Safety and Efficacy of Tolvaptan for Acute Refractive Hyponatremia Associated with Traumatic Brain Injury
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection
2.2. Tolvaptan Administration Protocol
2.3. Data Collection
2.4. End Points
2.5. Statistical Analysis
3. Results
3.1. Baseline Characteristics
3.2. Outcomes
3.3. Safety Data
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TBI | Traumatic Brain Injury |
| SIADH | Syndrome of Inappropriate Antidiuretic Hormone Secretion |
| AVP | Arginine Vasopressin |
| FDA | Food and Drug Administration |
| ICU | Intensive Care Unit |
| IRB | Institutional Review Board |
| CKD | Chronic Kidney Disease |
| CHF | Congestive Heart Failure |
| ODS | Osmotic Demyelination Syndrome |
| Na+ | Sodium |
| SD | Standard Deviation |
| OR | Odds Ratio |
| CI | Confidence Interval |
References
- Verbalis, J.G.; Goldsmith, S.R.; Greenberg, A.; Korzelius, C.; Schrier, R.W.; Sterns, R.H.; Thompson, C.J. Diagnosis, Evaluation, and Treatment of Hyponatremia: Expert Panel Recommendations. Am. J. Med. 2013, 126, S1–S42. [Google Scholar] [CrossRef]
- Jiang, L.; Cheng, J.; Li, M. The Association Between Hyponatremia and Prognosis in Traumatic Brain Injury: A Comprehensive Meta-Analysis. J. Craniofacial Surg. 2025, 36, 1715–1720. [Google Scholar] [CrossRef]
- Lohani, S.; Devkota, U.P. Hyponatremia in Patients with Traumatic Brain Injury: Etiology, Incidence, and Severity Correlation. World Neurosurg. 2011, 76, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Yumoto, T.; Sato, K.; Ugawa, T.; Ichiba, S.; Ujike, Y. Prevalence, risk factors, and short-term consequences of traumatic brain injury-associated hyponatremia. Acta Medica Okayama 2015, 69, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Krisanapan, P.; Tangpanithandee, S.; Thongprayoon, C.; Pattharanitima, P.; Kleindienst, A.; Miao, J.; Craici, I.M.; Mao, M.A.; Cheungpasitporn, W. Safety and Efficacy of Vaptans in the Treatment of Hyponatremia from Syndrome of Inappropriate Antidiuretic Hormone Secretion (SIADH): A Systematic Review and Meta-Analysis. J. Clin. Med. 2023, 12, 5483. [Google Scholar] [CrossRef] [PubMed]
- Galton, C.; Deem, S.; Yanez, N.D.; Souter, M.; Chesnut, R.; Dagal, A.; Treggiari, M. Open-Label Randomized Trial of the Safety and Efficacy of a Single Dose Conivaptan to Raise Serum Sodium in Patients with Traumatic Brain Injury. Neurocrit. Care 2011, 14, 354–360. [Google Scholar] [CrossRef]
- Jeon, S.-B.; Choi, H.A.; Lesch, C.; Kim, M.C.; Badjatia, N.; Claassen, J.; Mayer, S.A.; Lee, K. Use of Oral Vasopressin V2 Receptor Antagonist for Hyponatremia in Acute Brain Injury. Eur. Neurol. 2013, 70, 142–148. [Google Scholar] [CrossRef]
- Thomas, S.; Parvathi, U.; Sushama, J. An observational study on the effectiveness of tolvaptan in patients with euvolemic and hypervolemic hyponatremia admitted to a tertiary care center. Natl. J. Physiol. Pharm. Pharmacol. 2023, 13, 2218–2223. [Google Scholar] [CrossRef]
- Llewellyn, D.C.; Oštarijaš, E.; Sahadevan, S.; Nuamek, T.; Byrne, C.; Taylor, D.R.; Vincent, R.P.; Dimitriadis, G.K.; Aylwin, S.J.B. Efficacy and Safety of Low-Dose Tolvaptan (7.5 mg) in the Treatment of Inpatient Hyponatremia: A Retrospective Study. Endocr. Pract. 2025, 31, 419–425. [Google Scholar] [CrossRef]
- Lewellyn, D.; Nuamek, T.; Ostarijas, E.; Logan Ellis, H.; Drakou, E.E.; Aylwin, S.J.B.; Dimitriadis, G.K. Low-Dose Tolvaptan for the Treatment of Syndrome of Inappropriate Antidiuretic Hormone–Associated Hyponatremia: A Systematic Review, Meta-Analysis, and Meta-Regression Analysis of Clinical Effectiveness and Safety. Endocr. Pract. 2025, 31, 956–964. [Google Scholar] [CrossRef]
- Krisanapan, P.; Chinpraditsuk, S.; Jaiyen, N.; Tantiyavarong, P.; Pattharanitima, P. Efficacy of Low-Dosage Tolvaptan for Treatment of Profound Hyponatremia in Syndrome of Inappropriate Antidiuresis: An Open-Label Randomized Controlled Trial: TH-PO324. J. Am. Soc. Nephrol. 2024, 35, 10S. [Google Scholar] [CrossRef]
- Tzoulis, P.; Kaltsas, G.; Baldeweg, S.E.; Bouloux, P.-M.; Grossman, A.B. Tolvaptan for the treatment of the syndrome of inappropriate antidiuresis (SIAD). Ther. Adv. Endocrinol. Metab. 2023, 14, 20420188231173330. [Google Scholar] [CrossRef] [PubMed]
- Soerarso, R.; Yonas, E.; Sirait, S.P.; Hasanah, D.Y.; Raharjo, S.B.; Siswanto, B.B.; Cramer, M.J.; van der Harst, P.; Oerlemans, M.I.F.J. Importance of early use of tolvaptan in hyponatremic acutely decompensated heart failure patients, a retrospective study. Egypt. Heart J. 2025, 77, 10. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-J. Vasopressin Receptor Antagonist, Tolvaptan, for Treating Hyponatremia in Patients with Heart Failure. Cardiovasc. Prev. Pharmacother. 2021, 3, 10–14. [Google Scholar] [CrossRef]
- Saimiya, M.; Kaku, Y.; Nishimura, M. Efficacy of oral tolvaptan for severe edema and hyponatremia in a patient with refractory nephrotic syndrome. CEN Case Rep. 2021, 10, 523–526. [Google Scholar] [CrossRef]
- Chatzimavridou-Grigoriadou, V.; Al-Othman, S.; Brabant, G.; Kyriacou, A.; King, J.; Blackhall, F.; Trainer, P.J.; Higham, C.E. Clinical Experience of the Efficacy and Safety of Low-dose Tolvaptan Therapy in a UK Tertiary Oncology Setting. J. Clin. Endocrinol. Metab. 2021, 106, e4766–e4775. [Google Scholar] [CrossRef]
- Ren, P.; Yang, Q. The role of tolvaptan in managing hyponatremia in small cell lung cancer patients with SIADH: A retrospective study of 23 cases. Transl. Cancer Res. 2021, 10, 1229–1237. [Google Scholar] [CrossRef]
- Hoshi, A.; Momo, K.; Yamazaki, K.; Otsuka, K.; Aoki, T.; Murakami, M.; Sasaki, T. Tolvaptan for Hyponatremia Induced by Chemotherapy With Docetaxel, Cisplatin, and 5-Fluorouracil. Am. J. Ther. 2022, 29, e380–e383. [Google Scholar] [CrossRef]
- Indirli, R.; Ferreira de Carvalho, J.; Cremaschi, A.; Mantovani, B.; Sala, E.; Serban, A.L.; Locatelli, M.; Bertani, G.; Carosi, G.; Fiore, G.; et al. Tolvaptan in the Management of Acute Euvolemic Hyponatremia After Transsphenoidal Surgery: A Retrospective Single-Center Analysis. Front. Endocrinol. 2021, 12, 689887. [Google Scholar] [CrossRef]
- Bisiani, A.; Redmond, J.M.; Deane, A.M.; Plummer, M.P. Vaptans for the Management of Hyponatremia in Neurocritical Care: A Systematic Review. SN Compr. Clin. Med. 2022, 4, 12. [Google Scholar] [CrossRef]
- Smetana, K.S.; Wiss, A.L.; May, C.C. Efficacy and Safety of Conivaptan versus Tolvaptan in Neurocritically Ill Patients. J. Transl. Crit. Care Med. 2022, 4, 7. [Google Scholar] [CrossRef]
- Friedman, B.; Cirulli, J. Hyponatremia in critical care patients: Frequency, outcome, characteristics, and treatment with the vasopressin V2-receptor antagonist tolvaptan. J. Crit. Care 2013, 28, e1–e219. [Google Scholar] [CrossRef]
- Human, T.; Cook, A.M.; Anger, B.; Bledsoe, K.; Castle, A.; Deen, D.; Gibbs, H.; Lesch, C.; Liang, N.; McAllen, K.; et al. Treatment of Hyponatremia in Patients with Acute Neurological Injury. Neurocrit. Care 2017, 27, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Carney, N.; Totten, A.M.; O’Reilly, C.; Ullman, J.S.; Hawryluk, G.W.J.; Bell, M.J.; Bratton, S.L.; Chesnut, R.; Harris, O.A.; Kissoon, N.; et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery 2017, 80, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Cook, A.M.; Morgan Jones, G.; Hawryluk, G.W.J.; Mailloux, P.; McLaughlin, D.; Papangelou, A.; Samuel, S.; Tokumaru, S.; Venkatasubramanian, C.; Zacko, C.; et al. Guidelines for the Acute Treatment of Cerebral Edema in Neurocritical Care Patients. Neurocrit. Care 2020, 32, 647–666. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, R.; Swaminathan, G.; Nair, S.; Joseph, M. Hyponatremia in Traumatic Brain Injury: A Practical Management Protocol. World Neurosurg. 2017, 108, 529–533. [Google Scholar] [CrossRef]
- Wendt, R.; Fenves, A.Z.; Geisler, B.P. Use of Urea for the Syndrome of Inappropriate Secretion of Antidiuretic Hormone: A Systematic Review. JAMA Netw. Open 2023, 6, e2340313. [Google Scholar] [CrossRef]
- Delgado-Cuesta, J.; Escorial-Moya, C.; Vallejo-Vaz, A.J.; Santos-Ramos, B.; Varela, J.M.; Calderón, E.J.; Medrano, F.J. Effectiveness of Urea and Tolvaptan in the Treatment of Hypotonic Hyponatremia. J. Clin. Med. 2025, 14, 3315. [Google Scholar] [CrossRef]
- Scalla, P.A.; Palma, S.M.; Dittmar, E.; Zamora, F.J.; Trimino, E. Comparative Safety and Effectiveness of Urea and Tolvaptan for the Management of Hyponatremia. J. Pharm. Pract. 2024, 37, 1252–1257. [Google Scholar] [CrossRef]
- Martínez González, Á.; González Nunes, M.; Rodeiro Escobar, P.; Llópiz Castedo, J.; Cabaleiro Loureiro, A.; Martínez Espinosa, R.P.; Ruades Patiño, R.; Lorenzo Canda, G.; Aguayo Arjona, J.; Rodríguez Zorrilla, S. Comparative study of the effectiveness of tolvaptan versus urea in patients with hyponatremia caused by SIADH. Rev. Clínica Española (Engl. Ed.) 2025, 225, 85–91. [Google Scholar] [CrossRef]
- Warren, A.M.; Grossmann, M.; Christ-Crain, M.; Russell, N. Syndrome of Inappropriate Antidiuresis: From Pathophysiology to Management. Endocr. Rev. 2023, 44, 819–861. [Google Scholar] [CrossRef]


| Baseline Characteristics (Total Patients = 126) | |||
|---|---|---|---|
| Patient Characteristic | Trauma (73) | Non-Trauma (53) | p Value |
| Age (years) | 67.8 ± 18 | 71.1 ± 17 | 0.33 |
| % females | 42% (31) | 49% (26) | 0.46 |
| Diagnoses | Injury Severity Scores | Admission Diagnoses Category | NA |
| 10 (4–16) | Renal | 30.2% (16) | |
| Neurological | 30.2% (16) | ||
| Musculoskeletal | 11.3% (6) | ||
| Cardiac | 13.2% (7) | ||
| Pulmonary | 3.8% (2) | ||
| Others | 11.3% (6) | ||
| Outcome of Tolvaptan Administration in Trauma and Non-Trauma Patients | |||
|---|---|---|---|
| Measurement | Trauma (73) | Non-Trauma (53) | p Value |
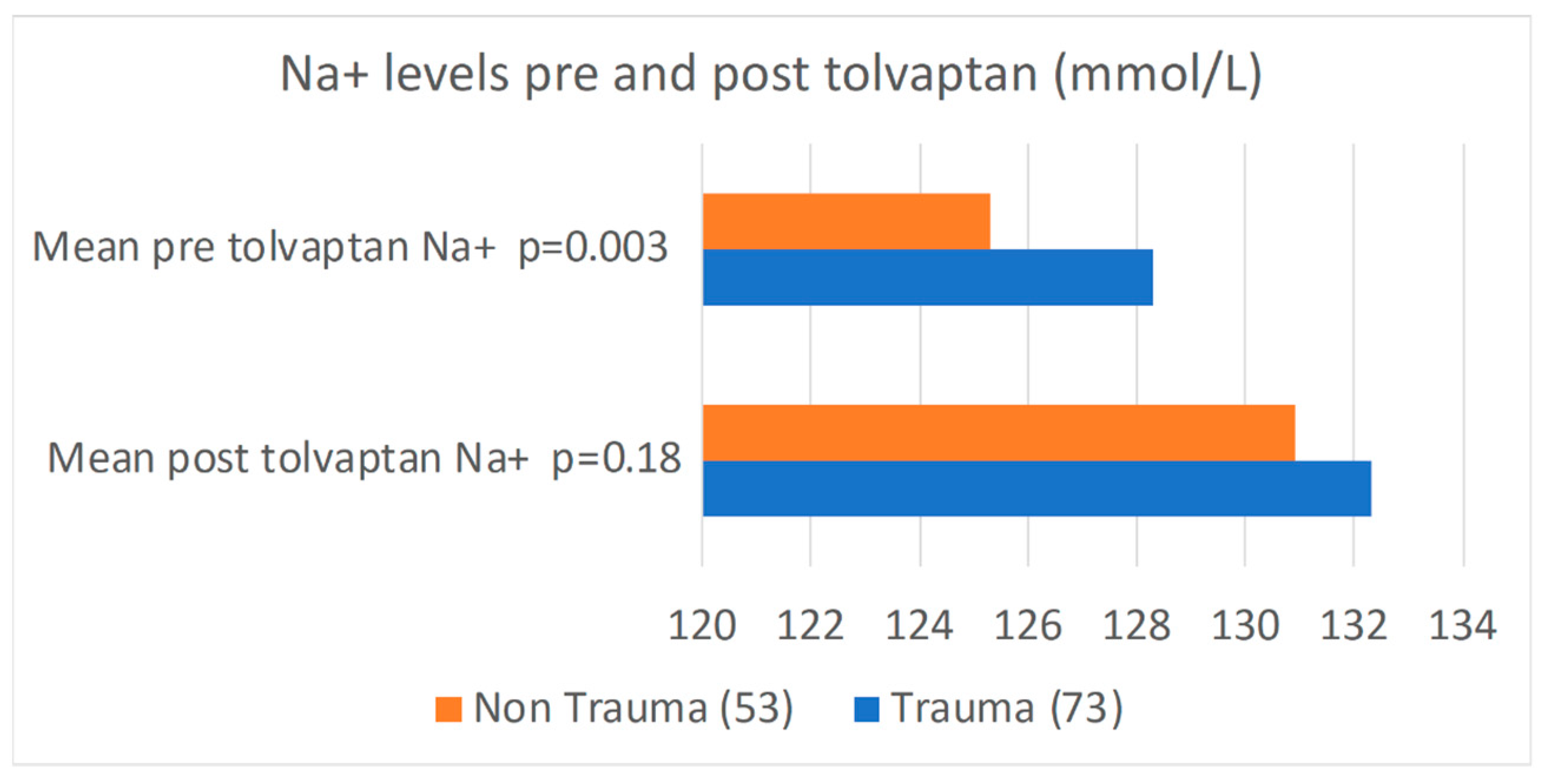
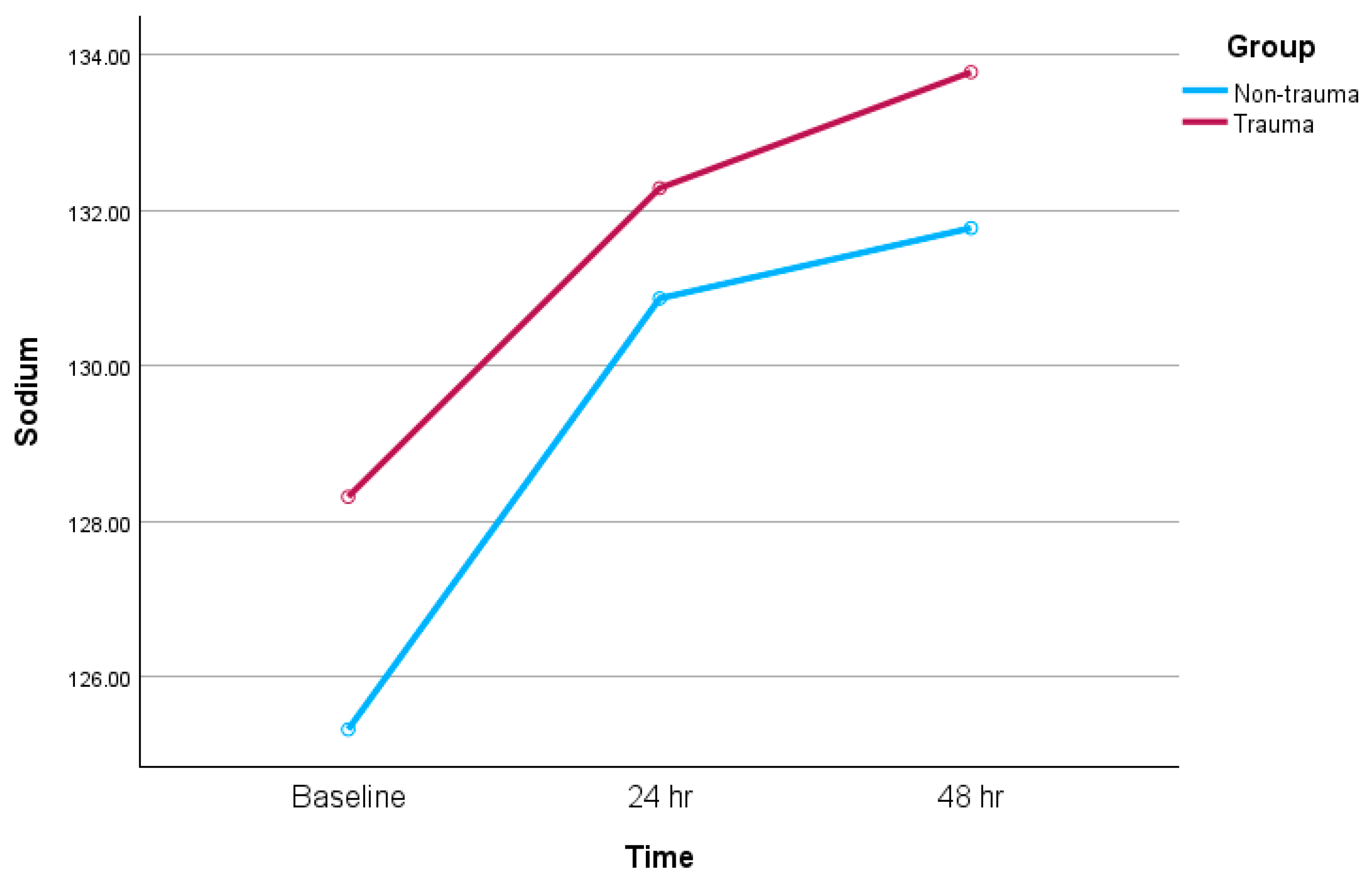
| Mean pre tolvaptan Na+ | 128.3 ± 4 | 125.3 ± 5 | 0.003 |
| Mean post tolvaptan Na+ (at 24 h) | 132.3 ±5 | 130.9 ± 7 | 0.18 |
| Mean post tolvaptan Na+ (at 48 h) | 133.8 ± 4 | 131.8 ± 6 | 0.036 |
| Median number of tolvaptan does | 1 (1–3) | 1 (1–3) | 1 |
| % patients with more than 1 Tolvaptan dose | 10.9% (8) | 56.6% (30) | <0.01 |
| % of patients with increase in Na+ post-tolvaptan therapy within 48 h of first dose | 95% (69) | 87% (46) | 0.12 |
| % patients with decrease or no change in Na+ post tolvaptan therapy | 5% (4) | 13% (7) | 0.12 |
| Amongst patients with an increase in Na+, mean % increase in Na+ compared to before tolvaptan therapy | 4.6% ± 2.7 | 6.5% ± 3.9 | 0.0018 |
| % patients with normalization of Na+ (135–145) within 24 h of first dose | 48% (35) | 30% (16) | 0.045 |
| % patients with Na+ level ≥ 130 | 86% (63) | 69.8% (37) | 0.02 |
| % patients with ≥ 8 meq increase in Na+ in 24 h | 30% (22) | 43.3% (23) | 0.12 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Desai, S.; Holsaeter, K.; Winski, A.; Barletta, J.F.; Bauer, F. Safety and Efficacy of Tolvaptan for Acute Refractive Hyponatremia Associated with Traumatic Brain Injury. J. Clin. Med. 2025, 14, 6138. https://doi.org/10.3390/jcm14176138
Desai S, Holsaeter K, Winski A, Barletta JF, Bauer F. Safety and Efficacy of Tolvaptan for Acute Refractive Hyponatremia Associated with Traumatic Brain Injury. Journal of Clinical Medicine. 2025; 14(17):6138. https://doi.org/10.3390/jcm14176138
Chicago/Turabian StyleDesai, Shashvat, Kathleen Holsaeter, Alexandra Winski, Jeffrey F. Barletta, and Frank Bauer. 2025. "Safety and Efficacy of Tolvaptan for Acute Refractive Hyponatremia Associated with Traumatic Brain Injury" Journal of Clinical Medicine 14, no. 17: 6138. https://doi.org/10.3390/jcm14176138
APA StyleDesai, S., Holsaeter, K., Winski, A., Barletta, J. F., & Bauer, F. (2025). Safety and Efficacy of Tolvaptan for Acute Refractive Hyponatremia Associated with Traumatic Brain Injury. Journal of Clinical Medicine, 14(17), 6138. https://doi.org/10.3390/jcm14176138







