The Role of Sex in Memory Function: Considerations and Recommendations in the Context of Exercise
Abstract
1. Overview
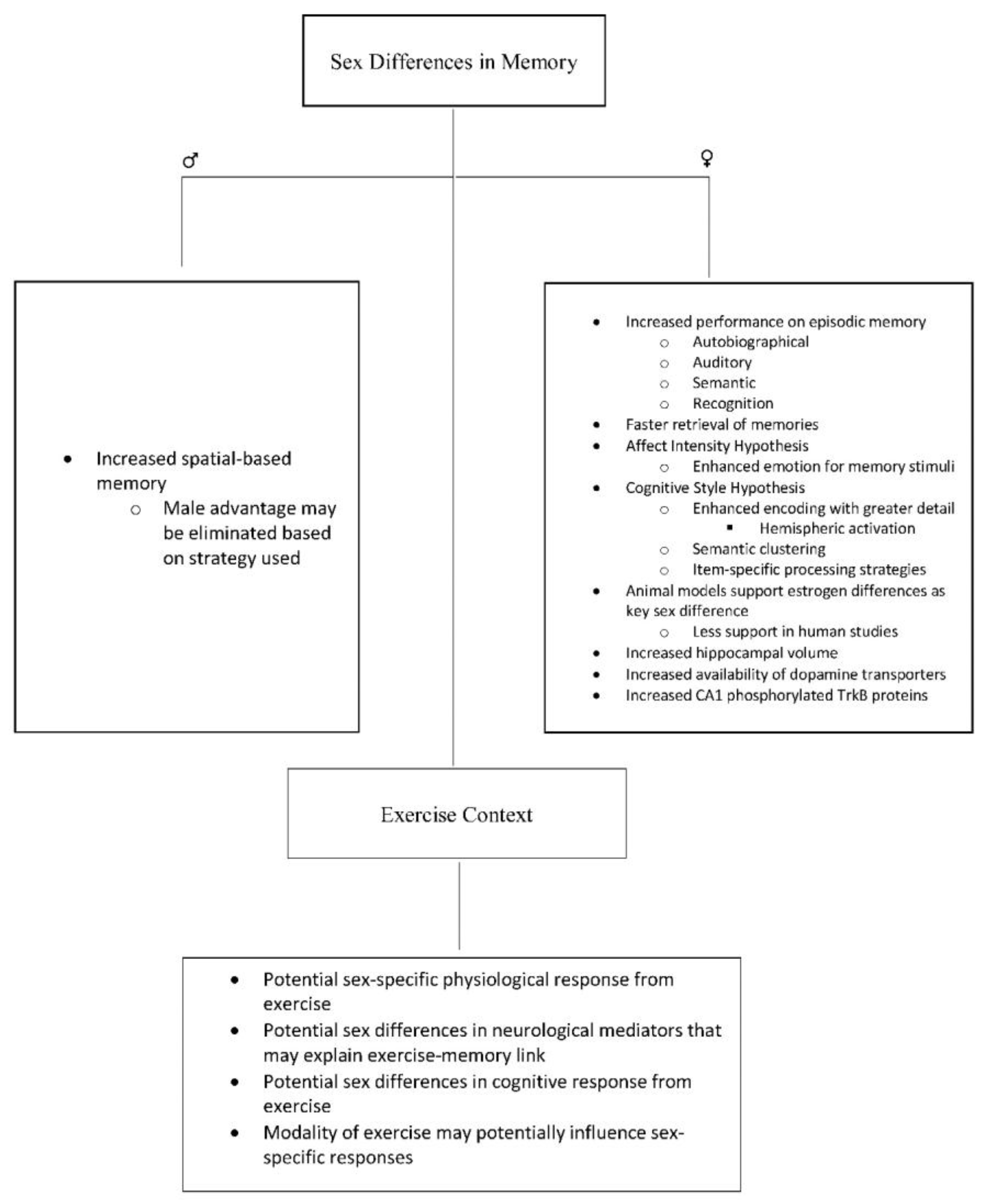
2. Sex Differences in Memory
3. Potential Psychological Explanations for Sex Differences in Memory
4. Potential Biological Explanations for Sex Differences in Memory
5. Sex Differences in Memory—Considerations for Exercise Research
Author Contributions
Conflicts of Interest
References
- Gale, S.D.; Baxter, L.; Thompson, J. Greater memory impairment in dementing females than males relative to sex-matched healthy controls. J. Clin. Exp. Neuropsychol. 2016, 38, 527–533. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, D.J.; Refsum, H.; Nurk, E.; Warden, D.R.; Tell, G.S.; Vollset, S.E.; Engedal, K.; Nygaard, H.A.; Smith, A.D. Apolipoprotein E epsilon4 and impaired episodic memory in community-dwelling elderly people: A marked sex difference. The Hordaland Health Study. J. Neurol. Neurosurg. Psychiatry 2006, 77, 902–908. [Google Scholar] [CrossRef] [PubMed]
- Hua, X.; Hibar, D.P.; Lee, S.; Toga, A.W.; Jack, C.R., Jr.; Weiner, M.W.; Thompson, P.M.; Alzheimer’s disease neuroimaging initiative. Sex and age differences in atrophic rates: An ADNI study with n = 1368 MRI scans. Neurobiol. Aging 2010, 31, 1463–1480. [Google Scholar] [CrossRef] [PubMed]
- Lundervold, A.J.; Wollschlager, D.; Wehling, E. Age and sex related changes in episodic memory function in middle aged and older adults. Scand. J. Psychol. 2014, 55, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Sunderaraman, P.; Blumen, H.M.; DeMatteo, D.; Apa, Z.L.; Cosentino, S. Task demand influences relationships among sex, clustering strategy, and recall: 16-word versus 9-word list learning tests. Cogn. Behav. Neurol. Off. J. Soc. Behav. Cogn. Neurol. 2013, 26, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Herlitz, A.; Nilsson, L.G.; Backman, L. Gender differences in episodic memory. Mem. Cogn. 1997, 25, 801–811. [Google Scholar] [CrossRef]
- Ferreira, L.; Ferreira Santos-Galduroz, R.; Ferri, C.P.; Fernandes Galduroz, J.C. Rate of cognitive decline in relation to sex after 60 years-of-age: A systematic review. Geriatr. Gerontol. Int. 2014, 14, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Maitland, S.B.; Herlitz, A.; Nyberg, L.; Backman, L.; Nilsson, L.G. Selective sex differences in declarative memory. Mem. Cogn. 2004, 32, 1160–1169. [Google Scholar] [CrossRef]
- Hyde, J.S. Sex and cognition: Gender and cognitive functions. Curr. Opin. Neurobiol. 2016, 38, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Herlitz, A.; Yonker, J.E. Sex differences in episodic memory: The influence of intelligence. J. Clin. Exp. Neuropsychol. 2002, 24, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Davis, P.J. Gender differences in autobiographical memory for childhood emotional experiences. J. Personal. Soc. Psychol. 1999, 76, 498–510. [Google Scholar] [CrossRef]
- Skowronski, J.J.; Betz, A.L.; Thompson, C.P.; Shannon, L. Social memory in everyday life: Recall of self-evenets and other-events. J. Pers. Soc. Psychol. 1991, 60, 831–843. [Google Scholar] [CrossRef]
- Fuentes, A.; Desrocher, M. The effects of gender on the retrieval of episodic and semantic components of autobiographical memory. Memory 2013, 21, 619–632. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Hou, Y.; Tang, H.; Wiprovnick, A. Travelling backwards and forwards in time: Culture and gender in the episodic specificity of past and future events. Memory 2011, 19, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.A.; Wahlin, A.; Maitland, S.B.; Hultsch, D.F.; Hertzog, C.; Backman, L. Episodic memory change in late adulthood: Generalizability across samples and performance indices. Mem. Cogn. 2004, 32, 768–778. [Google Scholar] [CrossRef]
- Pauls, F.; Petermann, F.; Lepach, A.C. Gender differences in episodic memory and visual working memory including the effects of age. Memory 2013, 21, 857–874. [Google Scholar] [CrossRef] [PubMed]
- Heisz, J.J.; Pottruff, M.M.; Shore, D.I. Females scan more than males: A potential mechanism for sex differences in recognition memory. Psychol. Sci. 2013, 24, 1157–1163. [Google Scholar] [CrossRef] [PubMed]
- Megreya, A.M.; Bindemann, M.; Havard, C. Sex differences in unfamiliar face identification: Evidence from matching tasks. Acta Psychol. 2011, 137, 83–89. [Google Scholar] [CrossRef] [PubMed]
- Lewin, C.; Herlitz, A. Sex differences in face recognition—Women’s faces make the difference. Brain Cogn. 2002, 50, 121–128. [Google Scholar] [CrossRef]
- Rehnman, J.; Herlitz, A. Women remember more faces than men do. Acta Psychol. (Amst) 2007, 124, 344–355. [Google Scholar] [CrossRef] [PubMed]
- Bothwell, R.K.; Brigham, J.C.; Malpass, R.S. Cross-racial identification. Personal. Soc. Psychol. Bull. 1989, 15, 19–25. [Google Scholar] [CrossRef]
- Andreano, J.M.; Cahill, L. Sex influences on the neurobiology of learning and memory. Learn. Mem. 2009, 16, 248–266. [Google Scholar] [CrossRef] [PubMed]
- Suchan, B.; Botko, R.; Gizewski, E.; Forsting, M.; Daum, I. Neural substrates of manipulation in visuospatial working memory. Neuroscience 2006, 139, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Maguire, E.A.; Burgess, N.; O’Keefe, J. Human spatial navigation: Cognitive maps, sexual dimorphism, and neural substrates. Curr. Opin. Neurobiol. 1999, 9, 171–177. [Google Scholar] [CrossRef]
- Postma, A.; Jager, G.; Kessels, R.P.; Koppeschaar, H.P.; van Honk, J. Sex differences for selective forms of spatial memory. Brain Cogn. 2004, 54, 24–34. [Google Scholar] [CrossRef]
- Saucier, D.M.; Green, S.M.; Leason, J.; MacFadden, A.; Bell, S.; Elias, L.J. Are sex differences in navigation caused by sexually dimorphic strategies or by differences in the ability to use the strategies? Behav. Neurosci. 2002, 116, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Silverman, I. Sexual dimorphism in spatial behaviors: Applications to route learning. Evol. Cogn. 1996, 2, 165–171. [Google Scholar]
- James, T.W.; Kimura, D. Sex differences in remembering the locations of objects in an array: Location-shifts versus location-exchanges. Evol. Hum. Behav. 1997, 18, 155–163. [Google Scholar] [CrossRef]
- Fujita, F.; Diener, E.; Sandvik, E. Gender differences in negative affect and well-being: The case for emotional intensity. J. Personal. Soc. Psychol. 1991, 61, 427–434. [Google Scholar] [CrossRef]
- Richards, J.M.; Gross, J.J. Emotion regulation and memory: The cognitive costs of keeping one’s cool. J. Personal. Soc. Psychol. 2000, 79, 410–424. [Google Scholar] [CrossRef]
- Bloise, S.M.; Johnson, M.K. Memory for emotional and neutral information: Gender and individual differences in emotional sensitivity. Memory 2007, 15, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Cahill, L.; Haier, R.J.; White, N.S.; Fallon, J.; Kilpatrick, L.; Lawrence, C.; Potkin, S.G.; Alkire, M.T. Sex-related difference in amygdala activity during emotionally influenced memory storage. Neurobiol. Learn. Mem. 2001, 75, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Seidlitz, L.; Diener, E. Sex differences in the recall of affective experiences. J. Personal. Soc. Psychol. 1998, 74, 262–271. [Google Scholar] [CrossRef]
- Cahill, L.; Uncapher, M.; Kilpatrick, L.; Alkire, M.T.; Turner, J. Sex-related hemispheric lateralization of amygdala function in emotionally influenced memory: An FMRI investigation. Learn. Mem. 2004, 11, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Fink, G.R.; Halligan, P.W.; Marshall, J.C.; Frith, C.D.; Frackowiak, R.S.; Dolan, R.J. Where in the brain does visual attention select the forest and the trees? Nature 1996, 382, 626–628. [Google Scholar] [CrossRef] [PubMed]
- Fink, G.R.; Marshall, J.C.; Halligan, P.W.; Dolan, R.J. Hemispheric asymmetries in global/local processing are modulated by perceptual salience. Neuropsychologia 1999, 37, 31–40. [Google Scholar] [CrossRef]
- Schulkind, M.; Schoppel, K.; Scheiderer, E. Gender differences in autobiographical narratives: He shoots and scores; she evaluates and interprets. Mem. Cogn. 2012, 40, 958–965. [Google Scholar] [CrossRef] [PubMed]
- Fivush, R.; Berlin, L.J.; Sales, J.M.; Mennuti-Washburn, J.; Cassidy, J. Functions of parent-child reminiscing about emotionally negative events. Memory 2003, 11, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Krueger, L.E.; Salthouse, T.A. Differences in acquisition, not retention, largely contribute to sex differences in multitrial word recall performance. Pers. Individ. Dif. 2010, 49, 768–772. [Google Scholar] [CrossRef] [PubMed]
- Kramer, J.H.; Delis, D.C.; Daniel, M. Sex differences in verbal learning. J. Clin. Psychol. 1988, 44, 907–915. [Google Scholar] [CrossRef]
- Berenbaum, S.A.; Baxter, L.; Seidenberg, M.; Hermann, B. Role of the hippocampus in sex differences in verbal memory: Memory outcome following left anterior temporal lobectomy. Neuropsychology 1997, 11, 585–591. [Google Scholar] [CrossRef] [PubMed]
- Mulligan, N.W.; Lozito, J.P. Self-generation and memory. Psychol. Learn. Motiv. 2004, 45, 175–214. [Google Scholar]
- Kimura, D. Sex differences in cerebral organization for speech and praxic functions. Can. J. Psychol. 1983, 37, 19–35. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Xue, G.; Dong, Q.; Jin, Z.; Li, T.; Xue, F.; Zhao, L.; Guo, Y. Sex determines the neurofunctional predictors of visual word learning. Neuropsychologia 2007, 45, 741–747. [Google Scholar] [CrossRef] [PubMed]
- McGlone, J. Sex differences in functional brain asymmetry. Cortex 1978, 14, 122–128. [Google Scholar] [CrossRef]
- Trenerry, M.R.; Jack, C.R., Jr.; Cascino, G.D.; Sharbrough, F.W.; Ivnik, R.J. Gender differences in post-temporal lobectomy verbal memory and relationships between MRI hippocampal volumes and preoperative verbal memory. Epilepsy Res. 1995, 20, 69–76. [Google Scholar] [CrossRef]
- Jacobs, D.M.; Tang, M.X.; Stern, Y.; Sano, M.; Marder, K.; Bell, K.L.; Schofield, P.; Dooneief, G.; Gurland, B.; Mayeux, R. Cognitive function in nondemented older women who took estrogen after menopause. Neurology 1998, 50, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Boss, L.; Kang, D.H.; Marcus, M.; Bergstrom, N. Endogenous sex hormones and cognitive function in older adults: A systematic review. West. J. Nurs. Res. 2014, 36, 388–426. [Google Scholar] [CrossRef] [PubMed]
- Haut, K.M.; Barch, D.M. Sex influences on material-sensitive functional lateralization in working and episodic memory: Men and women are not all that different. NeuroImage 2006, 32, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Piefke, M.; Weiss, P.H.; Markowitsch, H.J.; Fink, G.R. Gender differences in the functional neuroanatomy of emotional episodic autobiographical memory. Hum. Brain Mapp. 2005, 24, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Nyberg, L.; Habib, R.; Herlitz, A. Brain activation during episodic memory retrieval: Sex differences. Acta Psychol. 2000, 105, 181–194. [Google Scholar] [CrossRef]
- Ragland, J.D.; Coleman, A.R.; Gur, R.C.; Glahn, D.C.; Gur, R.E. Sex differences in brain-behavior relationships between verbal episodic memory and resting regional cerebral blood flow. Neuropsychologia 2000, 38, 451–461. [Google Scholar] [CrossRef]
- Berger, J.; Oltmanns, F.; Holtkamp, M.; Bengner, T. Sex differences in verbal and nonverbal learning before and after temporal lobe epilepsy surgery. Epilepsy Behav. E B 2017, 66, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Rentz, D.M.; Weiss, B.K.; Jacobs, E.G.; Cherkerzian, S.; Klibanski, A.; Remington, A.; Aizley, H.; Goldstein, J.M. Sex differences in episodic memory in early midlife: Impact of reproductive aging. Menopause 2017, 24, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Henderson, V.W.; Guthrie, J.R.; Dudley, E.C.; Burger, H.G.; Dennerstein, L. Estrogen exposures and memory at midlife: A population-based study of women. Neurology 2003, 60, 1369–1371. [Google Scholar] [CrossRef] [PubMed]
- Henderson, V.W.; Popat, R.A. Effects of endogenous and exogenous estrogen exposures in midlife and late-life women on episodic memory and executive functions. Neuroscience 2011, 191, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Yonker, J.E.; Eriksson, E.; Nilsson, L.G.; Herlitz, A. Sex differences in episodic memory: Minimal influence of estradiol. Brain Cogn. 2003, 52, 231–238. [Google Scholar] [CrossRef]
- Kramer, J.H.; Delis, D.C.; Kaplan, E.; O’Donnell, L.; Prifitera, A. Developmental sex differences in verbal learning. Neuropsychology 1997, 11, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Woolley, C.S.; Gould, E.; Frankfurt, M.; McEwen, B.S. Naturally occurring fluctuation in dendritic spine density on adult hippocampal pyramidal neurons. J. Neurosci. Off. J. Soc. Neurosci. 1990, 10, 4035–4039. [Google Scholar] [CrossRef]
- Woolley, C.S.; McEwen, B.S. Roles of estradiol and progesterone in regulation of hippocampal dendritic spine density during the estrous cycle in the rat. J. Comp. Neurol. 1993, 336, 293–306. [Google Scholar] [CrossRef] [PubMed]
- Woolley, C.S.; McEwen, B.S. Estradiol regulates hippocampal dendritic spine density via an N-methyl-D-aspartate receptor-dependent mechanism. J. Neurosci. Off. J. Soc. Neurosci. 1994, 14, 7680–7687. [Google Scholar] [CrossRef]
- Dumitriu, D.; Rapp, P.R.; McEwen, B.S.; Morrison, J.H. Estrogen and the aging brain: An elixir for the weary cortical network. Ann. N. Y. Acad. Sci. 2010, 1204, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y.; Park, C.S.; Janssen, W.G.; Roberts, M.T.; Morrison, J.H.; Rapp, P.R. Synaptic correlates of memory and menopause in the hippocampal dentate gyrus in rhesus monkeys. Neurobiol. Aging 2012, 33, 421.e17. [Google Scholar] [CrossRef] [PubMed]
- Fink, G.; Sumner, B.E.; Rosie, R.; Grace, O.; Quinn, J.P. Estrogen control of central neurotransmission: Effect on mood, mental state, and memory. Cell. Mol. Neurobiol. 1996, 16, 325–344. [Google Scholar] [CrossRef] [PubMed]
- Naftolin, F.; Leranth, C.; Perez, J.; Garcia-Segura, L.M. Estrogen induces synaptic plasticity in adult primate neurons. Neuroendocrinology 1994, 57, 935–939. [Google Scholar] [CrossRef] [PubMed]
- Brinton, R.D.; Tran, J.; Proffitt, P.; Montoya, M. 17 beta-Estradiol enhances the outgrowth and survival of neocortical neurons in culture. Neurochem. Res. 1997, 22, 1339–1351. [Google Scholar] [CrossRef] [PubMed]
- Filipek, P.A.; Richelme, C.; Kennedy, D.N.; Caviness, V.S., Jr. The young adult human brain: An MRI-based morphometric analysis. Cereb. Cortex 1994, 4, 344–360. [Google Scholar] [CrossRef] [PubMed]
- Paus, T.; Otaky, N.; Caramanos, Z.; MacDonald, D.; Zijdenbos, A.; D’Avirro, D.; Gutmans, D.; Holmes, C.; Tomaiuolo, F.; Evans, A.C. In vivo morphometry of the intrasulcal gray matter in the human cingulate, paracingulate, and superior-rostral sulci: Hemispheric asymmetries, gender differences and probability maps. J. Comp. Neurol. 1996, 376, 664–673. [Google Scholar] [CrossRef]
- Schlaepfer, T.E.; Harris, G.J.; Tien, A.Y.; Peng, L.; Lee, S.; Pearlson, G.D. Structural differences in the cerebral cortex of healthy female and male subjects: A magnetic resonance imaging study. Psychiatry Res. 1995, 61, 129–135. [Google Scholar] [CrossRef]
- Witelson, S.F.; Glezer, I.I.; Kigar, D.L. Women have greater density of neurons in posterior temporal cortex. J. Neurosci. Off. J. Soc. Neurosci. 1995, 15, 3418–3428. [Google Scholar] [CrossRef]
- Giedd, J.N.; Vaituzis, A.C.; Hamburger, S.D.; Lange, N.; Rajapakse, J.C.; Kaysen, D.; Vauss, Y.C.; Rapoport, J.L. Quantitative MRI of the temporal lobe, amygdala, and hippocampus in normal human development: Ages 4–18 years. J. Comp. Neurol. 1996, 366, 223–230. [Google Scholar] [CrossRef]
- Lavalaye, J.; Booij, J.; Reneman, L.; Habraken, J.B.; van Royen, E.A. Effect of age and gender on dopamine transporter imaging with [123I]FP-CIT SPET in healthy volunteers. Eur. J. Nucl. Med. 2000, 27, 867–869. [Google Scholar] [CrossRef] [PubMed]
- Laakso, A.; Vilkman, H.; Bergman, J.; Haaparanta, M.; Solin, O.; Syvälahti, E.; Salokangas, R.K.; Hietala, J. Sex differences in striatal presynaptic dopamine synthesis capacity in healthy subjects. Biol. Psychiatry 2002, 52, 759–763. [Google Scholar] [CrossRef]
- Park, H.; Poo, M.M. Neurotrophin regulation of neural circuit development and function. Nat. Rev. Neurosci. 2013, 14, 7–23. [Google Scholar] [CrossRef] [PubMed]
- Bakos, J.; Hlavacova, N.; Rajman, M.; Ondicova, K.; Koros, C.; Kitraki, E.; Steinbusch, H.W.; Jezova, D. Enriched environment influences hormonal status and hippocampal brain derived neurotrophic factor in a sex dependent manner. Neuroscience 2009, 164, 788–797. [Google Scholar] [CrossRef] [PubMed]
- Bland, S.T.; Schmid, M.J.; Der-Avakian, A.; Watkins, L.R.; Spencer, R.L.; Maier, S.F. Expression of c-fos and BDNF mRNA in subregions of the prefrontal cortex of male and female rats after acute uncontrollable stress. Brain Res. 2005, 1051, 90–99. [Google Scholar] [CrossRef] [PubMed]
- Hayley, S.; Du, L.; Litteljohn, D.; Palkovits, M.; Faludi, G.; Merali, Z.; Poulter, M.O.; Anisman, H. Gender and brain regions specific differences in brain derived neurotrophic factor protein levels of depressed individuals who died through suicide. Neurosci. Lett. 2015, 600, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Zhu, S.W.; Yee, B.K.; Nyffeler, M.; Winblad, B.; Feldon, J.; Mohammed, A.H. Influence of differential housing on emotional behaviour and neurotrophin levels in mice. Behav. Brain Res. 2006, 169, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Spencer-Segal, J.L.; Waters, E.M.; Bath, K.G.; Chao, M.V.; McEwen, B.S.; Milner, T.A. Distribution of phosphorylated TrkB receptor in the mouse hippocampal formation depends on sex and estrous cycle stage. J. Neurosci. Off. J. Soc. Neurosci. 2011, 31, 6780–6790. [Google Scholar] [CrossRef] [PubMed]
- Honack, D.; Loscher, W. Sex differences in NMDA receptor mediated responses in rats. Brain Res. 1993, 620, 167–170. [Google Scholar] [CrossRef]
- Qi, X.; Zhang, K.; Xu, T.; Yamaki, V.N.; Wei, Z.; Huang, M.; Rose, G.M.; Cai, X. Sex differences in long-term potentiation at Temporoammonic-CA1 synapses: Potential implications for memory consolidation. PLoS ONE 2016, 11, e0165891. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Edwards, M.K.; Frith, E. Potential avenues for exercise to activate episodic memory-related pathways: A narrative review. Eur. J. Neurosci. 2017, 46, 2067–2077. [Google Scholar] [CrossRef] [PubMed]
- Crush, E.A.; Loprinzi, P.D. Dose-Response Effects of exercise duration and recovery on cognitive functioning. Percept. Mot. Skills 2017. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Voss, M.W.; Prakash, R.S.; Basak, C.; Szabo, A.; Chaddock, L.; Kim, J.S.; Heo, S.; Alves, H.; White, S.M.; et al. Exercise training increases size of hippocampus and improves memory. Proc. Natl. Acad. Sci. USA 2011, 108, 3017–3022. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Frith, E.; Edwards, M.K.; Sng, E.; Ashpole, N. The effects of exercise on memory function among young to middle-aged adults: Systematic review and recommendations for future research. Am. J. Health Promot. AJHP 2017. [Google Scholar] [CrossRef] [PubMed]
- Frith, E.; Sng, E.; Loprinzi, P.D. Randomized controlled trial evaluating the temporal effects of high-intensity exercise on learning, short-term and long-term memory, and prospective memory. Eur. J. Neurosci. 2017, 46, 2557–2564. [Google Scholar] [CrossRef] [PubMed]
- Sng, E.; Frith, E.; Loprinzi, P.D. Temporal effects of acute walking exercise on learning and memory function. Am. J. Health Promot. AJHP 2017. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Kane, C.J. Exercise and cognitive function: A randomized controlled trial examining acute exercise and free-living physical activity and sedentary effects. Mayo Clin. Proc. 2015, 90, 450–460. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Edwards, M.K. Exercise and implicit memory: A brief systematic review. Psychol. Rep. 2017. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.K.; Loprinzi, P.D. Experimental effects of acute exercise and meditation on parameters of cognitive function. J. Clin. Med. 2018, 7, 125. [Google Scholar] [CrossRef] [PubMed]
- Roig, M.; Thomas, R.; Mang, C.S.; Snow, N.J.; Ostadan, F.; Boyd, L.A.; Lundbye-Jensen, J. Time-dependent effects of cardiovascular exercise on memory. Exerc. Sport Sci. Rev. 2016, 44, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Labban, J.D.; Etnier, J.L. Effects of acute exercise on long-term memory. Res. Q. Exerc. Sport. 2011, 82, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T. Developing the catecholamines hypothesis for the acute exercise-cognition interaction in humans: Lessons from animal studies. Phys. Behav. 2016, 165, 291–299. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T.; Turner, A.; Hale, B.J.; Sproule, J. Beyond the catecholamines hypothesis for an acute exercise-cognition interaction: A neurochemical perspective. In Exercise-Cognition Interaction: Neuroscience Perspectives; McMorris, T., Ed.; Academic Press: New York, NY, USA, 2016; pp. 65–103. [Google Scholar]
- McMorris, T.; Sproule, J.; Turner, A.; Hale, B.J. Acute, intermediate intensity exercise, and speed and accuracy in working memory tasks: A meta-analytical comparison of effects. Phys. Behav. 2011, 102, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Roig, M.; Nordbrandt, S.; Geertsen, S.S.; Nielsen, J.B. The effects of cardiovascular exercise on human memory: A review with meta-analysis. Neurosci. Biobehav. Rev. 2013, 37, 1645–1666. [Google Scholar] [CrossRef] [PubMed]
- Josefsson, M.; de Luna, X.; Pudas, S.; Nilsson, L.G.; Nyberg, L. Genetic and lifestyle predictors of 15-year longitudinal change in episodic memory. J. Am. Geriatr. Soc. 2012, 60, 2308–2312. [Google Scholar] [CrossRef] [PubMed]
- Sandstrom, N.J.; Kaufman, J.; Huettel, S.A. Males and females use different distal cues in a virtual environment navigation task. Brain Res. Cogn. Brain Res. 1998, 6, 351–360. [Google Scholar] [CrossRef]
- Vargha-Khadem, F.; Gadian, D.G.; Watkins, K.E.; Connelly, A.; Van Paesschen, W.; Mishkin, M. Differential effects of early hippocampal pathology on episodic and semantic memory. Science 1997, 277, 376–380. [Google Scholar] [CrossRef] [PubMed]
- Gron, G.; Wunderlich, A.P.; Spitzer, M.; Tomczak, R.; Riepe, M.W. Brain activation during human navigation: Gender-different neural networks as substrate of performance. Nat. Neurosci. 2000, 3, 404–408. [Google Scholar] [CrossRef] [PubMed]
- Bauer, P.J.; Stennes, L.; Haight, J.C. Representation of the inner self in autobiography: Women’s and men’s use of internal states language in personal narratives. Memory 2003, 11, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Barha, C.K.; Davis, J.C.; Falck, R.S.; Nagamatsu, L.S.; Liu-Ambrose, T. Sex differences in exercise efficacy to improve cognition: A systematic review and meta-analysis of randomized controlled trials in older humans. Front. Neuroendocrinol. 2017, 46, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Sheel, A.W. Sex differences in the physiology of exercise: An integrative perspective. Exp. Physiol. 2016, 101, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.B.; Ye, K. Sex differences in brain-derived neurotrophic factor signaling and functions. J. Neurosci. Res. 2017, 95, 328–335. [Google Scholar] [CrossRef] [PubMed]
- Barha, C.K.; Falck, R.S.; Davis, J.C.; Nagamatsu, L.S.; Liu-Ambrose, T. Sex differences in aerobic exercise efficacy to improve cognition: A systematic review and meta-analysis of studies in older rodents. Front Neuroendocr. 2017, 46, 86–105. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loprinzi, P.D.; Frith, E. The Role of Sex in Memory Function: Considerations and Recommendations in the Context of Exercise. J. Clin. Med. 2018, 7, 132. https://doi.org/10.3390/jcm7060132
Loprinzi PD, Frith E. The Role of Sex in Memory Function: Considerations and Recommendations in the Context of Exercise. Journal of Clinical Medicine. 2018; 7(6):132. https://doi.org/10.3390/jcm7060132
Chicago/Turabian StyleLoprinzi, Paul D., and Emily Frith. 2018. "The Role of Sex in Memory Function: Considerations and Recommendations in the Context of Exercise" Journal of Clinical Medicine 7, no. 6: 132. https://doi.org/10.3390/jcm7060132
APA StyleLoprinzi, P. D., & Frith, E. (2018). The Role of Sex in Memory Function: Considerations and Recommendations in the Context of Exercise. Journal of Clinical Medicine, 7(6), 132. https://doi.org/10.3390/jcm7060132




