The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial
Abstract
1. Introduction
2. Experimental Section
2.1. Participants/Study Population
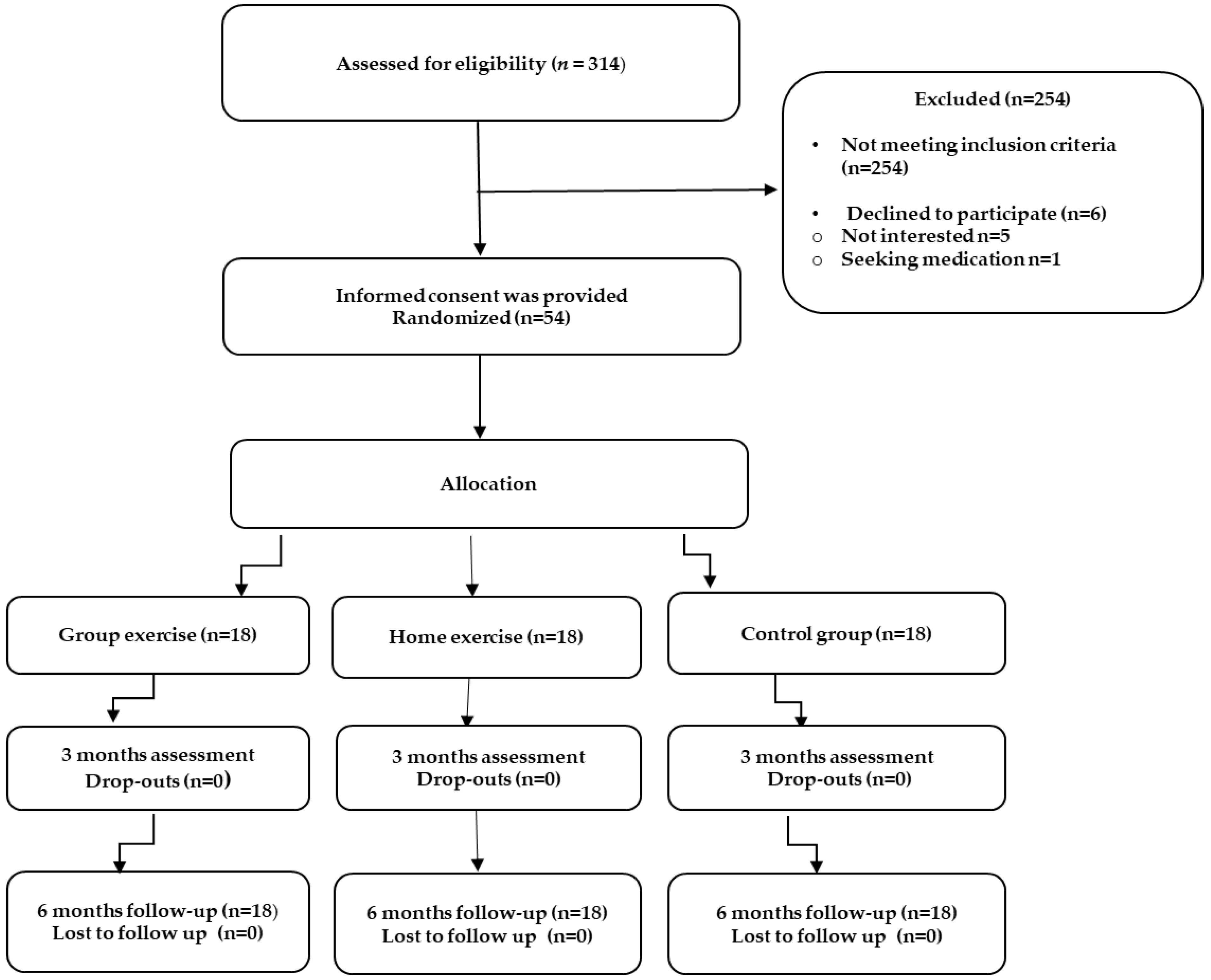
2.2. Randomized Group Assignment
2.3. Outcome Measures
2.3.1. Interview Survey
2.3.2. Body Composition Assessment
2.3.3. Muscular Strength Assessments
2.3.4. Functional Assessments
2.3.5. Quality of Life Assessment
2.4. Interventions
3. Statistical Analysis
4. Results
4.1. Within Group Differences
4.2. Group Differences
5. Discussion
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Cruz-Jentoft, A.J.; Landi, F.; Schneider, S.M.; Zúñiga, C.; Arai, H.; Boirie, Y.; Chen, L.K.; Fielding, R.A.; Martin, F.C.; Michel, J.P.; et al. Prevalence of and interventions for sarcopenia in ageing adults: A systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing 2014, 43, 748–759. [Google Scholar] [CrossRef] [PubMed]
- Zembroń-Łacny, A.; Dziubek, W.; Rogowski, Ł.; Skorupka, E.; Dąbrowska, G. Sarcopenia: Monitoring, molecular mechanisms, and physical intervention. Physiol. Res. 2014, 63, 683–691. [Google Scholar] [PubMed]
- Tsekoura, M.; Gliatis, J.; Billis, E. Sarcopenia. Literature update. Arch. Hell. Med. 2017, 34, 42–48. [Google Scholar]
- Beaudart, C.; Rizzoli, R.; Bruyère, O.; Reginster, J.Y.; Biver, E. Sarcopenia: Burden and challenges for public health. Arch. Public Health 2014, 72, 45. [Google Scholar] [CrossRef] [PubMed]
- Locquet, M.; Beaudart, C.; Reginster, J.Y.; Petermans, J.; Bruyère, O. Comparison of the performance of five screening methods for sarcopenia. Clin. Epidemiol. 2017, 2018, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Tsekoura, M.; Kastrinis, A.; Katsoulaki, M.; Billis, E.; Gliatis, J. Sarcopenia and Its Impact on Quality of Life. Adv. Exp. Med. Biol. 2017, 987, 213–218. [Google Scholar] [PubMed]
- Yu, S.C.Y.; Khow, K.S.F.; Jadszak, A.D.; Visvanathan, R. Clinical screening tools for sarcopenia and its management. Curr. Gerontol. Geriatr. Res. 2016, 2016, 5978523. [Google Scholar] [CrossRef] [PubMed]
- Martine, A.M.; Lattanzio, F.; Abbatecola, A.M.; Carpia, D.L.; Tosato, M.; Marzetti, E.; Calvani, R.; Onder, G.; Landi, F. Treating sarcopenia in older and oldest old. Curr. Pharm. Des. 2015, 21, 1715–1722. [Google Scholar]
- Kim, H.K.; Suzuki, T.; Saito, K.; Yoshida, H.; Kobayashi, H.; Kato, H.; Katayama, M. Effects of exercise and amino acid supplementation on body composition and physical function in community-dwelling elderly Japanese sarcopenic women: A randomized controlled trial. J. Am. Geriatr. Soc. 2012, 60, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Suzuki, T.; Saito, K.; Yoshida, H.; Kojima, N.; Kim, M.; Sudo, M.; Yamashiro, Y.; Tokimitsu, I. Effects of exercise and tea catechins on muscle mass, strength and walking ability in community-dwelling elderly Japanese sarcopenic women: A randomized controlled trial. Geriatr. Gerontol. Int. 2013, 13, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kim, M.; Kojima, N.; Fujino, K.; Hosoi, E.; Kobayashi, H.; Somekawa, S.; Niki, Y.; Yamashiro, Y.; Yoshida, H. Exercise and nutritional supplementation on community-dwelling elderly Japanese women with sarcopenic obesity: A randomized controlled trial. J. Am. Med. Dir. Assoc. 2016, 17, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.D.; Tsauo, J.Y.; Lin, L.F.; Huang, S.W.; Ku, J.W.; Chou, L.C.; Liou, T.H. Effects of elastic resistance exercise on body composition and physical capacity in older women with sarcopenic obesity: A CONSORT-compliant prospective randomized controlled trial. Medicine 2017, 96, e7115. [Google Scholar] [CrossRef] [PubMed]
- Balachandran, A.; Krawczyk, S.N.; Potiaumpai, M.; Signorile, J.F. High-speed circuit training vs hypertrophy training to improve physical function in sarcopenic obese adults: A randomized controlled trial. Exp. Gerontol. 2014, 60, 64–67. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J.; Latham, N.K. Progressive resistance strength training for improving physical function in older adults. Cochrane Database Syst. Rev. 2009, 3, CD002759. [Google Scholar] [CrossRef] [PubMed]
- Wei, N.; Shamay, S.M.; Gabriel, Y.F.; Ruby, S.Y.L.; Mary, C.K.L.; Marco, Y.C.P. Whole-body vibration training improves muscle and physical performance in community dwelling with sarcopenia: A randomized controlled trial. Int. J. Phys. Ther. Rehabil. 2016, 2, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Zdzieblik, D.; Oesser, S.; Baumstark, M.W.; Gollhofer, A.; König, D. Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: A randomised controlled trial. Br. J. Nutr. 2015, 114, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Montoya, I.L.; Correa-Perez, A.; Abraha, I.; Soiza, R.L.; Cherubin, A.; O’Mahony, D.; Crus-Jentoft, A.J. Nopharmacological interventions to treat physical frailty and sarcopenia in older patients: A systematic overview-the SENATOR Project ONTOP series. Clin. Interv. Aging 2017, 12, 721–740. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, Y.; Wakabayashi, H.; Yamada, M.; Kim, H.; Harada, A.; Arai, H. Interventions for Treating Sarcopenia: A Systematic Review and Meta-Analysis of Randomized Controlled Studies. Interventions for Treating Sarcopenia: A Systematic Review and Meta Analysis of Randomized Controlled Studies. J. Am. Med. Dir. Assoc. 2017, 18, 553.e1–553.e16. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Dawson, A.; Shaw, S.C.; Harvey, N.C.; Kanis, J.A.; Binkley, N.; Reginster, J.Y.; Chapurlat, R.; Chan, D.C.; Bruyère, O.; et al. Sarcopenia Working Group. Nutrition and physical activity in the prevention and treatment of sarcopenia: Systematic review. Osteoporos. Int. 2017, 28, 1817–1833. [Google Scholar] [CrossRef] [PubMed]
- El-Khoury, F.; Cassou, B.; Charles, M.A.; Dargent-Molina, P. The effect of fall prevention exercise programmes on fall induced injuries in community dwelling older adults: Systematic review and meta-analysis of randomised controlled trials. BMJ 2013, 347, f6234. [Google Scholar] [PubMed]
- Youssef, E.F.; Shanb, A.A. Supervised Versus Home Exercise Training Programs on Functional Balance in Older Subjects. Malays. J. Med. Sci. 2016, 23, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, N.L.; Chad, K.E.; Harrison, E.L.; Reeder, B.A.; Marshall, S.C. Home versus center based physical activity programs in older adults. Cochrane Database Syst. Rev. 2005, 25, CD004017. [Google Scholar] [CrossRef] [PubMed]
- Middleton, L.A.; Black, S.E.; Herrmann, N.; Oh, P.I.; Regan, K.; Lanctot, K.L. Centre- versus home-based exercise among people with mci and mild dementia: Study protocol for a randomized parallel-group trial. BMC Geriatr. 2018, 18, 27. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Suresh, K.P. An overview of randomization techniques: An unbiased assessment of outcome in clinical research. J. Hum. Reprod. Sci. 2011, 4, 8–11. [Google Scholar] [CrossRef] [PubMed]
- Iannuzzi-Sucich, M.; Prestwood, K.M.; Kenny, A.M. Prevalence of sarcopenia and predictors of skeletal muscle mass in healthy, older men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, M772–M777. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; Mc Hugh, P.R. Mini Mental State: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Bahat, G.; Tufan, A.; Tufan, F.; Kilic, S.; Akpinar, T.S.; Kose, M.; Erten, N.; Karan, M.A.; Cruz-Jentoft, A.J. Cut-off points to identify sarcopenia according to European Working Group on Sarcopenia in Older People (EWGSOP) definition. Clin. Nutr. 2016, 35, 1557–1563. [Google Scholar] [CrossRef] [PubMed]
- Tsekoura, M.; Billis, E.; Gliatis, J.; Tsepis, E.; Matzaroglou, C.; Sakkas, G.K.; Beaudart, C.; Bruyere, O.; Tyllianakis, M.; Panagiotopoulos, E. Cross cultural adaptation of the Greek Sarcopenia Quality of Life (SarQoL®) questionnaire. Disabil. Rehabil. 2018. accepted. [Google Scholar] [CrossRef] [PubMed]
- Pagotto, V.; Ferreira dos Santo, K.; Malaquias, S.G.; Bachion, M.M.; Silveira, E.E. Calf circumference: Clinical validation for evaluation of muscle mass in the elderly. Rev. Bras. Enferm. 2018, 71, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, R.; Murakami, H.; Sanada, K.; Tanaka, N.; Sawada, S.S.; Tabata, I.; Higuchi, M.; Miyachi, M. Calf circumference as a surrogate marker of muscle mass for diagnosing sarcopenia in Japanese men and women. Geriatr. Gerontol. Int. 2015, 15, 969–976. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, M.; Lee, Y.; Kim, N.; Yoon, T.Y.; Won, C.W. Calf Circumference as a Simple Screening Marker for Diagnosing Sarcopenia in Older Korean Adults: The Korean Frailty and Aging Cohort Study (KFACS). J. Korean Med. Sci. 2018, 33, e151. [Google Scholar] [CrossRef] [PubMed]
- Fox, B.; Henwood, T.; Schaap, L.; Bruyère, O.; Reginster, J.Y.; Beaudart, C.; Buckinx, F.; Roberts, H.; Cooper, C.; Cherubini, A.; et al. Adherence to a standardized protocol for measuring grip strength and appropriate cut-off values in adults over 65 years with sarcopenia: A systematic review protocol. JBI Database Syst. Rev. Implement. Rep. 2015, 13, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Gadelha, A.B.; Neri, S.G.R.; Oliveira, R.J.; Bottaro, M.; David, A.C.; Vainshelboim, B.; Lima, R.M. Severity of sarcopenia is associated with postural balance and risk of falls in community-dwelling older women. Exp. Aging Res. 2018, 44, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Guralnik, J.M.; Simonsicj, E.M.; Ferucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. Med. Sci. 1994, 49, M85–M94. [Google Scholar] [CrossRef]
- Beaudart, C.; Biver, E.; Reginster, J.Y.; Rizzoli, R.; Rolland, Y.; Bautmans, I.; Petermans, J.; Gillain, S.; Buckinx, F.; Van Beveren, J.; et al. Development of a self-administered quality of life questionnaire for sarcopenia in elderly subjects: The SarQoL. Age Ageing 2015, 44, 960–966. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, C. Rating of Perceived Exertion (RPE). J. Physiother. 2012, 58, 62. [Google Scholar] [CrossRef]
- Borg, G. Borg’s Perceived Exertion and Pain Scales; Human Kinetics: Champaign, IL, USA, 1998; pp. 47–77. [Google Scholar]
- Jansons, P.; Robins, L.; O’Brien, L.; Haines, T. Gym-based exercise was more costly compared with home-based exercise with telephone support when used as maintenance programs for adults with chronic health conditions: Cost-effectiveness analysis of a randomised trial. J. Physiother. 2018, 64, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, S.; Takamiya, T.; Inoue, S. Group exercise for adults and elderly: Determinants of participation in group exercise and its associations with health outcome. J. Phys. Fit. Sports Med. 2015, 4, 315–320. [Google Scholar] [CrossRef]
- Munro, J.F.; Nicholl, J.P.; Brazier, J.E.; Davey, R.; Cochrane, T. Cost effectiveness of a community based exercise programme in over 65 year olds: Cluster randomised trial. J. Epidemiol. Community Health 2004, 58, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M.; Hayes, A.; Mc Creesh, K.; Purtill, H.; O’Sullivan, K. Are group-based and individual physiotherapy exercise programmes equally effective for musculoskeletal conditions? A systematic review and meta-analysis. Br. J. Sports Med. 2017, 51, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2018. Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Rubenstein, L.Z.; Josephson, K.R.; Trueblood, P.R.; Loy, S.; Harker, J.O.; Pietruszka, F.M.; Robbins, A.S. Effects of a Group Exercise Program on Strength, Mobility, and Falls Among Fall-Prone Elderly Men. J. Gerontol. A Biol. Sci. Med. Sci. 2000, 55, M317–M321. [Google Scholar] [CrossRef] [PubMed]
- Maruya, K.; Asakawa, Y.; Ishibashi, H.; Fujita, H.; Arai, T.; Yamaguchi, H. Effect of a simple and adherent home exercise program on the physical function of community dwelling adults sixty years of age and older with pre-sarcopenia or sarcopenia. J. Phys. Ther. Sci. 2016, 28, 3183–3188. [Google Scholar] [CrossRef] [PubMed]
- Eakin, E.; Reeves, M.; Lawler, S.; Graves, N.; Oldenburg, B.; Del Mar, C.; Wilke, K.; Winkler, E.; Barnett, A. Telephone counseling for physical activity and diet in primary care patients. Am. J. Prev. Med. 2009, 36, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Farrance, C.; Tsofliou, F.; Clark, C. Adherence to community based group exercise interventions for older people: A mixed-methods systematic review. Prev. Med. 2016, 87, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, H.; Yagasaki, K.; Saito, Y.; Oguma, Y. Regular group exercise contributes to balanced health in older adults in Japan: A qualitative study. BMC Geriatr. 2017, 17, 190. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Biver, E.; Reginster, J.Y.; Rizzoli, R.; Rolland, Y.; Bautmans, I.; Petermans, J.; Gillain, S.; Buckinx, F.; Dardenne, N.; et al. Validation of the SarQoL®, a specific health-related quality of life questionnaire for Sarcopenia. J. Cachexia Sarcopenia Muscle 2017, 8, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Gasparik, A.I.; Mihai, G.; Beaudart, C.; Bruyere, O.; Pop, R.M.; Reginster, J.Y.; Pascanu, I.M. Psychometric performance of the Romanian version of the SarQoL, a health-related quality of life questionnaire for sarcopenia. Arch. Osteoporos. 2017, 12, 103. [Google Scholar] [CrossRef] [PubMed]
- Geerinck, A.; Scheppers, A.; Beaudart, C.; Bruyère, O.; Vandenbussche, W.; René Bautman, R.; Delye, S.; Bautmans, I. Translation and validation of the Dutch SarQoL®, a quality of life questionnaire specific to sarcopenia. J. Musculoskelet. Neuronal Interact. 2018, in press. [Google Scholar]
- Konstantynowicz, J.; Abramowicz, P.; Glinkowski, W.; Taranta, E.; Marcinowicz, L.; Dymitrowicz, M.; Reginster, J.-Y.; Bruyere, O.; Beaudart, C. Polish Validation of the SarQoL®, a Quality of Life Questionnaire Specific to Sarcopenia. J. Clin. Med. 2018, 7, 323. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Edwards, M.; Moss, C.; Reginster, J.Y.; Moon, R.; Parsons, C.; Demoulin, C.; Rizzoli, R.; Biver, E.; Dennison, E.; et al. English translation and validation of the SarQoL, a quality of life questionnaire specific for sarcopenia. Age Ageing 2017, 46, 271–276. [Google Scholar] [CrossRef] [PubMed]
- Beaudart, C.; Reginster, J.Y.; Geerinck, A.; MédéaLocquet, M.; Bruyère, O. Current review of the SarQoL®: A health-related quality of life questionnaire specific to sarcopenia. Expert Rev. Pharmacoeconom. Outcomes Res. 2017, 17, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Rejeski, W.J.; Mihalko, S.L. Physical activity and quality of life in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Nourhashémi, F.; Andrieu, S.; Gillette-Guyonnet, S.; Vellas, B.; Albarède, J.L.; Grandjean, H. Instrumental Activities of Daily Living as a Potential Marker of Frailty: A Study of 7364 Community-Dwelling Elderly Women (the EPIDOS Study). J. Gerontol. A Biol. Sci. Med. Sci. 2001, 25, M448–M453. [Google Scholar] [CrossRef]
- Millán-Calenti, J.C.; Tubío, J.; Pita-Fernández, S.; González-Abraldes, I.; Lorenzo, T.; Fernández-Arruty, T.; Maseda, A. Prevalence of functional disability in activities of daily living (ADL), instrumental activities of daily living (IADL) and associated factors, as predictors of morbidity and mortality. Arch. Gerontol. Geriatr. 2010, 50, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Rubio, E.; Lázaro, A.; Sánchez-Sánchez, A. Social participation and independence in activities of daily living: A cross sectional study. BMC Geriatr. 2009, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Iolascon, G.; Di Pietro, G.; Gimigliano, F.; Mauro, G.L.; Moretti, A.; Giamattei, M.T.; Ortolani, S.; Tarantino, U.; Brandi, M.L. Physical exercise and sarcopenia in older people: Position paper of the Italian Society of Orthopaedics and Medicine (OrtoMed). Clin. Cases Miner. Bone Metab. 2014, 11, 215–221. [Google Scholar] [CrossRef] [PubMed]
- Ziaaldini, M.M.; Marzetti, E.; Picca, A.; Murlasits, Z. Biochemical pathways of sarcopenia and their modulation by physical exercise: A narrative review. Front. Med. 2017, 4, 167. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.N.; Hardman, A.E. Walking to health. Sports Med. 1997, 23, 306–332. [Google Scholar] [CrossRef] [PubMed]
- Graves, J.E.; Pollock, M.L.; Montain, S.J.; Jackson, A.S.; O Keefe, J.M. The effects of hand held weights on the physiological responses to walking exercise. Med. Sci. Sports Exerc. 1987, 19, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidou, E.; Apostolidis, A.; Kondelidis, N.; Tsimtsiou, Z.; Hatzichristou, D.; Ioannides, E. Short-term efficacy of group pelvic floor training under intensive supervision versus unsupervised home training for female stress urinary incontinence: A randomized pilot study. Neurourol. Urodyn. 2007, 26, 486–491. [Google Scholar] [CrossRef] [PubMed]
- Dadgostar, H.; Firouzinezhad, S.; Ansari, M.; Younespour, S.; Mahmoudpour, A.; Khamseh, M.E. Supervised group-exercise therapy versus home-based exercise therapy: Their effects on Quality of Life and cardiovascular risk factors in women with type 2 diabetes. Diabetes Metab. Syndr. 2016, 10, S30–S36. [Google Scholar] [CrossRef] [PubMed]
- Shumway-Cook, A.; Woollacott, M.H. Motor Control: Theory and Practical Applications; Williams & Wilkins: Baltimore, MD, USA, 1995; pp. 99–114. [Google Scholar]
- Basharat, A.; Naeem, A.; Basharat, Z.; Qamar, M.M. A systematic review on the effectiveness of exercise in sarcopenia. J. Public Health Biol. Sci. 2012, 1, 61–67. [Google Scholar]
- Bauman, A.; Merom, D.; Bull, F.C.; Buchner, D.M.; Fiatarone Singh, M.A. Updating the Evidence for Physical Activity: Summative Reviews of the Epidemiological Evidence. Gerontologist 2016, 56, S268–S280. [Google Scholar] [CrossRef] [PubMed]
- Van Stralen, M.; De Vries, H.; Mudde, A.N.; Bolman, C.; Lechner, L. Determinants of initiation and maintenance of physical activity among older adults: A literature review. Health Psychol. Rev. 2009, 3, 147–207. [Google Scholar] [CrossRef]
- Mehra, S.; Dadema, T.; Krose, B.J.A.; Visser, B.; Engelbert, R.H.H.; Van den Helder, J.; Weijs, P.J.M. Attitudes of older dults in a Group- Based exercise program toward a blended intervention; a focus-group study. Front. Psychol. 2016, 7, 1827. [Google Scholar] [CrossRef] [PubMed]
- Freene, N.; Waddington, G.; Chesworth, W.; Davey, R.; Goss, J. Physical activity at home (PAAH)’, evaluation of a group versus home based physical activity program in community dwelling middle aged adults: Rationale and study design. BMC Public Health 2011, 11, 883. [Google Scholar] [CrossRef] [PubMed]
- Orbell, S.; Johnston, M.; Rowley, D.; Davey, P.; Espley, A. Self efficacy and goal importance in the prediction of physical disability in people following hospitalization: A prospective study. Br. J. Health Psychol. 2001, 6, 25–40. [Google Scholar] [CrossRef] [PubMed]
- Picorelli, A.M.; Pereira, L.S.; Pereira, D.S.; Felício, D.; Sherrington, C. Adherence to exercise programs for older people is influenced by program characteristics and personal factors: A systematic review. J. Physiother. 2014, 60, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Steffl, M.; Bohannon, R.W.; Petr, M.; Kohlikova, E.; Holmerova, I. Relation between cigarette smoking and sarcopenia: Meta-analysis. Physiol. Res. 2015, 64, 419–426. [Google Scholar] [PubMed]
- Buckinx, F.; Landi, F.; Cesari, M.; Fielding, R.A.; Fielding, R.A.; Engelke, K.; Maggi, S.; Dennison, E.; Al-Daghri, N.M.; Allepaerts, S.; et al. Pitfalls in the measurement of muscle mass: A need for a reference standard. J. Cachexia Sarcopenia Muscle 2018, 9, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.C.; Heymsfield, S.B. Bioelectrical impedance analysis for diagnosing sarcopenia and cachexia: What are we really estimating? J. Cachexia Sarcopenia Muscle 2017, 8, 187–189. [Google Scholar] [CrossRef] [PubMed]
| Warm up | Resistance | Balance | Cool down | |
|---|---|---|---|---|
| Phase 1 (weeks 1–4) | Activity: seated marching, forward walk and turn, semi-tandem walk, circle walking, sit to stand, double side arm raise, neck flexion, neck rotation, ankle—four-way Duration: 5 min | Activity: knee extensor-flexor, hip abductor-extension, ankle plantar flexors- dorsi flexors, wall push up, seated bicep curl, seated triceps extension, seated lateral shoulder raises, seated abdominal crunches Duration: 15 min Reps: 8 × 1 set Intensity: Easy (10–11 points in the Borg RPE) | Activity: walking and turning around, walking- backward- tandem-heel-toe- heel to toe-sideways, one leg stand, tandem stance Duration: 15 min Reps: 8 × 1 set Intensity: Easy (10–11 points in the Borg RPE) | Activity: forward walk/march, stretch-neck/shoulder/chest/rhomboid/, /tricep/lats/oblique, /hamstring, /gastrocnemius Duration: 5 min |
| Phase 2 (weeks 5–8) | Αctivity: As in phase 1 Duration: 5 min | Activity: As in phase 1 Added sit to stand exercise Duration: 20 min Reps: 10 × 2 sets Intensity: Easy (10–12 points in the Borg RPE) | Activity: As in phase 1 Added walking backward Duration: 20 min Reps: 10 × 2 sets Intensity: Easy (10–12 points in the Borg RPE) | Activity: As in phase 1 Duration: 5–10 min |
| Phase 3 (weeks 9–12) | Activity: As in phase 2 Duration: 5–10 min | Activity: As in phase 2 Duration: 20 min Reps: 12 × 2 sets Intensity: Medium to hard (12 points in the Borg RPE) | Activity: As in phase 2 Duration: 20 min Reps: 12 × 2 sets Intensity: Medium to hard (12 points in the Borg RPE) | Activity: As in phase 2 Duration: 5–10 min |
| Comorbidities | Group-Based | Home-Based | Control Group | Total | p-Value |
|---|---|---|---|---|---|
| (n = 18) | (n = 18) | (n = 18) | (n = 54) | ||
| mean ± SD | |||||
| Age | 74.56 ± 6.04 | 71.17 ± 6.47 | 72.89 ± 8.31 | 72.87 ± 7.02 | 0.35 |
| Body mass index (BMI) (kg/m2) | 21.95 ± 2.18 | 23.24 ± 2.78 | 22.98 ± 2.29 | 22.72 ± 2.45 | 0.25 |
| Drugs (n) | 3.55 ± 1.24 | 3.33 ± 1.37 | 2.77 ± 1.66 | 3.22 ± 1.44 | 0.25 |
| Comorbidities | 2.66 ± 1.08 | 2.5 ± 0.92 | 2.83 ± 1.2 | 2.66 ± 1.06 | 0.65 |
| Quality of life (SarQol_GR score) | 57.08 ± 13.24 | 56.51 ± 10.64 | 53.21 ± 13.15 | 56.6 ± 12.28 | 0.6 |
| Handgrip strength | 17.22 ± 4.8 | 19.2 ± 3.6 | 17.43 ± 4.04 | 17.95 ± 4.2 | 0.304 |
| Gait speed | 0.87 ± 0.13 | 0.9 ± 0.10 | 0.91 ± 0.12 ± | 0.89 ± 0.11 | 0.63 |
| number (percentage) | |||||
| Gender | 0.87 | ||||
| Female | 16 (89%) | 15 (84%) | 16 (89%) | 47 (87.3%) | |
| Male | 2 (11%) | 3 (16%) | 2 (11%) | 7 (12.7%) | |
| Smoking | 2 (11%) | 3 (16%) | 8 (44%) | 13 (24%) | 0.04 |
| Educational status | |||||
| Elementary | 3 (16.7%) | 2 (11.1%) | 2 (11.1%) | 7 (12.7%) | |
| High school | 11 (61.1%) | 8 (44.4%) | 11 (61.1%) | 30 (54.55) | 0.11 |
| University | 4 (22.2%) | 7 (38.9%) | 5 (27.8%) | 16 (29.1%) | |
| Marital status | |||||
| Married | 11 (61.1%) | 11 (61.1%) | 9 (50%) | 31 (56.4%) | |
| Widowed | 6 (33.3%) | 3 (16.7%) | 7 (38.9%) | 16 (29.1%) | 0.56 |
| Divorced | 0 (0%) | 2 (11.1%) | 0 (0%) | 2 (3.6%) | |
| Single | 1 (5.65) | 2 (11.1%) | 2 (11.1%) | 5 (9.1%) | |
| Falls | 9 (50%) | 7 (38.9%) | 9 (50%) | 25 (45.5%) | 0.75 |
| Variables | Total Sample * | Group | Baseline | After 3 Months Interventions | 6 Months Follow-Up | ANOVA (GxT) | |
|---|---|---|---|---|---|---|---|
| (n = 54) | p Value | ||||||
| BMI (kg/m2) | 22.72 ± 2.45 | GE | 21.95 ± 2.18 | 22.19 ± 2.12 | 22.08 ± 1.98 | 0.58 | |
| HE | 23.24 ± 2.78 | 23.44 ± 2.43 | 23.33 ± 2.29 | ||||
| C | 22.72 ± 2.45 | 22.95 ± 2.08 | 22.64 ± 2.02 | ||||
| SMMI | 5.64 ± 0.56 | GE | 5.7 ± 0.49 | 5.94 ± 0.51 | 5.86 ± 0.46 | 0.003 ** | |
| HE | 5.64 ± 0.55 | 5.71 ± 0.54 | 5.69 ± 0.5 | ||||
| C | 5.58 ± 0.66 | 5.57 ± 0.65 | 5.54 ± 0.65 | ||||
| Fat free mass | 25.05 ± 4.26 | GE | 25.59 ± 4.4 | 26.5 ± 4.4 | 25.95 ± 4.68 | 0.45 | |
| HE | 24.41 ± 4.9 | 24.73 ± 4.98 | 24.38 ± 3.1 | ||||
| C | 25.15 ± 3.52 | 24.57 ± 4.66 | 26.45 ± 10.46 | ||||
| Calf circumference (cm) | 32.76 ± 2.23 | GE | 33.17 ± 2.47 | 33.61 ± 2.32 | 33.72 ± 2.34 | <0.001 ** | |
| HE | 32.67 ± 2.52 | 32.76 ± 2.58 | 32.7 ± 2.51 | ||||
| C | 32.44 ± 1.65 | 32.38 ± 1.71 | 32.27 ± 1.84 | ||||
| TUG (sec) | 10.06 ± 1.98 | GE | 10.04 ± 2.68 | 8.27 ± 2.07 | 8.61 ± 2.48 | <0.001 ** | |
| HE | 9.67 ± 1.29 | 8.47 ± 1.22 | 8.82 ± 1.37 | ||||
| C | 10.48 ± 1.75 | 10.61 ± 1.67 | 10.52 ± 1.8 | ||||
| 4 m test (s) | 4.52 ± 0.65 | GE | 4.66 ± 0.84 | 3.34 ± 0.49 | 3.17 ± 0.75 | <0.001 ** | |
| HE | 4.47 ± 0.53 | 3.75 ± 0.54 | 3.71 ± 0.70 | ||||
| C | 4.42 ± 0.53 | 4.42 ± 0.69 | 4.64 ± 0.76 | ||||
| Gait speed (m/s2) | 0.89 ± 0.11 | GE | 0.87 ± 0.13 | 1.21 ± 0.15 | 1.32 ± 0.3 | 0.45 | |
| HE | 0.9 ± 0.10 | 1.08 ± 0.15 | 1.11 ± 0.2 | ||||
| C | 0.91 ± 0.12 | 0.92 ± 0.15 | 0.87 ± 0.15 | ||||
| Chair stand test (s) | 13.59 ± 3.52 | GE | 13.72 ± 5.07 | 10.78 ± 3.94 | 11.15 ± 4.82 | <0.001 ** | |
| HE | 12.29 ± 1.54 | 10.9 ± 1.98 | 11.41 ± 2.17 | ||||
| C | 14.76 ± 2.72 | 14.38 ± 3.19 | 14.61 ± 3.22 | ||||
| Handgrip strength (kg) | 17.95 ± 4.2 | GE | 17.22 ± 4.8 | 20.58 ± 4.29 | 20.07 ± 4.35 | <0.001 ** | |
| HE | 19.2 ± 3.6 | 19.33 ± 4.51 | 18.68 ± 3.34 | ||||
| C | 17.43 ± 4.04 | 17.92 ± 4.14 | 17.34 ± 3.77 | ||||
| Isokinetic measurements | Right knee ext 90° (Nm/kg) | 36.46 ± 9.1 | GE | 45.21 ± 12.87 | 51.69 ± 12.87 | 50.09 ± 11.94 | 0.003 * |
| HE | 47.73 ± 9.75 | 51.61 ± 12.66 | 50.48 ± 11.49 | ||||
| C | 44.48 ± 12.38 | 43.15 ± 10.76 | 41.88 ± 10.23 | ||||
| Right knee | 36.46 ± 9.1 | GE | 34.61 ± 10.06 | 39.87 ± 12.03 | 39.03 ± 10.51 | <0.001 ** | |
| ext 180° (Nm/kg) | HE | 38.31 ± 7.89 | 39.94 ± 11.81 | 37.69 ± 9.18 | |||
| C | 36.47 ± 9.35 | 33.63 ± 8.97 | 33.35 ± 7.7 | ||||
| Right knee | 30.74 ± 10.1 | GE | 30.15 ± 10.76 | 38.57 ± 7.52 | 38.25 ± 7.58 | 0.002 * | |
| flex 90° (Nm/kg) | HE | 31.67 ± 8.62 | 34.59 ± 10.12 | 35.05 ± 8.87 | |||
| C | 30.4 ± 11.48 | 27.15 ± 10.43 | 26.75 ± 10.36 | ||||
| Right knee | 26.92 ± 7.96 | GE | 27.38 ± 6.84 | 34.18 ± 7.1 | 32.62 ± 6.72 | <0.001 ** | |
| flex 180° (Nm/kg) | HE | 27.99 ± 7.6 | 29.6 ± 7.94 | 29.08 ± 7.29 | |||
| C | 25.38 ± 9.43 | 23.69 ± 9 | 25.53 ± 9.48 | ||||
| Left knee ext 90° (Nm/kg) | 43.06 ± 10.66 | GE | 45.59 ± 10.9 | 50.4 ± 10.91 | 49.66 ± 10.03 | 0.03 * | |
| HE | 44.51 ± 7.59 | 47.6 ± 10.1 | 47.24 ± 10.6 | ||||
| C | 39.07 ± 12.34 | 39.67 ± 11.1 | 36.43 ± 10.03 | ||||
| Left knee ext 180° (Nm/kg) | 34.4 ± 8.05 | GE | 34.86 ± 8.75 | 38.71 ± 8.88 | 39.03 ± 10.51 | 0.17 | |
| HE | 36.29 ± 6.79 | 38.93 ± 5.71 | 37.69 ± 9.18 | ||||
| C | 32.05 ± 8.33 | 31.17 ± 8.56 | 33.35 ± 7.7 | ||||
| Left knee flex 90° (Nm/kg) | 29.54 ± 9.25 | GE | 28.94 ± 9.74 | 36.26 ± 10.26 | 34.5 ± 8.6 | <0.001 ** | |
| HE | 31.67 ± 8.62 | 33.51 ± 7.81 | 32.4 ± 7.82 | ||||
| C | 28.06 ± 9.51 | 29.09 ± 9.47 | 26.14 ± 10.44 | ||||
| Left knee flex 180° (Nm/kg) | 23.58 ± 7.78 | GE | 26.95 ± 7.69 | 31.39 ± 8.03 | 29.17 ± 7.47 | 0.055 | |
| HE | 26.86 ± 7.5 | 31.56 ± 7.23 | 28.71 ± 7.29 | ||||
| C | 23.58 ± 8.11 | 24.41 ± 8.78 | 22.29 ± 9.07 | ||||
| Quality of life (SarQoL) | 56.6 ± 12.28 | GE | 57.08 ± 13.24 | 64.37 ± 11.77 | 61.4 ± 15.73 | <0.001 ** | |
| HE | 56.51 ± 10.64 | 59.93 ± 11.28 | 58.5 ± 11.23 | ||||
| C | 53.21 ± 13.15 | 58.44 ± 12.74 | 49.22 ± 10.49 |
| Variable | Difference Means | Exercise-Based Group | Home-Based Group | Control Group | |
|---|---|---|---|---|---|
| SMMI | baseline-12wks | −0.24 * | −0.07 | 0.01 | |
| baseline-24wks | −0.16 * | −0.05 | 0.03 | ||
| BMI | baseline-12wks | −0.24 | −0.19 | 0.02 | |
| baseline-24wks | −0.13 | −0.08 | 0.34 | ||
| TUG | baseline-12wks | 1.77 * | 1.20 * | −0.13 | |
| baseline-24wks | 1.42 * | 0.84 * | −0.14 | ||
| Handgrip strength (kg) | baseline-12wks | −3.36 * | −0.13 | −0.13 | |
| baseline-24wks | −2.85 * | 0.52 | 0.52 | ||
| Gait speed | baseline-12wks | −0.33 * | −0.17 * | −0.01 * | |
| baseline-24wks | −0.44 * | −0.21 * | 0.03 * | ||
| 4 m test | baseline-12wks | 1.32 * | 0.71 * | 0 | |
| baseline-24wks | 1.49 * | 0.75 * | −0.22 | ||
| Chair stand test | baseline-12wks | 2.94 * | 1.39 * | 0.37 | |
| baseline-24wks | 2.57 * | 1.87 | 0.14 | ||
| Quality of life (SarQoL) | baseline-12wks | −7.28 * | −3.41 * | 2.19 | |
| baseline-24wks | −4.32 | −1.99 | 3.99 | ||
| Calf circumference (cm) | baseline-12wks | −0.44 * | −0.11 | 0.05 | |
| baseline-24wks | −0.55 * | −0.05 | 0.16 | ||
| Isokinetic measurements | Right knee ext 90°/s | baseline-12wks | −6.47 * | −3.88 | 1.32 |
| baseline-24wks | −4.87 * | −2.75 | 2.59 | ||
| Right knee ext 180°/s | baseline-12wks | −5.26 * | −1.62 | 2.83 | |
| baseline-24wks | −4.42 * | 0.62 | 3.11 | ||
| Right knee flex 90°/s | baseline-12wks | −8.42 * | −2.91 | 3.25 | |
| baseline-24wks | −8.09 * | −3.37 | 3.64 | ||
| Right knee flex 180°/s | baseline-12wks | −6.8 * | −4.69 * | −0.82 | |
| baseline-24wks | −1.61 | −1.84 | 1.28 | ||
| Left knee ext 90°/s | baseline-12wks | −4.80 * | −3.08 | −0.60 | |
| baseline-24wks | −4.06 | −2.73 | 2.63 | ||
| Left knee ext 180°/s | baseline-12wks | −3.85 | −2.64 | 0.87 | |
| baseline-24wks | −4.17 | −1.40 | −1.29 | ||
| Left knee flex 90°/s | baseline-12wks | −7.31 * | −1.88 | −1.03 | |
| baseline-24wks | −5.55 * | −0.77 | 1.92 | ||
| Left knee flex 180°/s | baseline-12wks | −4.44 * | −4.69 * | −0.83 | |
| baseline-24wks | −2.22 | −1.84 | 1.28 |
| Variable | ANOVA (p Value) | Post Hoc (Bonferroni) Analysis | ||
|---|---|---|---|---|
| Group-Based vs. Home-Based Exercise Group | Group-Based vs. Control | Home-Based vs. Control | ||
| Skeletal muscle mass index | ≤0.001 | ≤0.001 | ≤0.001 | NS |
| Calf circumference | ≤0.001 | ≤0.001 | ≤0.001 | NS |
| Chair stand test | ≤0.001 | ≤0.05 | ≤0.001 | NS |
| Handgrip strength | ≤0.001 | ≤0.001 | ≤0.05 | NS |
| Right knee extension 90°/s | 0.005 | NS | ≤0.05 | NS |
| Right knee extension 180°/s | ≤0.001 | NS | ≤0.001 | NS |
| Right knee flexion 90°/s | 0.004 | NS | ≤0.05 | NS |
| Right knee flexion 180°/s | ≤0.001 | ≤0.001 | ≤0.001 | NS |
| Left knee extension 180°/s | 0.01 | NS | 0.02 | NS |
| Left knee flexion 90°/s | 0.004 | 0.02 | ≤0.05 | ΝS |
| 4m test | ≤0.001 | 0.003 | ≤0.001 | ≤0.001 |
| TUG | ≤0.001 | NS | ≤0.001 | ≤0.001 |
| Gait speed | ≤0.001 | ≤0.001 | ≤0.001 | ≤0.001 |
| Quality of life (SarQoL) | ≤0.001 | NS | ≤0.001 | ≤0.05 |
| Physical and mental health (Domain 1) | ≤0.001 | NS | ≤0.05 | NS |
| Locomotion (Domain 2) | 0.014 | NS | ≤0.05 | NS |
| Functionality (Domain 4) | 0.03 | NS | ≤0.05 | NS |
| Activities of daily living (Domain 5) | ≤0.001 | ≤0.05 | ≤0.001 | ≤0.05 |
| Fears (Domain 7) | 0.07 | NS | ≤0.05 | NS |
| Variable | ANOVA (p Value) | Post Hoc (Bonferroni) Analysis | ||
|---|---|---|---|---|
| Group-Based vs. Home-Based Exercise Group | Group-Based vs. Control | Home-Based vs. Control | ||
| Skeletal muscle mass index | ≤0.001 | NS | ≤0.001 | NS |
| Calf circumference | ≤0.001 | ≤0.05 | ≤0.001 | NS |
| Chair stand test | ≤0.001 | ≤0.001 | ≤0.05 | NS |
| Handgrip strength | ≤0.001 | 0.001 | ≤0.05 | NS |
| Right knee extension 90°/s | 0.005 | NS | ≤0.05 | NS |
| Right Knee extension 180°/s | 0.005 | ≤0.05 | ≤0.01 | NS |
| Right knee flexion 90°/s | 0.003 | NS | ≤0.05 | NS |
| Right knee flexion 180°/s | 0.013 | NS | ≤0.05 | NS |
| Left knee extension 180°/s | 0.49 | NS | ≤0.001 | NS |
| Left knee flexion 90°/s | ≤0.001 | ≤0.05 | ≤0.001 | ΝS |
| 4 m test | <0.001 | 0.003 | <0.001 | <0.001 |
| TUG | ≤0.001 | NS | ≤0.001 | ≤0.05 |
| Gait speed | ≤0.001 | ≤0.05 | ≤0.001 | ≤0.001 |
| Quality of life (SarQoL) | 0.002 | NS | ≤0.05 | ≤0.05 |
| Physical and mental health (Domain 1) | 0.05 | NS | ≤0.05 | NS |
| Locomotion (Domain 2) | 0.01 | NS | ≤0.05 | NS |
| Activities of daily living (Domain 3) | 0.02 | NS | ≤0.05 | NS |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsekoura, M.; Billis, E.; Tsepis, E.; Dimitriadis, Z.; Matzaroglou, C.; Tyllianakis, M.; Panagiotopoulos, E.; Gliatis, J. The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial. J. Clin. Med. 2018, 7, 480. https://doi.org/10.3390/jcm7120480
Tsekoura M, Billis E, Tsepis E, Dimitriadis Z, Matzaroglou C, Tyllianakis M, Panagiotopoulos E, Gliatis J. The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial. Journal of Clinical Medicine. 2018; 7(12):480. https://doi.org/10.3390/jcm7120480
Chicago/Turabian StyleTsekoura, Maria, Evdokia Billis, Elias Tsepis, Zacharias Dimitriadis, Charalampos Matzaroglou, Minos Tyllianakis, Elias Panagiotopoulos, and John Gliatis. 2018. "The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial" Journal of Clinical Medicine 7, no. 12: 480. https://doi.org/10.3390/jcm7120480
APA StyleTsekoura, M., Billis, E., Tsepis, E., Dimitriadis, Z., Matzaroglou, C., Tyllianakis, M., Panagiotopoulos, E., & Gliatis, J. (2018). The Effects of Group and Home-Based Exercise Programs in Elderly with Sarcopenia: A Randomized Controlled Trial. Journal of Clinical Medicine, 7(12), 480. https://doi.org/10.3390/jcm7120480




