Cancer Risk and Eicosanoid Production: Interaction between the Protective Effect of Long Chain Omega-3 Polyunsaturated Fatty Acid Intake and Genotype
Abstract
:1. Introduction
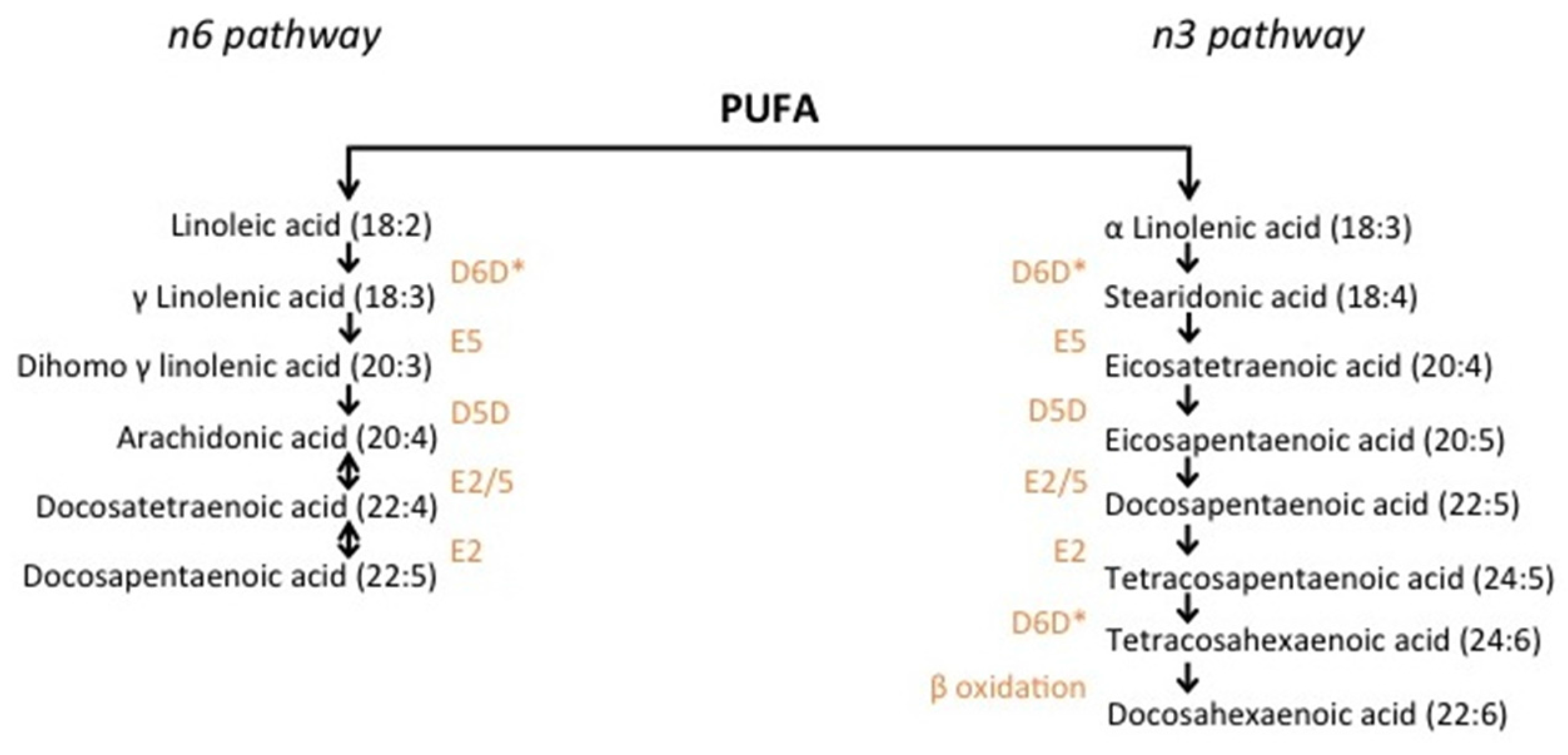

2. Methods
| Reference | n | Subjects/methods | Exposure Measurement (PC; CC, CS) | Intervention (RCT) | Cancer type | Gene/s | Locus | Effect |
|---|---|---|---|---|---|---|---|---|
| Al-Hilal et al., 2013 [14] | 367 | 6-month RCT/M + F, 45–70 year | EPA + DHA; 0.45, 0.9 or 1.9 g/day | FADS1 + FADS2 | rs174537 | ↓D5D activity associated with T variant allele; ↑D5D activity in TG, TT with increasing doses; no association for D6D | ||
| Fradet et al., 2009 [12] | Control 478; case 466 | CC/M, mean age 65 year | FFQ | Prostate | COX2 | rs4648310 | G allele correlated with aggressive PCa when n-3 FA intake was low, and low risk with high intake | |
| Gago-Dominguez et al., 2004 [49] | Control 670; case 258 | CC/F, 45–74 year | SQ FFQ | Breast | GSTM1, GSTT1, GSTP1 | multiple | Lower activity genotypes associated with higher BCa protection with ↑ intake of marine n-3 FA | |
| Habermann et al., 2013 [11] | Control 912; case 712 | CC/M + F, 30–79 year | CARDIA questionnaire | Rectal | ALOX12 | rs11571339 | G allele associated with↓rectal cancer risk in those with low n-3 PUFA intake (NS) | |
| Habermann et al., 2013 [11] | Control 1900; case 1543 | CC/M + F, 30–79 year | CARDIA questionnaire | Colon | ALOX15 | rs11568131 | AA genotype have ↓risk of colon cancer with↑ intakes of n-3 PUFA (NS) | |
| Habermann et al., 2013 [11] | Control 1900; case 1543 | CC/M + F, 30–79 year | CARDIA questionnaire | Colon | COX1 | rs10306110 | Low EPA/DHA intake associated with higher colon cancer risk in variant allele carriers only | |
| Hedelin et al., 2006 [50] | Control 1130; case1499 | CC/M, 35–79 year | FFQ | Prostate | COX2 | rs5275 | C allele at locus rs5275 correlated with ↓risk of PCa with high intake of fatty fish | |
| Hester et al., 2014 [13] | 30 | CS/Caucasian F, 21–65 year | Serum FA | FADS1 | rs174537 | T variant correlated with lower AA; GG genotype associated with ↑LTB4 + 5-HETE | ||
| Hog et al., 2013 [37] | 122 | 3 year PC/M, 35–59 year | Blood serum | FADS1, FADS2 + FADS3 | rs174537 (FADS1); rs174575, rs2727270 (FADS2), rs1000778 (FADS3) | rs174537GG had ↑AA, AA/DGLA, DPA, LDL, oxLDL + ↓ETA. Rs17453 had↓AA, AA/DGLA, EPA, DPA, EPA/ALA + urinary PGF2a | ||
| Poole et al., 2007 [51] | Control 626; case 716 | CC/M + F, 30–74 year | FFQ | Colorectal | COX1 | Phe17Leu | Modest↓risk of CRC for carriers of P17 with higher fish intake; L17 carriers have ↓risk of CRC with lower intake | |
| Poole et al., 2010 [52] | Control 582; case 483 | CC/M + F, 30–74 year | FFQ | Colorectal | PGES | rs7873087 | Carriers of T allele have ↓risk of CRC with ↑fish intake | |
| Poole et al., 2010 [52] | Control 582; case 483 | CC/M + F, 30–74 year | FFQ | Colorectal | EP4 | Val294Ile | Carriers of Ile variant showed correlation between ↑fish intake and ↑CRC risk | |
| Porenta et al., 2013 [53] | 108 | 6-month RCT/CRC at risk M + F | 2 day FR + 24 h recall | Healthy People 2010 diet or Mediterranean diet | Colon | FADS cluster | rs174556 and rs174561 in FADS1, rs383445 in FADS2 and rs174537 of the FADS1/2 intragenic region | Wild-type alleles associated with lower AA in colonic mucosa in persons on Mediterranean Diet |
3. The Role of Genetic Variation in Fatty Acid Desaturation
4. Genetic Polymorphisms Modulate Leukotriene Synthesis in Cancer
4.1 Lipoxygenases
4.2 Glutathione S-Transferases
5. Prostaglandin Synthesis
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ferlay, J.; Shin, H.-R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Zheng, S.; Zhang, S.; Jin, W.; Liu, H.; Jin, M.; Chen, Z.; Ding, Z.; Wang, L.; Chen, K. Polymorphisms of inflammation-related genes and colorectal cancer risk: A population-based case-control study in China. Int. J. Immunogenet. 2014, 41, 289–297. [Google Scholar] [CrossRef] [PubMed]
- De Marzo, A.M.; Platz, E.A.; Sutcliffe, S.; Xu, J.; Grönberg, H.; Drake, C.G.; Nakai, Y.; Isaacs, W.B.; Nelson, W.G. Inflammation in prostate carcinogenesis. Nat. Rev. Cancer 2007, 7, 256–269. [Google Scholar] [CrossRef] [PubMed]
- Baena, R.; Salinas, P. Diet and colorectal cancer. Maturitas 2015, 80, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Shivappa, N.; Hébert, J.R.; Zucchetto, A.; Montella, M.; Serraino, D.; La Vecchia, C.; Rossi, M. Dietary inflammatory index and endometrial cancer risk in an Italian case-control study. Br. J. Nutr. 2015, 115, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Dasilva, G.; Pazos, M.; García-Egido, E.; Gallardo, J.M.; Rodríguez, I.; Cela, R.; Medina, I. Healthy effect of different proportions of marine ω-3 PUFAs EPA and DHA supplementation in Wistar rats: Lipidomic biomarkers of oxidative stress and inflammation. J. Nutr. Biochem. 2015, 26, 1385–1392. [Google Scholar] [CrossRef] [PubMed]
- Pelser, C.; Mondul, A.M.; Hollenbeck, A.R.; Park, Y. Dietary fat, fatty acids, and risk of prostate cancer in the NIH-AARP diet and health study. Cancer Epidemiol. Biomark. Prev. 2013, 22, 697–707. [Google Scholar] [CrossRef] [PubMed]
- Richman, E.L.; Kenfield, S.A.; Chavarro, J.E.; Stampfer, M.J.; Giovannucci, E.L.; Willett, W.C.; Chan, J.M. Fat intake after diagnosis and risk of lethal prostate cancer and all-cause mortality. JAMA Intern. Med. 2013, 173, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Azrad, M.; Turgeon, C.; Demark-Wahnefried, W. Current evidence linking polyunsaturated Fatty acids with cancer risk and progression. Front. Oncol. 2013, 3, 224. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Calder, P.C. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev. 2005, 45, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Habermann, N.; Ulrich, C.M.; Lundgreen, A.; Makar, K.W.; Poole, E.M.; Caan, B.; Kulmacz, R.; Whitton, J.; Galbraith, R.; Potter, J.D.; et al. PTGS1, PTGS2, ALOX5, ALOX12, ALOX15, and FLAP SNPs: Interaction with fatty acids in colon cancer and rectal cancer. Genes Nutr. 2013, 8, 115–126. [Google Scholar] [CrossRef] [PubMed]
- Fradet, V.; Cheng, I.; Casey, G.; Witte, J.S. Dietary omega-3 fatty acids, cyclooxygenase-2 genetic variation, and aggressive prostate cancer risk. Clin. Cancer Res. 2009, 15, 2559–2566. [Google Scholar] [CrossRef] [PubMed]
- Hester, A.G.; Murphy, R.C.; Uhlson, C.J.; Ivester, P.; Lee, T.C.; Sergeant, S.; Miller, L.R.; Howard, T.D.; Mathias, R.A.; Chilton, F.H. Relationship between a common variant in the fatty acid desaturase (FADS) cluster and eicosanoid generation in humans. J. Biol. Chem. 2014, 289, 22482–22489. [Google Scholar] [CrossRef] [PubMed]
- Al-Hilal, M.; Alsaleh, A.; Maniou, Z.; Lewis, F.J.; Hall, W.L.; Sanders, T.A.B.; O’Dell, S.D. Genetic variation at the FADS1-FADS2 gene locus influences delta-5 desaturase activity and LC-PUFA proportions after fish oil supplement. J. Lipid Res. 2013, 54, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Lenihan-Geels, G.; Bishop, K.S.; Ferguson, L.R. Alternative sources of omega-3 fats: Can we find a sustainable substitute for fish? Nutrients 2013, 5, 1301–1315. [Google Scholar] [CrossRef] [PubMed]
- Szefel, J.; Piotrowska, M.; Kruszewski, W.J.; Jankun, J.; Lysiak-Szydlowska, W.; Skrzypczak-Jankun, E. Eicosanoids in Prevention and Management of Diseases. Curr. Mol. Med. 2011, 11, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Pischon, T.; Hankinson, S.E.; Hotamisligil, G.S.; Rifai, N.; Willett, W.C.; Rimm, E.B. Habitual dietary intake of n-3 and n-6 fatty acids in relation to inflammatory markers among US men and women. Circulation 2003, 108, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Yli-Jama, P.; Seljeflot, I.; Meyer, H.E.; Hjerkinn, E.M.; Arnesen, H.; Pedersen, J.I. Serum non-esterified very long-chain PUFA are associated with markers of endothelial dysfunction. Atherosclerosis 2002, 164, 275–281. [Google Scholar] [CrossRef]
- Ferrucci, L.; Cherubini, A.; Bandinelli, S.; Bartali, B.; Corsi, A.; Lauretani, F.; Martin, A.; Andres-Lacueva, C.; Senin, U.; Guralnik, J.M. Relationship of Plasma Polyunsaturated Fatty Acids to Circulating Inflammatory Markers. J. Clin. Endocrinol. Metable. 2006, 91, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Ahluwalia, N.; Brouns, F.; Buetler, T.; Clement, K.; Cunningham, K.; Esposito, K.; Jönsson, L.S.; Kolb, H.; Lansink, M.; et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011, 106, S5–S78. [Google Scholar] [CrossRef] [PubMed]
- Tuncer, S.; Banerjee, S. Eicosanoid pathway in colorectal cancer: Recent updates. World J. Gastroenterol. 2015, 21, 11748–11766. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Zhu, J.; Lyu, F.; Panigrahy, D.; Ferrara, K.W.; Hammock, B.; Zhang, G. ω-3 polyunsaturated fatty acids-derived lipid metabolites on angiogenesis, inflammation and cancer. Prostaglandins Other Lipid Mediat. 2014, 113, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: the next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Mazhar, D.; Ang, R.; Waxman, J. COX inhibitors and breast cancer. Br. J. Cancer 2006, 94, 346–350. [Google Scholar] [CrossRef] [PubMed]
- Eberhart, C.E.; Coffey, R.J.; Radhika, A.; Giardiello, F.M.; Ferrenbach, S.; DuBois, R.N. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994, 107, 1183–1188. [Google Scholar] [PubMed]
- Kleinstein, S.E.; Heath, L.; Makar, K.W.; Poole, E.M.; Seufert, B.L.; Slattery, M.L.; Xiao, L.; Duggan, D.J.; Hsu, L.; Curtin, K.; et al. Genetic variation in the lipoxygenase pathway and risk of colorectal neoplasia. Genes. Chromosomes Cancer 2013, 52, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Augustsson, K.; Michaud, D.S.; Rimm, E.B.; Leitzmann, M.F.; Stampfer, M.J.; Willett, W.C.; Giovannucci, E. A prospective study of intake of fish and marine fatty acids and prostate cancer. Cancer Epidemiol. Biomark. Prev. 2003, 12, 64–67. [Google Scholar]
- Kantor, E.D.; Lampe, J.W.; Peters, U.; Vaughan, T.L.; White, E. Long-chain omega-3 polyunsaturated fatty acid intake and risk of colorectal cancer. Nutr. Cancer 2014, 66, 716–727. [Google Scholar] [CrossRef] [PubMed]
- Catsburg, C.; Joshi, A.D.; Corral, R.; Lewinger, J.P.; Koo, J.; John, E.M.; Ingles, S.A.; Stern, M.C. Polymorphisms in carcinogen metabolism enzymes, fish intake, and risk of prostate cancer. Carcinogenesis 2012, 33, 1352–1359. [Google Scholar] [CrossRef] [PubMed]
- Wallström, P.; Bjartell, A.; Gullberg, B.; Olsson, H.; Wirfält, E. A prospective study on dietary fat and incidence of prostate cancer (Malmö, Sweden). Cancer Causes Control 2007, 18, 1107–1121. [Google Scholar] [CrossRef] [PubMed]
- Corella, D.; Ordovás, J.M. Interactions between dietary n-3 fatty acids and genetic variants and risk of disease. Br. J. Nutr. 2012, 107, S271–S283. [Google Scholar] [CrossRef] [PubMed]
- Von Schacky, C. Omega-3 index and cardiovascular health. Nutrients 2014, 6, 799–814. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Exposure to Dioxins and Dioxin-like Substances: A Major Public Health Concern. Available online: http://www.who.int/ipcs/features/dioxins.pdf (accessed on 14 January 2016).
- White, M.C.; Holman, D.M.; Boehm, J.E.; Peipins, L.A.; Grossman, M.; Jane Henley, S. Age and Cancer Risk. Am. J. Prev. Med. 2014, 46, S7–S15. [Google Scholar] [CrossRef] [PubMed]
- Tintle, N.L.; Pottala, J.V.; Lacey, S.; Ramachandran, V.; Westra, J.; Rogers, A.; Clark, J.; Olthoff, B.; Larson, M.; Harris, W.; Shearer, G.C. A genome-wide association study of saturated, mono- and polyunsaturated red blood cell fatty acids in the Framingham Heart Offspring Study. Prostaglandins Leukot. Essent. Fatty Acids 2015, 94, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Bishop, K.S.; Erdrich, S.; Karunasinghe, N.; Han, D.Y.; Zhu, S.; Jesuthasan, A.; Ferguson, L.R. An investigation into the association between DNA damage and dietary fatty acid in men with prostate cancer. Nutrients 2015, 7, 405–422. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.H.; Kwak, J.H.; Paik, J.K.; Chae, J.S.; Lee, J.H. Association of polymorphisms in FADS gene with age-related changes in serum phospholipid polyunsaturated fatty acids and oxidative stress markers in middle-aged nonobese men. Clin. Interv. Aging 2013, 8, 585–596. [Google Scholar] [PubMed]
- Chilton, F.H.; Murphy, R.C.; Wilson, B.A.; Sergeant, S.; Ainsworth, H.; Seeds, M.C.; Mathias, R.A. Diet-gene interactions and PUFA metabolism: A potential contributor to health disparities and human diseases. Nutrients 2014, 6, 1993–2022. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Genetic variants in the metabolism of omega-6 and omega-3 fatty acids: their role in the determination of nutritional requirements and chronic disease risk. Exp. Biol. Med. (Maywood) 2010, 235, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yu, H.; Ni, X.; Shen, S.; Das, U.N. Growth inhibitory effect of polyunsaturated fatty acids (PUFAs) on colon cancer cells via their growth inhibitory metabolites and fatty acid composition changes. PLoS ONE 2015, 10, e0123256. [Google Scholar] [CrossRef] [PubMed]
- Mansara, P.P.; Deshpande, R.A.; Vaidya, M.M.; Kaul-Ghanekar, R. Differential Ratios of Omega Fatty Acids (AA/EPA + DHA) Modulate Growth, Lipid Peroxidation and Expression of Tumor Regulatory MARBPs in Breast Cancer Cell Lines MCF7 and MDA-MB-231. PLoS ONE 2015, 10, e0136542. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.P.; Connolly, J.M. Effects of fatty acids and eicosanoid synthesis inhibitors on the growth of two human prostate cancer cell lines. Prostate 1991, 18, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Akinsete, J.A.; Ion, G.; Witte, T.R.; Hardman, W.E. Consumption of high ω-3 fatty acid diet suppressed prostate tumorigenesis in C3(1) Tag mice. Carcinogenesis 2012, 33, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Van Beelen, V.A.; Spenkelink, B.; Mooibroek, H.; Sijtsma, L.; Bosch, D.; Rietjens, I.M.; Alink, G.M. An n-3 PUFA-rich microalgal oil diet protects to a similar extent as a fish oil-rich diet against AOM-induced colonic aberrant crypt foci in F344 rats. Food Chem. Toxicol. 2009, 47, 316–320. [Google Scholar] [CrossRef] [PubMed]
- Leslie, M.A.; Abdelmagid, S.A.; Perez, K.; Muller, W.J.; Ma, D.W. Mammary tumour development is dose-dependently inhibited by n-3 polyunsaturated fatty acids in the MMTV-neu(ndl)-YD5 transgenic mouse model. Lipids Health Dis. 2014, 13, 96. [Google Scholar] [CrossRef] [PubMed]
- Glaser, C.; Rzehak, P.; Demmelmair, H.; Klopp, N.; Heinrich, J.; Koletzko, B. Influence of FADS polymorphisms on tracking of serum glycerophospholipid fatty acid concentrations and percentage composition in children. PLoS ONE 2011, 6, e21933. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.-B.; Drummen, G.P.C.; Jiang, Z.-P.; Qin, Y.-H. GSTT1 polymorphism and the risk of developing prostate cancer. Am. J. Epidemiol. 2014, 180, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, C. COX-2’s new role in inflammation. Nat. Chem. Biol. 2010, 6, 401–402. [Google Scholar] [CrossRef] [PubMed]
- Gago-Dominguez, M. Marine n-3 fatty acid intake, glutathione S-transferase polymorphisms and breast cancer risk in post-menopausal Chinese women in Singapore. Carcinogenesis 2004, 25, 2143–2147. [Google Scholar] [CrossRef] [PubMed]
- Hedelin, M.; Chang, E.T.; Wiklund, F.; Bellocco, R.; Klint, A.; Adolfsson, J.; Shahedi, K.; Xu, J.; Adami, H.-O.; Grönberg, H.; Bälter, K.A. Association of frequent consumption of fatty fish with prostate cancer risk is modified by COX-2 polymorphism. Int. J. Cancer 2007, 120, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Poole, E.M.; Bigler, J.; Whitton, J.; Sibert, J.G.; Kulmacz, R.J.; Potter, J.D.; Ulrich, C.M. Genetic variability in prostaglandin synthesis, fish intake and risk of colorectal polyps. Carcinogenesis 2007, 28, 1259–1263. [Google Scholar] [CrossRef] [PubMed]
- Poole, E.M.; Hsu, L.; Xiao, L.; Kulmacz, R.J.; Carlson, C.S.; Rabinovitch, P.S.; Makar, K.W.; Potter, J.D.; Ulrich, C.M. Genetic variation in prostaglandin E2 synthesis and signaling, prostaglandin dehydrogenase, and the risk of colorectal adenoma. Cancer Epidemiol. Biomark. Prev. 2010, 19, 547–557. [Google Scholar] [CrossRef] [PubMed]
- Porenta, S.R.; Ko, Y.-A.; Gruber, S.B.; Mukherjee, B.; Baylin, A.; Ren, J.; Djuric, Z. Interaction of fatty acid genotype and diet on changes in colonic fatty acids in a Mediterranean diet intervention study. Cancer Prev. Res. (Phila) 2013, 6, 1212–1221. [Google Scholar] [CrossRef] [PubMed]
- Schaeffer, L.; Gohlke, H.; Müller, M.; Heid, I.M.; Palmer, L.J.; Kompauer, I.; Demmelmair, H.; Illig, T.; Koletzko, B.; Heinrich, J. Common genetic variants of the FADS1 FADS2 gene cluster and their reconstructed haplotypes are associated with the fatty acid composition in phospholipids. Hum. Mol. Genet. 2006, 15, 1745–1756. [Google Scholar] [CrossRef] [PubMed]
- Harsløf, L.B.S.; Larsen, L.H.; Ritz, C.; Hellgren, L.I.; Michaelsen, K.F.; Vogel, U.; Lauritzen, L. FADS genotype and diet are important determinants of DHA status: a cross-sectional study in Danish infants. Am. J. Clin. Nutr. 2013, 97, 1403–1410. [Google Scholar] [CrossRef] [PubMed]
- Cormier, H.; Rudkowska, I.; Paradis, A.-M.; Thifault, E.; Garneau, V.; Lemieux, S.; Couture, P.; Vohl, M.-C. Association between polymorphisms in the fatty acid desaturase gene cluster and the plasma triacylglycerol response to an n-3 PUFA supplementation. Nutrients 2012, 4, 1026–1041. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet; A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Dubois, R.N. Eicosanoids and cancer. Nat. Rev. Cancer 2010, 10, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Shan, D.; Shen, K.; Zhu, J.; Feng, M.; Wu, Y.; Wan, C.; Shen, Y.; Xu, L. The polymorphism (Gln261Arg) of 12-lipoxygenase and cancer risk: A meta-analysis. Int. J. Clin. Exp. Med. 2015, 8, 488–495. [Google Scholar] [PubMed]
- Prasad, V.V.; Kolli, P.; Moganti, D. Association of a functional polymorphism (Gln261Arg) in 12-lipoxygenase with breast cancer. Exp. Ther. Med. 2011, 2, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Gong, Z.; Hebert, J.R.; Bostick, R.M.; Deng, Z.; Hurley, T.G.; Dixon, D.A.; Nitcheva, D.; Xie, D. Common polymorphisms in 5-lipoxygenase and 12-lipoxygenase genes and the risk of incident, sporadic colorectal adenoma. Cancer 2007, 109, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Hao, Z. Polymorphisms of glutathione S-transferase M1 (GSTM1) and T1 (GSTT1) in ovarian cancer risk. Tumour Biol. 2014, 35, 5267–5272. [Google Scholar] [CrossRef] [PubMed]
- Kanwal, R.; Pandey, M.; Bhaskaran, N.; Maclennan, G.T.; Fu, P.; Ponsky, L.E.; Gupta, S. Protection against oxidative DNA damage and stress in human prostate by glutathione S-transferase P1. Mol. Carcinog. 2014, 53, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Van Hemelrijck, M.; Rohrmann, S.; Steinbrecher, A.; Kaaks, R.; Teucher, B.; Linseisen, J. Heterocyclic aromatic amine [HCA] intake and prostate cancer risk: effect modification by genetic variants. Nutr. Cancer 2012, 64, 704–713. [Google Scholar] [CrossRef] [PubMed]
- John, E.; Stern, M.; Sinha, R.; Koo, J. Meat Consumption, Cooking Practices, Meat Mutagens, and Risk of Prostate Cancer. Nutr. Cancer 2011, 63, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Murray, N.R.; Weems, C.; Chen, L.; Guo, H.; Ethridge, R.; Ceci, J.D.; Evers, B.M.; Thompson, E.A.; Fields, A.P. Role of cyclooxygenase 2 in protein kinase C beta II-mediated colon carcinogenesis. J. Biol. Chem. 2003, 278, 11167–11174. [Google Scholar] [CrossRef] [PubMed]
- Thun, M.J.; Henley, S.J.; Patrono, C. Nonsteroidal anti-inflammatory drugs as anticancer agents: Mechanistic, pharmacologic, and clinical issues. J. Natl. Cancer Inst. 2002, 94, 252–266. [Google Scholar] [CrossRef] [PubMed]
- Cavazos, D.A.; Price, R.S.; Apte, S.S.; deGraffenried, L.A. Docosahexaenoic acid selectively induces human prostate cancer cell sensitivity to oxidative stress through modulation of NF-κB. Prostate 2011, 71, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Eser, P.O.; Vanden Heuvel, J.P.; Araujo, J.; Thompson, J.T. Marine- and plant-derived ω-3 fatty acids differentially regulate prostate cancer cell proliferation. Mol. Clin. Oncol. 2013, 1, 444–452. [Google Scholar] [PubMed]
- Shahedi, K.; Lindström, S.; Zheng, S.L.; Wiklund, F.; Adolfsson, J.; Sun, J.; Augustsson-Bälter, K.; Chang, B.-L.; Adami, H.-O.; Liu, W.; Grönberg, H.; Xu, J. Genetic variation in the COX-2 gene and the association with prostate cancer risk. Int. J. Cancer 2006, 119, 668–672. [Google Scholar] [CrossRef] [PubMed]
- Qiao, L.; Kozoni, V.; Tsioulias, G.J.; Koutsos, M.I.; Hanif, R.; Shiff, S.J.; Rigas, B. Selected eicosanoids increase the proliferation rate of human colon carcinoma cell lines and mouse colonocytes in vivo. Biochim. Biophys. Acta 1995, 1258, 215–223. [Google Scholar] [CrossRef]
- Sheng, H.; Shao, J.; Washington, M.K.; DuBois, R.N. Prostaglandin E2 increases growth and motility of colorectal carcinoma cells. J. Biol. Chem. 2001, 276, 18075–18081. [Google Scholar] [CrossRef] [PubMed]
- Stern, M.C.; Butler, L.M.; Corral, R.; Joshi, A.D.; Yuan, J.-M.; Koh, W.-P.; Yu, M.C. Polyunsaturated fatty acids, DNA repair single nucleotide polymorphisms and colorectal cancer in the Singapore Chinese Health Study. J. Nutrigenet. Nutrigenom. 2009, 2, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.; Mansel, R.E.; Jiang, W.G. Expression of human delta-6-desaturase is associated with aggressiveness of human breast cancer. Int. J. Mol. Med. 2003, 12, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Azrad, M.; Zhang, K.; Vollmer, R.T.; Madden, J.; Polascik, T.J.; Snyder, D.C.; Ruffin, M.T.; Moul, J.W.; Brenner, D.; Hardy, R.W.; et al. Prostatic alpha-linolenic acid (ALA) is positively associated with aggressive prostate cancer: a relationship which may depend on genetic variation in ALA metabolism. PLoS ONE 2012, 7, e53104. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenihan-Geels, G.; Bishop, K.S.; Ferguson, L.R. Cancer Risk and Eicosanoid Production: Interaction between the Protective Effect of Long Chain Omega-3 Polyunsaturated Fatty Acid Intake and Genotype. J. Clin. Med. 2016, 5, 25. https://doi.org/10.3390/jcm5020025
Lenihan-Geels G, Bishop KS, Ferguson LR. Cancer Risk and Eicosanoid Production: Interaction between the Protective Effect of Long Chain Omega-3 Polyunsaturated Fatty Acid Intake and Genotype. Journal of Clinical Medicine. 2016; 5(2):25. https://doi.org/10.3390/jcm5020025
Chicago/Turabian StyleLenihan-Geels, Georgia, Karen S. Bishop, and Lynnette R. Ferguson. 2016. "Cancer Risk and Eicosanoid Production: Interaction between the Protective Effect of Long Chain Omega-3 Polyunsaturated Fatty Acid Intake and Genotype" Journal of Clinical Medicine 5, no. 2: 25. https://doi.org/10.3390/jcm5020025
APA StyleLenihan-Geels, G., Bishop, K. S., & Ferguson, L. R. (2016). Cancer Risk and Eicosanoid Production: Interaction between the Protective Effect of Long Chain Omega-3 Polyunsaturated Fatty Acid Intake and Genotype. Journal of Clinical Medicine, 5(2), 25. https://doi.org/10.3390/jcm5020025








