Sweet Cherry Byproducts Processed by Green Extraction Techniques as a Source of Bioactive Compounds with Antiaging Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Plant Material
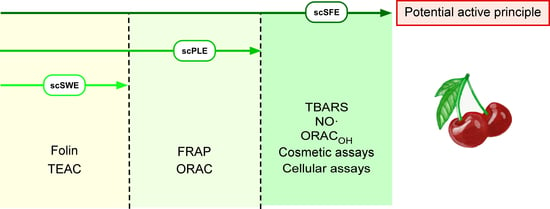
2.3. Green Extraction Techniques
2.3.1. Pressurized Solvent Extraction (PLE)
2.3.2. Supercritical Fluid Extraction (SFE)
2.3.3. Subcritical Water Extraction (SWE)
2.4. HPLC-ESI-QTOF-MS Analysis
2.5. Total Phenolic Content and Antioxidant Activity Assays
2.6. In Vitro Determination of Antiaging Properties by Enzymatic Assays
2.7. Cell Culturing
2.8. ROS Generation and Photoprotection Measurements
2.9. Statistical Analysis
3. Results and Discussion
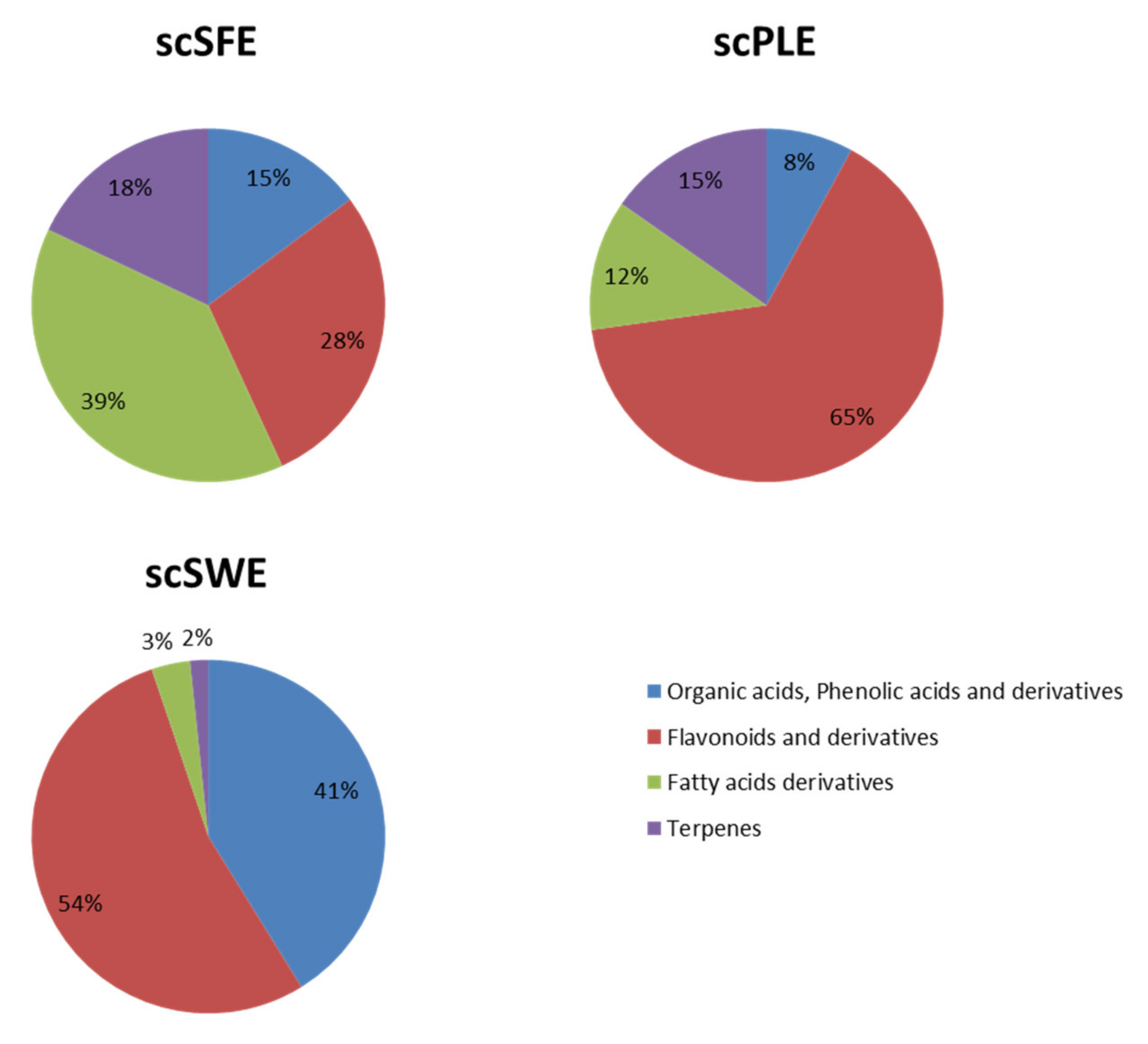
3.1. Characterization of Sweet Cherry Stem Extracts by HPLC-ESI-QTOF-MS
3.2. Total Phenolic Contents and Antioxidant Capacities of the SC Stem Extracts
3.3. Skin Aging-Related Enzymatic Assays
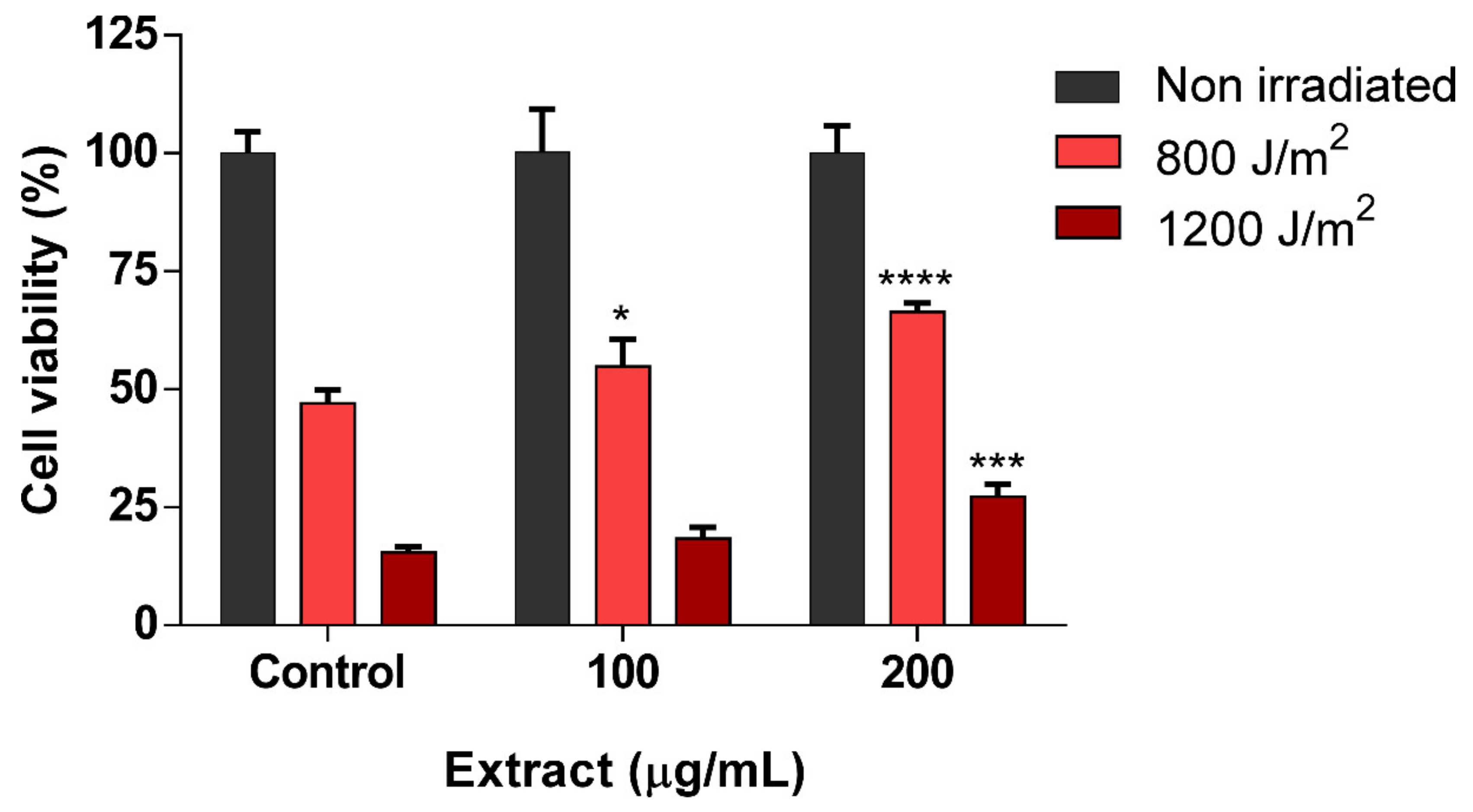
3.4. scSFE Showed Photoprotection Activity against UVB Irradiation
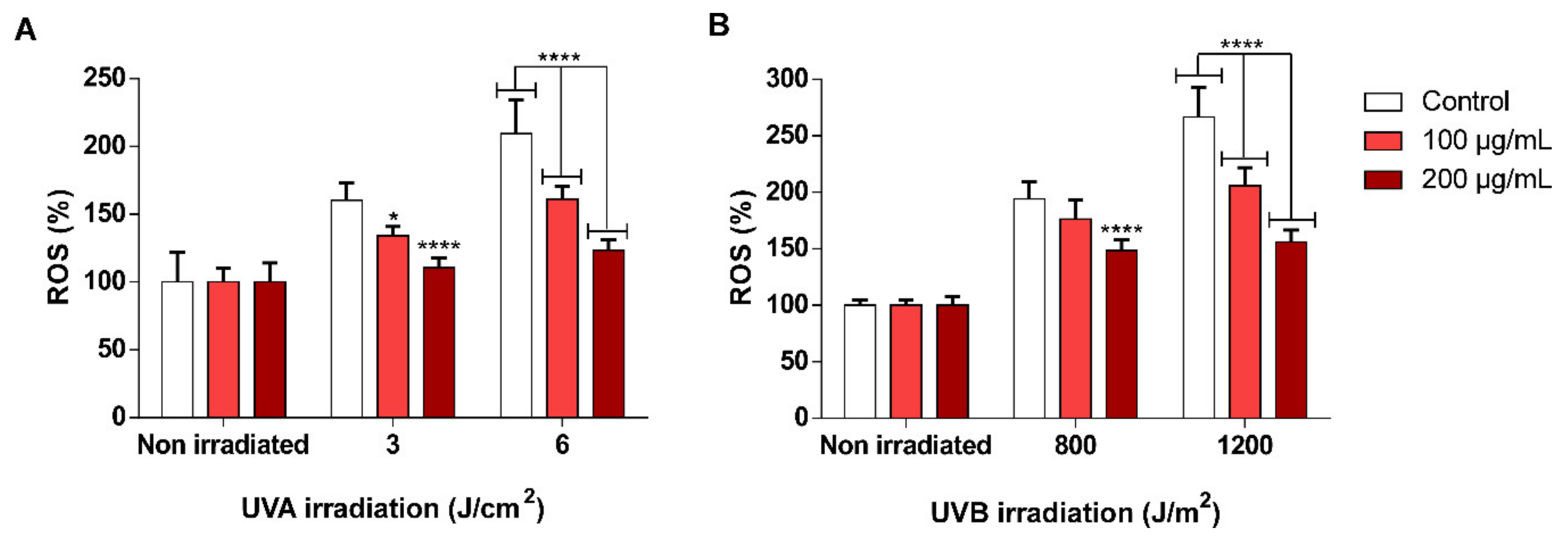
3.5. scSFE Inhibited Intracellular ROS Generation Induced by UVA and UVB Light in HaCaT Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kerscher, M.; Buntrock, H. Update on cosmeceuticals. J. Dtsch. Dermatol. Ges. 2011, 9, 314–326; quiz 327. [Google Scholar] [CrossRef] [PubMed]
- Peschel, W.; Sánchez, F.; Diekmann, W.; Plescher, A.; Gartzía, I.; Jiménez, D.; Lamuela-Raventós, R.M.; Buxaderas, S.; Codina, C. An industrial approach in the search of natural antioxidants from vegetable and fruit wastes. Food Chem. 2006, 97, 137–150. [Google Scholar] [CrossRef]
- Banerjee, J.; Singh, R.; Vijayaraghavan, R.; MacFarlane, D.; Patti, A.F.; Arora, A. Bioactives from fruit processing wastes: Green approaches to valuable chemicals. Food Chem. 2017, 225, 10–22. [Google Scholar] [CrossRef]
- Ayala-Zavala, J.F.; Vega-Vega, V.; Rosas-Domínguez, C.; Palafox-Carlos, H.; Villa-Rodriguez, J.A.; Siddiqui, M.W.; Dávila-Aviña, J.E.; González-Aguilar, G.A. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res. Int. 2011, 44, 1866–1874. [Google Scholar] [CrossRef]
- Sinmisola, A.; Oluwasesan, B.M.; Chukwuemeka, A.P. Blighia sapida k.D. Koenig: A review on its phytochemistry, pharmacological and nutritional properties. J. Ethnopharmacol. 2019, 235, 446–459. [Google Scholar] [CrossRef]
- Aizat, W.M.; Jamil, I.N.; Ahmad-Hashim, F.H.; Noor, N.M. Recent updates on metabolite composition and medicinal benefits of mangosteen plant. PeerJ 2019, 7, e6324. [Google Scholar] [CrossRef]
- Losada-Echeberria, M.; Herranz-Lopez, M.; Micol, V.; Barrajon-Catalan, E. Polyphenols as promising drugs against main breast cancer signatures. Antioxidants 2017, 6, 88. [Google Scholar] [CrossRef]
- Herranz-Lopez, M.; Olivares-Vicente, M.; Encinar, J.A.; Barrajon-Catalan, E.; Segura-Carretero, A.; Joven, J.; Micol, V. Multi-targeted molecular effects of hibiscus sabdariffa polyphenols: An opportunity for a global approach to obesity. Nutrients 2017, 9, 907. [Google Scholar] [CrossRef]
- Barbieri, R.; Coppo, E.; Marchese, A.; Daglia, M.; Sobarzo-Sanchez, E.; Nabavi, S.F.; Nabavi, S.M. Phytochemicals for human disease: An update on plant-derived compounds antibacterial activity. Microbiol. Res. 2017, 196, 44–68. [Google Scholar] [CrossRef]
- Boix-Castejon, M.; Herranz-Lopez, M.; Perez Gago, A.; Olivares-Vicente, M.; Caturla, N.; Roche, E.; Micol, V. Hibiscus and lemon verbena polyphenols modulate appetite-related biomarkers in overweight subjects: A randomized controlled trial. Food Funct. 2018, 9, 3173–3184. [Google Scholar] [CrossRef]
- Peyrol, J.; Meyer, G.; Obert, P.; Dangles, O.; Pechere, L.; Amiot, M.J.; Riva, C. Involvement of bilitranslocase and beta-glucuronidase in the vascular protection by hydroxytyrosol and its glucuronide metabolites in oxidative stress conditions. J. Nutr. Biochem. 2017, 51, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Quirantes-Pine, R.; Herranz-Lopez, M.; Funes, L.; Borras-Linares, I.; Micol, V.; Segura-Carretero, A.; Fernandez-Gutierrez, A. Phenylpropanoids and their metabolites are the major compounds responsible for blood-cell protection against oxidative stress after administration of lippia citriodora in rats. Phytomedicine 2013, 20, 1112–1118. [Google Scholar] [CrossRef] [PubMed]
- Joven, J.; Espinel, E.; Rull, A.; Aragones, G.; Rodriguez-Gallego, E.; Camps, J.; Micol, V.; Herranz-Lopez, M.; Menendez, J.A.; Borras, I.; et al. Plant-derived polyphenols regulate expression of mirna paralogs mir-103/107 and mir-122 and prevent diet-induced fatty liver disease in hyperlipidemic mice. Biochim. Biophys. Acta 2012, 1820, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Herranz-Lopez, M.; Fernandez-Arroyo, S.; Perez-Sanchez, A.; Barrajon-Catalan, E.; Beltran-Debon, R.; Menendez, J.A.; Alonso-Villaverde, C.; Segura-Carretero, A.; Joven, J.; Micol, V. Synergism of plant-derived polyphenols in adipogenesis: Perspectives and implications. Phytomedicine 2012, 19, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Goncalves, A.C.; Rodrigues, M.; Santos, A.O.; Alves, G.; Silva, L.R. Antioxidant status, antidiabetic properties and effects on caco-2 cells of colored and non-colored enriched extracts of sweet cherry fruits. Nutrients 2018, 10, 1688. [Google Scholar] [CrossRef]
- Overbeck, V.; Schmitz, M.; Blanke, M. Targeted forcing improves quality, nutritional and health value of sweet cherry fruit. J. Sci. Food Agric. 2017, 97, 3649–3655. [Google Scholar] [CrossRef]
- Acero, N.; Gradillas, A.; Beltran, M.; Garcia, A.; Munoz Mingarro, D. Comparison of phenolic compounds profile and antioxidant properties of different sweet cherry (Prunus avium L.) varieties. Food Chem. 2019, 279, 260–271. [Google Scholar] [CrossRef]
- Pacifico, S.; Di Maro, A.; Petriccione, M.; Galasso, S.; Piccolella, S.; Di Giuseppe, A.M.A.; Scortichini, M.; Monaco, P. Chemical composition, nutritional value and antioxidant properties of autochthonous Prunus avium cultivars from campania region. Food Res. Int. 2014, 64, 188–199. [Google Scholar] [CrossRef]
- Nastić, N.; Lozano-Sánchez, J.; Borrás-Linares, I.; Švarc-Gajić, J.; Segura-Carretero, A. New technological approaches for recovering bioactive food constituents from sweet cherry (Prunus avium L.) stems. Phytochem. Anal. 2020, 31, 119–130. [Google Scholar]
- Varc-Gajic, J.; Clavijo, S.; Suarez, R.; Cvetanovic, A.; Cerda, V. Simultaneous dispersive liquid-liquid microextraction derivatisation and gas chromatography mass spectrometry analysis of subcritical water extracts of sweet and sour cherry stems. Anal. Bioanal. Chem. 2018, 410, 1943–1953. [Google Scholar] [CrossRef]
- Švarc-Gajić, J.; Cerdà, V.; Clavijo, S.; Suárez, R.; Mašković, P.; Cvetanović, A.; Delerue-Matos, C.; Carvalho, A.P.; Novakov, V. Bioactive compounds of sweet and sour cherry stems obtained by subcritical water extraction. J. Chem. Technol. Biotechnol. 2018, 93, 1627–1635. [Google Scholar] [CrossRef]
- Perez-Sanchez, A.; Barrajon-Catalan, E.; Herranz-Lopez, M.; Castillo, J.; Micol, V. Lemon balm extract (Melissa officinalis, l.) promotes melanogenesis and prevents uvb-induced oxidative stress and DNA damage in a skin cell model. J. Dermatol. Sci. 2016, 84, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Perez-Sanchez, A.; Barrajon-Catalan, E.; Caturla, N.; Castillo, J.; Benavente-Garcia, O.; Alcaraz, M.; Micol, V. Protective effects of citrus and rosemary extracts on uv-induced damage in skin cell model and human volunteers. J. Photochem. Photobiol. B 2014, 136, 12–18. [Google Scholar] [CrossRef]
- Fernandez-Arroyo, S.; Herranz-Lopez, M.; Beltran-Debon, R.; Borras-Linares, I.; Barrajon-Catalan, E.; Joven, J.; Fernandez-Gutierrez, A.; Segura-Carretero, A.; Micol, V. Bioavailability study of a polyphenol-enriched extract from hibiscus sabdariffa in rats and associated antioxidant status. Mol. Nutr. Food Res. 2012, 56, 1590–1595. [Google Scholar] [CrossRef]
- Barrajon-Catalan, E.; Fernandez-Arroyo, S.; Saura, D.; Guillen, E.; Fernandez-Gutierrez, A.; Segura-Carretero, A.; Micol, V. Cistaceae aqueous extracts containing ellagitannins show antioxidant and antimicrobial capacity, and cytotoxic activity against human cancer cells. Food Chem. Toxicol. 2010, 48, 2273–2282. [Google Scholar] [CrossRef]
- Cao, G.; Alessio, H.M.; Cutler, R.G. Oxygen-radical absorbance capacity assay for antioxidants. Free Radic. Biol. Med. 1993, 14, 303–311. [Google Scholar] [CrossRef]
- Cao, G.; Sofic, E.; Prior, R.L. Antioxidant and prooxidant behavior of flavonoids: Structure-activity relationships. Free Radic. Biol. Med. 1997, 22, 749–760. [Google Scholar] [CrossRef]
- Alam, M.N.; Bristi, N.J.; Rafiquzzaman, M. Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharmacogn. J. 2013, 21, 143–152. [Google Scholar] [CrossRef]
- Silva, I.K.; Soysa, P. Evaluation of phytochemical composition and antioxidant capacity of a decoction containing Adenanthera pavonina L. and Thespesia populnea L. Pharmacogn. Mag. 2011, 7, 193–199. [Google Scholar]
- Roy, A.; Sahu, R.K.; Matlam, M.; Deshmukh, V.K.; Dwivedi, J.; Jha, A.K. In vitro techniques to assess the proficiency of skin care cosmetic formulations. Pharmacogn. Rev. 2013, 7, 97–106. [Google Scholar]
- Grosso, C.; Valentão, P.; Ferreres, F.; Andrade, P.B. Alternative and efficient extraction methods for marine-derived compounds. Mar. Drugs 2015, 13, 3182–3230. [Google Scholar] [CrossRef] [PubMed]
- Talmaciu, A.I.; Volf, I.; Popa, V.I. A comparative analysis of the ‘green’ techniques applied for polyphenols extraction from bioresources. Chem. Biodivers. 2015, 12, 1635–1651. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Maity, N.; Nema, N.K.; Sarkar, B.K. Bioactive compounds from natural resources against skin aging. Phytomedicine 2011, 19, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Marzo, N.; Perez-Sanchez, A.; Ruiz-Torres, V.; Martinez-Tebar, A.; Castillo, J.; Herranz-Lopez, M.; Barrajon-Catalan, E. Antioxidant and photoprotective activity of apigenin and its potassium salt derivative in human keratinocytes and absorption in caco-2 cell monolayers. Int. J. Mol. Sci. 2019, 20, 2148. [Google Scholar] [CrossRef]
- Rosero, J.C.; Cruz, S.; Osorio, C.; Hurtado, N. Analysis of phenolic composition of byproducts (seeds and peels) of avocado (persea americana mill.) cultivated in colombia. Molecules 2019, 24, 3209. [Google Scholar] [CrossRef]
- Zhao, C.N.; Tang, G.Y.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Liu, Q.; Mao, Q.Q.; Shang, A.; Li, H.B. Phenolic profiles and antioxidant activities of 30 tea infusions from green, black, oolong, white, yellow and dark teas. Antioxidants 2019, 8, 215. [Google Scholar] [CrossRef]
- Ahmad-Qasem, M.H.; Barrajon-Catalan, E.; Micol, V.; Cárcel, J.A.; Garcia-Perez, J.V. Influence of air temperature on drying kinetics and antioxidant potential of olive pomace. J. Food Eng. 2013, 119, 516–524. [Google Scholar] [CrossRef]
- Tomás-Menor, L.; Morales-Soto, A.; Barrajón-Catalán, E.; Roldán-Segura, C.; Segura-Carretero, A.; Micol, V. Correlation between the antibacterial activity and the composition of extracts derived from various spanish cistus species. Food Chem. Toxicol. 2013, 55, 313–322. [Google Scholar] [CrossRef]
- Ou, B.; Huang, D.; Hampsch-Woodill, M.; Flanagan, J.A.; Deemer, E.K. Analysis of antioxidant activities of common vegetables employing oxygen radical absorbance capacity (orac) and ferric reducing antioxidant power (frap) assays: A comparative study. J. Agric. Food Chem. 2002, 50, 3122–3128. [Google Scholar] [CrossRef]
- Umeno, A.; Horie, M.; Murotomi, K.; Nakajima, Y.; Yoshida, Y. Antioxidative and antidiabetic effects of natural polyphenols and isoflavones. Molecules 2016, 21, 708. [Google Scholar] [CrossRef]
- He, M.T.; Lee, A.Y.; Park, C.H.; Cho, E.J. Protective effect of cordyceps militaris against hydrogen peroxide-induced oxidative stress in vitro. Nutr. Res. Pract. 2019, 13, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Fokou, P.V.T.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of naringenin: A review of clinical trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Rashmi, R.; Bojan Magesh, S.; Mohanram Ramkumar, K.; Suryanarayanan, S.; Venkata SubbaRao, M. Antioxidant potential of naringenin helps to protect liver tissue from streptozotocin-induced damage. Rep. Biochem. Mol. Biol. 2018, 7, 76–84. [Google Scholar]
- Grzesik, M.; Naparlo, K.; Bartosz, G.; Sadowska-Bartosz, I. Antioxidant properties of catechins: Comparison with other antioxidants. Food Chem. 2018, 241, 480–492. [Google Scholar] [CrossRef]
- Koch, W.; Kukula-Koch, W.; Glowniak, K. Catechin composition and antioxidant activity of black teas in relation to brewing time. J. Aoac. Int. 2017, 100, 1694–1699. [Google Scholar] [CrossRef]
- Mani, R.; Natesan, V. Chrysin: Sources, beneficial pharmacological activities, and molecular mechanism of action. Phytochemistry 2018, 145, 187–196. [Google Scholar] [CrossRef]
- Niu, Q.; Gao, Y.; Liu, P. Optimization of microwave-assisted extraction, antioxidant capacity, and characterization of total flavonoids from the leaves of alpinia oxyphylla miq. Prep. Biochem. Biotechnol. 2020, 50, 82–90. [Google Scholar] [CrossRef]
- Naz, S.; Imran, M.; Rauf, A.; Orhan, I.E.; Shariati, M.A.; Iahtisham Ul, H.; Plygun, S.; Shahbaz, M.; Qaisrani, T.B.; Shah, Z.A.; et al. Chrysin: Pharmacological and therapeutic properties. Life Sci. 2019, 235, 116797. [Google Scholar] [CrossRef]
- Thring, T.S.; Hili, P.; Naughton, D.P. Anti-collagenase, anti-elastase and anti-oxidant activities of extracts from 21 plants. BMC Complement. Altern. Med. 2009, 9, 27. [Google Scholar] [CrossRef]
- Burlando, B.; Pastorino, G.; Salis, A.; Damonte, G.; Clericuzio, M.; Cornara, L. The bioactivity of hedysarum coronarium extracts on skin enzymes and cells correlates with phenolic content. Pharm. Biol. 2017, 55, 1984–1991. [Google Scholar] [CrossRef]
- Chatatikun, M.; Chiabchalard, A. Thai plants with high antioxidant levels, free radical scavenging activity, anti-tyrosinase and anti-collagenase activity. BMC Complement. Altern. Med. 2017, 17, 487. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Muramatsu, T.; Yamashina, Y.; Shirai, T.; Ohnishi, T.; Mori, T. Melanin reduces ultraviolet-induced DNA damage formation and killing rate in cultured human melanoma cells. J. Investig. Dermatol. 1993, 101, 685–689. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A.; Gostner, J.; Fuchs, J.E.; Chaita, E.; Aligiannis, N.; Skaltsounis, L.; Ganzera, M. Inhibition of collagenase by mycosporine-like amino acids from marine sources. Planta Med. 2015, 81, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Vostalova, J.; Tinkova, E.; Biedermann, D.; Kosina, P.; Ulrichova, J.; Rajnochova Svobodova, A. Skin protective activity of silymarin and its flavonolignans. Molecules 2019, 24, 1022. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F. Potential role of natural compounds against skin aging. Curr. Med. Chem. 2015, 22, 1515–1538. [Google Scholar] [CrossRef]
- Baiao, D.D.S.; de Freitas, C.S.; Gomes, L.P.; da Silva, D.; Correa, A.; Pereira, P.R.; Aguila, E.M.D.; Paschoalin, V.M.F. Polyphenols from root, tubercles and grains cropped in brazil: Chemical and nutritional characterization and their effects on human health and diseases. Nutrients 2017, 9, 1044. [Google Scholar] [CrossRef]
- Binic, I.; Lazarevic, V.; Ljubenovic, M.; Mojsa, J.; Sokolovic, D. Skin ageing: Natural weapons and strategies. Evid. Based Complement. Altern. Med. 2013, 2013, 827248. [Google Scholar] [CrossRef]
- Dzialo, M.; Mierziak, J.; Korzun, U.; Preisner, M.; Szopa, J.; Kulma, A. The potential of plant phenolics in prevention and therapy of skin disorders. Int. J. Mol. Sci. 2016, 17, 160. [Google Scholar] [CrossRef]
- Eun Lee, K.; Bharadwaj, S.; Yadava, U.; Gu Kang, S. Evaluation of caffeine as inhibitor against collagenase, elastase and tyrosinase using in silico and in vitro approach. J. Enzyme Inhib. Med. Chem. 2019, 34, 927–936. [Google Scholar] [CrossRef]
- Zaluski, D.; Olech, M.; Kuzniewski, R.; Verpoorte, R.; Nowak, R.; Smolarz, H.D. Lc-esi-ms/ms profiling of phenolics from eleutherococcus spp. Inflorescences, structure-activity relationship as antioxidants, inhibitors of hyaluronidase and acetylcholinesterase. Saudi Pharm. J. 2017, 25, 734–743. [Google Scholar] [CrossRef]
- Pedrosa, T.D.; Barros, A.O.; Nogueira, J.R.; Fruet, A.C.; Rodrigues, I.C.; Calcagno, D.Q.; Smith, M.A.; de Souza, T.P.; Barros, S.B.; de Vasconcellos, M.C.; et al. Anti-wrinkle and anti-whitening effects of juca (Libidibia ferrea mart.) extracts. Arch. Dermatol. Res. 2016, 308, 643–654. [Google Scholar] [CrossRef] [PubMed]
- Ochocka, R.; Hering, A.; Stefanowicz-Hajduk, J.; Cal, K.; Baranska, H. The effect of mangiferin on skin: Penetration, permeation and inhibition of ecm enzymes. PLoS ONE 2017, 12, e0181542. [Google Scholar] [CrossRef] [PubMed]
- Promden, W.; Viriyabancha, W.; Monthakantirat, O.; Umehara, K.; Noguchi, H.; De-Eknamkul, W. Correlation between the potency of flavonoids on mushroom tyrosinase inhibitory activity and melanin synthesis in melanocytes. Molecules 2018, 23, 1403. [Google Scholar] [CrossRef] [PubMed]
- Zolghadri, S.; Bahrami, A.; Hassan Khan, M.T.; Munoz-Munoz, J.; Garcia-Molina, F.; Garcia-Canovas, F.; Saboury, A.A. A comprehensive review on tyrosinase inhibitors. J. Enzyme Inhib. Med. Chem. 2019, 34, 279–309. [Google Scholar] [CrossRef] [PubMed]
- Herranz-López, M.; Losada-Echeberría, M.; Barrajón-Catalán, E. The multitarget activity of natural extracts on cancer: Synergy and xenohormesis. Medicines 2018, 6, 6. [Google Scholar] [CrossRef]
- Sánchez-Marzo, N.; Pérez-Sánchez, A.; Barrajón-Catalán, E.; Castillo, J.; Herranz-López, M.; Micol, V. Rosemary diterpenes and flavanone aglycones provide improved genoprotection against uv-induced DNA damage in a human skin cell model. Antioxidants 2020, 9, 255. [Google Scholar] [CrossRef] [PubMed]
- Kostyuk, V.; Potapovich, A.; Albuhaydar, A.R.; Mayer, W.; De Luca, C.; Korkina, L. Natural substances for prevention of skin photoaging: Screening systems in the development of sunscreen and rejuvenation cosmetics. Rejuvenation Res. 2018, 21, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Almeida, I.F.; Pinto, A.S.; Monteiro, C.; Monteiro, H.; Belo, L.; Fernandes, J.; Bento, A.R.; Duarte, T.L.; Garrido, J.; Bahia, M.F.; et al. Protective effect of c. Sativa leaf extract against uv mediated-DNA damage in a human keratinocyte cell line. J. Photochem. Photobiol. B 2015, 144, 28–34. [Google Scholar] [CrossRef]
- Moreira, L.C.; de Avila, R.I.; Veloso, D.; Pedrosa, T.N.; Lima, E.S.; do Couto, R.O.; Lima, E.M.; Batista, A.C.; de Paula, J.R.; Valadares, M.C. In vitro safety and efficacy evaluations of a complex botanical mixture of eugenia dysenterica dc. (myrtaceae): Prospects for developing a new dermocosmetic product. Toxicol. In Vitro 2017, 45, 397–408. [Google Scholar] [CrossRef]
- Cavinato, M.; Waltenberger, B.; Baraldo, G.; Grade, C.V.C.; Stuppner, H.; Jansen-Durr, P. Plant extracts and natural compounds used against uvb-induced photoaging. Biogerontology 2017, 18, 499–516. [Google Scholar] [CrossRef]
- Magcwebeba, T.; Swart, P.; Swanevelder, S.; Joubert, E.; Gelderblom, W. Anti-inflammatory effects of aspalathus linearis and cyclopia spp. Extracts in a uvb/keratinocyte (hacat) model utilising interleukin-1alpha accumulation as biomarker. Molecules 2016, 21, 1323. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.Y.; Huang, C.C.; Chu, Y.; Huang, Y.H.; Lin, P.; Liu, Y.H.; Wen, K.C.; Lin, C.Y.; Hsu, M.C.; Chiang, H.M. Alleviation of ultraviolet b-induced photodamage by coffea arabica extract in human skin fibroblasts and hairless mouse skin. Int. J. Mol. Sci. 2017, 18, 782. [Google Scholar] [CrossRef] [PubMed]
- El-Mahdy, M.A.; Zhu, Q.; Wang, Q.E.; Wani, G.; Patnaik, S.; Zhao, Q.; Arafa el, S.; Barakat, B.; Mir, S.N.; Wani, A.A. Naringenin protects hacat human keratinocytes against uvb-induced apoptosis and enhances the removal of cyclobutane pyrimidine dimers from the genome. Photochem. Photobiol. 2008, 84, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Hwang, K.; Lee, J.; Han, S.Y.; Kim, E.M.; Park, J.; Cho, J.Y. Skin protective effect of epigallocatechin gallate. Int. J. Mol. Sci. 2018, 19, 173. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.N.; Shankar, S.; Srivastava, R.K. Green tea catechin, epigallocatechin-3-gallate (egcg): Mechanisms, perspectives and clinical applications. Biochem. Pharmacol. 2011, 82, 1807–1821. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.E.; Choi-Kwon, S.; Park, K.A.; Lee, H.J.; Park, M.S.; Lee, J.H.; Kwon, S.B.; Park, K.C. Dietary supplementation of (+)-catechin protects against uvb-induced skin damage by modulating antioxidant enzyme activities. Photodermatol. Photoimmunol. Photomed. 2003, 19, 235–241. [Google Scholar] [CrossRef]
- Hu, Y.; Ma, Y.; Wu, S.; Chen, T.; He, Y.; Sun, J.; Jiao, R.; Jiang, X.; Huang, Y.; Deng, L.; et al. Protective effect of cyanidin-3-o-glucoside against ultraviolet b radiation-induced cell damage in human hacat keratinocytes. Front. Pharmacol. 2016, 7, 301. [Google Scholar] [CrossRef]
- Wu, N.L.; Fang, J.Y.; Chen, M.; Wu, C.J.; Huang, C.C.; Hung, C.F. Chrysin protects epidermal keratinocytes from uva- and uvb-induced damage. J. Agric. Food Chem. 2011, 59, 8391–8400. [Google Scholar] [CrossRef]
- Myhre, O.; Andersen, J.M.; Aarnes, H.; Fonnum, F. Evaluation of the probes 2′,7′-dichlorofluorescin diacetate, luminol, and lucigenin as indicators of reactive species formation. Biochem. Pharmacol. 2003, 65, 1575–1582. [Google Scholar] [CrossRef]
- Ichihashi, M.; Ueda, M.; Budiyanto, A.; Bito, T.; Oka, M.; Fukunaga, M.; Tsuru, K.; Horikawa, T. Uv-induced skin damage. Toxicology 2003, 189, 21–39. [Google Scholar] [CrossRef]
- Kawashima, S.; Funakoshi, T.; Sato, Y.; Saito, N.; Ohsawa, H.; Kurita, K.; Nagata, K.; Yoshida, M.; Ishigami, A. Protective effect of pre- and post-vitamin c treatments on uvb-irradiation-induced skin damage. Sci. Rep. 2018, 8, 16199. [Google Scholar] [CrossRef] [PubMed]
- Gęgotek, A.; Ambrożewicz, E.; Jastrząb, A.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Rutin and ascorbic acid cooperation in antioxidant and antiapoptotic effect on human skin keratinocytes and fibroblasts exposed to uva and uvb radiation. Arch. Dermatol. Res. 2019, 311, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, S.; Tada, M.; Yamada, K.; Takahata, K. Suppressive effects of ascorbate derivatives on ultraviolet-b-induced injury in hacat human keratinocytes. In Vitro Cell. Dev. Biol. Anim. 2004, 40, 71–73. [Google Scholar] [CrossRef]
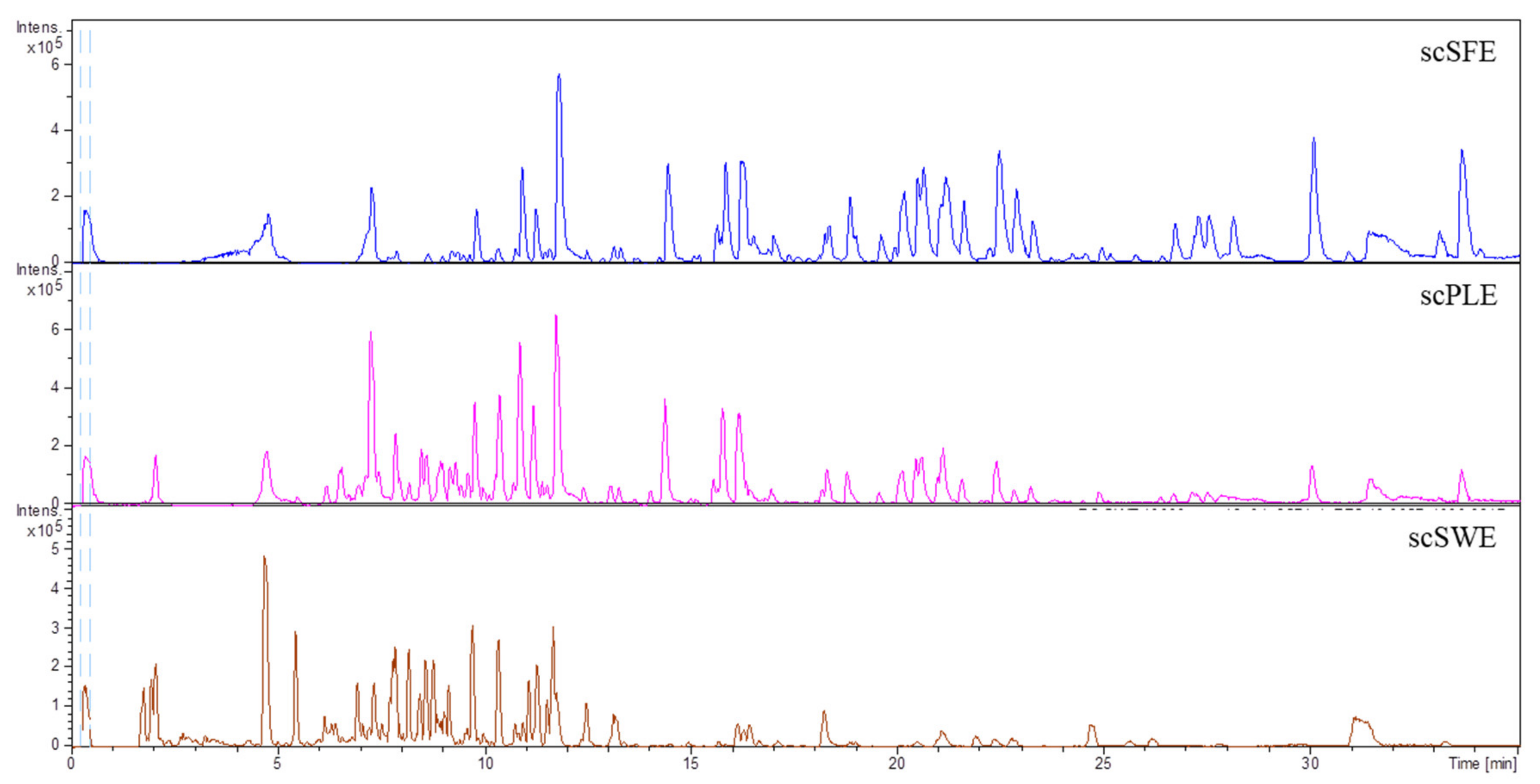
| Retention Time (min) | m/z Experimental | m/z Calculated | (M – H)– | Error (ppm) | Proposed Compound | Extracts |
|---|---|---|---|---|---|---|
| 1.91 | 195.0499 | 195.0510 | C6H11O7 | 5.7 | D-gluconic acid | scSWE |
| 1.99 | 191.0552 | 191.0561 | C7H11O6 | 5.0 | quinic acid | scSWE |
| 5.42 | 315.0702 | 315.0722 | C13H15O9 | 6.3 | protocatechuic acid hexoside | scSWE |
| 6.52 | 577.1381 | 577.1351 | C30H25O12 | −5 | proanthocyanidin B2 isomer 1 | scSFE |
| 6.81 | 341.0856 | 341.0878 | C15H17O9 | 6.6 | caffeic acid hexoside | scSFE, scSWE |
| 7.23 | 289.0732 | 289.0718 | C15H13O6 | −5.1 | (epi)catechin isomer 1 | scSFE, scPLE, scSWE |
| 7.31 | 401.1446 | 401.1453 | C18H25O10 | 1.7 | benzyl β-primeveroside | scSWE |
| 7.40 | 137.0243 | 137.0244 | C7H5O3 | 0.6 | salicylic acid | scSWE |
| 7.43 | 577.1381 | 577.1351 | C30H25O12 | −5.1 | proanthocyanidin B2 isomer 2 | scSFE |
| 7.54 | 771.1977 | 771.1989 | C33H39O21 | 1.6 | quercetin-rutinoside-glucoside | scSWE |
| 7.60 | 521.2002 | 521.2028 | C26H33O11 | 5.1 | dihydrodehydrodiconiferyl alcohol glucopyranoside | scSWE |
| 7.64 | 325.0930 | 325.0929 | C15H17O8 | −0.3 | p-coumaric acid O-hexoside | scSFE, scPLE |
| 7.74 | 449.1094 | 449.1089 | C21H21O11 | −1.1 | eriodictyol glucoside isomer 1 | scSWE |
| 7.81 | 165.0557 | 165.0557 | C15H17O8 | 0.4 | melilotic acid | scSWE |
| 7.83 | 289.0735 | 289.0718 | C15H13O6 | −5.9 | (epi)catechin isomer 2 | scSFE, scPLE |
| 7.88 | 449.1094 | 449.1089 | C21H21O11 | −1.1 | eriodictyol glucoside isomer 2 | scSWE |
| 8.16 | 195.0664 | 195.0663 | C10H11O4 | −0.8 | dihydroferulic acid | scSWE |
| 8.45 | 609.1482 | 609.1461 | C27H29O16 | −2.1 | rutin | scSFE, scSWE |
| 8.58 | 465.1073 | 465.1038 | C21H21O12 | −3.4 | epicatechin-O-glucuronide | scSFE, scPLE, scSWE |
| 8.85 | 463.0903 | 463.0882 | C21H19O12 | −2.1 | quercetin-glucoside | scPLE, scSWE |
| 8.91 | 431.1011 | 431.0984 | C21H19O10 | −6.2 | genistein-O-glucoside isomer 1 | scSFE, scPLE, scSWE |
| 8.95 | 593.1534 | 593.1512 | C27H29O15 | −3.7 | kaempferol-O-rutinoside | scSFE, scPLE, scSWE |
| 9.13 | 477.1076 | 477.1038 | C22H21O12 | −7.9 | isorhamnetin-glucoside | scSFE, scPLE |
| 9.15 | 431.1007 | 431.0984 | C21H19O10 | −5.3 | genistein-O-glucoside isomer 2 | scSFE, scPLE |
| 9.28 | 431.1004 | 431.0984 | C21H19O10 | −4.6 | genistein-O-glucoside isomer 3 | scSFE, scPLE, scSWE |
| 9.42 | 447.0955 | 447.0933 | C21H19O11 | −4.9 | kaempferol-O-glucoside | scSFE, scPLE, scSWE |
| 9.58 | 431.0995 | 431.0984 | C21H19O10 | −2.7 | genistein-O-glucoside isomer 4 | scSFE, scPLE |
| 9.75 | 433.1158 | 433.1140 | C21H21O10 | −4.1 | naringenin-O-glucoside isomer 1 | scSFE, scPLE, scSWE |
| 10.25 | 433.1123 | 433.1140 | C21H21O10 | 4.0 | naringenin-O-glucoside isomer 2 | scSFE, scPLE |
| 10.35 | 417.1182 | 417.1191 | C21H21O9 | 2.2 | liquiritin | scSWE |
| 10.69 | 433.1148 | 433.1140 | C21H21O10 | −1.7 | naringenin-O-glucoside isomer 3 | scSFE, scPLE |
| 10.74 | 447.1295 | 447.1297 | C22H23O10 | 0.3 | sakuranin | scSWE |
| 10.84 | 415.1064 | 415.1035 | C21H19O9 | −6.2 | chrysin-O-glucoside | scSFE, scPLE |
| 10.94 | 447.129 | 447.1297 | C22H23O10 | 1.6 | sakuranetin glucopyranoside | scSWE |
| 11.07 | 417.1204 | 417.1191 | C21H21O9 | −3.1 | sakuranetin xylopyranoside | scSWE |
| 11.17 | 433.1176 | 433.1140 | C21H21O10 | −8.2 | naringenin-O-glucoside isomer 4 | scSFE, scPLE |
| 11.27 | 417.1196 | 417.1191 | C21H21O9 | −1.2 | prupersin B | scSWE |
| 11.62 | 447.1307 | 447.1297 | C22H23O10 | −2.4 | dihydrowogonin glucoside | scSWE |
| 12.39 | 327.2175 | 327.2177 | C18H31O5 | 0.6 | trihydroxy-octadecadienoic acid | scSFE, scPLE, scSWE |
| 12.78 | 433.1161 | 433.1140 | C21H21O10 | −4.9 | naringenin-O-glucoside isomer 5 | scSFE, scPLE |
| 13.04 | 329.2346 | 329.2333 | C18H33O5 | −3.8 | trihydroxy-octadecenoic acid | scSFE, scPLE, scSWE |
| 13.23 | 271.0607 | 271.0612 | C15H11O5 | 1.7 | naringenin isomer 1 | scSFE, scPLE, scSWE |
| 13.53 | 271.0600 | 271.0612 | C15H11O5 | 4.2 | naringenin isomer 2 | scSFE, scPLE |
| 14.35 | 517.3187 | 517.3171 | C30H45O7 | −3.2 | jaligonic acid | scSFE, scPLE |
| 15.74 | 501.3250 | 501.3222 | C30H45O6 | −5.7 | hydroxyceanothic acid isomer 1 | scSFE, scPLE |
| 16.14 | 253.0503 | 253.0506 | C15H9O4 | 1.4 | chrysin | scSFE, scPLE, scSWE |
| 16.31 | 285.0775 | 285.0768 | C16H13O5 | −2.3 | methylnaringenin | scSFE, scPLE, scSWE |
| 16.44 | 255.0644 | 255.0663 | C15H11O4 | 7.3 | pinocembrin | scSFE, scPLE, scSWE |
| 16.77 | 501.3238 | 501.3222 | C30H45O6 | −3.2 | hydroxyceanothic acid isomer 2 | scSFE, scPLE |
| 19.53 | 293.2122 | 293.2122 | C18H29O3 | 0.0 | hydroxy-octadecatrienoic acid isomer 1 | scSFE, scPLE |
| 19.87 | 293.2104 | 293.2122 | C18H29O3 | 6.1 | hydroxy-octadecatrienoic acid isomer 2 | scSFE, scPLE |
| 21.52 | 295.2291 | 295.2279 | C18H31O3 | −4.2 | hydroxy-octadecadienoic acid | scSFE, scPLE |
| 23.21 | 293.2111 | 293.2122 | C18H29O3 | 3.7 | hydroxy-octadecatrienoic acid isomer 3 | scSFE, scPLE |
| 30.02 | 277.2182 | 277.2173 | C18H29O2 | −3.4 | linolenic acid | scSFE, scPLE |
| 31.41 | 455.3528 | 455.3531 | C30H47O3 | 0.7 | ursolic acid | scSWE |
| 33.64 | 279.2327 | 279.2330 | C18H31O2 | 1.1 | linoleic acid | scSFE, scPLE |
| 34.06 | 299.2590 | 299.2592 | C18H35O3 | 0.4 | hydroxy-octadecanoic acid | scSFE, scPLE |
| Proposed Compound | Peak Area x E+4 | ||
|---|---|---|---|
| PLE | SFE | SWE | |
| Organic acids, phenolic acids, and derivatives | |||
| D-gluconic acid | ND | ND | 22.8 ± 0.9 |
| quinic acid | ND | ND | 32.0 ± 2.0 |
| caffeic acid hexoside | 5.0 ± 0.3 | ND | 2.2 ± 0.2 |
| p-coumaric acid O-hexoside | 12.0 ± 2.0 | 4.0 ± 0.2 | ND |
| protocatechuic acid hexoside | ND | ND | 17.2 ± 0.2 |
| salicylic acid | ND | ND | 9.9 ± 0.5 |
| melilotic acid | ND | ND | 16.6 ± 0.7 |
| dihydroferulic acid | ND | ND | 22 ± 2 |
| Flavonoids and derivatives | |||
| (epi)catechin–(epi)catechin (proanthocyanidin B2) isomer 1 | 129.0 ± 8.0 | ND | ND |
| (epi)catechin isomer 1 | 573.0 ± 14.0 | 282.0 ± 23.0 | 5.5 ± 0.3 |
| (epi)catechin–(epi)catechin (proanthocyanidin B2) isomer 2 | 112.0 ± 9.0 | ND | ND |
| (epi)catechin isomer 2 | 214.0 ± 44.0 | 44 ± 4 | ND |
| rutin | 121.0 ± 3.0 | ND | 13.4 ± 0.4 |
| epicatechin-O-glucuronide | 113.0 ± 1.0 | 20.0 ± 0.5 | 25.0 ± 1.0 |
| quercetin-glucoside | 67.0 ± 0.6 | ND | 11.0 ± 1.0 |
| genistein-O-glucoside isomer 1 | 89.0 ± 0.1 | 12.0 ± 0.5 | 27.8 ± 0.9 |
| kaempferol-O-rutinoside | 91.0 ± 4.0 | 4.0 ± 0.2 | 6.3 ± 0.5 |
| isorhamnetin-glucoside | 15.0 ± 0.2 | 3.0 ± 0.3 | ND |
| genistein-O-glucoside isomer 2 | 65.0 ± 4.0 | 16.0 ± 1.0 | ND |
| genistein-O-glucoside isomer 3 | 102 ± 0.8 | 24.0 ± 1.0 | 8.4 ± 0.3 |
| kaempferol-O-glucoside | 41.0 ± 2.0 | 8.0 ± 0.2 | 4.6 ± 0.6 |
| genistein-O-glucoside isomer 4 | 82.0 ± 0.0 | 17.0 ± 1.0 | ND |
| naringenin-O-glucoside isomer 1 | 94.0 ± 0.2 | 34.0 ± 1.0 | 38 ± 1 |
| naringenin-O-glucoside isomer 2 | 89.0 ± 0.4 | 40.0 ± 1.0 | ND |
| naringenin-O-glucoside isomer 3 | 57.0 ± 1.0 | 34.0 ± 4.0 | ND |
| chrysin-O-glucoside | 432.0 ± 14.0 | 180.0 ± 1.0 | ND |
| naringenin-O-glucoside isomer 4 | 91.0 ± 0.1 | 49.0 ± 5.0 | ND |
| naringenin-O-glucoside isomer 5 | 6.0 ± 0.0 | 10.0 ± 0.5 | ND |
| naringenin isomer 1 | 14.0 ± 1.0 | 13.0 ± 0.8 | 8.8 ± 0.2 |
| naringenin isomer 2 | 9 ± 0.2 | 5 ± 0.4 | ND |
| chrysin | 143.0 ± 12.0 | 143 ± 3 | 3.8 ± 0.8 |
| methylnaringenin | 39.0 ± 2.0 | 42.0 ± 1.0 | 3.0 ± 0.3 |
| benzyl β-primeveroside | ND | ND | 24.6 ± 0.4 |
| quercetin-rutinoside-glucoside | ND | ND | 5.9 ± 0.6 |
| dihydrodehydrodiconiferyl alcohol glucopyranoside | ND | ND | 4.6 ± 0.3 |
| eriodictyol-glucoside isomer 1 | ND | ND | 14.4 ± 0.6 |
| eriodictyol-glucoside isomer 2 | ND | ND | 36 ± 1 |
| liquiritin | ND | ND | 24 ± 1 |
| sakuranin | ND | ND | 4.3 ± 0.3 |
| sakuranetin-glucopyranoside | ND | ND | 4.9 ± 0.3 |
| sakuranin-xylopyranoside | ND | ND | 18.2 ± 0.2 |
| prupersin B | ND | ND | 22.3 ± 0.4 |
| dihydrowogonin glucoside | ND | ND | 35 ± 2 |
| pinocembrin | ND | ND | 5.3 ± 0.1 |
| Fatty acid derivatives | |||
| trihydroxy-octadecadienoic acid | 42.0 ± 6.0 | 29.0 ± 2.0 | 11.6 ± 0.6 |
| trihydroxy-octadecenoic acid | 48.0 ± 2.0 | 40.0 ± 2.0 | 7.4 ± 0.2 |
| hydroxy-octadecatrienoic acid isomer 1 | 19.0 ± 0.3 | 59.0 ± 0.2 | ND |
| hydroxy-octadecatrienoic acid isomer 2 | 9.0 ± 0.5 | 40 ± 0.8 | ND |
| hydroxy-octadecadienoic acid | 76.0 ± 2.0 | 210.0 ± 12.0 | ND |
| hydroxy-octadecatrienoic acid isomer 3 | 57.0 ± 4.0 | 121.0 ± 5.0 | ND |
| linolenic acid | 129.0 ± 15.0 | 407.0 ± 39.0 | ND |
| linoleic acid | 129.0 ± 2.0 | 448.0 ± 39.0 | ND |
| hydroxy-octadecanoic acid | 13.0 ± 0.3 | 49.0 ± 3.0 | ND |
| Terpenes | |||
| jaligonic acid | 333.0 ± 2.0 | 297.0 ± 10.0 | ND |
| ursolic acid | ND | ND | 58.0 ± 11.0 |
| hydroxyceanothic acid isomer 1 | 279.0 ± 7.0 | 273.0 ± 6.0 | ND |
| hydroxyceanothic acid isomer 2 | 54.0 ± 3.0 | 78.0 ± 6.0 | ND |
| Extract | Folin % GAE (w/w) | TEAC mmol Trolox eq./100 g Extract |
|---|---|---|
| scSFE | 15.26 ± 2.94 | 240.61 ± 11.74 |
| scPLE | 18.81 ± 2.36 | 220.53 ± 13.57 |
| scSWE | 5.49 ± 1.16 | 70.38 ± 3.89 |
| Extract | FRAP mmol FeSO4 eq./100 g Extract | ORAC mmol Trolox eq./100 g Extract |
|---|---|---|
| scSFE | 64.83 ± 6.32 | 107.77 ± 5.76 |
| scPLE | 203.94 ± 8.37 | 64.15 ± 1.04 |
| TBARS | ORACOH | % NO· Depletion | |
|---|---|---|---|
| scSFE | 45.13 ± 10.84 | 189.10 ± 0.81 | 29.37 ± 0.01 |
| Percentage of Inhibition | |||||
|---|---|---|---|---|---|
| Extract | Collagenase | Tyrosinase | Elastase | Hyaluronidase | Glycosylation |
| scSFE | −11.76 ± 2.71 ns | 40.47 ± 19.35 ns | 164.11 ± 27.33 ** | 90.80 ± 5.93 **** | 51.76 ± 7.06 **** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agulló-Chazarra, L.; Borrás-Linares, I.; Lozano-Sánchez, J.; Segura-Carretero, A.; Micol, V.; Herranz-López, M.; Barrajón-Catalán, E. Sweet Cherry Byproducts Processed by Green Extraction Techniques as a Source of Bioactive Compounds with Antiaging Properties. Antioxidants 2020, 9, 418. https://doi.org/10.3390/antiox9050418
Agulló-Chazarra L, Borrás-Linares I, Lozano-Sánchez J, Segura-Carretero A, Micol V, Herranz-López M, Barrajón-Catalán E. Sweet Cherry Byproducts Processed by Green Extraction Techniques as a Source of Bioactive Compounds with Antiaging Properties. Antioxidants. 2020; 9(5):418. https://doi.org/10.3390/antiox9050418
Chicago/Turabian StyleAgulló-Chazarra, Luz, Isabel Borrás-Linares, Jesús Lozano-Sánchez, Antonio Segura-Carretero, Vicente Micol, María Herranz-López, and Enrique Barrajón-Catalán. 2020. "Sweet Cherry Byproducts Processed by Green Extraction Techniques as a Source of Bioactive Compounds with Antiaging Properties" Antioxidants 9, no. 5: 418. https://doi.org/10.3390/antiox9050418
APA StyleAgulló-Chazarra, L., Borrás-Linares, I., Lozano-Sánchez, J., Segura-Carretero, A., Micol, V., Herranz-López, M., & Barrajón-Catalán, E. (2020). Sweet Cherry Byproducts Processed by Green Extraction Techniques as a Source of Bioactive Compounds with Antiaging Properties. Antioxidants, 9(5), 418. https://doi.org/10.3390/antiox9050418