B.; Baixauli, I.; Miranda, A. The Impact of Inattention, Hyperactivity/Impulsivity Symptoms, and Executive Functions on Learning Behaviors of Children with ADHD. Front. Psychol. 2017, 8, 540. [Google Scholar] [CrossRef] [PubMed]
- Martel, M.; Nikolas, M.; Nigg, J.T. Executive Function in Adolescents With ADHD. J. Am. Acad. Child Adolesc. Psychiatry 2007, 46, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Biederman, J.; Petty, C.R.; Fried, R.; Doyle, A.E.; Spencer, T.; Seidman, L.J.; Gross, L.; Poetzl, K.; Faraone, S.V. Stability of executive function deficits into young adult years: A prospective longitudinal follow-up study of grown up males with ADHD. Acta Psychiatr. Scand. 2007, 116, 129–136. [Google Scholar] [CrossRef]
- Biederman, J.; Monuteaux, M.C.; Doyle, A.E.; Seidman, L.J.; Wilens, T.E.; Ferrero, F.; Morgan, C.L.; Faraone, S.V. Impact of executive function deficits and attention-deficit/hyperactivity disorder (ADHD) on academic outcomes in children. J. Consult. Clin. Psychol. 2004, 72, 757. [Google Scholar] [CrossRef] [PubMed]
- Tsatsoulis, A.; Fountoulakis, S. The Protective Role of Exercise on Stress System Dysregulation and Comorbidities. Ann. N. Y. Acad. Sci. 2006, 1083, 196–213. [Google Scholar] [CrossRef] [PubMed]
- Binder, E.; Droste, S.K.; Ohl, F.; Reul, J.M.H.M. Regular voluntary exercise reduces anxiety-related behaviour and impulsiveness in mice. Behav. Brain Res. 2004, 155, 197–206. [Google Scholar] [CrossRef]
- McKercher, C.M.; Schmidt, M.D.; Sanderson, K.A.; Patton, G.C.; Dwyer, T.; Venn, A.J. Physical activity and depression in young adults. Am. J. Prev. Med. 2009, 36, 161–164. [Google Scholar] [CrossRef]
- Garcia, D.; Archer, T.; Moradi, S.; Andersson-Arntén, A.-C. Exercise frequency, high activation positive affect, and psychological well-being: Beyond age, gender, and occupation. Psychology 2012, 3, 328. [Google Scholar] [CrossRef]
- Caspersen, C.J.; Powell, K.E.; Christenson, G.M. Physical activity, exercise, and physical fitness: Definitions and distinctions for health-related research. Public Health Rep. 1985, 100, 126–131. [Google Scholar] [PubMed]
- Diamond, A. Effects of physical exercise on executive functions: Going beyond simply moving to moving with thought. Ann. Sports Med. Res. 2015, 2, 1011. [Google Scholar] [PubMed]
- Ludyga, S.; Gerber, M.; Brand, S.; Holsboer-Trachsler, E.; Pühse, U. Acute effects of moderate aerobic exercise on specific aspects of executive function in different age and fitness groups: A meta-analysis. Psychophysiology 2016, 53, 1611–1626. [Google Scholar] [CrossRef] [PubMed]
- De Greeff, J.W.; Bosker, R.J.; Oosterlaan, J.; Visscher, C.; Hartman, E. Effects of physical activity on executive functions, attention and academic performance in preadolescent children: A meta-analysis. J. Sci. Med. Sport 2018, 21, 501–507. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Verburgh, L.; Königs, M.; Scherder, E.J.A.; Oosterlaan, J. Physical exercise and executive functions in preadolescent children, adolescents and young adults: A meta-analysis. Br. J. Sports Med. 2014, 48, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Tan, B.W.Z.; Pooley, J.A.; Speelman, C.P. A Meta-Analytic Review of the Efficacy of Physical Exercise Interventions on Cognition in Individuals with Autism Spectrum Disorder and ADHD. J. Autism Dev. Disord. 2016, 46, 3126–3143. [Google Scholar] [CrossRef] [PubMed]
- Cerrillo-Urbina, A.; García-Hermoso, A.; Sánchez-López, M.; Pardo-Guijarro, M.J.; Santos Gomez, J.; Martínez-Vizcaíno, V. The effects of physical exercise in children with attention deficit hyperactivity disorder: A systematic review and meta-analysis of randomized control trials. Child Carehealth Dev. 2015, 41, 779–788. [Google Scholar] [CrossRef] [PubMed]
- Cornelius, C.; Fedewa, A.L.; Ahn, S. The Effect of Physical Activity on Children With ADHD: A Quantitative Review of the Literature. J. Appl. Sch. Psychol. 2017, 33, 136–170. [Google Scholar] [CrossRef]
- Vysniauske, R.; Verburgh, L.; Oosterlaan, J.; Molendijk, M.L. The effects of physical exercise on functional outcomes in the treatment of ADHD: A meta-analysis. J. Atten. Disord. 2016. [Google Scholar] [CrossRef] [PubMed]
- Drollette, E.S.; Scudder, M.R.; Raine, L.B.; Moore, R.D.; Saliba, B.J.; Pontifex, M.B.; Hillman, C.H. Acute exercise facilitates brain function and cognition in children who need it most: An ERP study of individual differences in inhibitory control capacity. Dev. Cogn. Neurosci. 2014, 7, 53–64. [Google Scholar] [CrossRef]
- Silva, A.P.; Prado, S.O.; Scardovelli, T.A.; Boschi, S.R.; Campos, L.C.; Frère, A.F. Measurement of the effect of physical exercise on the concentration of individuals with ADHD. PLoS ONE 2015, 10, e0122119. [Google Scholar] [CrossRef]
- Gawrilow, C.; Stadler, G.; Langguth, N.; Naumann, A.; Boeck, A. Physical activity, affect, and cognition in children with symptoms of ADHD. J. Atten. Disord. 2016, 20, 151–162. [Google Scholar] [CrossRef]
- Chuang, L.-Y.; Tsai, Y.-J.; Chang, Y.-K.; Huang, C.-J.; Hung, T.-M. Effects of acute aerobic exercise on response preparation in a Go/No Go Task in children with ADHD: An ERP study. J. Sport Health Sci. 2015, 4, 82–88. [Google Scholar] [CrossRef]
- Ludyga, S.; Brand, S.; Gerber, M.; Weber, P.; Brotzmann, M.; Habibifar, F.; Pühse, U. An event-related potential investigation of the acute effects of aerobic and coordinative exercise on inhibitory control in children with ADHD. Dev. Cogn. Neurosci. 2017, 28, 21–28. [Google Scholar] [CrossRef]
- Benzing, V.; Chang, Y.-K.; Schmidt, M. Acute Physical Activity Enhances Executive Functions in Children with ADHD. Sci. Rep. 2018, 8, 12382. [Google Scholar] [CrossRef]
- Pontifex, M.B.; Saliba, B.J.; Raine, L.B.; Picchietti, D.L.; Hillman, C.H. Exercise improves behavioral, neurocognitive, and scholastic performance in children with attention-deficit/hyperactivity disorder. J. Pediatrics 2013, 162, 543–551. [Google Scholar] [CrossRef]
- Piepmeier, A.T.; Shih, C.-H.; Whedon, M.; Williams, L.M.; Davis, M.E.; Henning, D.A.; Park, S.; Calkins, S.D.; Etnier, J.L. The effect of acute exercise on cognitive performance in children with and without ADHD. J. Sport Health Sci. 2015, 4, 97–104. [Google Scholar] [CrossRef]
- Gapin, J.I.; Labban, J.D.; Bohall, S.C.; Wooten, J.S.; Chang, Y.-K. Acute exercise is associated with specific executive functions in college students with ADHD: A preliminary study. J. Sport Health Sci. 2015, 4, 89–96. [Google Scholar] [CrossRef]
- Chang, Y.-K.; Liu, S.; Yu, H.-H.; Lee, Y.-H. Effect of acute exercise on executive function in children with attention deficit hyperactivity disorder. Arch. Clin. Neuropsychol. 2012, 27, 225–237. [Google Scholar] [CrossRef]
- Hung, C.-L.; Huang, C.-J.; Tsai, Y.-J.; Chang, Y.-K.; Hung, T.-M. Neuroelectric and behavioral effects of acute exercise on task switching in children with attention-deficit/hyperactivity disorder. Front. Psychol. 2016, 7, 1589. [Google Scholar] [CrossRef]
- Ludyga, S.; Gerber, M.; Mücke, M.; Brand, S.; Weber, P.; Brotzmann, M.; Pühse, U. The acute effects of aerobic exercise on cognitive flexibility and task-related heart rate variability in children with ADHD and healthy controls. J. Atten. Disord. 2018. [Google Scholar] [CrossRef]
- Fritz, K.M.; O’Connor, P.J. Acute Exercise Improves Mood and Motivation in Young Men with ADHD Symptoms. Med. Sci. Sports Exerc. 2016, 48, 1153–1160. [Google Scholar] [CrossRef]
- Craft, D.H. Effect of prior exercise on cognitive performance tasks by hyperactive and normal young boys. Percept. Mot. Ski. 1983, 56, 979–982. [Google Scholar] [CrossRef]
- O’Halloran, L.; Cao, Z.; Ruddy, K.; Jollans, L.; Albaugh, M.D.; Aleni, A.; Potter, A.S.; Vahey, N.; Banaschewski, T.; Hohmann, S.; et al. Neural circuitry underlying sustained attention in healthy adolescents and in ADHD symptomatology. NeuroImage 2018, 169, 395–406. [Google Scholar] [CrossRef]
- Mahon, A.D.; Dean, R.S.; McIntosh, D.E.; Marjerrison, A.D.; Cole, A.S.; Woodruff, M.E.; Lee, M.P. Acute exercise effects on measures of attention and impulsivity in children with attention deficit/hyperactivity disorder. J. Educ. Dev. Psychol. 2013, 3, 65. [Google Scholar] [CrossRef]
- Rowlands, A.V. Accelerometer assessment of physical activity in children: An update. Pediatric Exerc. Sci. 2007, 19, 252–266. [Google Scholar] [CrossRef]
- Coe, D.P.; Pivarnik, J.M.; Womack, C.J.; Reeves, M.J.; Malina, R.M. Effect of physical education and activity levels on academic achievement in children. Med. Sci. Sports Exerc. 2006, 38, 1515–1519. [Google Scholar] [CrossRef]
- Chaddock, L.; Erickson, K.I.; Prakash, R.S.; VanPatter, M.; Voss, M.W.; Pontifex, M.B.; Raine, L.B.; Hillman, C.H.; Kramer, A.F. Basal ganglia volume is associated with aerobic fitness in preadolescent children. Dev. Neurosci. 2010, 32, 249–256. [Google Scholar] [CrossRef]
- Chaddock, L.; Erickson, K.I.; Prakash, R.S.; Voss, M.W.; VanPatter, M.; Pontifex, M.B.; Hillman, C.H.; Kramer, A.F. A functional MRI investigation of the association between childhood aerobic fitness and neurocognitive control. Biol. Psychol. 2012, 89, 260–268. [Google Scholar] [CrossRef]
- Chaddock, L.; Hillman, C.H.; Pontifex, M.B.; Johnson, C.R.; Raine, L.B.; Kramer, A.F. Childhood aerobic fitness predicts cognitive performance one year later. J. Sports Sci. 2012, 30, 421–430. [Google Scholar] [CrossRef]
- Chaddock-Heyman, L.; Erickson, K.I.; Chappell, M.A.; Johnson, C.L.; Kienzler, C.; Knecht, A.; Drollette, E.S.; Raine, L.B.; Scudder, M.R.; Kao, S.-C.; et al. Aerobic fitness is associated with greater hippocampal cerebral blood flow in children. Dev. Cogn. Neurosci. 2016, 20, 52–58. [Google Scholar] [CrossRef]
- Chaddock-Heyman, L.; Erickson, K.I.; Holtrop, J.L.; Voss, M.W.; Pontifex, M.B.; Raine, L.B.; Hillman, C.H.; Kramer, A.F. Aerobic fitness is associated with greater white matter integrity in children. Front. Hum. Neurosci. 2014, 8, 584. [Google Scholar] [CrossRef]
- Fedewa, A.L.; Ahn, S. The Effects of Physical Activity and Physical Fitness on Children’s Achievement and Cognitive Outcomes. Res. Q. Exerc. Sport 2011, 82, 521–535. [Google Scholar] [CrossRef]
- Etnier, J.L.; Salazar, W.; Landers, D.M.; Petruzzello, S.J.; Han, M.; Nowell, P. The influence of physical fitness and exercise upon cognitive functioning: A meta-analysis. J. Sport Exerc. Psychol. 1997, 19, 249–277. [Google Scholar] [CrossRef]
- Sibley, B.A.; Etnier, J.L. The relationship between physical activity and cognition in children: A meta-analysis. Pediatric Exerc. Sci. 2003, 15, 243–256. [Google Scholar] [CrossRef]
- Cook, B.G.; Li, D.; Heinrich, K.M. Obesity, Physical Activity, and Sedentary Behavior of Youth With Learning Disabilities and ADHD. J. Learn. Disabil. 2015, 48, 563–576. [Google Scholar] [CrossRef]
- Kim, J.; Mutyala, B.; Agiovlasitis, S.; Fernhall, B. Health behaviors and obesity among US children with attention deficit hyperactivity disorder by gender and medication use. Prev. Med. 2011, 52, 218–222. [Google Scholar] [CrossRef]
- Brassell, A.A.; Shoulberg, E.K.; Pontifex, M.B.; Smith, A.L.; Delli Paoli, A.G.; Hoza, B. Aerobic fitness and inhibition in young children: Moderating roles of ADHD status and age. J. Clin. Child Adolesc. Psychol. 2017, 46, 646–652. [Google Scholar] [CrossRef]
- Tsai, Y.-J.; Hung, C.-L.; Tsai, C.-L.; Chang, Y.-K.; Huang, C.-J.; Hung, T.-M. The Relationship between physical fitness and inhibitory ability in children with attention deficit hyperactivity disorder: An event-related potential study. Psychol. Sport Exerc. 2017, 31, 149–157. [Google Scholar] [CrossRef]
- Gapin, J.; Etnier, J.L. The relationship between physical activity and executive function performance in children with attention-deficit hyperactivity disorder. J. Sport Exerc. Psychol. 2010, 32, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Bueno, C.; Pesce, C.; Cavero-Redondo, I.; Sánchez-López, M.; Martínez-Hortelano, J.A.; Martínez-Vizcaíno, V. The Effect of Physical Activity Interventions on Children’s Cognition and Metacognition: A Systematic Review and Meta-Analysis. J. Am. Acad. Child Adolesc. Psychiatry 2017, 56, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Yang, Y.; Huang, T. Effects of chronic exercise interventions on executive function among children and adolescents: A systematic review with meta-analysis. Br. J. Sports Med. 2019. [Google Scholar] [CrossRef] [PubMed]
- Gallotta, M.C.; Emerenziani, G.P.; Iazzoni, S.; Meucci, M.; Baldari, C.; Guidetti, L. Impacts of coordinative training on normal weight and overweight/obese children’s attentional performance. Front. Hum. Neurosci. 2015, 9, 577. [Google Scholar] [CrossRef] [PubMed]
- Brickenkamp, R.; Schmidt-Atzert, L.; Liepmann, D.; Zillmer, E. d2-R: Test d2-Revision: Aufmerksamkeits-und Konzentrationstest; Hogrefe: Oxford, UK, 2010. [Google Scholar]
- Allen, J.I. Jogging can modify disruptive behaviors. Teach. Except. Child. 1980, 12, 66–70. [Google Scholar] [CrossRef]
- Zhang, L.; Ji, W.; Fan, Y.; Wu, H.; Liu, K. Effect of Physical Exercise on Cognition and Emotion in Children with ADHD. Destech Trans. Comput. Sci. Eng. 2015. [Google Scholar] [CrossRef]
- Verret, C.; Guay, M.-C.; Berthiaume, C.; Gardiner, P.; Béliveau, L. A Physical Activity Program Improves Behavior and Cognitive Functions in Children with ADHD: An Exploratory Study. J. Atten. Disord. 2012, 16, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.-Y.; Chu, C.-H.; Tsai, C.-L.; Lo, S.-Y.; Cheng, Y.-W.; Liu, Y.-J. A racket-sport intervention improves behavioral and cognitive performance in children with attention-deficit/hyperactivity disorder. Res. Dev. Disabil. 2016, 57, 1–10. [Google Scholar] [CrossRef]
- Pan, C.-Y.; Tsai, C.-L.; Chu, C.-H.; Sung, M.-C.; Huang, C.-Y.; Ma, W.-Y. Effects of physical exercise intervention on motor skills and executive functions in children with ADHD: A pilot study. J. Atten. Disord. 2015. [Google Scholar] [CrossRef]
- Memarmoghaddam, M.; Torbati, H.; Sohrabi, M.; Mashhadi, A.; Kashi, A. Effects of a selected exercise program on executive function of children with attention deficit hyperactivity disorder. J. Med. Life 2016, 9, 373. [Google Scholar]
- Geladé, K.; Janssen, T.; Bink, M.; Maras, A.; Oosterlaan, J. Behavioral effects of neurofeedback compared to stimulants and physical activity in attention-deficit/hyperactivity disorder: A randomized controlled trial. J. Clin. Psychiatry 2016, 77, e1270–e1277. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.W.; Han, D.H.; Kang, K.D.; Jung, H.Y.; Renshaw, P.F. Aerobic exercise and attention deficit hyperactivity disorder: Brain research. Med. Sci. Sports Exerc. 2015, 47, 33. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-K.; Hung, C.-L.; Huang, C.-J.; Hatfield, B.D.; Hung, T.-M. Effects of an aquatic exercise program on inhibitory control in children with ADHD: A preliminary study. Arch. Clin. Neuropsychol. 2014, 29, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, G.; Mohamed, S. Effect of regular aerobic exercises on behavioral, cognitive and psychological response in patients with attention deficit-hyperactivity disorder. Life Sci. J. 2011, 8, 366–371. [Google Scholar]
- Kim, J.-K. The effects of a home-based sensorimotor program on executive and motor functions in children with ADHD: A case series. J. Phys. Ther. Sci. 2018, 30, 1138–1140. [Google Scholar] [CrossRef] [PubMed]
- Frazier, W.; Wilson, S. Does physical activity in children improve ADHD symptoms? Evid.-Based Pract. 2018, 21, 12. [Google Scholar]
- Rommel, A.-S.; Halperin, J.M.; Mill, J.; Asherson, P.; Kuntsi, J. Protection From Genetic Diathesis in Attention-Deficit/Hyperactivity Disorder: Possible Complementary Roles of Exercise. J. Am. Acad. Child Adolesc. Psychiatry 2013, 52, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Reeves, M.J.; Bailey, R.P. The effects of physical activity on children diagnosed with attention deficit hyperactivity disorder: A review. Education 3-13 2016, 44, 591–603. [Google Scholar] [CrossRef]
- Halperin, J.M.; Berwid, O.G.; O’Neill, S. Healthy body, healthy mind? The effectiveness of physical activity to treat ADHD in children. Child Adolesc. Psychiatr. Clin. 2014, 23, 899–936. [Google Scholar] [CrossRef]
- Jeyanthi, S.; Arumugam, N.; Parasher, R.K. Effect of physical exercises on attention, motor skill and physical fitness in children with attention deficit hyperactivity disorder: A systematic review. ADHD Atten. Deficit Hyperact. Disord. 2018. [Google Scholar] [CrossRef]
- Suarez-Manzano, S.; Ruiz-Ariza, A.; De La Torre-Cruz, M.; Martínez-López, E.J. Acute and chronic effect of physical activity on cognition and behaviour in young people with ADHD: A systematic review of intervention studies. Res. Dev. Disabil. 2018, 77, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Neudecker, C.; Mewes, N.; Reimers, A.K.; Woll, A. Exercise interventions in children and adolescents with ADHD: A systematic review. J. Atten. Disord. 2015. [Google Scholar] [CrossRef] [PubMed]
- Kamp, C.F.; Sperlich, B.; Holmberg, H.C. Exercise reduces the symptoms of attention-deficit/hyperactivity disorder and improves social behaviour, motor skills, strength and neuropsychological parameters. Acta Paediatr. 2014, 103, 709–714. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Kareem, I.A.; Stancak, A.; Parkes, L.M.; Sluming, V. Increased gray matter volume of left pars opercularis in male orchestral musicians correlate positively with years of musical performance. J. Magn. Reson. Imaging 2011, 33, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Hyde, K.L.; Lerch, J.; Norton, A.; Forgeard, M.; Winner, E.; Evans, A.C.; Schlaug, G. Musical training shapes structural brain development. J. Neurosci. 2009, 29, 3019–3025. [Google Scholar] [CrossRef] [PubMed]
- Driemeyer, J.; Boyke, J.; Gaser, C.; Büchel, C.; May, A. Changes in Gray Matter Induced by Learning—Revisited. PLoS ONE 2008, 3, e2669. [Google Scholar] [CrossRef] [PubMed]
- Kooij, J.J.S.; Bijlenga, D.; Salerno, L.; Jaeschke, R.; Bitter, I.; Balázs, J.; Thome, J.; Dom, G.; Kasper, S.; Nunes Filipe, C.; et al. Updated European Consensus Statement on diagnosis and treatment of adult ADHD. Eur. Psychiatry 2019, 56, 14–34. [Google Scholar] [CrossRef] [PubMed]
- Muster, R.; Choudhury, S.; Sharp, W.; Kasparek, S.; Sudre, G.; Shaw, P. Mapping the neuroanatomic substrates of cognition in familial attention deficit hyperactivity disorder. Psychol. Med. 2018, 49, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Frodl, T.; Skokauskas, N. Meta-analysis of structural MRI studies in children and adults with attention deficit hyperactivity disorder indicates treatment effects. Acta Psychiatr. Scand. 2012, 125, 114–126. [Google Scholar] [CrossRef]
- Almeida, L.G.; Ricardo-Garcell, J.; Prado, H.; Barajas, L.; Fernández-Bouzas, A.; Ávila, D.; Martínez, R.B. Reduced right frontal cortical thickness in children, adolescents and adults with ADHD and its correlation to clinical variables: A cross-sectional study. J. Psychiatr. Res. 2010, 44, 1214–1223. [Google Scholar] [CrossRef]
- Amico, F.; Stauber, J.; Koutsouleris, N.; Frodl, T. Anterior cingulate cortex gray matter abnormalities in adults with attention deficit hyperactivity disorder: A voxel-based morphometry study. Psychiatry Res. Neuroimaging 2011, 191, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Makris, N.; Seidman, L.J.; Valera, E.M.; Biederman, J.; Monuteaux, M.C.; Kennedy, D.N.; Caviness, V.S., Jr.; Bush, G.; Crum, K.; Brown, A.B. Anterior cingulate volumetric alterations in treatment-naive adults with ADHD: A pilot study. J. Atten. Disord. 2010, 13, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Hoogman, M.; Bralten, J.; Hibar, D.P.; Mennes, M.; Zwiers, M.P.; Schweren, L.S.; van Hulzen, K.J.; Medland, S.E.; Shumskaya, E.; Jahanshad, N. Subcortical brain volume differences in participants with attention deficit hyperactivity disorder in children and adults: A cross-sectional mega-analysis. Lancet Psychiatry 2017, 4, 310–319. [Google Scholar] [CrossRef]
- Berquin, P.C.; Giedd, J.N.; Jacobsen, L.K.; Hamburger, S.D.; Krain, A.L.; Rapoport, J.L.; Castellanos, F.X. Cerebellum in attention-deficit hyperactivity disorder. A Morphometric MRI Study. Neurology 1998, 50, 1087–1093. [Google Scholar] [PubMed]
- Stoodley, C.J. Distinct regions of the cerebellum show gray matter decreases in autism, ADHD, and developmental dyslexia. Front. Syst. Neurosci. 2014, 8, 92. [Google Scholar] [CrossRef] [PubMed]
- McAlonan, G.M.; Cheung, V.; Cheung, C.; Chua, S.E.; Murphy, D.G.M.; Suckling, J.; Tai, K.-S.; Yip, L.K.C.; Leung, P.; Ho, T.P. Mapping brain structure in attention deficit-hyperactivity disorder: A voxel-based MRI study of regional grey and white matter volume. Psychiatry Res. Neuroimaging 2007, 154, 171–180. [Google Scholar] [CrossRef]
- Vilgis, V.; Sun, L.; Chen, J.; Silk, T.J.; Vance, A. Global and local grey matter reductions in boys with ADHD combined type and ADHD inattentive type. Psychiatry Res. Neuroimaging 2016, 254, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Valera, E.M.; Faraone, S.V.; Murray, K.E.; Seidman, L.J. Meta-analysis of structural imaging findings in attention-deficit/hyperactivity disorder. Biol. Psychiatry 2007, 61, 1361–1369. [Google Scholar] [CrossRef]
- Cha, J.; Fekete, T.; Siciliano, F.; Biezonski, D.; Greenhill, L.; Pliszka, S.R.; Blader, J.C.; Roy, A.K.; Leibenluft, E.; Posner, J. Neural correlates of aggression in medication-naive children with ADHD: Multivariate analysis of morphometry and tractography. Neuropsychopharmacology 2015, 40, 1717. [Google Scholar] [CrossRef]
- Nagel, B.J.; Bathula, D.; Herting, M.; Schmitt, C.; Kroenke, C.D.; Fair, D.; Nigg, J.T. Altered white matter microstructure in children with attention-deficit/hyperactivity disorder. J. Am. Acad. Child Adolesc. Psychiatry 2011, 50, 283–292. [Google Scholar] [CrossRef]
- Shaw, P.; Sudre, G.; Wharton, A.; Weingart, D.; Sharp, W.; Sarlls, J. White matter microstructure and the variable adult outcome of childhood attention deficit hyperactivity disorder. Neuropsychopharmacology 2015, 40, 746. [Google Scholar] [CrossRef] [PubMed]
- Almeida Montes, L.G.; Ricardo-Garcell, J.; Barajas De La Torre, L.B.; Prado Alcántara, H.; Martínez García, R.B.; Fernández-Bouzas, A.; Avila Acosta, D. Clinical correlations of grey matter reductions in the caudate nucleus of adults with attention deficit hyperactivity disorder. J. Psychiatry Neurosci. 2010, 35, 238–246. [Google Scholar] [PubMed]
- Bartus, R.T.; Levere, T. Frontal decortication in rhesus monkeys: A test of the interference hypothesis. Brain Res. 1977, 119, 233–248. [Google Scholar] [CrossRef]
- Wilkins, A.J.; Shallice, T.; McCarthy, R. Frontal lesions and sustained attention. Neuropsychologia 1987, 25, 359–365. [Google Scholar] [CrossRef]
- Woods, D.L.; Knight, R.T. Electrophysiologic evidence of increased distractibility after dorsolateral prefrontal lesions. Neurology 1986, 36, 212. [Google Scholar] [CrossRef] [PubMed]
- Chao, L.L.; Knight, R.T. Human prefrontal lesions increase distractibility to irrelevant sensory inputs. Neuroreport Int. J. Rapid Commun. Res. Neurosci. 1995, 6, 1605–1610. [Google Scholar] [CrossRef]
- Ma, C.-L.; Qi, X.-L.; Peng, J.-Y.; Li, B.-M. Selective deficit in no-go performance induced by blockade of prefrontal cortical α2-adrenoceptors in monkeys. NeuroReport 2003, 14, 1013–1016. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.-L.; Arnsten, A.F.; Li, B.-M. Locomotor hyperactivity induced by blockade of prefrontal cortical α2-adrenoceptors in monkeys. Biol. Psychiatry 2005, 57, 192–195. [Google Scholar] [CrossRef] [PubMed]
- Voss, M.W.; Vivar, C.; Kramer, A.F.; van Praag, H. Bridging animal and human models of exercise-induced brain plasticity. Trends Cogn. Sci. 2013, 17, 525–544. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H.; Christie, B.R.; Sejnowski, T.J.; Gage, F.H. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc. Natl. Acad. Sci. USA 1999, 96, 13427–13431. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H.; Kempermann, G.; Gage, F.H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nat. Neurosci. 1999, 2, 266. [Google Scholar] [CrossRef] [PubMed]
- Eddy, M.C.; Green, J.T. Running wheel exercise reduces renewal of extinguished instrumental behavior and alters medial prefrontal cortex neurons in adolescent, but not adult, rats. Behav. Neurosci. 2017, 131, 460–469. [Google Scholar] [CrossRef] [PubMed]
- Chaddock, L.; Erickson, K.I.; Prakash, R.S.; Kim, J.S.; Voss, M.W.; VanPatter, M.; Pontifex, M.B.; Raine, L.B.; Konkel, A.; Hillman, C.H. A neuroimaging investigation of the association between aerobic fitness, hippocampal volume, and memory performance in preadolescent children. Brain Res. 2010, 1358, 172–183. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, A.M.; Voss, M.W.; Prakash, R.S.; Chaddock, L.; Szabo, A.; White, S.M.; Wojcicki, T.R.; Mailey, E.; McAuley, E.; Kramer, A.F. The association between aerobic fitness and executive function is mediated by prefrontal cortex volume. Brainbehav. Immun. 2012, 26, 811–819. [Google Scholar] [CrossRef]
- Pontifex, M.B.; Raine, L.B.; Johnson, C.R.; Chaddock, L.; Voss, M.W.; Cohen, N.J.; Kramer, A.F.; Hillman, C.H. Cardiorespiratory Fitness and the Flexible Modulation of Cognitive Control in Preadolescent Children. J. Cogn. Neurosci. 2011, 23, 1332–1345. [Google Scholar] [CrossRef]
- Voss, M.W.; Chaddock, L.; Kim, J.S.; VanPatter, M.; Pontifex, M.B.; Raine, L.B.; Cohen, N.J.; Hillman, C.H.; Kramer, A.F. Aerobic fitness is associated with greater efficiency of the network underlying cognitive control in preadolescent children. Neuroscience 2011, 199, 166–176. [Google Scholar] [CrossRef]
- Colcombe, S.J.; Erickson, K.I.; Scalf, P.E.; Kim, J.S.; Prakash, R.; McAuley, E.; Elavsky, S.; Marquez, D.X.; Hu, L.; Kramer, A.F. Aerobic exercise training increases brain volume in aging humans. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 1166–1170. [Google Scholar] [CrossRef]
- Chaddock-Heyman, L.; Erickson, K.I.; Kienzler, C.; King, M.; Pontifex, M.B.; Raine, L.B.; Hillman, C.H.; Kramer, A.F. The role of aerobic fitness in cortical thickness and mathematics achievement in preadolescent children. PLoS ONE 2015, 10, e0134115. [Google Scholar]
- Sowell, E.R.; Thompson, P.M.; Leonard, C.M.; Welcome, S.E.; Kan, E.; Toga, A.W. Longitudinal Mapping of Cortical Thickness and Brain Growth in Normal Children. J. Neurosci. 2004, 24, 8223–8231. [Google Scholar] [CrossRef]
- Gogtay, N.; Giedd, J.N.; Lusk, L.; Hayashi, K.M.; Greenstein, D.; Vaituzis, A.C.; Nugent, T.F.; Herman, D.H.; Clasen, L.S.; Toga, A.W.; et al. Dynamic mapping of human cortical development during childhood through early adulthood. Proc. Natl. Acad. Sci. USA 2004, 101, 8174–8179. [Google Scholar] [CrossRef]
- Krafft, C.E.; Schaeffer, D.J.; Schwarz, N.F.; Chi, L.; Weinberger, A.L.; Pierce, J.E.; Rodrigue, A.L.; Allison, J.D.; Yanasak, N.E.; Liu, T.; et al. Improved Frontoparietal White Matter Integrity in Overweight Children Is Associated with Attendance at an After-School Exercise Program. Dev. Neurosci. 2014, 36, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, D.J.; Krafft, C.E.; Schwarz, N.F.; Chi, L.; Rodrigue, A.L.; Pierce, J.E.; Allison, J.D.; Yanasak, N.E.; Liu, T.; Davis, C.L.; et al. An 8-month exercise intervention alters frontotemporal white matter integrity in overweight children. Psychophysiology 2014, 51, 728–733. [Google Scholar] [CrossRef]
- Smith, S.M.; Fox, P.T.; Miller, K.L.; Glahn, D.C.; Fox, P.M.; Mackay, C.E.; Filippini, N.; Watkins, K.E.; Toro, R.; Laird, A.R. Correspondence of the brain’s functional architecture during activation and rest. Proc. Natl. Acad. Sci. USA 2009, 106, 13040–13045. [Google Scholar] [CrossRef] [PubMed]
- Greicius, M.D.; Krasnow, B.; Reiss, A.L.; Menon, V. Functional connectivity in the resting brain: A network analysis of the default mode hypothesis. Proc. Natl. Acad. Sci. USA 2003, 100, 253–258. [Google Scholar] [CrossRef] [PubMed]
- Fassbender, C.; Zhang, H.; Buzy, W.M.; Cortes, C.R.; Mizuiri, D.; Beckett, L.; Schweitzer, J.B. A lack of default network suppression is linked to increased distractibility in ADHD. Brain Res. 2009, 1273, 114–128. [Google Scholar] [CrossRef] [PubMed]
- Rubia, K.; Alegria, A.A.; Cubillo, A.I.; Smith, A.B.; Brammer, M.J.; Radua, J. Effects of Stimulants on Brain Function in Attention-Deficit/Hyperactivity Disorder: A Systematic Review and Meta-Analysis. Biol. Psychiatry 2014, 76, 616–628. [Google Scholar] [CrossRef] [PubMed]
- Liddle, E.B.; Hollis, C.; Batty, M.J.; Groom, M.J.; Totman, J.J.; Liotti, M.; Scerif, G.; Liddle, P.F. Task-related default mode network modulation and inhibitory control in ADHD: Effects of motivation and methylphenidate. J. Child Psychol. Psychiatry 2011, 52, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Jasper, H.H.; Solomon, P.; Bradley, C. Electroencephalographic analyses of behavior problem children. Am. J. Psychiatry 1938, 95, 641–658. [Google Scholar] [CrossRef]
- Satterfield, J.H.; Dawson, M.E. Electrodermal correlates of hyperactivity in children. Psychophysiology 1971, 8, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, A.E.; Holton, K.F. Measuring Treatment Response in Pharmacological and Lifestyle Interventions Using Electroencephalography in ADHD: A Review. Clin. EEG Neurosci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Arns, M.; Conners, C.K.; Kraemer, H.C. A Decade of EEG Theta/Beta Ratio Research in ADHD: A Meta-Analysis. J. Atten. Disord. 2013, 17, 374–383. [Google Scholar] [CrossRef]
- Arns, M. EEG-Based Personalized Medicine in ADHD: Individual Alpha Peak Frequency as an Endophenotype Associated with Nonresponse. J. Neurother. 2012, 16, 123–141. [Google Scholar] [CrossRef]
- Lansbergen, M.M.; Arns, M.; van Dongen-Boomsma, M.; Spronk, D.; Buitelaar, J.K. The increase in theta/beta ratio on resting-state EEG in boys with attention-deficit/hyperactivity disorder is mediated by slow alpha peak frequency. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2011, 35, 47–52. [Google Scholar] [CrossRef]
- Vollebregt, M.A.; van Dongen-Boomsma, M.; Slaats-Willemse, D.; Buitelaar, J.K.; Oostenveld, R. How the Individual Alpha Peak Frequency Helps Unravel the Neurophysiologic Underpinnings of Behavioral Functioning in Children With Attention-Deficit/Hyperactivity Disorder. Clin. EEG Neurosci. 2015, 46, 285–291. [Google Scholar] [CrossRef]
- Jann, K.; Koenig, T.; Dierks, T.; Boesch, C.; Federspiel, A. Association of individual resting state EEG alpha frequency and cerebral blood flow. NeuroImage 2010, 51, 365–372. [Google Scholar] [CrossRef]
- Jin, Y.; O’Halloran, J.P.; Plon, L.; Sandman, C.A.; Potkin, S.G. Alpha EEG predicts visual reaction time. Int. J. Neurosci. 2006, 116, 1035–1044. [Google Scholar] [CrossRef]
- Klimesch, W. EEG alpha and theta oscillations reflect cognitive and memory performance: A review and analysis. Brain Res. Rev. 1999, 29, 169–195. [Google Scholar] [CrossRef]
- Cortese, S.; Kelly, C.; Chabernaud, C.; Proal, E.; Di Martino, A.; Milham, M.P.; Xavier Castellanos, F. Toward Systems Neuroscience of ADHD: A Meta-Analysis of 55 fMRI Studies. Am. J. Psychiatry 2012, 169, 1038–1055. [Google Scholar] [CrossRef]
- Dickstein, S.G.; Bannon, K.; Xavier Castellanos, F.; Milham, M.P. The neural correlates of attention deficit hyperactivity disorder: An ALE meta-analysis. J. Child Psychol. Psychiatry 2006, 47, 1051–1062. [Google Scholar] [CrossRef]
- Sonuga-Barke, E.J.S.; Fairchild, G. Neuroeconomics of Attention-Deficit/Hyperactivity Disorder: Differential Influences of Medial, Dorsal, and Ventral Prefrontal Brain Networks on Suboptimal Decision Making? Biol. Psychiatry 2012, 72, 126–133. [Google Scholar] [CrossRef]
- Hart, H.; Radua, J.; Nakao, T.; Mataix-Cols, D.; Rubia, K. Meta-analysis of functional magnetic resonance imaging studies of inhibition and attention in attention-deficit/hyperactivity disorder: Exploring task-specific, stimulant medication, and age effects. JAMA Psychiatry 2013, 70, 185–198. [Google Scholar] [CrossRef]
- Plichta, M.M.; Scheres, A. Ventral–striatal responsiveness during reward anticipation in ADHD and its relation to trait impulsivity in the healthy population: A meta-analytic review of the fMRI literature. Neurosci. Biobehav. Rev. 2014, 38, 125–134. [Google Scholar] [CrossRef]
- Scheres, A.; Milham, M.P.; Knutson, B.; Castellanos, F.X. Ventral striatal hyporesponsiveness during reward anticipation in attention-deficit/hyperactivity disorder. Biol. Psychiatry 2007, 61, 720–724. [Google Scholar] [CrossRef]
- Van Hulst, B.M.; de Zeeuw, P.; Bos, D.J.; Rijks, Y.; Neggers, S.F.; Durston, S. Children with ADHD symptoms show decreased activity in ventral striatum during the anticipation of reward, irrespective of ADHD diagnosis. J. Child Psychol. Psychiatry 2017, 58, 206–214. [Google Scholar] [CrossRef]
- Luman, M.; Tripp, G.; Scheres, A. Identifying the neurobiology of altered reinforcement sensitivity in ADHD: A review and research agenda. Neurosci. Biobehav. Rev. 2010, 34, 744–754. [Google Scholar] [CrossRef]
- Wu, A.; Ying, Z.; Gomez-Pinilla, F. Docosahexaenoic acid dietary supplementation enhances the effects of exercise on synaptic plasticity and cognition. Neuroscience 2008, 155, 751–759. [Google Scholar] [CrossRef]
- Yeo, B.T.T.; Krienen, F.M.; Sepulcre, J.; Sabuncu, M.R.; Lashkari, D.; Hollinshead, M.; Roffman, J.L.; Smoller, J.W.; Zöllei, L.; Polimeni, J.R.; et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J. Neurophysiol. 2011, 106, 1125–1165. [Google Scholar]
- Tegelbeckers, J.; Kanowski, M.; Krauel, K.; Haynes, J.-D.; Breitling, C.; Flechtner, H.-H.; Kahnt, T. Orbitofrontal Signaling of Future Reward is Associated with Hyperactivity in Attention-Deficit/Hyperactivity Disorder. J. Neurosci. 2018, 38, 6779–6786. [Google Scholar] [CrossRef]
- Corbetta, M.; Shulman, G.L. Control of goal-directed and stimulus-driven attention in the brain. Nat. Rev. Neurosci. 2002, 3, 201. [Google Scholar] [CrossRef]
- Corbetta, M.; Patel, G.; Shulman, G.L. The reorienting system of the human brain: From environment to theory of mind. Neuron 2008, 58, 306–324. [Google Scholar] [CrossRef]
- McCarthy, H.; Skokauskas, N.; Mulligan, A.; Donohoe, G.; Mullins, D.; Kelly, J.; Johnson, K.; Fagan, A.; Gill, M.; Meaney, J.; et al. Attention Network Hypoconnectivity With Default and Affective Network Hyperconnectivity in Adults Diagnosed With Attention-Deficit/Hyperactivity Disorder in ChildhoodAttention and Affective Changes in Adult ADHDAttention and Affective Changes in Adult ADHD. JAMA Psychiatry 2013, 70, 1329–1337. [Google Scholar] [CrossRef]
- Berger, A.; Posner, M.I. Pathologies of brain attentional networks. Neurosci. Biobehav. Rev. 2000, 24, 3–5. [Google Scholar] [CrossRef]
- Posner, M.I.; Petersen, S.E. The attention system of the human brain. Annu. Rev. Neurosci. 1990, 13, 25–42. [Google Scholar] [CrossRef]
- Luck, S.J.; Woodman, G.F.; Vogel, E.K. Event-related potential studies of attention. Trends Cogn. Sci. 2000, 4, 432–440. [Google Scholar] [CrossRef]
- Picton, T.W. The P300 wave of the human event-related potential. J. Clin. Neurophysiol. 1992, 9, 456–479. [Google Scholar] [CrossRef]
- Johnstone, S.J.; Barry, R.J.; Clarke, A.R. Ten years on: A follow-up review of ERP research in attention-deficit/hyperactivity disorder. Clin. Neurophysiol. 2013, 124, 644–657. [Google Scholar] [CrossRef]
- Polich, J. Updating P300: An integrative theory of P3a and P3b. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 2007, 118, 2128–2148. [Google Scholar] [CrossRef]
- Gutmann, B.; Mierau, A.; Hülsdünker, T.; Hildebrand, C.; Przyklenk, A.; Hollmann, W.; Strüder, H.K. Effects of Physical Exercise on Individual Resting State EEG Alpha Peak Frequency. Neural Plast. 2015, 2015, 717312. [Google Scholar] [CrossRef]
- Moraes, H.; Ferreira, C.; Deslandes, A.; Cagy, M.; Pompeu, F.; Ribeiro, P.; Piedade, R. Beta and alpha electroencephalographic activity changes after acute exercise. Arq. De Neuro-Psiquiatr. 2007, 65, 637–641. [Google Scholar] [CrossRef]
- Choi, H.; Park, S.; Kim, K.K.; Lee, K.; Rhyu, H.-S. Acute effects of aerobic stretching, health and happiness improving movement exercise on cortical activity of children. J. Exerc. Rehabil. 2016, 12, 320–327. [Google Scholar] [CrossRef]
- Huang, C.-J.; Huang, C.-W.; Tsai, Y.-J.; Tsai, C.-L.; Chang, Y.-K.; Hung, T.-M. A preliminary examination of aerobic exercise effects on resting EEG in children with ADHD. J. Atten. Disord. 2017, 21, 898–903. [Google Scholar] [CrossRef]
- Huang, C.-J.; Huang, C.-W.; Hung, C.-L.; Tsai, Y.-J.; Chang, Y.-K.; Wu, C.-T.; Hung, T.-M. Effects of Acute Exercise on Resting EEG in Children with Attention-Deficit/Hyperactivity Disorder. Child Psychiatry Hum. Dev. 2018, 49, 993–1002. [Google Scholar] [CrossRef]
- Weng, T.B.; Pierce, G.L.; Darling, W.G.; Falk, D.; Magnotta, V.A.; Voss, M.W. The acute effects of aerobic exercise on the functional connectivity of human brain networks. Brain Plast. 2017, 2, 171–190. [Google Scholar] [CrossRef]
- Hillman, C.H.; Snook, E.M.; Jerome, G.J. Acute cardiovascular exercise and executive control function. Int. J. Psychophysiol. 2003, 48, 307–314. [Google Scholar] [CrossRef]
- Kao, S.-C.; Drollette, E.S.; Ritondale, J.P.; Khan, N.; Hillman, C.H. The acute effects of high-intensity interval training and moderate-intensity continuous exercise on declarative memory and inhibitory control. Psychol. Sport Exerc. 2018, 38, 90–99. [Google Scholar] [CrossRef]
- Li, L.; Men, W.-W.; Chang, Y.-K.; Fan, M.-X.; Ji, L.; Wei, G.-X. Acute Aerobic Exercise Increases Cortical Activity during Working Memory: A Functional MRI Study in Female College Students. PLoS ONE 2014, 9, e99222. [Google Scholar] [CrossRef]
- Chen, A.-G.; Zhu, L.-N.; Yan, J.; Yin, H.-C. Neural Basis of Working Memory Enhancement after Acute Aerobic Exercise: fMRI Study of Preadolescent Children. Front. Psychol. 2016, 7, 1804. [Google Scholar] [CrossRef]
- Bediz, C.S.; Oniz, A.; Guducu, C.; Ural Demirci, E.; Ogut, H.; Gunay, E.; Cetinkaya, C.; Ozgoren, M. Acute Supramaximal Exercise Increases the Brain Oxygenation in Relation to Cognitive Workload. Front. Hum. Neurosci. 2016, 10, 174. [Google Scholar] [CrossRef]
- Byun, K.; Hyodo, K.; Suwabe, K.; Ochi, G.; Sakairi, Y.; Kato, M.; Dan, I.; Soya, H. Positive effect of acute mild exercise on executive function via arousal-related prefrontal activations: An fNIRS study. NeuroImage 2014, 98, 336–345. [Google Scholar] [CrossRef]
- Kujach, S.; Byun, K.; Hyodo, K.; Suwabe, K.; Fukuie, T.; Laskowski, R.; Dan, I.; Soya, H. A transferable high-intensity intermittent exercise improves executive performance in association with dorsolateral prefrontal activation in young adults. NeuroImage 2018, 169, 117–125. [Google Scholar] [CrossRef]
- Yanagisawa, H.; Dan, I.; Tsuzuki, D.; Kato, M.; Okamoto, M.; Kyutoku, Y.; Soya, H. Acute moderate exercise elicits increased dorsolateral prefrontal activation and improves cognitive performance with Stroop test. NeuroImage 2010, 50, 1702–1710. [Google Scholar] [CrossRef]
- Lambrick, D.; Stoner, L.; Grigg, R.; Faulkner, J. Effects of continuous and intermittent exercise on executive function in children aged 8–10 years. Psychophysiology 2016, 53, 1335–1342. [Google Scholar] [CrossRef]
- Budde, H.; Voelcker-Rehage, C.; Pietraßyk-Kendziorra, S.; Ribeiro, P.; Tidow, G. Acute coordinative exercise improves attentional performance in adolescents. Neurosci. Lett. 2008, 441, 219–223. [Google Scholar] [CrossRef]
- Davis, C.L.; Tomporowski, P.D.; McDowell, J.E.; Austin, B.P.; Miller, P.H.; Yanasak, N.E.; Allison, J.D.; Naglieri, J.A. Exercise improves executive function and achievement and alters brain activation in overweight children: A randomized, controlled trial. Health Psychol. Off. J. Div. Health Psychol. Am. Psychol. Assoc. 2011, 30, 91–98. [Google Scholar] [CrossRef]
- Chaddock-Heyman, L.; Erickson, K.I.; Voss, M.; Knecht, A.; Pontifex, M.B.; Castelli, D.; Hillman, C.; Kramer, A. The effects of physical activity on functional MRI activation associated with cognitive control in children: A randomized controlled intervention. Front. Hum. Neurosci. 2013, 7, 72. [Google Scholar] [CrossRef]
- Krafft, C.E.; Schwarz, N.F.; Chi, L.; Weinberger, A.L.; Schaeffer, D.J.; Pierce, J.E.; Rodrigue, A.L.; Yanasak, N.E.; Miller, P.H.; Tomporowski, P.D.; et al. An 8-month randomized controlled exercise trial alters brain activation during cognitive tasks in overweight children. Obesity 2014, 22, 232–242. [Google Scholar] [CrossRef]
- Hillman, C.H.; Pontifex, M.B.; Castelli, D.M.; Khan, N.A.; Raine, L.B.; Scudder, M.R.; Drollette, E.S.; Moore, R.D.; Wu, C.-T.; Kamijo, K. Effects of the FITKids randomized controlled trial on executive control and brain function. Pediatrics 2014, 134, e1063–e1071. [Google Scholar] [CrossRef]
- Ludyga, S.; Koutsandréou, F.; Reuter, E.-M.; Voelcker-Rehage, C.; Budde, H. A Randomized Controlled Trial on the Effects of Aerobic and Coordinative Training on Neural Correlates of Inhibitory Control in Children. J. Clin. Med. 2019, 8, 184. [Google Scholar] [CrossRef]
- Ludyga, S.; Gerber, M.; Herrmann, C.; Brand, S.; Pühse, U. Chronic effects of exercise implemented during school-break time on neurophysiological indices of inhibitory control in adolescents. Trends Neurosci. Educ. 2018, 10, 1–7. [Google Scholar] [CrossRef]
- Lardon, M.T.; Polich, J. EEG changes from long-term physical exercise. Biol. Psychol. 1996, 44, 19–30. [Google Scholar] [CrossRef]
- Madsen, P.L.; Sperling, B.R.K.; Warming, T.; Schmidt, J.; Secher, N.; Wildschiodtz, G.; Holm, S.; Lassen, N. Middle cerebral artery blood velocity and cerebral blood flow and O2 uptake during dynamic exercise. J. Appl. Physiol. 1993, 74, 245–250. [Google Scholar] [CrossRef]
- Isaacs, K.R.; Anderson, B.J.; Alcantara, A.A.; Black, J.E.; Greenough, W.T. Exercise and the brain: Angiogenesis in the adult rat cerebellum after vigorous physical activity and motor skill learning. J. Cereb. Blood Flow Metab. 1992, 12, 110–119. [Google Scholar] [CrossRef]
- Black, J.E.; Isaacs, K.R.; Anderson, B.J.; Alcantara, A.A.; Greenough, W.T. Learning causes synaptogenesis, whereas motor activity causes angiogenesis, in cerebellar cortex of adult rats. Proc. Natl. Acad. Sci. USA 1990, 87, 5568–5572. [Google Scholar] [CrossRef]
- Swain, R.A.; Harris, A.B.; Wiener, E.C.; Dutka, M.V.; Morris, H.D.; Theien, B.E.; Konda, S.; Engberg, K.; Lauterbur, P.C.; Greenough, W.T. Prolonged exercise induces angiogenesis and increases cerebral blood volume in primary motor cortex of the rat. Neuroscience 2003, 117, 1037–1046. [Google Scholar] [CrossRef]
- Kleim, J.A.; Cooper, N.R.; VandenBerg, P.M. Exercise induces angiogenesis but does not alter movement representations within rat motor cortex. Brain Res. 2002, 934, 1–6. [Google Scholar] [CrossRef]
- Rhyu, I.; Bytheway, J.; Kohler, S.; Lange, H.; Lee, K.; Boklewski, J.; McCormick, K.; Williams, N.; Stanton, G.; Greenough, W. Effects of aerobic exercise training on cognitive function and cortical vascularity in monkeys. Neuroscience 2010, 167, 1239–1248. [Google Scholar] [CrossRef]
- Ding, Y.-H.; Luan, X.-D.; Li, J.; Rafols, J.A.; Guthinkonda, M.; Diaz, F.G.; Ding, Y. Exercise-induced overexpression of angiogenic factors and reduction of ischemia/reperfusion injury in stroke. Curr. Neurovascular Res. 2004, 1, 411–420. [Google Scholar] [CrossRef]
- Yun, H.-S.; Park, M.-S.; Ji, E.-S.; Kim, T.-W.; Ko, I.-G.; Kim, H.-B.; Kim, H. Treadmill exercise ameliorates symptoms of attention deficit/hyperactivity disorder through reducing Purkinje cell loss and astrocytic reaction in spontaneous hypertensive rats. J. Exerc. Rehabil. 2014, 10, 22–30. [Google Scholar] [CrossRef]
- Pickel, V.M.; Segal, M.; Bloom, F.E. A radioautographic study of the efferent pathways of the nucleus locus coeruleus. J. Comp. Neurol. 1974, 155, 15–41. [Google Scholar] [CrossRef]
- Fallon, J.H. Collateralization of monoamine neurons: Mesotelencephalic dopamine projections to caudate, septum, and frontal cortex. J. Neurosci. 1981, 1, 1361–1368. [Google Scholar] [CrossRef]
- Bush, G.; Valera, E.M.; Seidman, L.J. Functional neuroimaging of attention-deficit/hyperactivity disorder: A review and suggested future directions. Biol. Psychiatry 2005, 57, 1273–1284. [Google Scholar] [CrossRef]
- Marco, R.; Miranda, A.; Schlotz, W.; Melia, A.; Mulligan, A.; Müller, U.; Andreou, P.; Butler, L.; Christiansen, H.; Gabriels, I. Delay and reward choice in ADHD: An experimental test of the role of delay aversion. Neuropsychology 2009, 23, 367. [Google Scholar] [CrossRef]
- Luman, M.; Oosterlaan, J.; Sergeant, J.A. The impact of reinforcement contingencies on AD/HD: A review and theoretical appraisal. Clin. Psychol. Rev. 2005, 25, 183–213. [Google Scholar] [CrossRef]
- Schultz, W. Getting formal with dopamine and reward. Neuron 2002, 36, 241–263. [Google Scholar] [CrossRef]
- Faraone, S.V. The pharmacology of amphetamine and methylphenidate: Relevance to the neurobiology of attention-deficit/hyperactivity disorder and other psychiatric comorbidities. Neurosci. Biobehav. Rev. 2018, 87, 255–270. [Google Scholar] [CrossRef]
- Avelar, A.J.; Juliano, S.A.; Garris, P.A. Amphetamine augments vesicular dopamine release in the dorsal and ventral striatum through different mechanisms. J. Neurochem. 2013, 125, 373–385. [Google Scholar] [CrossRef]
- Easton, N.; Steward, C.; Marshall, F.; Fone, K.; Marsden, C. Effects of amphetamine isomers, methylphenidate and atomoxetine on synaptosomal and synaptic vesicle accumulation and release of dopamine and noradrenaline in vitro in the rat brain. Neuropharmacology 2007, 52, 405–414. [Google Scholar] [CrossRef]
- Ren, J.; Xu, H.; Choi, J.K.; Jenkins, B.G.; Chen, Y.I. Dopaminergic response to graded dopamine concentration elicited by four amphetamine doses. Synapse 2009, 63, 764–772. [Google Scholar] [CrossRef]
- Carson, R.E.; Breier, A.; De Bartolomeis, A.; Saunders, R.C.; Su, T.P.; Schmall, B.; Der, M.G.; Pickar, D.; Eckelman, W.C. Quantification of amphetamine-induced changes in [11C] raclopride binding with continuous infusion. J. Cereb. Blood Flow Metab. 1997, 17, 437–447. [Google Scholar] [CrossRef]
- Jedema, H.P.; Narendran, R.; Bradberry, C.W. Amphetamine-induced release of dopamine in primate prefrontal cortex and striatum: Striking differences in magnitude and timecourse. J. Neurochem. 2014, 130, 490–497. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Tomasi, D.; Kollins, S.H.; Wigal, T.L.; Newcorn, J.H.; Telang, F.W.; Fowler, J.S.; Logan, J.; Wong, C.T. Methylphenidate-elicited dopamine increases in ventral striatum are associated with long-term symptom improvement in adults with attention deficit hyperactivity disorder. J. Neurosci. 2012, 32, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Moeller, S.J.; Honorio, J.; Tomasi, D.; Parvaz, M.A.; Woicik, P.A.; Volkow, N.D.; Goldstein, R.Z. Methylphenidate enhances executive function and optimizes prefrontal function in both health and cocaine addiction. Cereb. Cortex 2012, 24, 643–653. [Google Scholar] [CrossRef] [PubMed]
- Tomasi, D.; Volkow, N.D.; Wang, G.-J.; Wang, R.; Telang, F.; Caparelli, E.C.; Wong, C.; Jayne, M.; Fowler, J.S. Methylphenidate enhances brain activation and deactivation responses to visual attention and working memory tasks in healthy controls. Neuroimage 2011, 54, 3101–3110. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.V.; Asherson, P.; Banaschewski, T.; Biederman, J.; Buitelaar, J.K.; Ramos-Quiroga, J.A.; Rohde, L.A.; Sonuga-Barke, E.J.S.; Tannock, R.; Franke, B. Attention-deficit/hyperactivity disorder. Nat. Rev. Dis. Primers 2015, 1, 15020. [Google Scholar] [CrossRef] [PubMed]
- Sagvolden, T. Behavioral validation of the spontaneously hypertensive rat (SHR) as an animal model of attention-deficit/hyperactivity disorder (AD/HD). Neurosci. Biobehav. Rev. 2000, 24, 31–39. [Google Scholar] [CrossRef]
- Russell, V.A.; Oades, R.D.; Tannock, R.; Killeen, P.R.; Auerbach, J.G.; Johansen, E.B.; Sagvolden, T. Response variability in attention-deficit/hyperactivity disorder: A neuronal and glial energetics hypothesis. Behav. Brain Funct. 2006, 2, 30. [Google Scholar] [CrossRef] [PubMed]
- Sagvolden, T.; Xu, T. l-Amphetamine improves poor sustained attention while d-amphetamine reduces overactivity and impulsiveness as well as improves sustained attention in an animal model of Attention-Deficit/Hyperactivity Disorder (ADHD). Behav. Brain Funct. 2008, 4, 3. [Google Scholar] [CrossRef]
- Sagvolden, T.; Johansen, E.B. Rat models of ADHD. In Behavioral Neuroscience of Attention Deficit Hyperactivity Disorder and Its Treatment; Springer: Berlin/Heidelberg, Germany, 2011; pp. 301–315. [Google Scholar]
- Sagvolden, T.; Russell, V.A.; Aase, H.; Johansen, E.B.; Farshbaf, M. Rodent models of attention-deficit/hyperactivity disorder. Biol. Psychiatry 2005, 57, 1239–1247. [Google Scholar] [CrossRef]
- Watanabe, Y.; Fujita, M.; Ito, Y.; Okada, T. Brain dopamine transporter in spontaneously hypertensive rats. J. Nucl. Med. 1997, 38, 470. [Google Scholar]
- Russell, V.A. Dopamine hypofunction possibly results from a defect in glutamate-stimulated release of dopamine in the nucleus accumbens shell of a rat model for attention deficit hyperactivity disorder—The spontaneously hypertensive rat. Neurosci. Biobehav. Rev. 2003, 27, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Russell, V.; de Villiers, A.; Sagvolden, T.; Lamm, M.; Taljaard, J. Altered dopaminergic function in the prefrontal cortex, nucleus accumbens and caudate-putamen of an animal model of attention-deficit hyperactivity disorder—The spontaneously hypertensive rat. Brain Res. 1995, 676, 343–351. [Google Scholar] [CrossRef]
- Faraone, S.V.; Larsson, H. Genetics of attention deficit hyperactivity disorder. Mol. Psychiatry 2018, 24, 562–575. [Google Scholar] [CrossRef] [PubMed]
- Demontis, D.; Walters, R.K.; Martin, J.; Mattheisen, M.; Als, T.D.; Agerbo, E.; Baldursson, G.; Belliveau, R.; Bybjerg-Grauholm, J.; Bækvad-Hansen, M. Discovery of the first genome-wide significant risk loci for attention deficit/hyperactivity disorder. Nat. Genet. Adv. Online Publ. 2018, 51, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Mortensen, O.V.; Larsen, M.B.; Prasad, B.M.; Amara, S.G. Genetic complementation screen identifies a mitogen-activated protein kinase phosphatase, MKP3, as a regulator of dopamine transporter trafficking. Mol. Biol. Cell 2008, 19, 2818–2829. [Google Scholar] [CrossRef] [PubMed]
- Gizer, I.R.; Ficks, C.; Waldman, I.D. Candidate gene studies of ADHD: A meta-analytic review. Hum. Genet. 2009, 126, 51–90. [Google Scholar] [CrossRef] [PubMed]
- Akutagava-Martins, G.C.; Salatino-Oliveira, A.; Kieling, C.; Genro, J.P.; Polanczyk, G.V.; Anselmi, L.; Menezes, A.M.; Gonçalves, H.; Wehrmeister, F.C.; Barros, F.C. COMT and DAT1 genes are associated with hyperactivity and inattention traits in the 1993 Pelotas Birth Cohort: Evidence of sex-specific combined effect. J. Psychiatry Neurosci. 2016, 41, 405. [Google Scholar] [CrossRef] [PubMed]
- Skriver, K.; Roig, M.; Lundbye-Jensen, J.; Pingel, J.; Helge, J.W.; Kiens, B.; Nielsen, J.B. Acute exercise improves motor memory: Exploring potential biomarkers. Neurobiol. Learn. Mem. 2014, 116, 46–58. [Google Scholar] [CrossRef]
- Arnsten, A.F.; Steere, J.C.; Hunt, R.D. The contribution of α2-noradrenergic mechanisms to prefrontal cortical cognitive function: Potential significance for attention-deficit hyperactivity disorder. Arch. Gen. Psychiatry 1996, 53, 448–455. [Google Scholar] [CrossRef]
- Russell, V.A.; Wiggins, T.M. Increased Glutamate-Stimulated Norepinephrine Release from Prefrontal Cortex Slices of Spontaneously Hypertensive Rats. Metab. Brain Dis. 2000, 15, 297–304. [Google Scholar] [CrossRef]
- Sara, S.J. The locus coeruleus and noradrenergic modulation of cognition. Nat. Rev. Neurosci. 2009, 10, 211. [Google Scholar] [CrossRef]
- Aston-Jones, G.; Chiang, C.; Alexinsky, T. Chapter 35—Discharge of noradrenergic locus coeruleus neurons in behaving rats and monkeys suggests a role in vigilance. In Progress in Brain Research; Barnes, C.D., Pompeiano, O., Eds.; Elsevier: Amsterdam, The Netherlands, 1991; Volume 88, pp. 501–520. [Google Scholar]
- Smith, A.; Nutt, D. Noradrenaline and attention lapses. Nature 1996. [Google Scholar] [CrossRef] [PubMed]
- Wigal, S.B.; Nemet, D.; Swanson, J.M.; Regino, R.; Trampush, J.; Ziegler, M.G.; Cooper, D.M. Catecholamine response to exercise in children with attention deficit hyperactivity disorder. Pediatric Res. 2003, 53, 756. [Google Scholar] [CrossRef] [PubMed]
- Finnema, S.J.; Hughes, Z.A.; Haaparanta-Solin, M.; Stepanov, V.; Nakao, R.; Varnäs, K.; Varrone, A.; Arponen, E.; Marjamäki, P.; Pohjanoksa, K. Amphetamine decreases α2C-adrenoceptor binding of [11C] ORM-13070: A PET study in the primate brain. Int. J. Neuropsychopharmacol. 2015, 18. [Google Scholar] [CrossRef]
- Scahill, L.; Aman, M.G.; McDougle, C.J.; McCracken, J.T.; Tierney, E.; Dziura, J.; Arnold, L.E.; Posey, D.; Young, C.; Shah, B. AProspective Open Trial of Guanfacine in Children with Pervasive Developmental Disorders. J. Child Adolesc. Psychopharmacol. 2006, 16, 589–598. [Google Scholar] [CrossRef] [PubMed]
- Kent, L.; Doerry, U.; Hardy, E.; Parmar, R.; Gingell, K.; Hawi, Z.; Kirley, A.; Lowe, N.; Fitzgerald, M.; Gill, M.; et al. Evidence that variation at the serotonin transporter gene influences susceptibility to attention deficit hyperactivity disorder (ADHD): Analysis and pooled analysis. Mol. Psychiatry 2002, 7, 908. [Google Scholar] [CrossRef]
- Malhotra, S.; Santosh, P.J. An Open Clinical Trial of Buspirone in Children With Attention-Deficit/Hyperactivity Disorder. J. Am. Acad. Child Adolesc. Psychiatry 1998, 37, 364–371. [Google Scholar] [CrossRef] [PubMed]
- Oades, R.D. Role of the serotonin system in ADHD: Treatment implications. Expert Rev. Neurother. 2007, 7, 1357–1374. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.S.; Naganawa, M.; Gallezot, J.D.; Nabulsi, N.; Lin, S.F.; Ropchan, J.; Weinzimmer, D.; McCarthy, T.J.; Carson, R.E.; Huang, Y.; et al. Clinical doses of atomoxetine significantly occupy both norepinephrine and serotonin transports: Implications on treatment of depression and ADHD. NeuroImage 2014, 86, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Shuai, L.; Yang, L.; Cao, Q.; Wang, Y.; Chan, R.C.K. Comparative study of OROS-MPH and atomoxetine on executive function improvement in ADHD: A randomized controlled trial. Int. J. Neuropsychopharmacol. 2012, 15, 15–26. [Google Scholar]
- Chamberlain, S.R.; del Campo, N.; Dowson, J.; Müller, U.; Clark, L.; Robbins, T.W.; Sahakian, B.J. Atomoxetine Improved Response Inhibition in Adults with Attention Deficit/Hyperactivity Disorder. Biol. Psychiatry 2007, 62, 977–984. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.-J.; Kollins, S.H.; Wigal, T.L.; Newcorn, J.H.; Telang, F.; Fowler, J.S.; Zhu, W.; Logan, J.; Ma, Y.; et al. Evaluating Dopamine Reward Pathway in ADHD: Clinical Implications. JAMA 2009, 302, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Heijnen, S.; Hommel, B.; Kibele, A.; Colzato, L.S. Neuromodulation of Aerobic Exercise—A Review. Front. Psychol. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Meeusen, R.; De Meirleir, K. Exercise and brain neurotransmission. Sports Med. 1995, 20, 160–188. [Google Scholar] [CrossRef] [PubMed]
- Koch, G.; Johansson, U.; Arvidsson, E. Radioenzymatic determination of epinephrine, norepinephrine and dopamine in 0.1 ml plasma samples. Plasma catecholamine response to submaximal and near maximal exercise. Clin. Chem. Lab. Med. 1980, 18, 367–372. [Google Scholar] [CrossRef]
- Van Loon, G.R.; Schwartz, L.; Sole, M.J. Plasma dopamine responses to standing and exercise in man. Life Sci. 1979, 24, 2273–2277. [Google Scholar] [CrossRef]
- Winter, B.; Breitenstein, C.; Mooren, F.C.; Voelker, K.; Fobker, M.; Lechtermann, A.; Krueger, K.; Fromme, A.; Korsukewitz, C.; Floel, A.; et al. High impact running improves learning. Neurobiol. Learn. Mem. 2007, 87, 597–609. [Google Scholar] [CrossRef] [PubMed]
- Sutoo, D.; Akiyama, K. Regulation of brain function by exercise. Neurobiol. Dis. 2003, 13, 1–14. [Google Scholar] [CrossRef]
- Sutoo, D.; Akiyama, K. The mechanism by which exercise modifies brain function. Physiol. Behav. 1996, 60, 177–181. [Google Scholar] [CrossRef]
- Meeusen, R.; Smolders, I.; Sarre, S.; De Meirleir, K.; Keizer, H.; Serneels, M.; Ebinger, G.; Michotte, Y. Endurance training effects on neurotransmitter release in rat striatum: An in vivo microdialysis study. Acta Physiol. Scand. 1997, 159, 335–341. [Google Scholar] [CrossRef]
- Heyes, M.P.; Garnett, E.S.; Coates, G. Nigrostriatal dopaminergic activity is increased during exhaustive exercise stress in rats. Life Sci. 1988, 42, 1537–1542. [Google Scholar] [CrossRef]
- Hattori, S.; Naoi, M.; Nishino, H. Striatal dopamine turnover during treadmill running in the rat: Relation to the speed of running. Brain Res. Bull. 1994, 35, 41–49. [Google Scholar] [CrossRef]
- Eddy, M.C.; Stansfield, K.J.; Green, J.T. Voluntary exercise improves performance of a discrimination task through effects on the striatal dopamine system. Learn. Mem. 2014, 21, 334–337. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dey, S.; Singh, R.; Dey, P. Exercise training: Significance of regional alterations in serotonin metabolism of rat brain in relation to antidepressant effect of exercise. Physiol. Behav. 1992, 52, 1095–1099. [Google Scholar] [CrossRef]
- Wang, G.-J.; Volkow, N.D.; Fowler, J.S.; Franceschi, D.; Logan, J.; Pappas, N.R.; Wong, C.T.; Netusil, N. PET studies of the effects of aerobic exercise on human striatal dopamine release. J. Nucl. Med. 2000, 41, 1352–1356. [Google Scholar] [PubMed]
- Blomstrand, E.; Perrett, D.; Parry-billings, M.; Newsholme, E.A. Effect of sustained exercise on plasma amino acid concentrations and on 5-hydroxytryptamine metabolism in six different brain regions in the rat. Acta Physiol. Scand. 1989, 136, 473–482. [Google Scholar] [CrossRef] [PubMed]
- Pagliari, R.; Peyrin, L. Norepinephrine release in the rat frontal cortex under treadmill exercise: A study with microdialysis. J. Appl. Physiol. 1995, 78, 2121–2130. [Google Scholar] [CrossRef] [PubMed]
- Foley, T.E.; Fleshner, M. Neuroplasticity of dopamine circuits after exercise: Implications for central fatigue. Neuromolecular Med. 2008, 10, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Baek, D.-J.; Lee, C.-B.; Baek, S.-S. Effect of treadmill exercise on social interaction and tyrosine hydroxylase expression in the attention-deficit/hyperactivity disorder rats. J. Exerc. Rehabil. 2014, 10, 252. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Heo, H.-I.; Kim, D.-H.; Ko, I.-G.; Lee, S.-S.; Kim, S.-E.; Kim, B.-K.; Kim, T.-W.; Ji, E.-S.; Kim, J.-D.; et al. Treadmill exercise and methylphenidate ameliorate symptoms of attention deficit/hyperactivity disorder through enhancing dopamine synthesis and brain-derived neurotrophic factor expression in spontaneous hypertensive rats. Neurosci. Lett. 2011, 504, 35–39. [Google Scholar] [CrossRef]
- Ji, E.-S.; Kim, C.-J.; Park, J.H.; Bahn, G.H. Duration-dependence of the effect of treadmill exercise on hyperactivity in attention deficit hyperactivity disorder rats. J. Exerc. Rehabil. 2014, 10, 75–80. [Google Scholar] [CrossRef]
- Cho, H.S.; Baek, D.J.; Baek, S.S. Effect of exercise on hyperactivity, impulsivity and dopamine D2 receptor expression in the substantia nigra and striatum of spontaneous hypertensive rats. J. Exerc. Nutr. Biochem. 2014, 18, 379. [Google Scholar] [CrossRef]
- Östman, I.; Nybäck, H. Adaptive changes in central and peripheral noradrenergic neurons in rats following chronic exercise. Neuroscience 1976, 1, 41–47. [Google Scholar] [CrossRef]
- Brown, B.S.; Payne, T.; Kim, C.; Moore, G.; Krebs, P.; Martin, W. Chronic response of rat brain norepinephrine and serotonin levels to endurance training. J. Appl. Physiol. 1979, 46, 19–23. [Google Scholar] [CrossRef]
- Robinson, A.M.; Buttolph, T.; Green, J.T.; Bucci, D.J. Physical exercise affects attentional orienting behavior through noradrenergic mechanisms. Behav. Neurosci. 2015, 129, 361. [Google Scholar] [CrossRef] [PubMed]
- Wigal, S.B.; Emmerson, N.; Gehricke, J.-G.; Galassetti, P. Exercise:Applications to Childhood ADHD. J. Atten. Disord. 2013, 17, 279–290. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, L.; Thomas, R.; Beck, M.M.; Pingel, J.; Andersen, J.D.; Mang, C.S.; Madsen, M.A.J.; Roig, M.; Lundbye-Jensen, J. The Beneficial Effect of Acute Exercise on Motor Memory Consolidation is Modulated by Dopaminergic Gene Profile. J. Clin. Med. 2019, 8, 578. [Google Scholar] [CrossRef] [PubMed]
- Mang, C.S.; McEwen, L.M.; MacIsaac, J.L.; Snow, N.J.; Campbell, K.L.; Kobor, M.S.; Ross, C.J.; Boyd, L.A. Exploring genetic influences underlying acute aerobic exercise effects on motor learning. Sci. Rep. 2017, 7, 12123. [Google Scholar] [CrossRef]
- Thomas, A.; Dennis, A.; Bandettini, P.; Johansen-Berg, H. The Effects of Aerobic Activity on Brain Structure. Front. Psychol. 2012, 3, 86. [Google Scholar] [CrossRef]
- Barde, Y.-A.; Edgar, D.; Thoenen, H. Purification of a new neurotrophic factor from mammalian brain. EMBO J. 1982, 1, 549–553. [Google Scholar] [CrossRef]
- Vaynman, S.; Ying, Z.; Gomez-Pinilla, F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur. J. Neurosci. 2004, 20, 2580–2590. [Google Scholar] [CrossRef]
- Fritsch, B.; Reis, J.; Martinowich, K.; Schambra, H.M.; Ji, Y.; Cohen, L.G.; Lu, B. Direct current stimulation promotes BDNF-dependent synaptic plasticity: Potential implications for motor learning. Neuron 2010, 66, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Christian, K.; Lu, B. BDNF: A key regulator for protein synthesis-dependent LTP and long-term memory? Neurobiol. Learn. Mem. 2008, 89, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Egan, M.F.; Kojima, M.; Callicott, J.H.; Goldberg, T.E.; Kolachana, B.S.; Bertolino, A.; Zaitsev, E.; Gold, B.; Goldman, D.; Dean, M. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 2003, 112, 257–269. [Google Scholar] [CrossRef]
- Tsai, S.-J. Role of neurotrophic factors in attention deficit hyperactivity disorder. Cytokine Growth Factor Rev. 2017, 34, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.-J. Attention-deficit hyperactivity disorder may be associated with decreased central brain-derived neurotrophic factor activity: Clinical and therapeutic implications. Med. Hypotheses 2007, 68, 896–899. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.-Y.; Shen, X.-M.; Yuan, F.-F.; Guo, O.-Y.; Zhong, Y.; Chen, J.-G.; Zhu, L.-Q.; Wu, J. The physiology of BDNF and its relationship with ADHD. Mol. Neurobiol. 2015, 52, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.J.; Nilsson, E.K.; Jacobsson, J.A.; Stein, D.J.; Fredriksson, R.; Lind, L.; Schiöth, H.B. BDNF Polymorphisms Are Linked to Poorer Working Memory Performance, Reduced Cerebellar and Hippocampal Volumes and Differences in Prefrontal Cortex in a Swedish Elderly Population. PLoS ONE 2014, 9, e82707. [Google Scholar] [CrossRef]
- Mandelman, S.D.; Grigorenko, E.L. BDNF Val66Met and cognition: All, none, or some? A meta-analysis of the genetic association. Genesbrain Behav. 2012, 11, 127–136. [Google Scholar]
- Lee, Y.H.; Song, G.G. BDNF 196 G/A and COMT Val158Met Polymorphisms and Susceptibility to ADHD: A Meta-Analysis. J. Atten. Disord. 2015, 22, 872–877. [Google Scholar] [CrossRef]
- Jasińska, K.K.; Molfese, P.J.; Kornilov, S.A.; Mencl, W.E.; Frost, S.J.; Lee, M.; Pugh, K.R.; Grigorenko, E.L.; Landi, N. The BDNF Val66Met polymorphism is associated with structural neuroanatomical differences in young children. Behav. Brain Res. 2017, 328, 48–56. [Google Scholar] [CrossRef]
- De Araujo, C.M.; Zugman, A.; Swardfager, W.; Belangero, S.I.N.; Ota, V.K.; Spindola, L.M.; Hakonarson, H.; Pellegrino, R.; Gadelha, A.; Salum, G.A.; et al. Effects of the brain-derived neurotropic factor variant Val66Met on cortical structure in late childhood and early adolescence. J. Psychiatr. Res. 2018, 98, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Bergman, O.; Westberg, L.; Lichtenstein, P.; Eriksson, E.; Larsson, H. Study on the possible association of brain-derived neurotrophic factor polymorphism with the developmental course of symptoms of attention deficit and hyperactivity. Int. J. Neuropsychopharmacol. 2011, 14, 1367–1376. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.-C.; Kim, H.-W.; Kim, B.-N.; Kim, J.-W.; Shin, M.-S.; Chung, S.; Cho, D.-Y.; Jung, S.-W.; Yoo, H.J.; Chung, I.-W.; et al. Gender-specific association of the brain-derived neurotrophic factor gene with attention-deficit/hyperactivity disorder. Psychiatry Investig. 2010, 7, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Bonvicini, C.; Faraone, S.V.; Scassellati, C. Common and specific genes and peripheral biomarkers in children and adults with attention-deficit/hyperactivity disorder. World J. Biol. Psychiatry 2018, 19, 80–100. [Google Scholar] [CrossRef] [PubMed]
- Hawi, Z.; Cummins, T.D.R.; Tong, J.; Arcos-Burgos, M.; Zhao, Q.; Matthews, N.; Newman, D.P.; Johnson, B.; Vance, A.; Heussler, H.S.; et al. Rare DNA variants in the brain-derived neurotrophic factor gene increase risk for attention-deficit hyperactivity disorder: A next-generation sequencing study. Mol. Psychiatry 2016, 22, 580. [Google Scholar] [CrossRef] [PubMed]
- Karege, F.; Schwald, M.; Cisse, M. Postnatal developmental profile of brain-derived neurotrophic factor in rat brain and platelets. Neurosci. Lett. 2002, 328, 261–264. [Google Scholar] [CrossRef]
- Li, H.; Liu, L.; Tang, Y.; Ji, N.; Yang, L.; Qian, Q.; Wang, Y. Sex-specific association of brain-derived neurotrophic factor (BDNF) Val66Met polymorphism and plasma BDNF with attention-deficit/hyperactivity disorder in a drug-naïve Han Chinese sample. Psychiatry Res. 2014, 217, 191–197. [Google Scholar] [CrossRef]
- Scassellati, C.; Zanardini, R.; Tiberti, A.; Pezzani, M.; Valenti, V.; Effedri, P.; Filippini, E.; Conte, S.; Ottolini, A.; Gennarelli, M.; et al. Serum brain-derived neurotrophic factor (BDNF) levels in attention deficit–hyperactivity disorder (ADHD). Eur. Child Adolesc. Psychiatry 2014, 23, 173–177. [Google Scholar] [CrossRef]
- Corominas-Roso, M.; Ramos-Quiroga, J.A.; Ribases, M.; Sanchez-Mora, C.; Palomar, G.; Valero, S.; Bosch, R.; Casas, M. Decreased serum levels of brain-derived neurotrophic factor in adults with attention-deficit hyperactivity disorder. Int. J. Neuropsychopharmacol. 2013, 16, 1267–1275. [Google Scholar] [CrossRef]
- Bilgiç, A.; Toker, A.; Işık, Ü.; Kılınç, İ. Serum brain-derived neurotrophic factor, glial-derived neurotrophic factor, nerve growth factor, and neurotrophin-3 levels in children with attention-deficit/hyperactivity disorder. Eur. Child Adolesc. Psychiatry 2017, 26, 355–363. [Google Scholar] [CrossRef]
- Zhang, J.; Luo, W.; Li, Q.; Xu, R.; Wang, Q.; Huang, Q. Peripheral brain-derived neurotrophic factor in attention-deficit/hyperactivity disorder: A comprehensive systematic review and meta-analysis. J. Affect. Disord. 2018, 227, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Amiri, A.; Torabi Parizi, G.; Kousha, M.; Saadat, F.; Modabbernia, M.-J.; Najafi, K.; Atrkar Roushan, Z. Changes in plasma Brain-derived neurotrophic factor (BDNF) levels induced by methylphenidate in children with Attention deficit–hyperactivity disorder (ADHD). Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2013, 47, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Banducci, S.E.; Weinstein, A.M.; MacDonald, A.W.; Ferrell, R.E.; Halder, I.; Flory, J.D.; Manuck, S.B. The Brain-Derived Neurotrophic Factor Val66Met Polymorphism Moderates an Effect of Physical Activity on Working Memory Performance. Psychol. Sci. 2013, 24, 1770–1779. [Google Scholar] [CrossRef] [PubMed]
- Altar, C.A.; Cai, N.; Bliven, T.; Juhasz, M.; Conner, J.M.; Acheson, A.L.; Lindsay, R.M.; Wiegand, S.J. Anterograde transport of brain-derived neurotrophic factor and its role in the brain. Nature 1997, 389, 856. [Google Scholar] [CrossRef] [PubMed]
- Yan, Q.; Rosenfeld, R.; Matheson, C.; Hawkins, N.; Lopez, O.; Bennett, L.; Welcher, A. Expression of brain-derived neurotrophic factor protein in the adult rat central nervous system. Neuroscience 1997, 78, 431–448. [Google Scholar] [CrossRef]
- Altar, C.A.; Siuciak, J.A.; Wright, P.; Ip, N.Y.; Lindsay, R.M.; Wiegand, S.J. In situ hybridization of trkB and trkC receptor mRNA in rat forebrain and association with high-affinity binding of [125I] BDNF, [125I] NT-4/5 and [125I] NT-3. Eur. J. Neurosci. 1994, 6, 1389–1405. [Google Scholar] [CrossRef] [PubMed]
- Merlio, J.P.; Ernfors, P.; Jaber, M.; Persson, H. Molecular cloning of rat trkC and distribution of cells expressing messenger RNAs for members of the trk family in the rat central nervous system. Neuroscience 1992, 51, 513–532. [Google Scholar] [CrossRef]
- Goggi, J.; Pullar, I.A.; Carney, S.L.; Bradford, H.F. Modulation of neurotransmitter release induced by brain-derived neurotrophic factor in rat brain striatal slices in vitro. Brain Res. 2002, 941, 34–42. [Google Scholar] [CrossRef]
- Goggi, J.; Pullar, I.A.; Carney, S.L.; Bradford, H.F. Signalling pathways involved in the short-term potentiation of dopamine release by BDNF. Brain Res. 2003, 968, 156–161. [Google Scholar] [CrossRef]
- D’Amore, D.E.; Tracy, B.A.; Parikh, V. Exogenous BDNF facilitates strategy set-shifting by modulating glutamate dynamics in the dorsal striatum. Neuropharmacology 2013, 75, 312–323. [Google Scholar] [CrossRef]
- Saylor, A.J.; McGinty, J.F. Amphetamine-induced locomotion and gene expression are altered in BDNF heterozygous mice. Genesbrain Behav. 2008, 7, 906–914. [Google Scholar] [CrossRef] [PubMed]
- Skodzik, T.; Holling, H.; Pedersen, A. Long-Term Memory Performance in Adult ADHD: A Meta-Analysis. J. Atten. Disord. 2017, 21, 267–283. [Google Scholar] [CrossRef] [PubMed]
- Szuhany, K.L.; Bugatti, M.; Otto, M.W. A meta-analytic review of the effects of exercise on brain-derived neurotrophic factor. J. Psychiatr. Res. 2015, 60, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, P.; Brassard, P.; Adser, H.; Pedersen, M.V.; Leick, L.; Hart, E.; Secher, N.H.; Pedersen, B.K.; Pilegaard, H. Evidence for a release of brain-derived neurotrophic factor from the brain during exercise. Exp. Physiol. 2009, 94, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.; Banks, W.A.; Fasold, M.B.; Bluth, J.; Kastin, A.J. Transport of brain-derived neurotrophic factor across the blood–brain barrier. Neuropharmacology 1998, 37, 1553–1561. [Google Scholar] [CrossRef]
- Poduslo, J.F.; Curran, G.L. Permeability at the blood-brain and blood-nerve barriers of the neurotrophic factors: NGF, CNTF, NT-3, BDNF. Mol. Brain Res. 1996, 36, 280–286. [Google Scholar] [CrossRef]
- Kyeremanteng, C.; James, J.; Mackay, J.; Merali, Z. A study of brain and serum brain-derived neurotrophic factor protein in Wistar and Wistar-Kyoto rat strains after electroconvulsive stimulus. Pharmacopsychiatry 2012, 45, 244–249. [Google Scholar] [CrossRef]
- Pardridge, W.M.; Wu, D.; Sakane, T. Combined use of carboxyl-directed protein pegylation and vector-mediated blood-brain barrier drug delivery system optimizes brain uptake of brain-derived neurotrophic factor following intravenous administration. Pharm. Res. 1998, 15, 576–582. [Google Scholar] [CrossRef]
- Thompson, A.B.; Stolyarova, A.; Ying, Z.; Zhuang, Y.; Gómez-Pinilla, F.; Izquierdo, A. Methamphetamine blocks exercise effects on Bdnf and Drd2 gene expression in frontal cortex and striatum. Neuropharmacology 2015, 99, 658–664. [Google Scholar] [CrossRef]
- Neeper, S.A.; Gómez-Pinilla, F.; Choi, J.; Cotman, C.W. Physical activity increases mRNA for brain-derived neurotrophic factor and nerve growth factor in rat brain. Brain Res. 1996, 726, 49–56. [Google Scholar] [CrossRef]
- Lee, J.K.W.; Koh, A.C.H.; Koh, S.X.T.; Liu, G.J.X.; Nio, A.Q.X.; Fan, P.W.P. Neck cooling and cognitive performance following exercise-induced hyperthermia. Eur. J. Appl. Physiol. 2014, 114, 375–384. [Google Scholar] [CrossRef]
- Tsai, C.-L.; Pan, C.-Y.; Chen, F.-C.; Wang, C.-H.; Chou, F.-Y. Effects of acute aerobic exercise on a task-switching protocol and brain-derived neurotrophic factor concentrations in young adults with different levels of cardiorespiratory fitness. Exp. Physiol. 2016, 101, 836–850. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-L.; Chen, F.-C.; Pan, C.-Y.; Wang, C.-H.; Huang, T.-H.; Chen, T.-C. Impact of acute aerobic exercise and cardiorespiratory fitness on visuospatial attention performance and serum BDNF levels. Psychoneuroendocrinology 2014, 41, 121–131. [Google Scholar] [CrossRef] [PubMed]
- Slusher, A.L.; Patterson, V.T.; Schwartz, C.S.; Acevedo, E.O. Impact of high intensity interval exercise on executive function and brain derived neurotrophic factor in healthy college aged males. Physiol. Behav. 2018, 191, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Tonoli, C.; Heyman, E.; Buyse, L.; Roelands, B.; Piacentini, M.F.; Bailey, S.; Pattyn, N.; Berthoin, S.; Meeusen, R. Neurotrophins and cognitive functions in T1D compared with healthy controls: Effects of a high-intensity exercise. Appl. Physiol. Nutr. Metab. 2014, 40, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Ferris, L.T.; Williams, J.S.; Shen, C.-L. The Effect of Acute Exercise on Serum Brain-Derived Neurotrophic Factor Levels and Cognitive Function. Med. Sci. Sports Exerc. 2007, 39, 728–734. [Google Scholar] [CrossRef] [PubMed]
- Griffin, É.W.; Mullally, S.; Foley, C.; Warmington, S.A.; O’Mara, S.M.; Kelly, Á.M. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol. Behav. 2011, 104, 934–941. [Google Scholar] [CrossRef]
- Faber Taylor, A.; Kuo, F.E. Children with attention deficits concentrate better after walk in the park. J. Atten. Disord. 2009, 12, 402–409. [Google Scholar] [CrossRef] [PubMed]
- García-Gómez, A.; Rodríguez-Jiménez, M.; Guerrero-Barona, E.; Rubio-Jiménez, J.C.; García-Peña, I.; Moreno-Manso, J.M. Benefits of an experimental program of equestrian therapy for children with ADHD. Res. Dev. Disabil. 2016, 59, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Jensen, P.S.; Kenny, D.T. The effects of yoga on the attention and behavior of boys with attention-deficit/hyperactivity disorder (ADHD). J. Atten. Disord. 2004, 7, 205–216. [Google Scholar] [CrossRef]
- Haffner, J.; Roos, J.; Goldstein, N.; Parzer, P.; Resch, F. The effectiveness of body-oriented methods of therapy in the treatment of attention-deficit hyperactivity disorder (ADHD): Results of a controlled pilot study. Z. Fur Kinder-Und Jugendpsychiatrie Und Psychother. 2006, 34, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Diamond, A.; Ling, D.S. Conclusions about interventions, programs, and approaches for improving executive functions that appear justified and those that, despite much hype, do not. Dev. Cogn. Neurosci. 2016, 18, 34–48. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-K.; Song, J.; Park, J.-H. Effects of combination exercises on electroencephalography and frontal lobe executive function measures in children with ADHD: A pilot study. Biomed. Res. 2017, S455–S460. [Google Scholar]
- Robertson, I.H.; Manly, T.; Andrade, J.; Baddeley, B.T.; Yiend, J. ‘Oops!’: Performance correlates of everyday attentional failures in traumatic brain injured and normal subjects. Neuropsychologia 1997, 35, 747–758. [Google Scholar] [CrossRef]
- Kortte, K.B.; Horner, M.D.; Windham, W.K. The Trail Making Test, Part B: Cognitive Flexibility or Ability to Maintain Set? Appl. Neuropsychol. 2002, 9, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Ziereis, S.; Jansen, P. Effects of physical activity on executive function and motor performance in children with ADHD. Res. Dev. Disabil. 2015, 38, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Den Heijer, A.E.; Groen, Y.; Tucha, L.; Fuermaier, A.B.; Koerts, J.; Lange, K.W.; Thome, J.; Tucha, O. Sweat it out? The effects of physical exercise on cognition and behavior in children and adults with ADHD: A systematic literature review. J. Neural Transm. 2017, 124, 3–26. [Google Scholar] [CrossRef] [PubMed]
- Grassmann, V.; Alves, M.V.; Santos-Galduróz, R.F.; Galduróz, J.C.F. Possible Cognitive Benefits of Acute Physical Exercise in Children With ADHD:A Systematic Review. J. Atten. Disord. 2017, 21, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Medina, J.A.; Netto, T.L.; Muszkat, M.; Medina, A.C.; Botter, D.; Orbetelli, R.; Scaramuzza, L.F.; Sinnes, E.G.; Vilela, M.; Miranda, M.C. Exercise impact on sustained attention of ADHD children, methylphenidate effects. Adhd Atten. Deficit Hyperact. Disord. 2010, 2, 49–58. [Google Scholar] [CrossRef]
- Hillman, C.H.; McAuley, E.; Erickson, K.I.; Liu-Ambrose, T.; Kramer, A.F. On mindful and mindless physical activity and executive function: A response to Diamond and Ling (2016). Dev. Cogn. Neurosci. 2018, 100529. [Google Scholar] [CrossRef]
- Best, J.R. Effects of physical activity on children’s executive function: Contributions of experimental research on aerobic exercise. Dev. Rev. 2010, 30, 331–351. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, V.; Saliasi, E.; de Groot, R.H.; Jolles, J.; Chinapaw, M.J.; Singh, A.S. Physical activity in the school setting: Cognitive performance is not affected by three different types of acute exercise. Front. Psychol. 2016, 7, 723. [Google Scholar] [CrossRef]
- Ng, Q.X.; Ho, C.Y.X.; Chan, H.W.; Yong, B.Z.J.; Yeo, W.-S. Managing childhood and adolescent attention-deficit/hyperactivity disorder (ADHD) with exercise: A systematic review. Complementary Ther. Med. 2017, 34, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Egger, F.; Conzelmann, A.; Schmidt, M. The effect of acute cognitively engaging physical activity breaks on children’s executive functions: Too much of a good thing? Psychol. Sport Exerc. 2018, 36, 178–186. [Google Scholar] [CrossRef]
- KoutsandrÉou, F.; Wegner, M.; Niemann, C.; Budde, H. Effects of motor versus cardiovascular exercise training on children’s working memory. Med. Sci. Sports Exerc. 2016, 48, 1144–1152. [Google Scholar] [CrossRef]
- Christiansen, L.; Larsen, M.N.; Grey, M.J.; Nielsen, J.B.; Lundbye-Jensen, J. Long-term progressive motor skill training enhances corticospinal excitability for the ipsilateral hemisphere and motor performance of the untrained hand. Eur. J. Neurosci. 2017, 45, 1490–1500. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, L.; Madsen, M.; Bojsen-Møller, E.; Thomas, R.; Nielsen, J.B.; Lundbye-Jensen, J. Progressive practice promotes motor learning and repeated transient increases in corticospinal excitability across multiple days. Brain Stimul. 2018, 11, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Crova, C.; Struzzolino, I.; Marchetti, R.; Masci, I.; Vannozzi, G.; Forte, R.; Pesce, C. Cognitively challenging physical activity benefits executive function in overweight children. J. Sports Sci. 2014, 32, 201–211. [Google Scholar] [CrossRef]
- Lambourne, K.; Tomporowski, P. The effect of exercise-induced arousal on cognitive task performance: A meta-regression analysis. Brain Res. 2010, 1341, 12–24. [Google Scholar] [CrossRef]
- Singh, A.S.; Saliasi, E.; van den Berg, V.; Uijtdewilligen, L.; de Groot, R.H.; Jolles, J.; Andersen, L.B.; Bailey, R.; Chang, Y.-K.; Diamond, A. Effects of physical activity interventions on cognitive and academic performance in children and adolescents: A novel combination of a systematic review and recommendations from an expert panel. Br. J. Sports Med. 2018. [Google Scholar] [CrossRef]
- Hart, J.L. Determining the Duration of Effects on Behavior and Academic Outcomes From Single and Multiple Bouts of Moderate Physical Activity for Students with ADHD. Ph.D. Thesis, Proquest LLC, Ann Arbor, MI, USA, 2015. [Google Scholar]
- Lundbye-Jensen, J.; Skriver, K.; Nielsen, J.B.; Roig, M. Acute Exercise Improves Motor Memory Consolidation in Preadolescent Children. Front. Hum. Neurosci. 2017, 11, 182. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Beck, M.M.; Lind, R.R.; Korsgaard Johnsen, L.; Geertsen, S.S.; Christiansen, L.; Ritz, C.; Roig, M.; Lundbye-Jensen, J. Acute exercise and motor memory consolidation: The role of exercise timing. Neural Plast. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R.; Flindtgaard, M.; Skriver, K.; Geertsen, S.S.; Christiansen, L.; Korsgaard Johnsen, L.; Busk, D.V.P.; Bojsen-Møller, E.; Madsen, M.; Ritz, C. Acute exercise and motor memory consolidation: Does exercise type play a role? Scand. J. Med. Sci. Sports 2017, 27, 1523–1532. [Google Scholar] [CrossRef]
- Thomas, R.; Johnsen, L.K.; Geertsen, S.S.; Christiansen, L.; Ritz, C.; Roig, M.; Lundbye-Jensen, J. Acute exercise and motor memory consolidation: The role of exercise intensity. PLoS ONE 2016, 11, e0159589. [Google Scholar] [CrossRef]
- Caterina, P.; Claudia, C.; Rosalba, M.; Ilaria, S.; Ilaria, M.; Giuseppe, V.; Roberta, F. Searching for cognitively optimal challenge point in physical activity for children with typical and atypical motor development. Ment. Health Phys. Act. 2013, 6, 172–180. [Google Scholar]
- Hoza, B.; Smith, A.L.; Shoulberg, E.K.; Linnea, K.S.; Dorsch, T.E.; Blazo, J.A.; Alerding, C.M.; McCabe, G.P. A randomized trial examining the effects of aerobic physical activity on attention-deficit/hyperactivity disorder symptoms in young children. J. Abnorm. Child Psychol. 2015, 43, 655–667. [Google Scholar] [CrossRef] [PubMed]
- Meßler, C.F.; Holmberg, H.-C.; Sperlich, B. Multimodal Therapy Involving High-Intensity Interval Training Improves the Physical Fitness, Motor Skills, Social Behavior, and Quality of Life of Boys With ADHD: A Randomized Controlled Study. J. Atten. Disord. 2018, 22, 806–812. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.-Y.; Chang, Y.-K.; Tsai, C.-L.; Chu, C.-H.; Cheng, Y.-W.; Sung, M.-C. Effects of Physical Activity Intervention on Motor Proficiency and Physical Fitness in Children With ADHD: An Exploratory Study. J. Atten. Disord. 2017, 21, 783–795. [Google Scholar] [CrossRef]
- Torabi, F.; Farahani, A.; Safakish, S.; Ramezankhani, A.; Dehghan, F. Evaluation of motor proficiency and adiponectin in adolescent students with attention deficit hyperactivity disorder after high-intensity intermittent training. Psychiatry Res. 2018, 261, 40–44. [Google Scholar] [CrossRef]
- Banaschewski, T.; Besmens, F.; Zieger, H.; Rothenberger, A. Evaluation of Sensorimotor Training in Children with Adhd. Percept. Mot. Ski. 2001, 92, 137–149. [Google Scholar] [CrossRef]
- Kiluk, B.D.; Weden, S.; Culotta, V.P. Sport Participation and Anxiety in Children with ADHD. J. Atten. Disord. 2009, 12, 499–506. [Google Scholar] [CrossRef] [PubMed]
- Etscheidt, M.A.; Ayllon, T. Contingent exercise to decrease hyperactivity. J. Child Adolesc. Psychother. 1987, 4, 192–198. [Google Scholar]
- Smith, A.L.; Hoza, B.; Linnea, K.; McQuade, J.D.; Tomb, M.; Vaughn, A.J.; Shoulberg, E.K.; Hook, H. Pilot physical activity intervention reduces severity of ADHD symptoms in young children. J. Atten. Disord. 2013, 17, 70–82. [Google Scholar] [CrossRef] [PubMed]
- Vibholm, H.A.; Pedersen, J.; Faltinsen, E.; Marcussen, M.H.; Gluud, C.; Storebø, O.J. Training, executive, attention and motor skills (TEAMS) training versus standard treatment for preschool children with attention deficit hyperactivity disorder: A randomised clinical trial. BMC Res. Notes 2018, 11, 366. [Google Scholar] [CrossRef] [PubMed]
- Silverstein, J.M.; Allison, D.B. The comparative efficacy of antecedent exercise and methylphenidate: A single-case randomized trial. Child: Care Health Dev. 1994, 20, 47–60. [Google Scholar] [CrossRef]
- McKune, A.; Pautz, J.; Lomjbard, J. Behavioural response to exercise in children with attention-deficit/hyperactivity disorder. South Afr. J. Sports Med. 2003, 15, 17–21. [Google Scholar] [CrossRef]
- Åberg, M.A.; Pedersen, N.L.; Torén, K.; Svartengren, M.; Bäckstrand, B.; Johnsson, T.; Cooper-Kuhn, C.M.; Åberg, N.D.; Nilsson, M.; Kuhn, H.G. Cardiovascular fitness is associated with cognition in young adulthood. Proc. Natl. Acad. Sci. USA 2009, 106, 20906–20911. [Google Scholar] [CrossRef]
- Tandon, P.S.; Tovar, A.; Jayasuriya, A.T.; Welker, E.; Schober, D.J.; Copeland, K.; Dev, D.A.; Murriel, A.L.; Amso, D.; Ward, D.S. The relationship between physical activity and diet and young children’s cognitive development: A systematic review. Prev. Med. Rep. 2016, 3, 379–390. [Google Scholar] [CrossRef]
- Berger, N.A.; Müller, A.; Brähler, E.; Philipsen, A.; de Zwaan, M. Association of symptoms of attention-deficit/hyperactivity disorder with symptoms of excessive exercising in an adult general population sample. BMC Psychiatry 2014, 14, 250. [Google Scholar] [CrossRef]
- Rommel, A.-S.; Lichtenstein, P.; Rydell, M.; Kuja-Halkola, R.; Asherson, P.; Kuntsi, J.; Larsson, H. Is physical activity causally associated with symptoms of attention-deficit/hyperactivity disorder? J. Am. Acad. Child Adolesc. Psychiatry 2015, 54, 565–570. [Google Scholar] [CrossRef]
- Labban, J.D.; Gapin, J.I.; Etnier, J.L. Physical activity and cognitive performance in children with attention deficit hyperactivity disorder (ADHD)-A randomized controlled trial testing the effects of a single bout of aerobic exercise on executive function. J. Sport Exerc. Psychol. 2019, 31, S12–S13. [Google Scholar]
- Bustamante, E.E.; Davis, C.L.; Frazier, S.L.; Rusch, D.; Fogg, L.F.; Atkins, M.S.; Marquez, D.X. Randomized controlled trial of exercise for ADHD and disruptive behavior disorders. Med. Sci. Sports Exerc. 2016, 48, 1397. [Google Scholar] [CrossRef] [PubMed]
- Benzing, V.; Schmidt, M. The effect of exergaming on executive functions in children with ADHD: A randomized clinical trial. Scand. J. Med. Sci. Sports 2019. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Green, S. (Eds.) Cochrane Collaboration: Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 [updated March 2011]. Available online: www.handbook.cochrane.org (accessed on 15 February 2019).
- Diamond, A.; Ling, D.S. Aerobic-exercise and resistance-training interventions have been among the least effective ways to improve executive functions of any method tried thus far. Dev. Cogn. Neurosci. 2018, 10, 100572. [Google Scholar] [CrossRef] [PubMed]
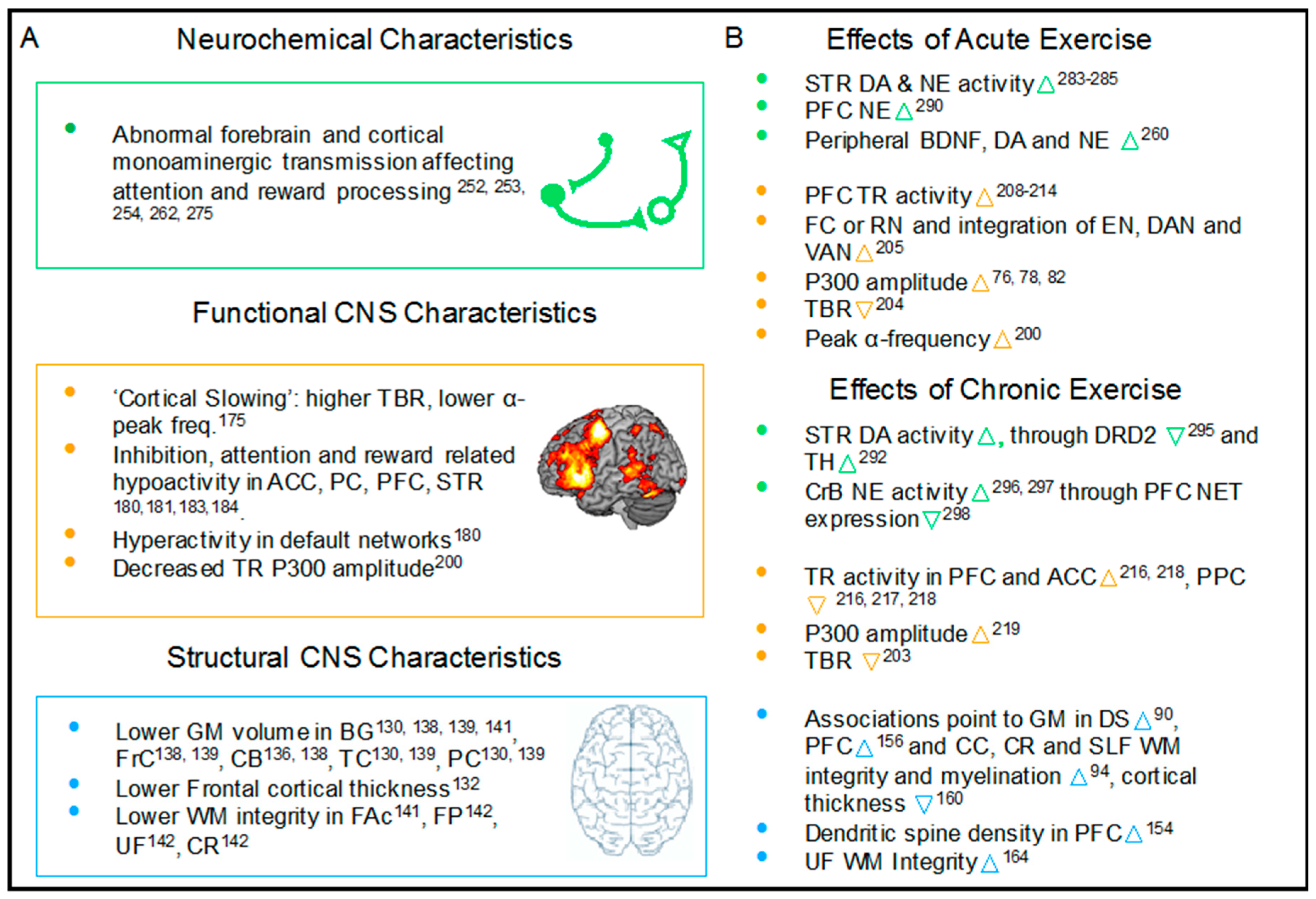
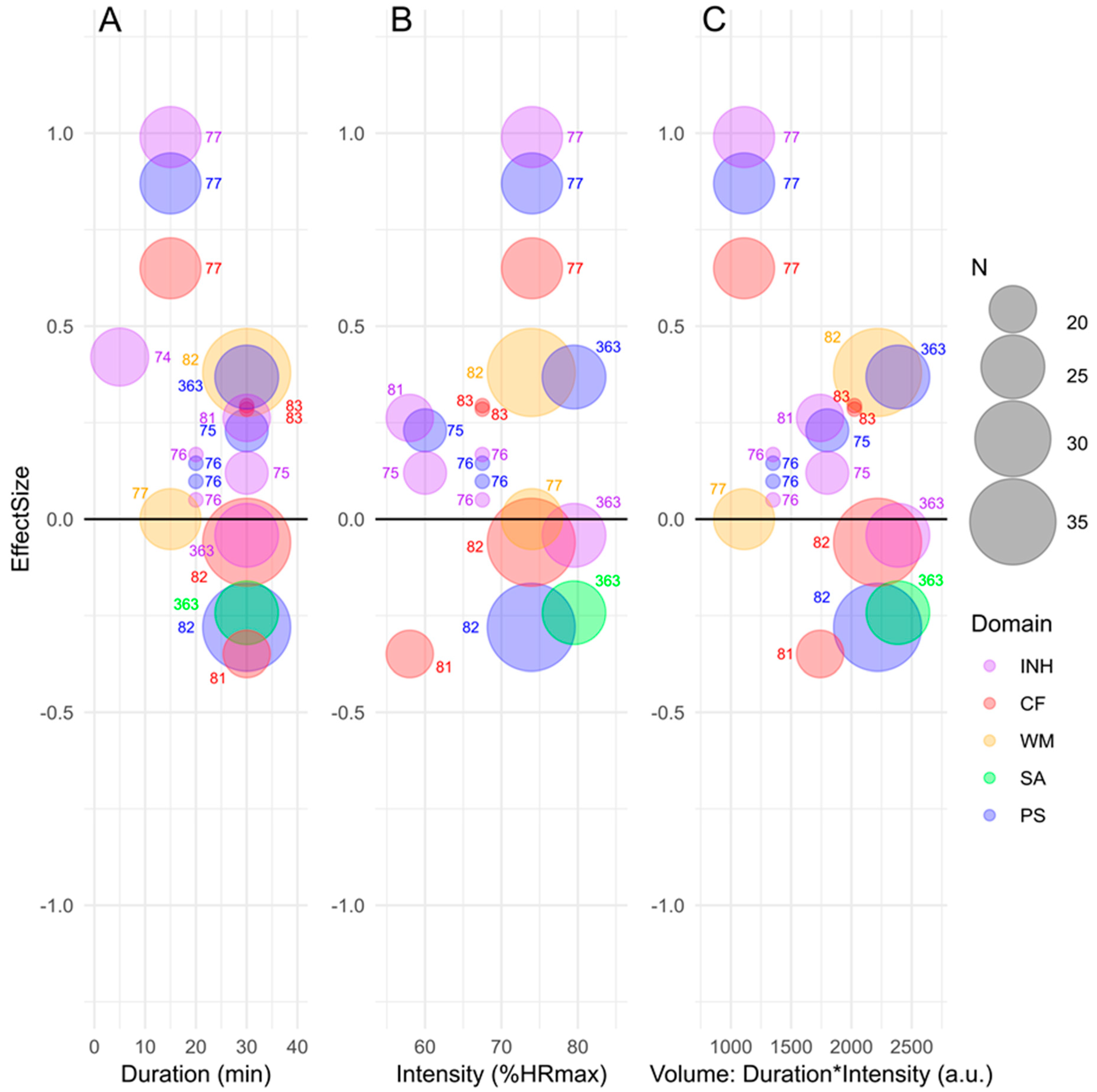
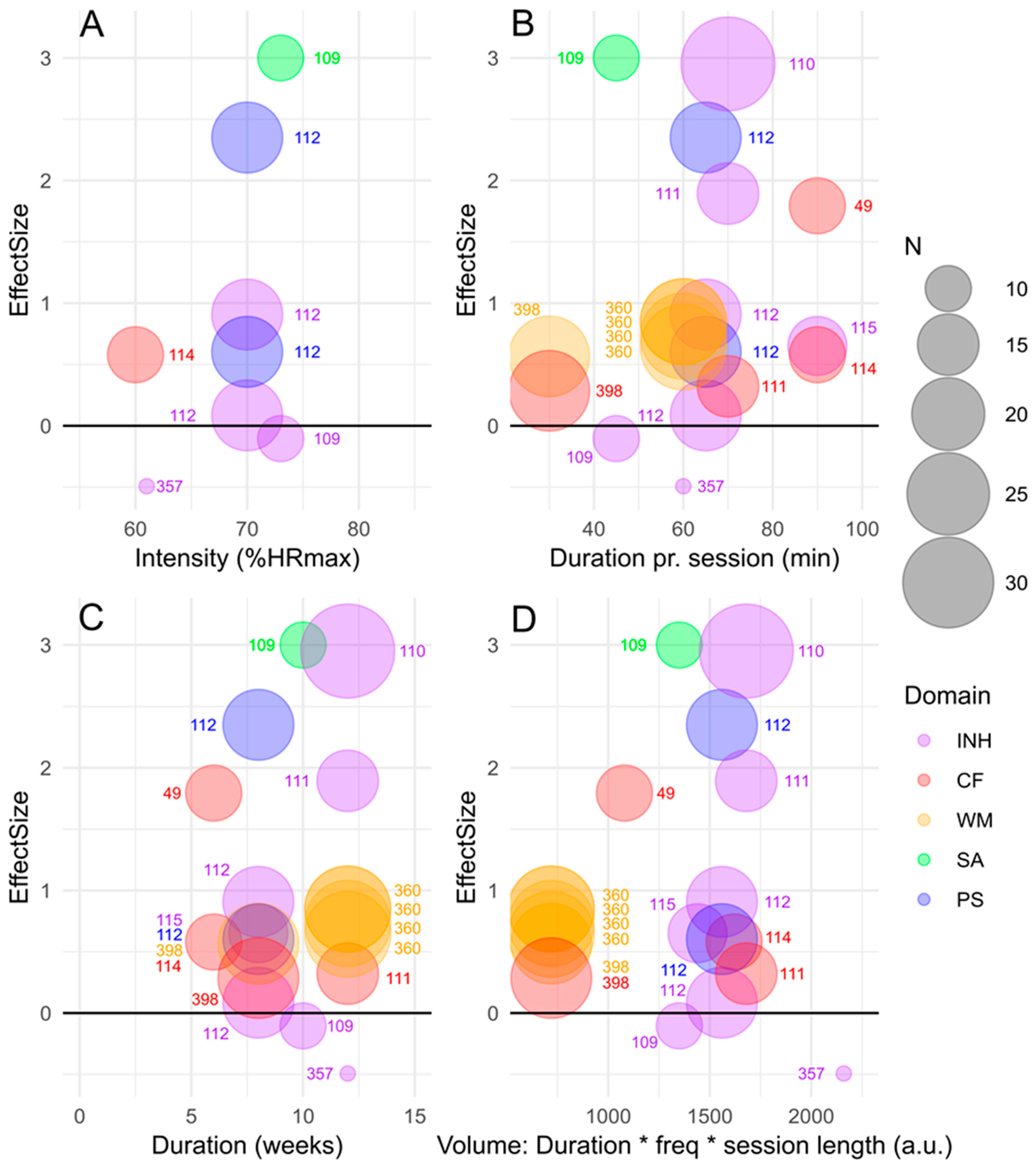
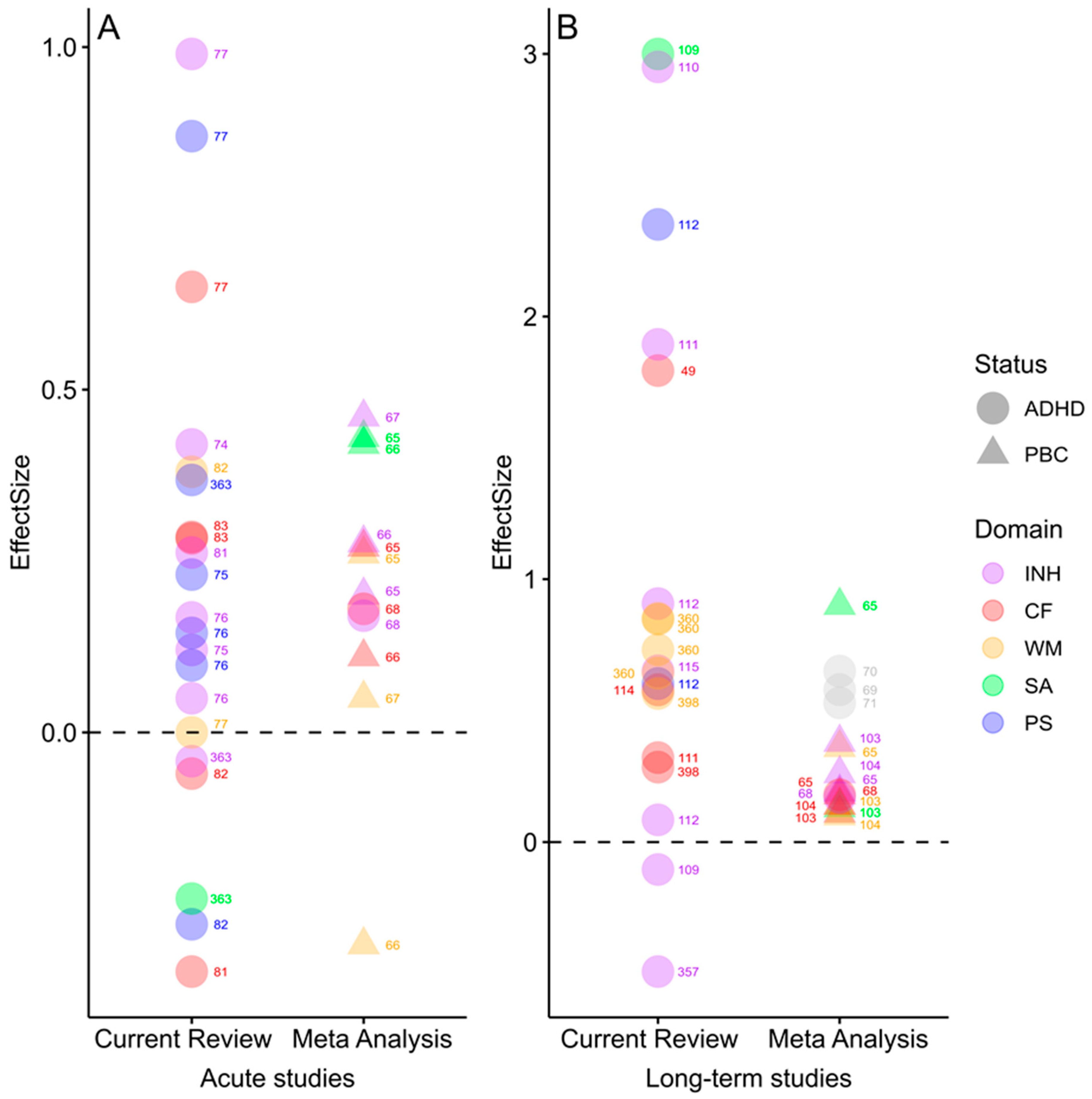
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Christiansen, L.; Beck, M.M.; Bilenberg, N.; Wienecke, J.; Astrup, A.; Lundbye-Jensen, J. Effects of Exercise on Cognitive Performance in Children and Adolescents with ADHD: Potential Mechanisms and Evidence-based Recommendations. J. Clin. Med. 2019, 8, 841. https://doi.org/10.3390/jcm8060841
Christiansen L, Beck MM, Bilenberg N, Wienecke J, Astrup A, Lundbye-Jensen J. Effects of Exercise on Cognitive Performance in Children and Adolescents with ADHD: Potential Mechanisms and Evidence-based Recommendations. Journal of Clinical Medicine. 2019; 8(6):841. https://doi.org/10.3390/jcm8060841
Chicago/Turabian StyleChristiansen, Lasse, Mikkel M. Beck, Niels Bilenberg, Jacob Wienecke, Arne Astrup, and Jesper Lundbye-Jensen. 2019. "Effects of Exercise on Cognitive Performance in Children and Adolescents with ADHD: Potential Mechanisms and Evidence-based Recommendations" Journal of Clinical Medicine 8, no. 6: 841. https://doi.org/10.3390/jcm8060841
APA StyleChristiansen, L., Beck, M. M., Bilenberg, N., Wienecke, J., Astrup, A., & Lundbye-Jensen, J. (2019). Effects of Exercise on Cognitive Performance in Children and Adolescents with ADHD: Potential Mechanisms and Evidence-based Recommendations. Journal of Clinical Medicine, 8(6), 841. https://doi.org/10.3390/jcm8060841





