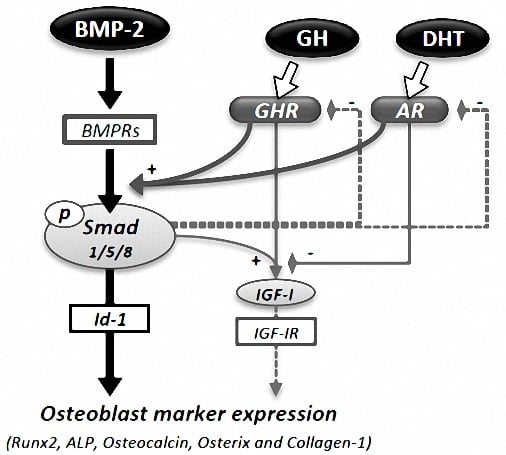
Combined Effects of Androgen and Growth Hormone on Osteoblast Marker Expression in Mouse C2C12 and MC3T3-E1 Cells Induced by Bone Morphogenetic Protein
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Supplies
2.2. Cell Culture and Morphological Examination
2.3. RNA Extraction, RT-PCR, and Quantitative Real-Time PCR Analysis
2.4. Western Immunoblot Analysis
2.5. ALP Activity Assay
2.6. Statistical Analysis
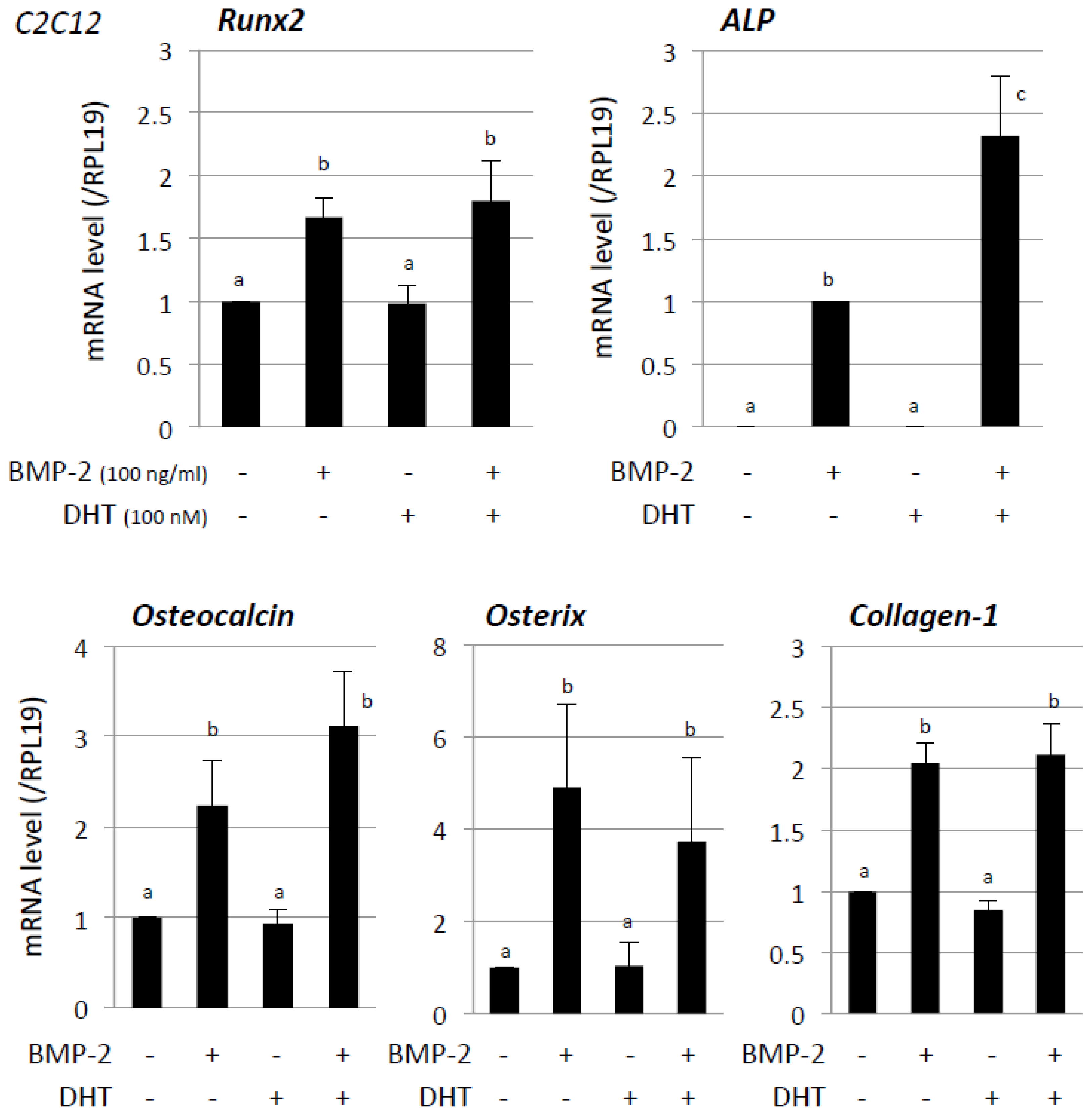
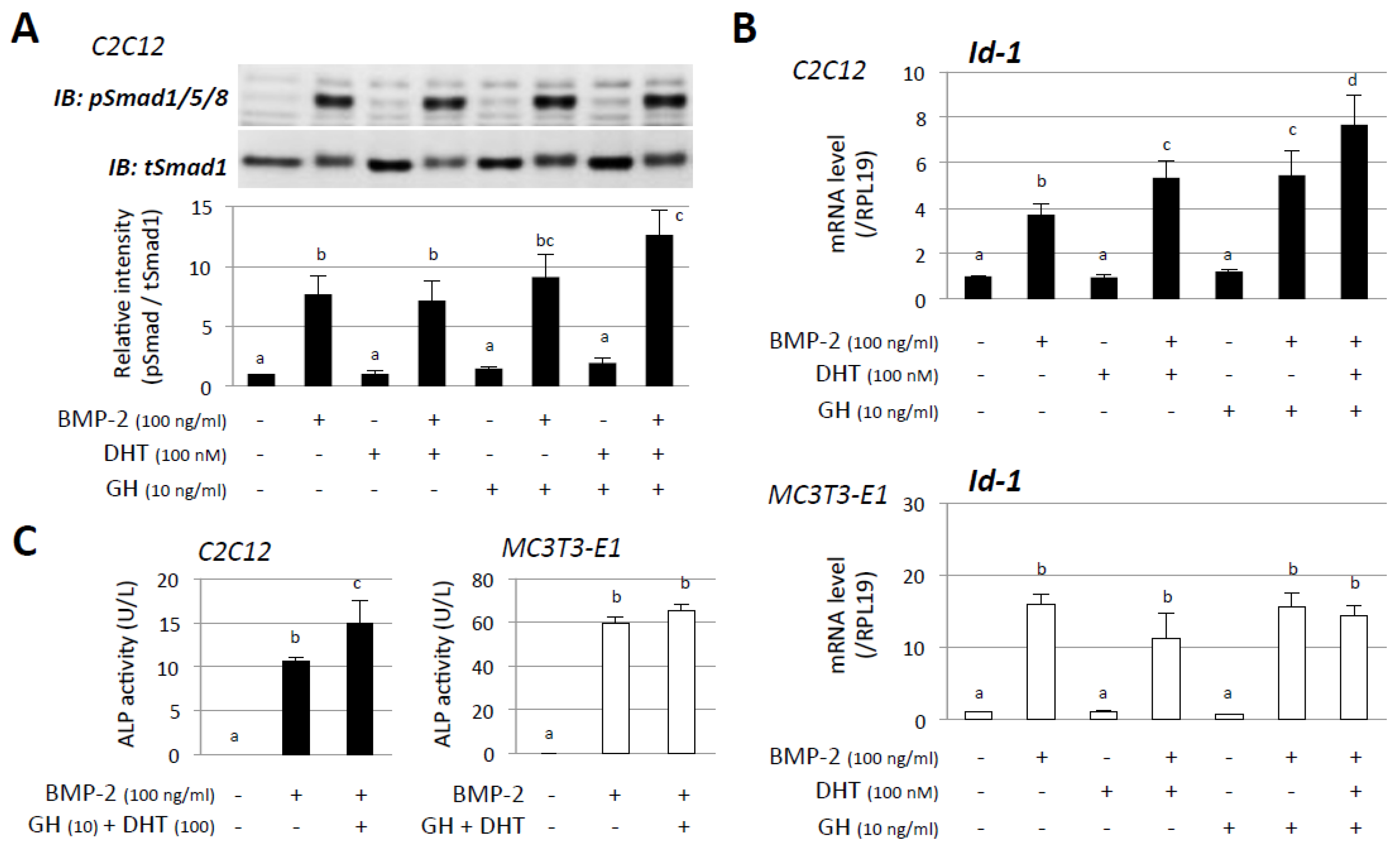
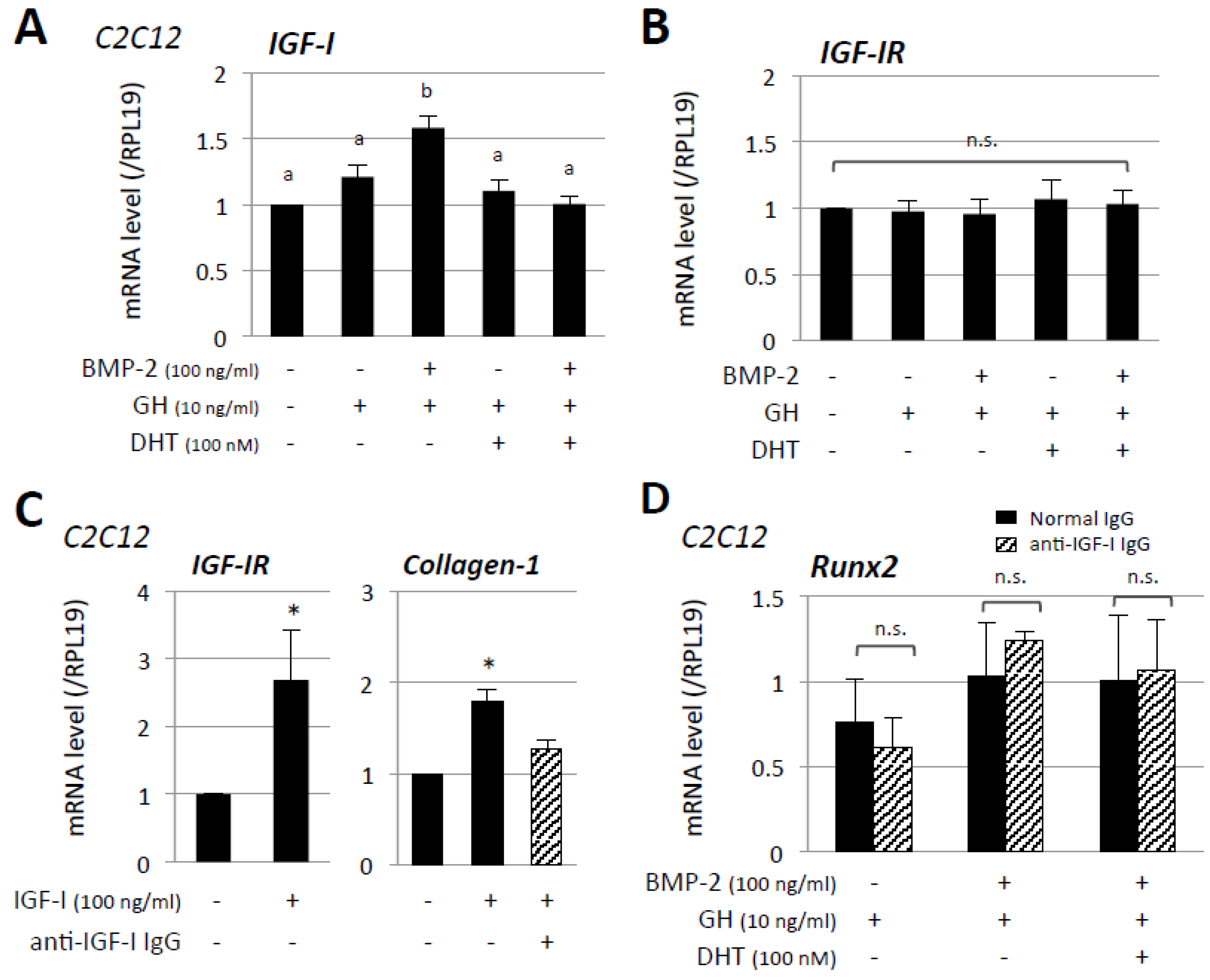
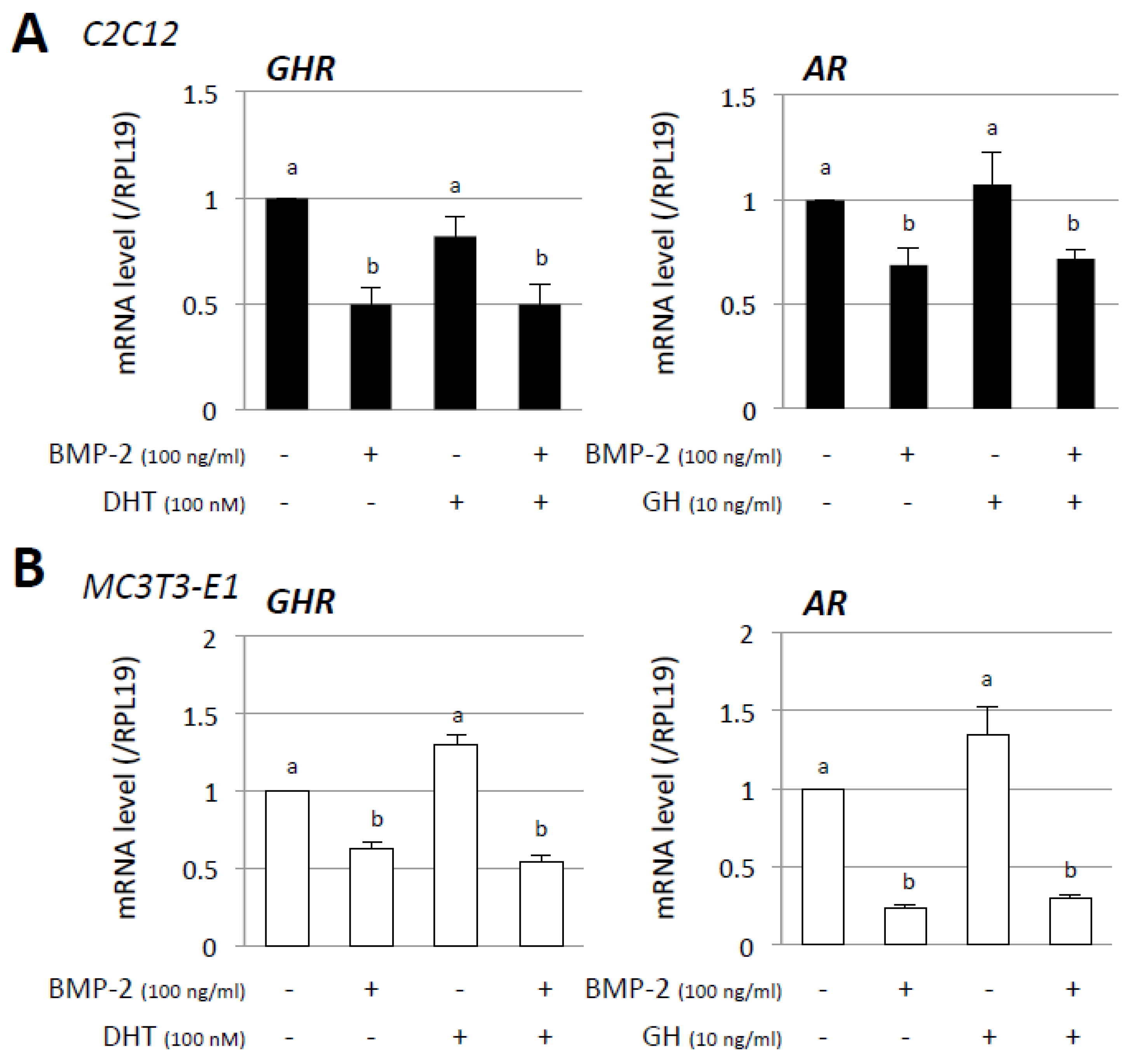
3. Results
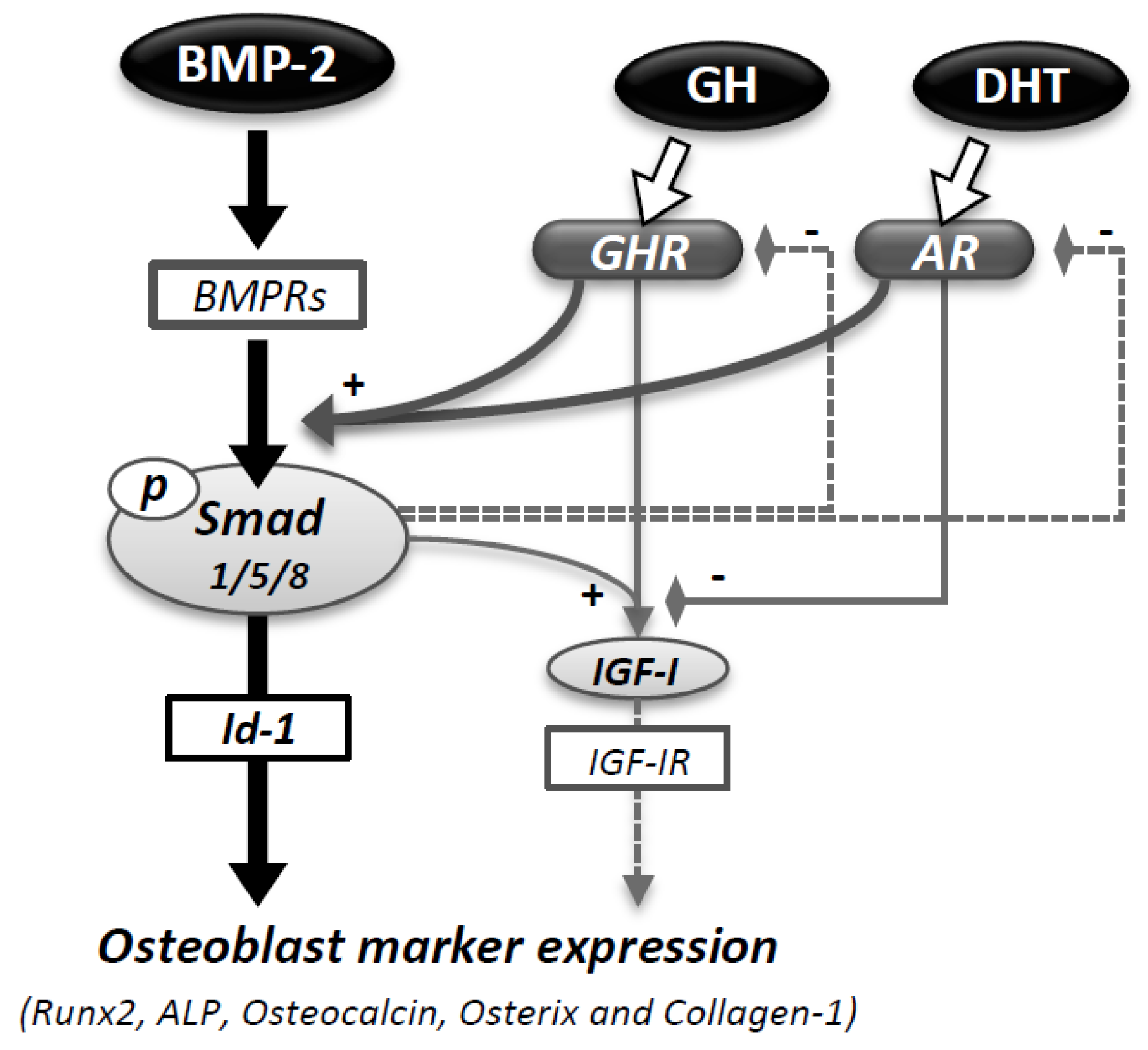
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AR | androgen receptor |
| BMD | bone mineral density |
| BMP | bone morphogenetic protein |
| DHT | dihydrotestosterone |
| GH | growth hormone |
| GHR | GH receptor |
| IGFBP | IGF binding protein |
| IGF-I | Insulin-like growth factor-I |
| IGF-IR | IGF-I receptor |
References
- Spelsberg, T.C.; Subramaniam, M.; Riggs, B.L.; Khosla, S. The actions and interactions of sex steroids and growth factors/cytokines on the skeleton. Mol. Endocrinol. 1999, 13, 819–828. [Google Scholar] [CrossRef] [PubMed]
- Gurlek, A.; Pittelkow, M.R.; Kumar, R. Modulation of growth factor/cytokine synthesis and signaling by 1α,25-dihydroxyvitamin D3: Implications in cell growth and differentiation. Endocr. Rev. 2002, 23, 763–786. [Google Scholar] [CrossRef] [PubMed]
- Sinnesael, M.; Claessens, F.; Boonen, S.; Vanderschueren, D. Novel insights in the regulation and mechanism of androgen action on bone. Curr. Opin. Endocrinol. Diabetes Obes. 2013, 20, 240–244. [Google Scholar] [CrossRef] [PubMed]
- Cauley, J.A. Estrogen and bone health in men and women. Steroids 2015, 99, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Khosla, S.; Melton, L.J.; Riggs, B.L., 3rd. Clinical review 144: Estrogen and the male skeleton. J. Clin. Endocrinol. Metab. 2002, 87, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Tritos, N.A.; Greenspan, S.L.; King, D.; Hamrahian, A.; Cook, D.M.; Jonsson, P.J.; Wajnrajch, M.P.; Koltowska-Haggstrom, M.; Biller, B.M. Unreplaced sex steroid deficiency, corticotropin deficiency, and lower IGF-I are associated with lower bone mineral density in adults with growth hormone deficiency: A KIMS database analysis. J. Clin. Endocrinol. Metab. 2011, 96, 1516–1523. [Google Scholar] [CrossRef] [PubMed]
- Wuster, C.; Abs, R.; Bengtsson, B.A.; Bennmarker, H.; Feldt-Rasmussen, U.; Hernberg-Stahl, E.; Monson, J.P.; Westberg, B.; Wilton, P.; Group, K.S.; et al. The influence of growth hormone deficiency, growth hormone replacement therapy, and other aspects of hypopituitarism on fracture rate and bone mineral density. J. Bone Miner. Res. 2001, 16, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Snyder, P.J.; Biller, B.M.; Zagar, A.; Jackson, I.; Arafah, B.M.; Nippoldt, T.B.; Cook, D.M.; Mooradian, A.D.; Kwan, A.; Scism-Bacon, J.; et al. Effect of growth hormone replacement on BMD in adult-onset growth hormone deficiency. J. Bone Miner. Res. 2007, 22, 762–770. [Google Scholar] [CrossRef] [PubMed]
- Snyder, P.J.; Peachey, H.; Berlin, J.A.; Hannoush, P.; Haddad, G.; Dlewati, A.; Santanna, J.; Loh, L.; Lenrow, D.A.; Holmes, J.H.; et al. Effects of testosterone replacement in hypogonadal men. J. Clin. Endocrinol. Metab. 2000, 85, 2670–2677. [Google Scholar] [CrossRef] [PubMed]
- Katagiri, T.; Yamaguchi, A.; Komaki, M.; Abe, E.; Takahashi, N.; Ikeda, T.; Rosen, V.; Wozney, J.M.; Fujisawa-Sehara, A.; Suda, T. Bone morphogenetic protein-2 converts the differentiation pathway of C2C12 myoblasts into the osteoblast lineage. J. Cell Biol. 1994, 127, 1755–1766. [Google Scholar] [CrossRef] [PubMed]
- Shimasaki, S.; Moore, R.K.; Otsuka, F.; Erickson, G.F. The bone morphogenetic protein system in mammalian reproduction. Endocr. Rev. 2004, 25, 72–101. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, F. Multiple endocrine regulation by bone morphogenetic protein system. Endocr. J. 2010, 57, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Otsuka, F.; Takano, M.; Mukai, T.; Yamanaka, R.; Takeda, M.; Miyoshi, T.; Inagaki, K.; Sada, K.E.; Makino, H. Estrogen and glucocorticoid regulate osteoblast differentiation through the interaction of bone morphogenetic protein-2 and tumor necrosis factor-α in C2C12 cells. Mol. Cell Endocrinol. 2010, 325, 118–127. [Google Scholar] [CrossRef] [PubMed]
- Takano, M.; Otsuka, F.; Matsumoto, Y.; Inagaki, K.; Takeda, M.; Nakamura, E.; Tsukamoto, N.; Miyoshi, T.; Sada, K.E.; Makino, H. Peroxisome proliferator-activated receptor activity is involved in the osteoblastic differentiation regulated by bone morphogenetic proteins and tumor necrosis factor-α. Mol. Cell Endocrinol. 2012, 348, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Otsuka, F.; Takano-Narazaki, M.; Katsuyama, T.; Nakamura, E.; Tsukamoto, N.; Inagaki, K.; Sada, K.E.; Makino, H. Estrogen facilitates osteoblast differentiation by upregulating bone morphogenetic protein-4 signaling. Steroids 2013, 78, 513–520. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Otsuka, F.; Otani, H.; Yamashita, M.; Takasugi, K.; Inagaki, K.; Yamamura, M.; Makino, H. TNF-α inhibits BMP-induced osteoblast differentiation through activating SAPK/JNK signaling. Biochem. Biophys. Res. Commun. 2007, 356, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, E.; Otsuka, F.; Inagaki, K.; Miyoshi, T.; Matsumoto, Y.; Ogura, K.; Tsukamoto, N.; Takeda, M.; Makino, H. Mutual regulation of growth hormone and bone morphogenetic protein system in steroidogenesis by rat granulosa cells. Endocrinology 2012, 153, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Katsuyama, T.; Otsuka, F.; Terasaka, T.; Inagaki, K.; Takano-Narazaki, M.; Matsumoto, Y.; Sada, K.E.; Makino, H. Regulatory effects of fibroblast growth factor-8 and tumor necrosis factor-α on osteoblast marker expression induced by bone morphogenetic protein-2. Peptides 2015, 73, 88–94. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, M.; Otsuka, F.; Mukai, T.; Otani, H.; Inagaki, K.; Miyoshi, T.; Goto, J.; Yamamura, M.; Makino, H. Simvastatin antagonizes tumor necrosis factor-α inhibition of bone morphogenetic proteins-2-induced osteoblast differentiation by regulating Smad signaling and Ras/Rho-mitogen-activated protein kinase pathway. J. Endocrinol. 2008, 196, 601–613. [Google Scholar] [CrossRef] [PubMed]
- Diel, P.; Baadners, D.; Schlupmann, K.; Velders, M.; Schwarz, J.P. C2C12 myoblastoma cell differentiation and proliferation is stimulated by androgens and associated with a modulation of myostatin and Pax7 expression. J. Mol. Endocrinol. 2008, 40, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Kanno, Y.; Ota, R.; Someya, K.; Kusakabe, T.; Kato, K.; Inouye, Y. Selective androgen receptor modulator, YK11, regulates myogenic differentiation of C2C12 myoblasts by follistatin expression. Biol. Pharm. Bull. 2013, 36, 1460–1465. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lee, N.K.; Zajac, J.D.; MacLean, H.E. Generation and analysis of an androgen-responsive myoblast cell line indicates that androgens regulate myotube protein accretion. J. Endocrinol. Investig. 2008, 31, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Frost, R.A.; Nystrom, G.J.; Lang, C.H. Regulation of IGF-I mRNA and signal transducers and activators of transcription-3 and -5 (Stat-3 and -5) by GH in C2C12 myoblasts. Endocrinology 2002, 143, 492–503. [Google Scholar] [CrossRef] [PubMed]
- Sadowski, C.L.; Wheeler, T.T.; Wang, L.H.; Sadowski, H.B. GH regulation of IGF-I and suppressor of cytokine signaling gene expression in C2C12 skeletal muscle cells. Endocrinology 2001, 142, 3890–3900. [Google Scholar] [CrossRef] [PubMed]
- Hansen, M.; Boesen, A.; Holm, L.; Flyvbjerg, A.; Langberg, H.; Kjaer, M. Local administration of insulin-like growth factor-I (IGF-I) stimulates tendon collagen synthesis in humans. Scand. J. Med. Sci. Sports 2013, 23, 614–619. [Google Scholar] [CrossRef] [PubMed]
- Xin, X.; Hou, Y.T.; Li, L.; Schmiedlin-Ren, P.; Christman, G.M.; Cheng, H.L.; Bitar, K.N.; Zimmermann, E.M. IGF-I increases IGFBP-5 and collagen α1(I) mRNAs by the MAPK pathway in rat intestinal smooth muscle cells. Am. J. Physiol. Gastrointest. Liver Physiol. 2004, 286, G777–G783. [Google Scholar] [CrossRef] [PubMed]
- Bex, M.; Abs, R.; Maiter, D.; Beckers, A.; Lamberigts, G.; Bouillon, R. The effects of growth hormone replacement therapy on bone metabolism in adult-onset growth hormone deficiency: A 2-year open randomized controlled multicenter trial. J. Bone Miner. Res. 2002, 17, 1081–1094. [Google Scholar] [CrossRef] [PubMed]
- Singhal, V.; Goh, B.C.; Bouxsein, M.L.; Faugere, M.C.; DiGirolamo, D.J. Osteoblast-restricted Disruption of the Growth Hormone Receptor in Mice Results in Sexually Dimorphic Skeletal Phenotypes. Bone Res. 2013, 1, 85–97. [Google Scholar] [CrossRef] [PubMed]
- Blackman, M.R.; Sorkin, J.D.; Munzer, T.; Bellantoni, M.F.; Busby-Whitehead, J.; Stevens, T.E.; Jayme, J.; O'Connor, K.G.; Christmas, C.; Tobin, J.D.; et al. Growth hormone and sex steroid administration in healthy aged women and men: A randomized controlled trial. JAMA 2002, 288, 2282–2292. [Google Scholar] [CrossRef] [PubMed]
- Mauras, N.; Rini, A.; Welch, S.; Sager, B.; Murphy, S.P. Synergistic effects of testosterone and growth hormone on protein metabolism and body composition in prepubertal boys. Metabolism 2003, 52, 964–969. [Google Scholar] [CrossRef]
- Al Mukaddam, M.; Rajapakse, C.S.; Bhagat, Y.A.; Wehrli, F.W.; Guo, W.; Peachey, H.; LeBeau, S.O.; Zemel, B.S.; Wang, C.; Swerdloff, R.S.; et al. Effects of testosterone and growth hormone on the structural and mechanical properties of bone by micro-MRI in the distal tibia of men with hypopituitarism. J. Clin. Endocrinol. Metab. 2014, 99, 1236–1244. [Google Scholar] [CrossRef] [PubMed]
- Bikle, D.; Majumdar, S.; Laib, A.; Powell-Braxton, L.; Rosen, C.; Beamer, W.; Nauman, E.; Leary, C.; Halloran, B. The skeletal structure of insulin-like growth factor I-deficient mice. J. Bone Miner. Res. 2001, 16, 2320–2329. [Google Scholar] [CrossRef] [PubMed]
- Olson, L.E.; Ohlsson, C.; Mohan, S. The role of GH/IGF-I-mediated mechanisms in sex differences in cortical bone size in mice. Calcif. Tissue Int. 2011, 88, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Klover, P.; Chen, W.; Zhu, B.M.; Hennighausen, L. Skeletal muscle growth and fiber composition in mice are regulated through the transcription factors STAT5a/b: Linking growth hormone to the androgen receptor. FASEB J. 2009, 23, 3140–3148. [Google Scholar] [CrossRef] [PubMed]
- Iglesias-Gato, D.; Chuan, Y.C.; Wikstrom, P.; Augsten, S.; Jiang, N.; Niu, Y.; Seipel, A.; Danneman, D.; Vermeij, M.; Fernandez-Perez, L.; et al. SOCS2 mediates the cross talk between androgen and growth hormone signaling in prostate cancer. Carcinogenesis 2014, 35, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Venken, K.; Moverare-Skrtic, S.; Kopchick, J.J.; Coschigano, K.T.; Ohlsson, C.; Boonen, S.; Bouillon, R.; Vanderschueren, D. Impact of androgens, growth hormone, and IGF-I on bone and muscle in male mice during puberty. J. Bone Miner. Res. 2007, 22, 72–82. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kimura, K.; Terasaka, T.; Iwata, N.; Katsuyama, T.; Komatsubara, M.; Nagao, R.; Inagaki, K.; Otsuka, F. Combined Effects of Androgen and Growth Hormone on Osteoblast Marker Expression in Mouse C2C12 and MC3T3-E1 Cells Induced by Bone Morphogenetic Protein. J. Clin. Med. 2017, 6, 6. https://doi.org/10.3390/jcm6010006
Kimura K, Terasaka T, Iwata N, Katsuyama T, Komatsubara M, Nagao R, Inagaki K, Otsuka F. Combined Effects of Androgen and Growth Hormone on Osteoblast Marker Expression in Mouse C2C12 and MC3T3-E1 Cells Induced by Bone Morphogenetic Protein. Journal of Clinical Medicine. 2017; 6(1):6. https://doi.org/10.3390/jcm6010006
Chicago/Turabian StyleKimura, Kosuke, Tomohiro Terasaka, Nahoko Iwata, Takayuki Katsuyama, Motoshi Komatsubara, Ryota Nagao, Kenichi Inagaki, and Fumio Otsuka. 2017. "Combined Effects of Androgen and Growth Hormone on Osteoblast Marker Expression in Mouse C2C12 and MC3T3-E1 Cells Induced by Bone Morphogenetic Protein" Journal of Clinical Medicine 6, no. 1: 6. https://doi.org/10.3390/jcm6010006
APA StyleKimura, K., Terasaka, T., Iwata, N., Katsuyama, T., Komatsubara, M., Nagao, R., Inagaki, K., & Otsuka, F. (2017). Combined Effects of Androgen and Growth Hormone on Osteoblast Marker Expression in Mouse C2C12 and MC3T3-E1 Cells Induced by Bone Morphogenetic Protein. Journal of Clinical Medicine, 6(1), 6. https://doi.org/10.3390/jcm6010006