Systematic Review on Antidepressant Use and Bleeding Risk After Dental Extractions: Evidence Gaps and Clinical Implications
Abstract
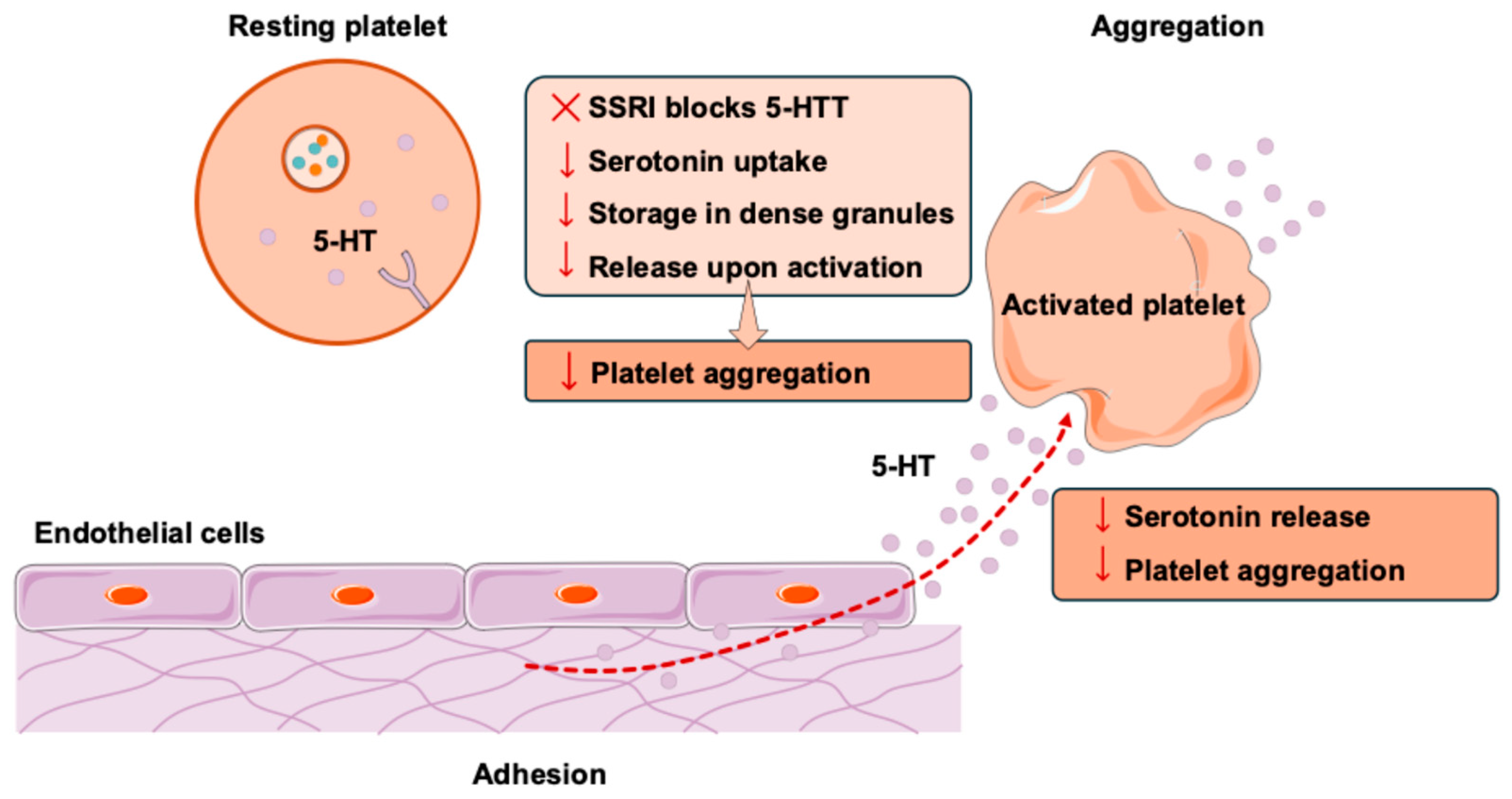
1. Introduction
2. Materials and Methods
2.1. Focused Question and Outcome Definition
2.2. Eligibility Criteria
- P: Patients undergoing dental extraction procedures;
- I: Patients taking antidepressants;
- C: Patients not taking antidepressants;
- O: Bleeding;
- S: Randomized clinical trials.
2.3. Search Strategy
- (((antidepressants) OR (SSRI)) OR (SNRI)) OR (wellbutrin)) AND ((bleeding) OR (hemorrhage) OR (bleeding events)) AND (dental OR extractions OR tooth).
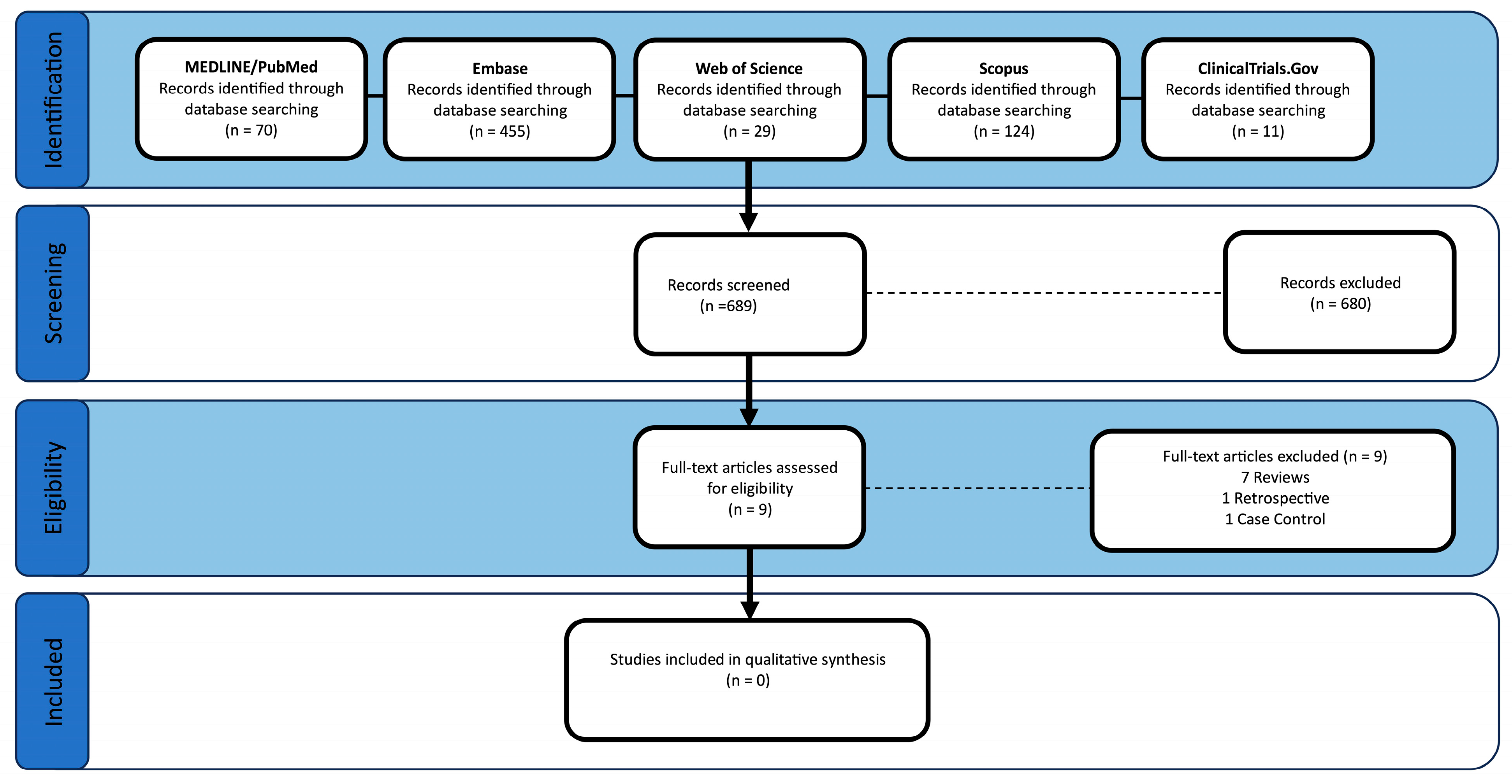
2.4. Study Selection and Data Extraction
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| SSRIs | Serotonin reuptake inhibitors |
| SNRIs | Serotonin–norepinephrine reuptake inhibitors |
| TCAs | Tricyclic antidepressants |
| MAOIs | Monoamine oxidase inhibitors |
| 5-HT | Serotonin |
| 5-HTT | 5-HT transporters |
| NSAIDs | Non-steroidal anti-inflammatory drugs |
| PRISMA | Preferred Reporting Items for Systematic Reviews and Meta-Analyses |
| PROSPERO | International Prospective Register of Systematic Reviews |
| PICOS | Patients, Intervention, Comparison, Outcome, and Study design |
| RCTs | Randomized controlled trials |
| ADP | Adenosine diphosphate |
| DOAC | Direct oral anticoagulant |
References
- Sheffler, Z.M.; Patel, P.; Abdijadid, S. Antidepressants. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Brody, D.J.; Gu, Q. Antidepressant Use Among Adults: United States, 2015–2018. NCHS Data Brief 2020, 377, 1–8. [Google Scholar]
- De Crescenzo, F.; De Giorgi, R.; Garriga, C.; Liu, Q.; Fazel, S.; Efthimiou, O.; Hippisley-Cox, J.; Cipriani, A. Real-world effects of antidepressants for depressive disorder in primary care: Population-based cohort study. Br. J. Psychiatry 2025, 226, 278–287. [Google Scholar] [CrossRef]
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Strawn, J.R.; Mills, J.A.; Poweleit, E.A.; Ramsey, L.B.; Croarkin, P.E. Adverse Effects of Antidepressant Medications and Their Management in Children and Adolescents. Pharmacotherapy 2023, 43, 675–690. [Google Scholar] [CrossRef]
- Ferguson, J.M. SSRI Antidepressant Medications: Adverse Effects and Tolerability. Prim. Care Companion J. Clin. Psychiatry 2001, 3, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Moraczewski, J.; Awosika, A.O.; Aedma, K.K. Tricyclic Antidepressants. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Trindade, E.; Menon, D.; Topfer, L.A.; Coloma, C. Adverse effects associated with selective serotonin reuptake inhibitors and tricyclic antidepressants: A meta-analysis. CMAJ 1998, 159, 1245–1252. [Google Scholar] [PubMed]
- Sub Laban, T.; Saadabadi, A. Monoamine Oxidase Inhibitors (MAOI). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Remick, R.A.; Froese, C.; Keller, F.D. Common side effects associated with monoamine oxidase inhibitors. Prog. Neuropsychopharmacol. Biol. Psychiatry 1989, 13, 497–504. [Google Scholar] [CrossRef]
- Huecker, M.R.; Smiley, A.; Saadabadi, A. Bupropion. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Laporte, S.; Chapelle, C.; Caillet, P.; Beyens, M.N.; Bellet, F.; Delavenne, X.; Mismetyti, P.; Bertoletti, L. Bleeding risk under selective serotonin reuptake inhibitor (SSRI) antidepressants: A meta-analysis of observational studies. Pharmacol. Res. 2017, 118, 19–32. [Google Scholar] [CrossRef]
- Sayadipour, A.; Mago, R.; Kepler, C.K.; Chambliss, R.B.; Certa, K.M.; Vaccaro, A.R.; Albert, T.J.; Anderson, D.G. Antidepressants and the risk of abnormal bleeding during spinal surgery: A case-control study. Eur. Spine J. 2012, 21, 2070–2078. [Google Scholar] [CrossRef]
- Schadler, P.; Shue, J.; Moawad, M.; Girardi, F.P.; Cammisa, F.P.; Sama, A.A.; Huang, R.C.; Lebl, D.R.; Craig, C.M.; Hughes, A.P. Serotonergic Antidepressants Are Associated with Increased Blood Loss and Risk for Transfusion in Single-Level Lumbar Fusion Surgery. Asian Spine J. 2017, 11, 601–609. [Google Scholar] [CrossRef]
- Basile, F.V.; Basile, A.R.; Basile, V.V. Use of selective serotonin reuptake inhibitors antidepressants and bleeding risk in breast cosmetic surgery. Aesthetic Plast. Surg. 2013, 37, 561–566. [Google Scholar] [CrossRef]
- Gärtner, R.; Cronin-Fenton, D.; Hundborg, H.H.; Pedersen, L.; Lash, T.L.; Sørensen, H.T.; Kroman, N. Use of selective serotonin reuptake inhibitors and risk of re-operation due to post-surgical bleeding in breast cancer patients: A Danish population-based cohort study. BMC Surg. 2010, 10, 3. [Google Scholar] [CrossRef]
- Movig, K.L.; Janssen, M.W.; de Waal Malefijt, J.; Kabel, P.J.; Leufkens, H.G.; Egberts, A.C. Relationship of serotonergic antidepressants and need for blood transfusion in orthopedic surgical patients. Arch. Intern. Med. 2003, 163, 2354–2358. [Google Scholar] [CrossRef] [PubMed]
- Schutte, H.J.; Jansen, S.; Schafroth, M.U.; Goslings, J.C.; van der Velde, N.; de Rooij, S.E. SSRIs increase risk of blood transfusion in patients admitted for hip surgery. PLoS ONE 2014, 9, e95906. [Google Scholar] [CrossRef] [PubMed]
- Salkeld, E.; Ferris, L.E.; Juurlink, D.N. The risk of postpartum hemorrhage with selective serotonin reuptake inhibitors and other antidepressants. J. Clin. Psychopharmacol. 2008, 28, 230–234. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.; Okamoto, T.; Sangu, N.M.; Kudo, S.; Ando, T. Mandibular Fracture in a Patient Taking a Direct Oral Anticoagulant. J. Craniofac. Surg. 2021, 32, 1421–1422. [Google Scholar] [CrossRef]
- Chisci, G.; Chisci, D.; Chisci, E.; Chisci, V.; Stumpo, M.; Chisci, E. The Management of a Geriatric Patient Using Dabigatran Therapy on Dentigerous Cyst with Oral Bleeding. J. Clin. Med. 2024, 13, 1499. [Google Scholar] [CrossRef]
- Toledano-Serrabona, J.; Sánchez-Torres, A.; Camps-Font, O.; Figueiredo, R.; Gay-Escoda, C.; Valmaseda-Castellón, E. Effect of an Informative Video on Anxiety and Hemodynamic Parameters in Patients Requiring Mandibular Third Molar Extraction: A Randomized Clinical Trial. J. Oral Maxillofac. Surg. 2020, 78, 1933–1941. [Google Scholar] [CrossRef]
- Li, X.; He, X.; Li, M.; Gu, X.; Wang, P.; Wu, Y.; Chen, Y. The effect of esketamine combined with sufentanil based patient-controlled intravenous analgesia for postoperative pain in patients undergoing third molar surgery and maxillofacial trauma: A randomized clinical trial. BMC Oral Health 2024, 24, 1460. [Google Scholar] [CrossRef]
- Steenen, S.A.; van Wijk, A.J.; van Westrhenen, R.; de Lange, J.; de Jongh, A. Effects of propranolol on fear of dental extraction: Study protocol for a randomized controlled trial. Trials 2015, 16, 536. [Google Scholar] [CrossRef]
- Perez, A.R.; Destiné, H.; Patel, N.K.; Campbell, R.E.; Muchintala, R.; Hall, A.T.; Pepe, M.D.; Tucker, B.S.; Tjoumakaris, F.P. Effects of Melatonin on Sleep Quality and Patient-Reported Outcomes After Arthroscopic Rotator Cuff Surgery: A Prospective Randomized Controlled Trial. Am. J. Sports Med. 2024, 52, 3075–3083. [Google Scholar] [CrossRef]
- El-Refai, N.A.; Shehata, J.H.; Lotfy, A.; Elbadawy, A.M.; Abdel Rahman, R.A.; Gamaleldin, T.M.; Dobal, N.M.; Mohamed, A.A.; Farrag, T.A.; Shafik, Y.M.; et al. Role of preoperative pregabalin in reducing inhalational anesthetic requirements in abdominal hysterectomy: Randomized controlled trial. Minerva Anestesiol. 2020, 86, 56–63. [Google Scholar] [CrossRef]
- Müller, J.; Nowak, S.; Vogelgesang, A.; von Sarnowski, B.; Rathmann, E.; Schmidt, S.; Rehberg, S.; Usichenko, T.; Kertscho, H.; Hahnenkamp, K.; et al. Evaluating Mechanisms of Postoperative Delirium and Cognitive Dysfunction Following Elective Spine Surgery in Elderly Patients (CONFESS): Protocol for a Prospective Observational Trial. JMIR Res. Protoc. 2020, 9, e15488. [Google Scholar] [CrossRef]
- Müller, J.; Nowak, S.; Weidemeier, M.; Vogelgesang, A.; Ruhnau, J.; von Sarnowski, B.; Saar, A.; Veser, Y.; Behr, F.; Gross, S.; et al. Duration of Surgery and Intraoperative Blood Pressure Management Are Modifiable Risk Factors for Postoperative Neurocognitive Disorders After Spine Surgery: Results of the Prospective CONFESS Study. Spine 2023, 48, 1127–1137. [Google Scholar] [CrossRef]
- Bonatto, M.D.S.; Feltran, G.D.S.; Barbosa, T.P.; Pereira, D.A.; Santos, S.D.S.; Mendes, P.G.J.; Sales e Pessoa, R.; Barbosa Bezerra, F.J.; Zambuzzi, W.F.; de Oliveira, G.J.P.L. Green tea and hyaluronic acid gel enhance fibroblast activation and improves the gingival healing post-third molar extraction. Sci. Rep. 2024, 14, 7124. [Google Scholar] [CrossRef]
- Weinrieb, R.M.; Auriacombe, M.; Lynch, K.G.; Lewis, J.D. Selective serotonin re-uptake inhibitors and the risk of bleeding. Expert Opin. Drug Saf. 2005, 4, 337–344. [Google Scholar] [CrossRef]
- Edinoff, A.N.; Raveendran, K.; Colon, M.A.; Thomas, B.H.; Trettin, K.A.; Hunt, G.W.; Kaye, A.M.; Cornett, E.M.; Kaye, A.D. Selective Serotonin Reuptake Inhibitors and Associated Bleeding Risks: A Narrative and Clinical Review. Health Psychol. Res. 2022, 10, 39580. [Google Scholar] [CrossRef] [PubMed]
- Galan, A.M.; Lopez-Vilchez, I.; Diaz-Ricart, M.; Navalon, F.; Gomez, E.; Gasto, C.; Escolar, G. Serotonergic mechanisms enhance platelet-mediated thrombogenicity. Thromb. Haemost. 2009, 102, 511–519. [Google Scholar] [CrossRef]
- de Abajo, F.J. Effects of selective serotonin reuptake inhibitors on platelet function: Mechanisms, clinical outcomes and implications for use in elderly patients. Drugs Aging 2011, 28, 345–367. [Google Scholar] [CrossRef] [PubMed]
- Weng, J.; Lan, R. Does Concomitant Use of Antidepressants and Direct Oral Anticoagulants Increase the Risk of Bleeding? A Systematic Review and Meta-Analysis. J. Clin. Psychopharmacol. 2025, 45, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Quinn, G.R.; Hellkamp, A.S.; Hankey, G.J.; Becker, R.C.; Berkowitz, S.D.; Breithardt, G.; Fava, M.; Fox, K.A.A.; Halperin, J.L.; Mahaffey, K.W.; et al. Selective Serotonin Reuptake Inhibitors and Bleeding Risk in Anticoagulated Patients with Atrial Fibrillation: An Analysis from the ROCKET AF Trial. J. Am. Heart Assoc. 2018, 7, e008755. [Google Scholar] [CrossRef] [PubMed]
- Anglin, R.; Yuan, Y.; Moayyedi, P.; Tse, F.; Armstrong, D.; Leontiadis, G.I. Risk of upper gastrointestinal bleeding with selective serotonin reuptake inhibitors with or without concurrent nonsteroidal anti-inflammatory use: A systematic review and meta-analysis. Am. J. Gastroenterol. 2014, 109, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Napeñas, J.J.; Hong, C.H.; Kempter, E.; Brennan, M.T.; Furney, S.L.; Lockhart, P.B. Selective serotonin reuptake inhibitors and oral bleeding complications after invasive dental treatment. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2011, 112, 463–467. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. (Eds.) Cochrane Handbook for Systematic Reviews of Interventions, Version 6.5; Cochrane: London, UK, 2024; Available online: www.cochrane.org/handbook (accessed on 17 August 2025).
| Database | Search Strategy |
|---|---|
| PubMed/MEDLINE | (“antidepressent”[All Fields] OR “antidepression”[All Fields] OR “antidepressive agents”[Pharmacological Action] OR “antidepressive agents”[Supplementary Concept] OR “antidepressive agents”[All Fields] OR “antidepressant”[All Fields] OR “antidepressive agents”[MeSH Terms] OR (“antidepressive”[All Fields] AND “agents”[All Fields]) OR “antidepressants”[All Fields] OR “antidepressive”[All Fields] OR “antidepressives”[All Fields] OR “SSRI 1”[All Fields] OR (“serotonin and noradrenaline reuptake inhibitors”[Pharmacological Action] OR “serotonin and noradrenaline reuptake inhibitors”[Supplementary Concept] OR “serotonin and noradrenaline reuptake inhibitors”[All Fields] OR “SNRI 2”[All Fields] OR “serotonin and noradrenaline reuptake inhibitors”[MeSH Terms] OR (“serotonin”[All Fields] AND “noradrenaline”[All Fields] AND “reuptake”[All Fields] AND “inhibitors”[All Fields])) OR (“bupropion”[Supplementary Concept] OR “bupropion”[All Fields] OR “amfebutamone”[All Fields] OR “bupropion”[MeSH Terms] OR “wellbutrin”[All Fields] OR “bupropion s”[All Fields] OR “bupropione”[All Fields])) AND (“bleedings”[All Fields] OR “hemorrhage”[MeSH Terms] OR “hemorrhage”[All Fields] OR “bleed”[All Fields] OR “bleeding”[All Fields] OR “bleeds”[All Fields] OR “bleeding event”[All Fields]) AND (“dental health services”[MeSH Terms] OR (“dental”[All Fields] AND “health”[All Fields] AND “services”[All Fields]) OR “dental health services”[All Fields] OR “dental”[All Fields] OR “dentally”[All Fields] OR “dentals”[All Fields] OR (“extract”[All Fields] OR “extract s”[All Fields] OR “extractabilities”[All Fields] OR “extractability”[All Fields] OR “extractable”[All Fields] OR “extractables”[All Fields] OR “extractant”[All Fields] OR “extractants”[All Fields] OR “extracted”[All Fields] OR “extractibility”[All Fields] OR “extractible”[All Fields] OR “extracting”[All Fields] OR “extraction”[All Fields] OR “extractions”[All Fields] OR “extractive”[All Fields] OR “extractives”[All Fields] OR “extracts”[All Fields]) OR (“teeth s”[All Fields] OR “teeths”[All Fields] OR “tooth”[MeSH Terms] OR “tooth”[All Fields] OR “teeth”[All Fields] OR “tooth s”[All Fields] OR “tooths”[All Fields])) |
| EMBASE | (‘antidepressants’/exp OR antidepressants OR ‘ssri’/exp OR ssri OR ‘snri’/exp OR snri OR ‘wellbutrin’/exp OR wellbutrin) AND (‘bleeding’/exp OR bleeding OR ‘hemorrhage’/exp OR hemorrhage OR ‘bleeding events’/exp OR ‘bleeding events’) AND (‘dental’/exp OR dental OR extractions OR ‘tooth’/exp OR tooth) |
| Web of Science | (((((antidepressants) OR (SSRI)) OR (SNRI)) OR (wellbutrin)) AND (bleeding OR hemorrhage OR bleeding events) AND (dental OR extractions OR tooth)) |
| Scopus | TITLE-ABS-KEY((antidepressants OR ssri OR snri OR wellbutrin) AND (bleeding OR hemorrhage OR (bleeding events)) AND (dental OR extractions OR tooth)) |
| ClinicalTrials.gov | ((((antidepressants) OR (SSRI)) OR (SNRI)) OR (wellbutrin)) AND (bleeding OR hemorrhage OR (bleeding events)) AND (dental OR extractions OR tooth) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nemeth, A.; Rahimi, K.; Karande, S.; Romasco, T.; Lowenstein, A.; Pereira, R.d.S.; Mourão, C.F. Systematic Review on Antidepressant Use and Bleeding Risk After Dental Extractions: Evidence Gaps and Clinical Implications. J. Clin. Med. 2025, 14, 6140. https://doi.org/10.3390/jcm14176140
Nemeth A, Rahimi K, Karande S, Romasco T, Lowenstein A, Pereira RdS, Mourão CF. Systematic Review on Antidepressant Use and Bleeding Risk After Dental Extractions: Evidence Gaps and Clinical Implications. Journal of Clinical Medicine. 2025; 14(17):6140. https://doi.org/10.3390/jcm14176140
Chicago/Turabian StyleNemeth, Alexander, Kayvon Rahimi, Sachin Karande, Tea Romasco, Adam Lowenstein, Rodrigo dos Santos Pereira, and Carlos Fernando Mourão. 2025. "Systematic Review on Antidepressant Use and Bleeding Risk After Dental Extractions: Evidence Gaps and Clinical Implications" Journal of Clinical Medicine 14, no. 17: 6140. https://doi.org/10.3390/jcm14176140
APA StyleNemeth, A., Rahimi, K., Karande, S., Romasco, T., Lowenstein, A., Pereira, R. d. S., & Mourão, C. F. (2025). Systematic Review on Antidepressant Use and Bleeding Risk After Dental Extractions: Evidence Gaps and Clinical Implications. Journal of Clinical Medicine, 14(17), 6140. https://doi.org/10.3390/jcm14176140









