Antioxidant Activity of Graptopetalum paraguayense E. Walther Leaf Extract Counteracts Oxidative Stress Induced by Ethanol and Carbon Tetrachloride Co-Induced Hepatotoxicity in Rats
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of GE50 Extract of G. Paraguayense
2.2. Animal Treatment
2.3. Serum and Liver Tissue Preparation
2.4. Determination of AST, ALT, LDH, and TAS Serum Levels in Rats
2.5. Measurement of MDA and GSH Levels, and GPx, SOD, and CAT, and GST Activities
2.6. Measurement of Antioxidants
2.7. Histopathology
2.8. Characterization of Phenolic Compounds in GE50 Extracts
2.9. Statistical Analysis
3. Results
3.1. Effects of GE50 on Body Weight Gain, Feed Efficiency, and Liver Index in Ethanol + CCl4 -Treated Group
3.2. Effects of GE50 on Ethanol + CCl4 -Induced Hepatotoxicity
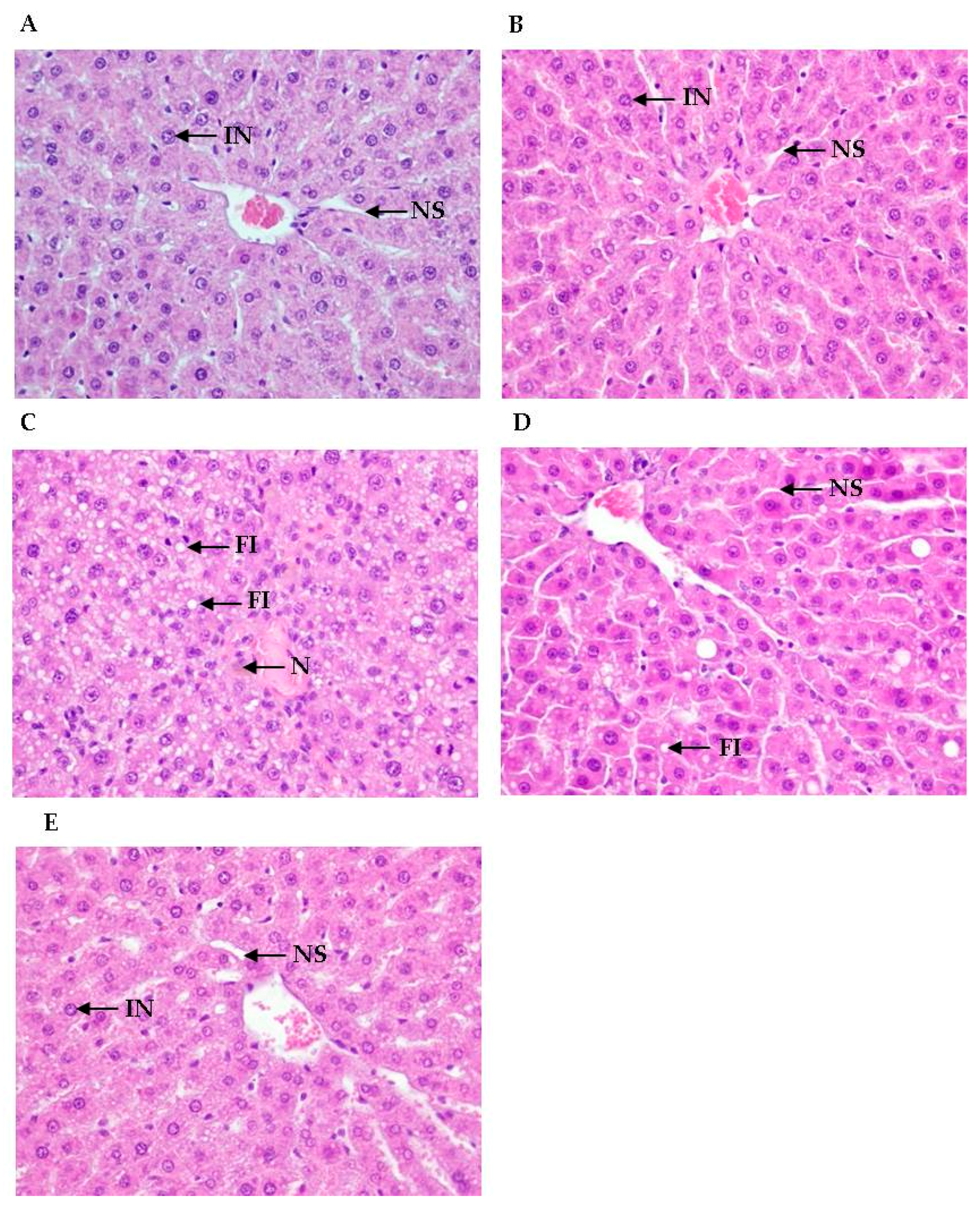
3.3. Histological Analyses
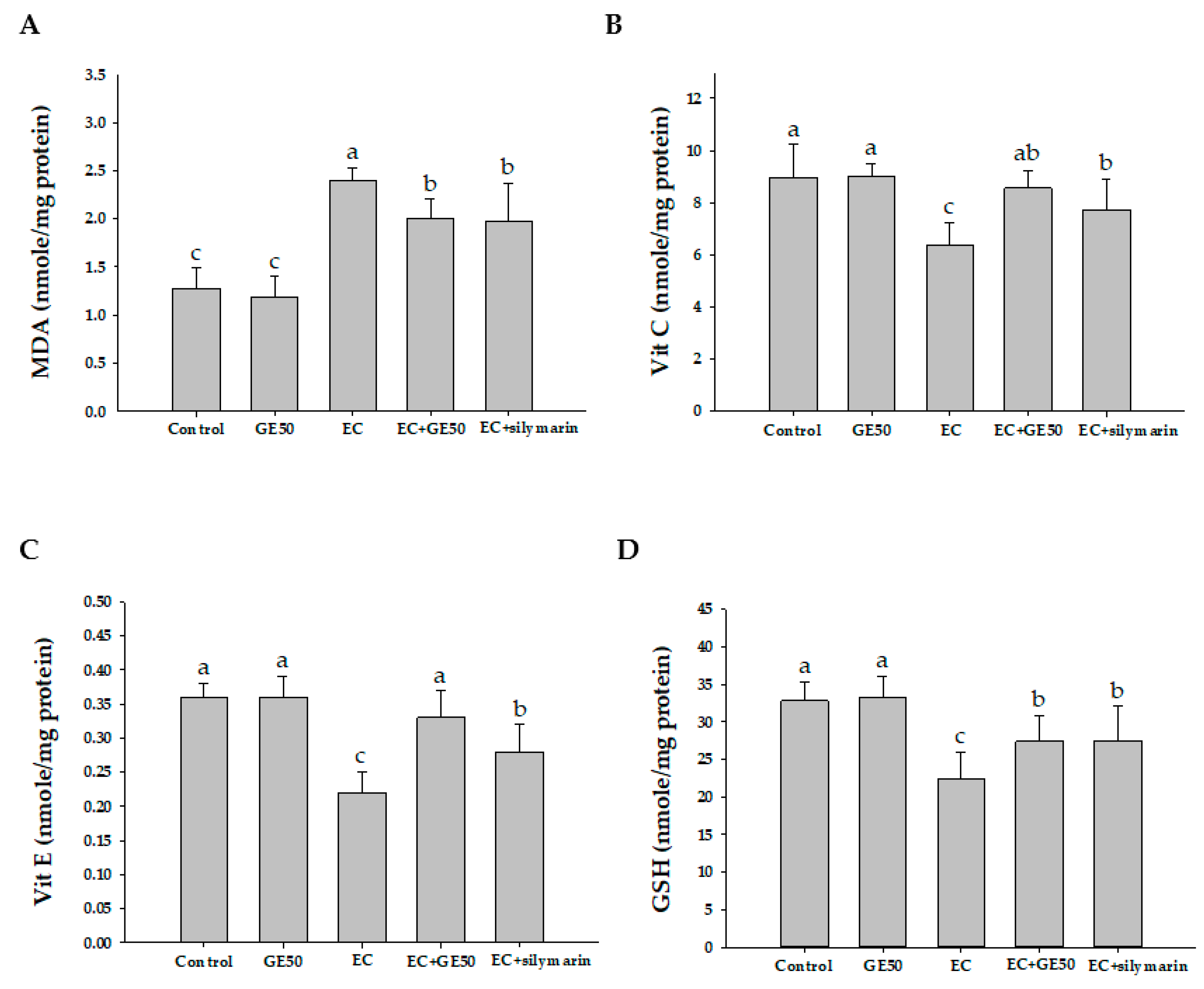
3.4. Effects of GE50 on Hepatic MDA, Vitamin C, Vitamin E, and GSH Levels in Ethanol + CCl4-Treated Group
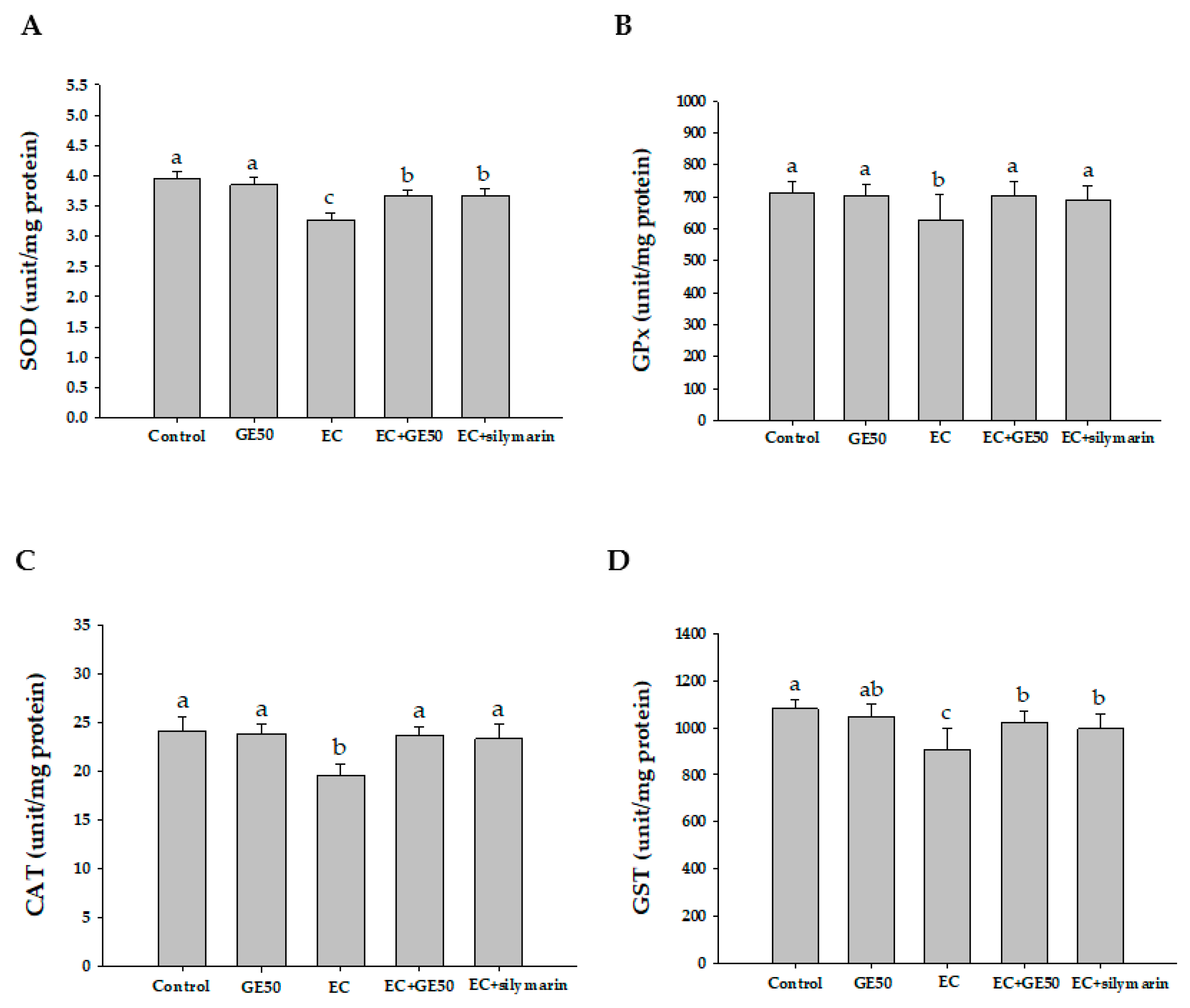
3.5. Effects of GE50 on Antioxidant Enzymatic Activities in Ethanol + CCl4-Treated Group
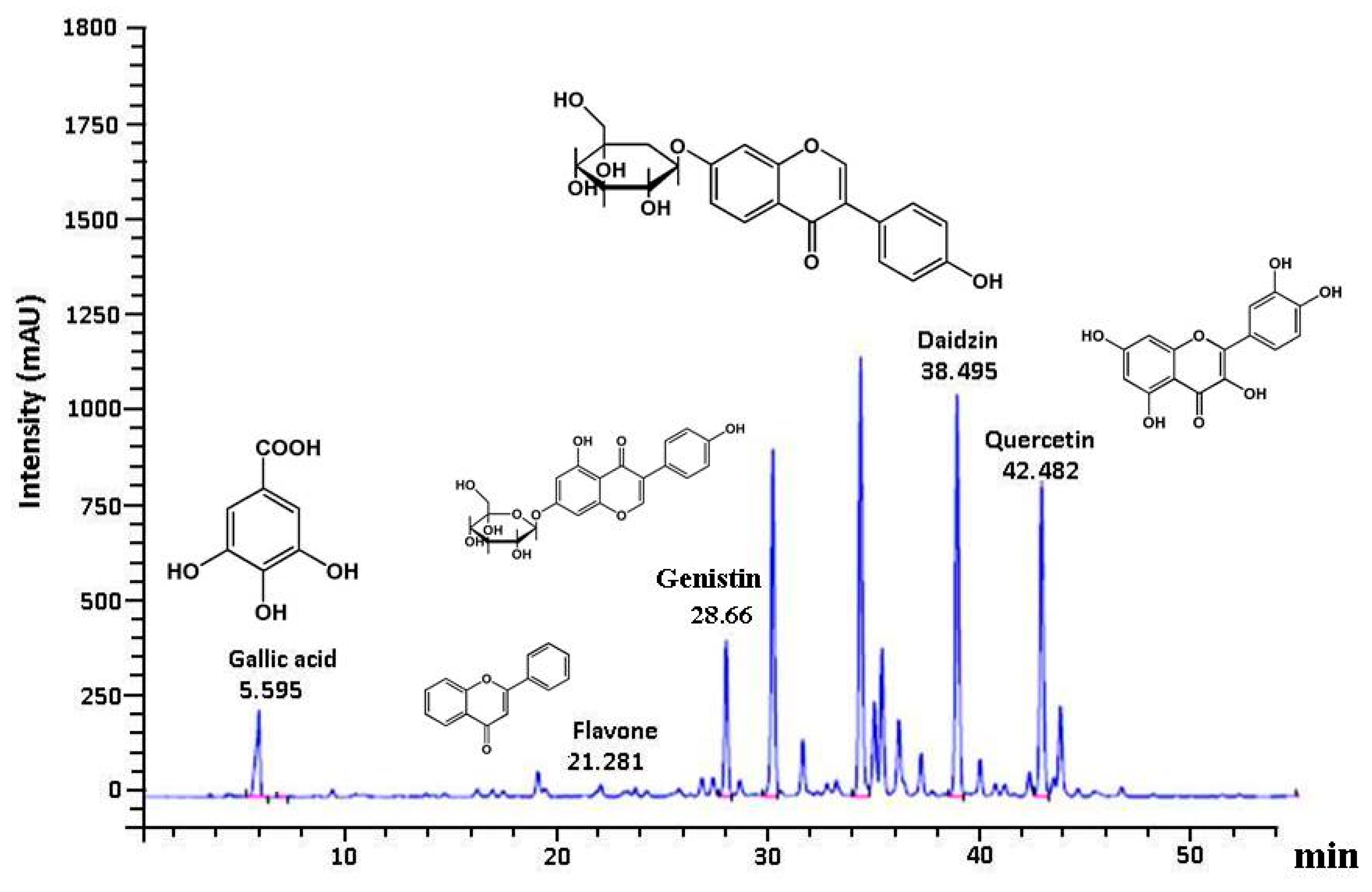
3.6. Polyphenolic Profile in GE50
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zhang, H.Y.; Wang, H.L.; Zhong, G.Y.; Zhu, J.X. Molecular mechanism and research progress on pharmacology of traditional Chinese medicine in liver injury. Pharm. Biol. 2018, 56, 594–611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingawale, D.K.; Mandlik, S.K.; Naik, S.R. Models of hepatotoxicity and the underlying cellular, biochemical and immunological mechanism(s): A critical discussion. Environ. Toxicol. Pharmacol. 2014, 37, 118–133. [Google Scholar] [CrossRef] [PubMed]
- Stoyanovsky, D.A.; Cederbaum, A.I. Thiol oxidation and cytochrome P450-dependent metabolism of CCl4 triggers Ca2+ release from liver microsomes. Biochemistry 1996, 35, 15839–15845. [Google Scholar] [CrossRef] [PubMed]
- Chao, W.W.; Lin, B.F. Hepatoprotective diterpenoids isolated from Andrographis paniculata. Chin. Med. 2012, 3, 136–143. [Google Scholar] [CrossRef]
- Wang, M.Y.; Srinivasan, M.; Dasari, S.; Narvekar, P.; Samy, A.L.P.; Dontaraju, V.S.; Peng, L.; Anderson, G.L.; Munirathinam, G. Antioxidant activity of Yichun Blue Honeysuckle (YBHS) berry counteracts CCl4-induced toxicity in liver injury model of mice. Antioxidants 2017, 6, 50. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Yang, Z.; Zhang, J.; Mu, J.; Zhou, X.; Zhao, X. Liver injury induced by carbon tetrachloride in mice is prevented by the antioxidant capacity of Anji white tea polyphenols. Antioxidants 2019, 8, 64. [Google Scholar] [CrossRef] [PubMed]
- Albano, E. Oxidative mechanisms in the pathogenesis of alcoholic liver disease. Mol. Asp. Med. 2008, 29, 9–16. [Google Scholar] [CrossRef]
- Das, M.; Basu, S.; Banerjee, B.; Sen, A.; Jana, K.; Datta, G. Hepatoprotective effects of green Capsicum annum against ethanol induced oxidative stress, inflammation and apoptosis in rats. J. Ethnopharmacol. 2018, 227, 69–81. [Google Scholar] [CrossRef]
- Choi, Y.; Abdelmegeed, M.A.; Song, B.J. Preventive effects of indole-3-carbinol against alcohol-induced liver injury in mice via antioxidant, anti-inflammatory, and anti-apoptotic mechanisms: Role of gut-liver-adipose tissue axis. J. Nutr. Biochem. 2018, 55, 12–25. [Google Scholar] [CrossRef]
- Hellerbrand, C.; Schattenberg, J.M.; Peterburs, P.; Lechner, A.; Brignoli, R. The potential of silymarin for the treatment of hepatic disorders. Clin. Phytosc. 2016, 2, 7–20. [Google Scholar] [CrossRef]
- Federico, A.; Dallio, M.; Loguercio, C. Silymarin/silybin and chronic liver disease: A marriage of many years. Molecules 2017, 22, 191. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.C.; Chen, S.J.; Hsu, C.K.; Chang, C.T.; Chou, S.T. Studies on the antioxidative activity of Graptopetalum paraguayense E. Walther. Food Chem. 2005, 91, 419–424. [Google Scholar] [CrossRef]
- Chen, S.J.; Chang, C.T.; Chung, Y.C.; Chou, S.T. Studies on the inhibitory effect of Graptopetalum paraguayense E. Walther extracts on the angiotensin converting enzyme. Food Chem. 2007, 100, 1032–1036. [Google Scholar] [CrossRef]
- Chen, S.J.; Chung, J.G.; Chung, Y.C.; Chou, S.T. In vitro antioxidant and antiproliferative activity of the stem extracts from Graptopetalum paraguayense. Am. J. Chin. Med. 2008, 36, 369–383. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.C.; Chou, S.T.; Jhan, J.K.; Liao, J.W.; Chen, S.J. In vitro and in vivo safety of aqueous extracts of Graptopetalum paraguayense E. Walther. J. Ethnopharmacol. 2012, 140, 91–97. [Google Scholar] [CrossRef]
- Liu, H.Y.; Peng, H.Y.; Hsu, S.L.; Jong, T.T.; Chou, S.T. Chemical characterization and antioxidative activity of four 3-hydroxyl-3-methylglutaroyl (HMG)-substituted flavonoid glycosides from Graptopetalum paraguayense E. Walther. Bot. Stud. 2015, 56, 8–16. [Google Scholar] [CrossRef]
- Kao, T.K.; Ou, Y.C.; Raung, S.L.; Chen, W.Y.; Yen, Y.J.; Lai, C.Y.; Chou, S.T.; Chen, C.J. Graptopetalum paraguayense E. Walther leaf extracts protect against brain injury in ischemic rats. Am. J. Chin. Med. 2010, 38, 495–516. [Google Scholar] [CrossRef]
- Lin, Y.L.; Peng, H.Y.; Hsieh, H.M.; Lin, C.H.; Chou, S.T. Effects of Graptopetalum paraguayense consumption on serum lipid profiles and antioxidative status in hypercholesteremic subjects. J. Sci. Food Agric. 2011, 91, 1230–1235. [Google Scholar]
- Duh, P.D.; Lin, S.L.; Wu, S.C. Hepatoprotection of Graptopetalum paraguayense E. Walther on CCl4-induced liver damage and inflammation. J. Ethnopharmacol. 2011, 134, 379–385. [Google Scholar] [CrossRef]
- Hsu, W.H.; Chang, C.C.; Huang, K.W.; Chen, Y.C.; Hsu, S.L.; Wu, L.C.; Tsou, A.P.; Lai, J.M.; Huang, C.Y. Evaluation of the medicinal herb Graptopetalum paraguayense as a treatment for liver cancer. PLoS ONE 2015, 10, e0121298. [Google Scholar] [CrossRef]
- Su, L.J.; Chang, C.C.; Yang, C.H.; Hsieh, S.J.; Wu, Y.C.; Lai, J.M.; Tseng, T.L.; Huang, C.Y.F.; Hsu, S.L. Graptopetalum paraguayense ameliorates chemical-induced rat hepatic fibrosis in vivo and inactivates stellate cells and kupffer cells in vitro. PLoS ONE 2013, 8, e53988. [Google Scholar] [CrossRef] [PubMed]
- Chou, S.T.; Lai, C.C.; Lai, C.P.; Chao, W.W. Chemical composition, antioxidant, anti-melanogenic and anti-inflammatory activities of Glechoma hederacea (Lamiaceae) essential oil. Ind. Crops Prod. 2018, 122, 675–685. [Google Scholar] [CrossRef]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferase: The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [PubMed]
- Mitton, K.P.; Trevithick, J.R. High-performance liquid chromatography-electrochemical detection of antioxidants in vertebrate lens: Glutathione, α-tocopherol and ascorbate. Meth. Enzymol. 1994, 233, 523–539. [Google Scholar] [PubMed]
- Chou, S.T.; Ko, L.E.; Yang, C.S. Determination of tissue α- tocopherol in senescence-accelerated mice by high-performance liquid chromatography with fluorimetric detection. Anal. Chim. Acta 2000, 419, 81–86. [Google Scholar] [CrossRef]
- Yang, C.S.; Chou, S.T.; Lin, L.; Tsai, P.J.; Kuo, J.S. Effect of ageing on human plasma glutathione concentrations as determined by high-performance liquid chromatography with fluorimetric detection. J. Chromatogr. B 1995, 674, 23–30. [Google Scholar] [CrossRef]
- Li, Q.; Jia, Y.; Xu, L.; Wang, X.; Shen, Z.; Liu, Y.; Bi, K. Simultaneous determination of protocatechuic acid, syringin, chlorogenic acid, caffeic acid, liriodendrin and isofraxidin in Acanthopanax senticosus Harms by HPLC-DAD. Biol. Pharm. Bull. 2006, 29, 532–534. [Google Scholar] [CrossRef]
- Chang, Y.Y.; Liu, Y.C.; Kuo, Y.H.; Lin, Y.L.; Wu, Y.H.S.; Chen, J.W.; Chen, Y.C. Effects of antrosterol from Antrodia camphorata submerged whole broth on lipid homeostasis, antioxidation, alcohol clearance, and antiinflammation in livers of chronic-alcohol fed mice. J. Ethnopharmacol. 2017, 202, 200–207. [Google Scholar] [CrossRef]
- Tiwary, B.K.; Dutta, S.; Dey, P.; Hossain, M.; Kumar, A.; Bihani, S.; Nanda, A.K.; Chaudhuri, T.K. Radical scavenging activities of Lagerstroemia speciosa (L.) Pers. Petal extracts and its hepatoprotection in CCl4-intoxicated mice. BMC Complement. Altern. Med. 2017, 17, 55–67. [Google Scholar] [CrossRef]
- Kim, T.W.; Lee, D.R.; Choi, B.K.; Kang, H.K.; Jung, J.Y.; Lim, S.W.; Yang, S.H.; Suh, J.W. Hepatoprotective effects of polymethoxyflavones against acute and chronic carbon tetrachloride intoxication. Food Chem. Toxicol. 2016, 91, 91–99. [Google Scholar] [CrossRef]
- Bishayee, A.; Mandal, A.; Atterjee, M. Prevention of alcohol-carbon tetrachloride-induced signs of early hepatotoxicity in mice by Trianthema portulacastrum L. Phytornedicine 1996, 3, 155–161. [Google Scholar] [CrossRef]
- McCay, P.B.; Lai, E.K.; Poyer, J.L.; DuBose, C.M.; Janzen, E.G. Oxygen- and carboncentered free radical formation during carbon tetrachloride metabolism. Observation of lipid radicals in vivo and in vitro. J. Biol. Chem. 1984, 259, 2135–2143. [Google Scholar]
- Basu, S. Carbon tetrachloride-induced lipid peroxidation: Eicosanoid formation and their regulation by antioxidant nutrients. Toxicology 2003, 189, 113–127. [Google Scholar] [CrossRef]
- Turkodgan, M.K.; Agaoglu, Z.; Yener, Z.; Sekeroglu, R.; Akkan, H.A.; Avci, M.E. The role of antioxidant vitamins (C and E), selenium and nigella sativa in the prevention of liver fibrosis and cirrhosis in rabbits: New hopes. Dtsch. Tierarztl. Wochenschr. 2001, 108, 71–73. [Google Scholar]
- Cheeseman, K.H.; Davies, M.J.; Emery, S.; Maddix, S.P.; Slater, T.F. Effects of alpha-tocopherol on carbon tetrachloride metabolism in rat liver microsomes. Free Radic. Res. Commun. 1987, 3, 325–330. [Google Scholar] [CrossRef]
- McBean, G. Cysteine, glutathione, and thiol redox balance in astrocytes. Antioxidants 2017, 6, 62. [Google Scholar] [CrossRef]
- Fraschini, F.; Demartini, G.; Esposti, D. Pharmacology of silymarin. Clin. Drug Investig. 2002, 22, 51–65. [Google Scholar] [CrossRef]
- Koch, O.; Farre, S.; De Leo, M.E.; Palozza, P.; Palazzotti, B.; Borrelo, S.; Palombini, G.; Cravero, A.; Galeotti, T. Regulation of manganese superoxide dismutase (MnSOD) in chronic experimental alcoholism: Effects of vitamin E supplemented and -deficient diets. Alcohol Alcohol. 2000, 35, 159–163. [Google Scholar] [CrossRef]
- Hayes, P.C.; Bouchier, J.A.D.; Beckett, G.J. Glutathione S-transferase in humans in heath and disease. Gut 1991, 32, 813–818. [Google Scholar] [CrossRef]
- Hiraganahalli, D.; Chandrasekaran, C.; Dethe, S.; Mundkinajeddu, D.; Pandre, M.; Balachandran, J. Hepatoprotective and antioxidant activity of standardized herbal extracts. Pharm. Mag. 2012, 8, 116–123. [Google Scholar]
- Kerimi, A.; Williamson, G. At the interface of antioxidant signaling and cellular function: Key polyphenol effects. Mol. Nutr. Food Res. 2016, 60, 1770–1788. [Google Scholar] [CrossRef]
- Sun, X.; Yamasaki, M.; Katsube, T.; Shiwaku, K. Effects of quercetin derivatives from mulberry leaves: Improved gene expression related hepatic lipid and glucose metabolism in short-term high-fat fed mice. Nutr. Res. Pract. 2015, 9, 137–143. [Google Scholar] [CrossRef] [Green Version]
- Miltonprabu, S.; Tomczyk, M.; Skalicka-Woaniak, K.; Rastrelli, L.; Daglia, M.; Nabavi, S.F.; Alavian, S.M.; Nabavi, S.M. Hepatoprotective effect of quercetin: From chemistry to medicine. Food Chem. Toxicol. 2017, 108, 365–374. [Google Scholar] [CrossRef]
- Pu, X.; Fan, W.; Yu, S.; Li, Y.; Ma, X.; Liu, L.; Ren, J.; Zhang, W. Polysaccharides from Angelica and Astragalus exert hepatoprotective effects against carbon-tetrachloride-induced intoxication in mice. Can. J. Physiol. Pharmacol. 2015, 93, 39–43. [Google Scholar] [CrossRef]
- Pareek, A.; Godavarthi, A.; Issarani, R.; Nagori, B.P. Antioxidant and hepatoprotective activity of Fagonia schweinfurthii (Hadidi) Hadidi extract in carbon tetrachloride induced hepatotoxicity in HepG2 cell line and rats. J. Ethnopharmacol. 2013, 150, 973–981. [Google Scholar] [CrossRef]
- Ao, Z.H.; Xu, Z.H.; Lu, Z.M.; Xu, H.Y.; Zhang, X.M.; Dou, W.F. Niuchangchih (Antrodia camphorata) and its potential in treating liver diseases. J. Ethnopharmacol. 2009, 121, 194–212. [Google Scholar] [CrossRef]
- Li, Z.W.; Kuang, Y.; Tang, S.N.; Li, K.; Huang, Y.; Qiao, X.; Yu, S.W.; Tzeng, Y.M.; Lo, J.Y.; Ye, M. Hepatoprotective activities of Antrodia camphorata and its triterpenoid compounds against CCl4-induced liver injury in mice. J. Ethnopharmacol. 2017, 206, 31–39. [Google Scholar] [CrossRef]
- Wu, Y.; Tian, W.J.; Gao, S.; Liao, Z.J.; Wang, G.H.; Lo, J.M.; Lin, P.H.; Zeng, D.Q.; Qiu, D.R.; Liu, X.Z.; et al. Secondary metabolites of petri-dish cultured Antrodia camphorata and their hepatoprotective activities against alcohol-induced liver injury in mice. Chin. J. Nat. Med. 2019, 17, 33–42. [Google Scholar] [CrossRef]
- Wang, J.; Tang, L.; White, J.; Fang, J. Inhibitory effect of gallic acid on CCl4-mediated liver fibrosis in mice. Cell Biochem. Biophys. 2014, 69, 21–26. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, Z.; Mo, Q.; Zhou, G.; Wang, Y. Gallic acid attenuates dimethylnitrosamine-induced liver fibrosis by alteration of Smad phosphoisoform signaling in rats. BioMed Res. Int. 2018, 2018, 1682743. [Google Scholar] [CrossRef]
- Hsu, W.H.; Liao, S.C.; Chyan, Y.J.; Huang, K.W.; Hsu, S.L.; Chen, Y.C.; Siu, M.L.; Chang, C.C.; Chung, Y.S.; Huang, C.Y.F. Graptopetalum paraguayense inhibits liver fibrosis by blocking TGF-β signaling in vivo and in vitro. Int. J. Mol. Sci. 2019, 20, 2592. [Google Scholar] [CrossRef]
- Chiang, C.M.; Wang, D.S.; Chang, T.S. Improving free radical scavenging activity of soy isoflavone glycosides daidzin and genistin by 30-hydroxylation using recombinant Escherichia coli. Molecules 2016, 21, 1723. [Google Scholar] [CrossRef]
- Zhao, L.; Wang, Y.; Liu, J.; Wang, K.; Guo, X.X.; Ji, B.; Wu, W.; Zhou, F. Protective effects of genistein and puerarin against chronic alcohol-induced liver injury in mice via antioxidant, anti-inflammatory, and anti-apoptotic mechanisms. J. Agric. Food Chem. 2016, 64, 7291–7297. [Google Scholar] [CrossRef]
- Qiu, P.; Dong, Y.; Zhu, T.; Luo, Y.Y.; Kang, X.J.; Pang, M.X.; Li, H.Z.; Xu, H.; Gu, C.; Pan, S.H.; et al. Semen hoveniae extract ameliorates alcohol-induced chronic liver damage in rats via modulation of the abnormalities of gut-liver axis. Phytomedicine 2019, 52, 40–50. [Google Scholar] [CrossRef]
- Surapaneni, K.M.; Jainu, M. Comparative effect of pioglitazone, quercetin and hydroxy citric acid on the status of lipid peroxidation and antioxidants in experimental non-alcoholic steatohepatitis. J. Physiol. Pharmacol. 2014, 65, 67–74. [Google Scholar]
- Kemelo, M.K.; Pierzynova, A.; Canova, N.K.; Kucera, T.; Farghali, H. The involvement of sirtuin 1 and heme oxygenase 1 in the hepatoprotective effects of quercetin against carbon tetrachloride-induced sub-chronic liver toxicity in rats. Chem. Biol. Interact. 2017, 269, 1–8. [Google Scholar] [CrossRef]
- Ma, J.Q.; Li, Z.; Xie, W.R.; Liu, C.M.; Liu, S.S. Quercetin protectsmouse liver against CCl4-induced inflammation by the TLR2/4 and MAPK/NF-κB pathway. Int. Immunopharmacol. 2015, 28, 531–539. [Google Scholar] [CrossRef]
- Zakarua, Z.A.; Mahmood, N.D.; Omar, M.H.; Taher, M.; Basir, R. Methanol extract of Muntingia calabura leaves attenuates CCl4-induced liver injury: Possible synergistic action of flavonoids and volatile bioactive compounds on endogenous defence system. Pharm. Biol. 2019, 57, 335–344. [Google Scholar] [CrossRef]
| Groups | Daily Body Weight Gain (g/Day/Rat) | Feed Efficiency (g Gain/g Feed) | Relative Liver Weight (g/100 g Body Weight) |
|---|---|---|---|
| Control | 5.58 ± 0.66 a | 0.21 ± 0.02 a | 2.96 ± 0.14 c |
| GE50 | 5.92 ± 0.81 a | 0.21 ± 0.02 ab | 3.05 ± 0.19 bc |
| Ethanol + CCl4 (EC) | 3.56 ± 0.66 c | 0.15 ± 0.01 c | 3.30 ± 0.11 a |
| EC + GE50 | 4.68 ± 0.59 b | 0.20 ± 0.01 b | 3.10 ± 0.12 b |
| EC + silymarin | 4.32 ± 0.74 b | 0.20 ± 0.02 ab | 3.17 ± 0.11 b |
| Groups | AST (U/L) | ALT (U/L) | LDH (U/L) | TAS (nmole/L) |
|---|---|---|---|---|
| Control | 103.98 ± 14.0 c | 44.10 ± 5.4 c | 273.82 ± 94.9 c | 0.39 ± 0.1 a |
| GE50 | 97.25 ± 19.8 c | 44.28 ± 3.8 c | 218.39 ± 35.2 c | 0.37 ± 0.1 a |
| Ethanol + CCl4 (EC) | 184.40 ± 25.5 a | 66.89 ± 4.9 a | 563.04 ± 103.7 a | 0.15 ± 0.1 c |
| EC + GE50 | 110.95 ± 14.0 bc | 50.01 ± 4.2 b | 403.20 ± 79.4 b | 0.28 ± 0.1 b |
| EC + silymarin | 122.28 ± 24.2 b | 42.05 ± 6.6 c | 347.13 ± 43.9 b | 0.26 ± 0.1 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chao, W.-W.; Chen, S.-J.; Peng, H.-C.; Liao, J.-W.; Chou, S.-T. Antioxidant Activity of Graptopetalum paraguayense E. Walther Leaf Extract Counteracts Oxidative Stress Induced by Ethanol and Carbon Tetrachloride Co-Induced Hepatotoxicity in Rats. Antioxidants 2019, 8, 251. https://doi.org/10.3390/antiox8080251
Chao W-W, Chen S-J, Peng H-C, Liao J-W, Chou S-T. Antioxidant Activity of Graptopetalum paraguayense E. Walther Leaf Extract Counteracts Oxidative Stress Induced by Ethanol and Carbon Tetrachloride Co-Induced Hepatotoxicity in Rats. Antioxidants. 2019; 8(8):251. https://doi.org/10.3390/antiox8080251
Chicago/Turabian StyleChao, Wen-Wan, Shu-Ju Chen, Hui-Chen Peng, Jiunn-Wang Liao, and Su-Tze Chou. 2019. "Antioxidant Activity of Graptopetalum paraguayense E. Walther Leaf Extract Counteracts Oxidative Stress Induced by Ethanol and Carbon Tetrachloride Co-Induced Hepatotoxicity in Rats" Antioxidants 8, no. 8: 251. https://doi.org/10.3390/antiox8080251





