Neurostimulation in People with Oropharyngeal Dysphagia: A Systematic Review and Meta-Analysis of Randomised Controlled Trials—Part II: Brain Neurostimulation
Abstract
:1. Introduction
2. Methods
2.1. Information Sources and Search Strategies
2.2. Inclusion and Exclusion Criteria
2.3. Systematic Review
2.4. Meta-Analysis
3. Results
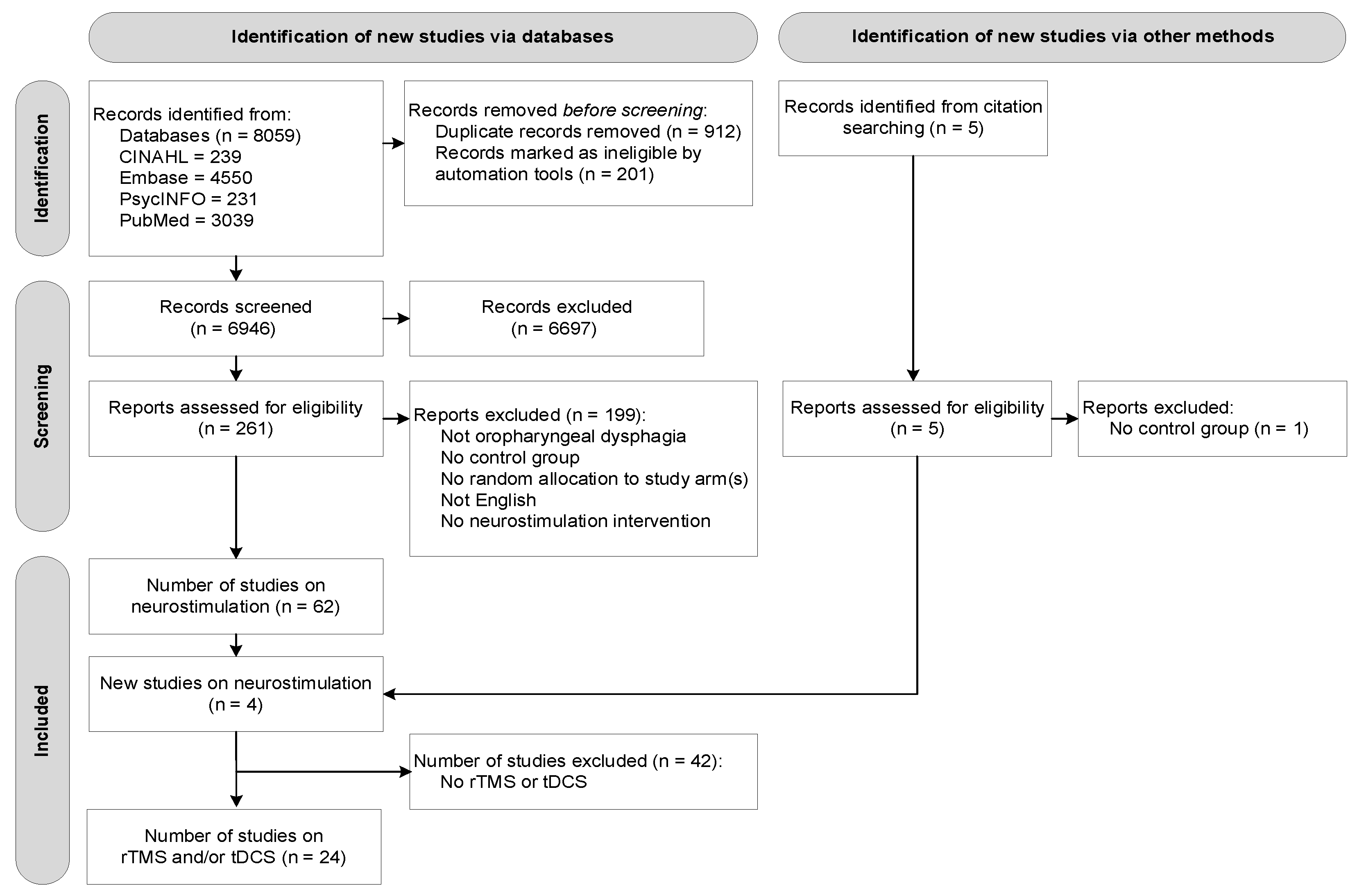
3.1. Study Selection
3.2. Description of Studies
Study
| Inclusion/Exclusion Criteria | Sample (n)
| Group Descriptives (Mean ± SD) Age, Gender, Medical Diagnoses | Procedure, Delivery and Dosage per Intervention Group a |
|---|---|---|---|---|
| repetitive Transcranial Magnetic Stimulation (rTMS)—n = 11 | ||||
Cheng et al. (2017) [46]
|
| n = 15
| Treatment group: Age = 65.1 ± 8.3 Male = 64% Sham group: Age = 63.3 ± 7.8 Male = 100% NS difference between groups in age or post-stroke duration. | Procedure: rTMS (Magstim Rapid) daily for 10 days over 2 weeks
|
Du et al. (2016) [34]
|
| n = 40
| Treatment group 1: Age 58.2 ± 2.8 87% male Location of lesion: cortical (1), subcortical (10), massive (4) Treatment group 2: Age 57.9 ± 2.5 54% male Location of lesion: cortical (0), subcortical (9), massive (4) Sham group: Age 58.8 ± 3.4 50% male Location of lesion: cortical (2), subcortical (5), massive (5) NS differences between groups. | Procedure:
|
Khedr et al. (2009) [47]
|
| n = 26
| Treatment group: Age 58.9 ± 11.7 Sham group: Age 56.2 ± 13.4 No group specific descriptors given. Overall, 38.5% male. 14 with right-sided hemiplegia and 12 patients with left-sided hemiplegia. NS difference between groups. | Procedure:
|
Khedr and Abo-Elfetoh (2010) [48]
|
| n = 22
| Group statistics given based on infarction type divided into treatment versus sham. Lateral medullary infarction group: Treatment group (6): Age 56.7 ± 16 100% male Sham (5): Age: 58 ± 17.5 100% male Other brainstem infarction group: Treatment group (5): Age: 55.4 ± 9.7 40% male Sham (6): Age: 60.5 ± 11 50% male NS difference between groups. | Procedure:
|
Khedr et al. (2019) [30]
|
| n = 30
| Treatment group: Age 60.7 ± 8.8 duration of illness 5.7 +/− 3.9 Hoehn and Yahr 3.1 +/− 1.1 Sham group: Age 57.4 ± 10.0 duration of illness 6.5 +/− 3.7 Hoehn and Yahr 3.5 +/− 1.0 Gender distribution not given. NS difference between groups. | Procedure:
|
Kim et al. (2011) [32]
|
| n = 30
| Treatment group 1: Age: 69.8 ± 8.0 50% male Stroke (9), TBI (1) Treatment group 2: Age: 66.4 ± 12.3 66.6% maleStroke (10), TBI (0) Sham group: Age: 68.2 ± 12.6 66.6% male Stroke (9), TBI (1) NS difference between groups. | Procedure:
|
Momosaki et al. (2014) [49]
|
| n = 20
| Treatment group: Age 61 ± 22 80% male Duration post-stroke 19 +/− 8 months Lesion: cerebrum 2, cerebellum 2, brainstem 5, mixed 1 Sham group: Age 66 ± 9 60% male Duration post-stroke 21 +/− 8 months Lesion: cerebrum 1, cerebellum 3, brainstem 2, mixed 4. NS difference between groups. | Procedure:
Same parameters with the coil held on its lateral side |
Park et al. (2013) [50]
|
| n = 18
| Treatment group: Age 73.7 ± 3.8 56% male Infarct = 7, haemorrhage = 2Right lesion = 6 Sham group: Age 68.9 ± 9.354% male Infarct = 8, haemorrhage = 1Right lesion = 5 NS difference between groups. | Procedure: rTMS (Magstim Rapid2)
Same rTMS dosage, however Magstim coil positioned at 90 degree tilt (same noise, no motor cortical stimulation) |
Park et al. (2017) [35]
|
| n = 33
| Treatment group 1: Age 60.2 ± 13.8 73% male Infarct = 7, haemorrhage = 4 Treatment group 2: Age 67.5 ± 13.4 73% male Infarct = 9, haemorrhage = 2 Sham group: 69.6 ± 8.6 64% male Infarct = 7, haemorrhage = 4 NS difference between groups. | Procedure: rTMS (Magstim Rapid 2) to cortical representation of the mylohyoid muscle, identified by EMG. Applied 10 Hz and 90% of RMT for 5 s with a 55 s inter-train interval.
|
Tarameshlu et al. (2019) [33]
|
| n = 18
| Treatment group 1: Age 55.33 ± 19.55 67% male 67% cortical stroke, 33% subcortical Treatment group 2: Age 74.67 ± 5.92 17% male 83% cortical stroke, 17% subcortical Treatment group 3: Age 66 ± 5.55 67% male 67% cortical stroke, 33% subcortical NS difference between groups. | Treatment group 1: rTMS (Magstim super-rapid stimulator).
|
Ünlüer et al. (2019) [51]
|
| n = 28
| Treatment group: Age 67.80 ± 11.88 60% male 7% haemorrhage, 93% ischaemic stroke Sham group: Age 69.31 ± 12.89 46% male 8% haemorrhage, 92% ischaemic stroke NS difference between groups. | Procedure: DT for 30–45 min, 3 days/week (+2 days home exercises) for 4 weeks
|
| transcranial Direct Current Stimulation (tDCS)—n = 9 | ||||
Ahn et al. (2017) [36]
|
| n = 26
| Treatment group: Age 61.6 ± 10.3 69.2% male 38.5% infarction, 61.5% haemorrhage Sham group: Age 66.4 ± 10.7 46.2% male 84.6% infarction, 15.4% haemorrhage Statistical difference between groups = NR | Procedure:
|
Cosentino et al. (2020) [31]
|
| n = 40
Both groups crossed over to sham treatment, also. Order randomised. | Treatment group 1: Age 71.5 ± 5.2 53% male 70.5% primary presbydysphagia, 72.4% secondary presbydysphagia Treatment group 2: Age 75.2 ± 4.8 (p = 0.025) 57% male 76.4% primary presbydysphagia, 74.0% secondary presbydysphagia Statistical difference between groups = NR | Procedure:
|
Kumar et al. (2011) [27]
|
| n = 14 (pilot study)
| Treatment group: Average age 79.7 43% male Average NIHHS score 13.6Sham group: Average age 70 57% male Average NIHHS score 13.1Statistical difference between groups = NR | Procedure:
Treatment parameters not described in detail |
Pingue et al. (2018) [37]
|
| n = 40
| Treatment group: Age 63.5 (range = 54.5–75.25) 40% male Infarct = 11, haemorrhage = 11 (NB. Note numeral errors reported here, n= 20, not 22) Sham group: Age 68.5 (range = 62–73) 40% male Infarct = 4, haemorrhage = 16 NS difference between groups. | Procedure: tDCS by a battery-driven constant current stimulator (HDCkit Newronika, Italy). Stimulation targeted the pharyngeal motor cortex (site location method not described).
2 mA of anodal tDCS over the lesioned hemisphere and cathodal stimulation to the contralesional hemisphere. Sham + DT: Same protocol except current was delivered for only 30 s through 2 electrodes, producing initial tingling sensation but no cortical excitability. |
Sawan et al. (2020) [29]
|
| n = 40
| Treatment group: Age 53.3 ± 5.0 50% unilateral stroke, 50% bilateral stroke Sham group: Age 50.3 ± 5.2 50% unilateral stroke, 50% bilateral stroke NS difference between groups. | Procedure:
Group 1 (unilateral hemispheric stroke) anode placed on healthy hemisphere with reference electrode over contralateral supraorbital region.Group 2 (bilateral hemispheric stroke) stimulation first applied to the dominant hemisphere, then non-dominant hemisphere. Sham + DT: Same protocol producing tingling sensation but no cortical excitability. |
Shigematsu et al. (2013) [38]
|
| n = 20
| Treatment group: Age: 66.9 ± 6.3 70% male; Time post-stroke: 12.9 ± 7.8 Site of lesion: 20% putamen; 20% medulla oblongata; 10% corona radiata; 10% frontotemporal; 10% frontoparietal; 10% pons; 10% thalamus; 10% internal capsule Sham group: Age 64.7 ± 8.9 70% male Time post-stroke: 12.1 ± 9.0 Site of lesion: 40% pons; 20% frontoparietal; 10% putamen; 10% thalamus; 10% internal capsule; 10% caudate nucleus NS difference between groups. | Procedure: stimulation by DC stimulator (NeuroConn)
|
Suntrup-Krueger et al. (2018) [39]
|
| n = 59
| Treatment group: Age 68.9 ± 11.5 58.6 % male 72.4% supratentorial stroke 27.6% infratentorial stroke Sham group: Age 67.2 ± 14.5 56.7 % male 80.0% supratentorial stroke 20.0% infratentorial stroke NS difference between groups. | Procedure: tDCS stimulation delivered by battery-driven constant current stimulator (NeuroConn)
|
Wang et al. (2020) [40]
|
| n = 28
| Treatment group: Age 61.43 ± 11.24 79% male Time post-stroke: 66.79 ± 38.62 days Sham group: Age 62.00 ± 10.46 71% male Time post-stroke: 67.50 ± 47.62 days NS difference between groups. | Procedure: anodal tDCS + catheter balloon dilatation + standard swallow therapy (based on VFSS, details not described)
|
Yang et al. (2012) [41]
|
| n = 16
| Treatment group: Age 70.44 ± 12.59 66.7% male 44.4% right lesion, NIHSS = 9.7 ± 5.4 Sham group: Age 70.57 ± 8.46 42.9% male 57.1% right lesion, NIHSS = 13.9 ± 6.3 NS differences between groups. | Procedure: anodal tDCS (Phoresor II)
|
| Combined Neurostimulation Interventions—n = 4 | ||||
Cabib et al. (2020) [44]
|
| n = 36
| Treatment group 1: Age 70.0 ± 8.6 75% male 0% haemorrhage, 100% infarction Treatment group 2: Age 74.3 ± 7.8 58% male 8% haemorrhage, 92% infarction Treatment group 3: Age 70.0 ± 14.2 92% male 25% haemorrhage, 75% infarction NS differences between groups, except shorter time since stroke for capsaicin group. | Procedure: All patients received both treatment and sham, cross-over active/sham in visits 1 week apart (randomised). Assessment occurred immediately prior to treatment and within 2 h post-treatment.Treatment group 1: rTMS (Magstim rapid stimulator)
Treatment group 3: PES via two-ring electrode naso-pharyngeal catheter (Gaeltec Ltd.)
|
Lim et al. (2014) [43]
|
| n = 47
| Treatment group 1: Age 62.5 ± 8.2 60% male 34% haemorrhage, 66% infarction Treatment group 2: Age 59.8 ± 11.8 43% male 71% haemorrhage, 29% infarction Treatment group 3: Age 66.3 ± 15.4 67% male 66% haemorrhage, 44% infarction NS difference between groups. | Procedure:
|
Michou et al. (2014) [42]
|
| n = 18
| Treatment group: Avg age 60.3 83% male Treatment group 2: Avg age 67.3 100% male Treatment group 3: Avg age 67.8 66.7% male Overall 63 +/− 15 weeks post-stroke with 7.6 +/− 1 on NIHHS Statistical difference between groups = NR | Procedure:
|
Zhang et al. (2019) [45]
|
| n = 64
| Treatment group 1: Age 55.9 ± 8.9 43% male 61.5% subcortical, 38.5% brainstem Treatment group 2: Age 56.8 ± 9.7 54% male 30.8% subcortical, 69.2% brainstem Treatment group 3: Age 56.5 ± 10.1 50% male 58.3% subcortical, 41.7% brainstem Treatment group 4: Age 53.1 ± 10.6 31% male 61.5% subcortical, 38.5% brainstem All data given on participants that finished the trial and follow-up period (n = 52) | Procedure:
10-Hz real rTMS was delivered to the hot spot for the mylohyoid muscle at the ipsilesional hemisphere followed by 1-Hz sham rTMS over the corresponding position of the contralesional hemisphere. Treatment group 3: Contralateral rTMS + NMES 10-Hz sham rTMS was delivered to the hot spot for the mylohyoid muscle at the ipsilesional hemisphere followed by 1-Hz real rTMS over the corresponding position of the contralesional hemisphere. Treatment group 4: Bilateral rTMS + NMES 10-Hz real rTMS was delivered to the hot spot for the mylohyoid muscle at the ipsilesional hemisphere followed by 1-Hz real rTMS over the corresponding position of the contralesional hemisphere. |
| Study | Intervention Goal | Outcome Measures | Intervention Outcomes &Conclusions |
|---|---|---|---|
| repetitive Transcranial Magnetic Stimulation (rTMS)—n = 11 | |||
| Cheng et al. (2017) [46] | To investigate the short-(2-months) and long-term (6 and 12 months) effects of 5 Hz rTMS on chronic post-stroke dysphagia | Primary outcomes: Maximum tongue strength, VFSS (oral transit time, stage transit time, pharyngeal transit time, pharyngeal constriction ratio), and SAPP [52]. Assessed: 1 week pre-, and 2, 6 and 12 months post-intervention. |
|
| Du et al. (2016) [34] | To investigate the effects of high-frequency versus low-frequency rTMS on poststroke dysphagia during early rehabilitation | Primary outcome: SSA [53]. Secondary outcomes: WST [54], DD [55], NIHSS score [56], BI [57], mRS, measures of mylohyoid MEPs evoked from both hemispheres before and after treatment. Assessed: before treatment, after 5th rTMS session, and at 1-, 2-, and 3-months post-treatment. | Primary outcomes:
|
| Khedr et al. (2009) [47] | To investigate the therapeutic effect of rTMS on post-stroke dysphagia | Primary outcome: Dysphagia rating scale [58] (swallowing questionnaire + bedside examination). Secondary outcomes: Motor power of hand grip, BI [57], measures of oesophageal MEPs from both hemispheres. Assessed: before and immediately after treatment, and at 1- and 2-months post-treatment. |
|
| Khedr and Abo-Elfetoh (2010) [48] | To assess the effect of rTMS on dysphagia in patients with acute lateral medullary or other brainstem infarction | Primary outcome: DD [55] Secondary outcomes: Hand grip strength, NIHHS [56] and BI [57]. Assessed: before treatment, after 5th rTMS session, and at 1- and 2-months post-treatment. | Results given based on infarction type divided into treatment versus sham. rTMS and lateral medullary infarction
|
| Khedr et al. (2019) [30] | To investigate the therapeutic effect of rTMS on dysphagia with Parkinson’s Disease | Primary outcomes: Hoen and Yahr staging [59], UPDRS [60] part III, IADL [61], Self-Assessment Scale [62], SDQ [63], Arabic-DHI [64]. VFSS was conducted on 9 rTMS and 6 sham group patients. Assessed: before treatment, post treatment, and at 1-, 2-, and 3-months post-treatment. |
|
| Kim et al. (2011) [32] | To investigate the effect of rTMS on dysphagia recovery in patients with brain injury | Primary outcomes: FDS [65], PAS [66] and ASHA-NOMS [67] before and after treatment Assessed: before and after treatment, times unspecified. |
|
| Momosaki et al. (2014) [49] | To assess the effectiveness of a single functional magnetic stimulation session on post-stroke dysphagia | Primary outcomes: Timed WST [54] before and after stimulation Secondary outcome: N/R |
|
| Park et al. (2013) [50] | To find the therapeutic effect of high-frequency repetitive TMS on a contra-lesional intact pharyngeal motor cortex inpost-stroke dysphagic patients | Primary outcome: VDS [68], PAS [66] (as per VFSS), pre- and post- treatment. 2 and 4 weeks from baseline. Secondary outcomes: Oral and pharyngeal components of VDS | Treatment group:
|
| Park et al. (2017) [35] | to investigate the effects of high-frequency rTMS at the bilateral motor cortices over the cortical representation of the mylohyoid muscles in the patients with post-stroke dysphagia. | Primary outcomes: Immediately post-treatment and 3 weeks post-treatment: using CDS [69], DOSS [58], PAS [66], and VDS [68]. Secondary outcome: N/R |
|
| Tarameshlu et al. (2019) [33] | To compare the effects of standard swallow therapy (DT), rTMS and a combined intervention (CI)on swallowing function in patients with poststroke dysphagia | Primary outcome: MASA [70]. Secondary outcomes: FOIS [71] assessed (a) before treatment, (b) after 5th session and after 10th, 15th and 18th session. | Primary outcome: MASA
|
| Ünlüer et al. (2019) [51] | To identify whether applying low-frequency rTMS can enhance the effect of conventional swallowing treatment and quality of life of chronic (2–6 months) stroke patients suffering from dysphagia | Primary outcome: PAS [66], pre-post treatment, 1 and 3 months post-treatment. Secondary outcomes: VFSS parameters (including oral parameters, tongue retraction, hyolaryngeal elevation, delayed swallow reflex, residue, nutritional status, SWAL-QOL). |
|
| transcranial Direct Current Stimulation (tDCS)—n = 9 | |||
| Ahn et al. (2017) [36] | To investigate the effect of bihemispheric anodal tDCS with conventional dysphagia therapy on chronic post-stroke dysphagia | Primary outcome: DOSS [58] score based on VFSS pre- and post-treatment Secondary outcome: N/R |
|
| Cosentino et al. (2020) [31] | To investigate the therapeutic potential of tDCS and theta-burst stimulation on primary or secondary presbydysphagia | Primary outcomes: DOSS [58] based on bedside assessment and FEES. Similarity Index based on Electrokinesiographic/electromyographic Study (EES) for Laryngeal-pharyngeal Mechanogram (LPM) and electromyographic activity of the submental/suprahyoid muscles complex (SHEMG). Secondary outcome: N/R Outcomes assessed at baseline, 1 month and 3 months post-treatment |
|
| Kumar et al. (2011) [27] | To investigate whether anodal tDCS in combination with swallowing manoeuvres facilitates dysphagia recovery in stroke patients during early stroke convalescence | Primary outcome: DOSS [58]. Secondary outcome: N/R | Treatment group had significantly improved DOSS scores compared to sham group (p = 0.019). |
| Pingue et al. (2018) [37] | To evaluate whether anodal tDCS over the lesioned hemisphere and cathodal tDCS to the contralateral one during the early stage of rehabilitation can improve poststroke dysphagia | Primary outcome: DOSS [58], PAS [66] post-treatment. Secondary outcome: N/R |
|
| Sawan et al. (2020) [29] | To assess the effect of tDCS on improving dysphagia in stroke patients | Primary outcomes: DOSS [58]; Oral Transit Time; laryngeal and hyoid elevation; oesophageal sphincter spasm; aspiration Secondary outcome: N/R |
|
| Shigematsu et al. (2013) [38] | To investigate if the application of tDCS to the ipsilateral cortical motor and sensory pharyngeal areas can improve swallowing function in poststroke patients | Primary outcome: DOSS [58] immediately post-treatment and 1 month post-treatment Secondary outcomes: PAS [66], oral intake status. |
|
| Suntrup-Krueger et al. (2013) [39] | To evaluate the efficacy of a pathophysiologically reasonable tDCS protocol to improve stroke-related OD, via a randomized controlled trial (RCT) in a sufficiently large patient sample with objective clinical outcome measures alongside functional neuroimaging | Primary outcome: Improved FEDSS 4 days post-treatment Secondary outcomes: DSRS [72]; final FEDSS, and FOIS [71] scores prior to discharge; pneumonia rate until discharge; length of stay (in hospital). Activation changes in the swallowing network as measured with MEG. | Primary outcome:
|
| Wang et al. (2020) [40] | To investigate the effects of tDCS combined with conventional swallowing training on the swallowing function in brainstem stroke patients with cricopharyngeal muscle dysfunction. | Primary outcome: FDS [65] (before and immediately after intervention). Secondary outcomes: FOIS [71], MBSImp [73], PESO measurement [74]. | Primary outcomes: Statistical difference between the groups at endpoint not reported.
|
| Yang et al. (2012) [41] | To investigate the effects of anodal tDCS combined with swallowing training for post-stroke dysphagia. | Primary outcome: FDS [65] immediately post-treatment and at 3 months Secondary outcomes: Oral Transit Time, Pharyngeal Transit Time and total transit time. |
|
| Combined Neurostimulation Interventions—n = 4 | |||
| Cabib et al. (2020) [44] | To investigate the effect of rTMS of the primary sensory cortex (A), oral capsaicin (B) and intra-pharyngeal electrical stimulation (IPES; C) on post-stroke dysphagia | Primary outcomes: Effect size pre-post treatment for neurophysiological variables (pharyngeal and thenar RMT and MEP). Secondary outcomes: Effects on the biomechanics of swallow (PAS [66], impaired efficiency + more) VFSS before and after treatment |
|
| Lim et al. (2014) [43] | To investigate the effect of low-frequency rTMS and NMES on post-stroke dysphagia. | Primary outcomes: VFSS baseline, 2- + 4-weeks post-treatment (for semi-solids and liquids): FDS [65], PAS [66], Pharyngeal Transit Time. Secondary outcome: N/R |
|
| Michou et al. (2014) [42] | To compare the effects of a single application of one of three neurostimulation techniques (PES, paired stimulation, rTMS) on swallow safety and neurophysiological mechanisms in chronic post-stroke dysphagia. | Primary Outcome: VFSS before and after treatment Secondary outcomes: Percentage change in cortical excitability; Oral Transit Time, pharyngeal response time, Pharyngeal Transit Time, airway closure time and upper oesophageal opening time as per VFSS | Treatment group 1 (PES): significant excitability increase immediately post-Tx in the unaffected hemisphere (real vs. sham p = 0.043) and in the affected hemisphere 30 min post-Tx (real vs. sham p = 0.04).
Corticobulbar excitability of pharyngeal motor cortex was beneficially modulated by PES, Paired Stimulation and to a lesser extent by rTMS. |
| Zhang et al. (2019) [45] | To determine whether rTMS NMES effectively ameliorates dysphagia and how rTMS protocols (bilateral vs. unilateral) combined with NMES can be optimized. | Primary outcome: Cortical excitability(amplitude of the motor evoked potential) Secondary outcomes: SSA [53] and DD [55]. | Compared with group 2 or 3 in the affected hemisphere, group 4 displayed a significantly greater percentage change (p.0.017 and p.0.024, respectively). All groups displayed significant improvements in SSA and DD scores after treatment and at 1-month follow-up. The percentage change in cortical excitability increased over time in either the affected or unaffected hemisphere in treatment groups 1, 2 and 4 (p < 0.05). In Group 3, the percentage change in cortical excitability in the unaffected hemisphere significantly decreased after the stimulation course (p < 0.05). Change in SSA and DD scores in group 4 was markedly higher than that in the other three groups at the end of stimulation (p.0.02, p.0.03, and p.0.005) and still higher than that in group 1 at the 1-month follow-up (p.0.01). |
3.3. Risk of Bias Assessment and Methodological Quality
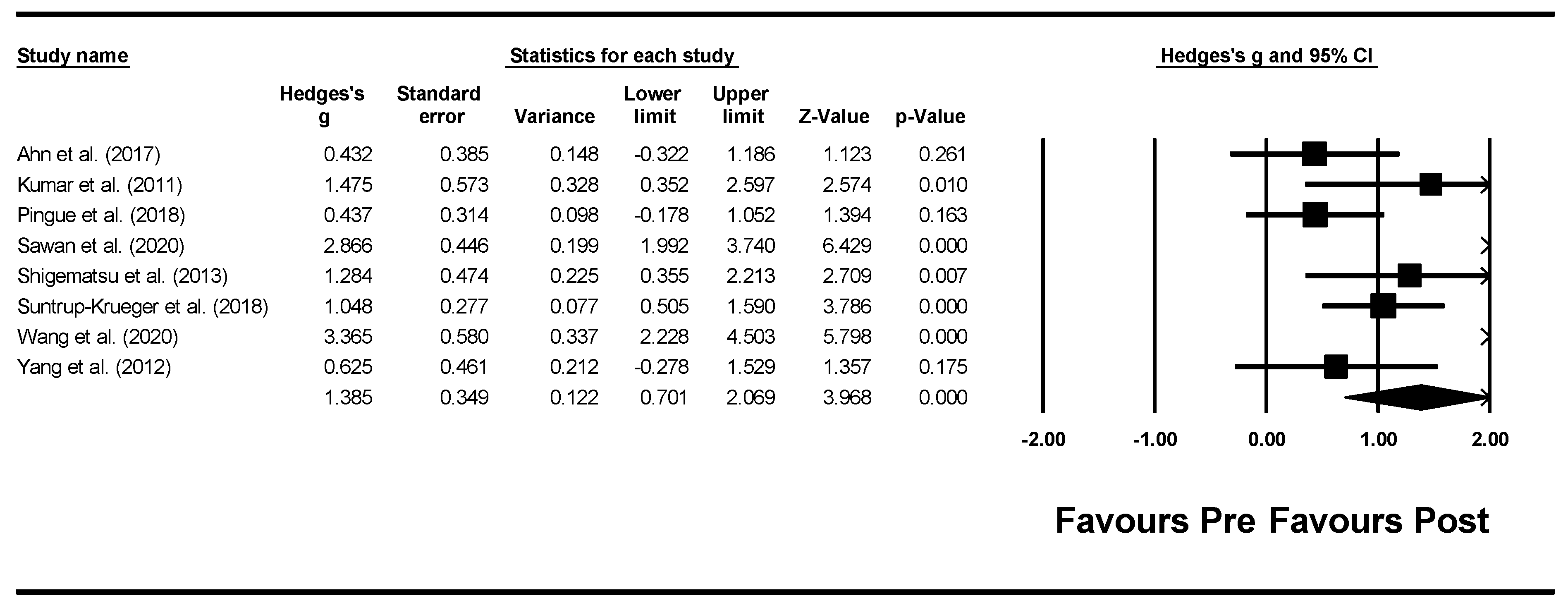
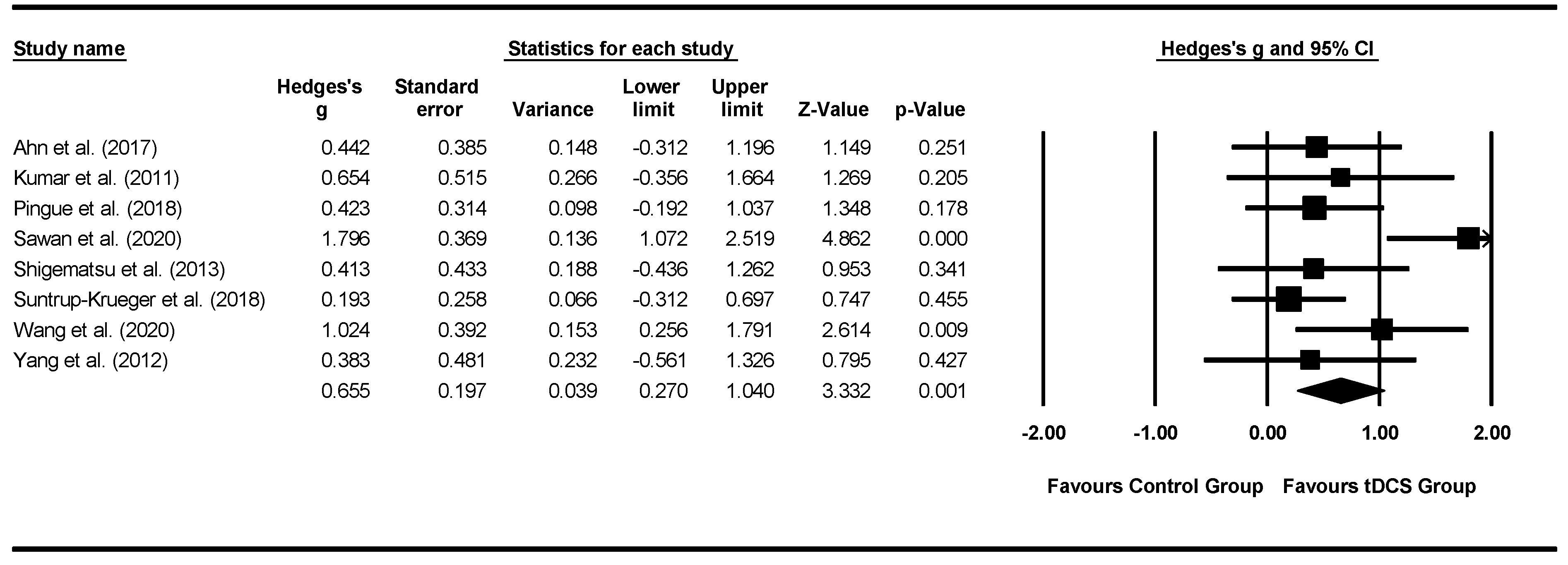
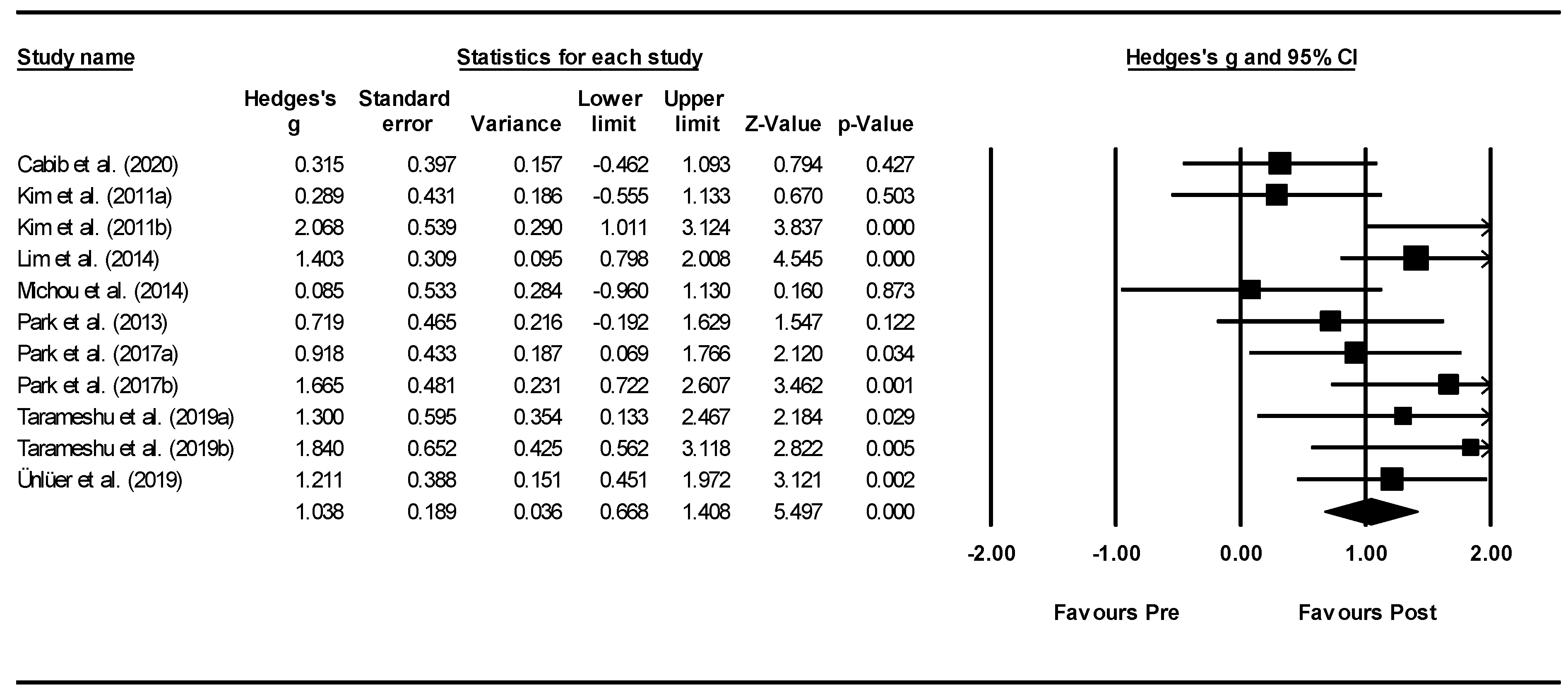
3.4. Meta-Analysis: Effects of interventions
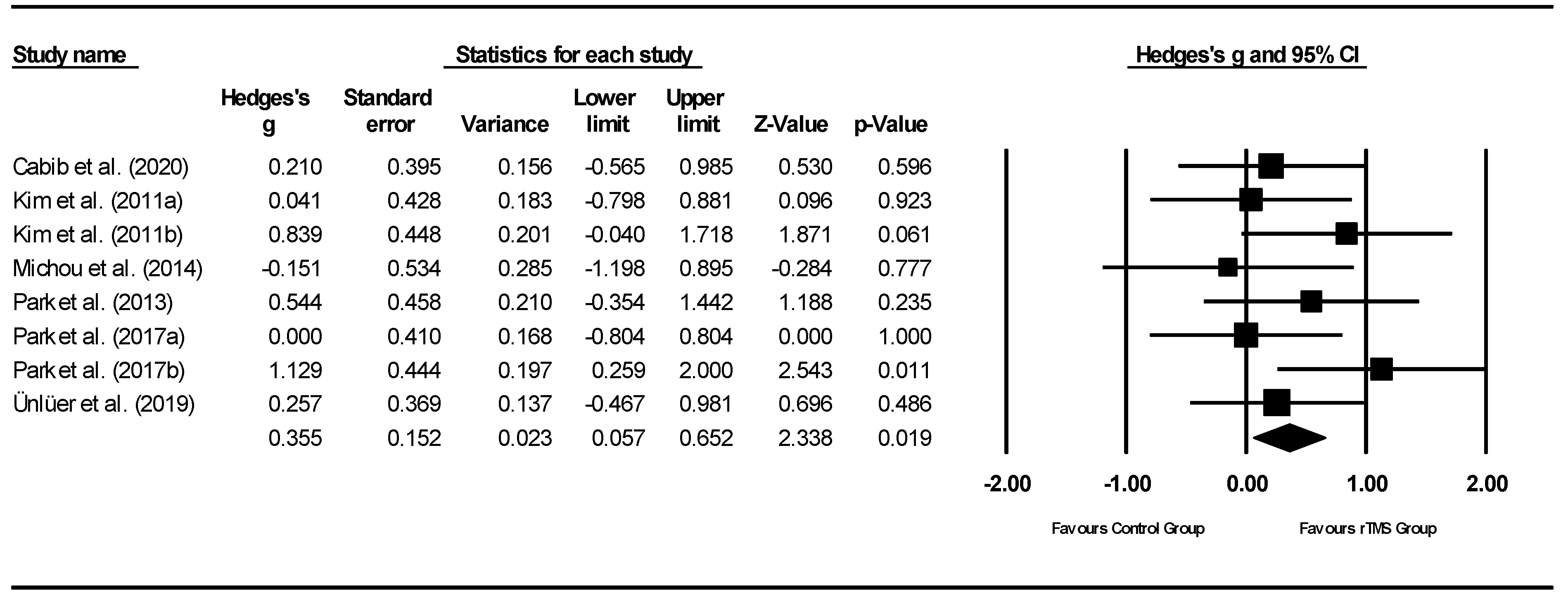
3.4.1. rTMS Meta-Analysis
3.4.2. tDCS Meta-Analysis
4. Discussion
4.1. Systematic Review Findings
4.1.1. rTMS
4.1.2. tDCS
4.1.3. Moderators
4.2. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Melgaard, D.; Westergren, A.; Skrubbeltrang, C.; Smithard, D. Interventions for nursing home residents with dyspaghia—A scoping review. Geriatrics 2021, 6, 55. [Google Scholar] [CrossRef]
- Baijens, L.W.; Clave, P.; Cras, P.; Ekberg, O.; Forster, A.; Kolb, G.F.; Leners, J.C.; Masiero, S.; Mateos-Nozal, J.; Ortego, O.; et al. European Society for Swallowing Disorders—European Union Geriatric Medicine Society white paper: Oropharyngeal dysphagia as a geriatric syndrome. Clin. Interv. Aging 2016, 11, 1403–1428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takizawa, C.; Gemmell, E.; Kenworthy, J.; Speyer, R. A systematic review of the prevalence of oropharyngeal dysphagia in stroke, Parkinson’s disease, Alzheimer’s disease, head injury, and pneumonia. Dysphagia 2016, 31, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Ciarán, K.; Regan, R.; Balding, L.; Higgins, S.; O’Leary, N.; Kelleher, F.; McDermott, R.; Armstrong, J.; Mihai, A.; Tiernan, E.; et al. Dysphagia prevalence and predictors in cancers outside the head, neck, and upper gastrointestinal tract. J. Pain Symptom Manag. 2019, 58, 949–958.e942. [Google Scholar]
- Seo, Z.W.; Min, J.H.; Huh, S.; Shin, Y.I.; Ko, H.Y.; Ko, S.H. Prevalence and severity of dysphagia using videofluoroscopic swallowing study in patients with aspiration pneumonia. Lung 2021, 199, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Eytan, D.F.; Blackford, A.L.; Eisele, D.W.; Fakhry, C. Prevalence of comorbidities among older head and neck cancer survivors in the United States. Head Neck Surg. 2019, 160, 85–92. [Google Scholar] [CrossRef]
- da Silva, A.F.; Moreira, E.A.M.; Barni, G.C.; Panza, V.S.P.; Furkim, A.M.; Moreno, Y.M.F. Relationships between high comorbidity index and nutritional parameters in patients with oropharyngeal dysphagia. Clin. Nutr. ESPEN 2020, 38, 218–222. [Google Scholar] [CrossRef]
- Jones, E.; Speyer, R.; Kertscher, B.; Swan, K.; Wagg, B.; Cordier, R. Health-related quality of life in oropharyngeal dysphagia. Dysphagia 2018, 33, 141–172. [Google Scholar] [CrossRef] [Green Version]
- Speyer, R. (Ed.) Behavioural Treatment of Oropharyngeal Dysphagia; Springer: Berlin/Heidelberg, Germany, 2018. [Google Scholar]
- Cheng, I.; Sasegbon, A.; Hamdy, S. Effects of neurostimulation on poststroke dysphagia: A synthesis of current evidence from randomised controlled trials. Neuromodulation Technol. Neural Interface 2021, 24, 1388–1401. [Google Scholar] [CrossRef]
- Cheng, I.; Hamdy, S. Metaplasticity in the human swallowing system: Clinical implications for dysphagia rehabilitation. Neurol. Sci. 2021, 1–11. [Google Scholar] [CrossRef]
- Chiang, C.-F.; Lin, M.-T.; Hsiao, M.-Y.; Yeh, Y.-C.; Liang, Y.-C.; Wang, T.-G. Comparative efficacy of noninvasive neurostimulation therapies for acute and subacute poststroke dysphagia: A systematic review and network meta-analysis. Arch. Phys. Med. Rehabil. 2019, 100, 739–750. [Google Scholar] [CrossRef] [PubMed]
- Bath, P.; Lee, H.; Everton, L. Swallowing therapy for dysphagia in acute and subacute stroke (Review). Cochrane Database Syst. Rev. 2018, 10, CD000323. [Google Scholar] [CrossRef] [PubMed]
- Dionísioa, A.; Duartea, I.; Patrícioc, M.; Castelo-Brancoa, M. Transcranial magnetic stimulation as an intervention tool to recover from language, swallowing and attentional deficits after stroke: A systematic review. Cerebrovasc. Dis. 2018, 18, 176–183. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Xing, G.; Guo, Z.; Jin, Y.; Tang, Q.; He, B.; McClure, M.; Liu, H.; Chen, H.; Mu, Q. Repetitive transcranial magnetic stimulation as an alternative therapy for dysphagia after stroke: A systematic review and meta-analysis. Clin. Rehabil. 2017, 31, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Momosaki, R.; Kinoshita, S.; Kakuda, W.; Yamada, N.; Abo, M. Noninvasive brain stimulation for dysphagia after acquired brain injury: A systematic review. J. Med. Investig. 2016, 63, 153–158. [Google Scholar] [CrossRef] [Green Version]
- Pisegna, J.; Kaneoka, A.; Pearson, W., Jr.; Kumar, S.; Langmore, S. Effects of non-invasive brain stimulation on post-stroke dysphagia: A systematic review and meta-analysis of randomized controlled trials. Clin. Neurophysiol. 2016, 127, 956–968. [Google Scholar] [CrossRef] [Green Version]
- Marchina, S.; Pisegna, J.; Massaro, J.M.; Langmore, S.E.; McVey, C.; Wang, J.; Kumar, S. Transcranial direct current stimulation for post-stroke dysphagia: A systematic review and meta-analysis of randomized controlled trials. J. Neurol. 2021, 268, 293–304. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Moher, D.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Int. J. Surg. 2021, 88, 105906. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; McKenzie, J.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372. [Google Scholar] [CrossRef]
- Sterne, J.; Savović, J.; Page, M.; Elbers, R.; Blencowe, N.; Boutron, I.; Cates, C.; Cheng, H.-Y.; Corbett, M.; Eldridge, S.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366. [Google Scholar] [CrossRef] [Green Version]
- Borenstein, M.; Hedges, L.; Higgins, J.; Rothstein, H. Comprehensive Meta-Analysis; SBiostat, Inc.: Englewood, NJ, USA, 2014; Volume 3. [Google Scholar]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, J. Statistical Power Analysis for the Behavioural Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Begg, C.B.; Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 1994, 50, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- Rosenthal, R. The file drawer problem and tolerance for null results. Psychol. Bull. 1979, 86, 638–664. [Google Scholar] [CrossRef]
- Kumar, S.; Wagner, C.W.; Frayne, C.; Zhu, L.; Selim, M.; Feng, W.; Schlaug, G. Noninvasive brain stimulation may improve stroke-related dysphagia: A pilot study. Stroke 2011, 42, 1035–1040. [Google Scholar] [CrossRef] [Green Version]
- Zhang, R.; Ju, X.-M. Clinical improvement of nursing intervention in swallowing dysfunction of elderly stroke patients. Biomed. Res. 2018, 29, 1099–1102. [Google Scholar] [CrossRef] [Green Version]
- Sawan, S.A.E.; Reda, A.M.; Kamel, A.H.; Ali, M.A.M. Transcranial direct current stimulation (tDCS): Its effect on improving dysphagia in stroke patients. Egypt. J. Neurol. Psychiatry Neurosurg. 2020, 56, 1–7. [Google Scholar] [CrossRef]
- Khedr, E.M.; Mohamed, K.O.; Soliman, R.K.; Hassan, A.M.M.; Rothwell, J. The Effect of high-frequency repetitive transcranial magnetic stimulation on advancing Parkinson’s Disease with dyspahagia: Double blind randomised clinical trial. Neurorehabilit. Neural Repair 2019, 33, 442–452. [Google Scholar] [CrossRef]
- Cosentino, G.; Tassorelli, C.; Preunetti, P.; Bertino, G.; De Icco, R.; Todisco, M.; Di Marco, S.; Brighina, F.; Schindler, A.; Rondanelli, M.; et al. Anodal transcranial direct current stimulation and intermittent theta-burst stimulation deglutition and swallowing reproducibility in elderly patients with dysphagia. Neurogastroenterol. Motil. 2020, 32, e13791. [Google Scholar] [CrossRef]
- Kim, L.; Chun, M.H.; Kim, B.R.; Lee, S.J. Effect of repetitive transcranial magnetic stimulation on patients with brain injury and dysphagia. Ann. Rehabil. Med. 2011, 35, 765–771. [Google Scholar] [CrossRef]
- Tarameshlu, M.; Ansari, N.N.; Ghelichi, L.; Jalaei, S. The effect of repetitive transcranial magnetic stimulation combined with traditional dysphagia therapy on poststroke dysphagia: A pilot double-blinded randomized-controlled trial. Int. J. Rehabil. Res. 2019, 42, 133–138. [Google Scholar] [CrossRef]
- Du, J.; Yang, F.; Liu, L.; Hu, J.; Cai, B.; Liu, W.; Xu, G.; Liu, X. Repetitive transcranial magnetic stimulation for poststroke dysphagia: A randomised, double-blind clinical trial. Clin. Neurophysiol. 2016, 127, 1907–1913. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Kim, M.S.; Chang, W.H.; Oh, S.M.; Kim, Y.K.; Lee, A.; Kim, Y.-H. Effects of bilateral repetitive transcranial magnetic stimulation on post-stroke dysphagia. Brain Stimul. 2017, 10, 75–82. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.H.; Sohn, H.-J.; Park, J.-S.; Ahn, T.G.; Shin, Y.B.; Park, M.; Ko, S.-H.; Shin, Y.-I. Effect of bihemispheric anodal transcranial direct current stimulation for dysphagia in chronic stroke patients: A randomized control trial. J. Rehabil. Med. 2017, 49, 30–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pingue, V.; Priori, A.; Malovini, A.; Pistarini, C. Dual transcranial direct current stimulation for post-stroke dysphagia: A randomised controlled trial. Neurorehabilit. Neural Repair 2018, 32, 635–644. [Google Scholar] [CrossRef] [Green Version]
- Shigematsu, T.; Fujishima, I.; Ohno, K. Transcranial direct current stimulation improves swallowing function in stroke patients. Neurorehabilit. Neural Repair 2013, 27, 363–369. [Google Scholar] [CrossRef]
- Suntrup-Krueger, S.; Ringmaier, C.; Muhle, P.; Wollbrink, A.; Kemmling, A.; Hanning, U.; Claus, I.; Warnecke, T.; Teismann, I.; Pantev, C.; et al. Randomized trial of transcranial direct current stimulation for poststroke dysphagia. Ann. Neurol. 2018, 83, 328–340. [Google Scholar] [CrossRef]
- Wang, Z.-Y.; Chen, J.-M.; Lin, Z.-k.; Ni, G.-X. Transcranial direct current stimulation improves the swallowing function in patients with cricopharyngeal muscle dysfunction following a brainstem stroke. Neurol. Sci. 2020, 41, 569–574. [Google Scholar] [CrossRef]
- Yang, E.J.; Baek, S.-R.; Shin, J.; Lim, J.Y.; Jang, H.J.; Kim, Y.K.; Paik, N.-J. Effects of transcranial direct current stimulation (tDCS) on post-stroke dysphagia. Restor. Neurol. Neurosci. 2012, 30, 303–311. [Google Scholar] [CrossRef]
- Michou, E.; Mistry, S.; Jefferson, S.; Tyrrell, P.; Hamdy, S. Characterizing the mechanisms of central and peripheral forms of neurostimulation in chronic dysphagic stroke patients. Brain Stimul. 2014, 7, 66–73. [Google Scholar] [CrossRef]
- Lim, K.-L.; Lee, H.-J.; Yoo, J.; Kwon, Y.-G. Effect of low-frequency rTMS and NMES on subacute unilateral hemispheric stroke with dysphagia. Ann. Rehabil. Med. 2014, 38, 592–602. [Google Scholar] [CrossRef]
- Cabib, C.; Nascimento, W.; Rofes, L.; Arreola, V.; Tomsen, N.; Mundet, L.; Palomeras, E.; Michou, E.; Clavé, P.; Ortega, O. Short-term neurophysiological effects of sensory pathway neurorehabilitation strategies on chronic poststroke oropharyngeal dysphagia. Neurogastroenterol. Motil. 2020, 32, e13887. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zheng, X.; Lu, R.; Yun, W.; Yun, H.; Zhou, X. Repetitive transcranial magnetic stimulation in combination with neuromuscular electrical stimulation for treatment of post-stroke dysphagia. J. Int. Med. Res. 2019, 47, 662–672. [Google Scholar] [CrossRef] [PubMed]
- Cheng, I.K.Y.; Chan, K.M.K.; Wong, C.S.; Li, L.S.W.; Chiu, K.M.Y.; Cheung, R.T.F.; Yiu, E.M.L. Neronavigated high-frequency repetitive transcranial magnetic stimulation for chronic post-stroke dysphagia: A randomized controlled trial. J. Rehabil. Med. 2017, 49, 476–481. [Google Scholar] [CrossRef] [Green Version]
- Khedr, E.M.; Abo-Elfetoh, N.; Rothwell, J. Treatment of post-stroke dysphagia with repetitive transcranial magnetic stimulation. Acta Neurol. Scand. 2009, 119, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Khedr, E.M.; Abo-Elfetoh, N. Therapeutic role of rTMS on recovery of dysphagia in patients with lateral medullary syndrome and brainstem infarction. J. Neurol. Neurosurg. Psychiatry 2010, 81, 495–499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Momosaki, R.; Abo, M.; Watanabe, S.; Kakuda, W.; Yamada, N.; Mochio, K. Functional magnetic stimulation using a parabolic coil for dysphagia after stroke. Neuromodulation 2014, 17, 637–641. [Google Scholar] [CrossRef]
- Park, J.-W.; Oh, J.-C.; Lee, J.-W.; Yeo, J.-S.; Ryu, K.H. The effect of 5Hz high-frequency rTMS over contralesional pharyngeal motor cortex in post-stroke oropharyngeal dysphagia: A randomized controlled study. Neurogastroenterol. Motil. 2013, 25, 324-e250. [Google Scholar] [CrossRef]
- Ünlüer, N.O.; Temuçin, C.M.; Demir, N.; Arslan, S.S.; Karaduman, A. Effects of low-frequency repetitive transcranial magnetic stimulation on swallowing function and quality of life of post-stroke patients. Dysphagia 2019, 34, 360–371. [Google Scholar] [CrossRef]
- Chan, K.; Yiu, E.; Ho, E. The impact of swallowing problems on nursing home residents’ quality of life. In Proceedings of the International Symposium on Healthy Aging, ISHA, Hongkong, China, 5–6 March 2011. [Google Scholar]
- Perry, L. Screening swallowing function of patients with acute stroke. Part two: Detailed evaluation of the tool used by nurses. J. Clin. Nurs. 2001, 10, 474–481. [Google Scholar] [CrossRef]
- Suiter, D.M.; Leder, S.B. Clinical utility of the 3-ounce water swallow test. Dysphagia 2008, 23, 244–250. [Google Scholar] [CrossRef]
- Ertekin, C.; Aydogdu, I.; Yüceyar, N.; Kiylioglu, N.; Tarlaci, S.; Uludag, B. Pathophysiological mechanisms of oropharyngeal dysphagia in amyotrophic lateral sclerosis. Brain 2000, 123, 125–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brott, T.; Adams, H.P., Jr.; Olinger, C.P.; Marler, J.R.; Barsan, W.G.; Biller, J.; Spilker, J.; Holleran, R.; Eberle, R.; Hertzberg, V. Measurements of acute cerebral infarction: A clinical examination scale. Stroke 1989, 20, 864–870. [Google Scholar] [CrossRef] [Green Version]
- Mahoney, F.I. Functional evaluation: The Barthel index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar]
- O’Neil, K.H.; Purdy, M.; Falk, J.; Gallo, L. The dysphagia outcome and severity scale. Dysphagia 1999, 14, 139–145. [Google Scholar] [CrossRef]
- Zhao, Y.J.; Wee, H.L.; Chan, Y.H.; Seah, S.H.; Au, W.L.; Lau, P.N.; Pica, E.C.; Li, S.C.; Luo, N.; Tan, L.C. Progression of Parkinson’s disease as evaluated by Hoehn and Yahr stage transition times. Mov. Disord. 2010, 25, 710–716. [Google Scholar] [CrossRef]
- Fahn, S.; Elton, R. UPDRS program members. Unified Parkinsons disease rating scale. Recent Dev. Parkinson’s Dis. 1987, 2, 153–163. [Google Scholar]
- Lawton, M.; Brody, E.; Médecin, U. Instrumental activities of daily living (IADL). Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Brown, R.G.; MacCarthy, B.; Jahanshahi, M.; Marsden, C.D. Accuracy of self-reported disability in patients with parkinsonism. Arch. Neurol. 1989, 46, 955–959. [Google Scholar] [CrossRef]
- Cohen, J.T.; Manor, Y. Swallowing disturbance questionnaire for detecting dysphagia. Laryngoscope 2011, 121, 1383–1387. [Google Scholar] [CrossRef] [PubMed]
- Farahat, M.; Malki, K.H.; Mesallam, T.A.; Bukhari, M.; Alharethy, S. Development of the arabic version of dysphagia handicap index (dhi). Dysphagia 2014, 29, 459–467. [Google Scholar] [CrossRef]
- Han, T.R.; Paik, N.-J.; Park, J.W. Quantifying swallowing function after stroke: A functional dysphagia scale based on videofluoroscopic studies. Arch. Phys. Med. Rehabil. 2001, 82, 677–682. [Google Scholar] [CrossRef] [PubMed]
- Rosenbek, J.C.; Robbins, J.A.; Roecker, E.B.; Coyle, J.L.; Wood, J.L. A penetration-aspiration scale. Dysphagia 1996, 11, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Mullen, R. Evidence for whom?: ASHA’s National outcomes measurement system. J. Commun. Disord. 2004, 37, 413–417. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Oh, B.-M.; Kim, J.Y.; Lee, G.J.; Lee, S.A.; Han, T.R. Validation of the videofluoroscopic dysphagia scale in various etiologies. Dysphagia 2014, 29, 438–443. [Google Scholar] [CrossRef]
- Jung, S.H.; Lee, K.J.; Hong, J.B.; Han, T.R. Validation of clinical dysphagia scale: Based on videofluoroscopic swallowing study. J. Korean Acad. Rehabil. Med. 2005, 29, 343–350. [Google Scholar]
- Carnaby-Mann, G.; Lenius, K.; Crary, M.A. Update on assessment and management of dysphagia post stroke. Northeast Fla. Med. 2007, 58, 31–34. [Google Scholar]
- Crary, M.A.; Mann, G.D.C.; Groher, M.E. Initial psychometric assessment of a functional oral intake scale for dysphagia in stroke patients. Arch. Phys. Med. Rehabil. 2005, 86, 1516–1520. [Google Scholar] [CrossRef]
- Scutt, P.; Lee, H.S.; Hamdy, S.; Bath, P.M. Pharyngeal electrical stimulation for treatment of poststroke dysphagia: Individual patient data meta-analysis of randomised controlled trials. Stroke Res. Treat. 2015, 2015. [Google Scholar] [CrossRef] [Green Version]
- Martin-Harris, B.; Brodsky, M.B.; Michel, Y.; Castell, D.O.; Schleicher, M.; Sandidge, J.; Maxwell, R.; Blair, J. MBS measurement tool for swallow impairment—MBSImp: Establishing a standard. Dysphagia 2008, 23, 392–405. [Google Scholar] [CrossRef] [Green Version]
- Ertekin, C.; Turman, B.; Tarlaci, S.; Celik, M.; Aydogdu, I.; Secil, Y.; Kiylioglu, N. Cricopharyngeal sphincter muscle responses to transcranial magnetic stimulation in normal subjects and in patients with dysphagia. Clin. Neurophysiol. 2001, 112, 86–94. [Google Scholar] [CrossRef]
- Speyer, R.; Sutt, A.-l.; Bergström, L.; Hamdy, S.; Heijnen, B.J.; Remijn, L.; Wilkes-Gillan, S.; Cordier, R. Neurostimulation in people with oropharyngeal dysphagia: A systematic review and meta-analyses of randomised controlled trials. Part I: Pharyngeal and neuromuscular electrical stimulation. J. Clin. Med. 2022, 11, 776. [Google Scholar] [CrossRef]


| Database and Search Terms | Number of Records |
|---|---|
| Cinahl: ((MH “Deglutition”) OR (MH “Deglutition Disorders”)) AND (MH “Randomized Controlled Trials”) | 239 |
| Embase: (swallowing/OR dysphagia/) AND (randomization/or randomized controlled trial/OR “randomized controlled trial (topic)”/OR controlled clinical trial/) | 4550 |
| PsycINFO: (swallowing/OR dysphagia/) AND (RCT OR (Randomised AND Controlled AND Trial) OR (Randomized AND Clinical AND Trial) OR (Randomised AND Clinical AND Trial) OR (Controlled AND Clinical AND Trial)).af. | 231 |
| PubMed: (“Deglutition” [Mesh] OR “Deglutition Disorders” [Mesh]) AND (“Randomized Controlled Trial” [Publication Type] OR “Randomized Controlled Trials as Topic” [Mesh] OR “Controlled Clinical Trial” [Publication Type] OR “Pragmatic Clinical Trials as Topic” [Mesh]) | 3039 |
| Neurostimulation | Subgroup | Hedges’ g | Lower Limit CI | Upper Limit CI | Z-Value | p-Value |
|---|---|---|---|---|---|---|
| rTMS | Time between pre-post (days) | |||||
| 1 (n = 2) | 0.082 | −0.541 | 0.704 | 0.257 | 0.797 | |
| 5 (n = 1) | 0.257 | −0.467 | −0.981 | 0.696 | 0.486 | |
| 14 (n = 5) | 0.491 | 0.054 | 0.929 | 2.202 | 0.028 * | |
| Stimulation site | ||||||
| Bilateral (n = 2) | 0.523 | −0.730 | 1.776 | 0.818 | 0.413 | |
| Contra-lesional (n = 3) | 0.315 | −0.141 | 0.771 | 1.353 | 0.176 | |
| Ipsi-lesional (n = 3) | 0.272 | −0.251 | 0.795 | 1.020 | 0.308 | |
| Pulse range | ||||||
| Low [≤ 600] (n = 2) | 0.082 | −0.541 | 0.704 | 0.257 | 0.797 | |
| Medium [> 600 and < 10000] (n = 3) | 0.248 | −0.213 | 0.710 | 1.054 | 0.292 | |
| High [≥ 10000] (n = 3) | 0.660 | 0.014 | 1.306 | 2.004 | 0.045 * | |
| Stimulation frequency (Hz) | ||||||
| 1 (n = 2) | 0.492 | −0.067 | 1.052 | 1.726 | 0.084 | |
| 5 (n = 4) | 0.180 | −0.257 | 0.617 | 0.809 | 0.419 | |
| 10 (n = 2) | 0.552 | −0.555 | 1.658 | 0.978 | 0.328 | |
| Behavioural training | ||||||
| rTMS + DT (n = 1) | 0.257 | −0.467 | 0.981 | 0.696 | 0.486 | |
| rTMS (n = 7) | 0.375 | 0.031 | 0.720 | 2.135 | 0.033 * | |
| tDCS | Time between pre-post (days) | |||||
| 4 (n = 1) | 0.193 | −0.312 | 0.697 | 0.747 | 0.455 | |
| 5 (n = 1) | 0.654 | −0.356 | 1.664 | 1.269 | 0.205 | |
| 10 (n = 1) | 0.432 | −0.192 | 1.037 | 1.348 | 0.178 | |
| 14 (n = 4) | 0.784 | 0.056 | 1.512 | 2.112 | 0.035 * | |
| 28 (n = 1) | 1.024 | 0.256 | 1.791 | 2.614 | 0.009 * | |
| Outcome measures | ||||||
| DOSS (n = 5) | 0.753 | 0.195 | 1.311 | 2.644 | 0.008 * | |
| DSRS (n = 1) | 0.193 | −0.312 | 0.697 | 0.747 | 0.455 | |
| FDS (n = 2) | 0.764 | 0.147 | 1.381 | 2.428 | 0.015 * | |
| Total stimulation time (min) | ||||||
| 80 (n = 1) | 0.193 | −0.312 | 0.697 | 0.747 | 0.455 | |
| 150 (n = 1) | 0.654 | −0.356 | 1.664 | 1.269 | 0.205 | |
| 200 (n = 4) | 0.419 | 0.039 | 0.799 | 2.161 | 0.031 * | |
| 300 (n = 1) | 1.796 | 1.072 | 2.519 | 4.862 | <0.001 * | |
| 400 (n = 1) | 1.024 | 0.256 | 1.791 | 2.614 | 0.009 * | |
| Stimulation current (mA) | ||||||
| 1 (n = 6) | 0.430 | 0.148 | 0.712 | 2.985 | 0.003 * | |
| 2 (n = 2) | 1.281 | 0.168 | 2.395 | 2.256 | 0.024 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Speyer, R.; Sutt, A.-L.; Bergström, L.; Hamdy, S.; Pommée, T.; Balaguer, M.; Kaale, A.; Cordier, R. Neurostimulation in People with Oropharyngeal Dysphagia: A Systematic Review and Meta-Analysis of Randomised Controlled Trials—Part II: Brain Neurostimulation. J. Clin. Med. 2022, 11, 993. https://doi.org/10.3390/jcm11040993
Speyer R, Sutt A-L, Bergström L, Hamdy S, Pommée T, Balaguer M, Kaale A, Cordier R. Neurostimulation in People with Oropharyngeal Dysphagia: A Systematic Review and Meta-Analysis of Randomised Controlled Trials—Part II: Brain Neurostimulation. Journal of Clinical Medicine. 2022; 11(4):993. https://doi.org/10.3390/jcm11040993
Chicago/Turabian StyleSpeyer, Renée, Anna-Liisa Sutt, Liza Bergström, Shaheen Hamdy, Timothy Pommée, Mathieu Balaguer, Anett Kaale, and Reinie Cordier. 2022. "Neurostimulation in People with Oropharyngeal Dysphagia: A Systematic Review and Meta-Analysis of Randomised Controlled Trials—Part II: Brain Neurostimulation" Journal of Clinical Medicine 11, no. 4: 993. https://doi.org/10.3390/jcm11040993
APA StyleSpeyer, R., Sutt, A.-L., Bergström, L., Hamdy, S., Pommée, T., Balaguer, M., Kaale, A., & Cordier, R. (2022). Neurostimulation in People with Oropharyngeal Dysphagia: A Systematic Review and Meta-Analysis of Randomised Controlled Trials—Part II: Brain Neurostimulation. Journal of Clinical Medicine, 11(4), 993. https://doi.org/10.3390/jcm11040993