Head-to-Head Comparison of Rapid and Automated Antigen Detection Tests for the Diagnosis of SARS-CoV-2 Infection
Abstract
1. Introduction
2. Material and Methods
2.1. Patients and Sample Collection
2.2. Analytical Procedures
2.3. Statistical Analyses
3. Results
3.1. Population
3.2. RAD Tests Sensitivity
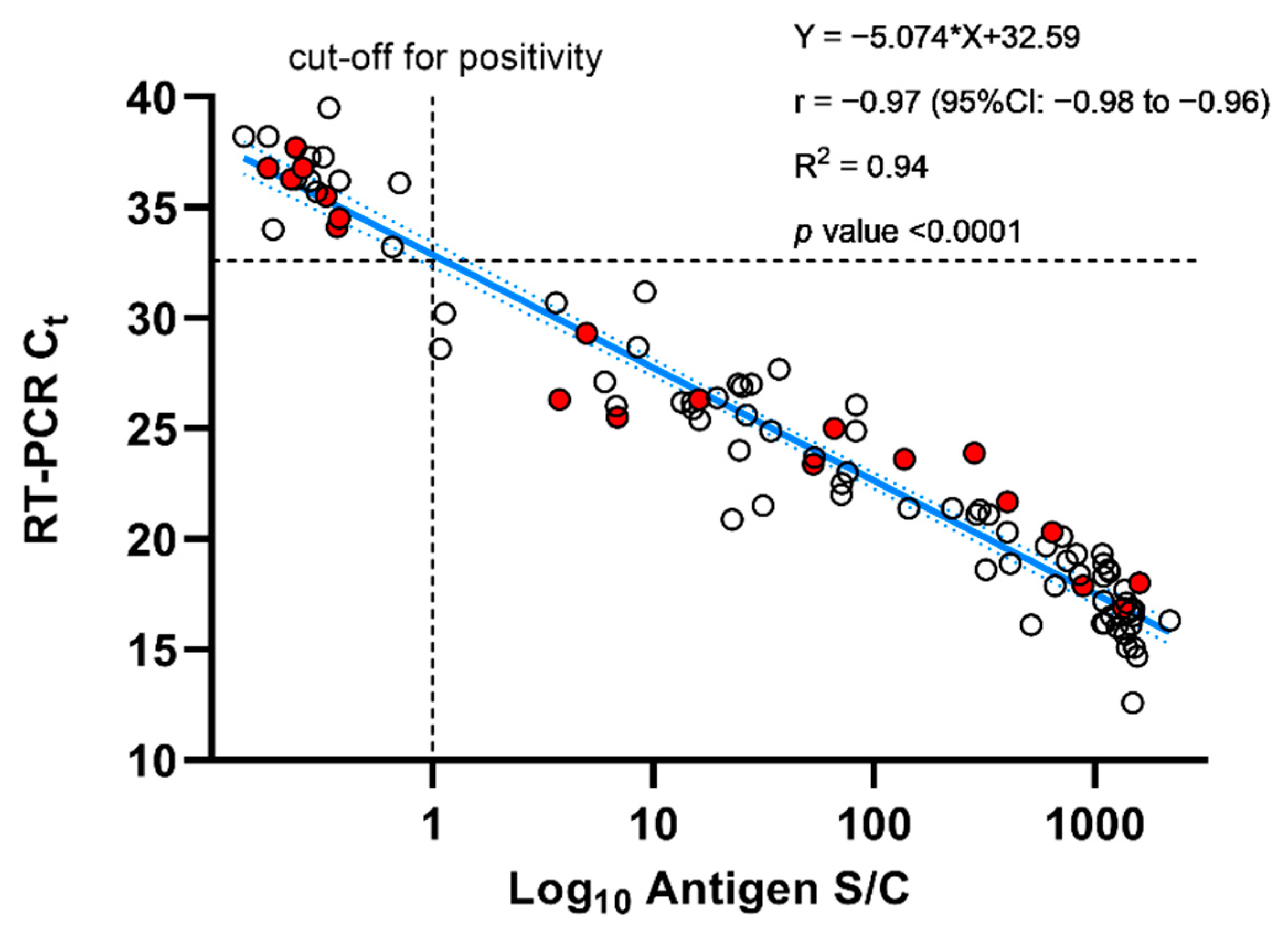
3.3. Ortho Sensitivity
3.4. Antigen Detection Tests Specificity
3.5. Negative Predictive Value, Positive Predictive Value, and Accuracy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vashist, S.K. In Vitro Diagnostic Assays for COVID-19: Recent Advances and Emerging Trends. Diagnostics 2020, 10, 202. [Google Scholar] [CrossRef] [PubMed]
- Buchta, C.; Görzer, I.; Chiba, P.; Camp, J.V.; Holzmann, H.; Puchhammer-Stöckl, E.; Mayerhofer, M.; Muller, M.M.; Aberle, S.W. Variability of cycle threshold values in an external quality assessment scheme for detection of the SARS-CoV-2 virus genome by RT-PCR. Clin. Chem. Lab. Med. (CCLM) 2020. [Google Scholar] [CrossRef]
- Lanser, L.; Bellmann-Weiler, R.; Ottl, K.W.; Huber, L.; Griesmacher, A.; Theurl, I.; Weiss, G. Evaluating the clinical utility and sensitivity of SARS-CoV-2 antigen testing in relation to RT-PCR Ct values. Infection 2020. [Google Scholar] [CrossRef] [PubMed]
- Bullard, J.; Dust, K.; Funk, D.; Strong, J.E.; Alexander, D.; Garnett, L.; Boodman, C.; Bello, A.; Hedley, A.; Schiffman, Z.; et al. Predicting infectious SARS-CoV-2 from diagnostic samples. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Bohn, M.K.; Loh, T.P.; Wang, C.B.; Mueller, R.; Koch, D.; Sethi, S.; Rawlinson, W.D.; Clementi, M.; Erasmus, R.; Lepotier, M.; et al. IFCC interim guidelines on serological testing of antibodies against SARS-CoV-2. Clin. Chem. Lab. Med. 2020, 58, 2001–2008. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control. Interim Guidelines for Covid-Antibody Testing. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antibody-tests-guidelines.html (accessed on 25 December 2020).
- FindDx. Sars-CoV-2 Diagnostics Pipeline. 2020. Available online: https://www.finddx.org/covid-19/pipeline/ (accessed on 25 December 2020).
- Albert, E.; Torres, I.; Bueno, F.; Huntley, D.; Molla, E.; Fernandez-Fuentes, M.A.; Martinez, M.; Poujois, S.; Forque, L.; Valdivia, A.; et al. Field evaluation of a rapid antigen test (Panbio COVID-19 Ag Rapid Test Device) for COVID-19 diagnosis in primary healthcare centers. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Cerutti, F.; Burdino, E.; Milia, M.G.; Allice, T.; Gregori, G.; Bruzzone, B.; Ghisetti, V. Urgent need of rapid tests for SARS CoV-2 antigen detection: Evaluation of the SD-Biosensor antigen test for SARS-CoV-2. J. Clin. Virol. 2020, 132, 104654. [Google Scholar] [CrossRef]
- Diao, B.; Wen, K.; Zhang, J.; Chen, J.; Han, C.; Chen, Y.; Want, S.; Deng, G.; Zhou, H.; Wu, Y. Accuracy of a nucleocapsid protein antigen rapid test in the diagnosis of SARS-CoV-2 infection. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef]
- Dinnes, J.; Deeks, J.J.; Adriano, A.; Berhane, S.; Davenport, C.; Dittrich, S.; Emperador, D.; Takwoingi, Y.; Cunningham, J.; Beese, S.; et al. Rapid, point-of-care antigen and molecular-based tests for diagnosis of SARS-CoV-2 infection. Cochrane Database Syst. Rev. 2020. [Google Scholar] [CrossRef]
- Fenollar, F.; Bouam, A.; Ballouche, M.; Fuster, L.; Prudent, E.; Colson, P.; Tissot-Dupont, H.; Million, M.; Drancourt, M.; Raoult, D.; et al. Evaluation of the Panbio Covid-19 rapid antigen detection test device for the screening of patients with Covid-19. J. Clin. Microbiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kruttgen, A.; Cornelissen, C.G.; Dreher, M.; Hornef, M.W.; Imohl, M.; Kleines, M. Comparison of the SARS-CoV-2 Rapid Antigen Test to the Real Star Sars-CoV-2 RT PCR Kit. J. Virol. Methods 2020, 288, 114024. [Google Scholar] [CrossRef] [PubMed]
- Linares, M.; Perez-Tanoira, R.; Carrero, A.; Romanyk, J.; Perez-Garcia, F.; Gomez-Herruz, P.; Arroyo, T.; Cuadros, J. Panbio antigen rapid test is reliable to diagnose SARS-CoV-2 infection in the first 7 days after the onset of symptoms. J. Clin. Virol. 2020, 133, 104659. [Google Scholar] [CrossRef] [PubMed]
- Liotti, F.M.; Menchinelli, G.; Lalle, E.; Palucci, I.; Marchetti, S.; Colavita, F.; La Sorda, M.; Sberna, G.; Bordi, L.; Sanguinetti, M.; et al. Performance of a novel diagnostic assay for rapid SARS-CoV-2 antigen detection in nasopharynx samples. Clin. Microbiol. Infect. 2020. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Ju, C.; Han, C.; Shi, R.; Chen, X.; Duan, D.; Yan, J.; Yan, X. Nanozyme chemiluminescence paper test for rapid and sensitive detection of SARS-CoV-2 antigen. Biosens. Bioelectron. 2020, 173, 112817. [Google Scholar] [CrossRef] [PubMed]
- Mak, G.C.; Lau, S.S.; Wong, K.K.; Chow, N.L.; Lau, C.S.; Lam, E.T.; Chan, R.C.W.; Tsnag, D.N.C. Analytical sensitivity and clinical sensitivity of the three rapid antigen detection kits for detection of SARS-CoV-2 virus. J. Clin. Virol. 2020, 133, 104684. [Google Scholar] [CrossRef]
- Ogata, A.F.; Maley, A.M.; Wu, C.; Gilboa, T.; Norman, M.; Lazarovits, R.; Mao, C.-P.; Newton, G.; Chang, M.; Nguyen, K.; et al. Ultra-sensitive Serial Profiling of SARS-CoV-2 Antigens and Antibodies in Plasma to Understand Disease Progression in COVID-19 Patients with Severe Disease. Clin. Chem. 2020. [Google Scholar] [CrossRef]
- Scohy, A.; Anantharajah, A.; Bodeus, M.; Kabamba-Mukadi, B.; Verroken, A.; Rodriguez-Villalobos, H. Low performance of rapid antigen detection test as frontline testing for COVID-19 diagnosis. J. Clin. Virol. 2020, 129, 104455. [Google Scholar] [CrossRef]
- Yamayoshi, S.; Sakai-Tagawa, Y.; Koga, M.; Akasaka, O.; Nakachi, I.; Koh, H.; Maeda, K.; Adachi, E.; Saito, M.; Nagai, H.; et al. Comparison of Rapid Antigen Tests for COVID-19. Viruses 2020, 12, 1420. [Google Scholar] [CrossRef]
- Mak, G.C.; Cheng, P.K.; Lau, S.S.; Wong, K.K.; Lau, C.; Lam, E.T.; Chan, R.C.W.; Tsang, D.N.C. Evaluation of rapid antigen test for detection of SARS-CoV-2 virus. J. Clin. Virol. 2020, 129, 104500. [Google Scholar] [CrossRef]
- Mak, G.C.K.; Lau, S.S.Y.; Wong, K.K.Y.; Chow, N.L.S.; Lau, C.S.; Lam, E.T.K.; Chan, R.C.W.; Tsang, D.N.C. Evaluation of rapid antigen detection kit from the WHO Emergency Use List for detecting SARS-CoV-2. J. Clin. Virol. 2021, 134, 104712. [Google Scholar] [CrossRef]
- Salvagno, G.L.; Gianfilippi, G.; Bragantini, D.; Henry, B.M.; Lippi, G. Clinical assessment of the Roche SARS-CoV-2 rapid antigen test. Diagnosis 2020. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Common Investigation Protocol for Investigating Suspected SARS-CoV-2 Reinfection. 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/php/reinfection.html (accessed on 25 December 2020).
- Jaafar, R.; Aherfi, S.; Wurtz, N.; Grimaldier, C.; Hoang, V.T.; Colson, P.; Raoult, D.; La Scola, B. Correlation between 3790 qPCR positives samples and positive cell cultures including 1941 SARS-CoV-2 isolates. Clin. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Jefferson, T.; Spencer, E.; Brassey, J.; Heneghan, C. Viral cultures for COVID-19 infectivity assessment—A systematic review. medRxiv. 2020. [Google Scholar] [CrossRef]
- La Scola, B.; Le Bideau, M.; Andreani, J.; Hoang, V.T.; Grimaldier, C.; Colson, P.; Gautret, P.; Raoult, D. Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1059–1061. [Google Scholar] [CrossRef]
- Singanayagam, A.; Patel, M.; Charlett, A.; Lopez Bernal, J.; Saliba, V.; Ellis, J.; Ladhani, S.; Zambon, M.; Gopal, R. Duration of infectiousness and correlation with RT-PCR cycle threshold values in cases of COVID-19, England, January to May 2020. Eurosurveillance 2020, 25, 2001483. [Google Scholar] [CrossRef] [PubMed]
- Hirotsu, Y.; Maejima, M.; Shibusawa, M.; Nagakubo, Y.; Hosaka, K.; Amemiya, K.; Sueki, H.; Hayakawa, M.; Mochizuki, H.; Tsutsui, T.; et al. Comparison of automated SARS-CoV-2 antigen test for COVID-19 infection with quantitative RT-PCR using 313 nasopharyngeal swabs, including from seven serially followed patients. Int. J. Infect. Dis. 2020, 99, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, T.; Fukumori, T.; Nishihara, Y.; Sekine, T.; Okuda, N.; Nishimura, T.; Fujikura, H.; Hirai, N.; Imakita, N.; Kasahara, K. Another false-positive problem for a SARS-CoV-2 antigen test in Japan. J. Clin. Virol. 2020, 131, 104612. [Google Scholar] [CrossRef] [PubMed]
- Kost, G.J. Designing and Interpreting COVID-19 Diagnostics: Mathematics, Visual Logistics, and Low Prevalence. Arch. Pathol. Lab. Med. 2020. [Google Scholar] [CrossRef]

| Test | Manufacturer | Process | Target | Technological | Procedure |
|---|---|---|---|---|---|
| Biotical SARS-CoV-2 Ag card | Biotical health | Manual | Nucleocapsid | Immunochromatographic | 50 parts sample: 50 parts buffer; reading at 10 min. Positive if both control and test line are present. Negative if only the control band is present |
| Panbio™ COVID-19 Ag Rapid Test Device | Abbott | Manual | Nucleocapsid | Immunochromatographic | 46 parts sample: 54 parts buffer; reading between 15 and 20 min. Positive if both control and test line are present. Negative if only the control band is present |
| Coronavirus Ag Rapid Test Cassette | Healgen Scientific | Manual | Nucleocapsid | Immunochromatographic | 50 parts sample: 50 parts buffer; reading between 15 and 20 min. Positive if both control and test line are present. Negative if only the control band is present |
| Roche SARS-CoV-2 Rapid Antigen Test | Roche Diagnostics | Manual | Nucleocapsid | Immunochromatographic | 50 parts sample: 50 parts buffer; reading between 15 and 30 min. Positive if both control and test line are present. Negative if only the control band is present |
| VITROS Immunodiagnostic Products SARS-CoV-2 Antigen test | Ortho Clinical Diagnostics | Automated | Nucleocapsid | Chemiluminescence | 80 parts sample: 20 parts buffer. Positive if signal ≥1. Negative if signal <1. Time first result = 48 min, ≅150 samples per hour |
| Sensitivity | No. of Positive Patients | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ct Range | n | Biotical (10 min) | Panbio (15 min) | Panbio (20 min) | Healgen (15 min) | Healgen (20 min) | Roche (15 min) | Roche (30 min) | Ortho |
| <15 | 2 | 2 (100%) | 1 (50.0%) | 1 (50.0%) | 2 (100%) | 2 (100%) | 2 (100%) | 2 (100%) | 2 (100%) |
| >15–20 | 34 | 33 (97.1%) | 32 (94.1%) | 32 (94.1%) | 33 (97.1%) | 33 (97.1%) | 33 (97.1%) | 33 (97.1%) | 34 (100%) |
| >20–25 | 22 | 19 (86.4%) | 21 (95.5%) | 21 (95.5%) | 21 (95.5%) | 21 (95.5%) | 21 (95.5%) | 21 (95.5%) | 22 (100%) |
| >25–30 | 19 | 6 (31.6%) | 10 (52.6%) | 10 (52.6%) | 11 (57.9%) | 13 (68.4%) | 10 (52.6%) | 10 (52.6%) | 19 (100%) |
| >30–33 | 3 | 1 (33.3%) | 0 (0.0%) | 0 (0.0%) | 2 (66.7%) | 2 (66.7%) | 0 (0.0%) | 0 (0.0%) | 3 (100%) |
| Total <33 † [95% CI] | 80 | 61 (76.2%) [65.4–85.1%] | 64 (80.0%) [69.6–88.1%] | 64 (80.0%) [69.6–88.1%] | 69 (86.3%) [76.7–92.9%] | 71 (88.8%) [79.7–94.7%] | 66 (82.5%) [72.4–90.1%] | 66 (82.5%) [72.4–90.1%] | 80 (100%) [95.5–100%] |
| >33 | 16 | 3 (18.8%) | 1 (6.3%) | 1 (6.3%) | 1 (6.3%) | 3 (18.8%) | 1 (6.3%) | 1 (6.3%) | 0 (0.0%) |
| Total (n, %) [95% CI] | 96 | 64 (66.7%) [56.3–76.0%] | 65 (67.7%) [57.4–76.9%] | 65 (67.7%) [57.4–76.9%] | 70 (72.9%) [62.9–81.5%] | 74 (77.1%) [67.4–85.1%] | 67 (69.8%) [59.6–78.8%] | 67 (69.8%) [59.6–78.8%] | 80 (83.3%) [74.4–90.2%] |
| Specificity | No. of Negative Patients | ||||||||
| Neg. RT-PCR ‡ [95% CI] | 108 | 104 (96.3%) [90.8–99.0%] | 107 (99.1%) [95.0–100%] | 107 (99.1%) [95.0–100%] | 105 (97.2%) [92.1–99.4%] | 102 (94.4%) [88.3–97.9%] | 107 (99.1%) [95.0–100%] | 107 (99.1%) [95.0–100%] | 108 (100%) [96.6–100%] |
| Neg. RT-PCR [95% CI] | 92 | 91 (98.9%) [94.1–99.9%] | 92 (100%) [96.1–100%] | 92 (100%) [96.1–100%] | 90 (97.8%) [92.4–99.7%] | 89 (96.7%) [90.8–99.3%] | 92 (100%) [96.1–100%] | 92 (100%) [96.1–100%] | 92 (100%) [96.1–100%] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Favresse, J.; Gillot, C.; Oliveira, M.; Cadrobbi, J.; Elsen, M.; Eucher, C.; Laffineur, K.; Rosseels, C.; Van Eeckhoudt, S.; Nicolas, J.-B.; et al. Head-to-Head Comparison of Rapid and Automated Antigen Detection Tests for the Diagnosis of SARS-CoV-2 Infection. J. Clin. Med. 2021, 10, 265. https://doi.org/10.3390/jcm10020265
Favresse J, Gillot C, Oliveira M, Cadrobbi J, Elsen M, Eucher C, Laffineur K, Rosseels C, Van Eeckhoudt S, Nicolas J-B, et al. Head-to-Head Comparison of Rapid and Automated Antigen Detection Tests for the Diagnosis of SARS-CoV-2 Infection. Journal of Clinical Medicine. 2021; 10(2):265. https://doi.org/10.3390/jcm10020265
Chicago/Turabian StyleFavresse, Julien, Constant Gillot, Maxime Oliveira, Julie Cadrobbi, Marc Elsen, Christine Eucher, Kim Laffineur, Catherine Rosseels, Sandrine Van Eeckhoudt, Jean-Baptiste Nicolas, and et al. 2021. "Head-to-Head Comparison of Rapid and Automated Antigen Detection Tests for the Diagnosis of SARS-CoV-2 Infection" Journal of Clinical Medicine 10, no. 2: 265. https://doi.org/10.3390/jcm10020265
APA StyleFavresse, J., Gillot, C., Oliveira, M., Cadrobbi, J., Elsen, M., Eucher, C., Laffineur, K., Rosseels, C., Van Eeckhoudt, S., Nicolas, J.-B., Morimont, L., Dogné, J.-M., & Douxfils, J. (2021). Head-to-Head Comparison of Rapid and Automated Antigen Detection Tests for the Diagnosis of SARS-CoV-2 Infection. Journal of Clinical Medicine, 10(2), 265. https://doi.org/10.3390/jcm10020265






