Beyond Traditional Risk Factors: Integrating Epicardial Adipose Tissue into the Comorbidity Landscape of HFpEF
Abstract
1. Introduction
2. Methods
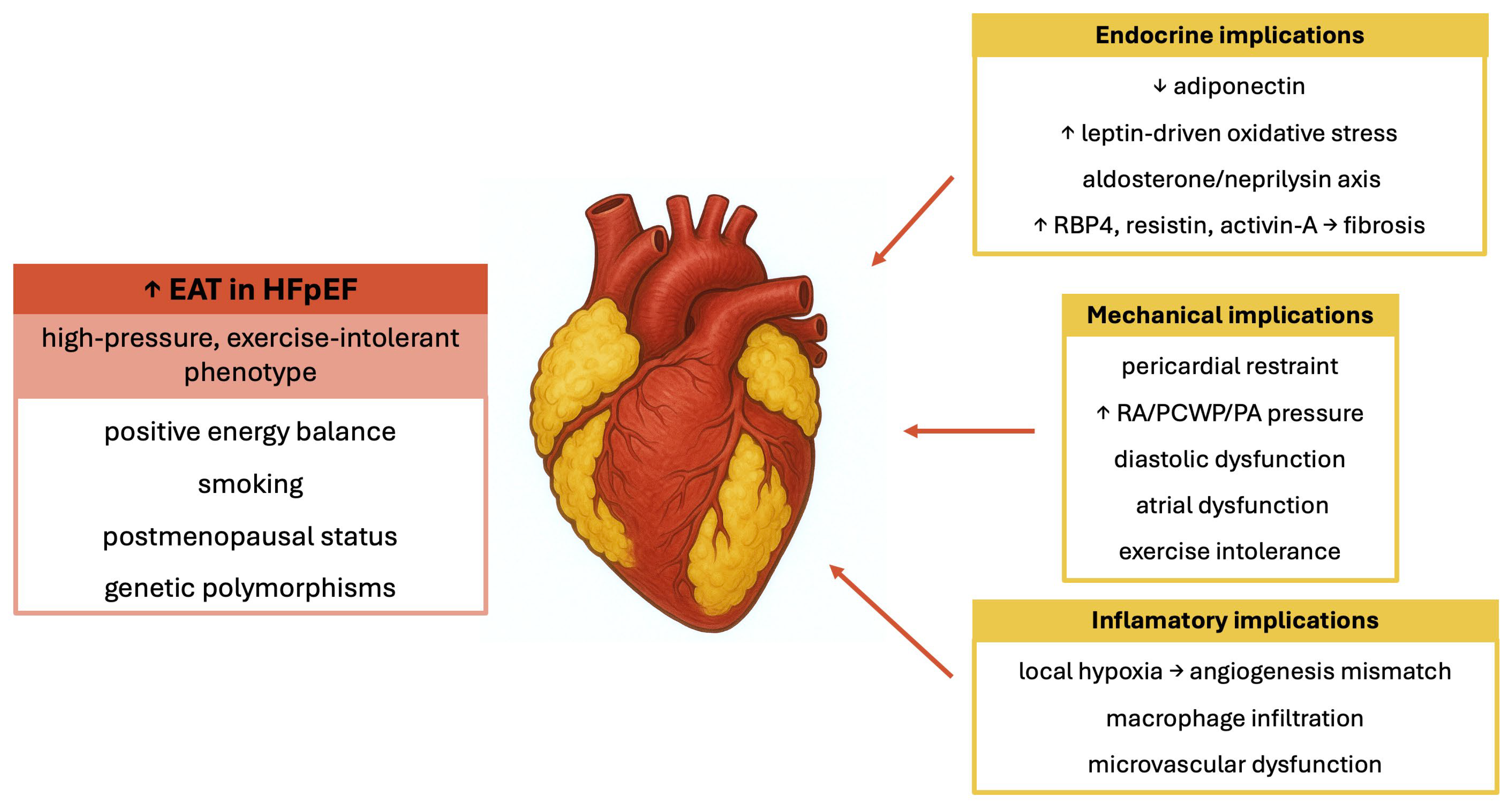
3. EAT in HFpEF vs. HFrEF
4. Pathophysiology of EAT in HFpEF
4.1. Mechanical Effects of EAT
4.2. Metabolic and Endocrine Effects of EAT
5. Imaging-Based Evaluation of EAT
5.1. Transthotacic Echocardiography
5.2. Cardiac Magnetic Resonance Imaging
5.3. Cardiac Computed Tomography
6. Therapeutic Strategies Targeting EAT
6.1. Non-Pharmacological Interventions
6.2. Lipid-Lowering Therapies
6.3. Anti-Inflammatory Therapies
6.4. Anti-Hyperglycaemic Therapies
| Non-Pharmacological Interventions | Pharmacological Interventions | |||
|---|---|---|---|---|
| Lifestyle Changes | Bariatric Surgery | Lipid-Lowering | Anti-Inflammatory | Anti-Hyperglycaemic |
| Caloric restriction + aerobic exercise | Improves [10]:
| |||
7. Future Directions in EAT Research
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| EAT | Epicardial adipose tissue |
| HFpEF | Heart failure with preserved ejection fraction |
| HF | Heart failure |
| SGLT2 | Sodium–glucose cotransporter 2 |
| CT | Computed tomography |
| MRI | Magnetic resonance imaging |
| NT-proBNP | N-terminal pro-B-type natriuretic peptide |
| GLP-1 | Glucagon-like peptide-1 |
| HFrEF | Heart failure with reduced ejection fraction |
| LVEDV | Left ventricular end-diastolic volume |
| VAT | Visceral adipose tissue |
| UCP1 | Uncoupling Protein 1 |
| ATP | Adenosine triphosphate |
| RA | Right atrium |
| PCWP | Pulmonary capillary wedge pressure |
| PA | Pulmonary artery |
| BMI | Body mass index |
| RV | Right ventricle |
| LA | Left atrium |
| CMR | Cardiac magnetic resonance |
| PKC | Protein Kinase C |
| NADPH | Nicotinamide Adenine Dinucleotide Phosphate |
| ROS | Reactive oxygen species |
| PPAR-γ | Peroxisome proliferator-activated receptor gamma |
| SAT | Subcutaneous adipose tissue |
| RBP4 | Retinol-binding protein 4 |
| TLR-4 | Toll-like receptor 4 |
| NF-κB | Nuclear Factor kappa B |
| ICD | Implantable cardioverter–defibrillator |
| cCT | Cardiac-gated computed tomography |
| FAI | Fat attenuation index |
| CCTA | Coronary CT angiography |
| PCAT | Pericoronary adipose tissue |
| PAAT | Periatrial adipose tissue |
| TNF-α | Tumor Necrosis Factor alpha |
| PCSK9 | Pro-protein convertase subtilisin/kexin type-9 |
| GIP | Glucose-dependent insulinotropic polypeptide |
| DPP-4 | Dipeptidyl peptidase-4 |
| CRP | C-reactive protein |
| GCG | Glucagon gene |
| ASCs | Adipose-derived mesenchymal stem cells |
References
- Goldman, S.A.; Requena-Ibanez, J.A.; Devesa, A.; Santos-Gallego, C.G.; Badimon, J.J.; Fuster, V. Uncovering the Role of Epicardial Adipose Tissue in Heart Failure With Preserved Ejection Fraction. JACC Adv. 2023, 2, 100657. [Google Scholar] [CrossRef] [PubMed]
- van Woerden, G.; van Veldhuisen, D.J.; Westenbrink, B.D.; de Boer, R.A.; Rienstra, M.; Gorter, T.M. Connecting epicardial adipose tissue and heart failure with preserved ejection fraction: Mechanisms, management and modern perspectives. Eur. J. Heart Fail. 2022, 24, 2238–2250. [Google Scholar] [CrossRef]
- Whitman, J.; Kozaily, E.; Michos, E.D.; Silverman, D.N.; Fudim, M.; Mentz, R.J.; Tedford, R.J.; Rao, V.N. Epicardial Fat in Heart Failure and Preserved Ejection Fraction: Novel Insights and Future Perspectives. Curr. Heart Fail. Rep. 2025, 22, 13. [Google Scholar] [CrossRef]
- Borruel, S.; Moltó, J.F.; Alpañés, M.; Fernández-Durán, E.; Álvarez-Blasco, F.; Luque-Ramírez, M.; Escobar-Morreale, H.F. Surrogate markers of visceral adiposity in young adults: Waist circumference and body mass index are more accurate than waist hip ratio, model of adipose distribution and visceral adiposity index. PLoS ONE 2014, 9, e114112. [Google Scholar] [CrossRef]
- Abusnina, W.; Merdler, I.; Cellamare, M.; Chitturi, K.R.; Chaturvedi, A.; Feuerstein, I.M.; Zhang, C.; Ozturk, S.T.; Deksissa, T.; Sawant, V.; et al. Epicardial Fat Tissue: A Potential Marker for Coronary Microvascular Dysfunction. J. Am. Heart Assoc. 2025, 14, e038484. [Google Scholar] [CrossRef]
- Gorter, T.M.; van Woerden, G.; Rienstra, M.; Dickinson, M.G.; Hummel, Y.M.; Voors, A.A.; Hoendermis, E.S.; van Veldhuisen, D.J. Epicardial Adipose Tissue and Invasive Hemodynamics in Heart Failure wth Preserved Ejection Fraction. JACC Heart Fail. 2020, 8, 667–676. [Google Scholar] [CrossRef]
- Fu, Z.; Wang, Y.; Wang, Y.; Shi, S.; Li, Y.; Zhang, B.; Wu, H.; Song, Q. Linking abnormal fat distribution with HFpEF and diastolic dysfunction: A systematic review, meta-analysis, and meta-regression of observational studies. Lipids Health Dis. 2024, 23, 277. [Google Scholar] [CrossRef] [PubMed]
- Chong, B.; Jayabaskaran, J.; Ruban, J.; Goh, R.; Chin, Y.H.; Kong, G.; Ng, C.H.; Lin, C.; Loong, S.; Muthiah, M.D.; et al. Epicardial Adipose Tissue Assessed by Computed Tomography and Echocardiography Are Associated With Adverse Cardiovascular Outcomes: A Systematic Review and Meta-Analysis. Circ. Cardiovasc. Imaging 2023, 16, e015159. [Google Scholar] [CrossRef]
- Wang, W.; Gao, Y.; Wang, J.; Ji, C.; Gu, H.; Yuan, X.; Yang, S.; Wang, X. Prognostic Value of Epicardial Adipose Tissue in Heart Failure With Mid-Range and Preserved Ejection Fraction: A Multicenter Study. J. Am. Heart Assoc. 2024, 13, e036789. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.J.; Dong, G.J.; Guo, M. Targeting epicardial adipose tissue: A potential therapeutic strategy for heart failure with preserved ejection fraction with type 2 diabetes mellitus. World J. Diabetes 2023, 14, 724–740. [Google Scholar] [CrossRef]
- Packer, M.; Lam, C.S.P.; Lund, L.H.; Maurer, M.S.; Borlaug, B.A. Characterization of the inflammatory-metabolic phenotype of heart failure with a preserved ejection fraction: A hypothesis to explain influence of sex on the evolution and potential treatment of the disease. Eur. J. Heart Fail. 2020, 22, 1551–1567. [Google Scholar] [CrossRef]
- Rossi, V.A.; Nebunu, D.; Haider, T.; Laptseva, N.; Naegele, M.P.; Ruschitzka, F.; Sudano, I.; Flammer, A.J. Diverging role of epicardial adipose tissue across the entire heart failure spectrum. ESC Heart Fail. 2023, 10, 3419–3429. [Google Scholar] [CrossRef]
- Cundari, G.; Marchitelli, L.; Pambianchi, G.; Catapano, F.; Conia, L.; Stancanelli, G.; Catalano, C.; Galea, N. Imaging biomarkers in cardiac CT: Moving beyond simple coronary anatomical assessment. Radiol. Med. 2024, 129, 380–400. [Google Scholar] [CrossRef]
- Pandey, A.K.; Bhatt, D.L.; Pandey, A.; Marx, N.; Cosentino, F.; Pandey, A.; Verma, S. Mechanisms of benefits of sodium-glucose cotransporter 2 inhibitors in heart failure with preserved ejection fraction. Eur. Heart J. 2023, 44, 3640–3651. [Google Scholar] [CrossRef]
- Doesch, C.; Haghi, D.; Flüchter, S.; Suselbeck, T.; Schoenberg, S.O.; Michaely, H.; Borggrefe, M.; Papavassiliu, T. Epicardial adipose tissue in patients with heart failure. J. Cardiovasc. Magn. Reson. 2010, 12, 40. [Google Scholar] [CrossRef]
- Obokata, M.; Reddy, Y.N.V.; Pislaru, S.V.; Melenovsky, V.; Borlaug, B.A. Evidence Supporting the Existence of a Distinct Obese Phenotype of Heart Failure with Preserved Ejection Fraction. Circulation 2017, 136, 6–19. [Google Scholar] [CrossRef] [PubMed]
- Guan, B.; Liu, L.; Li, X.; Huang, X.; Yang, W.; Sun, S.; Ma, Y.; Yu, Y.; Luo, J.; Cao, J. Association between epicardial adipose tissue and blood pressure: A systematic review and meta-analysis. Nutr. Metab. Cardiovasc. Dis. 2021, 31, 2547–2556. [Google Scholar] [CrossRef] [PubMed]
- Song, D.K.; Hong, Y.S.; Lee, H.; Oh, J.Y.; Sung, Y.A.; Kim, Y. Increased Epicardial Adipose Tissue Thickness in Type 2 Diabetes Mellitus and Obesity. Diabetes Metab. J. 2015, 39, 405–413. [Google Scholar] [CrossRef]
- Nesti, L.; Pugliese, N.R.; Chiriacò, M.; Trico, D.; Baldi, S.; Natali, A. Epicardial adipose tissue thickness is associated with reduced peak oxygen consumption and systolic reserve in patients with type 2 diabetes and normal heart function. Diabetes Obes. Metab. 2023, 25, 177–188. [Google Scholar] [CrossRef]
- Couselo-Seijas, M.; Rodríguez-Mañero, M.; González-Juanatey, J.R.; Eiras, S. Updates on epicardial adipose tissue mechanisms on atrial fibrillation. Obes. Rev. 2021, 22, e13277. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, G.S.; Rabkin, S.W. Epicardial Fat in Heart Failure with Preserved Ejection Fraction Compared with Reduced Ejection Fraction. J. Clin. Med. 2024, 13, 5533. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Hung, C.L.; Tay, W.T.; Soon, D.; Sim, D.; Sung, K.T.; Loh, S.Y.; Lee, S.; Jaufeerally, F.; Ling, L.H.; et al. Epicardial adipose tissue related to left atrial and ventricular function in heart failure with preserved versus reduced and mildly reduced ejection fraction. Eur. J. Heart Fail. 2022, 24, 1346–1356. [Google Scholar] [CrossRef]
- Pugliese, N.R.; Paneni, F.; Mazzola, M.; De Biase, N.; Del Punta, L.; Gargani, L.; Mengozzi, A.; Virdis, A.; Nesti, L.; Taddei, S.; et al. Impact of epicardial adipose tissue on cardiovascular haemodynamics, metabolic profile, and prognosis in heart failure. Eur. J. Heart. Fail. 2021, 23, 1858–1871. [Google Scholar] [CrossRef]
- Cho, D.H.; Park, S.M. Epicardial Adipose Tissue and Heart Failure, Friend or Foe? Diabetes Metab. J. 2024, 48, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Van Bortel, L.M.; Laurent, S.; Boutouyrie, P.; Chowienczyk, P.; Cruickshank, J.K.; De Backer, T.; Filipovsky, J.; Huybrechts, S.; Mattace-Raso, F.U.; Protogerou, A.D.; et al. Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J. Hypertens. 2012, 30, 445–448. [Google Scholar] [CrossRef]
- Lin, J.L.; Sung, K.T.; Lai, Y.H.; Yen, C.H.; Yun, C.H.; Su, C.H.; Kuo, J.Y.; Liu, C.Y.; Chien, C.Y.; Cury, R.C.; et al. Epicardial Adiposity in Relation to Metabolic Abnormality, Circulating Adipocyte FABP, and Preserved Ejection Fraction Heart Failure. Diagnostics 2021, 11, 397. [Google Scholar] [CrossRef] [PubMed]
- van Woerden, G.; van Veldhuisen, D.J.; Manintveld, O.C.; van Empel, V.P.M.; Willems, T.P.; de Boer, R.A.; Rienstra, M.; Westenbrink, B.D.; Gorter, T.M. Epicardial Adipose Tissue and Outcome in Heart Failure With Mid-Range and Preserved Ejection Fraction. Circ. Heart Fail. 2022, 15, e009238. [Google Scholar] [CrossRef]
- Ebong, I.A.; Goff DCJr Rodriguez, C.J.; Chen, H.; Bertoni, A.G. Mechanisms of heart failure in obesity. Obes Res Clin Pract. 2014, 8, e540-8. [Google Scholar] [CrossRef]
- Lembo, M.; Strisciuglio, T.; Fonderico, C.; Mancusi, C.; Izzo, R.; Trimarco, V.; Bellis, A.; Barbato, E.; Esposito, G.; Morisco, C.; et al. Obesity: The perfect storm for heart failure. ESC Heart Fail. 2024, 11, 1841–1860. [Google Scholar] [CrossRef]
- Van Gaal, L.F.; Maggioni, A.P. Overweight, obesity, and outcomes: Fat mass and beyond. Lancet 2014, 383, 935–936. [Google Scholar] [CrossRef]
- Savji, N.; Meijers, W.C.; Bartz, T.M.; Bhambhani, V.; Cushman, M.; Nayor, M.; Kizer, J.R.; Sarma, A.; Blaha, M.J.; Gansevoort, R.T.; et al. The Association of Obesity and Cardiometabolic Traits With Incident HFpEF and HFrEF. JACC Heart Fail. 2018, 6, 701–709. [Google Scholar] [CrossRef]
- Haregu, T.N.; Nanayakkara, S.; Carrington, M.; Kaye, D. Prevalence and correlates of normal body mass index central obesity among people with cardiovascular diseases in Australia. Public Health 2020, 183, 126–131. [Google Scholar] [CrossRef]
- Hassan, S.; Oladele, C.; Galusha, D.; Adams, O.P.; Maharaj, R.G.; Nazario, C.M.; Nunez, M.; Nunez-Smith, M.; ECHORN Writing Group. Anthropometric measures of obesity and associated cardiovascular disease risk in the Eastern Caribbean Health Outcomes Research Network (ECHORN) Cohort Study. BMC Public Health 2021, 21, 399. [Google Scholar] [CrossRef] [PubMed]
- Nooyens, A.C.; Koppes, L.L.; Visscher, T.L.; Twisk, J.W.; Kemper, H.C.; Schuit, A.J.; van Mechelen, W.; Seidell, J.C. Adolescent skinfold thickness is a better predictor of high body fatness in adults than is body mass index: The Amsterdam Growth and Health Longitudinal Study. Am. J. Clin. Nutr. 2007, 85, 1533–1539. [Google Scholar] [CrossRef]
- Iacobellis, G.; Bianco, A.C. Epicardial adipose tissue: Emerging physiological, pathophysiological and clinical features. Trends Endocrinol. Metab. 2011, 22, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, G.; Corradi, D.; Sharma, A.M. Epicardial adipose tissue: Anatomic, biomolecular and clinical relationships with the heart. Nat. Clin. Pract. Cardiovasc. Med. 2005, 2, 536–543. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, G. Epicardial and pericardial fat: Close, but very different. Obesity 2009, 17, 625; author reply 626-7. [Google Scholar] [CrossRef]
- Iacobellis, G.; Malavazos, A.E. Pericardial adipose tissue, atherosclerosis, and cardiovascular disease risk factors: The Jackson Heart Study: Comment on Liu et Al. Diabetes Care. 2010, 33, e127; author reply e128. [Google Scholar] [CrossRef]
- Rabkin, S.W. Epicardial fat: Properties, function and relationship to obesity. Obes. Rev. 2007, 8, 253–261. [Google Scholar] [CrossRef]
- Fedorenko, A.; Lishko, P.V.; Kirichok, Y. Mechanism of fatty-acid-dependent UCP1 uncoupling in brown fat mitochondria. Cell 2012, 151, 400–413. [Google Scholar] [CrossRef]
- Song, Y.; Tan, Y.; Deng, M.; Shan, W.; Zheng, W.; Zhang, B.; Cui, J.; Feng, L.; Shi, L.; Zhang, M.; et al. Epicardial adipose tissue, metabolic disorders, and cardiovascular diseases: Recent advances classified by research methodologies. MedComm 2023, 4, e413. [Google Scholar] [CrossRef]
- Lopaschuk, G.D.; Ussher, J.R. Evolving Concepts of Myocardial Energy Metabolism: More Than Just Fats and Carbohydrates. Circ. Res. 2016, 119, 1173–1176. [Google Scholar] [CrossRef]
- Pezeshkian, M.; Noori, M.; Najjarpour-Jabbari, H.; Abolfathi, A.; Darabi, M.; Darabi, M.; Shaaker, M.; Shahmohammadi, G. Fatty acid composition of epicardial and subcutaneous human adipose tissue. Metab. Syndr. Relat. Disord. 2009, 7, 125–131. [Google Scholar] [CrossRef]
- Zatterale, F.; Longo, M.; Naderi, J.; Raciti, G.A.; Desiderio, A.; Miele, C.; Beguinot, F. Chronic Adipose Tissue Inflammation Linking Obesity to Insulin Resistance and Type 2 Diabetes. Front. Physiol. 2020, 10, 1607. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, K.A.; Lönnroth, P.; Parkkola, R.; Peltoniemi, P.; Asola, M.; Viljanen, T.; Tolvanen, T.; Knuuti, J.; Rönnemaa, T.; Huupponen, R.; et al. Glucose uptake and perfusion in subcutaneous and visceral adipose tissue during insulin stimulation in nonobese and obese humans. J. Clin. Endocrinol. Metab. 2002, 87, 3902–3910. [Google Scholar] [CrossRef]
- Pasarica, M.; Sereda, O.R.; Redman, L.M.; Albarado, D.C.; Hymel, D.T.; Roan, L.E.; Rood, J.C.; Burk, D.H.; Smith, S.R. Reduced adipose tissue oxygenation in human obesity: Evidence for rarefaction, macrophage chemotaxis, and inflammation without an angiogenic response. Diabetes 2009, 58, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Koepp, K.E.; Obokata, M.; Reddy, Y.N.V.; Olson, T.P.; Borlaug, B.A. Hemodynamic and Functional Impact of Epicardial Adipose Tissue in Heart Failure With Preserved Ejection Fraction. JACC Heart Fail. 2020, 8, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Crum, Y.; Hoendermis, E.S.; van Veldhuisen, D.J.; van Woerden, G.; Lobeek, M.; Dickinson, M.G.; Meems, L.M.; Voors, A.A.; Rienstra, M.; Gorter, T.M. Epicardial adipose tissue and pericardial constraint in heart failure with preserved ejection fraction. ESC Heart Fail. 2024, 11, 1698–1706. [Google Scholar] [CrossRef]
- Lobeek, M.; Gorter, T.M.; Westenbrink, B.D.; Van Veldhuisen, D.J.; Rienstra, M. Increased epicardial adipose tissue is associated with left atrial mechanical dysfunction in patients with heart failure with mildly reduced and preserved ejection fraction. Clin. Res. Cardiol. 2025, 114, 601–608. [Google Scholar] [CrossRef]
- Haykowsky, M.J.; Kouba, E.J.; Brubaker, P.H.; Nicklas, B.J.; Eggebeen, J.; Kitzman, D.W. Skeletal muscle composition and its relation to exercise intolerance in older patients with heart failure and preserved ejection fraction. Am. J. Cardiol. 2014, 113, 1211–1216. [Google Scholar] [CrossRef]
- Lima-Martínez, M.M.; Blandenier, C.; Iacobellis, G. Tejido adiposo epicárdico: ¿más que un simple depósito de grasa? [Epicardial adipose tissue: More than a simple fat deposit?]. Endocrinol Nutr. 2013, 60, 320–328. (In Spanish) [Google Scholar] [CrossRef]
- Chen, H.; Liu, L.; Li, M.; Zhu, D.; Tian, G. Epicardial Adipose Tissue-Derived Leptin Promotes Myocardial Injury in Metabolic Syndrome Rats Through PKC/NADPH Oxidase/ROS Pathway. J. Am. Heart Assoc. 2023, 12, e029415. [Google Scholar] [CrossRef] [PubMed]
- Ansaldo, A.M.; Montecucco, F.; Sahebkar, A.; Dallegri, F.; Carbone, F. Epicardial adipose tissue and cardiovascular diseases. Int J Cardiol. 2019, 278, 254–260. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Wang, H.; Zhang, L.; Cao, Y.; Bao, J.Z.; Liu, Z.X.; Wang, L.S.; Yang, Q.; Lu, X. Retinol-Binding Protein 4 Induces Cardiomyocyte Hypertrophy by Activating TLR4/MyD88 Pathway. Endocrinology 2016, 157, 2282–2293. [Google Scholar] [CrossRef]
- Kawanami, D.; Maemura, K.; Takeda, N.; Harada, T.; Nojiri, T.; Imai, Y.; Manabe, I.; Utsunomiya, K.; Nagai, R. Direct reciprocal effects of resistin and adiponectin on vascular endothelial cells: A new insight into adipocytokine-endothelial cell interactions. Biochem. Biophys. Res. Commun. 2004, 314, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Greulich, S.; Maxhera, B.; Vandenplas, G.; de Wiza, D.H.; Smiris, K.; Mueller, H.; Heinrichs, J.; Blumensatt, M.; Cuvelier, C.; Akhyari, P.; et al. Secretory products from epicardial adipose tissue of patients with type 2 diabetes mellitus induce cardiomyocyte dysfunction. Circulation 2012, 126, 2324–2334. [Google Scholar] [CrossRef]
- Ying, H.; Guo, W.; Tang, X.; Pan, J.; Yu, P.; Fan, H.; Wang, X.; Jiang, R.; Jiang, C.; Liang, P. Colchicine attenuates the electrical remodeling of post-operative atrial fibrillation through inhibited expression of immune-related hub genes and stabilization of microtubules. Int. J. Biol. Sci. 2023, 19, 2934–2956. [Google Scholar] [CrossRef]
- Borlaug, B.A.; Reddy, Y.N.V. The Role of the Pericardium in Heart Failure: Implications for Pathophysiology and Treatment. JACC Heart Fail. 2019, 7, 574–585. [Google Scholar] [CrossRef]
- Tedford, R.J.; Houston, B.A. HFpEF, Obesity, and Epicardial Adipose Tissue: Don’t Have Your Cake and EAT It, Too. JACC Heart Fail. 2020, 8, 677–680. [Google Scholar] [CrossRef]
- Welch, T.D. Constrictive pericarditis: Diagnosis, management and clinical outcomes. Heart 2018, 104, 725–731. [Google Scholar] [CrossRef]
- Choi, J.H.; Choi, J.O.; Ryu, D.R.; Lee, S.C.; Park, S.W.; Choe, Y.H.; Oh, J.K. Mitral and tricuspid annular velocities in constrictive pericarditis and restrictive cardiomyopathy: Correlation with pericardial thickness on computed tomography. JACC Cardiovasc. Imaging 2011, 4, 567–575. [Google Scholar] [CrossRef]
- Gaeta, M.; Bandera, F.; Tassinari, F.; Capasso, L.; Cargnelutti, M.; Pelissero, G.; Malavazos, A.E.; Ricci, C. Is epicardial fat depot associated with atrial fibrillation? A systematic review and meta-analysis. Europace 2017, 19, 747–752. [Google Scholar] [CrossRef]
- Cacciapuoti, F.; Caso, I.; Crispo, S.; Verde, N.; Capone, V.; Gottilla, R.; Materazzi, C.; Volpicelli, M.; Ziviello, F.; Mauro, C.; et al. Linking Epicardial Adipose Tissue to Atrial Remodeling: Clinical Implications of Strain Imaging. Hearts 2025, 6, 3. [Google Scholar] [CrossRef]
- Patel, K.H.K.; Hwang, T.; Se Liebers, C.; Ng, F.S. Epicardial adipose tissue as a mediator of cardiac arrhythmias. Am. J. Physiol. Heart Circ. Physiol. 2022, 322, H129–H144. [Google Scholar] [CrossRef]
- Lau, D.H.; Linz, D.; Sanders, P. New Findings in Atrial Fibrillation Mechanisms. Card. Electrophysiol. Clin. 2019, 11, 563–571. [Google Scholar] [CrossRef]
- Zhen, X.W.; Li, W.C.; Wang, H.; Song, N.P.; Zhong, L. Does types of atrial fibrillation matter in the impairment of global and regional left ventricular mechanics and intra-ventricular dyssynchrony? Front. Cardiovasc. Med. 2022, 9, 1019472. [Google Scholar] [CrossRef]
- Janovska, P.; Melenovsky, V.; Svobodova, M.; Havlenova, T.; Kratochvilova, H.; Haluzik, M.; Hoskova, E.; Pelikanova, T.; Kautzner, J.; Monzo, L.; et al. Dysregulation of epicardial adipose tissue in cachexia due to heart failure: The role of natriuretic peptides and cardiolipin. J. Cachexia Sarcopenia Muscle 2020, 11, 1614–1627. [Google Scholar] [CrossRef] [PubMed]
- Janssen-Telders, C.; Eringa, E.C.; de Groot, J.R.; de Man, F.S.; Handoko, M.L. The role of epicardial adipose tissue remodelling in heart failure with preserved ejection fraction. Cardiovasc. Res. 2025, 121, 860–870. [Google Scholar] [CrossRef] [PubMed]
- Schiattarella, G.G.; Alcaide, P.; Condorelli, G.; Gillette, T.G.; Heymans, S.; Jones, E.A.V.; Kallikourdis, M.; Lichtman, A.; Marelli-Berg, F.; Shah, S.J.; et al. Immunometabolic Mechanisms of Heart Failure with Preserved Ejection Fraction. Nat. Cardiovasc. Res. 2022, 1, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Guo, Z.; Wang, P.; Zheng, M.; Yang, X.; Liu, Y.; Ma, Z.; Chen, M.; Yang, X. Proteomics of epicardial adipose tissue in patients with heart failure. J. Cell Mol. Med. 2020, 24, 511–520. [Google Scholar] [CrossRef]
- Agra, R.M.; Teijeira-Fernández, E.; Pascual-Figal, D.; Sánchez-Más, J.; Fernández-Trasancos, A.; González-Juanatey, J.R.; Eiras, S. Adiponectin and p53 mRNA in epicardial and subcutaneous fat from heart failure patients. Eur. J. Clin. Investig. 2014, 44, 29–37. [Google Scholar] [CrossRef]
- Gaborit, B.; Venteclef, N.; Ancel, P.; Pelloux, V.; Gariboldi, V.; Leprince, P.; Amour, J.; Hatem, S.N.; Jouve, E.; Dutour, A.; et al. Human epicardial adipose tissue has a specific transcriptomic signature depending on its anatomical peri-atrial, peri-ventricular, or peri-coronary location. Cardiovasc. Res. 2015, 108, 62–73. [Google Scholar] [CrossRef]
- Miroshnikova, V.V.; Polyakova, E.A.; Pobozheva, I.A.; Panteleeva, A.A.; Razgildina, N.D.; Kolodina, D.A.; Belyaeva, O.D.; Berkovich, O.A.; Pchelina, S.N.; Baranova, E.I. FABP4 and omentin-1 gene expression in epicardial adipose tissue from coronary artery disease patients. Genet. Mol. Biol. 2021, 44, e20200441. [Google Scholar] [CrossRef]
- Wang, Q.; Yu, Y.; Zhang, P.; Chen, Y.; Li, C.; Chen, J.; Wang, Y.; Li, Y. The crucial role of activin A/ALK4 pathway in the pathogenesis of Ang-II-induced atrial fibrosis and vulnerability to atrial fibrillation. Basic. Res. Cardiol. 2017, 112, 47. [Google Scholar] [CrossRef] [PubMed]
- Iacobellis, G.; Assael, F.; Ribaudo, M.C.; Zappaterreno, A.; Alessi, G.; Di Mario, U.; Leonetti, F. Epicardial fat from echocardiography: A new method for visceral adipose tissue prediction. Obes. Res. 2003, 11, 304–310. [Google Scholar] [CrossRef]
- Iacobellis, G.; Willens, H.J. Echocardiographic epicardial fat: A review of research and clinical applications. J. Am. Soc. Echocardiogr. 2009, 22, 1311–1319; 1417-8. [Google Scholar] [CrossRef]
- Iacobellis, G.; Willens, H.J.; Barbaro, G.; Sharma, A.M. Threshold values of high-risk echocardiographic epicardial fat thickness. Obesity 2008, 16, 887–892. [Google Scholar] [CrossRef] [PubMed]
- Saura, D.; Oliva, M.J.; Rodríguez, D.; Pascual-Figal, D.A.; Hurtado, J.A.; Pinar, E.; de la Morena, G.; Valdés, M. Reproducibility of echocardiographic measurements of epicardial fat thickness. Int. J. Cardiol. 2010, 141, 311–313. [Google Scholar] [CrossRef]
- Jeong, J.W.; Jeong, M.H.; Yun, K.H.; Oh, S.K.; Park, E.M.; Kim, Y.K.; Rhee, S.J.; Lee, E.M.; Lee, J.; Yoo, N.J.; et al. Echocardiographic epicardial fat thickness and coronary artery disease. Circ. J. 2007, 71, 536–539. [Google Scholar] [CrossRef] [PubMed]
- Parisi, V.; Conte, M.; Petraglia, L.; Grieco, F.V.; Bruzzese, D.; Caruso, A.; Grimaldi, M.G.; Campana, P.; Gargiulo, P.; Paolillo, S.; et al. Echocardiographic Epicardial Adipose Tissue Thickness for Risk Stratification of Patients With Heart Failure. Front. Physiol. 2020, 11, 43. [Google Scholar] [CrossRef]
- Parisi, V.; Petraglia, L.; Formisano, R.; Caruso, A.; Grimaldi, M.G.; Bruzzese, D.; Grieco, F.V.; Conte, M.; Paolillo, S.; Scatteia, A.; et al. Validation of the echocardiographic assessment of epicardial adipose tissue thickness at the Rindfleisch fold for the prediction of coronary artery disease. Nutr. Metab. Cardiovasc. Dis. 2020, 30, 99–105. [Google Scholar] [CrossRef]
- Carerj, M.L.; Restelli, D.; Poleggi, C.; Di Bella, G.; Zito, C.; Manganaro, R.; Piccione, M.C.; Trimarchi, G.; Farina, A.; Micari, A.; et al. The Role of Imaging in Cardiovascular Prevention: A Comprehensive Review. J. Cardiovasc. Echogr. 2025, 35, 8–18. [Google Scholar] [CrossRef]
- Malahfji, M.; Al-Mallah, M. Epicardial adipose tissue on cardiac MRI and cardiovascular risk: Another reason to watch what you “EAT”. Atherosclerosis 2024, 397, 118523. [Google Scholar] [CrossRef]
- Duca, F.; Mascherbauer, K.; Donà, C.; Koschutnik, M.; Binder, C.; Nitsche, C.; Halavina, K.; Beitzke, D.; Loewe, C.; Bartko, P.; et al. Association of epicardial adipose tissue on magnetic resonance imaging with cardiovascular outcomes: Quality over quantity? Obesity 2024, 32, 1670–1679. [Google Scholar] [CrossRef] [PubMed]
- Trimarchi, G.; Carerj, M.L.; Zito, C.; Bella, G.D.; Taverna, G.; Cusmà Piccione, M.; Crea, P.; Lo Giudice, S.; Buonpane, A.; Bonanni, M.; et al. Epicardial Adipose Tissue: A Multimodal Imaging Diagnostic Perspective. Medicina 2025, 61, 961. [Google Scholar] [CrossRef] [PubMed]
- van Woerden, G.; van Veldhuisen, D.J.; Gorter, T.M.; van Empel, V.P.M.; Hemels, M.E.W.; Hazebroek, E.J.; van Veldhuisen, S.L.; Willems, T.P.; Rienstra, M.; Westenbrink, B.D. Importance of epicardial adipose tissue localization using cardiac magnetic resonance imaging in patients with heart failure with mid-range and preserved ejection fraction. Clin. Cardiol. 2021, 44, 987–993. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, S.; Wang, Y.; Zhou, N.; Shu, J.; Stamm, C.; Jiang, M.; Luo, F. Association of epicardial adipose tissue attenuation with coronary atherosclerosis in patients with a high risk of coronary artery disease. Atherosclerosis 2019, 284, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Daudé, P.; Ancel, P.; Confort Gouny, S.; Jacquier, A.; Kober, F.; Dutour, A.; Bernard, M.; Gaborit, B.; Rapacchi, S. Deep-Learning Segmentation of Epicardial Adipose Tissue Using Four-Chamber Cardiac Magnetic Resonance Imaging. Diagnostics 2022, 12, 126. [Google Scholar] [CrossRef]
- Granér, M.; Siren, R.; Nyman, K.; Lundbom, J.; Hakkarainen, A.; Pentikäinen, M.O.; Lauerma, K.; Lundbom, N.; Adiels, M.; Nieminen, M.S.; et al. Cardiac steatosis associates with visceral obesity in nondiabetic obese men. J. Clin. Endocrinol. Metab. 2013, 98, 1189–1197. [Google Scholar] [CrossRef]
- Perseghin, G.; Lattuada, G.; De Cobelli, F.; Esposito, A.; Belloni, E.; Ntali, G.; Ragogna, F.; Canu, T.; Scifo, P.; Del Maschio, A.; et al. Increased mediastinal fat and impaired left ventricular energy metabolism in young men with newly found fatty liver. Hepatology 2008, 47, 51–58. [Google Scholar] [CrossRef]
- Hell, M.M.; Achenbach, S.; Schuhbaeck, A.; Klinghammer, L.; May, M.S.; Marwan, M. CT-based analysis of pericoronary adipose tissue density: Relation to cardiovascular risk factors and epicardial adipose tissue volume. J. Cardiovasc. Comput. Tomogr. 2016, 10, 52–60. [Google Scholar] [CrossRef]
- Nogajski, Ł.; Mazuruk, M.; Kacperska, M.; Kurpias, M.; Mączewski, M.; Nowakowski, M.; Mączewski, M.; Michałowska, I.; Leszek, P.; Paterek, A. Epicardial fat density obtained with computed tomography imaging—More important than volume? Cardiovasc. Diabetol. 2024, 23, 389. [Google Scholar] [CrossRef] [PubMed]
- Cho, I.; Al’Aref, S.J.; Berger, A.; Ó Hartaigh, B.; Gransar, H.; Valenti, V.; Lin, F.Y.; Achenbach, S.; Berman, D.S.; Budoff, M.J.; et al. Prognostic value of coronary computed tomographic angiography findings in asymptomatic individuals: A 6-year follow-up from the prospective multicentre international CONFIRM study. Eur. Heart J. 2018, 39, 934–941. [Google Scholar] [CrossRef]
- Antonopoulos, A.S.; Sanna, F.; Sabharwal, N.; Thomas, S.; Oikonomou, E.K.; Herdman, L.; Margaritis, M.; Shirodaria, C.; Kampoli, A.M.; Akoumianakis, I.; et al. Detecting human coronary inflammation by imaging perivascular fat. Sci Transl Med. 2017, 9, eaal2658. [Google Scholar] [CrossRef] [PubMed]
- Kitagawa, T.; Sentani, K.; Ikegami, Y.; Takasaki, T.; Takahashi, S.; Nakano, Y. Relationship Between Clinical Parameters and Histological Features of Epicardial Adipose Tissue and Aortic Valve Calcification Assessed on Computed Tomography. Circ. J. 2024, 88, 1986–1995. [Google Scholar] [CrossRef]
- Oikonomou, E.K.; Marwan, M.; Desai, M.Y.; Mancio, J.; Alashi, A.; Hutt Centeno, E.; Thomas, S.; Herdman, L.; Kotanidis, C.P.; Thomas, K.E.; et al. Non-invasive detection of coronary inflammation using computed tomography and prediction of residual cardiovascular risk (the CRISP CT study): A post-hoc analysis of prospective outcome data. Lancet 2018, 392, 929–939. [Google Scholar] [CrossRef]
- Chan, K.; Wahome, E.; Tsiachristas, A.; Antonopoulos, A.S.; Patel, P.; Lyasheva, M.; Kingham, L.; West, H.; Oikonomou, E.K.; Volpe, L.; et al. ORFAN Consortium. Inflammatory risk and cardiovascular events in patients without obstructive coronary artery disease: The ORFAN multicentre, longitudinal cohort study. Lancet 2024, 403, 2606–2618. [Google Scholar] [CrossRef] [PubMed]
- Masuda, M.; Mizuno, H.; Enchi, Y.; Minamiguchi, H.; Konishi, S.; Ohtani, T.; Yamaguchi, O.; Okuyama, Y.; Nanto, S.; Sakata, Y. Abundant epicardial adipose tissue surrounding the left atrium predicts early rather than late recurrence of atrial fibrillation after catheter ablation. J. Interv. Card. Electrophysiol. 2015, 44, 31–37. [Google Scholar] [CrossRef]
- Takahashi, N.; Abe, I.; Kira, S.; Ishii, Y. Role of epicardial adipose tissue in human atrial fibrillation. J. Arrhythm. 2023, 39, 93–110. [Google Scholar] [CrossRef]
- Huber, A.T.; Fankhauser, S.; Wittmer, S.; Chollet, L.; Lam, A.; Maurhofer, J.; Madaffari, A.; Seiler, J.; Servatius, H.; Haeberlin, A.; et al. Epicardial adipose tissue dispersion at CT and recurrent atrial fibrillation after pulmonary vein isolation. Eur. Radiol. 2024, 34, 4928–4938. [Google Scholar] [CrossRef]
- Iacobellis, G.; Singh, N.; Wharton, S.; Sharma, A.M. Substantial changes in epicardial fat thickness after weight loss in severely obese subjects. Obesity 2008, 16, 1693–1697. [Google Scholar] [CrossRef]
- Serrano-Ferrer, J.; Crendal, E.; Walther, G.; Vinet, A.; Dutheil, F.; Naughton, G.; Lesourd, B.; Chapier, R.; Courteix, D.; Obert, P. Effects of lifestyle intervention on left ventricular regional myocardial function in metabolic syndrome patients from the RESOLVE randomized trial. Metabolism 2016, 65, 1350–1360. [Google Scholar] [CrossRef]
- Tunc Suygun, E.; Vardar Yagli, N.; Suygun, H. Relationship between epicardial adipose tissue thickness and sedentary time, physical activity level, and physical performance in patients with hypertension. J. Hum. Hypertens. 2025, 39, 274–278. [Google Scholar] [CrossRef]
- Johnson, N.A.; Sultana, R.N.; Brown, W.J.; Bauman, A.E.; Gill, T. Physical activity in the management of obesity in adults: A position statement from Exercise and Sport Science Australia. J. Sci. Med. Sport. 2021, 24, 1245–1254. [Google Scholar] [CrossRef] [PubMed]
- Saco-Ledo, G.; Valenzuela, P.L.; Castillo-García, A.; Arenas, J.; León-Sanz, M.; Ruilope, L.M.; Lucia, A. Physical exercise and epicardial adipose tissue: A systematic review and meta-analysis of randomized controlled trials. Obes. Rev. 2021, 22, e13103. [Google Scholar] [CrossRef]
- Gaborit, B.; Jacquier, A.; Kober, F.; Abdesselam, I.; Cuisset, T.; Boullu-Ciocca, S.; Emungania, O.; Alessi, M.C.; Clément, K.; Bernard, M.; et al. Effects of bariatric surgery on cardiac ectopic fat: Lesser decrease in epicardial fat compared to visceral fat loss and no change in myocardial triglyceride content. J. Am. Coll. Cardiol. 2012, 60, 1381–1389. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, A.; Rawat, U.; Kumar, P.; Mittal, P. Pleotropic effects of statins: The dilemma of wider utilization of statin. Egypt. Heart J. 2023, 75, 1. [Google Scholar] [CrossRef]
- Parisi, V.; Petraglia, L.; D’Esposito, V.; Cabaro, S.; Rengo, G.; Caruso, A.; Grimaldi, M.G.; Baldascino, F.; De Bellis, A.; Vitale, D.; et al. Statin therapy modulates thickness and inflammatory profile of human epicardial adipose tissue. Int. J. Cardiol. 2019, 274, 326–330. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Park, Y.S.; Kim, Y.J.; Lee, I.S.; Kim, J.H.; Lee, J.H.; Choi, S.W.; Jeong, J.O.; Seong, I.W. Effects of statins on the epicardial fat thickness in patients with coronary artery stenosis underwent percutaneous coronary intervention: Comparison of atorvastatin with simvastatin/ezetimibe. J. Cardiovasc. Ultrasound 2010, 18, 121–126. [Google Scholar] [CrossRef]
- Soucek, F.; Covassin, N.; Singh, P.; Ruzek, L.; Kara, T.; Suleiman, M.; Lerman, A.; Koestler, C.; Friedman, P.A.; Lopez-Jimenez, F.; et al. Effects of Atorvastatin (80 mg) Therapy on Quantity of Epicardial Adipose Tissue in Patients Undergoing Pulmonary Vein Isolation for Atrial Fibrillation. Am. J. Cardiol. 2015, 116, 1443–1446. [Google Scholar] [CrossRef]
- Raggi, P.; Gadiyaram, V.; Zhang, C.; Chen, Z.; Lopaschuk, G.; Stillman, A.E. Statins Reduce Epicardial Adipose Tissue Attenuation Independent of Lipid Lowering: A Potential Pleiotropic Effect. J. Am. Heart Assoc. 2019, 8, e013104. [Google Scholar] [CrossRef]
- Dong, L.; Kerwin, W.S.; Chen, H.; Chu, B.; Underhill, H.R.; Neradilek, M.B.; Hatsukami, T.S.; Yuan, C.; Zhao, X.Q. Carotid artery atherosclerosis: Effect of intensive lipid therapy on the vasa vasorum--evaluation by using dynamic contrast-enhanced MR imaging. Radiology 2011, 260, 224–231. [Google Scholar] [CrossRef]
- Balaz, M.; Becker, A.S.; Balazova, L.; Straub, L.; Müller, J.; Gashi, G.; Maushart, C.I.; Sun, W.; Dong, H.; Moser, C.; et al. Inhibition of Mevalonate Pathway Prevents Adipocyte Browning in Mice and Men by Affecting Protein Prenylation. Cell Metab. 2019, 29, 901–916.e8. [Google Scholar] [CrossRef]
- Toia, P.; La Grutta, L.; Vitabile, S.; Punzo, B.; Cavaliere, C.; Militello, C.; Rundo, L.; Matranga, D.; Filorizzo, C.; Maffei, E.; et al. Epicardial Adipose Tissue Changes during Statin Administration in Relation to the Body Mass Index: A Longitudinal Cardiac CT Study. Appl. Sci. 2023, 13, 10709. [Google Scholar] [CrossRef]
- Jamialahmadi, T.; Simental-Mendia, L.E.; Eid, A.H.; Almahmeed, W.; Salehabadi, S.; Al-Rasadi, K.; Banach, M.; Sahebkar, A. Efficacy of statin therapy in reducing epicardial adipose tissue: A systematic review and meta-analysis. Arch. Med. Sci. 2024, 20, 997–1001. [Google Scholar] [CrossRef] [PubMed]
- Dozio, E.; Ruscica, M.; Vianello, E.; Macchi, C.; Sitzia, C.; Schmitz, G.; Tacchini, L.; Corsi Romanelli, M.M. PCSK9 Expression in Epicardial Adipose Tissue: Molecular Association with Local Tissue Inflammation. Mediat. Inflamm. 2020, 2020, 1348913. [Google Scholar] [CrossRef] [PubMed]
- Rivas Galvez, R.E.; Morales Portano, J.D.; Trujillo Cortes, R.; Gomez Alvarez, E.B.; Sanchez Cubias, S.M.; Zelaya, S.M. Reduction of epicardial adipose tissue thickness with PCSK9 inhibitors. Eur. Heart. J. 2020, 41 (Suppl. S2), ehaa946–3008. [Google Scholar] [CrossRef]
- Salvatore, T.; Galiero, R.; Caturano, A.; Vetrano, E.; Rinaldi, L.; Coviello, F.; Di Martino, A.; Albanese, G.; Colantuoni, S.; Medicamento, G.; et al. Dysregulated Epicardial Adipose Tissue as a Risk Factor and Potential Therapeutic Target of Heart Failure with Preserved Ejection Fraction in Diabetes. Biomolecules 2022, 12, 176. [Google Scholar] [CrossRef]
- Egami, Y.; Nishino, M.; Ryu Shutta Makino, N.; Tanouchi, J. Abstract 12012: Relation Between Colchicine and Epicardial Adipose Tissue Volume Surrounding Left Atrium in Atrial Fibrillation Recurrence After Ablation. Circulation 2015, 132 (Suppl. S3), A12012. [Google Scholar] [CrossRef]
- Masson, W.; Lavalle-Cobo, A.; Nogueira, J.P. Effect of SGLT2-Inhibitors on Epicardial Adipose Tissue: A Meta-Analysis. Cells 2021, 10, 2150. [Google Scholar] [CrossRef]
- Wu, P.; Wen, W.; Li, J.; Xu, J.; Zhao, M.; Chen, H.; Sun, J. Systematic Review and Meta-Analysis of Randomized Controlled Trials on the Effect of SGLT2 Inhibitor on Blood Leptin and Adiponectin Level in Patients with Type 2 Diabetes. Horm. Metab. Res. 2019, 51, 487–494. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Maleki, M.; Butler, A.E.; Jamialahmadi, T.; Sahebkar, A. Sodium-glucose co-transporter-2 inhibitors and epicardial adiposity. Eur. J. Pharm. Sci. 2023, 180, 106322. [Google Scholar] [CrossRef]
- Morciano, C.; Gugliandolo, S.; Capece, U.; Di Giuseppe, G.; Mezza, T.; Ciccarelli, G.; Soldovieri, L.; Brunetti, M.; Avolio, A.; Splendore, A.; et al. SGLT2 inhibition and adipose tissue metabolism: Current outlook and perspectives. Cardiovasc. Diabetol. 2024, 23, 449. [Google Scholar] [CrossRef]
- Buttice, L.; Ghani, M.; Suthakar, J.; Gnanalingham, S.; Carande, E.; Kennedy, B.W.C.; Pitcher, A.; Gamble, J.H.; Ahmad, M.; Lewis, A.; et al. The effect of sodium-glucose cotransporter-2 inhibitors on inflammatory biomarkers: A meta-analysis of randomized controlled trials. Diabetes Obes. Metab. 2024, 26, 2706–2721. [Google Scholar] [CrossRef]
- Malavazos, A.E.; Iacobellis, G.; Dozio, E.; Basilico, S.; Di Vincenzo, A.; Dubini, C.; Menicanti, L.; Vianello, E.; Meregalli, C.; Ruocco, C.; et al. Human epicardial adipose tissue expresses glucose-dependent insulinotropic polypeptide, glucagon, and glucagon-like peptide-1 receptors as potential targets of pleiotropic therapies. Eur. J. Prev. Cardiol. 2023, 30, 680–693. [Google Scholar] [CrossRef]
- Yaribeygi, H.; Maleki, M.; Nasimi, F.; Jamialahmadi, T.; Stanford, F.C.; Sahebkar, A. Benefits of GLP-1 Mimetics on Epicardial Adiposity. Curr. Med. Chem. 2023, 30, 4256–4265. [Google Scholar] [CrossRef]
- Dozio, E.; Vianello, E.; Malavazos, A.E.; Tacchini, L.; Schmitz, G.; Iacobellis, G. Epicardial adipose tissue GLP-1 receptor is associated with genes involved in fatty acid oxidation and white-to-brown fat differentiation: A target to modulate cardiovascular risk? Int. J. Cardiol. 2019, 292, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Wang, X.; Wang, Y.; Yao, J.; Shi, C.; Du, J.; Bai, R. The Effect of Liraglutide on Epicardial Adipose Tissue in Type 2 Diabetes. J. Diabetes Res. 2021, 2021, 5578216. [Google Scholar] [CrossRef] [PubMed]
- Kosiborod, M.N.; Abildstrøm, S.Z.; Borlaug, B.A.; Butler, J.; Rasmussen, S.; Davies, M.; Hovingh, G.K.; Kitzman, D.W.; Lindegaard, M.L.; Møller, D.V.; et al. STEP-HFpEF Trial Committees and Investigators. Semaglutide in Patients with Heart Failure with Preserved Ejection Fraction and Obesity. N. Engl. J. Med. 2023, 389, 1069–1084. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, M. Epicardial Fat Paradox and Differential Effects of GLP-1 Receptor Agonists Across Heart Failure Phenotypes. Circ. Heart Fail. 2023, 16, e010966. [Google Scholar] [CrossRef] [PubMed]
- Qi, Z.; Wu, D.; Yan, Z.; Wang, Q.; Li, Y.; Zhao, J.; Xu, F. Role of epicardial adipose tissue in heart failure with preserved ejection fraction: An emerging molecular mechanism and therapeutic potential. Obes. Rev. 2025, 26, e13912. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.J.H.; Shanbhag, A.; Killekar, A.; Lemley, M.; Bednarski, B.; Kriekinge, V.; Kavanagh, P.B.; Feher, A.; Miller, E.J.; Einstein, A.J.; et al. AI-derived epicardial fat measurements improve cardiovascular risk prediction from myocardial perfusion imaging. NPJ Digit. Med. 2024, 7, 24. [Google Scholar] [CrossRef]
- Imazio, M.; Agrimi, C.; Cescon, L.; Panzolli, G.; Collini, V.; Sinagra, G. Colchicine for the treatment of the spectrum of cardiovascular diseases: Current evidence and ongoing perspectives. J. Cardiovasc. Med. 2024, 25, 653–663. [Google Scholar] [CrossRef]
- Marzolla, V.; Feraco, A.; Gorini, S.; Mammi, C.; Marrese, C.; Mularoni, V.; Boitani, C.; Lombès, M.; Kolkhof, P.; Ciriolo, M.R.; et al. The novel non-steroidal MR antagonist finerenone improves metabolic parameters in high-fat diet-fed mice and activates brown adipose tissue via AMPK-ATGL pathway. FASEB J. 2020, 34, 12450–12465. [Google Scholar] [CrossRef]
- Barrio-Lopez, M.T.; Ruiz-Canela, M.; Goni, L.; Valiente, A.M.; Garcia, S.R.; de la O, V.; Anton, B.D.; Fernandez-Friera, L.; Castellanos, E.; Martínez-González, M.A.; et al. Mediterranean diet and epicardial adipose tissue in patients with atrial fibrillation treated with ablation: A substudy of the ‘PREDIMAR’ trial. Eur. J. Prev. Cardiol. 2024, 31, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Nyawo, T.A.; Pheiffer, C.; Mazibuko-Mbeje, S.E.; Mthembu, S.X.H.; Nyambuya, T.M.; Nkambule, B.B.; Sadie-Van Gijsen, H.; Strijdom, H.; Tiano, L.; Dludla, P.V. Physical Exercise Potentially Targets Epicardial Adipose Tissue to Reduce Cardiovascular Disease Risk in Patients with Metabolic Diseases: Oxidative Stress and Inflammation Emerge as Major Therapeutic Targets. Antioxidants 2021, 10, 1758. [Google Scholar] [CrossRef] [PubMed]
- Hoang, D.M.; Pham, P.T.; Bach, T.Q.; Ngo, A.T.L.; Nguyen, Q.T.; Phan, T.T.K.; Nguyen, G.H.; Le, P.T.; Hoang, V.T.; Forsyth, N.R.; et al. Stem cell-based therapy for human diseases. Signal Transduct. Target. Ther. 2022, 7, 272. [Google Scholar] [CrossRef]
| Imaging Modality | Diagnostic Accuracy | Reproducibility | Prognostic Value | Practical Feasibility |
|---|---|---|---|---|
| Transthoracic Echocardiography (TTE) | Visualises EAT as an echo-free space between the outer myocardial wall and visceral pericardium; optimal accuracy with end-systolic measurement [75,76]. Moderate agreement with CT when end-diastolic values used [78]. | Excellent interobserver and intraobserver agreement [77] | EAT thickness at Rindfleisch fold correlates with CMR measurements and independently predicts HF-related death, HF admissions, and arrhythmic events [80,81] | Widely available, low cost, no radiation; already integrated into HFpEF assessments; quick acquisition |
| Cardiac Magnetic Resonance (CMR) | Gold standard for quantifying visceral fat; excellent delineation of pericardial layers; cine steady-state free-precession acquisitions provide accurate, reproducible volumes [82,83,84,85]. | Highly reproducible, especially with volumetric analysis; automated algorithms enhance consistency [86]. | EAT-T1 independently predicts HF hospitalisation, all-cause mortality, and MI beyond traditional risk factors [84,87]. Four-chamber cine-derived EAT area correlates with diastolic dysfunction and metabolic risk [88,89,90]. | No radiation, non-invasive; limited by scanner availability, cost, exam time, and contraindications [84,85]. |
| Cardiac Computed Tomography (cCT/CCTA) | High-fidelity visualisation of EAT; enables volumetric and thickness quantification; ability to measure FAI as surrogate for inflammation [91,92]. | High reproducibility with manual/automated methods; allows 3D rendering of EAT [91]. | Elevated pericoronary FAI predicts acute coronary events and is modifiable by anti-inflammatory therapy [95,96]; PAAT volume/FAI predicts atrial fibrillation incidence and recurrence [98,99,100]. | Excellent anatomical detail; can be integrated into CCTA for CAD evaluation with minimal added time; limited by radiation and contrast exposure [85,91,92,93]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mihăilă, M.-D.; Caloian, B.; Frîngu, F.I.; Irimie, D.A.; Minciună, I.A.; Pop, D. Beyond Traditional Risk Factors: Integrating Epicardial Adipose Tissue into the Comorbidity Landscape of HFpEF. J. Clin. Med. 2025, 14, 6139. https://doi.org/10.3390/jcm14176139
Mihăilă M-D, Caloian B, Frîngu FI, Irimie DA, Minciună IA, Pop D. Beyond Traditional Risk Factors: Integrating Epicardial Adipose Tissue into the Comorbidity Landscape of HFpEF. Journal of Clinical Medicine. 2025; 14(17):6139. https://doi.org/10.3390/jcm14176139
Chicago/Turabian StyleMihăilă, Marius-Dragoș, Bogdan Caloian, Florina Iulia Frîngu, Diana Andrada Irimie, Ioan Alexandru Minciună, and Dana Pop. 2025. "Beyond Traditional Risk Factors: Integrating Epicardial Adipose Tissue into the Comorbidity Landscape of HFpEF" Journal of Clinical Medicine 14, no. 17: 6139. https://doi.org/10.3390/jcm14176139
APA StyleMihăilă, M.-D., Caloian, B., Frîngu, F. I., Irimie, D. A., Minciună, I. A., & Pop, D. (2025). Beyond Traditional Risk Factors: Integrating Epicardial Adipose Tissue into the Comorbidity Landscape of HFpEF. Journal of Clinical Medicine, 14(17), 6139. https://doi.org/10.3390/jcm14176139






