Composition of Royal Jelly (RJ) and Its Anti-Androgenic Effect on Reproductive Parameters in a Polycystic Ovarian Syndrome (PCOS) Animal Model
Abstract
1. Introduction
2. Materials and Methods
2.1. Royal Jelly
2.2. Phytochemical Screening and Liquid Chromatography–Mass Spectrometry (LC–MS) Analysis
2.3. Animal Experimental Design
- (a)
- Control group: given SC olive oil (10 mg/kg bodyweight) and oral distilled water (0.5 mL);
- (b)
- T group: given SC testosterone and oral distilled water;
- (c)
- T+100RJ group: given SC testosterone and oral 100 mg/kg RJ;
- (d)
- T+200RJ group: given SC testosterone and oral 200 mg/kg RJ;
- (e)
- T+400RJ group: given SC testosterone and oral 400 mg/kg RJ.
2.4. Measurement of Reproductive Hormones
2.5. Evaluation of Estrus Cycle
2.6. Measurement of Oxidant-Antioxidant Status
2.7. Histology of Ovary
2.8. Statistical Analysis
3. Results
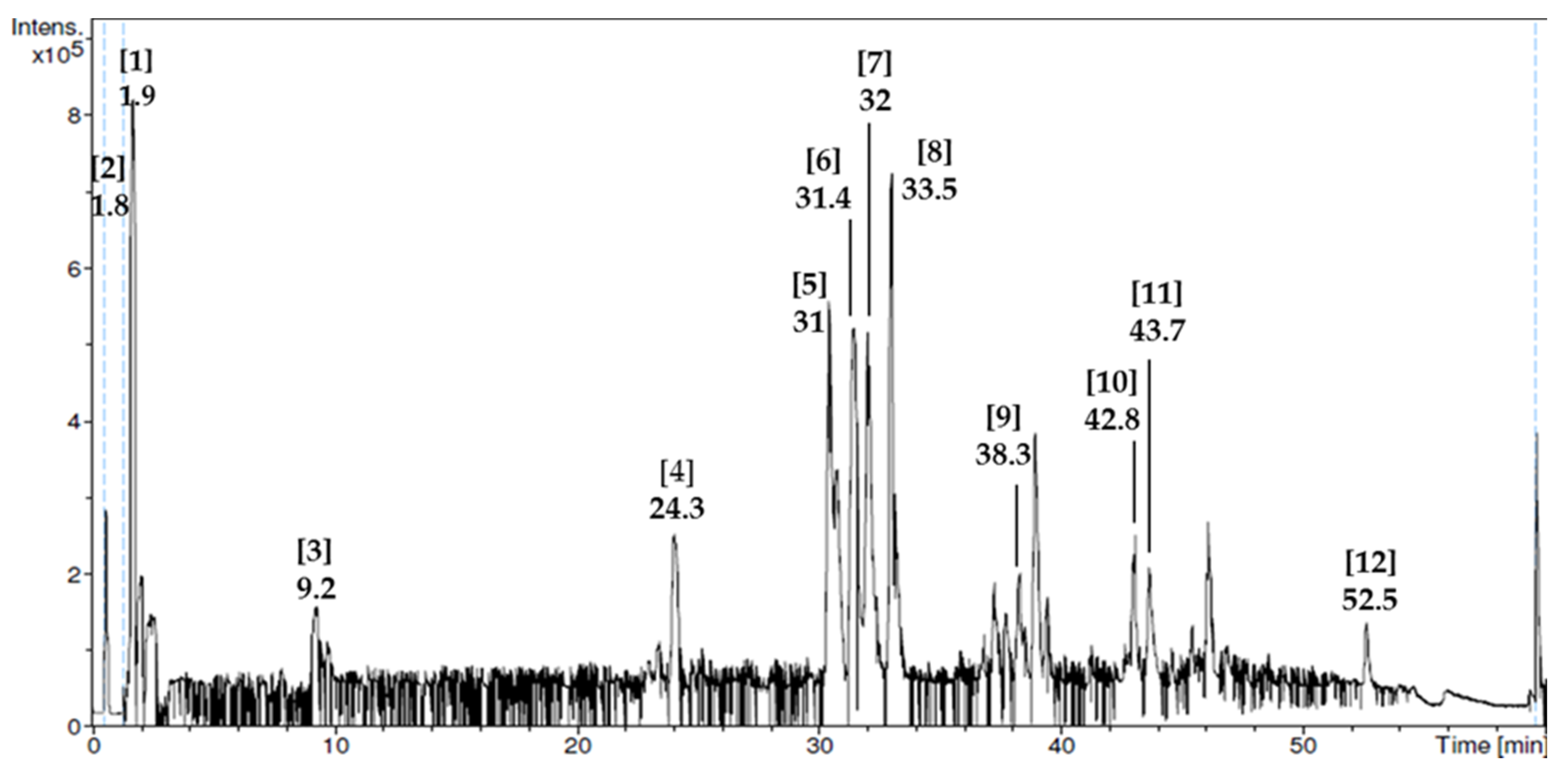
3.1. Phytochemical Screening and Liquid Chromatography–Mass Spectrometry (LC–MS) Analysis
3.2. Effect of Royal Jelly on Reproductive Hormones Levels in PCOS Rats
3.3. Effect of Royal Jelly on Regularity of Estrus Cycle in PCOS Rats
3.4. Effect of Royal Jelly on Ovarian Oxidant/Antioxidant Status in PCOS Rats
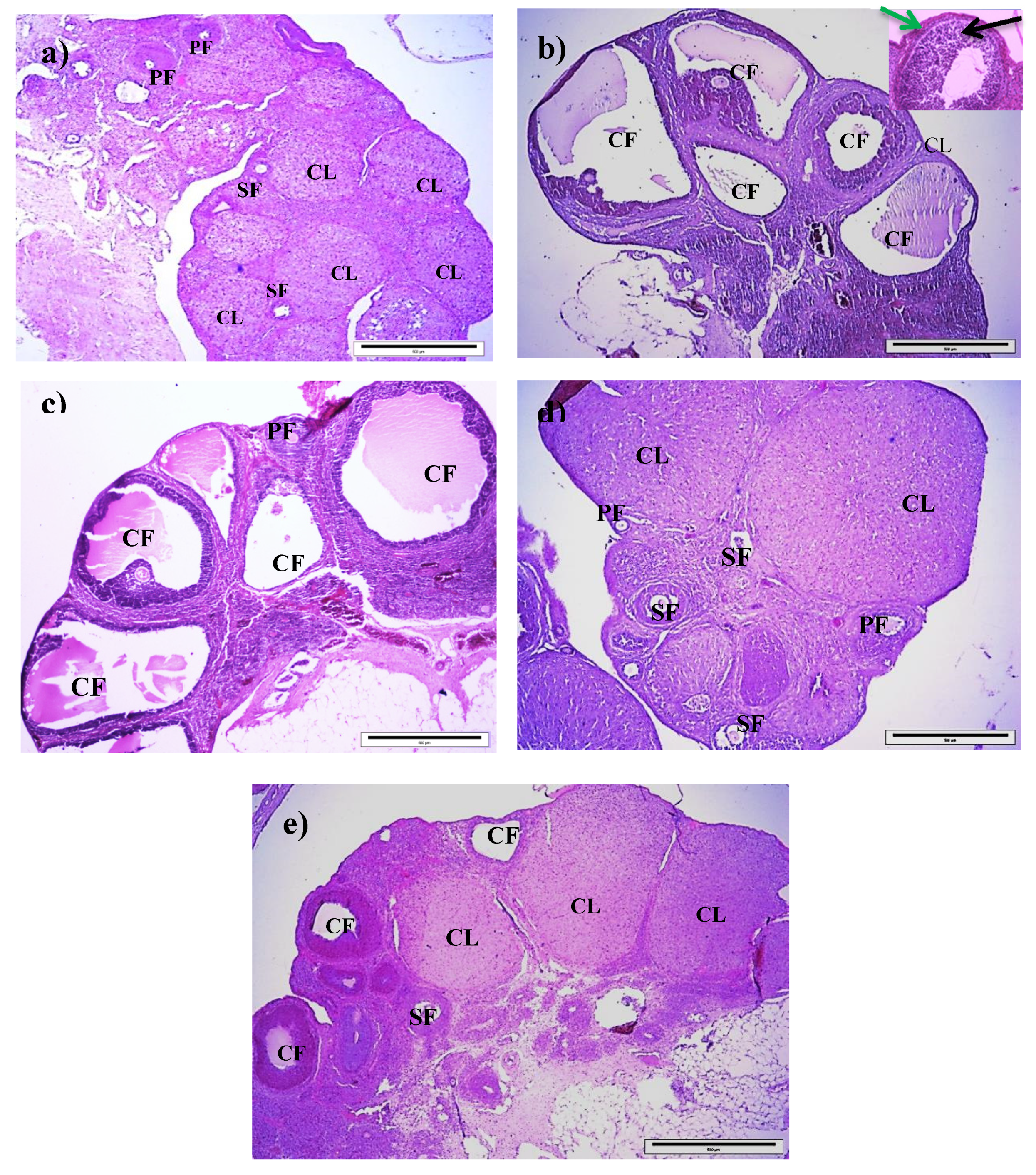
3.5. Effect of Royal Jelly on Ovarian Histology in PCOS Rats
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dashti, S.; Abdul Hamid, H.; Mohamad Saini, S.; Shah Abu Bakar, A.; Sabri, N.A.I.; Ismail, M.; Esfehani, A.J. Prevalence of Polycyctic ovary syndrome among Malaysian female university staff. J. Midwifery Reprod. Health 2019, 7, 1560–1568. [Google Scholar]
- Ding, T.; Hardiman, P.J.; Petersen, I.; Wang, F.F.; Qu, F.; Baio, G. The prevalence of polycystic ovary syndrome in reproductive-aged women of different ethnicity: A systematic review and meta-analysis. Oncotarget 2017, 8, 96351–96358. [Google Scholar] [CrossRef] [PubMed]
- Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil. Steril. 2004, 81, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Wachs, D.S.; Coffler, M.S.; Malcom, P.J.; Shimasaki, S.; Chang, R.J. Increased androgen response to follicle-stimulating hormone administration in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 2008, 93, 1827–1833. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, H.; Bai, H.; Zhang, Y.; Liu, Q.; Guan, L.; Fan, P. Oxidative stress status in Chinese women with different clinical phenotypes of polycystic ovary syndrome. Clin. Endocrinol. 2017, 86, 88–96. [Google Scholar] [CrossRef]
- Vitek, W.; Alur, S.; Hoeger, K.M. Off-label drug use in the treatment of polycystic ovary syndrome. Fertil. Steril. 2015, 103, 605–611. [Google Scholar] [CrossRef]
- Ramadan, M.F.; Al-Ghamdi, A. Bioactive compounds and health-promoting properties of royal jelly: A review. J. Funct. Foods 2012, 4, 39–52. [Google Scholar] [CrossRef]
- Sabatini, A.G.; Marcazzan, G.L.; Caboni, M.F.; Bogdanov, S.; Almeida-Muradian, L.B.D. Quality and standardisation of royal jelly. J. Apiproduct. Apimedical. Sci. 2009, 1, 1–6. [Google Scholar] [CrossRef]
- Park, M.J.; Kim, B.Y.; Park, H.G.; Deng, Y.; Yoon, H.J.; Choi, Y.S.; Lee, K.S.; Jin, B.R. Major royal jelly protein 2 acts as an antimicrobial agent and antioxidant in royal jelly. J. Asia Pac. Entomol. 2019, 22, 684–689. [Google Scholar] [CrossRef]
- Shirzad, M.; Kordyazdi, R.; Shahinfard, N.; Nikokar, M. Does royal jelly affect tumor cells? J. Herbmed. Pharm. 2013, 2, 45–48. [Google Scholar]
- Gasic, S.; Vucevic, D.; Vasilijic, S.; Antunovic, M.; Chinou, I.; Colic, M. Evaluation of the immunomodulatory activities of royal jelly components in vitro. Immunopharmacol. Immunotoxicol. 2007, 29, 521–536. [Google Scholar] [CrossRef] [PubMed]
- Silici, S.; Ekmekcioglu, O.; Kanbur, M.; Deniz, K. The protective effect of royal jelly against cisplatin-induced renal oxidative stress in rats. World J. Urol. 2011, 29, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Petelin, A.; Kenig, S.; Kopinc, R.; Dezelak, M.; Bizjak, M.C.; Praznikar, Z.J. Effects of royal jelly administration on lipid profile, satiety, inflammation, and antioxidant capacity in asymptomatic overweight adults. Evid. Based Complement. Altern. Med. 2019, 2019, 4969720. [Google Scholar] [CrossRef] [PubMed]
- Sharif, S.N.; Darsareh, F. Effect of royal jelly on menopausal symptoms: A randomized placebo-controlled clinical trial. Complement. Clin. Pr. 2019, 37, 47–50. [Google Scholar] [CrossRef]
- Elnagar, S.A. Royal jelly counteracts bucks’ “summer infertility”. Anim. Reprod. Sci. 2010, 121, 174–180. [Google Scholar] [CrossRef]
- El-Hanoun, A.M.; Elkomy, A.E.; Fares, W.A.; Shahien, E. Impact of royal jelly to improve reproductive performance of male rabbits under hot summer conditions. World Rabbit Sci. 2014, 22, 241–248. [Google Scholar] [CrossRef]
- Husein, M.Q.; Kridli, R.T. Reproductive responses following royal jelly treatment administered orally or intramuscularly into progesterone-treated Awassi ewes. Anim. Reprod. Sci. 2002, 74, 45–53. [Google Scholar] [CrossRef]
- Kridli, R.T.; Husein, M.Q.; Humphrey, W.D. Effect of royal jelly and GnRH on estrus synchronization and pregnancy rate in ewes using intravaginal sponges. Small Rumin. Res. 2003, 49, 25–30. [Google Scholar] [CrossRef]
- Mishima, S.; Suzuki, K.-M.; Isohama, Y.; Kuratsu, N.; Araki, Y.; Inoue, M.; Miyata, T. Royal jelly has estrogenic effects in vitro and in vivo. J. Ethnopharmacol. 2005, 101, 215–220. [Google Scholar] [CrossRef]
- Suzuki, K.M.; Isohama, Y.; Maruyama, H.; Yamada, Y.; Narita, Y.; Ohta, S.; Araki, Y.; Miyata, T. Estrogenic activities of fatty acids and a sterol isolated from royal jelly. Evid. Based Complement. Altern. Med. 2008, 5, 295–302. [Google Scholar] [CrossRef]
- Reagen-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. Faseb. J. 2008, 22, 659–661. [Google Scholar] [CrossRef] [PubMed]
- Raaman, N. Phytochemical Techniques; New India Publishing Agency: Pitam Pura, New Delhi, India, 2006. [Google Scholar]
- Ajayi, G.O.; Olagunju, J.A.; Ademuyiwa, O.; Martins, O.C. Gas chromatography-mass spectrometry analysis and phytochemical screening of ethanolic root extract of Plumbago Zeylanica, Linn. J. Med. Plants Res. 2011, 5, 1756–1761. [Google Scholar]
- Ganesh, S.; Vennila, J.J. Phytochemical analysis of Acanthus Ilicifolius and Avicennia officinalis by GC-MS. Res. J. Phytochem. 2011, 5, 60–65. [Google Scholar] [CrossRef]
- Abdulghani, M.; Hussin, A.H.; Sulaiman, S.A.; Chan, K.L. The ameliorative effects of Eurycoma longifolia Jack on testosterone-induced reproductive disorders in female rats. Reprod. Biol. 2012, 12, 247–255. [Google Scholar] [CrossRef]
- Marcondes, F.K.; Bianchi, F.J.; Tanno, A.P. Determination of the estrous cycle phases of rats: Some helpful considerations. Braz. J. Biol. 2002, 62, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Antonisamy, J.M.; Aparna, J.S.; Jeeva, S.; Sukumaran, S.; Anantham, B. Preliminary phytochemical studies on the methanolic flower extracts of some selected medicinal plants from India. Asian Pac. J. Trop. Biomed. 2012, 2, S79–S82. [Google Scholar] [CrossRef]
- Rao, M.R.K.; Kumar, M.H.; Amutha, A.; Prabhu, K.; Chatterjee, B.; Selva Kumar, S. Phytochemical analysis and antioxidant efficacy of the resin of Bombax ceiba (Salmali). Int. J. Pharm. Sci. Rev. Res. 2015, 30, 335–339. [Google Scholar]
- Ramalakshmi, S.; Muthuchelian, K. Analysis of bioactive constituents from the ethanolic leaf extract of Tabebuia rosea (Bertol.) DC by gas chromatography-mass spectrometry. Int. J. ChemTech Res. 2011, 3, 1054–1059. [Google Scholar]
- Termentzi, A.; Fokialakis, N.; Leandros Skaltsounis, A. Natural resins and bioactive natural products thereof as potential anitimicrobial agents. Curr. Pharm. Des. 2011, 17, 1267–1290. [Google Scholar] [CrossRef]
- Hattori, N.; Nomoto, H.; Fukumitsu, H.; Mishima, S.; Furukawa, S. AMP N1 -Oxide, a unique compound of royal jelly, induces neurite outgrowth from PC12 cells via signaling by protein kinase A independent of that by mitogen-activated protein kinase. Evid. Based Complement. Altern. Med. 2010, 7, 63–68. [Google Scholar] [CrossRef]
- Coyral-Castel, S.; Tosca, L.; Ferreira, G.; Jeanpierre, E.; Rame, C.; Lomet, D.; Caraty, A.; Monget, P.; Chabrolle, C.; Dupont, J. The effect of AMP-activated kinase activation on gonadotrophin-releasing hormone secretion in GT1-7 cells and its potential role in hypothalamic regulation of the oestrous cyclicity in rats. J. Neuroendocr. 2008, 20, 335–346. [Google Scholar] [CrossRef]
- Ratner, A. Effects of follicle stimulating hormone and luteinizing hormone upon cyclic AMP and cyclic GMP levels in rat ovaries in vitro. Endocrinology 1976, 99, 1496–1500. [Google Scholar] [CrossRef] [PubMed]
- Daval, J.-L.; Nehlig, A.; Nicolas, F. Physiological and pharmacological properties of adenosine: Therapeutic implications. Life Sci. 1991, 49, 1435–1453. [Google Scholar] [CrossRef]
- Kuo, Y.M.; Hayflick, S.J.; Gitschier, J. Deprivation of pantothenic acid elicits a movement disorder and azoospermia in a mouse model of pantothenate kinase-associated neurodegeneration. J. Inherit. Metab. Dis. 2007, 30, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Rumberger, J.A.; Napolitano, J.; Azumano, I.; Kamiya, T.; Evans, M. Pantethine, a derivative of vitamin B5 used as a nutritional supplement, favorably alters low-density lipoprotein cholesterol metabolism in low- to moderate-cardiovascular risk North American subjects: A triple-blinded placebo and diet-controlled investigation. Nutr. Res. 2011, 31, 608–615. [Google Scholar]
- Jung, S.; Kim, M.K.; Choi, B.Y. The long-term relationship between dietary pantothenic acid (vitamin B5) intake and C-reactive protein concentration in adults aged 40 years and older. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 806–816. [Google Scholar] [CrossRef]
- Layden, B.T.; Angueira, A.R.; Brodsky, M.; Durai, V.; Lower, W.L., Jr. Short chain fatty acids and their receptors: New metabolic targets. Transl. Res. 2013, 161, 131–140. [Google Scholar] [CrossRef]
- Iaconelli, A.; Gastaldelli, A.; Chiellini, C.; Gniuli, D.; Favuzzi, A.; Binnert, C.; Mace, K.; Mingrone, G. Effect of oral sebacic acid on postprandial glycemia, insulinemia, and glucose rate of appearance in type 2 diabetes. Diabetes Care 2010, 33, 2327–2332. [Google Scholar] [CrossRef]
- Chen, Y.-F.; Wang, K.; Zhang, Y.-Z.; Zheng, Y.-F.; Hu, F.-L. In vitro anti-inflammatory effects of three fatty acids from royal jelly. Mediat. Inflamm. 2016, 2016, 3583684. [Google Scholar] [CrossRef]
- Cho, B.N.; Kim, K. Differential effect of baclofen on hypothalamic GnRH and pituitary LHβ gene expression in steroid-treated rats. Mol. Cells 1997, 7, 605–609. [Google Scholar]
- Nakajima, Y.; Tsuruma, K.; Shimazawa, M.; Mishima, S.; Hara, H. Comparison of bee products based on assays of antioxidant capacities. BMC Complement. Altern. Med. 2009, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Qiu, S.; Wang, L.; Zhang, N.; Shi, Y.; Zhou, H.; Liu, X.; Shao, L.; Liu, X.; Chen, J.; et al. Reproductive and developmental toxicity study of caffeic acid in mice. Food Chem. Toxicol. 2019, 123, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Zanyatkin, I.; Stroylova, Y.; Tishina, S.; Stroylov, V.; Melnikova, A.; Haertle, T.; Muronetz, V. Inhibition of prion propagation by 3,4-dimethoxycinnamic acid. Phytother. Res. 2017, 31, 1046–1055. [Google Scholar] [CrossRef] [PubMed]
- Medvedeva, M.; Barinova, K.; Melnikova, A.; Semenyuk, P.; Kolmogorov, V.; Gorelkin, P.; Erofeev, A.; Muronetz, V. Naturally occuring cinnamic acid derivatives prevent amyloid transformation of alpha-synuclein. Biochimie 2020, 170, 128–139. [Google Scholar] [CrossRef]
- Thompson, D.; Pepys, M.B.; Wood, S.P. The physiological structure of human C-reactive protein and its complex with phosphocholine. Structure 1999, 7, 169–177. [Google Scholar] [CrossRef]
- Nabas, Z.; Haddadin, M.S.Y.; Haddadin, J.; Nazer, I.K. Chemical composition of royal jelly and effects of synbiotic with two different locally isolated probiotic strains on antioxidant activities. Pol. J. Food Nutr. Sci. 2014, 64, 171–180. [Google Scholar] [CrossRef]
- Nagai, T.; Inoue, R. Preparation and the functional properties of water extract and alkaline extract of royal jelly. Food Chem. 2004, 84, 181–186. [Google Scholar] [CrossRef]
- Beloosesky, R.; Gold, R.; Almog, B.; Sasson, R.; Dantes, A.; Land-Bracha, A.; Hirsh, L.; Itskovitz-Eldor, J.; Lessing, J.B.; Homburg, R.; et al. Induction of polycystic ovary by testosterone in immature female rats: Modulation of apoptosis and attenuation of glucose/insulin ratio. Int. J. Mol. Med. 2004, 14, 207–215. [Google Scholar] [CrossRef]
- Pandey, V.; Shukla, R.; Krishna, A.; Tripathi, Y.B. Effect of combined treatment of modern and herbal supplement in the management of letrozole induced polycystic ovary syndrome. J. Endocrinol. Diabetes 2017, 4, 1–6. [Google Scholar]
- Kavitha, A.; Babu, A.N.; Kumar, S.M.; Kiran, S.V. Evaluation of effects of Commiphora wightii in dehydroepiandrosterone (DHEA) induced polystic ovary syndrome (PCOS) in rats. PharmaTutor 2016, 4, 47–55. [Google Scholar]
- Tehrani, F.R.; Noroozzadeh, M.; Zahediasl, S.; Piryaei, A.; Azizi, F. Introducing a rat model of prenatal androgen-induced polycystic ovary syndrome in adulthood. Exp. Physiol. 2014, 99, 792–801. [Google Scholar] [CrossRef] [PubMed]
- Kandasamy, S.; Sivagamasundari, R.I.; Bupathy, A.; Sethubathy, S.; Gobal, V. Evaluation of insulin resistance and oxidative stress in obese patients with polycystic ovary syndrome. Int. J. Appl. Biol. Pharm. Technol. 2010, 1, 391–398. [Google Scholar]
- Yaba, A.; Demir, N. The mechanism of MTOR (mammalian target of rapamycin) in a mouse model of polycystic ovary syndrome (PCOS). J. Ovarian Res. 2012, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bartolone, L.; Smedile, G.; Arcoraci, V.; Trimarchi, F.; Benvenga, S. Extremely high levels of estradiol and testosterone in a case of polycystic ovarian syndrome. Hormone and clinical similarities with the phenotype of the α estrogen receptor null mice. J. Endocrinol. Investig. 2000, 23, 467–472. [Google Scholar] [CrossRef]
- Karimzadeh, L.; Nabiuni, M.; Sheikholeslami, A.; Irian, S. Bee venom treatment reduced C-reactive protein and improved follicle quality in a rat model of estradiol valerate-induced polycystic ovarian syndrome. J. Venom. Anim. Toxins Incl. Trop. Dis. 2012, 18, 384–392. [Google Scholar] [CrossRef]
- Taylor, A.E.; McCourt, B.; Martin, K.A.; Anderson, E.J.; Adams, J.M.; Schoenfeld, D.; Hall, J.E. Determinants of abnormal gonadotropin secretion in clinically defined women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 1997, 82, 2248–2256. [Google Scholar] [CrossRef]
- Laven, J.S.E.; Imani, B.; Eijkemans, M.J.C.; Fauser, B.C. New approach to polycystic ovary syndrome and other forms of anovulatory infertility. Obs. Gynecol. Surv. 2002, 57, 755–767. [Google Scholar] [CrossRef]
- Ehrmann, D.A. Polycystic Ovary Syndrome. N. Engl. J. Med. 2005, 352, 1223–1236. [Google Scholar] [CrossRef]
- Genazzani, A.D.; Lanzoni, C.; Ricchieri, F.; Jasonni, V.M. Myo-inositol administration positively affects hyperinsulinemia and hormonal parameters in overweight patients with polycystic ovary syndrome. Gynecol. Endocrinol. 2008, 24, 139–144. [Google Scholar] [CrossRef]
- Al-Sanafi, A.E.; Mohssin, S.A.; Abdulla, S.M. Effect of royal jelly on male infertility. Thi-Qar Med. J. 2007, 1, 1–12. [Google Scholar]
- Manas, G.E.; Najafi, G. Protective effects of royal jelly on the histomorphologic, oxidative stress and sperm parameters in ofloxacin treated rat. Comp. Clin. Path. 2017, 26, 1111–1115. [Google Scholar] [CrossRef]
- Sun, F.; Yu, J. The effect of a special herbal tea on obesity and anovulation in androgen-sterilized rats. Proc. Soc. Exp. Biol. Med. 2000, 223, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S.D.; Moenter, S.M. Prenatal androgens alter GABAergic drive to gonadotropin-releasing hormone neurons: Implications for a common fertility disorder. Proc. Natl. Acad. Sci. USA 2004, 101, 7129–7134. [Google Scholar] [CrossRef] [PubMed]
- Bhuvaneshwari, S.; Poornima, R.; Averal, H.I. Detection of polycystic ovary syndrome and its treatment with Pergulariadaemia in rat models. IOSR J. Pharm. 2015, 5, 42–49. [Google Scholar]
- Hamid, N.A.; Abu Bakar, A.B.; Mohamed, M. Phytochemical analysis and GC-MS profile of royal jelly from selected areas in Malaysia. Malays. Appl. Biol. 2018, 47, 101–107. [Google Scholar]
- Maleedhu, P.; Vijayabhaskar, M.; Rao, P.; Kodumuri, P. Antioxidant status in women with polycystic ovary syndrome. J. Med. Health Sci. 2014, 3, 91–96. [Google Scholar]
- Ghanbari, E.; Nejati, V.; Najafi, G.; Khazaei, M.; Babaei, M. Study on the effect of royal jelly on reproductive parameters in streptozotocin-induced diabetic rats. Int. J. Fertil. Steril. 2015, 9, 113–120. [Google Scholar]
- Najafi, G.; Nejati, V.; Jalali, A.S.; Zahmatkesh, E. Protective role of royal jelly in oxymetholone-induced oxidative injury in mouse testis. Iran. J. Toxicol. 2014, 8, 1073–1080. [Google Scholar]
- Ghasemzadeh, A.; Farzadi, L.; Khaki, A.; Ahmadi, S.K. Effect of Allium cepa seeds ethanolic extract on experimental polycystic ovary syndrome (PCOS) apoptosis induced by estradiol-valerate. Life Sci. J. 2013, 10, 170–175. [Google Scholar]
- Malekinejad, H.; Ahsan, S.; Delkhosh-Kasmaie, F.; Cheraghi, H.; Rezaei-Golmisheh, A.; Janbaz-Acyabar, H. Cardioprotective effect of royal jelly on paclitaxel-induced cardio-toxicity in rats. Iran. J. Basic Med. Sci. 2016, 19, 221–227. [Google Scholar]
- Shidfar, F.; Jazayeri, S.; Mousavi, S.N.; Malek, M.; Hosseini, A.F.; Khoshpey, B. Does supplementation with royal jelly improve oxidative stress and insulin resistance in type 2 diabetic patients? Iran. J. Public Health 2015, 44, 797–803. [Google Scholar] [PubMed]
- Kurdoglu, Z.; Ozkol, H.; Tuluce, Y.; Koyuncu, I. Oxidative status and its relation with insulin resistance in young non-obese women with polycystic ovary syndrome. J. Endocrinol. Investig. 2012, 35, 317–321. [Google Scholar]
- Jamnik, P.; Goranovič, D.; Raspor, P. Antioxidative action of royal jelly in the yeast cell. Exp. Gerontol. 2007, 42, 594–600. [Google Scholar] [CrossRef]
- Gómez-Caravaca, A.M.; Gómez-Romero, M.; Arráez-Román, D.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Advances in the analysis of phenolic compounds in products derived from bees. J. Pharm. Biomed. Anal. 2006, 41, 1220–1234. [Google Scholar] [CrossRef] [PubMed]
- Gleicher, N.; Weghofer, A.; Barad, D.H. The role of androgens in follicle maturation and ovulation induction: Friend or foe of infertility treatment? Reprod. Biol. Endocrinol. 2011, 9, 1–12. [Google Scholar] [CrossRef]
- Agarwal, A.; Gupta, S.; Sharma, R. Oxidative stress and its implications in female infertility—A clinician’s perspective. Reprod. Biomed. Online 2005, 11, 641–650. [Google Scholar] [CrossRef]
- Kamakura, M. Royalactin induces queen differentiation in honeybees. Nature 2011, 473, 478–483. [Google Scholar] [CrossRef] [PubMed]
| Compounds | Intensity | Activities |
|---|---|---|
| Alkaloids | + | Anti-inflammatory effect, anti-asthmatic and anti-anaphylactic activities [27]. |
| Phenols | + | Antibacterial and anti-hemolytic activities [28]. |
| Flavonoids | ++ | Antimicrobial, anti-proliferative [29], and antioxidant activities [27]. |
| Glycosides | ++ | Preservative [29]. |
| Resins | ++ | Antimicrobial activity [30]. |
| Saponins | ++ | Anti-hypercholesterolemia and antibiotic activities [28]. |
| Tannins | ++ | Anti-hemolytic activity [28]. |
| Terpenoids | ++ | Cytotoxic, anti-fungal, antibacterial, and anti-inflammatory activities [29]. |
| Xanthoproteins | + | No activity reported. |
| No. | Compound Name | Retention Time (min) | Mass Spectrum (m/z) | Bioactivities |
|---|---|---|---|---|
| 1 | Adenosine-5-monophosphate | 1.9 | 348 | Induces neurite outgrowth (neuritegenesis) [31], modulates gonadotrophin-releasing hormone (GnRH) release and estrus activity [32]. |
| 2 | Guanosine-5-monophosphate | 1.8 | 195 | Regulates oocytes maturation [33]. |
| 3 | Adenosine | 9.2 | 220 | Anticonvulsant, anti-ischemic, analgesic, and neuroprotective activities [34]. |
| 4 | d-pantothenic acid hemicalcium salt | 24.3 | 227 | Improves sperm motility and maturity [35], reduces cardiovascular risk [36] and inflammation [37] |
| 5 | Homovanillic acid | 31 | 155 | No activity reported. |
| 6 | Carboxylic acid | 31.4 | 151 | Required for formation of other important components in body such as fatty acid [38]. |
| 7 | Sebacic acid | 32 | 185 | Anti-diabetic [39] and anti-inflammatory effects [40]. |
| 8 | Methoxybenzoic acid derivative | 33.5 | 187 | No activity reported. |
| 9 | Baclofen | 38.3 | 237 | Enhances GnRH mRNA level [41] |
| 10 | Caffeic acid derivative | 42.8 | 206 | Antioxidant [42] and anti-implantation activities during early pregnancy in mice [43]. |
| 11 | Dimethoxycinnamic acid derivative | 43.7 | 295 | Inhibits prion propagation [44] and alpha-synuclein amyloid aggregation [45] |
| 12 | Phosphocholine derivative | 52.5 | 206 | One of the C-reactive protein-binding targets to carry out immunologic response [46] |
| Groups | T (ng/mL) | E2 (pg/mL) | LH (ng/mL) | FSH (ng/mL) |
|---|---|---|---|---|
| Control | 1.99 (0.07) | 52.73 (1.3) | 3.72 (0.05) | 81.23 (1.8) |
| T | 3.35 (0.18) a | 88.89 (2.98) a | 3.69 (0.03) | 56.52 (3.09) a |
| T+100RJ | 2.95 (0.25) a | 60.15 (1.39) b | 3.56 (0.03) | 62.95 (3.15) a |
| T+200RJ | 2.07 (0.06) b,c | 57.43 (3.74) b | 3.45 (0.19) a,b | 85.39 (1.7) b,c |
| T+400RJ | 2.96 (0.08) b,d | 60.96 (1.8) b | 3.49 (0.2) a | 70.49 (8.56) |
| Groups | Rats with Regular Estrus Cycle (%) |
|---|---|
| Control | 87.5 |
| T group | 25.0 a |
| T+100RJ | 25.0 a |
| T+200RJ | 87.5 b,c |
| T+400RJ | 50.0 |
| Group | MDA (nmol/mg Protein) | TAC µM Trolox Equivalents | SOD (U/mg Protein) | GPx (U/mg Protein) | CAT (U/mg Protein) |
|---|---|---|---|---|---|
| Control | 0.09 (0.18) | 0.25 (0.01) | 1.92 (0.32) | 34.1 (4.75) | 63.61 (0.63) |
| T | 0.39 (0.14) a | 0.10 (0.01) a | 2.93 (0.34) | 100.69 (14.39) a | 64.47 (0.41) |
| T+100RJ | 0.15 (0.05) b | 0.12 (0.01) a | 3.67 (0.15) a | 66.76 (21.81) | 59.07 (4.16) |
| T+ 200RJ | 0.15 (0.07) b | 0.21 (0.03) b | 2.19 (0.15) c | 40.09 (5.89) b | 54.10 (4.46) |
| T+400RJ | 0.3 (0.06) a | 0.12 (0.03) a,d | 2.57 (0.53) | 54.15 (14.53) | 53.10 (7.83) |
| Variables | No. of Primary Follicles | No. of Secondary Follicles | No. of Corpora Lutea | No. of Cystic Follicles |
|---|---|---|---|---|
| Control | 3.25 (0.37) | 7.88 (0.8) | 6.64 (0.93) | 1.13 (0.3) |
| T | 7.25 (0.53) a | 2.25 (0.36) a | 3.13 (0.35) a | 9.25 (0.77) a |
| T+100RJ | 5.88 (0.40) a | 2.38 (0.42) a | 4.75 (0.59) b | 5.50 (0.42) a,b |
| T+200RJ | 3.25 (0.37) b,c | 7.00 (0.57) b,c | 8.75 (0.36) b,c | 3.00 (0.423) b,c |
| T+400RJ | 6.00 (0.33) a,d | 2.13 (0.3) a,d | 3.75 (0.45) a,d | 4.38 (0.26) a,b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ab Hamid, N.; Abu Bakar, A.B.; Mat Zain, A.A.; Nik Hussain, N.H.; Othman, Z.A.; Zakaria, Z.; Mohamed, M. Composition of Royal Jelly (RJ) and Its Anti-Androgenic Effect on Reproductive Parameters in a Polycystic Ovarian Syndrome (PCOS) Animal Model. Antioxidants 2020, 9, 499. https://doi.org/10.3390/antiox9060499
Ab Hamid N, Abu Bakar AB, Mat Zain AA, Nik Hussain NH, Othman ZA, Zakaria Z, Mohamed M. Composition of Royal Jelly (RJ) and Its Anti-Androgenic Effect on Reproductive Parameters in a Polycystic Ovarian Syndrome (PCOS) Animal Model. Antioxidants. 2020; 9(6):499. https://doi.org/10.3390/antiox9060499
Chicago/Turabian StyleAb Hamid, Norhamidar, Ainul Bahiyah Abu Bakar, Anani Aila Mat Zain, Nik Hazlina Nik Hussain, Zaidatul Akmal Othman, Zaida Zakaria, and Mahaneem Mohamed. 2020. "Composition of Royal Jelly (RJ) and Its Anti-Androgenic Effect on Reproductive Parameters in a Polycystic Ovarian Syndrome (PCOS) Animal Model" Antioxidants 9, no. 6: 499. https://doi.org/10.3390/antiox9060499
APA StyleAb Hamid, N., Abu Bakar, A. B., Mat Zain, A. A., Nik Hussain, N. H., Othman, Z. A., Zakaria, Z., & Mohamed, M. (2020). Composition of Royal Jelly (RJ) and Its Anti-Androgenic Effect on Reproductive Parameters in a Polycystic Ovarian Syndrome (PCOS) Animal Model. Antioxidants, 9(6), 499. https://doi.org/10.3390/antiox9060499









