Mechanical Property of Sn-58Bi Solder Paste Strengthened by Resin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Specimen Preparation
2.2. Testing Device
3. Results and Discussion
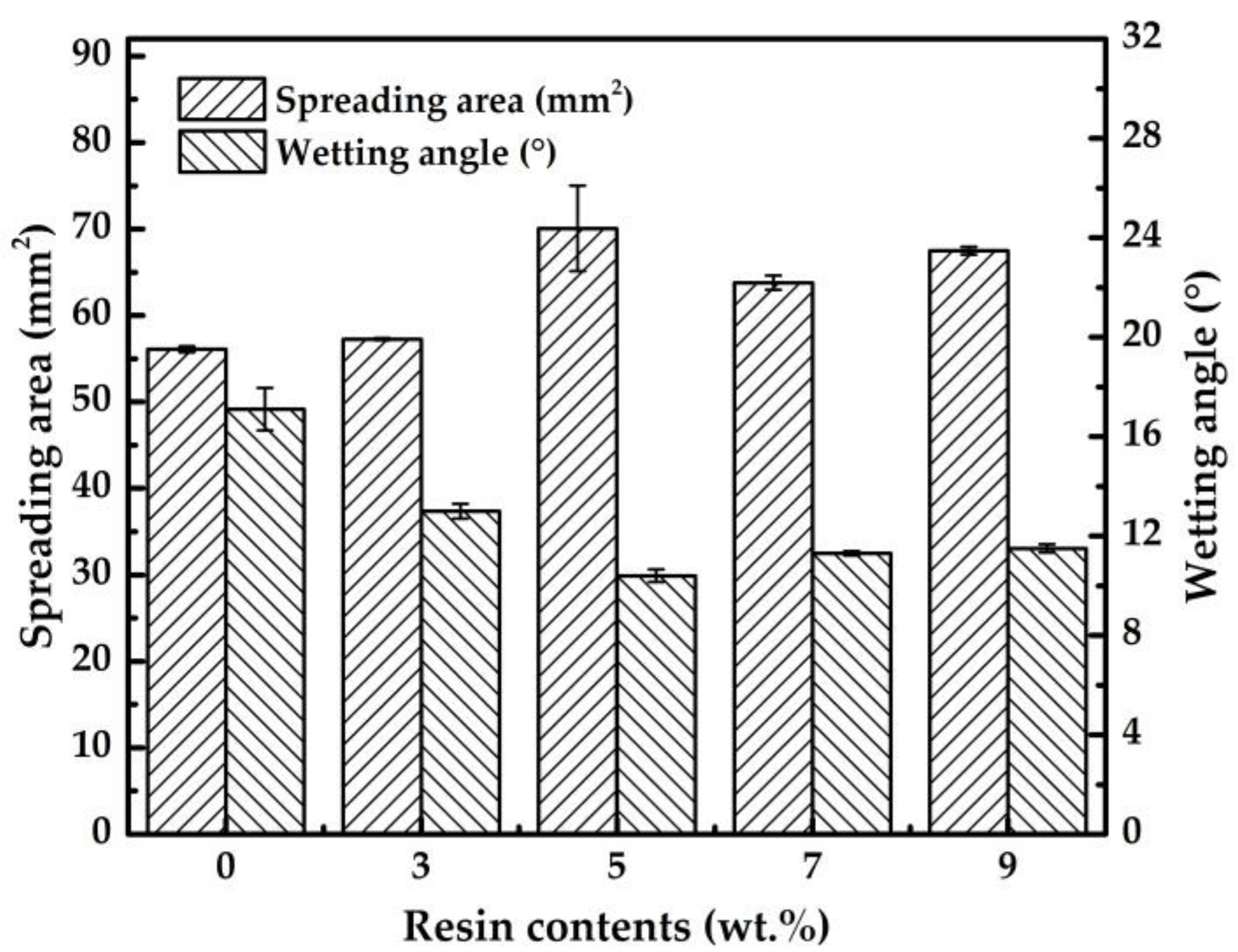
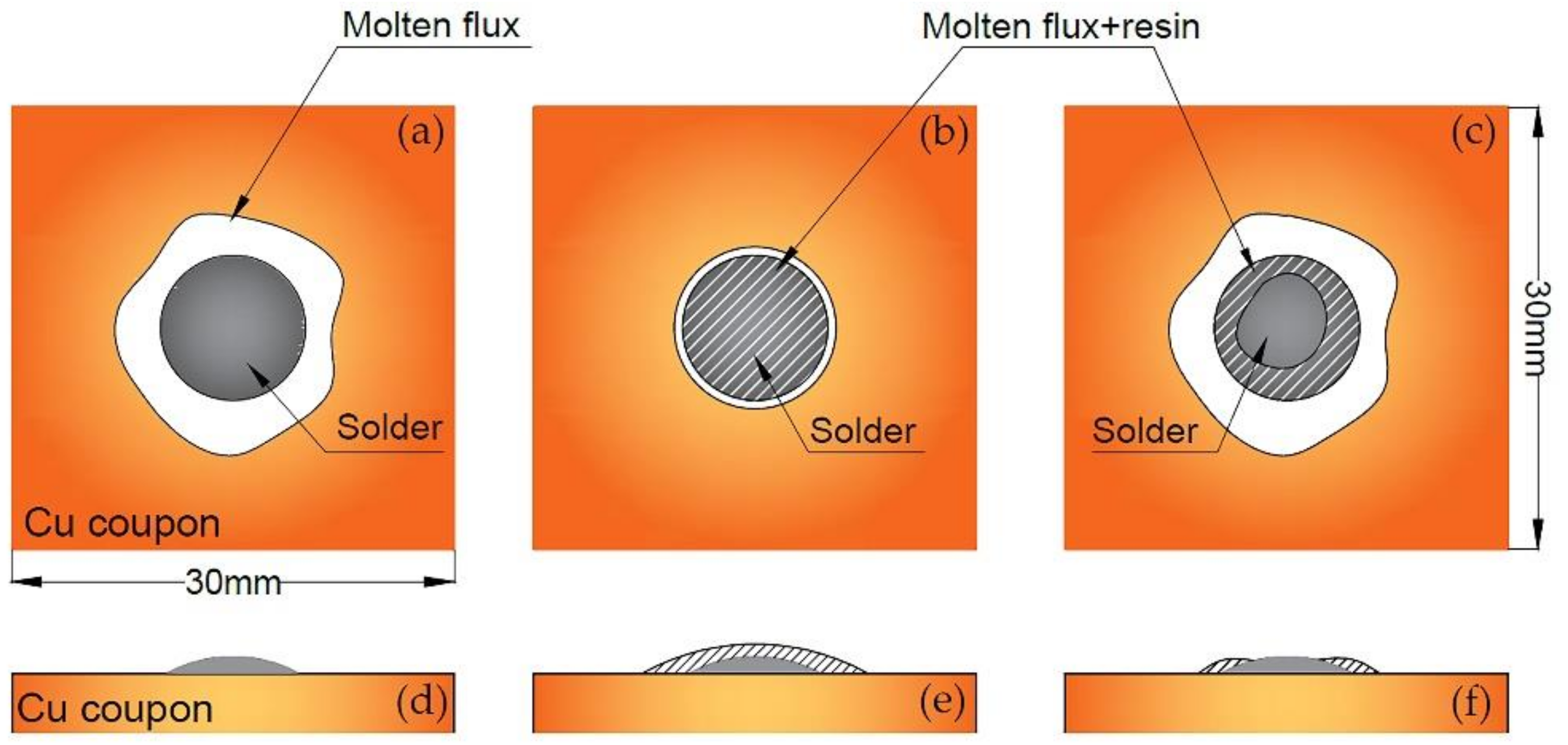
3.1. Spreading Performance of Solder Paste
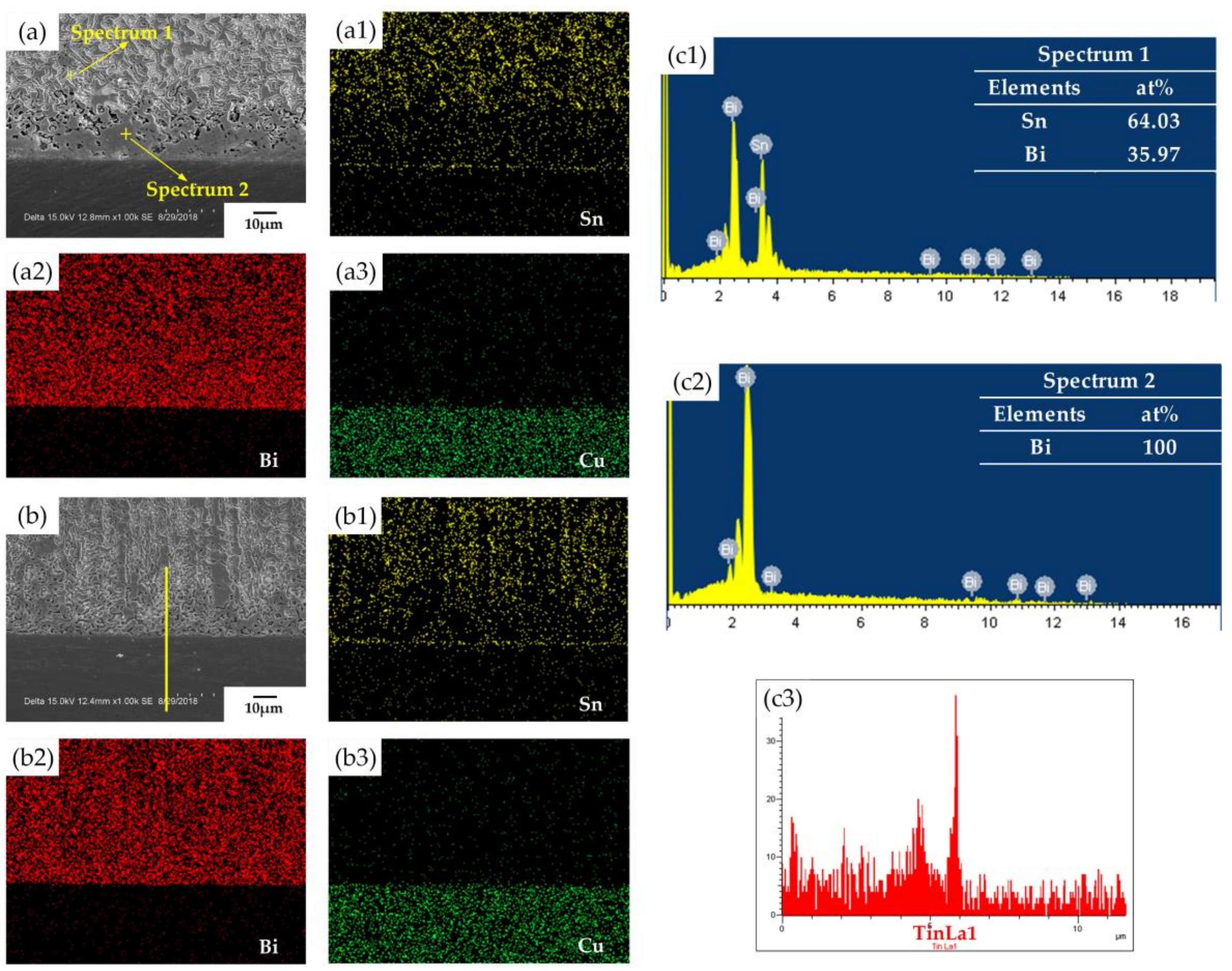
3.2. Microstructure Observation and Analysis
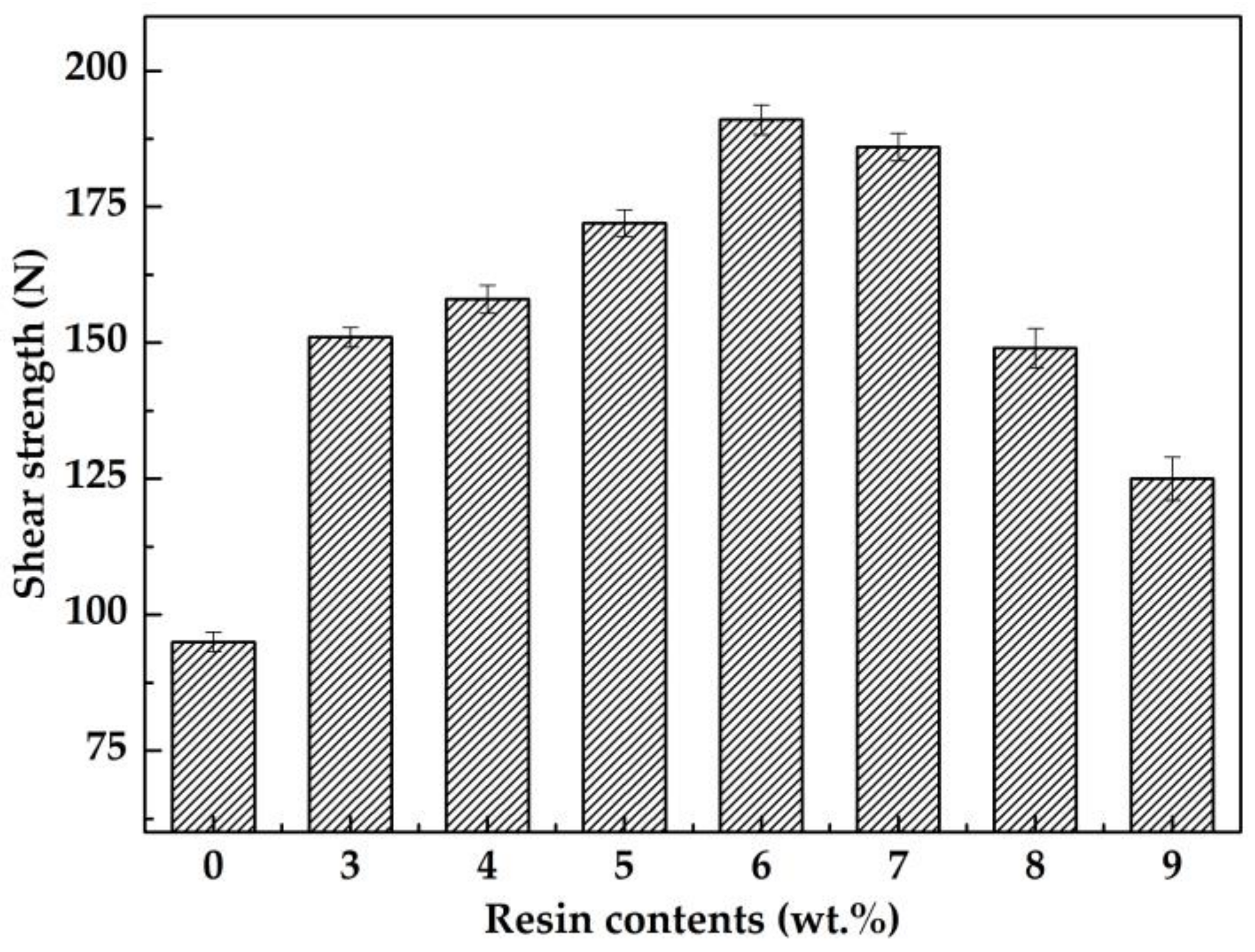
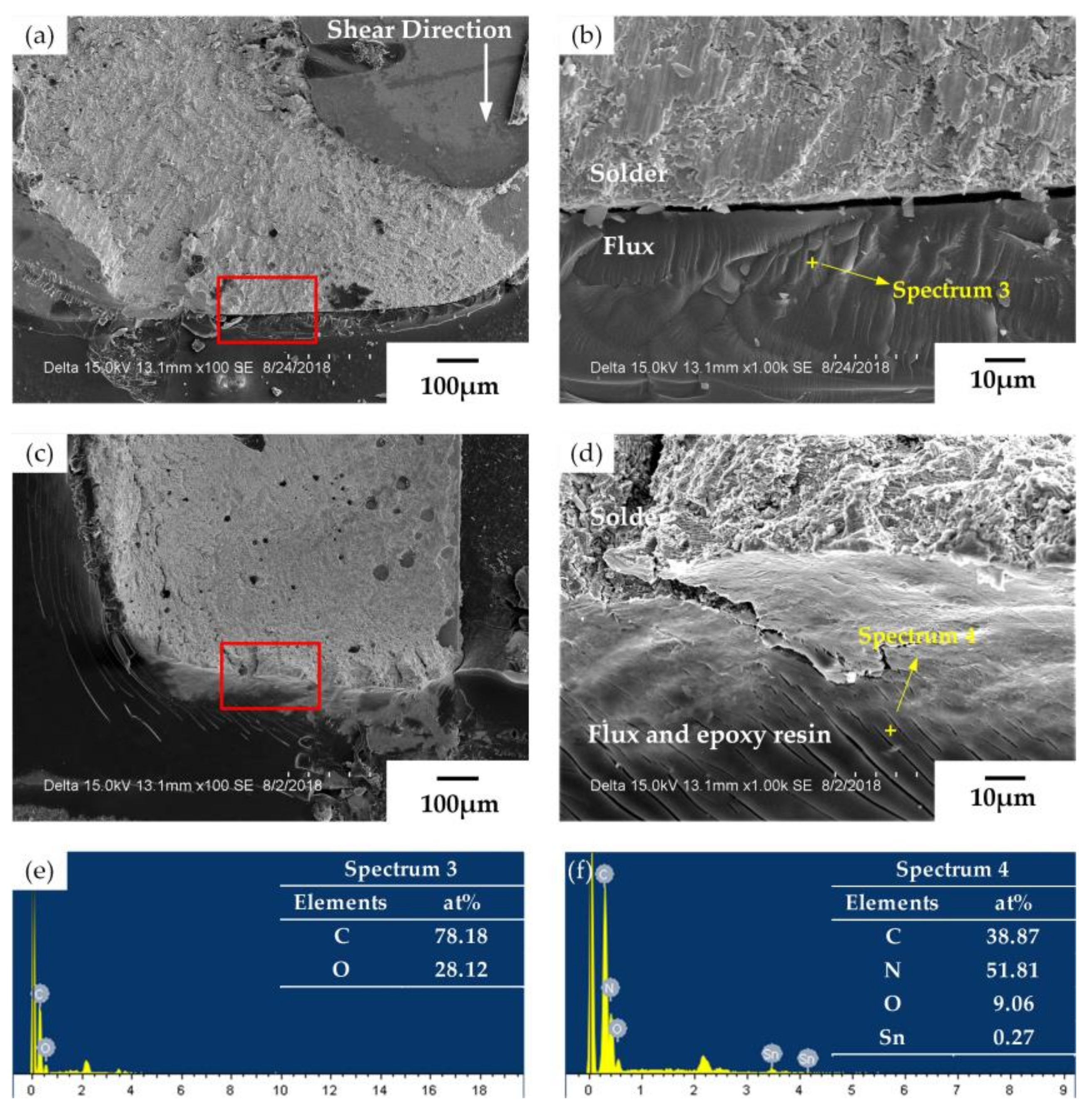
3.3. Fracture Analysis and Shear Strength
4. Conclusions
- (1)
- The Sn-58Bi solder paste with an appropriate amount epoxy of resin exhibited increased wettability because of the surface tension was reduced by the epoxy resin.
- (2)
- After reflow, it was observed that the epoxy resin promoted the reaction between Sn-58Bi and Cu and improved the excessive enrichment of bismuth near the interface.
- (3)
- The addition of epoxy resin increased the shear strength of Sn-58Bi solder joints; the shear strength of the Sn-58Bi + 6 wt.% ER solder joint was found to be nearly twice as high as that of the Sn-58Bi solder joint. Deformation caused by shear force was observed from the fracture of the flux with epoxy resin, which affected the solder joint shear strength by mechanical resistance.
- (4)
- Shear test results revealed that all solder joints had brittle fracture behaviors, while the fracture tended to exhibit a toughness and brittleness mixing morphology due to the addition of epoxy resin. However, carbon and oxygen were observed in the holes in the fracture when the epoxy resin was excessive, indicating that excess epoxy resin could decrease the shear strength of solder joints.
Author Contributions
Funding
Conflicts of Interest
References
- Lu, B.; Yang, J.X.; Ijomah, W.; Wu, W.J.; Zlamparet, G. Perspectives on reuse of WEEE in China: Lessons from the EU. Resour. Conserv. Recycl. 2018, 135, 83–92. [Google Scholar] [CrossRef]
- Balaram, V.; Rambabu, U.; Reddy, M.R.P.; Munirathnam, N.R.; Chatterjee, S. RoHS Regulation: Challenges in the Measurement of Substances of Concern in Industrial Products by Different Analytical Techniques. MAPAN-J. Metrol. Soc. India 2018, 33, 329–346. [Google Scholar] [CrossRef]
- Zhang, L.; Tu, K.N. Structure and properties of lead-free solders bearing micro and nano particles. Mater. Sci. Eng. R Rep. 2014, 82, 1–32. [Google Scholar] [CrossRef]
- Kotadia, H.R.; Howes, P.D.; Mannan, S.H. A review: On the development of low melting temperature Pb-free solders. Microelectron. Reliab. 2014, 54, 1253–1273. [Google Scholar] [CrossRef]
- Gain, A.K.; Zhang, L. Microstructure, thermal analysis and damping properties of Ag and Ni nano-particles doped Sn–8Zn–3Bi solder on OSP–Cu substrate. J. Alloy. Compd. 2014, 617, 779–786. [Google Scholar] [CrossRef]
- Gain, A.K.; Zhang, L.; Quadir, M.Z. Thermal aging effects on microstructures and mechanical properties of an environmentally friendly eutectic tin-copper solder alloy. Mater. Des. 2016, 110, 275–283. [Google Scholar] [CrossRef]
- Tsao, L.C.; Chang, S.Y. Effects of Nano-TiO2 additions on thermal analysis, microstructure and tensile properties of Sn3.5Ag0.25Cu solder. Mater. Des. 2010, 31, 990–993. [Google Scholar] [CrossRef]
- Cheng, S.F.; Huang, C.M.; Michael, P. A review of lead-free solders for electronics applications. Microelectron. Reliab. 2017, 75, 77–95. [Google Scholar] [CrossRef]
- Jovanovski, V.; Hocevar, S.B.; Ogorevc, B. Bismuth electrodes in contemporary electroanalysis. Curr. Opin. Electrochem. 2017, 3, 114–122. [Google Scholar] [CrossRef]
- Wislei, R.O.; Leandro, C.P.; Lenoardo, R.G.; Nathalie, M.; Amauri, G. Microstructure and mechanical properties of Sn-Bi, Sn-Ag and Sn-Zn lead-free solder alloys. J. Alloy. Compd. 2013, 572, 97–106. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, J.; Evans, J.; Johnson, W.; Vardaman, J.; Fujimura, I.; Tseng, A.; Knight, J. Tin-Bismuth Plating for Component Finishes. In Proceedings of the IEEE 61st Electronic Components and Technology Conference (ECTC), Lake Buena Vista, FL, USA, 31 May–3 June 2011; pp. 2060–2066. [Google Scholar] [CrossRef]
- Belyakov, S.A.; Gourlay, C.M. Recommended values for the βSn solidus line in Sn-Bi alloys panel. Thermochim. Acta 2017, 654, 65–69. [Google Scholar] [CrossRef]
- Liu, S.Q.; McDonald, S.; Sweatman, K.; Nogita, K. The effects of precipitation strengthening and solid solution strengthening on strain rate sensitivity of lead-free solders: Review. Microelectron. Reliab. 2018, 84, 170–180. [Google Scholar] [CrossRef]
- Wang, F.J.; Chen, H.; Huang, Y.; Yan, C. Interfacial behavior and joint strength of Sn–Bi solder with solid solution compositions. J. Mater. Sci. Mater. Electron. 2018, 29, 11409–11420. [Google Scholar] [CrossRef]
- Gain, A.K.; Zhang, L. Growth mechanism of intermetallic compound and mechanical properties of nickel (Ni) nanoparticle doped low melting temperature tin-bismuth (Sn-Bi) solder, J. Mater. Sci. Mater. Electron. 2016, 27, 781–794. [Google Scholar] [CrossRef]
- Mokhtari, O.; Nishikawa, H. Correlation between microstructure and mechanical properties of Sn–Bi–X solders. Mater. Sci. Eng. A Struct. Mater. 2016, 651, 831–839. [Google Scholar] [CrossRef]
- Hu, F.Q.; Zhang, Q.K.; Jiang, J.J.; Song, Z.L. Influences of Ag addition to Sn-58Bi solder on SnBi/Cu interfacial reaction. Mater. Lett. 2018, 214, 142–145. [Google Scholar] [CrossRef]
- Liu, Y.; Fu, H.F.; Sun, F.L.; Zhang, H.; Kong, X.X.; Xin, T. Microstructure and mechanical properties of as-reflowed composite solder pastes. J. Mater. Process. Technol. 2016, 238, 290–296. [Google Scholar] [CrossRef]
- Zhang, C.; Liu, S.D.; Qian, G.T.; Zhou, J.; Xue, F. Effect of Sb content on properties of Sn-Bi solders. Trans. Nonferrous Met. Soc. China 2014, 24, 184–191. [Google Scholar] [CrossRef]
- Hakiki, F.; Nuraeni, N.; Salam, D.D.; Akbari, A.; Alqhisty, N.; Aditya, W.; Siregar, S. Is Epoxy-Based Polymer Suitable for Water Shut-Off Application. In Proceedings of the 2015 SPE/IATMI Asia Pacific Oil & Gas Conference and Exhibition, Nusa Dua, Bali, Indonesia, 20–22 October 2015. [Google Scholar] [CrossRef]
- Satyanarayan, P.K.N. Effect of temperature and substrate surface texture on wettability and morphology of IMCs between Sn-0.7Cu solder alloy and copper substrate. J. Mater. Sci.-Mater. Electron. 2012, 23, 1664–1672. [Google Scholar] [CrossRef]
- Sona, M.; Prabhu, K.N. Spreading Behavior and Joint Reliability of Sn-0.3Ag-0.7Cu Lead-Free Solder Alloy on Nickel Coated Copper Substrate as a Function of Reflow Time. Trans. Indian Inst. Met. 2015, 68, 1027–1031. [Google Scholar] [CrossRef]
- Chen, X.; Xue, F.; Zhou, J.; Yao, Y. Effect of In on microstructure, thermodynamic characteristic and mechanical properties of Sn-Bi based lead-free solder. J. Alloy. Compd. 2015, 633, 377–383. [Google Scholar] [CrossRef]
- Geranmayeh, A.R.; Nayyeri, G.; Mahmudi, R. Microstructure and impression creep behavior of lead-free Sn-5Sb solder alloy containing Bi and Ag. Mater. Sci. Eng. A-Struct. Mater. Prop. Microstruct. Process. 2012, 547, 110–119. [Google Scholar] [CrossRef]
- Myung, W.R.; Kim, Y.; Jung, S.B. Mechanical property of the epoxy-contained Sn-58Bi solder with OSP surface finish. J. Alloy. Compd. 2014, 615, 411–417. [Google Scholar] [CrossRef]
- Ma, D.L.; Wu, P. Improved microstructure and mechanical properties for Sn58Bi0.7Zn solder joint by addition of graphene nanosheets. J. Alloy. Compd. 2016, 671, 127–136. [Google Scholar] [CrossRef]
- Yang, M.; Li, M.Y.; Wang, C.Q. Interfacial reactions of eutectic Sn3.5Ag and pure tin solders with Cu substrates during liquid-state soldering. Intermetallics 2012, 25, 86–94. [Google Scholar] [CrossRef]
- Mokhtari, O.; Nishikawa, H. The shear strength of transient liquid phase bonded Sn-Bi solder joint with added Cu patricles. Adv. Powder Technol. 2016, 27, 1000–1005. [Google Scholar] [CrossRef]
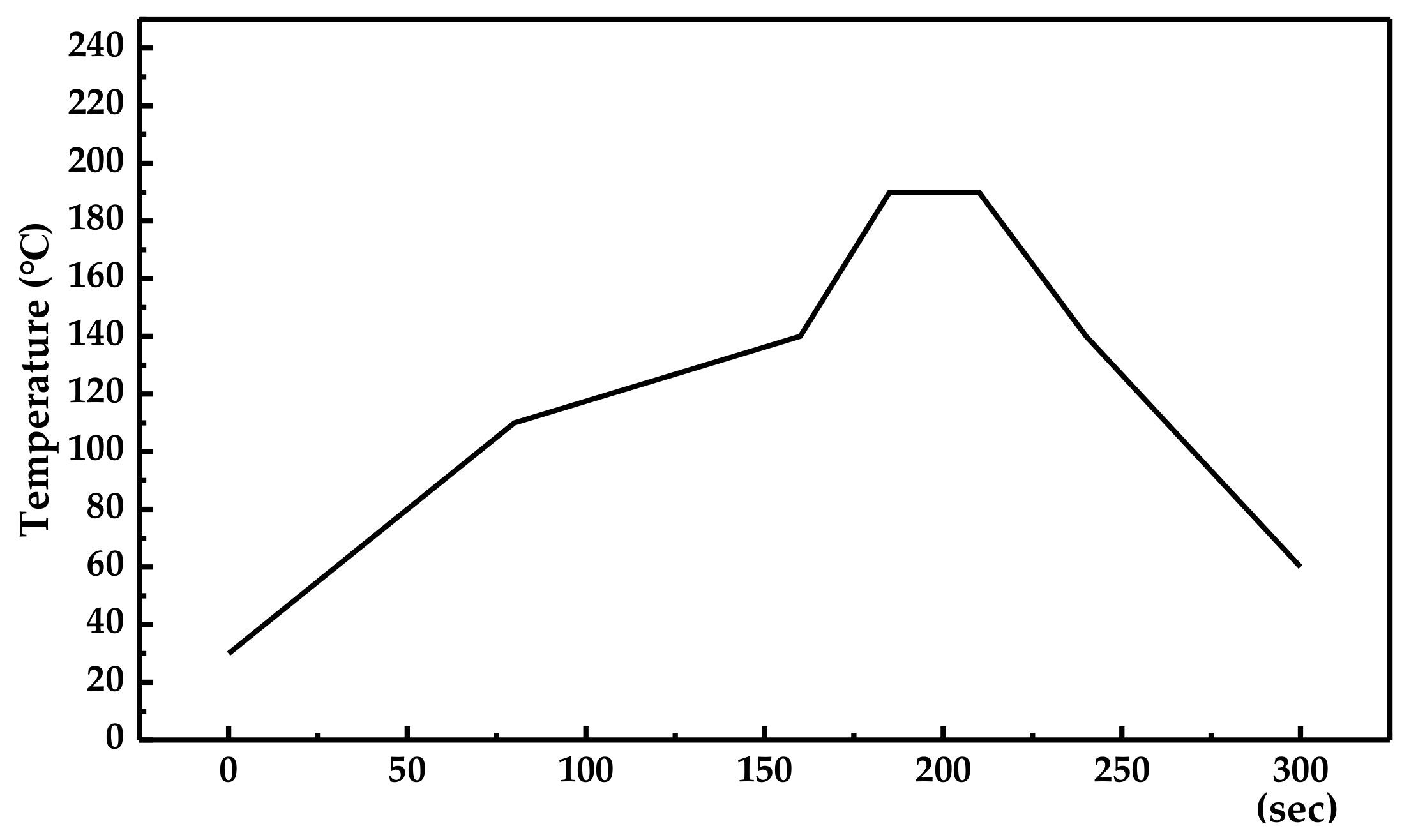

| Resin Category | Epoxy Resin |
|---|---|
| Curing temperature (°C) | 160~180 |
| Curing time (s) | 120~300 |
| Curing agent | Y |
| Resin Content | 0 wt.% | 3 wt.% | 5 wt.% | 7 wt.% | 9 wt.% |
|---|---|---|---|---|---|
| Spreading area (mm2) |  |  |  |  |  |
| Wetting angle (°) |  |  |  |  |  |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, L.; Xue, S.; Liu, S. Mechanical Property of Sn-58Bi Solder Paste Strengthened by Resin. Appl. Sci. 2018, 8, 2024. https://doi.org/10.3390/app8112024
Liu L, Xue S, Liu S. Mechanical Property of Sn-58Bi Solder Paste Strengthened by Resin. Applied Sciences. 2018; 8(11):2024. https://doi.org/10.3390/app8112024
Chicago/Turabian StyleLiu, Lu, Songbai Xue, and Siyi Liu. 2018. "Mechanical Property of Sn-58Bi Solder Paste Strengthened by Resin" Applied Sciences 8, no. 11: 2024. https://doi.org/10.3390/app8112024
APA StyleLiu, L., Xue, S., & Liu, S. (2018). Mechanical Property of Sn-58Bi Solder Paste Strengthened by Resin. Applied Sciences, 8(11), 2024. https://doi.org/10.3390/app8112024





