Peptic Ulcer Disease: A Brief Review of Conventional Therapy and Herbal Treatment Options
Abstract
1. Introduction
2. Pathogenesis of Peptic Ulcer
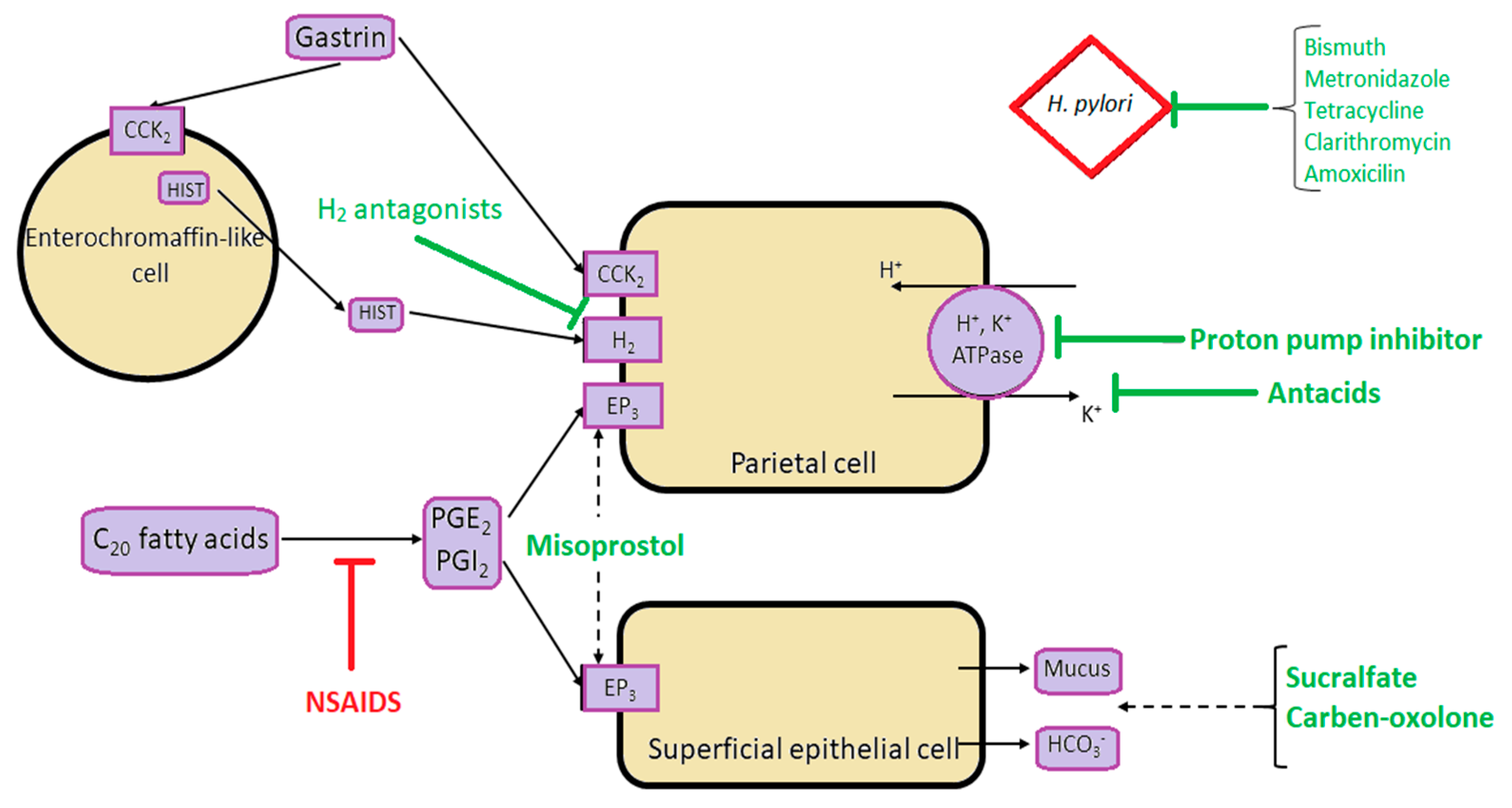
3. Treatment
3.1. Helicobacter pylori Eradication
3.2. NSAID-Associated Ulcer Disease and the Use of PPIs
3.3. Potassium-Competitive Acid Blockers
3.4. Future Research Questions
4. Alternative Therapy for Peptic Ulcer
4.1. The Effect on H. pylori Eradication
4.2. Korean Red Ginseng
4.3. Allium Sativum
4.4. Cistus Laurifolius
4.5. Zingiber Officinalis and Zingiber Zerumbet
4.6. Camellia Sinensis (Green Tea Polyphenols)
4.7. Curcuma Longa and Artemisia Asiatica
5. Herb–Drug Interactions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| IL1B | interleukin 1 beta |
| COX-1 | cyclooxygenase 1 |
| COX-2 | cyclooxygenase 2 |
| CYP | cytochrome |
| FDA | Food and drug administration |
| H. pylori | Helicobacter pylori |
| CCK2 | cholecystokinin receptor |
| PGE2 | prostaglandin E2 |
| PGI2 | prostaglandin I2 |
| EP3 | prostaglandin E receptor 3 |
| HIST | histamine |
| H2 receptor agonists | histamine-2 receptor agonists |
| NSAIDs | non-steroidal anti-inflammatory drugs |
| PPIs | proton pump inhibtors |
| 5-LOX | 5-lypoxigenase |
| iNOS | inducible nitric oxide synthase |
| SAC | S-allyl-L-cysteine |
| EGCG | epigallocatechin gallate |
| vacA | vacuolating cytotoxin A |
| CRS | cold restraint stress |
| PL | pylorus ligation |
| GERD | gastroesophageal reflux disease |
| LES | lower esophageal sphincter |
| IL | interleukin |
| ROS | reactive oxygen species |
| TNF-α | tumor necrosis factor-alpha |
| ICAM 1 | intercellular adhesion molecule-1 |
| CINC 2-beta | cytokine-induced neutrophil chemoattractant-2-beta |
| OATP1A1 | organic anion transporting protein 1a1 |
| OATP1A2 | anion transporting protein 1a2 |
| STW 5 | a complex herbal combination preparation composed of 9 different herbal extracts |
| CYP3A4 | cytochrome P450 3A4 |
References
- Narayanan, M.; Reddy, K.M.; Marsicano, E. Peptic ulcer disease and Helicobacter pylori infection. Mo. Med. 2018, 115, 219–224. [Google Scholar] [PubMed]
- Lanas, A.; Chan, F.K.L. Peptic ulcer disease. Lancet 2017, 390, 613–624. [Google Scholar] [CrossRef]
- Lanas, A.; García-Rodríguez, L.A.; Polo-Tomás, M.; Ponce, M.; Quintero, E.; Perez-Aisa, M.A.; Gisbert, J.P.; Bujanda, L.; Castro, M.; Muñoz, M.; et al. The changing face of hospitalisation due to gastrointestinal bleeding and perforation. Aliment. Pharmacol. Ther. 2011, 33, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Sonnenberg, A. Review article: Historic changes of helicobacter pylori-associated diseases. Aliment. Pharmacol. Ther. 2013, 38, 329–342. [Google Scholar] [CrossRef] [PubMed]
- Søreide, K.; Thorsen, K.; Harrison, E.M.; Bingener, J.; Møller, M.H.; Ohene-Yeboah, M.; Søreide, J.A. Perforated peptic ulcer. Lancet 2015, 386, 1288–1298. [Google Scholar] [CrossRef]
- Zhang, B.B.; Li, Y.; Liu, X.Q.; Wang, P.J.; Yang, B.; Bian, D.L. Association between vacA genotypes and the risk of duodenal ulcer: A meta-analysis. Mol. Biol. Rep. 2014, 41, 7241–7254. [Google Scholar] [CrossRef] [PubMed]
- Datta De, D.; Roychoudhury, S. To be or not to be: The host genetic factor and beyond in Helicobacter pylori mediated gastro-duodenal diseases. World J. Gastroenterol. 2015, 21, 2883–2895. [Google Scholar] [CrossRef]
- Lanas, Á.; Carrera-Lasfuentes, P.; Arguedas, Y.; García, S.; Bujanda, L.; Calvet, X.; Ponce, J.; Perez-Aísa, Á.; Castro, M.; Muñoz, M.; et al. Risk of upper and lower gastrointestinal bleeding in patients taking nonsteroidal anti-inflammatory drugs, antiplatelet agents, or anticoagulants. Clin. Gastroenterol. Hepatol. 2015, 13, 906–912.e2. [Google Scholar] [CrossRef]
- Masclee, G.M.; Valkhoff, V.E.; Coloma, P.M.; de Ridder, M.; Romio, S.; Schuemie, M.J.; Herings, R.; Gini, R.; Mazzaglia, G.; Picelli, G.; et al. Risk of upper gastrointestinal bleeding from different drug combinations. Gastroenterology 2014, 147, 784–792. [Google Scholar] [CrossRef]
- Huang, J.Q.; Sridhar, S.; Hunt, R.H. Role of helicobacter pylori infection and non-steroidal anti-inflammatory drugs in peptic-ulcer disease: A meta-analysis. Lancet 2002, 359, 14–22. [Google Scholar] [CrossRef]
- Charpignon, C.; Lesgourgues, B.; Pariente, A.; Nahon, S.; Pelaquier, A.; Gatineau-Sailliant, G.; Roucayrol, A.M.; Courillon-Mallet, A.; Group de l’Observatoire National des Ulcères de l’Association Nationale des HépatoGastroentérologues des Hôpitaux Généraux (ANGH). Peptic ulcer disease: One in five is related to neither Helicobacter pylori nor aspirin/NSAID intake. Aliment. Pharmacol. Ther. 2013, 38, 946–954. [Google Scholar] [CrossRef] [PubMed]
- Levenstein, S.; Rosenstock, S.; Jacobsen, R.K.; Jorgensen, T. Psychological stress increases risk for peptic ulcer, regardless of Helicobacter pylori infection or use of nonsteroidal anti-inflammatory drugs. Clin. Gastroenterol. Hepatol. 2015, 13, 498–506.e1. [Google Scholar] [CrossRef] [PubMed]
- McColl, K.E. Helicobacter pylori-negative nonsteroidal anti-inflammatory drug-negative ulcer. Gastroenterol. Clin. N. Am. 2009, 38, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Siddique, O.; Ovalle, A.; Siddique, A.S.; Moss, S.F. Helicobacter pylori infection: An update for the internist in the age of increasing global antibiotic resistance. Am. J. Med. 2018, 131, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Hooi, J.K.Y.; Lai, W.Y.; Ng, W.K.; Suen, M.M.Y.; Underwood, F.E.; Tanyingoh, D.; Malfertheiner, P.; Graham, D.Y.; Wong, V.W.S.; Wu, J.C.Y.; et al. Global prevalence of Helicobacter pylori infection: Systematic review and meta-analysis. Gastroenterology 2017, 153, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.; Coudron, P.E.; McCuen, R.W.; Harrington, L.; Chu, S.; Schubert, M.L. H. Pylori acutely inhibits gastric secretion by activating CGRP sensory neurons coupled to stimulation of somatostatin and inhibition of histamine secretion. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G715–G722. [Google Scholar] [CrossRef] [PubMed]
- El-Omar, E.M.; Oien, K.; El-Nujumi, A.; Gillen, D.; Wirz, A.; Dahill, S.; Williams, C.; Ardill, J.E.; McColl, K.E. Helicobacter pylori infection and chronic gastric acid hyposecretion. Gastroenterology 1997, 113, 15–24. [Google Scholar] [CrossRef]
- Moss, S.F.; Legon, S.; Bishop, A.E.; Polak, J.M.; Calam, J. Effect of helicobacter pylori on gastric somatostatin in duodenal ulcer disease. Lancet 1992, 340, 930–932. [Google Scholar] [CrossRef]
- Bhala, N.; Emberson, J.; Merhi, A.; Abramson, S.; Arber, N.; Baron, J.A.; Bombardier, C.; Cannon, C.; Farkouh, M.E.; FitzGerald, G.A.; et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet 2013, 382, 769–779. [Google Scholar]
- Bjarnason, I.; Scarpignato, C.; Takeuchi, K.; Rainsford, K.D. Determinants of the short-term gastric damage caused by NSAIDs in man. Aliment. Pharmacol. Ther. 2007, 26, 95–106. [Google Scholar] [CrossRef]
- Mössner, J. The indications, applications, and risks of proton pump inhibitors. Dtsch. Arztebl. Int. 2016, 113, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.L.; Fixen, D.R.; Linnebur, S.A. Adverse effects of proton-pump inhibitor use in older adults: A review of the evidence. Ther. Adv. Drug Saf. 2017, 8, 273–297. [Google Scholar] [CrossRef] [PubMed]
- Pension, J.; Wormsley, K.G. Adverse reactions and interactions with H2-receptor antagonists. Med. Toxicol. 1986, 1, 192–216. [Google Scholar] [CrossRef]
- Maton, P.N.; Burton, M.E. Antacids revisited: A review of their clinical pharmacology and recommended therapeutic use. Drugs 1999, 57, 855–870. [Google Scholar] [CrossRef] [PubMed]
- Mizokami, Y.; Oda, K.; Funao, N.; Nishimura, A.; Soen, S.; Kawai, T.; Ashida, K.; Sugano, K. Vonoprazan prevents ulcer recurrence during long-term NSAID therapy: Randomised, lansoprazole-controlled non-inferiority and single-blind extension study. Gut 2018, 67, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, A.; Yoshio, T.; Muramatsu, Y.; Horiuchi, Y.; Ishiyama, A.; Hirasawa, T.; Tsuchida, T.; Sasaki, Y.; Fujisaki, J. Vonoprazan is superior to rabeprazole for healing endoscopic submucosal dissection: Induced ulcers. Digestion 2018, 97, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Kawai, T.; Oda, K.; Funao, N.; Nishimura, A.; Matsumoto, Y.; Mizokami, Y.; Ashida, K.; Sugano, K. Vonoprazan prevents low-dose aspirin-associated ulcer recurrence: Randomised phase 3 study. Gut 2018, 67, 1033–1041. [Google Scholar] [CrossRef]
- Kagawa, T.; Iwamuro, M.; Ishikawa, S.; Ishida, M.; Kuraoka, S.; Sasaki, K.; Sakakihara, I.; Izumikawa, K.; Yamamoto, K.; Takahashi, S.; et al. Vonoprazan prevents bleeding from endoscopic submucosal dissection-induced gastric ulcers. Aliment. Pharmacol. Ther. 2016, 44, 583–591. [Google Scholar] [CrossRef]
- Tsuchiya, I.; Kato, Y.; Tanida, E.; Masui, Y.; Kato, S.; Nakajima, A.; Izumi, M. Effect of vonoprazan on the treatment of artificial gastric ulcers after endoscopic submucosal dissection: Prospective randomized controlled trial. Dig. Endosc. 2017, 29, 576–583. [Google Scholar] [CrossRef]
- Marks, I.N. Sucralfate-safety and side effects. Scand. J. Gastroenterol. Suppl. 1991, 26, 36–42. [Google Scholar] [CrossRef]
- Aubert, J.; Bejan-Angoulvant, T.; Jonville-Bera, A.P. [pharmacology of misoprostol (pharmacokinetic data, adverse effects and teratogenic effects)]. J. Gynecol. Obstet. Biol. Reprod. (Paris) 2014, 43, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Gisbert, J.P.; Kuipers, E.J.; Axon, A.T.; Bazzoli, F.; Gasbarrini, A.; Atherton, J.; Graham, D.Y.; et al. Management of Helicobacter pylori infection-the maastricht V/Florence consensus report. Gut 2017, 66, 6–30. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.Y.; Wu, M.S.; Chen, C.Y.; Bair, M.J.; Chou, C.K.; Lin, J.T.; Liou, J.M.; Taiwan Gastrointestinal Disease and Helicobacter Consortium. Systematic review with meta-analysis: The efficacy of levofloxacin triple therapy as the first- or second-line treatments of Helicobacter pylori infection. Aliment. Pharmacol. Ther. 2016, 44, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Shiota, S.; Reddy, R.; Alsarraj, A.; El-Serag, H.B.; Graham, D.Y. Antibiotic resistance of Helicobacter pylori among male united states veterans. Clin. Gastroenterol. Hepatol. 2015, 13, 1616–1624. [Google Scholar] [CrossRef] [PubMed]
- Graham, D.Y.; Lee, Y.C.; Wu, M.S. Rational Helicobacter pylori therapy: Evidence-based medicine rather than medicine-based evidence. Clin. Gastroenterol. Hepatol. 2014, 12, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Fallone, C.A.; Chiba, N.; van Zanten, S.V.; Fischbach, L.; Gisbert, J.P.; Hunt, R.H.; Jones, N.L.; Render, C.; Leontiadis, G.I.; Moayyedi, P.; et al. The toronto consensus for the treatment of Helicobacter pylori infection in adults. Gastroenterology 2016, 151, 51–69. [Google Scholar] [CrossRef] [PubMed]
- Dore, M.P.; Lu, H.; Graham, D.Y. Role of bismuth in improving Helicobacter pylori eradication with triple therapy. Gut 2016, 65, 870–878. [Google Scholar] [CrossRef]
- Sun, Q.; Liang, X.; Zheng, Q.; Liu, W.; Xiao, S.; Gu, W.; Lu, H. High efficacy of 14-day triple therapy-based, bismuth-containing quadruple therapy for initial Helicobacter pylori eradication. Helicobacter 2010, 15, 233–238. [Google Scholar] [CrossRef]
- Chey, W.D.; Leontiadis, G.I.; Howden, C.W.; Moss, S.F. Acg clinical guideline: Treatment of Helicobacter pylori infection. Am. J. Gastroenterol. 2017, 112, 212–239. [Google Scholar] [CrossRef]
- Gisbert, J.P.; Calvet, X. Review article: Rifabutin in the treatment of refractory Helicobacter pylori infection. Aliment. Pharmacol. Ther. 2012, 35, 209–221. [Google Scholar] [CrossRef]
- Strand, D.S.; Kim, D.; Peura, D.A. 25 years of proton pump inhibitors: A comprehensive review. Gut Liver 2017, 11, 27–37. [Google Scholar] [CrossRef] [PubMed]
- DaCosta DiBonaventura, M.; Yuan, Y.; Wagner, J.S.; L’Italien, G.J.; Lescrauwaet, B.; Langley, P. The burden of viral hepatitis C in Europe: A propensity analysis of patient outcomes. Eur. J. Gastroenterol. Hepatol. 2012, 24, 869–877. [Google Scholar] [CrossRef] [PubMed]
- Rostom, A.; Muir, K.; Dube, C.; Lanas, A.; Jolicoeur, E.; Tugwell, P. Prevention of NSAID-related upper gastrointestinal toxicity: A meta-analysis of traditional NSAIDs with gastroprotection and COX-2 inhibitors. Drug Healthc. Patient Saf. 2009, 1, 47–71. [Google Scholar] [CrossRef] [PubMed]
- Spechler, S.J. Proton pump inhibitors: What the internist needs to know. Med. Clin. N. Am. 2019, 103, 1–14. [Google Scholar] [CrossRef]
- Lambert, A.A.; Lam, J.O.; Paik, J.J.; Ugarte-Gil, C.; Drummond, M.B.; Crowell, T.A. Risk of community-acquired pneumonia with outpatient proton-pump inhibitor therapy: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0128004. [Google Scholar] [CrossRef]
- Kwok, C.S.; Arthur, A.K.; Anibueze, C.I.; Singh, S.; Cavallazzi, R.; Loke, Y.K. Risk of clostridium difficile infection with acid suppressing drugs and antibiotics: Meta-analysis. Am. J. Gastroenterol. 2012, 107, 1011–1019. [Google Scholar] [CrossRef]
- Deshpande, A.; Pasupuleti, V.; Thota, P.; Pant, C.; Mapara, S.; Hassan, S.; Rolston, D.D.; Sferra, T.J.; Hernandez, A.V. Acid-suppressive therapy is associated with spontaneous bacterial peritonitis in cirrhotic patients: A meta-analysis. J. Gastroenterol. Hepatol. 2013, 28, 235–242. [Google Scholar] [CrossRef]
- Haigh, C.R.; Attwood, S.E.; Thompson, D.G.; Jankowski, J.A.; Kirton, C.M.; Pritchard, D.M.; Varro, A.; Dimaline, R. Gastrin induces proliferation in Barrett’s metaplasia through activation of the CCK2 receptor. Gastroenterology 2003, 124, 615–625. [Google Scholar] [CrossRef] [PubMed]
- Laine, L.; Ahnen, D.; McClain, C.; Solcia, E.; Walsh, J.H. Review article: Potential gastrointestinal effects of long-term acid suppression with proton pump inhibitors. Aliment. Pharmacol. Ther. 2000, 14, 651–668. [Google Scholar] [CrossRef]
- Lam, J.R.; Schneider, J.L.; Zhao, W.; Corley, D.A. Proton pump inhibitor and histamine 2 receptor antagonist use and vitamin B12 deficiency. JAMA 2013, 310, 2435–2442. [Google Scholar] [CrossRef] [PubMed]
- Koivisto, T.T.; Rautelin, H.I.; Voutilainen, M.E.; Heikkinen, M.T.; Koskenpato, J.P.; Färkkilä, M.A. First-line eradication therapy for Helicobacter pylori in primary health care based on antibiotic resistance: Results of three eradication regimens. Aliment. Pharmacol. Ther. 2005, 21, 773–782. [Google Scholar] [CrossRef] [PubMed]
- Lew, E.A. Review article: Pharmacokinetic concerns in the selection of anti-ulcer therapy. Aliment. Pharmacol. Ther. 1999, 13 (Suppl. S5), 11–16. [Google Scholar] [CrossRef] [PubMed]
- Gilard, M.; Arnaud, B.; Le Gal, G.; Abgrall, J.F.; Boschat, J. Influence of omeprazol on the antiplatelet action of clopidogrel associated to aspirin. J. Thromb. Haemost. 2006, 4, 2508–2509. [Google Scholar] [CrossRef] [PubMed]
- Ghebremariam, Y.T.; Lee, J.C.; LePendu, P.; Erlanson, D.A.; Slaviero, A.; Shah, N.H.; Leiper, J.M.; Cooke, J.P. Response to letters regarding article, “unexpected effect of proton pump inhibitors: Elevation of the cardiovascular risk factor asymmetric dimethylarginine”. Circulation 2014, 129, e428. [Google Scholar] [CrossRef] [PubMed]
- Merwat, S.N.; Spechler, S.J. Might the use of acid-suppressive medications predispose to the development of eosinophilic esophagitis? Am. J. Gastroenterol. 2009, 104, 1897–1902. [Google Scholar] [CrossRef] [PubMed]
- Lanas, A. We are using too many PPIs, and we need to stop: A European perspective. Am. J. Gastroenterol. 2016, 111, 1085–1086. [Google Scholar] [CrossRef] [PubMed]
- Wong, G.L.; Wong, V.W.; Chan, Y.; Ching, J.Y.; Au, K.; Hui, A.J.; Lai, L.H.; Chow, D.K.; Siu, D.K.; Lui, Y.N.; et al. High incidence of mortality and recurrent bleeding in patients with helicobacter pylori-negative idiopathic bleeding ulcers. Gastroenterology 2009, 137, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Debraekeleer, A.; Remaut, H. Future perspective for potential helicobacter pylori eradication therapies. Future Microbiol. 2018, 13, 671–687. [Google Scholar] [CrossRef] [PubMed]
- Rates, S.M. Plants as source of drugs. Toxicon 2001, 39, 603–613. [Google Scholar] [CrossRef]
- Yesilada, E.; Gürbüz, I.; Shibata, H. Screening of Turkish antiulcerogenic folk remedies for anti-Helicobacter pylori activity. J. Ethnopharmacol. 1999, 66, 289–293. [Google Scholar] [CrossRef]
- Falcão, H.S.; Mariath, I.R.; Diniz, M.F.; Batista, L.M.; Barbosa-Filho, J.M. Plants of the American continent with antiulcer activity. Phytomedicine 2008, 15, 132–146. [Google Scholar] [CrossRef] [PubMed]
- Chanda, S.; Baravalia, Y.; Kaneria, M. Protective effect of Polyalthia longifolia var. Pendula leaves on ethanol and ethanol/HCL induced ulcer in rats and its antimicrobial potency. Asian Pac. J. Trop. Med. 2011, 4, 673–679. [Google Scholar] [CrossRef]
- Palle, S.; Kanakalatha, A.; Kavitha, C.N. Gastroprotective and antiulcer effects of Celastrus paniculatus seed oil against several gastric ulcer models in rats. J. Diet. Suppl. 2018, 15, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, E.M. Plants: An alternative source for antimicrobials. J. Appl. Pharm. Sci. 2011, 1, 16–20. [Google Scholar]
- Silva, N.C.C.; Fernandes Júnior, A. Biological properties of medicinal plants: A review of their antimicrobial activity. J. Venom. Anim. Toxins Include. Trop. Dis. 2010, 16, 402–413. [Google Scholar] [CrossRef]
- Dikid, T.; Jain, S.K.; Sharma, A.; Kumar, A.; Narain, J.P. Emerging & re-emerging infections in India: An overview. Indian J. Med. Res. 2013, 138, 19–31. [Google Scholar] [PubMed]
- Abdallah, E.M. Medicinal plants with antibacterial properties against helicobacter pylori: A brief review. Curr. Trends Nutraceuticals 2016, 1, 3. [Google Scholar]
- Langmead, L.; Rampton, D.S. Review article: Herbal treatment in gastrointestinal and liver disease—Benefits and dangers. Aliment. Pharmacol. Ther. 2001, 15, 1239–1252. [Google Scholar] [CrossRef] [PubMed]
- Meshram, N.; Ojha, M.; Singh, A.; Alexander, A.; Sharma, M. Significance of medicinal plant used for the treatment of peptic ulcer. Asian J. Pharm. Technol. 2015, 5, 32–37. [Google Scholar] [CrossRef]
- Ricci, V.; Zarrilli, R.; Romano, M. Voyage of helicobacter pylori in human stomach: Odyssey of a bacterium. Dig. Liver Dis. 2002, 34, 2–8. [Google Scholar] [CrossRef]
- Mital, B.; Kansara, A.J.J. Possible interactions between garlic and conventional drugs: A review. Pharm. Biol. Eval. 2017, 4, 73–81. [Google Scholar]
- Tuorkey, M.; Karolin, K. Anti-ulcer activity of curcumin one experimental gastric ulcer in rats and its effect on oxidative stress/antioxidant, IL-6 and enzyme activities. Biomed. Environ. Sci. 2009, 22, 488–495. [Google Scholar] [CrossRef]
- Pan, M.H.; Hsieh, M.C.; Hsu, P.C.; Ho, S.Y.; Lai, C.S.; Wu, H.; Sang, S.; Ho, C.T. 6-shogaol suppressed lipopolysaccharide-induced up-expression of inos and cox-2 in murine macrophages. Mol. Nutr. Food Res. 2008, 52, 1467–1477. [Google Scholar] [CrossRef] [PubMed]
- Siddaraju, M.N.; Dharmesh, S.M. Inhibition of gastric H+, K+-ATPase and helicobacter pylori growth by phenolic antioxidants of Zingiber officinale. Mol. Nutr. Food Res. 2007, 51, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Sripramote, M.; Lekhyananda, N. A randomized comparison of ginger and vitamin B6 in the treatment of nausea and vomiting of pregnancy. J. Med. Assoc. Thail. 2003, 86, 846–853. [Google Scholar]
- Ustün, O.; Ozçelik, B.; Akyön, Y.; Abbasoglu, U.; Yesilada, E. Flavonoids with anti-Helicobacter pylori activity from Cistus laurifolius leaves. J. Ethnopharmacol. 2006, 108, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Asher, G.N.; Corbett, A.H.; Hawke, R.L. Common herbal dietary supplement-drug interactions. Am. Fam. Physician 2017, 96, 101–107. [Google Scholar] [PubMed]
- Amber Nawab, N.F. Review on green tea constituents and its negative effects. Pharm. Innov. J. 2015, 4, 21–24. [Google Scholar]
- Vakil, N. Helicobacter pylori treatment: A practical approach. Am. J. Gastroenterol. 2006, 101, 497–499. [Google Scholar] [CrossRef] [PubMed]
- Campo, S.M.; Zullo, A.; Hassan, C.; Morini, S. Antibiotic treatment strategies for Helicobacter pylori infection. Recent Pat. Antiinfect. Drug Discov. 2007, 2, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Han, S.U.; Kim, Y.B.; Joo, H.J.; Hahm, K.B.; Lee, W.H.; Cho, Y.K.; Kim, D.Y.; Kim, M.W. Helicobacter pylori infection promotes gastric carcinogenesis in a mice model. J. Gastroenterol. Hepatol. 2002, 17, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Shin, Y.W.; Hahm, K.B. Phytoceuticals: Mighty but ignored weapons against Helicobacter pylori infection. J. Dig. Dis. 2008, 9, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lee, J.A.; Kim, Y.B.; Lee, K.M.; Hahm, K.B. A randomized controlled trial assessing Korea red ginseng treatment of Helicobacter pylori-associated chronic gastritis. Korean J. Med. 2007, 72, 20–28. [Google Scholar]
- Park, S.; Yeo, M.; Jin, J.H.; Lee, K.M.; Jung, J.Y.; Choue, R.; Cho, S.W.; Hahm, K.B. Rescue of Helicobacter pylori-induced cytotoxicity by red ginseng. Dig. Dis. Sci. 2005, 50, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Park, J.M.; Han, Y.M.; Kangwan, N.; Lee, S.Y.; Jung, M.K.; Kim, E.H.; Hahm, K.B. S-allyl cysteine alleviates nonsteroidal anti-inflammatory drug-induced gastric mucosal damages by increasing cyclooxygenase-2 inhibition, heme oxygenase-1 induction, and histone deacetylation inhibition. J. Gastroenterol. Hepatol. 2014, 29 (Suppl. S4), 80–92. [Google Scholar] [CrossRef] [PubMed]
- Iimuro, M.; Shibata, H.; Kawamori, T.; Matsumoto, T.; Arakawa, T.; Sugimura, T.; Wakabayashi, K. Suppressive effects of garlic extract on Helicobacter pylori-induced gastritis in Mongolian gerbils. Cancer Lett. 2002, 187, 61–68. [Google Scholar] [CrossRef]
- Trio, P.Z.; You, S.; He, X.; He, J.; Sakao, K.; Hou, D.X. Chemopreventive functions and molecular mechanisms of garlic organosulfur compounds. Food Funct. 2014, 5, 833–844. [Google Scholar] [CrossRef]
- Cañizares, P.; Gracia, I.; Gómez, L.A.; Martín de Argila, C.; Boixeda, D.; García, A.; de Rafael, L. Allyl-thiosulfinates, the bacteriostatic compounds of garlic against Helicobacter pylori. Biotechnol. Prog. 2004, 20, 397–401. [Google Scholar] [CrossRef]
- Bovicelli, P.; D’Angelo, V.; Collalto, D.; Verzina, A.; D’Antona, N.; Lambusta, D. Efficient synthesis of polyoxygenated flavones from naturally occurring flavanones. J. Pharm. Pharmacol. 2007, 59, 1697–1701. [Google Scholar] [CrossRef]
- Li, H.Q.; Xu, C.; Li, H.S.; Xiao, Z.P.; Shi, L.; Zhu, H.L. Metronidazole-flavonoid derivatives as anti-Helicobacter pylori agents with potent inhibitory activity against HPE-induced interleukin-8 production by AGS cells. ChemMedChem 2007, 2, 1361–1369. [Google Scholar] [CrossRef]
- Afdhal, N.; Reddy, K.R.; Nelson, D.R.; Lawitz, E.; Gordon, S.C.; Schiff, E.; Nahass, R.; Ghalib, R.; Gitlin, N.; Herring, R.; et al. Ledipasvir and sofosbuvir for previously treated HCV genotype 1 infection. N. Engl. J. Med. 2014, 370, 1483–1493. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Mullick, H.I.; Banerjee, J.; Ghosh, A. Zingiber officinale: A natural gold. Int. J. Pharm. Bio-Sci. 2011, 2, 283–294. [Google Scholar]
- Jiang, S.Z.; Wang, N.S.; Mi, S.Q. Plasma pharmacokinetics and tissue distribution of [6]-gingerol in rats. Biopharm. Drug Dispos. 2008, 29, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Sidahmed, H.M.; Hashim, N.M.; Abdulla, M.A.; Ali, H.M.; Mohan, S.; Abdelwahab, S.I.; Taha, M.M.; Fai, L.M.; Vadivelu, J. Antisecretory, gastroprotective, antioxidant and anti-Helicobcter pylori activity of Zerumbone from Zingiber zerumbet (l.) smith. PLoS ONE 2015, 10, e0121060. [Google Scholar] [CrossRef] [PubMed]
- Fujiki, H.; Suganuma, M.; Okabe, S.; Kurusu, M.; Imai, K.; Nakachi, K. Involvement of TNF-alpha changes in human cancer development, prevention and palliative care. Mech. Ageing Dev. 2002, 123, 1655–1663. [Google Scholar] [CrossRef]
- Matsubara, S.; Shibata, H.; Ishikawa, F.; Yokokura, T.; Takahashi, M.; Sugimura, T.; Wakabayashi, K. Suppression of helicobacter pylori-induced gastritis by green tea extract in Mongolian gerbils. Biochem. Biophys. Res. Commun. 2003, 310, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, P.; Rossi, G.; Tombola, F.; Pancotto, L.; Lauretti, L.; Del Giudice, G.; Zoratti, M. Red wine and green tea reduce H pylori- or VacA-induced gastritis in a mouse model. World J. Gastroenterol. 2007, 13, 349–354. [Google Scholar] [CrossRef]
- Rao, C.V.; Verma, A.R.; Vijayakumar, M.; Rastogi, S. Gastroprotective effect of standardized extract of Ficus glomerata fruit on experimental gastric ulcers in rats. J. Ethnopharmacol. 2008, 115, 323–326. [Google Scholar] [CrossRef]
- Salehi, M. Medicinal plants for management of gastroesophageal reflux disease: A review of animal and human studies. J. Altern. Complement. Med. 2010, 23, 82–95. [Google Scholar] [CrossRef]
- Oh, T.Y.; Lee, J.S.; Ahn, B.O.; Cho, H.; Kim, W.B.; Kim, Y.B.; Surh, Y.J.; Cho, S.W.; Hahm, K.B. Oxidative damages are critical in pathogenesis of reflux esophagitis: Implication of antioxidants in its treatment. Free Radic. Biol. Med. 2001, 30, 905–915. [Google Scholar] [CrossRef]
- Mahattanadul, S.; Radenahmad, N.; Phadoongsombut, N.; Chuchom, T.; Panichayupakaranant, P.; Yano, S.; Reanmongkol, W. Effects of curcumin on reflux esophagitis in rats. J. Nat. Med. 2006, 60, 198–205. [Google Scholar] [CrossRef] [PubMed]
- Hajda, J.; Rentsch, K.M.; Gubler, C.; Steinert, H.; Stieger, B.; Fattinger, K. Garlic extract induces intestinal P-glycoprotein, but exhibits no effect on intestinal and hepatic CYP3A4 in humans. Eur. J. Pharm. Sci. 2010, 41, 729–735. [Google Scholar] [CrossRef] [PubMed]
- Alissa, E.M. Medicinal herbs and therapeutic drugs interactions. Ther. Drug Monit. 2014, 36, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Williams, K.M.; Liauw, W.S.; Ammit, A.J.; Roufogalis, B.D.; Duke, C.C.; Day, R.O.; McLachlan, A.J. Effect of ginkgo and ginger on the pharmacokinetics and pharmacodynamics of warfarin in healthy subjects. Br. J. Clin. Pharmacol. 2005, 59, 425–432. [Google Scholar] [CrossRef]
- Abebe, W. Herbal medication: Potential for adverse interactions with analgesic drugs. J. Clin. Pharm. Ther. 2002, 27, 391–401. [Google Scholar] [CrossRef]
- Malati, C.Y.; Robertson, S.M.; Hunt, J.D.; Chairez, C.; Alfaro, R.M.; Kovacs, J.A.; Penzak, S.R. Influence of panax ginseng on cytochrome p450 (CYP)3a and p-glycoprotein (P-gp) activity in healthy participants. J. Clin. Pharmacol. 2012, 52, 932–939. [Google Scholar] [CrossRef]
- Zhou, S.; Lim, L.Y.; Chowbay, B. Herbal modulation of p-glycoprotein. Drug Metab. Rev. 2004, 36, 57–104. [Google Scholar] [CrossRef]
- Werba, J.P.; Giroli, M.; Cavalca, V.; Nava, M.C.; Tremoli, E.; Dal Bo, L. The effect of green tea on simvastatin tolerability. Ann. Int. Med. 2008, 149, 286–287. [Google Scholar] [CrossRef]
- Sorkin, E.M.; Darvey, D.L. Review of cimetidine drug interactions. Drug Intell. Clin. Pharm. 1983, 17, 110–120. [Google Scholar] [CrossRef]
- Feely, J. Interaction of cimetidine with other drugs. S. Med. J. 1983, 76, 753–758. [Google Scholar] [CrossRef]
- Broughton, L.J.; Rogers, H.J. Decreased systemic clearance of caffeine due to cimetidine. Br. J. Clin. Pharmacol. 1981, 12, 155–159. [Google Scholar] [CrossRef] [PubMed]
| Medicine | Mechanism of Action | Adverse Effects | References | |
|---|---|---|---|---|
| Proton Pump Inhibitors (PPIs) | Omeprazole | Inhibition of the gastric H+/K+-ATPase (proton pump) enzyme system | Headache Abdominal pain Diarrhea Nausea Vomiting Constipation Flatulence Vitamin B12 deficiency Osteoporosis | [21,22] |
| Lansoprazole | ||||
| Rabeprazole | ||||
| Esomeprazole | ||||
| Pantoprazole | ||||
| H2 Receptor Blockers | Cimetidine | Blocking the action of histamine at the histamine H2 receptors of parietal cells | Headache Anxiety Depression Dizziness Cardiovascular events Thrombocytopenia | [23] |
| Famotidine | ||||
| Nizatidine | ||||
| Ranitidine | ||||
| Antacids | Aluminum hydroxide | Increases gastric pH to greater than four, and inhibits the proteolytic activity of pepsin | Frequency not defined: Nausea Vomiting Hypophosphatemia Chalky taste Constipation Abdominal cramping Diarrhea Electrolyte imbalance | [24] |
| Magnesium hydroxide | Causes osmotic retention of fluid | |||
| Potassium-Competitive Acid Blocker | Vonoprazan | Inhibits H+, K+-ATPase in gastric parietal cells at the final stage of the acid secretory pathway | Nasopharyngitis Fall Contusion Diarrhea Upper respiratory tract inflammation Eczema Constipation Back pain | [25,26,27,28,29] |
| Cytoprotective Agents | Misoprostol | Stimulate mucus production and enhance blood flow throughout the lining of the gastrointestinal tract | Diarrhea Abdominal pain Headache Constipation | [30,31] |
| Sucralfate | ||||
| Type | Duration | Efficiency | References |
|---|---|---|---|
| First line | |||
| Standard triple therapy: | |||
| PPI + two antibiotics (clarithromycin + metronidazile or amoxicilin) | 7–14 days | 70–85% | [32] |
| Second line | |||
| Bismuth-containing quadruple therapy: | |||
| PPI + bismuth salt + tetracycline + metronidazole | 14 days | 77–93% | [33,34] |
| Non-bismuth based concomitant therapy: | |||
| PPI + clarithromycin + amoxicillin + metronidazole | 14 days | 75–90% | |
| Levofloxacin triple therapy: | |||
| PPI + amoxicillin + levofloxacin | 14 days | 74–81% | |
| Salvage regimens | |||
| Rifabutin-based triple therapy: | |||
| PPI + rifabutin + amoxicillin | 10 days | 66–70% | [35] |
| Medicinal Plant | Possible Mechanisms | Effect | Adverse Effects | References |
|---|---|---|---|---|
| Korean red ginseng | Inhibition of H. pylori-induced 5-lipoxygenase (5-LOX) activity; preventing pro-inflammatory interleukin (IL)-8 or 5-LOX mRNA | Anti-inflammatory effect; increase eradication rates of H. pylori; reduction of gastric inflammation and oxidative DNA damage | Interaction with conventional drugs | [69,70] |
| Allium sativum | Inhibition of lipoprotein oxidation and lower serum glucose induction of antioxidant enzymes; mechanisms need to be more investigated | Antioxidant; suppressive effect of H. pylori-induced gastric inflammation in vivo and in vitro | Interaction with conventional drugs | [71] |
| Curcuma loga | Inhibition of H. pylori-induced 5-LOX activity | Anti-inflammatory; antioxidant | Not determined | [72] |
| Zingiber officinalis | Inhibition of PGE2 and parietal cell H+, K+-ATPase | Anti-inflammatory effect; antioxidant | Nausea and vomiting in pregnant women; restless, heartburn; interaction with conventional drugs (anticoagulants, analgesics) | [73,74,75] |
| Zingiber zerumbet | Gastroprotective mechanism of zerumbone (significant increased in the endogenous antioxidant GSH, reduction of lipid peroxidation level); other mechanism need to be investigated | Antioxidant, antiproliferative, anti-inflammatory, antisecretory effect; reduction of ulcer area formation | Nausea and vomiting in pregnant women; restless, heartburn; interaction with conventional drugs (anticoagulants, analgesics) | [75,76] |
| Camellia sinensis (Green tea polyphenols) | Suppression of tumor necrosis factor-alpha (TNF-α) gene expression; inhibition of urease | Antioxidant; improvement in the function of intestinal bacterial flora | Interaction with conventional drugs; dizziness, diarrhea, headaches, insomnia, heartbeat, may cause deficiency of iron | [77,78] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuna, L.; Jakab, J.; Smolic, R.; Raguz-Lucic, N.; Vcev, A.; Smolic, M. Peptic Ulcer Disease: A Brief Review of Conventional Therapy and Herbal Treatment Options. J. Clin. Med. 2019, 8, 179. https://doi.org/10.3390/jcm8020179
Kuna L, Jakab J, Smolic R, Raguz-Lucic N, Vcev A, Smolic M. Peptic Ulcer Disease: A Brief Review of Conventional Therapy and Herbal Treatment Options. Journal of Clinical Medicine. 2019; 8(2):179. https://doi.org/10.3390/jcm8020179
Chicago/Turabian StyleKuna, Lucija, Jelena Jakab, Robert Smolic, Nikola Raguz-Lucic, Aleksandar Vcev, and Martina Smolic. 2019. "Peptic Ulcer Disease: A Brief Review of Conventional Therapy and Herbal Treatment Options" Journal of Clinical Medicine 8, no. 2: 179. https://doi.org/10.3390/jcm8020179
APA StyleKuna, L., Jakab, J., Smolic, R., Raguz-Lucic, N., Vcev, A., & Smolic, M. (2019). Peptic Ulcer Disease: A Brief Review of Conventional Therapy and Herbal Treatment Options. Journal of Clinical Medicine, 8(2), 179. https://doi.org/10.3390/jcm8020179




