Profiling of Polyphenol Composition and Antiradical Capacity of Erica cinerea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Extraction
2.3. Polyphenol Composition by HPLC-DAD/UV-Vis
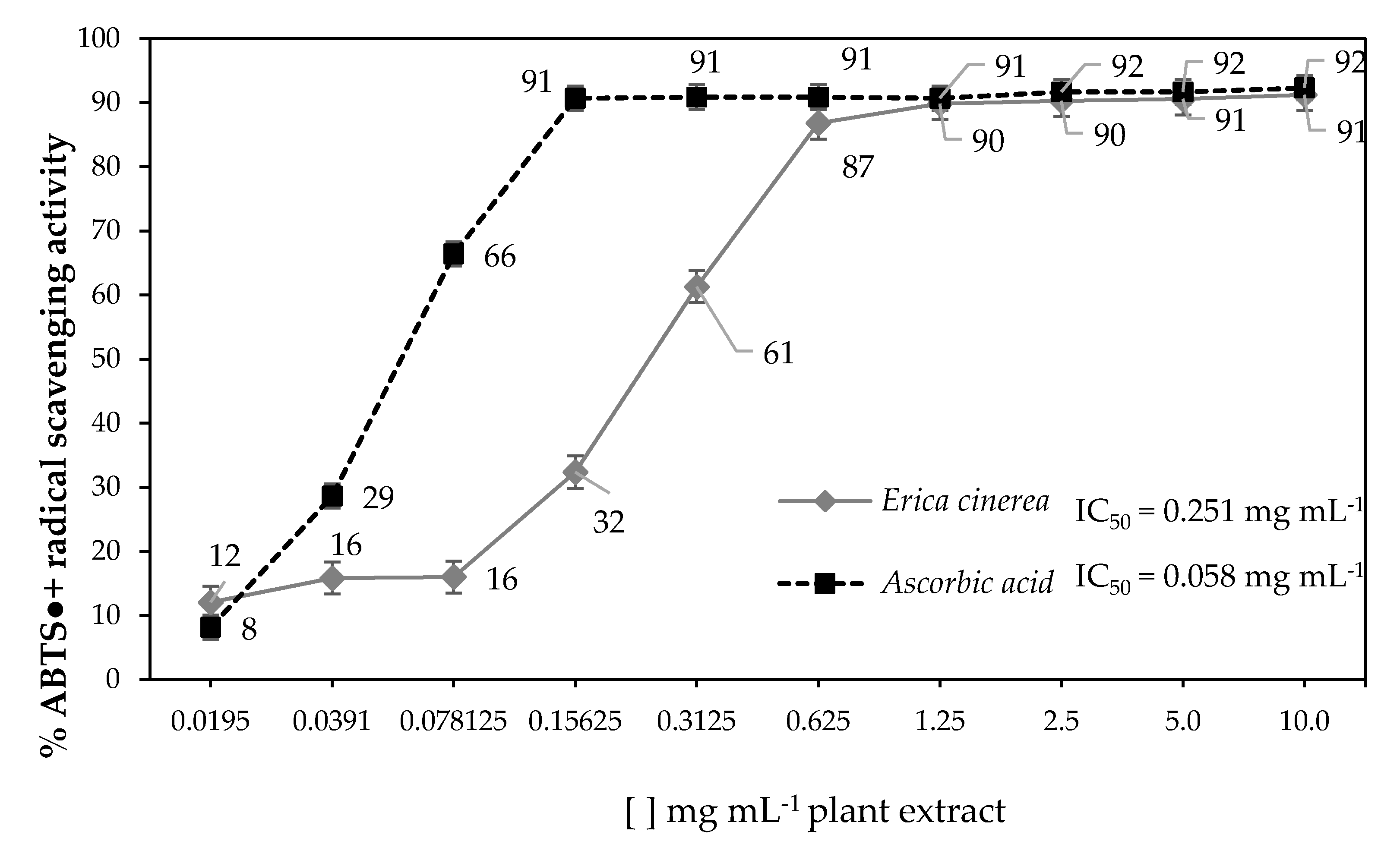
2.4. ABTS (2,2′-Azinobis-(3-Ethylbenzothiazoline-6-Sulfonic Acid)) Radical Scavenging Activity
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Akkol, E.K.; Yeşilada, E.; Güvenç, A. Valuation of anti-inflammatory and antinociceptive activities of Erica species native to Turkey. J. Ethnopharmacol. 2008, 116, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Nunes, R.; Anastácio, A.; Carvalho, I.S. Antioxidant and free radical scavenging activities of different plant parts from two Erica Species. J. Food Qual. 2012, 35, 307–314. [Google Scholar] [CrossRef]
- Luís, Â.; Domingues, F.; Duarte, A.P. Bioactive compounds, RP-HPLC analysis of polyphenols, and antioxidant activity of some Portuguese shrub species extracts. Nat. Prod. Commun. 2011, 6, 1863–1872. Available online: https://www.researchgate.net/publication/221813046 (accessed on 10 January 2017). [PubMed]
- Maleš, Ž.; Šarić, D.; Bojić, M. Quantitative determination of flavonoids and chlorogenic acid in the leaves of Arbutus unedo L. Using thin layer chromatography. J. Anal. Methods Chem. 2013, 2013, 385473. [Google Scholar] [CrossRef] [PubMed]
- De Jesus, N.Z.T.; de Souza Falcão, H.; Gomes, I.F.; de Almeida Leite, T.J.; de Morais Lima, G.R.; Barbosa-Filho, J.M.; Batista, L.M. Tannins, Peptic Ulcers and Related Mechanisms. Int. J. Mol. Sci. 2012, 13, 3203–3228. [Google Scholar] [CrossRef] [PubMed]
- Nunes, R.; Carvalho, I.S. Antioxidant activities, distribution of polyphenols and free amino acids of Erica australis L. leaves and flowers collected in Algarve, Portugal. Nat. Prod. Res. 2013, 27, 1664–1667. [Google Scholar] [CrossRef] [PubMed]
- Nunes, R.; Rodrigues, S.; Pasko, P.; Tyszka-Czochara, M.; Grenha, A.; de Carvalho, I.S. Effect of Erica australis extract on Caco-2 cells, fibroblasts and selected pathogenic bacteria responsible for wound infection. Ind. Crop. Prod. 2014, 52, 99–104. [Google Scholar] [CrossRef]
- Aires, A.; Carvalho, R.; Rosa, E.A.S.; Saavedra, M.J. Phytochemical characterization and antioxidant properties of organic baby-leaf watercress produced under organic production system. CyTA-J. Food 2013, 11, 343–351. [Google Scholar] [CrossRef]
- Sakakibara, H.; Honda, Y.; Nakagawa, S.; Ashida, H.; Kanazawa, K. Simultaneously determination of all polyphenols in vegetables, fruits and teas. J. Agric. Food Chem. 2003, 51, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.-Z.; Harnly, J.M. A screening method for the identification of glycosylated flavonoids and other phenolic compounds using standard analytical approach for all plant materials. J. Agric. Food Chem. 2007, 55, 1084–1096. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Amezouar, F.; Badri, W.; Hsaine, M.; Bourhim, N.; Fougrach, H. Antioxidant and anti-inflammatory activities of Moroccan Erica arborea L. Pathol. Biol. 2013, 61, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.-R.; Qiao, W.-L.; Liu, Z.-Z.; Wang, X.-H.; Jiang, R.; Li, S.-Y.; Shi, R.-B.; She, G.-M. Gaultheria: Phytochemical and Pharmacological Characteristics. Molecules 2013, 18, 12071–12108. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, G.; Kamiloglu, S.; Ozdal, T.; Boyacioglu, D.; Capanoglu, E. Potential use of Turkish medicinal plants in the treatment of various diseases. Molecules 2016, 21, 257. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Lia, H.; Sun, J.; Wang, S. Protective effects of hydroxycinnamic acid and cinnamic aldehyde on isoproterenol-induced acute myocardial ischemia in rats. J. Ethnopharmacol. 2013, 150, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Z.; Mi, S.; Zhou, L.; Zhou, Y.; Li, J.; Liu, W.; Deng, Z.; Yin, Y. Chlorogenic acid enhances intestinal barrier by decreasing MLCK expression and promoting dynamic distribution of tight junction proteins in colitic rats. J. Funct. Foods 2016, 26, 698–708. [Google Scholar] [CrossRef]
- Oboh, G.; Agunloye, O.M.; Akinyemi, A.J.; Ademiluyi, A.O.; Adefegha, S.A. Comparative study on the inhibitory effect of caffeic and chlorogenic acids on key enzymes linked to Alzheimer’s disease and some pro-oxidant induced oxidative stress in rats’ brain-in vitro. Neurochem. Res. 2013, 38, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, B.; Li, H.; Lu, H.; Qiu, F.; Xiong, L.; Xu, Y.; Wang, G.; Liu, X.; Wu, H.; et al. Quercetin, a flavonoid with anti-inflammatory activity, suppresses the development of abdominal aortic aneurysms in mice. Eur. J. Pharmacol. 2012, 690, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.; Chen, L.; Ma, X.; Jiao, R.; Li, X.; Wang, Y. Protective effects of kaempferol against reactive oxygen species-induced hemolysis and its antiproliferative activity on human cancer cells. Eur. J. Med. Chem. 2016, 114, 24–32. [Google Scholar] [CrossRef] [PubMed]
| Polyphenols | Rt (min) | UV Detection (nm) | UV λmax (nm) | Quantity (µg g−1 dw) | Percentage in the Total (%) |
|---|---|---|---|---|---|
| trans-Cinnamic acid | 15.05 | 320 | 274,318 | 5.0 ± 0.1 | 0.3 |
| Neochlorogenic acid | 15.94 | 320 | 308,322 | 104.4 ± 0.2 | 6.3 |
| Epigallocatechin | 16.55 | 280 | 279 | 21.7 ± 0.9 | 1.3 |
| 5-O-Caffeoylquinic acid | 17.58 | 320 | 284,311 | 10.0 ± 0.9 | 0.6 |
| Chlorogenic acid | 17.86 | 320 | 303,322 | 240.7 ± 1.1 | 14.6 |
| Catechin | 18.00 | 320 | 279 | 18.5 ± 0.1 | 1.1 |
| Epicatechin | 18.36 | 280 | 280 | 29.8 ± 0.4 | 1.8 |
| Epigallocatechin gallate | 18.55 | 280 | 279 | 18.8 ± 3.3 | 1.1 |
| Gallocatechin gallate | 18.75 | 280 | 279 | 8.0 ± 0.6 | 0.5 |
| Myricetin-3-O-rutinoside | 19.04 | 370 | 275,339,380 | 28.6 ± 1.0 | 1.7 |
| Cyanidin-3-O-rutinoside | 19.18 | 520 | 237,279,517 | 14.0 ± 0.2 | 0.9 |
| Caffeic acid | 19.31 | 320 | 280,324 | 34.3 ± 1.5 | 2.1 |
| Epicatechin gallate | 19.51 | 280 | 279 | 36.7 ± 0.0 | 2.2 |
| Cyanidin-3-O-arabinoside | 20.39 | 520 | 237,279,518 | 1.9 ± 0.1 | 0.1 |
| Kaempferol-3-O-galactoside | 20.59 | 370 | 266,355 | 25.6 ± 0.9 | 1.6 |
| Myricetin-3-O-glucoside | 20.74 | 370 | 271,354 | 7.7 ± 0.4 | 0.5 |
| Myricetin-3-O-galactoside | 21.17 | 370 | 273,358 | 9.9 ± 0.3 | 0.6 |
| Quercetin-3-O-galactoside | 21.34 | 370 | 254,358 | 104.2 ± 0.2 | 6.3 |
| Gossypetin | 21.63 | 370 | 273,356 | 19.8 ± 0.3 | 1.2 |
| Hesperidin | 21.85 | 280 | 285,336 | 26.0 ± 0.3 | 1.6 |
| Luteolin-3-O-rutinoside | 22.02 | 370 | 253,350 | 182.0 ± 0.1 | 11.1 |
| Luteolin-3-O-glucoside | 22.15 | 370 | 254,351 | 57.7 ± 1.6 | 3.5 |
| Quercetin-3-O-glucoside | 22.60 | 370 | 256,359 | 92.2 ± 3.0 | 5.6 |
| Kaempferol-3-O-glucoside | 23.03 | 370 | 254,355 | 73.4 ± 0.9 | 4.5 |
| Petunidin | 23.19 | 520 | 278,530 | 37.4 ± 0.1 | 2.3 |
| Luteolin-3-O-galactoside | 23.31 | 370 | 269,346 | 163.7 ± 0.3 | 9.9 |
| Rosmarinic acid | 23.55 | 320 | 282,328 | 26.2 ± 0.4 | 1.6 |
| Apigenin-7-O-rutinoside | 24.13 | 370 | 265,333 | 145.7 ± 1.1 | 8.9 |
| Isorhamnetin-3-O-rutinoside | 24.92 | 370 | 265,342 | 57.1 ± 3.2 | 3.5 |
| Quercetin-3-O-rhamnoside | 28.02 | 370 | 255,369 | 45.2 ± 3.7 | 2.7 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aires, A.; Carvalho, R. Profiling of Polyphenol Composition and Antiradical Capacity of Erica cinerea. Antioxidants 2017, 6, 72. https://doi.org/10.3390/antiox6030072
Aires A, Carvalho R. Profiling of Polyphenol Composition and Antiradical Capacity of Erica cinerea. Antioxidants. 2017; 6(3):72. https://doi.org/10.3390/antiox6030072
Chicago/Turabian StyleAires, Alfredo, and Rosa Carvalho. 2017. "Profiling of Polyphenol Composition and Antiradical Capacity of Erica cinerea" Antioxidants 6, no. 3: 72. https://doi.org/10.3390/antiox6030072
APA StyleAires, A., & Carvalho, R. (2017). Profiling of Polyphenol Composition and Antiradical Capacity of Erica cinerea. Antioxidants, 6(3), 72. https://doi.org/10.3390/antiox6030072






