Potential Benefits of Omega-3 Fatty Acids in Non-Melanoma Skin Cancer
Abstract
:1. Introduction
2. Essential Fatty Acids
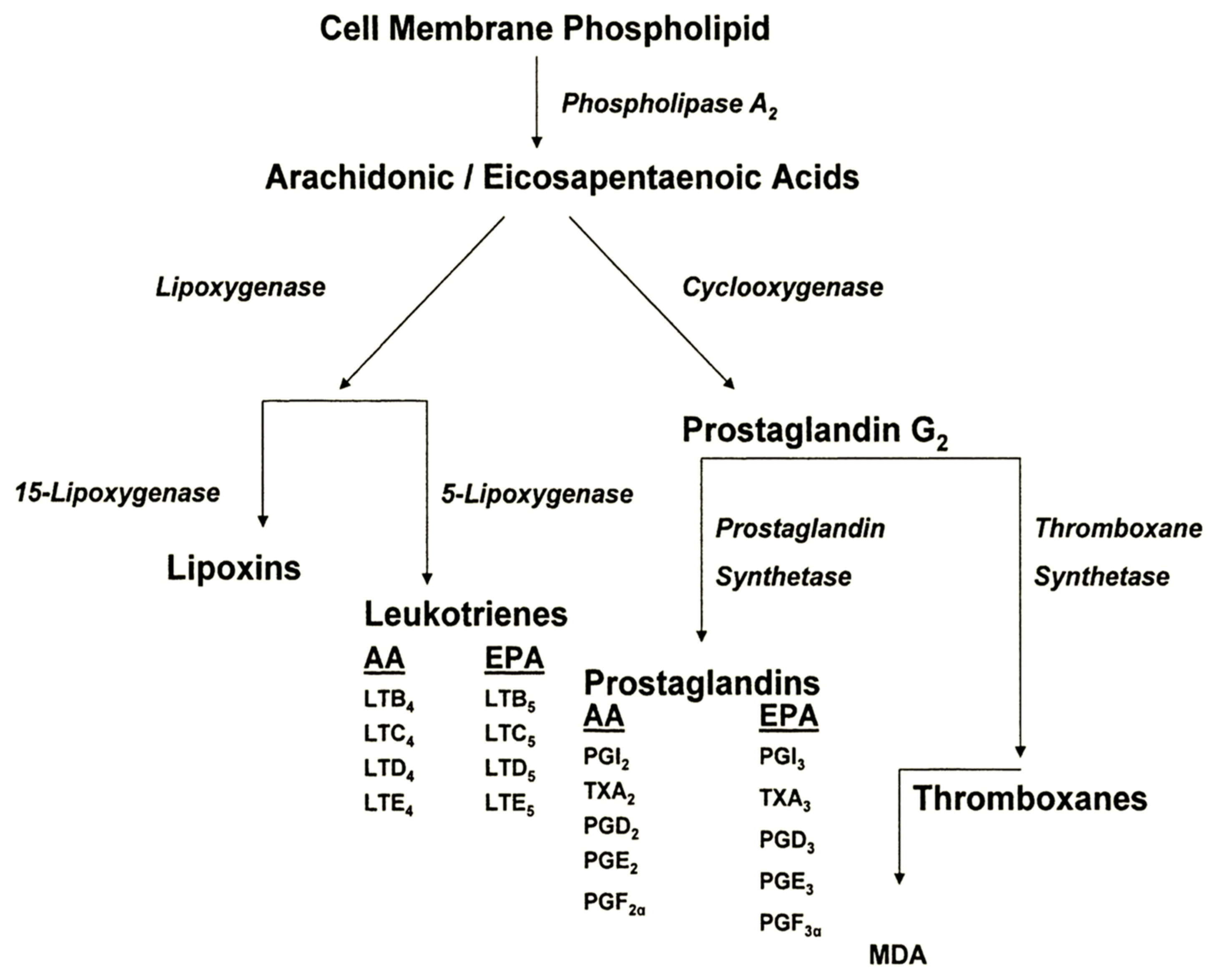
3. Evidence for Participation of Dietary PUFA in UVR-Induced Skin Cancer
3.1. Animal Studies
- Increasing levels of dietary omega-6 FA exacerbate UVR carcinogenic expression, with respect to both shortened tumor latent period and increased tumor multiplicity.
- Dietary omega-3 FA inhibit UVR carcinogenic expression.
- Omega-6 FA exert their principal effect upon the post-initiation, or promotion/progression stages of UVR carcinogenesis.
- Omega-3 FA appear to exert their principal effects during the initiation stage of the carcinogenic continuum.
- Pro-inflammatory and immunosuppressive PGE2 levels are increased linearly as dietary omega-6 FA levels increase.
- Pro-inflammatory and immunosuppressive PGE2 levels are dramatically reduced by dietary omega-3 FA intake.
- Dietary omega-6 FA suppress the immunologic responses involved in tumor transplant rejection and the immunologic pathways involved in DTH and CHS.
- Dietary omega-3 FA inhibit UVR-induced suppression of DTH and CHS.
3.2. Clinical Studies
- Omega-3 FA supplementation significantly increases the erythema threshold to UVR.
- Omega-3 FA modulate a number of cytokines (in human cells in vitro only) and eicosanoids that mediate inflammatory and immune responses.
- Omega-3 FA inhibit certain genotoxic markers of UVR-induced DNA damage, e.g., UVR- induced cutaneous p53.
- Omega-3 FA abrogate UVR-induced immunosuppression of cell mediated immunity assessed as nickel CHS
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Bang, H.O.; Dyerberg, J. Plasma lipid and lipoprotein pattern in Greenlandic West Coast Eskimos. Lancet 1971, 1, 1143–1145. [Google Scholar] [CrossRef]
- Bang, H.O.; Dyerberg, J.; Hjorne, N. The composition of food consumed by Greenland Eskimos. Acta Med. Scand. 1976, 200, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Bang, H.O.; Dyerberg, J.; Sinclair, H.M. The composition of the Eskimo food in North Western Greenland. Am. J. Clin. Nutr. 1980, 33, 2657–2661. [Google Scholar] [PubMed]
- Jordan, H.; Matthan, N.; Chung, M.; Balk, E.; Chew, P.; Kupelnick, B.; Lawrence, A.; Lichtenstein, A.; Lau, J. Effects of Omega-3 Fatty Acids on Arrhythmogenic Mechanisms in Animal and Isolated Organ/Cell Culture Studies; Evidence Report/Technology Assessment No. 92; Publication No. 04-EO11-2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- Balk, E.; Chung, M.; Lichtenstein, A.; Chew, P.; Kupelnick, B.; DeVine, D.; Lawrence, A.; Lau, J. Effects of Omega-3 Fatty Acids on Cardiovascular Risk Factors and Intermediate Markers of Cardiovascular Disease; Evidence Report/Technology Assessment No. 93; Publication No. 04-EO10-2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- Wang, C.; Chung, M.; Lichtenstein, A.; Balk, E.; Kupelnick, B.; DeVine, D.; Lawrence, A.; Lau, J. Effects of Omega-3 Fatty Acids on Cardiovascular Disease; Evidence Report/Technology Assessment No. 94; Publication No. 04-EO09-2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- Schacter, H.; Reisman, J.; Tran, K.; Dales, B.; Kourad, K.; Barnes, D.; Sampson, M.; Morrison, A.; Gaboury, I.; Blackman, J. Health Effects of Omega-3 Fatty Acids on Asthma; Evidence Report/Technology Assessment No. 94; Publication No. 04-EO13-2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- MacLean, C.H.; Mojica, W.A.; Morton, S.C.; Pencharz, J.; Hasenfeld Garland, R.; Tu, W.; Newberry, S.J.; Jungvig, L.K.; Khanna, P.; Rhodes, S.; et al. Effects of Omega-3 Fatty Acids on Lipids and Glycemic Control in Type II Diabetes and the Metabolic Syndrome and on Inflammatory Bowel Disease, Rheumatoid Arthritis, Renal Disease, Systemic Lupus Erythematosus, and Osteoporosis; Evidence Report/Technology Assessment No. 89; Publication No. 04-EO12–2; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2004.
- Nixon, D.W.; Rodgers, K. Breast Cancer. In Nutritional Oncology, 2nd ed.; Heber, D., Blackburn, G.L., Go, V.L.W., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 447–452. [Google Scholar]
- Aronson, W.; Yip, I.; Dekernion, J. Prostate Cancer. In Nutritional Oncology, 2nd ed.; Heber, D., Blackburn, G.L., Go, V.L.W., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 453–461. [Google Scholar]
- Clinton, S.K.; Michaud, D.; Giovannucci, E. Nutrition and Bladder Cancer. In Nutritional Oncology, 2nd ed.; Heber, D., Blackburn, G.L., Go, V.L.W., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 463–475. [Google Scholar]
- Harris, D.M.; Kang, S.Y.; Go, V.L.W. Nutrient-Gene Interactions and Prevention of Colorectal, Liver, and Pancreatic Cancer. In Nutritional Oncology, 2nd ed.; Heber, D., Blackburn, G.L., Go, V.L.W., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 469–500. [Google Scholar]
- Caygill, C.P.J.; Charlett, A.; Hill, M.J. Fat, fish, fish oil and cancer. Br. J. Cancer 1996, 74, 159–164. [Google Scholar] [CrossRef] [PubMed]
- MacLean, C.H.; Newberry, S.J.; Mojica, W.A.; Issa, A.; Khanna, P.; Lim, Y.S.W.; Morton, S.C.; Suttorp, M.; Tu, W.; Hilton, L.G.; et al. Effects of Omega-3 Fatty Acids on Cancer; Evidence Report/Technology Assessment No. 113; Publication No. 05-EO10-1; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2006.
- MacLean, C.H.; Newberry, S.J.; Mojica, W.A.; Khanna, P.; Issa, A.M.; Suttorp, M.J.; Lim, Y.W.; Traina, S.B.; Hilton, L.; Garland, R.; et al. Effects of omega-3 fatty acids on cancer risk: a systematic review. JAMA 2006, 295, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Fabian, C.J.; Kimler, B.F.; Hursting, S.D. Omega-3 fatty acids for breast cancer prevention and survivorship. Breast Cancer Res. 2015, 17, 62–77. [Google Scholar] [CrossRef] [PubMed]
- Brasky, T.M.; Darke, A.K.; Song, X.; Tangen, C.M.; Goodman, P.J.; Thompson, I.M.; Mayskens, F.L., Jr.; Goodman, G.E.; Minasian, L.M.; Parnes, H.L.; et al. Plasma phospholipid fatty acids and prostate cancer risk in the SELECT trial. J. Natl. Cancer Inst. 2013, 105, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.D.; Bassett, J.K.; Weed, D.L.; Barrett, E.C.; Watson, H.; Harris, W. Meta-analysis of long-chain omega-3 polyunsaturated fatty acids (LCω-3 PUFA) and prostate cancer. Nutr. Cancer 2015, 67, 543–554. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.; Ouyang, W.; Huang, C. Inflammation, a key event in cancer development. Mol. Cancer Res. 2006, 4, 221–233. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Cancer Facts and Figures, Atlanta, GA. 2014. Available online: http://www.cancer.org (accessed on 8 November 2015).
- Vanderveen, E.; Grekin, R.; Swanson, N.; Kragballe, K. Arachidonic acid metabolites in cutaneous carcinomas. Evidence suggesting that elevated levels of prostaglandins in basal cell carcinomas are associated with aggressive growth pattern. Arch Dermatol. 1986, 122, 407–412. [Google Scholar] [CrossRef] [PubMed]
- Malmsten, C. Leukotrienes: Mediators of inflammation and immediate hypersensitivity reactions. Crit. Rev. Immunol. 1984, 4, 307–334. [Google Scholar] [PubMed]
- Werner, E.; Walenga, R.; Dubowy, R.; Boone, S.; Stuart, M. Inhibition of human malignant neuroblastoma cell DNA synthesis by lipoxygenase metabolites of arachidonic acid. Cancer Res. 1985, 45, 561–563. [Google Scholar] [PubMed]
- Black, H.S.; Rhodes, L.E. The potential of omega-3 fatty acids in the prevention of non- melanoma skin cancer. Cancer Det. Prev. 2006, 30, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Baumann, C.; Rusch, H. Effect of diet on tumors induced by ultraviolet light. Am. J. Cancer 1939, 35, 213–221. [Google Scholar]
- Black, H.S.; Lenger, W.; Phelps, A.W.; Thornby, J.I. Influence of dietary lipid upon ultraviolet-light carcinogenesis. Nutr. Cancer 1983, 5, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Black, H.S.; Lenger, W.A.; Gerguis, J.; Thornby, J.I. Relation of antioxidants and level of dietary lipid to epidermal lipid peroxidation and ultraviolet carcinogenesis. Cancer Res. 1985, 45, 6254–6259. [Google Scholar] [PubMed]
- Reeve, V.; Bosnic, M.; Boehm-Wilcox, C. Dependence of photocarcinogenesis and photoimmunosuppression in the hairless mouse on dietary polyunsaturated fat. Cancer Lett. 1966, 108, 271–279. [Google Scholar] [CrossRef]
- Black, H.S.; Thornby, J.I.; Gerguis, J.; Lenger, W. Influence of dietary omega-6, -3 fatty acid sources on the initiation and promotion stages of photocarcinogenesis. Photochem. Photobiol. 1992, 56, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Orengo, I.F.; Black, H.S.; Kettler, A.H.; Wolf, J.E., Jr. Influence of dietary menhaden oil upon carcinogenesis and various cutaneous responses to ultraviolet radiation. Photochem. Photobiol. 1989, 49, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Black, H.S. Omega-3 fatty acids and non-melanoma skin cancer. In Handbook of Diet, Nutrition and the Skin; Preedy, V.R., Ed.; Wageningen Academic Publishers: Wageningen, The Netherlands, 2012; pp. 367–378. [Google Scholar]
- Fischer, M.A.; Black, H.S. Modification of membrane composition, eicosanoid metabolism, and immunoresponsiveness by dietary omega-3 and omega-6 fatty acid sources, modulators of ultraviolet-carcinogenesis. Photochem. Photobiol. 1991, 54, 381–387. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.X. Fat-1 transgenic mice: A new model for omega-3 research. Prostaglandins Leukot Essent Fatty Acids 2007, 77, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Xia, S.; Lu, Y.; Wang, J.; He, C.; Hong, S.; Serhan, C.N.; Kang, J.X. Melanoma growth is reduced in fat-1 transgenic mice: Impact of omega-6/omega-3 essential fatty acids. Proc. Natl. Acad. Sci. USA 2006, 103, 12499–12504. [Google Scholar] [CrossRef] [PubMed]
- Strickland, P.; Creasia, D.; Kripke, M. Enhancement of two-stage skin carcinogenesis by exposure of distant skin to UV-radiation. J. Natl. Cancer Inst. 1985, 74, 1129–1134. [Google Scholar] [PubMed]
- Chung, H.; Burnham, D.; Robertson, B.; Roberts, I.; Daynes, R. Involvement of prostaglandins in the immune alterations caused by exposure of mice to ultraviolet radiation. J. Immunol. 1986, 137, 2478–2884. [Google Scholar] [PubMed]
- Moison, R.; Beijersbergen Van Henegouwen, G. Dietary eicosapentaenoic acid prevents systemic immunosuppression in mice induced by UVB radiation. Radiat. Res 2001, 156, 36–44. [Google Scholar] [CrossRef]
- Black, H.S.; Okotie-Eboh, G.; Gerguis, J.; Urban, J.I.; Thornby, J.I. Dietary fat modulates immunoresponsiveness in UV-irradiated mice. Photochem. Photobiol. 1995, 62, 964–969. [Google Scholar] [CrossRef] [PubMed]
- Black, H.S.; Thornby, J.I.; Wolf, J.E.; Goldberg, L.H.; Herd, J.A.; Rosen, T.; Bruce, S.; Tschen, J.A.; Scott, L.W.; Jaax, S.; et al. Evidence that a low-fat diet reduces the occurrence of non-melanoma skin cancer. Int. J. Cancer 1995, 62, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Jaax, S.; Scott, L.W.; Wolf, J.E.; Thornby, J.I.; Black, H.S. General guidelines for a low-fat diet effective in the management and prevention of nonmelanoma skin cancer. Nutr. Cancer 1997, 27, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Black, H.S.; Herd, J.A.; Goldberg, L.H.; Wolf, J.E.; Thornby, J.I.; Rosen, T.; Bruce, S.; Tschen, J.A.; Foreyt, J.P.; Scott, L.W.; et al. Effect of a low-fat diet on the incidence of actinic keratosis. N. Engl. J. Med. 1994, 330, 1272–1275. [Google Scholar] [CrossRef] [PubMed]
- Hakim, I.A.; Harris, R.B.; Ritenbaugh, C. Fat intake and risk of squamous cell carcinomas of the skin. Nutr. Cancer 2000, 36, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Orengo, I.F.; Black, H.S.; Wolf, J.E. Influence of fish oil supplementation on the minimal erythema dose in humans. Arch. Dermatol. 1992, 284, 219–221. [Google Scholar] [CrossRef]
- Rhodes, L.E.; O’Farrell, S.; Jackson, M.J.; Friedmann, P.S. Dietary fish-oil supplementation in humans reduces UVB-erythemal sensitivity but increases epidermal lipid peroxidation. J. Investig. Dermatol. 1994, 103, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Pupe, A.; Moison, R.; De Haes, P.; Beijersbergen van Henegouwen, G.; Rhodes, L.E.; Degreef, H.; Garmyn, M. Eicosapentaenoic acid, a n-3 polyunsaturated fatty acid differentially modulates TNF-α, IL-1α, IL-6 and PGE2 expression in UVB-irradiated human keratinocytes. J. Investig. Dermatol. 2002, 118, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Storey, A.; McArdle, F.; Friedmann, P.S.; Jackson, M.J.; Rhodes, L.E. Eicosapentaenoic acid and docosahexaenoic acid reduce UVB- and TNF-alpha-induced IL-8 secretion in keratinocytes and UVB-induced IL-8 in fibroblasts. J. Investig. Dermatol. 2005, 124, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Shahbakhti, H.; Watson, R.; Azurdia, R.; Ferreira, R.; Garmyn, M.; Rhodes, L.E. Influence of eicosapentaenoic acid, an omega-3 fatty acid, on ultraviolet B generation of prostaglandin E2 and proinflammatory cytokines interleukin-1 beta, tumor necrosis factor-alpha, interleukin-6 and interleukin-8 in human skin in vivo. Photochem. Photobiol. 2004, 80, 231–235. [Google Scholar] [CrossRef] [PubMed]
- Pilkington, S.M.; Rhodes, L.E.; Al-Aasswad, N.M.I.; Massey, K.A.; Nicolaou, A. Impact of EPA ingestion on COX- and LOX-mediated eicosanoid synthesis in skin with and without a pro- inflammatory UVR challenge. Report of a randomised controlled study in humans. Mol. Nutr. Food Res. 2014, 58, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Pilkington, S.M.; Massey, K.A.; Bennett, S.P.; Al-Aasswad, N.M.; Roshdy, K.; Gibbs, N.K.; Friedmann, P.S.; Nicolaou, A.; Rhodes, L.E. Randomized controlled trial of oral omega-3 PUFA in solar-simulated radiation-induced suppression of human cutaneous immune responses. Am. J. Clin. Nutr. 2013, 97, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.E.; Shahbakhti, H.; Azurdia, R.; Moison, R.; Steenwinkel, M.-J.S.T.; Homburg, M.; Dean, M.; McArdle, F.; Beijersbergen van Henegouwen, G.; Epe, B.; et al. Effect of eicosapentaenoic acid, an omega-3 polyunsaturated fatty acid, on UVR-related cancer risk in humans. An assessment of early genotoxic markers. Carcinogenesis 2003, 24, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Pilkington, S.M.; Watson, R.E.B.; Nicolaou, A.; Rhodes, L.E. Omega-3 Polyunsaturated Fatty Acids: Photoprotective Macronutrients. Exp. Dermatol. 2011, 20, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Rhodes, L.E. Preventive oncology. Lancet 2004, 363, 1736–1737. [Google Scholar] [CrossRef]
- Noel, S.E.; Stoneham, A.C.S.; Olsen, C.M.; Rhodes, L.E.; Green, A.C. Consumption of omega-3 fatty acids and the risk of skin cancers: A systemic review and meta-analysis. Int. J. Cancer 2014, 135, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Lyon, J.Y.; Gardner, J.W.; West, D.W.; Mahoney, A.M. Methodological issues in epidemiological studies of the diet and cancer. Cancer Res. 1992, 52, 2040–2048. [Google Scholar]
- Black, H.S. Skin Cancer. In Nutritional Oncology, 2nd ed.; Heber, D., Blackburn, G.L., Go, V.L.W., Eds.; Academic Press: San Diego, CA, USA, 1999; pp. 405–422. [Google Scholar]
- Wallingford, S.C.; Pilkington, S.M.; Massey, K.A.; Al-Aasswad, N.M.I.; Ibiebele, T.I.; Hughes, M.C.; Bennett, S.; Nicolaou, A.; Rhodes, L.E.; Green, A.C. Three-way assessment of long chain omega-3 polyunsaturated fatty acid nutrition: By questionnaire and matched blood and skin samples. Br. J. Nutr. 2013, 109, 701–708. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Black, H.S.; Rhodes, L.E. Potential Benefits of Omega-3 Fatty Acids in Non-Melanoma Skin Cancer. J. Clin. Med. 2016, 5, 23. https://doi.org/10.3390/jcm5020023
Black HS, Rhodes LE. Potential Benefits of Omega-3 Fatty Acids in Non-Melanoma Skin Cancer. Journal of Clinical Medicine. 2016; 5(2):23. https://doi.org/10.3390/jcm5020023
Chicago/Turabian StyleBlack, Homer S., and Lesley E. Rhodes. 2016. "Potential Benefits of Omega-3 Fatty Acids in Non-Melanoma Skin Cancer" Journal of Clinical Medicine 5, no. 2: 23. https://doi.org/10.3390/jcm5020023
APA StyleBlack, H. S., & Rhodes, L. E. (2016). Potential Benefits of Omega-3 Fatty Acids in Non-Melanoma Skin Cancer. Journal of Clinical Medicine, 5(2), 23. https://doi.org/10.3390/jcm5020023






