Assessment of Two Novel Live-Attenuated Vaccine Candidates for Herpes Simplex Virus 2 (HSV-2) in Guinea Pigs
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells & Viruses
2.2. Guinea Pig Inoculation, Clinical Assessment and Tissue Collection
2.3. Assessment Of HSV-2 Shedding in Vaginal Secretions
2.4. Explant Reactivation
2.5. Thymidine kinase (TK)-specific qPCR and LAT-specific RT-qPCR
2.6. Immunofluorescence (IF) and Fluorescent in Situ Hybridization (FISH)
2.7. Serum Immunoassays
2.8. Statistical Analysis
3. Results
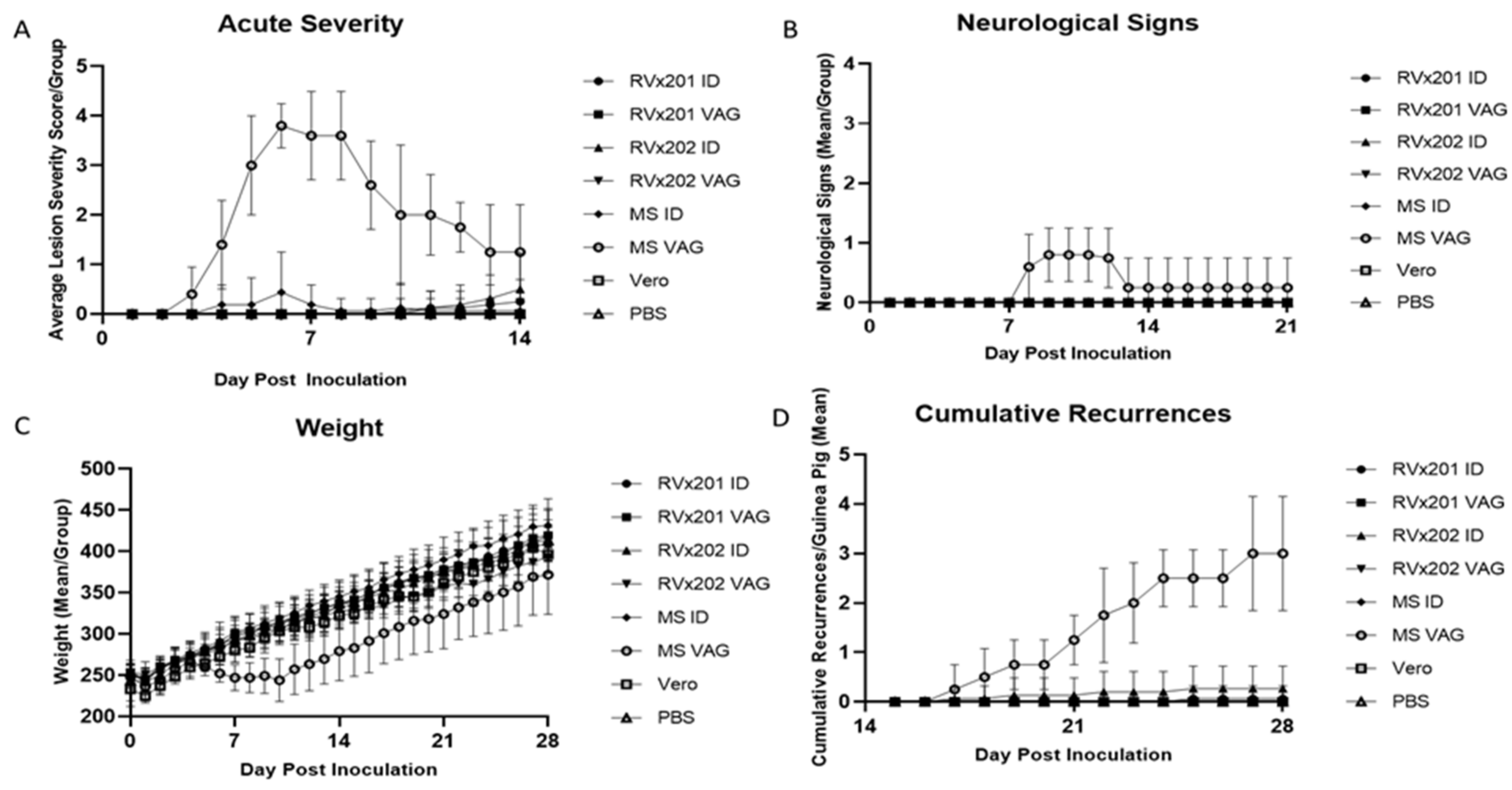
3.1. Clinical Assessment in Guinea Pigs
3.2. Low Level Transient Viral Replication Occurred after Vaginal Inoculation with RVx202 but Not with RVx201 or with Intradermal Inoculation
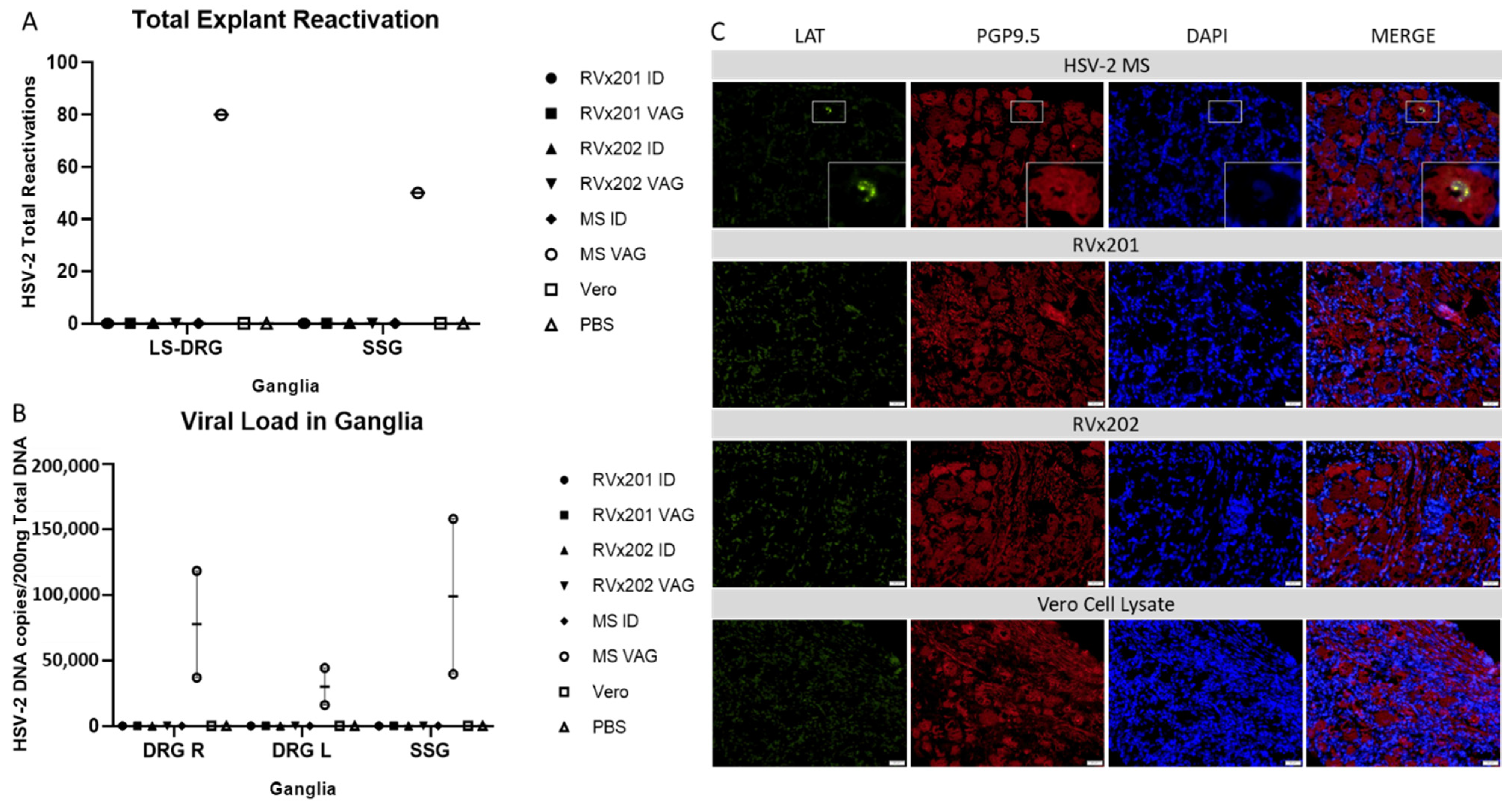
3.3. Neither Vaccine Candidate Established Latency or Detectable Replicating Virus in Lumbosacral Dorsal Root Ganglia
3.4. Vaccine Candidates Induce Serum Antibody Response Following Intradermal Inoculation
4. Discussion
| Vaccine Type | Agency | Antigen | Trial Phases Completed | Efficacy of HSV-2 Infection Prevention or Disease Amelioration |
|---|---|---|---|---|
| Subunit (monovalent) | GlaxoSmithKline | gD2 | 1–3 [53,54,55,56,57,58,59,60,61] | Infection: Not effective at preventing HSV-2 mediated infection/disease. Mild efficacy at preventing HSV-1 mediated infection/disease but not in men or HSV-1+ women [62,63,64]. |
| Subunit (bivalent) | Genocea Biosciences | gD2, ICP4 [65] | 1–2 [66,67,68,69,70,71] | Disease: Transient six-month reduction in viral shedding. Reduced recurrence rates and duration in 20% up to 12 months [72]. |
| Immune Design | gD2, UL19, UL25 [73] | 1–2 (active) [74] | Active study | |
| Chiron (Novartis) | gD2, gB2 | 1–2 | Infection: 9% reduction over 18 months (Transient 50% reduction during first 5 months vs. placebo) Disease: Frequency of recurrence not reduced [75,76]. | |
| Subunit (polyvalent) | Agenus | 22 HSV-2 Proteins [77] | 1–2 [78,79] | CD4+ and CD8+ T cell responses detected [80]. Phase 2 results not published. |
| DNA (plasmid) | PowderMed (Pfizer) | ICP0, ICP4, ICP22, ICP27 | 1 [81,82] | Not published |
| Vical | gD2, Tegument [83,84] | 1–2 [85,86] | Not published | |
| Admedus | gD2 [87] | 1–2 | Disease: Reduced viral shedding ≈50% after booster compared to placebo [51,52]. | |
| Replication deficient virus | Sanofi Pasteur | HSV-2 [88] | 1 2 (active) [74,89,90] | Active study. Shown to be immunogenic in HSV-1/HSV-2 seronegative participants [91] |
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Institutional Animal Care and Use Committee Statement
References
- Johnston, C.; Corey, L. Current Concepts for Genital Herpes Simplex Virus Infection: Diagnostics and Pathogenesis of Genital Tract Shedding. Clin. Microbiol. Rev. 2016, 29, 149–161. [Google Scholar] [CrossRef]
- Woestenberg, P.J.; Tjhie, J.H.; de Melker, H.E.; van der Klis, F.R.; van Bergen, J.E.; van der Sande, M.A.; van Benthem, B.H. Herpes simplex virus type 1 and type 2 in the Netherlands: Seroprevalence, risk factors and changes during a 12-year period. BMC Infect. Dis. 2016, 16, 364. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.M.; Pfister, J.R.; Spear, S.J. Increasing proportion of herpes simplex virus type 1 as a cause of genital herpes infection in college students. Sex. Transm. Dis. 2003, 30, 797–800. [Google Scholar] [CrossRef]
- Aravantinou, M.; Frank, I.; Arrode-Bruses, G.; Szpara, M.; Grasperge, B.; Blanchard, J.; Gettie, A.; Derby, N.; Martinelli, E. A model of genital herpes simplex virus Type 1 infection in Rhesus Macaques. J. Med. Primatol. 2017, 46, 121–128. [Google Scholar] [CrossRef]
- Scoular, A.; Norrie, J.; Gillespie, G.; Mir, N.; Carman, W.F. Longitudinal study of genital infection by herpes simplex virus type 1 in Western Scotland over 15 years. BMJ 2002, 324, 1366–1367. [Google Scholar] [CrossRef] [PubMed]
- Engelberg, R.; Carrell, D.; Krantz, E.; Corey, L.; Wald, A. Natural history of genital herpes simplex virus type 1 infection. Sex. Transm. Dis. 2003, 30, 174–177. [Google Scholar] [CrossRef]
- James, C.; Harfouche, M.; Welton, J.; Turner, K.; Abu-Raddad, L.; Gottliebd, S.; Lookera, K. Herpes simplex virus: Global infection prevalence and incidence estimates, 2016. Bull. World Health Organ. 2020, 98, 315–329. [Google Scholar] [CrossRef]
- Looker, K.J.; Magaret, A.S.; Turner, K.M.; Vickerman, P.; Gottlieb, S.L.; Newman, L.M. Global estimates of prevalent and incident herpes simplex virus type 2 infections in 2012. PLoS ONE 2015, 10, e114989. [Google Scholar] [CrossRef] [PubMed]
- McQuillan, G.M.; Kruszon-Moran, D.; Flagg, E.W.; Paulose-Ram, R. Prevalence of Herpes Simplex Virus Type 1 and Type 2 in Persons Aged 14–49: United States, 2015–2016. NCHS Data Brief. 2018, 304. [Google Scholar]
- Widener, R.; Whitley, R. Herpes simplex virus. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 251–263. [Google Scholar]
- Vassantachart, J.M.; Menter, A. Recurrent lumbosacral herpes simplex virus infection. Proc. (Bayl. Univ. Med. Cent.) 2016, 29, 48–49. [Google Scholar] [CrossRef]
- Nakamura, Y.; Nakajima, H.; Kano, Y.; Unoda, K.; Ishida, S.; Kimura, F. Herpes simplex virus type 2-associated recurrent aseptic meningitis (Mollaret’s meningitis) with a recurrence after 11-year interval: A case report. Rinsho Shinkeigaku 2016, 56, 785–787. [Google Scholar] [CrossRef][Green Version]
- Bergstrom, T.; Vahlne, A.; Alestig, K.; Jeansson, S.; Forsgren, M.; Lycke, E. Primary and recurrent herpes simplex virus type 2-induced meningitis. J. Infect. Dis. 1990, 162, 322–330. [Google Scholar] [CrossRef]
- Skoldenberg, B.; Jeansson, S.; Wolontis, S. Herpes simplex virus type 2 and acute aseptic meningitis. Clinical features of cases with isolation of herpes simplex virus from cerebrospinal fluids. Scand. J. Infect. Dis. 1975, 7, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Tedder, D.G.; Ashley, R.; Tyler, K.L.; Levin, M.J. Herpes simplex virus infection as a cause of benign recurrent lymphocytic meningitis. Ann. Intern. Med. 1994, 121, 334–338. [Google Scholar] [CrossRef]
- Chambers, S.T.; Powell, K.F.; Croxson, M.C.; Krishnan, S.; Weir, R.P. Demonstration of herpes simplex type 2 in the cerebrospinal fluid of two patients with recurrent lymphocytic meningitis. N. Z. Med. J. 1994, 107, 367–369. [Google Scholar] [PubMed]
- Bookhout, C.; Moylan, V.; Thorne, L.B. Two fatal herpesvirus cases: Treatable but easily missed diagnoses. IDCases 2016, 6, 65–67. [Google Scholar] [CrossRef] [PubMed]
- Arinze, F.; Shaver, A.; Raffanti, S. Surgical excision for recurrent herpes simplex virus 2 (HSV-2) anogenital infection in a patient with human immunodeficiency virus (HIV). Infection 2017, 45, 705–707. [Google Scholar] [CrossRef]
- Hu, K.; He, S.; Xiao, J.; Li, M.; Luo, S.; Zhang, M.; Hu, Q. Interaction between herpesvirus entry mediator and HSV-2 glycoproteins mediates HIV-1 entry of HSV-2-infected epithelial cells. J. Gen. Virol. 2017, 98, 2351–2361. [Google Scholar] [CrossRef]
- Desai, D.V.; Kulkarni, S.S. Herpes Simplex Virus: The Interplay Between HSV, Host, and HIV-1. Viral Immunol. 2015, 28, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Akinyi, B.; Odhiambo, C.; Otieno, F.; Inzaule, S.; Oswago, S.; Kerubo, E.; Ndivo, R.; Zeh, C. Prevalence, incidence and correlates of HSV-2 infection in an HIV incidence adolescent and adult cohort study in western Kenya. PLoS ONE 2017, 12, e0178907. [Google Scholar] [CrossRef] [PubMed]
- Freeman, E.E.; Weiss, H.A.; Glynn, J.R.; Cross, P.L.; Whitworth, J.A.; Hayes, R.J. Herpes simplex virus 2 infection increases HIV acquisition in men and women: Systematic review and meta-analysis of longitudinal studies. AIDS 2006, 20, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Corey, L.; Wald, A. Maternal and neonatal herpes simplex virus infections. N. Engl. J. Med. 2009, 361, 1376–1385. [Google Scholar] [CrossRef] [PubMed]
- Muller, W.J.; Zheng, X. Laboratory Diagnosis of Neonatal Herpes Simplex Virus Infections. J. Clin. Microbiol. 2019, 57. [Google Scholar] [CrossRef] [PubMed]
- Sen, P.; Barton, S.E. Genital herpes and its management. BMJ 2007, 334, 1048–1052. [Google Scholar] [CrossRef] [PubMed]
- Sauerbrei, A. Herpes Genitalis: Diagnosis, Treatment and Prevention. Geburtshilfe Frauenheilkd 2016, 76, 1310–1317. [Google Scholar] [CrossRef] [PubMed]
- Whitley, R.; Baines, J. Clinical management of herpes simplex virus infections: Past, present, and future. F1000Res 2018, 7. [Google Scholar] [CrossRef]
- Piret, J.; Boivin, G. Resistance of herpes simplex viruses to nucleoside analogues: Mechanisms, prevalence, and management. Antimicrob. Agents Chemother. 2011, 55, 459–472. [Google Scholar] [CrossRef]
- Andrei, G.; Snoeck, R. Herpes simplex virus drug-resistance: New mutations and insights. Curr. Opin. Infect. Dis. 2013, 26, 551–560. [Google Scholar] [CrossRef]
- Mertz, G.J. Asymptomatic shedding of herpes simplex virus 1 and 2: Implications for prevention of transmission. J. Infect. Dis. 2008, 198, 1098–1100. [Google Scholar] [CrossRef]
- Schiffer, J.T.; Mayer, B.T.; Fong, Y.; Swan, D.A.; Wald, A. Herpes simplex virus-2 transmission probability estimates based on quantity of viral shedding. J. R. Soc. Interface 2014, 11, 20140160. [Google Scholar] [CrossRef]
- Tronstein, E.; Johnston, C.; Huang, M.L.; Selke, S.; Magaret, A.; Warren, T.; Corey, L.; Wald, A. Genital shedding of herpes simplex virus among symptomatic and asymptomatic persons with HSV-2 infection. JAMA 2011, 305, 1441–1449. [Google Scholar] [CrossRef]
- Wald, A.; Zeh, J.; Selke, S.; Warren, T.; Ryncarz, A.J.; Ashley, R.; Krieger, J.N.; Corey, L. Reactivation of genital herpes simplex virus type 2 infection in asymptomatic seropositive persons. N. Engl. J. Med. 2000, 342, 844–850. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.; Koelle, D.M.; Wald, A. Current status and prospects for development of an HSV vaccine. Vaccine 2014, 32, 1553–1560. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.P.; Muhammad, Z.S.; Wang, J.G.; Lin, W.; Guo, S.K.; Zhang, W. HSV-2 vaccine: Current status and insight into factors for developing an efficient vaccine. Viruses 2014, 6, 371–390. [Google Scholar] [CrossRef] [PubMed]
- Halford, W.P.; Puschel, R.; Gershburg, E.; Wilber, A.; Gershburg, S.; Rakowski, B. A live-attenuated HSV-2 ICP0 virus elicits 10 to 100 times greater protection against genital herpes than a glycoprotein D subunit vaccine. PLoS ONE 2011, 6, e17748. [Google Scholar] [CrossRef]
- Delagrave, S.; Hernandez, H.; Zhou, C.; Hamberger, J.F.; Mundle, S.T.; Catalan, J.; Baloglu, S.; Anderson, S.F.; DiNapoli, J.M.; Londono-Hayes, P.; et al. Immunogenicity and efficacy of intramuscular replication-defective and subunit vaccines against herpes simplex virus type 2 in the mouse genital model. PLoS ONE 2012, 7, e46714. [Google Scholar] [CrossRef]
- Kemble, G.; Spaete, R. Human Herpesviruses: Biology, Therapy, and Immunoprophylaxis. In Herpes Simplex Vaccines; Cambridge University Press: London, UK, 2007. [Google Scholar]
- Gudnadottir, M.; Helgadottir, H.; Bjarnason, O.; Jonsdottir, K. Virus Isolated from the Brain of a Patient with Multiple Sclerosis. Exp. Neurol. 1964, 9, 85–95. [Google Scholar] [CrossRef]
- Halford, W.P.; Puschel, R.; Rakowski, B. Herpes simplex virus 2 ICP0 mutant viruses are avirulent and immunogenic: Implications for a genital herpes vaccine. PLoS ONE 2010, 5, e12251. [Google Scholar] [CrossRef]
- Samaniego, L.A.; Neiderhiser, L.; DeLuca, N.A. Persistence and expression of the herpes simplex virus genome in the absence of immediate-early proteins. J. Virol. 1998, 72, 3307–3320. [Google Scholar] [CrossRef]
- Bertke, A.S.; Swanson, S.M.; Chen, J.; Imai, Y.; Kinchington, P.R.; Margolis, T.P. A5-positive primary sensory neurons are nonpermissive for productive infection with herpes simplex virus 1 in vitro. J. Virol. 2011, 85, 6669–6677. [Google Scholar] [CrossRef]
- Halford, W.P.; Veress, L.A.; Gebhardt, B.M.; Carr, D.J. Innate and acquired immunity to herpes simplex virus type 1. Virology 1997, 236, 328–337. [Google Scholar] [CrossRef]
- Warren-Gash, C.; Forbes, H.; Breuer, J. Varicella and herpes zoster vaccine development: Lessons learned. Expert Rev. Vaccines 2017, 16, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhu, L.; Zhu, H. Efficacy of varicella (VZV) vaccination: An update for the clinician. Ther. Adv. Vaccines 2016, 4, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Wilck, M.B.; Seaman, M.S.; Baden, L.R.; Walsh, S.R.; Grandpre, L.E.; Devoy, C.; Giri, A.; Kleinjan, J.A.; Noble, L.C.; Stevenson, K.E.; et al. Safety and immunogenicity of modified vaccinia Ankara (ACAM3000): Effect of dose and route of administration. J. Infect. Dis. 2010, 201, 1361–1370. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.M.; Atmar, R.L.; El Sahly, H.M.; Cate, T.R.; Keitel, W.A. A phase I evaluation of inactivated influenza A/H5N1 vaccine administered by the intradermal or the intramuscular route. Vaccine 2010, 28, 3025–3029. [Google Scholar] [CrossRef]
- Chi, R.C.; Rock, M.T.; Neuzil, K.M. Immunogenicity and safety of intradermal influenza vaccination in healthy older adults. Clin. Infect. Dis 2010, 50, 1331–1338. [Google Scholar] [CrossRef]
- Handisurya, A.; Schellenbacher, C.; Haitel, A.; Senger, T.; Kirnbauer, R. Human papillomavirus vaccination induces neutralising antibodies in oral mucosal fluids. Br. J. Cancer 2016, 114, 409–416. [Google Scholar] [CrossRef]
- Mariani, L.; Venuti, A. HPV vaccine: An overview of immune response, clinical protection, and new approaches for the future. J. Transl. Med. 2010, 8, 105. [Google Scholar] [CrossRef] [PubMed]
- Dutton, J.L.; Woo, W.P.; Chandra, J.; Xu, Y.; Li, B.; Finlayson, N.; Griffin, P.; Frazer, I.H. An escalating dose study to assess the safety, tolerability and immunogenicity of a Herpes Simplex Virus DNA vaccine, COR-1. Hum. Vaccin Immunother. 2016, 12, 3079–3088. [Google Scholar] [CrossRef] [PubMed]
- Chandra, J.; Woo, W.P.; Dutton, J.L.; Xu, Y.; Li, B.; Kinrade, S.; Druce, J.; Finlayson, N.; Griffin, P.; Laing, K.J.; et al. Immune responses to a HSV-2 polynucleotide immunotherapy COR-1 in HSV-2 positive subjects: A randomized double blinded phase I/IIa trial. PLoS ONE 2019, 14, e0226320. [Google Scholar] [CrossRef] [PubMed]
- Allergy, N.I.o.; Diseases, I. Herpevac Neonatal Substudy. Available online: https://ClinicalTrials.gov/show/NCT00138320:2005 (accessed on 2 March 2021).
- GlaxoSmithKline. Evaluation of Safety of Candidate gD Vaccine, With or Without MPL in Healthy Herpes Simplex Virus-positive Adults. Available online: https://ClinicalTrials.gov/show/NCT00698893:1992 (accessed on 2 March 2021).
- GlaxoSmithKline. Evaluation of Immunogenicity, Reactogenicity and Safety of Herpes Simplex (gD) Candidate Vaccine With/Without Adjuvant. Available online: https://ClinicalTrials.gov/show/NCT00697567:1992 (accessed on 2 March 2021).
- GlaxoSmithKline. Humoral and Cellular Immune Response of Herpes Simplex (gD) Candidate Vaccines From 2 Different Cell Lines. Available online: https://ClinicalTrials.gov/show/NCT00698490:1995 (accessed on 2 March 2021).
- GlaxoSmithKline. Safety of a Herpes Simplex Candidate Vaccine (gD2t) With MPL and Its Efficacy to Prevent Genital Herpes Disease. Available online: https://ClinicalTrials.gov/show/NCT00699764:1996 (accessed on 2 March 2021).
- GlaxoSmithKline. Safety Evaluation of Herpes Simplex Candidate Vaccine (gD2t) With Adjuvant in HSV Seropositive Seronegative Subjects. Available online: https://ClinicalTrials.gov/show/NCT00698568:1996 (accessed on 2 March 2021).
- GlaxoSmithKline. Study to Compare Safety and Immunogenicity of Commercial Scale Consistency Lots of Herpes Simplex Vaccine. Available online: https://ClinicalTrials.gov/show/NCT00224471:2003 (accessed on 2 March 2021).
- GlaxoSmithKline. HerpeVac Trial for Young Women. Available online: https://ClinicalTrials.gov/show/NCT00057330:2003 (accessed on 2 March 2021).
- GlaxoSmithKline. Safety Study of Herpes Simplex Vaccine in HSV Seronegative and Seropositive Females Between 10 and 17 Years Old. Available online: https://ClinicalTrials.gov/show/NCT00224484:2004 (accessed on 2 March 2021).
- Belshe, R.B.; Leone, P.A.; Bernstein, D.I.; Wald, A.; Levin, M.J.; Stapleton, J.T.; Gorfinkel, I.; Morrow, R.L.; Ewell, M.G.; Stokes-Riner, A.; et al. Efficacy results of a trial of a herpes simplex vaccine. N. Engl. J. Med. 2012, 366, 34–43. [Google Scholar] [CrossRef] [PubMed]
- Stanberry, L.R.; Spruance, S.L.; Cunningham, A.L.; Bernstein, D.I.; Mindel, A.; Sacks, S.; Tyring, S.; Aoki, F.Y.; Slaoui, M.; Denis, M.; et al. Glycoprotein-D-adjuvant vaccine to prevent genital herpes. N. Engl. J. Med. 2002, 347, 1652–1661. [Google Scholar] [CrossRef] [PubMed]
- Group, H.S.V.S.; Abu-Elyazeed, R.R.; Heineman, T.; Dubin, G.; Fourneau, M.; Leroux-Roels, I.; Leroux-Roels, G.; Richardus, J.H.; Ostergaard, L.; Diez-Domingo, J.; et al. Safety and immunogenicity of a glycoprotein D genital herpes vaccine in healthy girls 10-17 years of age: Results from a randomised, controlled, double-blind trial. Vaccine 2013, 31, 6136–6143. [Google Scholar] [CrossRef]
- Skoberne, M.; Cardin, R.; Lee, A.; Kazimirova, A.; Zielinski, V.; Garvie, D.; Lundberg, A.; Larson, S.; Bravo, F.J.; Bernstein, D.I.; et al. An adjuvanted herpes simplex virus 2 subunit vaccine elicits a T cell response in mice and is an effective therapeutic vaccine in Guinea pigs. J. Virol. 2013, 87, 3930–3942. [Google Scholar] [CrossRef]
- Genocea Biosciences, Inc. Safety and Immunogenicity Study of Therapeutic HSV-2 Vaccine. Available online: https://ClinicalTrials.gov/show/NCT01667341:2012 (accessed on 2 March 2021).
- Genocea Biosciences, Inc. Dose Ranging Safety and Efficacy of Therapeutic HSV-2 Vaccine. Available online: https://ClinicalTrials.gov/show/NCT02114060:2014 (accessed on 2 March 2021).
- Genocea Biosciences, Inc. Evaluating New Formulation of Therapeutic HSV-2 Vaccine. Available online: https://ClinicalTrials.gov/show/NCT02515175:2015 (accessed on 2 March 2021).
- Genocea Biosciences, Inc. Rollover Trial for Placebo Subjects Previously Enrolled Into GEN-003-002 Study. Available online: https://ClinicalTrials.gov/show/NCT02300142:2015 (accessed on 2 March 2021).
- Genocea Biosciences, Inc. Long-term Follow-up of GEN-003-002 Subjects for Efficacy and Immunogenicity. Available online: https://ClinicalTrials.gov/show/NCT02910284:2016 (accessed on 2 March 2021).
- Genocea Biosciences, Inc. Maintenance Dose Study of GEN-003 in Subjects With Genital Herpes Infection. Available online: https://ClinicalTrials.gov/show/NCT03146403:2017 (accessed on 2 March 2021).
- Bernstein, D.I.; Flechtner, J.B.; McNeil, L.K.; Heineman, T.; Oliphant, T.; Tasker, S.; Wald, A.; Hetherington, S.; Genocea Study Group. Therapeutic HSV-2 vaccine decreases recurrent virus shedding and recurrent genital herpes disease. Vaccine 2019, 37, 3443–3450. [Google Scholar] [CrossRef]
- Odegard, J.M.; Flynn, P.A.; Campbell, D.J.; Robbins, S.H.; Dong, L.; Wang, K.; Ter Meulen, J.; Cohen, J.I.; Koelle, D.M. A novel HSV-2 subunit vaccine induces GLA-dependent CD4 and CD8 T cell responses and protective immunity in mice and guinea pigs. Vaccine 2016, 34, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Sanofi Pasteur; Immune Design. Safety and Efficacy of 4 Investigational HSV 2 Vaccines in Adults With Recurrent Genital Herpes Caused by HSV 2. Available online: https://ClinicalTrials.gov/show/NCT04222985:2019 (accessed on 2 March 2021).
- Langenberg, A.G.; Burke, R.L.; Adair, S.F.; Sekulovich, R.; Tigges, M.; Dekker, C.L.; Corey, L. A recombinant glycoprotein vaccine for herpes simplex virus type 2: Safety and immunogenicity [corrected]. Ann. Intern. Med. 1995, 122, 889–898. [Google Scholar] [CrossRef]
- Corey, L.; Langenberg, A.G.; Ashley, R.; Sekulovich, R.E.; Izu, A.E.; Douglas, J.M., Jr.; Handsfield, H.H.; Warren, T.; Marr, L.; Tyring, S.; et al. Recombinant glycoprotein vaccine for the prevention of genital HSV-2 infection: Two randomized controlled trials. Chiron HSV Vaccine Study Group. JAMA 1999, 282, 331–340. [Google Scholar] [CrossRef]
- Mo, A.; Musselli, C.; Chen, H.; Pappas, J.; Leclair, K.; Liu, A.; Chicz, R.M.; Truneh, A.; Monks, S.; Levey, D.L.; et al. A heat shock protein based polyvalent vaccine targeting HSV-2: CD4(+) and CD8(+) cellular immunity and protective efficacy. Vaccine 2011, 29, 8530–8541. [Google Scholar] [CrossRef] [PubMed]
- Agenus Inc. Trial Evaluating Safety, Tolerability and Immune Response of AG-707. Available online: https://ClinicalTrials.gov/show/NCT00231049:2006 (accessed on 2 March 2021).
- Agenus Inc. Biological Efficacy Study of HerpV Vaccine With QS-21 to Treat Subjects With Recurrent Genital Herpes. Available online: https://ClinicalTrials.gov/show/NCT01687595:2012 (accessed on 2 March 2021).
- Wald, A.; Koelle, D.M.; Fife, K.; Warren, T.; Leclair, K.; Chicz, R.M.; Monks, S.; Levey, D.L.; Musselli, C.; Srivastava, P.K. Safety and immunogenicity of long HSV-2 peptides complexed with rhHsc70 in HSV-2 seropositive persons. Vaccine 2011, 29, 8520–8529. [Google Scholar] [CrossRef] [PubMed]
- PowderMed. Safety Study of HSV2 DNA Vaccine to Treat Patients With Recurrent Genital Herpes Caused by HSV-2. Available online: https://ClinicalTrials.gov/show/NCT00274300:2004 (accessed on 2 March 2021).
- PowderMed. A Safety Study to Assess a DNA Vaccine Administered by Particle Mediated Delivery to the Skin in Healthy Subjects. Available online: https://ClinicalTrials.gov/show/NCT00310271:2006 (accessed on 2 March 2021).
- Shlapobersky, M.; Marshak, J.O.; Dong, L.; Huang, M.L.; Wei, Q.; Chu, A.; Rolland, A.; Sullivan, S.; Koelle, D.M. Vaxfectin-adjuvanted plasmid DNA vaccine improves protection and immunogenicity in a murine model of genital herpes infection. J. Gen. Virol. 2012, 93, 1305–1315. [Google Scholar] [CrossRef]
- Muller, W.J.; Dong, L.; Vilalta, A.; Byrd, B.; Wilhelm, K.M.; McClurkan, C.L.; Margalith, M.; Liu, C.; Kaslow, D.; Sidney, J.; et al. Herpes simplex virus type 2 tegument proteins contain subdominant T-cell epitopes detectable in BALB/c mice after DNA immunization and infection. J. Gen. Virol. 2009, 90, 1153–1163. [Google Scholar] [CrossRef]
- Vical. Safety and Efficacy Trial of DNA Vaccines to Treat Genital Herpes in Adults. Available online: https://ClinicalTrials.gov/show/NCT02030301:2013 (accessed on 2 March 2021).
- Vical. Safety and Efficacy Study of Herpes Simplex Virus Type 2 (HSV-2) Therapeutic DNA Vaccine. Available online: https://ClinicalTrials.gov/show/NCT02837575:2016 (accessed on 2 March 2021).
- Dutton, J.L.; Li, B.; Woo, W.P.; Marshak, J.O.; Xu, Y.; Huang, M.L.; Dong, L.; Frazer, I.H.; Koelle, D.M. A novel DNA vaccine technology conveying protection against a lethal herpes simplex viral challenge in mice. PLoS ONE 2013, 8, e76407. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, X.; Kramer, M.F.; Zhu, J.; Brockman, M.A.; Knipe, D.M. Construction, phenotypic analysis, and immunogenicity of a UL5/UL29 double deletion mutant of herpes simplex virus 2. J. Virol. 2000, 74, 7963–7971. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Allergy and Infectious Diseases; Sanofi Pasteur; National Institutes of Health Clinical Center (CC). Study of the Safety of a Particular Herpes Vaccine in Adults With or Without Herpes Infection. Available online: https://ClinicalTrials.gov/show/NCT01915212:2013 (accessed on 2 March 2021).
- Fred Hutchinson Cancer Research Center; University of Washington; Sanofi Pasteur. HSV529 Vaccine in HSV-2 Seropositive Adults. Available online: https://ClinicalTrials.gov/show/NCT02571166:2015 (accessed on 2 March 2021).
- Dropulic, L.K.; Oestreich, M.C.; Pietz, H.L.; Laing, K.J.; Hunsberger, S.; Lumbard, K.; Garabedian, D.; Turk, S.P.; Chen, A.; Hornung, R.L.; et al. A Randomized, Double-Blinded, Placebo-Controlled, Phase 1 Study of a Replication-Defective Herpes Simplex Virus (HSV) Type 2 Vaccine, HSV529, in Adults With or Without HSV Infection. J. Infect. Dis. 2019, 220, 990–1000. [Google Scholar] [CrossRef]
- Burn Aschner, C.; Pierce, C.; Knipe, D.M.; Herold, B.C. Vaccination Route as a Determinant of Protective Antibody Responses against Herpes Simplex Virus. Vaccines 2020, 8, 277. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.C.; Peng, T.; Kulka, M.; Aurelian, L. The PK domain of the large subunit of herpes simplex virus type 2 ribonucleotide reductase (ICP10) is required for immediate-early gene expression and virus growth. J. Virol. 1998, 72, 9131–9141. [Google Scholar] [CrossRef] [PubMed]
- Wachsman, M.; Kulka, M.; Smith, C.C.; Aurelian, L. A growth and latency compromised herpes simplex virus type 2 mutant (ICP10DeltaPK) has prophylactic and therapeutic protective activity in guinea pigs. Vaccine 2001, 19, 1879–1890. [Google Scholar] [CrossRef]
- Gyotoku, T.; Ono, F.; Aurelian, L. Development of HSV-specific CD4+ Th1 responses and CD8+ cytotoxic T lymphocytes with antiviral activity by vaccination with the HSV-2 mutant ICP10DeltaPK. Vaccine 2002, 20, 2796–2807. [Google Scholar] [CrossRef]
- Casanova, G.; Cancela, R.; Alonzo, L.; Benuto, R.; Magana Mdel, C.; Hurley, D.R.; Fishbein, E.; Lara, C.; Gonzalez, T.; Ponce, R.; et al. A double-blind study of the efficacy and safety of the ICP10deltaPK vaccine against recurrent genital HSV-2 infections. Cutis 2002, 70, 235–239. [Google Scholar]
- Awasthi, S.; Zumbrun, E.E.; Si, H.; Wang, F.; Shaw, C.E.; Cai, M.; Lubinski, J.M.; Barrett, S.M.; Balliet, J.W.; Flynn, J.A.; et al. Live attenuated herpes simplex virus 2 glycoprotein E deletion mutant as a vaccine candidate defective in neuronal spread. J. Virol. 2012, 86, 4586–4598. [Google Scholar] [CrossRef] [PubMed]
- Visalli, R.J.; Natuk, R.J.; Kowalski, J.; Guo, M.; Blakeney, S.; Gangolli, S.; Cooper, D. Vaccination with a HSV-2 UL24 mutant induces a protective immune response in murine and guinea pig vaginal infection models. Vaccine 2014, 32, 1398–1406. [Google Scholar] [CrossRef] [PubMed]
- Halford, W.P.; Geltz, J.; Gershburg, E. Pan-HSV-2 IgG antibody in vaccinated mice and guinea pigs correlates with protection against herpes simplex virus 2. PLoS ONE 2013, 8, e65523. [Google Scholar] [CrossRef] [PubMed]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Joyce, J.D.; Patel, A.K.; Murphy, B.; Carr, D.J.J.; Gershburg, E.; Bertke, A.S. Assessment of Two Novel Live-Attenuated Vaccine Candidates for Herpes Simplex Virus 2 (HSV-2) in Guinea Pigs. Vaccines 2021, 9, 258. https://doi.org/10.3390/vaccines9030258
Joyce JD, Patel AK, Murphy B, Carr DJJ, Gershburg E, Bertke AS. Assessment of Two Novel Live-Attenuated Vaccine Candidates for Herpes Simplex Virus 2 (HSV-2) in Guinea Pigs. Vaccines. 2021; 9(3):258. https://doi.org/10.3390/vaccines9030258
Chicago/Turabian StyleJoyce, Jonathan D., Anant K. Patel, Brandie Murphy, Daniel J.J. Carr, Edward Gershburg, and Andrea S. Bertke. 2021. "Assessment of Two Novel Live-Attenuated Vaccine Candidates for Herpes Simplex Virus 2 (HSV-2) in Guinea Pigs" Vaccines 9, no. 3: 258. https://doi.org/10.3390/vaccines9030258
APA StyleJoyce, J. D., Patel, A. K., Murphy, B., Carr, D. J. J., Gershburg, E., & Bertke, A. S. (2021). Assessment of Two Novel Live-Attenuated Vaccine Candidates for Herpes Simplex Virus 2 (HSV-2) in Guinea Pigs. Vaccines, 9(3), 258. https://doi.org/10.3390/vaccines9030258







