The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis
Abstract
:1. Introduction
2. Methods
2.1. Data Sources and Search Strategy
2.2. Study Selection
2.3. Inclusionary Criteria
2.4. Methodological Quality of Evaluated Studies
2.5. Data Extraction of Included Studies
2.6. Categorization of Temporal Period
2.7. Categorization of Moderators
2.8. Data Synthesis
3. Results
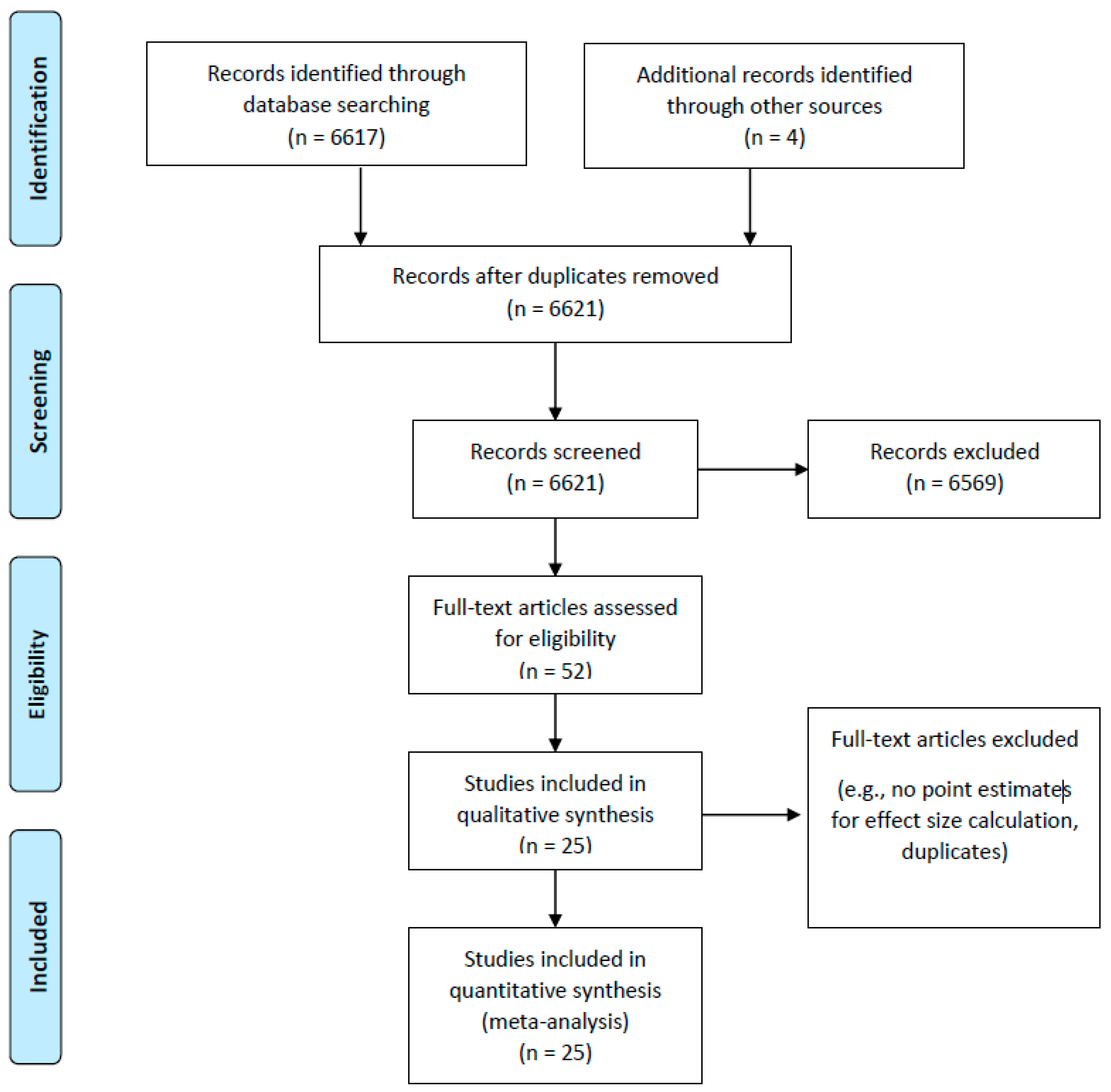
3.1. Retrieved Articles
3.2. Study Quality
3.3. Article Synthesis
3.4. Article Reference
3.5. Quantitative Analysis
4. Discussion
Funding
Conflicts of Interest
References
- Tulving, E. Elements of Episodic Memory; Oxford University Press: Oxford, UK, 1983. [Google Scholar]
- Mahr, J.; Csibra, G. Why do we remember? The communicative function of episodic memory. Behav. Brain Sci. 2017, 41, 1–93. [Google Scholar] [CrossRef]
- Frith, E.; Addoh, O.; Mann, J.R.; Windham, B.G.; Loprinzi, P.D. Individual and combined associations of cognitive and mobility limitations on mortality risk in older adults. Mayo Clin. Proc. 2017, 92, 1494–1501. [Google Scholar] [CrossRef]
- Dickerson, B.C.; Eichenbaum, H. The episodic memory system: Neurocircuitry and disorders. Neuropsychopharmacology 2010, 35, 86–104. [Google Scholar] [CrossRef] [PubMed]
- Etnier, J.L.; Wideman, L.; Labban, J.D.; Piepmeier, A.T.; Pendleton, D.M.; Dvorak, K.K.; Becofsky, K. The Effects of acute exercise on memory and brain-derived neurotrophic factor (BDNF). J. Sport Exerc. Psychol. 2016, 38, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Labban, J.D.; Etnier, J.L. Effects of acute exercise on long-term memory. Res. Q Exerc. Sport 2011, 82, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.I.; Voss, M.W.; Prakash, R.S.; Basak, C.; Szabo, A.; Chaddock, L.; Kim, J.S.; Heo, S.; Alves, H.; White, S.M.; et al. Exercise training increases size of hippocampus and improves memory. Proc. Natl. Acad. Sci. 2011, 108, 3017–3022. [Google Scholar] [CrossRef] [PubMed]
- Hillman, C.H.; Erickson, K.I.; Kramer, A.F. Be smart, exercise your heart: Exercise effects on brain and cognition. Nat. Rev. Neurosci. 2008, 9, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; Gwizdala, K.L.; Parks, A.C.; Pfeiffer, K.A.; Fenn, K.M. The Association between physical activity during the day and long-term memory stability. Sci. Rep. 2016, 6, 38148. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Kassow, M.; Zink, N.; Mock, J.; Thiel, C.; Vogt, L.; Abel, C.; Kaiser, J. Treadmill walking during vocabulary encoding improves verbal long-term memory. Behav. Brain Funct. 2014, 10, 24. [Google Scholar] [CrossRef] [PubMed]
- Schmidt-Kassow, M.; Deusser, M.; Thiel, C.; Otterbein, S.; Montag, C.; Revter, M.; Banzer, W.; Kaiser, J. Physical exercise during encoding improves vocabulary learning in young female adults: A neuroendocrinological study. PLoS ONE 2013, 8, e64172. [Google Scholar] [CrossRef] [PubMed]
- Rendeiro, C.; Rhodes, J.S. A new perspective of the hippocampus in the origin of exercise-brain interactions. Brain Struct. Funct. 2018, 223, 2527–2545. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T.; Sproule, J.; Turner, A.; Hale, B.J. Acute, intermediate intensity exercise, and speed and accuracy in working memory tasks: A meta-analytical comparison of effects. Physiol. Behav. 2011, 102, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Roig, M.; Thomas, R.; Mang, C.S.; Snow, N.J.; Ostadan, F.; Boyd, L.A.; Lundbye, J.J. Time-dependent effects of cardiovascular exercise on memory. Exerc. Sport Sci Rev. 2016, 44, 81–88. [Google Scholar] [CrossRef]
- Roig, M.; Nordbrandt, S.; Geertsen, S.S.; Nielsen, J.B. The effects of cardiovascular exercise on human memory: A review with meta-analysis. Neurosci. Biobehav. Rev. 2013, 37, 1645–1666. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.K.; Labban, J.D.; Gapin, J.I.; Etnier, J.L. The effects of acute exercise on cognitive performance: A meta-analysis. Brain Res. 2012, 1453, 87–101. [Google Scholar] [CrossRef] [PubMed]
- Heisz, J.J.; Clark, I.B.; Bonin, K.; Paolucci, M.E. The effects of physical exercise and cognitive training on memory and neurotrophic factors. J. Cogn. Neurosci. 2017, 29, 1895–1907. [Google Scholar] [CrossRef]
- Heisz, J.J.; Vandermorris, S.; Wu, J.; McIntosh, A.R.; Ryan, J.D. Age differences in the association of physical activity, sociocognitive engagement, and TV viewing on face memory. Health Psychol. 2015, 34, 83–88. [Google Scholar] [CrossRef]
- Van Dongen, E.V.; Kersten, I.H.; Wagner, I.C.; Morris, R.G.; Fernandez, G. Physical exercise performed four hours after learning improves memory retention and increases hippocampal pattern similarity during retrieval. Curr. Biol. 2016, 26, 1722–1777. [Google Scholar] [CrossRef]
- Wenderoth, N. Motor learning triggers neuroplastic processes while awake and during sleep. Exerc. Sport Sci. Rev. 2018, 46, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.B.; McGowan, A.L.; Chandler, M.C.; Gwizdala, M.C.; Gwizdala, K.L.; Parks, A.C.; Fenn, K.; Kamijo, K. A primer on investigating the after effects of acute bouts of physical activity on cognition. Psychol. Sport Exerc. 2019, 40, 1–22. [Google Scholar] [CrossRef]
- Moore, R.D.; Romine, M.W.; O’Connor, P.J.; Tomporowski, P.D. The influence of exercise-induced fatigue on cognitive function. J. Sports Sci. 2012, 30, 841–850. [Google Scholar] [CrossRef]
- Tomporowski, P.D.; Pendleton, D.M. Effects of the timing of acute exercise and movement complexity on young adults’ psychomotor learning. J. Sport Exerc. Psychol. 2018, 40, 240–248. [Google Scholar] [CrossRef]
- Coles, K.; Tomporowski, P.D. Effects of acute exercise on executive processing, short-term and long-term memory. J. Sports Sci. 2008, 26, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Tomporowski, P.D.; Ganio, M.S. Short-term effects of aerobic exercise on executive processing, memory, and emotional reactivity. Int. J. Sport Exerc. Psychol. 2006, 4, 57–72. [Google Scholar] [CrossRef]
- Tomporowski, P.D.; Ellis, N.R.; Stephens, R. The immediate effects of strenuous exercise on free-recall memory. Ergonomics 1987, 30, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Tomporowski, P.D. Effects of acute bouts of exercise on cognition. Acta Psychol. (Amst). 2003, 112, 297–324. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Edwards, M.K.; Frith, E. Potential avenues for exercise to activate episodic memory-related pathways: A narrative review. Eur. J. Neurosci. 2017, 46, 2067–2077. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Ponce, P.; Frith, E. Hypothesized mechanisms through which acute exercise influences episodic memory. Physiol. Int. 2018, 105, 285–297. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E.; Edwards, M.K.; Sng, E.; Ashpole, N. The effects of exercise on memory function among young to middle-aged adults: Systematic review and recommendations for future research. Am. J. Health Promot. 2018, 32, 691–704. [Google Scholar] [CrossRef]
- Frith, E.; Sng, E.; Loprinzi, P.D. Randomized controlled trial evaluating the temporal effects of high-intensity exercise on learning, short-term and long-term memory, and prospective memory. Eur. J. Neurosci. 2017, 46, 2557–2564. [Google Scholar] [CrossRef]
- Sng, E.; Frith, E.; Loprinzi, P.D. Temporal effects of acute walking exercise on learning and memory function. Am. J. Health Promot. 2018, 32, 1518–1525. [Google Scholar] [CrossRef]
- Haynes Iv, J.T.; Frith, E.; Sng, E.; Loprinzi, P.D. Experimental effects of acute exercise on episodic memory function: Considerations for the timing of exercise. Psychol. Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D. Intensity-specific effects of acute exercise on human memory function: Considerations for the timing of exercise and the type of memory. Health Promot. Perspect. 2018, 8, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D. An integrated model of acute exercise on memory function. Med. Hypotheses 2019, 126, 51–59. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E. A brief primer on the mediational role of BDNF in the exercise-memory link. Clin. Physiol. Funct. Imaging 2019, 39, 9–14. [Google Scholar] [CrossRef]
- Dietrich, A. Transient hypofrontality as a mechanism for the psychological effects of exercise. Psychiatry Res. 2006, 145, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Audiffren, M. The reticular-activating hypofrontality (RAH) model of acute exercise. Neurosci. Biobehav. Rev. 2011, 35, 1305–1325. [Google Scholar] [CrossRef]
- Bramer, W.M.; Rethlefsen, M.L.; Kleijnen, J.; Franco, O.H. Optimal database combinations for literature searches in systematic reviews: A prospective exploratory study. Syst. Rev. 2017, 6, 245. [Google Scholar] [CrossRef]
- Delancey, D.; Frith, E.; Sng, E.; Loprinzi, P.D. Randomized controlled trial examining the long-term memory effects of acute exercise during the memory consolidation stage of memory. J. Cogn. Enhanc. 2018, 1–6. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Frith, E. The role of sex in memory function: Considerations and recommendations in the context of exercise. J. Clin. Med. 2018, 7, 132. [Google Scholar] [CrossRef]
- Barha, C.K.; Davis, J.C.; Falck, R.S.; Nagamatsu, L.S.; Liu-Ambrose, T. Sex differences in exercise efficacy to improve cognition: A systematic review and meta-analysis of randomized controlled trials in older humans. Front. Neuroendocrinol. 2017, 46, 71–85. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Neiman, D.C.; Swain, D.P. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Potter, D.; Keeling, D. Effects of moderate exercise and circadian rhythms on human memory. J. Sport Exerc. Psychol. 2005, 27, 117–125. [Google Scholar] [CrossRef]
- Coleman, M.; Offen, K.; Markant, J. Exercise similarly facilitates men and women’s selective attention task response times but differentially affects memory task performance. Front. Psychol. 2018, 9, 1405. [Google Scholar] [CrossRef] [PubMed]
- Covassin, T.; Weiss, L.; Powell, J.; Womack, C. Effects of a maximal exercise test on neurocognitive function. Br. J. Sports Med. 2007, 41, 370–374. [Google Scholar] [CrossRef]
- Davey, C.P. Mental performance after physical activity. Aust. J. Sports Mede. 1972, 4, 25–33. [Google Scholar]
- Davey, C.P. Physical exertion and mental performance. Ergonomics 1973, 16, 595–599. [Google Scholar] [CrossRef]
- Dietrich, A.; Sparling, P.B. Endurance exercise selectively impairs prefrontal-dependent cognition. Brain Cogn. 2004, 55, 516–524. [Google Scholar] [CrossRef]
- Green, D.; Loprinzi, P.D. Experimental effects of acute exercise on prospective memory and false memory. Psychol. Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Griffin, E.W.; Mullally, S.; Foley, C.; Warmington, S.A.; O’Mara, S.M.; Kelly, A.M. Aerobic exercise improves hippocampal function and increases BDNF in the serum of young adult males. Physiol. Behav. 2011, 104, 934–941. [Google Scholar] [CrossRef]
- Hancock, S.; McNaughton, L. Effects of fatigue on ability to process visual information by experienced orienteers. Percept. Mot. Skills 1986, 62, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, M.E.; Davis, F.C.; Vantieghem, M.R.; Whalen, P.J.; Bucci, D.J. Differential effects of acute and regular physical exercise on cognition and affect. Neuroscience 2012, 215, 59–68. [Google Scholar] [CrossRef]
- Hotting, K.; Schickert, N.; Kaiser, J.; Roder, B.; Schmidt-Kassow, M. The effects of acute physical exercise on memory, peripheral BDNF, and cortisol in young adults. Neural. Plast. 2016, 2016, 6860573. [Google Scholar] [CrossRef]
- Nielson, K.A.; Wulff, L.L.; Arentsen, T.J. Muscle tension induced after learning enhances long-term narrative and visual memory in healthy older adults. Neurobiol. Learn. Mem. 2014, 109, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Segal, S.K.; Cotman, C.W.; Cahill, L.F. Exercise-induced noradrenergic activation enhances memory consolidation in both normal aging and patients with amnestic mild cognitive impairment. J. Alzheimers Dis. 2012, 32, 1011–1018. [Google Scholar] [CrossRef]
- Sjoberg, H. Physical fitness and mental performance during and after work. Ergonomics 1980, 23, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Libkuman, T.M.; Nichols-Whitehead, P.; Griffith, J.; Thomas, R. Source of arousal and memory for detail. Mem. Cognit. 1999, 27, 166–190. [Google Scholar] [CrossRef]
- Tomporowski, P.D.; Albrecht, C.; Pendleton, D.M. Effects of isometric hand-grip muscle contraction on young adults’ free recall and recognition memory. Res. Q Exerc. Sport. 2017, 88, 95–100. [Google Scholar] [CrossRef]
- Suwabe, K.; Hyodo, K.; Byun, K.; Ochi, G.; Yassa, M.A.; Soya, H. Acute moderate exercise improves mnemonic discrimination in young adults. Hippocampus 2017, 27, 229–234. [Google Scholar] [CrossRef]
- Winter, B.; Breitenstein, C.; Mooren, F.C.; Voelker, K.; Fobker, M.; Lechtermann, A.; Krueger, K.; Fromme, A.; Korsukewitz, C.; Floel, A.; et al. High impact running improves learning. Neurobiol. Learn Mem. 2007, 87, 597–609. [Google Scholar] [CrossRef]
- Stones, M.J.; Dawe, D. Acute exercise facilitates semantically cued memory in nursing home residents. J. Am. Geriatr. Soc. 1993, 41, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Schramke, C.J.; Bauer, R.M. State-dependent learning in older and younger adults. Psychol. Aging 1997, 12, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Salas, C.R.; Minakata, K.; Kelemen, W.L. Walking before study enhances free recall but not judgement-of-learning magnitude. J. Cogn. Psychol. 2011, 23, 507–513. [Google Scholar] [CrossRef]
- Nanda, B.; Balde, J.; Manjunatha, S. The acute effects of a single bout of moderate-intensity aerobic exercise on cognitive functions in healthy adult males. J. Clin. Diagn. Res. 2013, 7, 1883–1885. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, L.; Hasni, A.; Shinohara, M.; Duarte, A. A single bout of resistance exercise can enhance episodic memory performance. Acta Psychol. (Amst). 2014, 153, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Basso, J.C.; Shang, A.; Elman, M.; Karmouta, R.; Suzuki, W.A. Acute exercise improves prefrontal cortex but not hippocampal function in healthy adults. J. Int. Neuropsychol. Soc. 2015, 21, 791–801. [Google Scholar] [CrossRef]
- Loprinzi, P.D.; Kane, C.J. Exercise and cognitive function: A randomized controlled trial examining acute exercise and free-living physical activity and sedentary effects. Mayo Clin. Proc. 2015, 90, 450–460. [Google Scholar] [CrossRef]
- Bantoft, C.; Summers, M.J.; Tranent, P.J.; Palmer, M.A.; Cooley, P.D.; Pedersen, S.J. Effect of standing or walking at a workstation on cognitive function: A randomized counterbalanced trial. Hum. Factors 2016, 58, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Crush, E.A.; Loprinzi, P.D. Dose-response effects of exercise duration and recovery on cognitive functioning. Percept. Mot. Skills 2017, 124, 1164–1193. [Google Scholar] [CrossRef]
- Keyan, D.; Bryant, R.A. Brief exercise enhances intrusive memories of traumatic stimuli. Neurobiol. Learn. Mem. 2017, 141, 9–13. [Google Scholar] [CrossRef]
- Keyan, D.; Bryant, R.A. Acute physical exercise in humans enhances reconsolidation of emotional memories. Psychoneuroendocrinology 2017, 86, 144–151. [Google Scholar] [CrossRef] [PubMed]
- McNerney, M.W.; Radvansky, G.A. Mind racing: The influence of exercise on long-term memory consolidation. Memory 2015, 23, 1140–1151. [Google Scholar] [CrossRef] [PubMed]
- Most, S.B.; Kennedy, B.L.; Petras, E.A. Evidence for improved memory from 5 minutes of immediate, post-encoding exercise among women. Cogn. Res. Princ. Implic. 2017, 2, 33. [Google Scholar] [CrossRef] [PubMed]
- Labban, J.D.; Etnier, J.L. The effect of acute exercise on encoding and consolidation of long-term memory. J. Sport Exerc. Psychol. 2018, 40, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, A.; Loprinzi, P.D. Experimental investigation of the time course effects of acute exercise on false episodic memory. J. Clin. Med. 2018, 7, 157. [Google Scholar] [CrossRef]
- Wade, B.; Loprinzi, P.D. The experimental effects of acute exercise on long-term emotional memory. J. Clin. Med. 2018, 7, 486. [Google Scholar] [CrossRef]
- Yanes, D.; Loprinzi, P.D. Experimental effects of acute exercise on iconic memory, short-term episodic, and long-term episodic memory. J. Clin. Med. 2018, 7, 146. [Google Scholar] [CrossRef]
- Zuniga, K.E.; Mueller, M.; Santana, A.R.; Kelemen, W.L. Acute aerobic exercise improves memory across intensity and fitness levels. Memory 2018, 27, 1–9. [Google Scholar] [CrossRef]
- Winstein, C.J.; Grafton, S.T.; Pohl, P.S. Motor task difficulty and brain activity: Investigation of goal-directed reciprocal aiming using positron emission tomography. J. Neurophysiol. 1997, 77, 1581–1594. [Google Scholar] [CrossRef]
- Carey, J.R.; Bhatt, E.; Nagpal, A. Neuroplasticity promoted by task complexity. Exerc. Sport Sci. Rev. 2005, 33, 24–31. [Google Scholar]
- Gur, R.C.; Jaggi, J.L.; Ragland, J.D.; Resnick, S.M.; Shtasel, D.; Muenz, L.; Gur, E.R. Effects of memory processing on regional brain activation: Cerebral blood flow in normal subjects. Int. J. Neurosci. 1993, 72, 31–44. [Google Scholar] [CrossRef]
- Voss, M.W.; Soto, C.; Yoo, S.; Sodoma, M.; Vivar, C.; van Praag, H. Exercise and hippocampal memory systems. Trends Cogn. Sci. 2019, 23, 318–333. [Google Scholar] [CrossRef]
- Ikuta, T.; Loprinzi, P.D. Association of cardiorespiratory fitness on interhemispheric hippocampal and parahippocampal functional connectivity. Eur. J. Neur. 2019. [Google Scholar] [CrossRef]
- Ikuta, T.; Frith, E.; Ponce, P.; Loprinzi, P.D. Association of physical activity on the functional connectivity of the hippocampal-orbitofrontal pathway. Phys. Sportsmed. 2018. [Google Scholar] [CrossRef]
- Zheng, G.; Ye, B.; Zheng, Y.; Xiong, Z.; Xia, R.; Qiu, P.; Tao, J.; Chen, L. The effects of exercise on the structure of cognitive related brain regions: A meta-analysis of functional neuroimaging data. Int. J. Neurosci. 2018, 129, 406–415. [Google Scholar] [CrossRef]
- El-Sayes, J.; Harasym, D.; Turco, C.V.; Locke, M.B.; Nelson, A.J. Exercise-induced neuroplasticity: A mechanistic model and prospects for promoting plasticity. Neuroscientist 2018, 25, 1–21. [Google Scholar] [CrossRef]
| Author | Random Allocation/Counterbalance | Concealed Allocation | Baseline Comparability | Blinding of Assessors to Outcome | Between/Within Group Analysis | Reported Objective Measure of Exercise Intensity | Outcome Point Estimate | Total |
|---|---|---|---|---|---|---|---|---|
| Stones et al. (1993) [62] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Schramke et al. (1997) [63] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Labban et al. (2011) [6] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Salas et al. (2011) [64] | ✓ | ✓ | ✓ | 3 | ||||
| Nanda et al. (2013) [65] | ✓ | ✓ | 2 | |||||
| Schmidt-Kassow et al. (2014) [10] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Weinberg et al. (2014) [66] | ✓ | ✓ | ✓ | ✓ | 4 | |||
| Basso et al. (2015) [67] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Loprinzi et al. (2015) [68] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Bantoft et al. (2016) [69] | ✓ | ✓ | ✓ | ✓ | 4 | |||
| van Dongen et al. (2016) [19] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Crush et al. (2017) [70] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Frith et al. (2017) [31] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Keyan et al. (2017) [71] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Keyan et al. (2017) [72] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| McNerney et al. (2017) [73] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Most et al. (2017) [74] | ✓ | ✓ | ✓ | ✓ | 4 | |||
| Sng et al. (2017) [32] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Delancey et al. (2018) [40] | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | 6 | |
| Haynes et al. (2018) [33] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Labban et al. (2018) [75] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Siddiqui et al. (2018) [76] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Wade et al. (2018) [77] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Yanes et al. (2018) [78] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 | ||
| Zuniga et al. (2018) [79] | ✓ | ✓ | ✓ | ✓ | ✓ | 5 |
| Author | Sample | Study Design | Exercise Temporality | Exercise Protocol | Memory Assessment | Results |
|---|---|---|---|---|---|---|
| Stones et al. (1993) [62] | 20 older adults, Mage = 84.5 | Experimental; between-subject | Memory battered occurred before exercise, immediately after exercise and then 30-min post-exercise | 15-min exercises, which occurred while sitting in a chair (e.g., stretching, low-intensity aerobic activity, slow rhythmical movement) | Word fluency | Exercise was associated with greater semantically cued memory (p < 0.01). |
| Schramke et al. (1997) [63] | Two age groups, each including 48 adults. Younger group, 18–38 year. Older group, 60–80 year. | Experimental; within-subject | Exercise occurred either at rest or during encoding, and similarly, either during retrieval or not. | 5–7 minutes of walking in a long internal corridor. | CVLT; California verbal learning test | There was no difference in learning that was due to initial exercise condition, but both age groups showed greater recall when state was congruent before learning and delayed recall. |
| Labban et al. (2011) [6] | 48 young adults (Mage = 22.0) | Experimental; between-subject | Exercise occurred before and after encoding | 30-min of cycle ergometer exercise, with 20-min at moderate-intensity | Paragraph recall, with participants listening to two paragraphs and then recalling as much information as possible from the paragraphs | Exercise occurring prior to the memory task was effective in enhancing memory (p < 0.05). |
| Salas et al. (2011) [64] | 80 college undergraduate students (46 women). Mage = 19.3, SD = 2.3 | Experimental; between-subject factorial design. A 2 (encoding condition: walking vs. sitting) × 2 (retrieval condition: walking vs. sitting). | Exercise occurred either at rest or during encoding, and similarly, either during retrieval or not. | 10 minutes of walking outside at a brisk pace | Word-list memory task (10 nouns presented sequentially for 6 s each) | Students who walked before encoding had significantly higher recall (M = 0.45, SD = 0.17) compared to students who sat before encoding (M = 0.36, SD = 0.15), F(1,76) = 6.34, ƞ2p = 0.08. |
| Nanda et al. (2013) [65] | 10 healthy adult male medical students. Mage = 19.5, SD = 0.9 | Quasi-experimental; within-subject | Exercise occurred between pre- and post- memory assessments. | Cycle ergometer exercise for 30-min at moderate-intensity of 70% of heart rate reserve | Spatial span and paired associates memory task | Spatial span did not increase from pre- to post, but paired associates was significantly higher after the exercise bout. |
| Schmidt-Kassow et al. (2014) [10] | 49 right-handed German young adults (18–30 year) | Experimental; within-subject | Exercised during encoding | Self-selected walking pace during memory encoding | 40-item (Polish) word list. | Experiment 1: words recalled during walking was higher than non-walking (5.5, SD = 3.3; vs. 4.8, SD = 4.2), F = 6.98, p = 0.02, ƞ2p = 0.31. Experiment 2: words recalled during walking was higher than non-walking (5.3, SD = 4.6; vs. 4.1, SD = 3.5), F = 6.44, p = 0.02, ƞ2p = 0.19. |
| Weinberg et al. (2014) [66] | 23 participants (Mage = 20.6 year) in the exercise group and 23 (Mage = 20.2 year) in the control group. | Experimental; between-subject | Exercised during early consolidation | Isokinetic dynamometer knee extension exercise. Session consisted of submaximal voluntary dynamic contractions for a warm-up, maximal voluntary isometric contractions, and 6 sets of 10 repetitions of maximal voluntary knee extension contractions. Both legs were exercised. In the control (passive) group, the experimenter passively moved the participant leg between extension and flexion. | 180 images from the IAPS. Follow-up memory recall assessment took place 48-h later. The retrieval task included 90 studied images and 90 new images. Participants were instructed to indicate “remember”, “familiar”, or “new” after seeing each image. | There was no valence × group interaction effect. There was a main effect for valence in that participants remembered more positive and negative images than neutral images. |
| Basso et al. (2015) [67] | 85 young adults, Mage = 22.1 | Experimental; between-subject | Memory tasks occurred before exercise and at various time-points after exercise (30–120 min) | 50-min of vigorous-intensity exercise on cycle ergometer | Hopkins verbal learning test revised, modified Benton visual retention test, Digit span | Acute exercise improved prefrontal-cortex, but not hippocampal-dependent memory function. |
| Loprinzi et al. (2015) [68] | 87 young adults, Mage = 21.4 year | Experimental; between-subject | Exercise before memory task | Light, moderate, and vigorous exercise | Spatial span and paired associates | Acute exercise was not associated with either memory outcome. |
| Bantoft et al. (2016) [69] | 45 undergraduate students, Mage = 22.6 year (6.2) | Experimental; within-subject | Sitting, standing or walking during memory task | Low-intensity walking | Digit span | There were no differences in memory performance across the three conditions. |
| van Dongen et al. (2016) [19] | 72 young adults, approximately 22 years | Experimental; between-subject | Exercise immediately after encoding and 4 hours after encoding | 35 min of intermittent high-intensity exercise on cycle ergometer | Paired associates learning task | Exercising 4 hours after memory encoding was advantageous in improving memory function. |
| Crush et al. (2017) [70] | 352 participants, mean age approximately 21 years | Experimental; between-subject | Exercise occurring before memory assessment | 16 total groups, with groups ranging from 10 min of exercise to 60 min of exercise, including resting periods of either 5, 15, or 30 min | Spatial span | Shorter exercise recovery periods had a greater effect on memory performance. |
| Frith et al. (2017) [31] | 88 participants (22 per group), approximate age = 21 years. | Experimental; between-subject | Exercise occurring before, during, and after memory encoding | 15-min treadmill bout of progressive high-intensity aerobic exercise | RAVLT | High-intensity exercise prior to memory encoding was effective in enhancing long-term memory, for both 20-min delay (F = 3.36, p = 0.02, ƞ2p = 0.11) and 24-h delay (F =2.80, p = 0.04, ƞ2p = 0.09). |
| Keyan et al. (2017) [71] | 49 undergraduates between 18–29 years | Experimental; between-subject | Exercise occurred during the early memory consolidation period | Stepping exercise for 10-min on a 15 cm stepper, with a goal of exercising at 50%–85% of max. | Viewed a film depicting a car accident. Involves 10 min of live footage depicting emergency workers attending the scene of a motor vehicle accident. | Exercise (vs. control) did not induce more recall of central (t = 0.11, p > 0.05) or peripheral (t = 0.42, p > 0.05) details of the accident film. However, those that exercise recalled more intrusive memories of the car accident (t = 2.36, p = 0.02, d = 0.68). |
| Keyan et al. (2017) [72] | 54 healthy undergraduate students, Mage = 19.5 (3.0) | Experimental; between-subject | During a memory reconsolidation paradigm, participants either exercised or did not exercise after memory reactivation | 20–25 min of incremental cycling | Trauma film depicting the aftermath of a highway car crash | The exercise with reactivation condition recalled more central details of the trauma film. |
| McNerney et al. (2017) [73] | Experiment 1: 136 young adults, Mage = 19.2 (1.2) Experiment 2: 132 young adults, Mage = 19.1 (1.2) | Experimental; between-subject | Exercise occurring before and after memory encoding | 2-min of sprints | Paired associate learning, procedural learning, and text memory | Improvements in procedural and situation model memory occurred, regardless of whether exercise occurred before or after memory encoding. |
| Most et al. (2017) [74] | Experiment 1: 82 undergraduate psychology students (Mage = 19.9). Experiment 2: 83 undergraduate psychology students (Mage = 19.9). Experiment 3: 48 undergraduate psychology students (Mage = 19.2). Experiment 4: 75 undergraduate psychology students (Mage = 21.1). | Experimental; between-subject | Exercise occurring after memory encoding | 5-min of step exercise | Paired faces and names. | Acute exercise in the early consolidation period enhanced memory. |
| Sng et al. (2017) [32] | 88 participants, approximately 21–25 years (mean for each group) | Experimental; between-subject | Exercise occurred before, during and immediately after memory encoding | 15-min moderate intensity brisk walking (self-selected) | RAVLT | Exercising before memory encoding was superior for enhancing learning (p = 0.05), 24-h memory recognition (p = 0.05) and 24-h memory attribution (p = 0.006). |
| Delancey et al. (2018) [40] | 40 participants, approximately 20 years of age | Experimental; between-subject | Exercise occurring 4 hours after memory encoding | High-intensity bout of exercise for 15 minutes | RAVLT | Those who exercise during the consolidation period have a greater 24-h follow-up memory attribution (p = 0.04). |
| Haynes et al. (2018) [33] | 24 participants (Mage = 20.9; SD = 1.9), with 66.7% being female. | Experimental; within-subject | Exercise occurring before, during, and after memory encoding | Self-selected brisk walking pace for 15-min | RAVLT | Short-term memory was greater in the visit that involved exercise prior to the memory task (F= 3.76, p = 0.01, ƞ2p = 0.79). Similar results occurred for long-term memory, but there were no exercise effects on prospective memory. |
| Labban et al. (2018) [75] | 15 Participants; Mage = 22.7, SD = 3.1 | Experimental; within-subject | Exercise occurring both before and after memory encoding. | 30-min of moderate intensity cycling | RAVLT | Exercise that occurred before encoding (vs. control) was advantageous in enhancing long-term memory, including both 60-min delayed memory (p = 0.03) and 24-h delayed recall (p = 0.03). |
| Siddiqui et al. (2018) [76] | 20 participants (60% male). Mage = 21.1; SD = 1.0 | Experimental; within-subject | Exercise occurring both before and during memory encoding. | 20-min treadmill walk at a self-selected brisk walking pace | The Deese-Roediger-McDermott (DRM) paradigm. Included a 15-item word list. | For both short-term and long-term memory, the visit the involved exercise before the memory task resulted in the greatest memory performance (F = 11.56, p < 0.001, ƞ2p = 0.38) |
| Wade et al. (2018) [77] | 34 female participants; Mage = 20.5 (1.2) in the exercise group and 20.8 (1.8) in the control group. | Experimental; between-subject | Exercise occurred before memory encoding | 15-min treadmill walk at a self-selected brisk walking pace | Emotional memory assessment using images from the IAPS (International Affective Picture System). | There were no statistically significant group differences across any of the assessment periods (i.e., 1-day, 7-day, and 14-day follow-up assessments). |
| Yanes et al. (2018) [78] | 40 participants, Mage = 21.0 | Experimental; between-subject | Exercise occurred before memory encoding | 15-min treadmill walk at a self-selected brisk walking pace | 6-paragraph passage for memory recall | Exercise before encoding had greater scores on the short-term and long-term memory assessments, but this did not reach statistical significance (F = 1.0, p = 0.32, ƞ2p = 0.03). |
| Zuniga et al. (2018) [79] | Experiment 1 (N = 30), Mage = 20.4 (1.8); Experiment 2 (N = 57), Mage = 20.6 (4.1) in low-fit group and Mage = 19.4 (1.6) in high-fit group. | Experimental; within-subject | Exercise occurred before memory encoding | 3-min warm-up period on the treadmill, followed by 10-min of walking at either light or moderate-intensity. | Three lists of 30 concrete English nouns from the MRC Psycholinguistic database. | Both light-intensity (t = 2.79, p = 0.01) and moderate-intensity (t = 3.02, p = 0.006) recalled more words than the sedentary condition. Results were similar when comparing high-fit to low-fit individuals. |
| Moderator | Exercise Before Memory Encoding vs. Control | ||||
|---|---|---|---|---|---|
| Reference | Number of Effect Size Contributions | Effect Size (Cohen’s d) | Lower CI | Upper CI | |
| Age | |||||
| Young Adult | [6,31,32,33,63,64,65,68,70,71,72,73,74,75,76,77,78,79] | 66 | 0.18 * | 0.06 * | 0.29 * |
| Older Adults | [62,63] | 9 | −0.53 * | −0.88 * | −0.18 * |
| Sex | |||||
| Male | [65] | 2 | 0.32 | −0.41 | 1.05 |
| Female | [77] | 3 | −0.14 | −0.75 | 0.46 |
| Mixed | [6,31,32,33,64,68,71,72,73,74,75,76,78,79] | 42 | 0.28 * | 0.14 * | 0.43 * |
| Predominately Female | [62,70,79] | 22 | −0.06 | −0.27 | 0.15 |
| Race-Ethnicity | |||||
| Predominately white | [33,68,76,78,79] | 17 | 0.26 * | 0.02 * | 0.50 * |
| Mixed | [6,31,32,70,75,77,79] | 31 | 0.10 | -0.08 | 0.29 |
| Memory Type | |||||
| Short-term | [31,32,33,65,70,73,74,76] | 23 | −0.01 | −0.22 | 0.21 |
| Long-term | [6,31,32,33,62,63,64,71,72,73,75,76,77,78,79] | 46 | 0.19 * | 0.03 * | 0.34 * |
| Exercise Intensity | |||||
| Light | [62,63,68,79] | 17 | −0.20 | −0.45 | 0.04 |
| Moderate | [6,32,33,64,68,70,73,74,75,76,77,78,79] | 45 | 0.14 | −0.01 | 0.28 |
| Vigorous | [31,65,68,71] | 9 | 0.54 * | 0.19 * | 0.89 * |
| Exercise Duration | |||||
| Short | [31,32,33,62,63,64,70,71,73,74,77,78,79] | 45 | 0.07 | −0.09 | 0.22 |
| Medium | [6,65,68,70,72,75,76] | 24 | 0.20 | −0.02 | 0.41 |
| Long | [70] | 6 | 0.04 | −0.37 | 0.45 |
| Exercise Modality | |||||
| Walking/Running | [31,32,33,63,64,68,70,73,76,77,78,79] | 57 | 0.06 | −0.07 | 0.19 |
| Cycling | [6,65,72,75] | 10 | 0.46 * | 0.12 * | 0.81 * |
| Moderator | Exercise During Memory Encoding vs. Control | ||||
|---|---|---|---|---|---|
| Reference | Number of Effect Size Contributions | Effect Size (Cohen’s d) | Lower CI | Upper CI | |
| Sex | |||||
| Mixed | [10,31,32,33,76] | 16 | −0.13 * | −0.26 * | 0.00 * |
| Predominately Female | [69] | 2 | −0.09 | −0.33 | 0.15 |
| Race-Ethnicity | |||||
| Predominately white | [33,76] | 6 | 0.00 | −0.17 | 0.17 |
| Mixed | [31,32] | 8 | −0.27 * | −0.48 * | −0.06 * |
| Memory Type | |||||
| Short-term | [31,32,33,69,76] | 6 | 0.02 | -0.14 | 0.18 |
| Long-term | [10,31,32,33,76] | 12 | −0.23 * | −0.36 * | −0.09 * |
| Exercise Intensity | |||||
| Light | [10,69] | 4 | −0.15 | −0.35 | 0.05 |
| Moderate | [32,33,76] | 10 | −0.09 | −0.26 | 0.07 |
| Vigorous | [31] | 4 | −0.18 | −0.49 | 0.14 |
| Exercise Duration | |||||
| Short | [31,32,33] | 12 | −0.20 * | −0.35 * | −0.04 * |
| Medium | [10,76] | 4 | 0.00 | −0.22 | 0.22 |
| Moderator | Exercise During Early Consolidation vs. Control | ||||
|---|---|---|---|---|---|
| Reference | Number of Effect Size Contributions | Effect Size (Cohen’s d) | Lower CI | Upper CI | |
| Age | |||||
| Young Adult | [6,19,31,32,33,63,64,67,73,74,75] | 59 | 0.54 * | 0.35 * | 0.73 * |
| Older Adults | [63] | 3 | −0.95 | −1.76 | −0.15 |
| Sex | |||||
| Mixed | [6,19,31,32,33,64,67,73,74,75] | 55 | 0.60 * | 0.40 * | 0.80 * |
| Race-Ethnicity | |||||
| Predominately white | [33] | 4 | −0.07 | −0.76 | 0.63 |
| Mixed | [6,31,32,75] | 12 | −0.14 | −0.56 | 0.28 |
| Memory Type | |||||
| Short-term | [31,32,33,67,73,74] | 35 | 1.05 * | 0.79 * | 1.30 * |
| Long-term | [6,19,31,32,33,63,64,73,74,75] | 26 | −0.14 | −0.40 | 0.11 |
| Exercise Intensity | |||||
| Light | [63] | 6 | −0.59 * | −1.12 * | −0.06* |
| Moderate | [6,32,33,64,73,75] | 19 | −0.02 | −0.31 | 0.27 |
| Vigorous | [19,31,67] | 33 | 1.09 * | 0.83 * | 1.35 * |
| Exercise Duration | |||||
| Short | [31,32,33,63,64,73,74] | 29 | −0.13 | −0.34 | 0.09 |
| Medium | [6,19,75] | 5 | 0.21 | −0.32 | 0.74 |
| Long | [67] | 28 | 1.36 * | 1.09 * | 1.64 * |
| Exercise Modality | |||||
| Walking/Running | [31,32,33,63,64,73,75] | 26 | −0.19 | −0.43 | 0.05 |
| Cycling | [6,19,67] | 32 | 1.17 * | 0.91 * | 1.43 * |
| Moderator | Exercise During Late Consolidation vs. Control | ||||
|---|---|---|---|---|---|
| Reference | Number of Effect Size Contributions | Effect Size (Cohen’s d) | Lower CI | Upper CI | |
| Memory Type | |||||
| Long-term | [19,40] | 3 | 1.20 * | 0.13 * | 2.27 * |
| Exercise Duration | |||||
| Short | [40] | 3 | 1.31 * | 0.20 * | 2.43 * |
| Exercise Modality | |||||
| Walking/Running | [40] | 3 | 1.31 * | 0.20 * | 2.43 * |
| Exercise and Memory Temporal Periods | ||||
|---|---|---|---|---|
| Moderator | Before vs. Control | During vs. Control | Early vs. Control | Late vs. Control |
| Demographic Characteristic | ||||
| Young adults | + | + | ||
| Older adults | − | |||
| Mixed-sex sample | + | − | + | |
| Predominately white | + | |||
| Racially-Ethnically mixed sample | − | |||
| Exercise Characteristic | ||||
| Light-intensity | − | |||
| Vigorous-intensity | + | + | ||
| Short-duration | − | + | ||
| Long-duration | + | |||
| Cycling | + | + | ||
| Memory | ||||
| Short-term memory | + | |||
| Long-term memory | + | − | + | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Loprinzi, P.D.; Blough, J.; Crawford, L.; Ryu, S.; Zou, L.; Li, H. The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis. Brain Sci. 2019, 9, 87. https://doi.org/10.3390/brainsci9040087
Loprinzi PD, Blough J, Crawford L, Ryu S, Zou L, Li H. The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis. Brain Sciences. 2019; 9(4):87. https://doi.org/10.3390/brainsci9040087
Chicago/Turabian StyleLoprinzi, Paul D., Jeremiah Blough, Lindsay Crawford, Seungho Ryu, Liye Zou, and Hong Li. 2019. "The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis" Brain Sciences 9, no. 4: 87. https://doi.org/10.3390/brainsci9040087
APA StyleLoprinzi, P. D., Blough, J., Crawford, L., Ryu, S., Zou, L., & Li, H. (2019). The Temporal Effects of Acute Exercise on Episodic Memory Function: Systematic Review with Meta-Analysis. Brain Sciences, 9(4), 87. https://doi.org/10.3390/brainsci9040087





