Changes in EEG Recordings in COVID-19 Patients as a Basis for More Accurate QEEG Diagnostics and EEG Neurofeedback Therapy: A Systematic Review
Abstract
1. Introduction
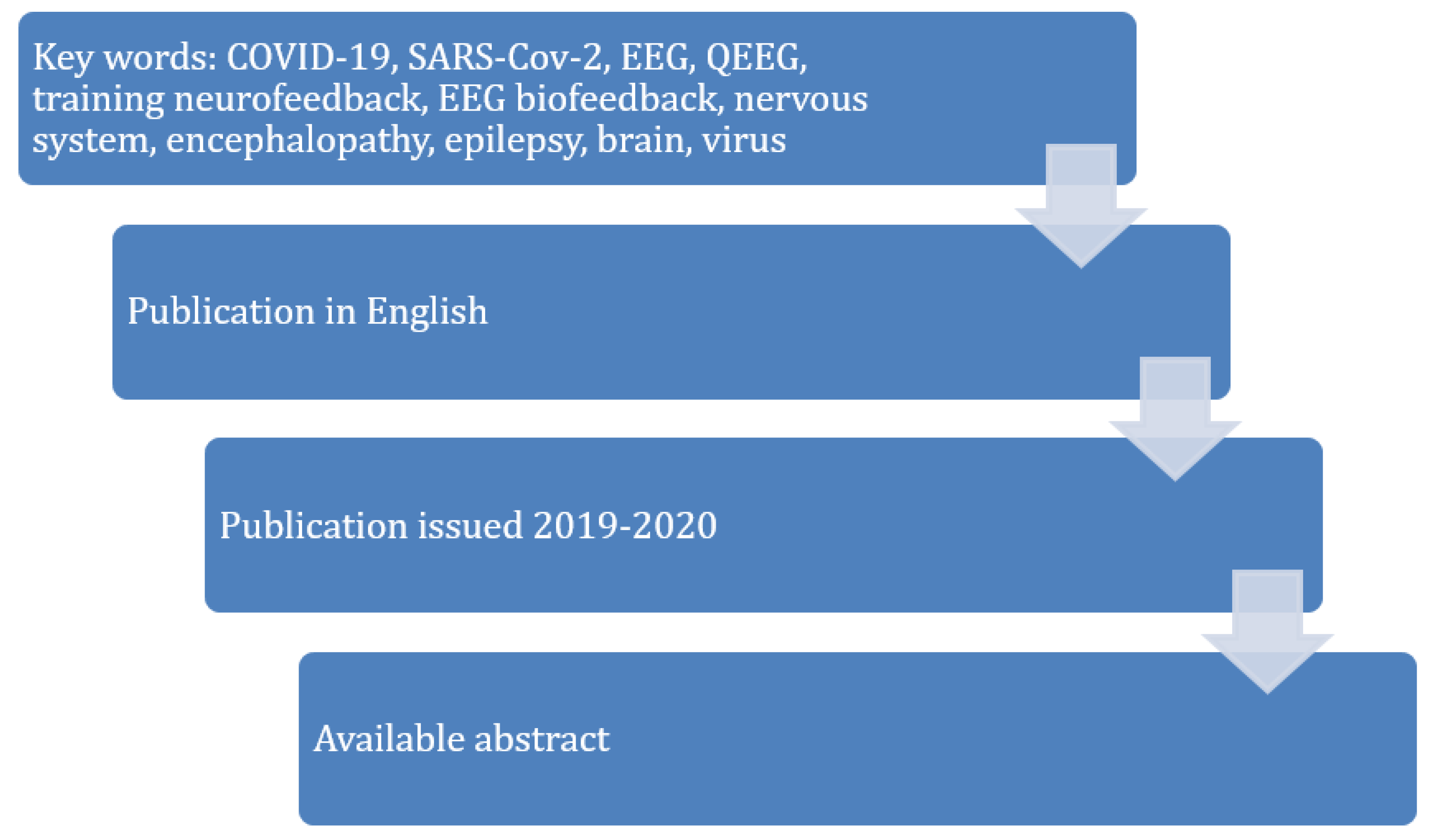
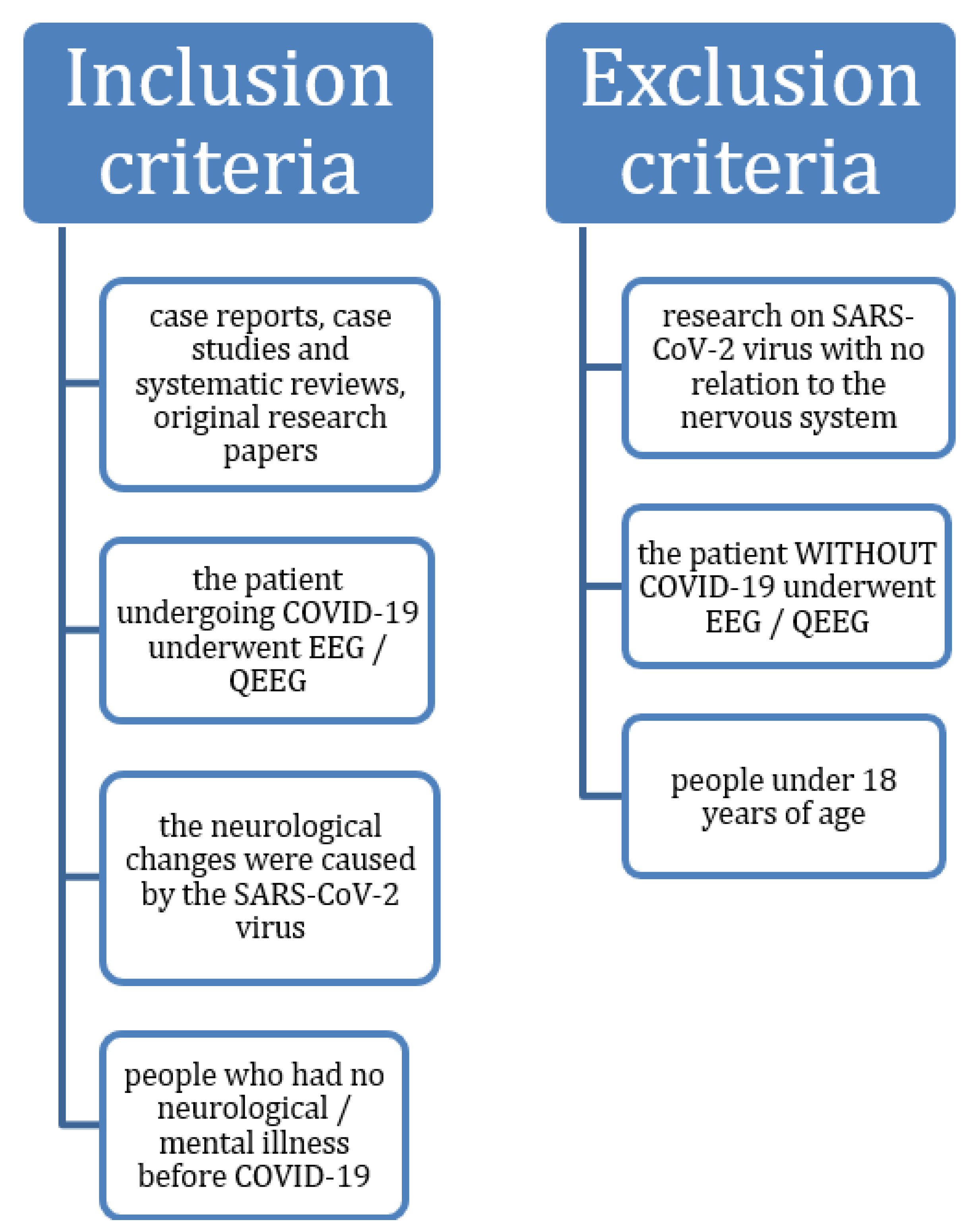
2. Objective
3. Literature Review
4. Description of the State of Knowledge
5. Results
6. Discussion/Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- St. Louis, E.K.; Frey, L.C.; Britton, J.W.; Frey, M.D.; Hopp, J.L.; Korb, P.; Koubeissi, M.Z.; Lievens, W.E.; Pestana-Knight, E.M. Electroencephalography (EEG): An Introductory Text and Atlas of Normal and Abnormal Findings in Adults, Children, and Infants; American Epilepsy Society: Chicago, IL, USA, 2016. [Google Scholar]
- Borkowski, P. EEG and QEEG Atlas, 1st ed.; Biomed Neurotechnologie: Wrocław, Poland, 2017; pp. 13–264. [Google Scholar]
- Kesebir, S.; Yosmaoğlu, A. QEEG in affective disorder: About to be a biomarker, endophenotype and predictor of treatment response. Heliyon 2018, 4, e00741. [Google Scholar] [CrossRef]
- Hasey, G.M.; Kiang, M. A Review of Recent Literature Employing Electroencephalographic Techniques to Study the Pathophysiology, Phenomenology and Treatment Response of Schizophrenia. Curr. Psychiatry Rep. 2013, 15, 388. [Google Scholar] [CrossRef]
- McVoy, M.; Lytle, S.; Fulchiero, E.; Aebi, M.E.; Adeleye, O.; Sajatovic, M. A systematic review of quantitative EEG as a possible biomarker in child psychiatric disorders. Psychiatry Res. 2019, 279, 331–344. [Google Scholar] [CrossRef]
- Bartel, P.; Schutte, C.M.; Becker, P.; van der Meyden, C. Discrimination between viral and nonviral meningitis by visually analyzed and quantitative electroencephalography. Clin. Electroencephalogr. 1999, 30, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Baldeweg, T.; Gruzelier, J.H. Alpha EEG activity and subcortical pathology in HIV infection. Int. J. Psychophysiol. 1997, 26, 431–442. [Google Scholar] [CrossRef]
- Adamis, D.; Sahu, S.; Treloar, A. The utility of EEG in dementia: A clinical perspective. Int. J. Geriatr. Psychiatry 2005, 20, 1038–1045. [Google Scholar] [CrossRef] [PubMed]
- Appel, S.; Cohen, O.S.; Chapman, J.; Gilat, S.; Rosenmann, H.; Nitsan, Z.; Kahan, E.; Blatt, I. The association of quantitative EEG and MRI in Creutzfeldt-Jakob Disease. Acta Neurol. Scand. 2019, 140, 366–371. [Google Scholar] [CrossRef]
- Luijmes, R.E.; Pouwels, S.; Boonman, J. The effectiveness of neurofeedback on cognitive functioning in patients with Alzheimer’s disease: Preliminary results. Neurophysiol. Clin. 2016, 46, 179–187. [Google Scholar] [CrossRef]
- Coben, R.; Myers, T.E. The relative efficacy of connectivity guided and symptom based EEG biofeedback for autistic disorders. Appl. Psychophysiol. Biofeedback 2010, 35, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Krepel, N.; Egtberts, T.; Sack, A.T.; Heinrich, H.; Ryan, M.; Arns, M. A multicenter effectiveness trial of QEEG-informed neurofeedback in ADHD: Replication and treatment prediction. Neuroimage Clin. 2020, 28, 102399. [Google Scholar] [CrossRef]
- Pasini, E.; Bisulli, F.; Volpi, L.; Minardi, I.; Tappatà, M.; Muccioli, L.; Pensato, U.; Riguzzi, P.; Tinuper, P.; Michelucci, R. EEG findings in COVID-19 related encephalopathy. Clin. Neurophysiol. 2020, 131, 2265–2267. [Google Scholar] [CrossRef] [PubMed]
- Vellieux, G.; Rouvel-Tallec, A.; Jaquet, P.; Grinea, A.; Sonneville, R.; d’Ortho, M.P. COVID-19 associated encephalopathy: Is there a specific EEG pattern? Clin. Neurophysiol. 2020, 131, 1928–1930. [Google Scholar] [CrossRef] [PubMed]
- Pastor, J.; Vega-Zelaya, L.; Martín Abad, E. Specific EEG Encephalopathy Pattern in SARS-CoV-2 Patients. J. Clin. Med. 2020, 9, 1545. [Google Scholar] [CrossRef] [PubMed]
- Koutroumanidis, M.; Gratwicke, J.; Sharma, S.; Whelan, A.; Tan, S.V.; Glover, G. Alpha coma EEG pattern in patients with severe COVID-19 related encephalopathy. Clin. Neurophysiol. 2021, 132, 218–225. [Google Scholar] [CrossRef]
- Flamand, M.; Perron, A.; Buron, Y.; Szurhaj, W. Pay more attention to EEG in COVID-19 pandemic. Clin. Neurophysiol. 2021, 131, 2062–2064. [Google Scholar] [CrossRef]
- Galanopoulou, A.S.; Ferastraoaru, V.; Correa, D.J.; Cherian, K.; Duberstein, S.; Gursky, J.; Hanumanthu, R.; Hung, C.; Molinero, I.; Khodakivska, O.; et al. EEG findings in acutely ill patients investigated for SARS-CoV-2/COVID-19: A small case series preliminary report. Epilepsia Open 2020, 5, 314–324. [Google Scholar] [CrossRef]
- Chen, W.; Toprani, S.; Werbaneth, K.; Falco-Walter, J. Status epilepticus and other EEG findings in patients with COVID-19: A case series. Seizure 2020, 81, 198–200. [Google Scholar] [CrossRef]
- De Stefano, P.; Nencha, U.; De Stefano, L.; Mégevand, P.; Seeck, M. Focal EEG changes indicating critical illness associated cerebral microbleeds in a Covid-19 patient. Clin. Neurophysiol. Pract. 2020, 5, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Cecchetti, G.; Vabanesi, M.; Chieffo, R.; Fanelli, G.; Minicucci, F.; Agosta, F.; Tresoldi, M.; Zangrillo, A.; Filippi, M. Cerebral involvement in COVID-19 is associated with metabolic and coagulation derangements: An EEG study. J. Neurol. 2020, 267, 3130–3134. [Google Scholar] [CrossRef] [PubMed]
- Antony, A.R.; Haneef, Z. Systematic review of EEG findings in 617 patients diagnosed with COVID-19. Seizure 2020, 83, 234–241. [Google Scholar] [CrossRef]
- Vespignani, H.; Colas, D.; Lavin, B.S.; Soufflet, C.; Maillard, L.; Pourcher, V.; Paccoud, O.; Medjebar, S.; Frouin, P.Y. Report on Electroencephalographic Findings in Critically Ill Patients with COVID-19. Ann. Neurol. 2020, 88, 626–630. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, T.; Yang, N.; Han, D.; Mi, X.; Li, Y.; Liu, K.; Vuylsteke, A.; Xiang, H.; Guo, X. Neurological manifestations of patients with COVID-19: Potential routes of SARS-CoV-2 neuroinvasion from the periphery to the brain. Front. Med. 2020, 14, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Asadi-Pooya, A.A.; Simani, L. Central nervous system manifestations of COVID-19: A systematic review. J. Neurol. Sci. 2020, 413, 116832. [Google Scholar] [CrossRef] [PubMed]
- Baig, A.M. Neurological manifestations in COVID-19 caused by SARS-CoV-2. CNS Neurosci. Ther. 2020, 26, 499–501. [Google Scholar] [CrossRef]
- Dixon, L.; Varley, J.; Gontsarova, A.; Mallon, D.; Tona, F.; Muir, D.; Luqmani, A.; Jenkins, I.H.; Nicholas, R.; Jones, B.; et al. COVID-19-related acute necrotizing encephalopathy with brain stem involvement in a patient with aplastic anemia. Neurol. Neuroimmunol. Neuroinflamm. 2020, 7, 789. [Google Scholar] [CrossRef]
- Gklinos, P. Neurological manifestations of COVID-19: A review of what we know so far. J. Neurol. 2020, 267, 2485–2489. [Google Scholar] [CrossRef] [PubMed]
- Egbert, A.R.; Cankurtaran, S.; Karpiak, S. Brain abnormalities in COVID-19 acute/subacute phase: A rapid systematic review. Brain Behav. Immun. 2020, 89, 543–554. [Google Scholar] [CrossRef] [PubMed]
| Researchers | Aim | Materials and Methods | Results | Conclusions |
|---|---|---|---|---|
| Pasini E et al. (2020) (experimental research paper) [13] | Checking for the presence of an encephalopathy pattern in patients with COVID-19 with diagnosed neurological symptoms. | 15 patients with suspected COVID-19 related encephalopathy. | EEG abnormalities similar to those found in patients with encephalopathy. | The SARS-CoV-2 virus can cause EEG changes; however, more research is needed. |
| Vellieux G et al. (2020) [14] (experimental research paper) | Checking whether the EEG record in patients infected with the SARS-CoV-2 virus manifests changes in the form of a new, unprecedented pattern. | A case report of two COVID-19 patients with neurological symptoms. | The EEG examination showed a new, unprecedented pattern. | EEG changes in patients with COVID-19 may be specific to their disease; however, more research is needed. |
| Pastor J et al. (2020) (experimental research paper) [15] | Using a QEEG to determine the characteristics of encephalopathy in patients with COVID-19. | A QEEG study was performed. The study group consisted of 20 hospitalized patients who had COVID-19. They were compared with two control groups. | Changes that were similar to the abnormalities found in encephalopathy were seen in patients who had a traumatic time with COVID-19. | The SARS-CoV-2 virus can cause neurological changes that are visible in the QEEG record. |
| Koutroumanidis M et al. (2020) (experimental research paper) [16] | Checking if COVID-19 related neurological changes are caused by viral neurotropism or due to hypoxia in the course of the disease. | An analysis of the encephalograms of 19 patients who underwent COVID-19. | Severe encephalopathy was found in 13 patients in the EEG. | A relatively high frequency of the rare alpha pattern may reflect direct SARS-CoV-2 neurotropism. |
| Flamand M et al. (2020) (experimental research paper) [17] | Check for EEG changes related to the SARS-CoV-2 virus. | A case report of an 80-year-old patient suffering from COVID-19 who underwent several EEG tests during her hospitalization. | The EEG record showed the development of three-phase waves. | EEG changes indicated a progressive neurological process that was possibly associated with SARS-CoV-2. This case indicated that more attention should be paid to the EEG patterns in patients during the COVID-19 pandemic. |
| Galanopoulou, A.S et al. (2020) (experimental research paper) [18] | Checking the neurological changes appearing in the EEG record in patients infected with the SARS-CoV-2 virus. | Twenty six adults were examined with an EEG (20 positive for the SARS-CoV-2 virus, six negative). | EEG changes/patterns similar to those in epilepsy appeared in 40.9% of COVID-19 positive patients. Changes with dominant frontal brain sharp waves were noticed in the record. | Future research must determine if COVID-19 infection increases the risk of the epilepsy-like abnormalities and investigate their pathogenesis. |
| Chen W et al. (2020) (experimental research paper) [19] | Assessment of EEG abnormalities in COVID-19 patients and assessment of epilepsy-like activity and seizures. | Five critically ill adults with COVID-19 underwent EEG monitoring. | All EEG recordings showed non-specific markers of encephalopathy as well as diffuse slowing and generalized rhythmic delta activity. Two patients also had epileptic discharges ranging from 2–3 Hz. The improvement in the EEG record and clinical symptoms was followed with anti-epileptic drugs. | The results showed the importance of EEG monitoring in COVID-19 patients and showed the positive effect of anti-epileptic drugs. However, more research is needed on a larger study group. |
| De Stefano P et al. (2020) (experimental research paper) [20] | Description of a patient suffering from COVID-19-associated acute respiratory syndrome (ARDS) to highlight the diagnostic role of the EEG in the ICU (intensive care unit). | A patient with COVID-19 underwent mechanical ventilation due to acute respiratory distress syndrome (ARDS) and had an altered mental state in the ICU. An EEG, magnetic resonance imaging and an analysis of the cerebrospinal fluid were performed. | A video EEG revealed a focal monomorphic theta slowing down in the bilateral fronto-central regions. In line with the localization of the EEG, MRI showed numerous microbleeds located in the bilateral junction of white matter and different areas of the corpus callosum and the inner capsule suggesting the presence of cerebral microhemorrhage. A cerebrospinal fluid analysis ruled out the presence of encephalitis. | An EEG allowed the detection of neurological dysfunctions in the ICU in a situation where it was difficult due to the severity of the respiratory ailments. |
| Cecchetti G et al. (2020) (experimental research paper) [21] | Presentation of neurological changes in COVID-19 patients and changes in the EEG recording. | Eighteen COVID-19 patients. | The presence of slow waves in the front in the EEG record was common. Lower values of oxygen saturation on admission were associated with more serious EEG abnormalities suggesting that higher level hypoxemia and possibly longer periods without treatment may contribute to brain dysfunction. | The EEG can be a useful tool for assessing early brain involvement in COVID-19 especially in severe cases. |
| Antony AR et al. (2020) (systematic review paper) [22] | A systematic literature review was carried out to synthesize data on EEG scores in COVID-19. | The available data from 617 patients were analyzed with the EEG results reported in 84 studies. | The occurrence of abnormal EEG patterns in COVID-19 patients. | EEG abnormalities were common in COVID-19 related encephalopathy and correlated with disease severity, pre-existing neurological conditions including epilepsy and prolonged EEG monitoring. Pre-frontal changes were common and were proposed as a biomarker for COVID-19 encephalopathy. |
| Vespignani H et al. (2020) (experimental research paper) [23] | Assessment of unexplained altered mental state, loss of consciousness or poor agitation and sensitivity in critically ill COVID-19 patients. | Twenty six critically ill hospitalized patients infected with SARS-CoV-2 underwent electroencephalography to evaluate an unexplained altered mental state, loss of consciousness or poor agitation and sensitivity. | Of the 26 patients tested, five patients had electroencephalograms that showed periodic discharges consisting of frontal brain monomorphic high amplitude delta waves in the absence of epileptic activity. | These findings could suggest central nervous system damage potentially related to COVID-19 in these patients. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kopańska, M.; Banaś-Ząbczyk, A.; Łagowska, A.; Kuduk, B.; Szczygielski, J. Changes in EEG Recordings in COVID-19 Patients as a Basis for More Accurate QEEG Diagnostics and EEG Neurofeedback Therapy: A Systematic Review. J. Clin. Med. 2021, 10, 1300. https://doi.org/10.3390/jcm10061300
Kopańska M, Banaś-Ząbczyk A, Łagowska A, Kuduk B, Szczygielski J. Changes in EEG Recordings in COVID-19 Patients as a Basis for More Accurate QEEG Diagnostics and EEG Neurofeedback Therapy: A Systematic Review. Journal of Clinical Medicine. 2021; 10(6):1300. https://doi.org/10.3390/jcm10061300
Chicago/Turabian StyleKopańska, Marta, Agnieszka Banaś-Ząbczyk, Anna Łagowska, Barbara Kuduk, and Jacek Szczygielski. 2021. "Changes in EEG Recordings in COVID-19 Patients as a Basis for More Accurate QEEG Diagnostics and EEG Neurofeedback Therapy: A Systematic Review" Journal of Clinical Medicine 10, no. 6: 1300. https://doi.org/10.3390/jcm10061300
APA StyleKopańska, M., Banaś-Ząbczyk, A., Łagowska, A., Kuduk, B., & Szczygielski, J. (2021). Changes in EEG Recordings in COVID-19 Patients as a Basis for More Accurate QEEG Diagnostics and EEG Neurofeedback Therapy: A Systematic Review. Journal of Clinical Medicine, 10(6), 1300. https://doi.org/10.3390/jcm10061300








