In-Hospital LSVT BIG Training Versus Structured Rehabilitation Treatment in Parkinson’s Disease: Feasibility and Primary Evaluation on Functional and Respiratory Outcomes
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
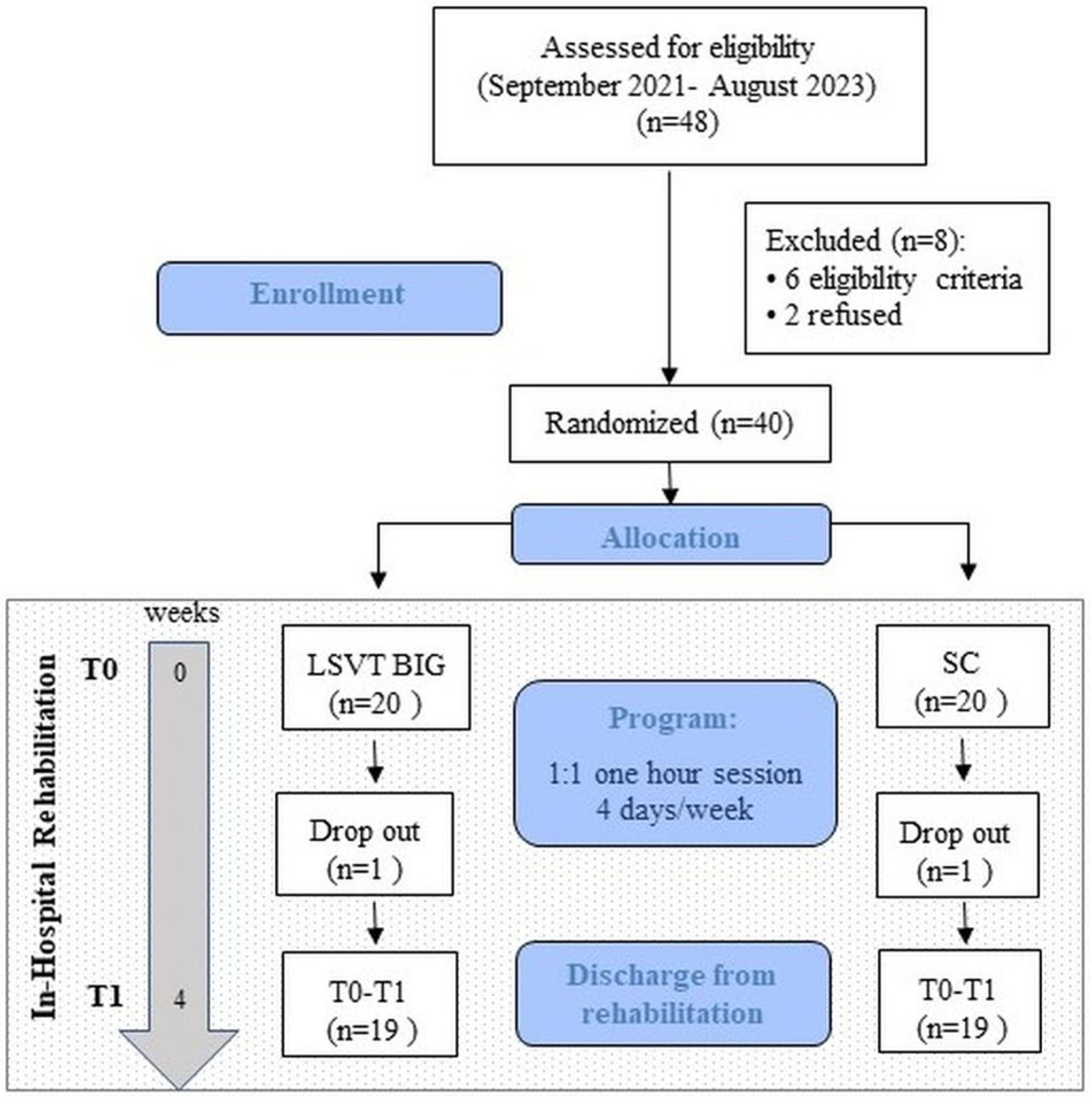
2.1. Design of the Study
2.2. Patients
2.3. Rehabilitation Program
2.4. Study Groups and Specific Training Programs
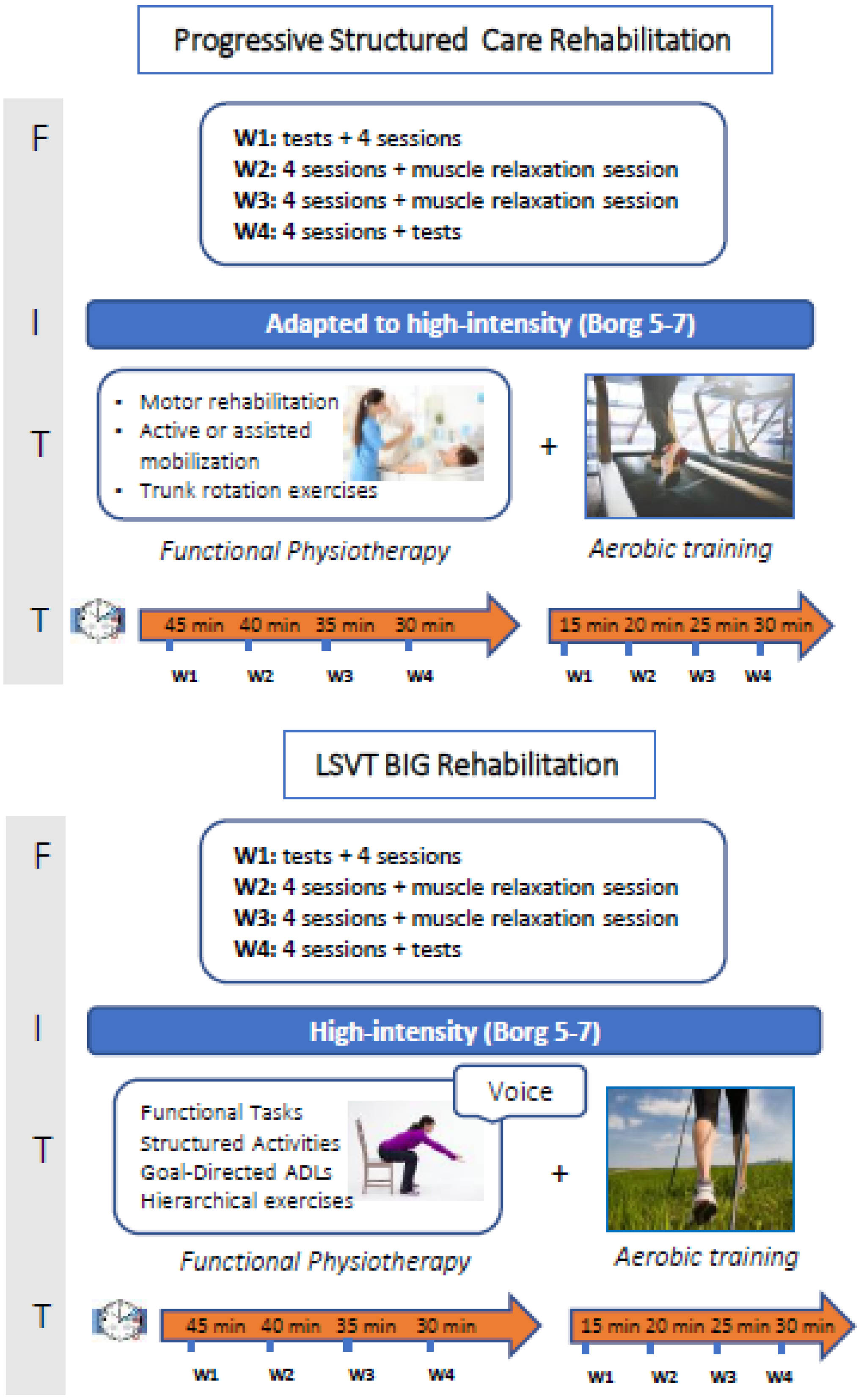
2.4.1. Progressive Structured Care (SC) Rehabilitation Program
- Motor rehabilitation focusing on gradual, full-range passive mobilization exercises for the trunk and limbs, performed on a physical therapist’s couch.
- Active or assisted mobilization exercises, such as active head and trunk extension exercises, muscle activation for upper and lower limbs, and stretching exercises.
- Trunk rotation exercises, sit-to-stand and stand-to-sit transitions, and balance exercises in both sitting and standing positions.
- Aerobic training consisted of walking on a treadmill, with speeds ranging from 1.8 to 3.5 km/h. The intensity of the treadmill training was carefully monitored using the Borg Fatigue Scale [23] (range 5–7), using Maltais protocol [24], ensuring it was as intensive as the LSVT BIG program. The heart rate was constantly monitored to be less than 90% of predicted (220 beats per minute—age). This aerobic component also incorporated a progression in intensity throughout the program.
- Time for free-body functional activity decreased from 45 to 30 min (5 min less each week, starting from week 1 to week 4).
- Time for aerobic treadmill activity increased from 15 to 30 min (5 min more each week, starting from week 1 to week 4).
- By week 4, an equal amount of time (30 min) was spent on both types of activities (Figure 2).
2.4.2. LSVT BIG
- Functional Tasks and Structured Activities: Patients practice large, exaggerated movements with their whole body. This includes repetitive movements in various directions, like stepping and reaching, and gentle stretching. The goal is to maximize the amplitude of their movements.
- Goal-Directed Activities of Daily Living (ADLs): This component integrates those “bigger” movements into everyday tasks. Patients practice ADLs (like getting dressed or eating) with high-amplitude movements and an aerobic component to improve their overall physical endurance.
- The “BIG Walk” is a crucial aerobic part of the program. Patients are encouraged to walk with large, exaggerated movements of both their arms and legs. This walking exercise is personalized to each patient’s abilities and adapted to various real-world situations. For instance, they might practice changing direction, navigating narrow spaces, stepping over obstacles, or walking through doorways, all while maintaining those big, purposeful movements.
2.5. Measures
2.5.1. Primary Outcome
2.5.2. Secondary Outcomes
- (a)
- Disease progression by motor experiences of daily living (UPDRSII, score from 0 to 52—lowest level of function) and PD-related motor examinations (UPDRSIII score from 0 to 132—lowest level of function) [25].
- (b)
- (c)
- Balance by Mini-BESTest [28] (Mini-BESTest, score from 0 to 28—highest level of function), Timed Up and Go test [29] as part of Mini-BESTest (Mini-BESTest-TUG, expressed in seconds to do the task), Timed Up and Go dual task as part of Mini-BESTest (Mini-BESTest-TUG cog, expressed in seconds to do the task with cognitive involvement) and Berg Balance Scale (score from 0 to 56—highest level of function) [30].
- (d)
- Lower limb strength by the 5 repetitions Sit-to-Stand test (5STS, expressed in seconds to do the task) [31].
- (e)
- Lower limb and upper limb exercise tolerance by 6 min walking test (6MWT) [32] and the Unsupported Upper Limb Exercise test (UULEX) [33], respectively. For 6MWT, the distance walked in meters was collected as % of predicted meters [34] calculated, and the Borg Fatigue scale [23] was recorded at the beginning and end of the test. UULEX maximum level reached and the total time of duration were recorded. For both tests, mean oximetry (SpO2%) (Spirodoc, MIR Medical International Research, USA) was recorded.
- (f)
- Respiratory muscle strength by maximal inspiratory pressure (MIP) and maximal expiratory pressure (MEP) [35] (MicroRPM, Care-Fusion, Hoechberg, Germany), both expressed in cmH2O and % of predicted value and respiratory function by peak cough expiratory flow was recorded (PCEF, expressed in L/min) [36] (Spirodoc, MIR Medical International Research, Suite O. Waukesha, WI, USA with Ambu Ultraseal mask, Ballierup, Denmark).
- (g)
2.6. Statistical Analysis
3. Results
3.1. Primary Outcome: Feasibility
3.2. Sample Characteristics
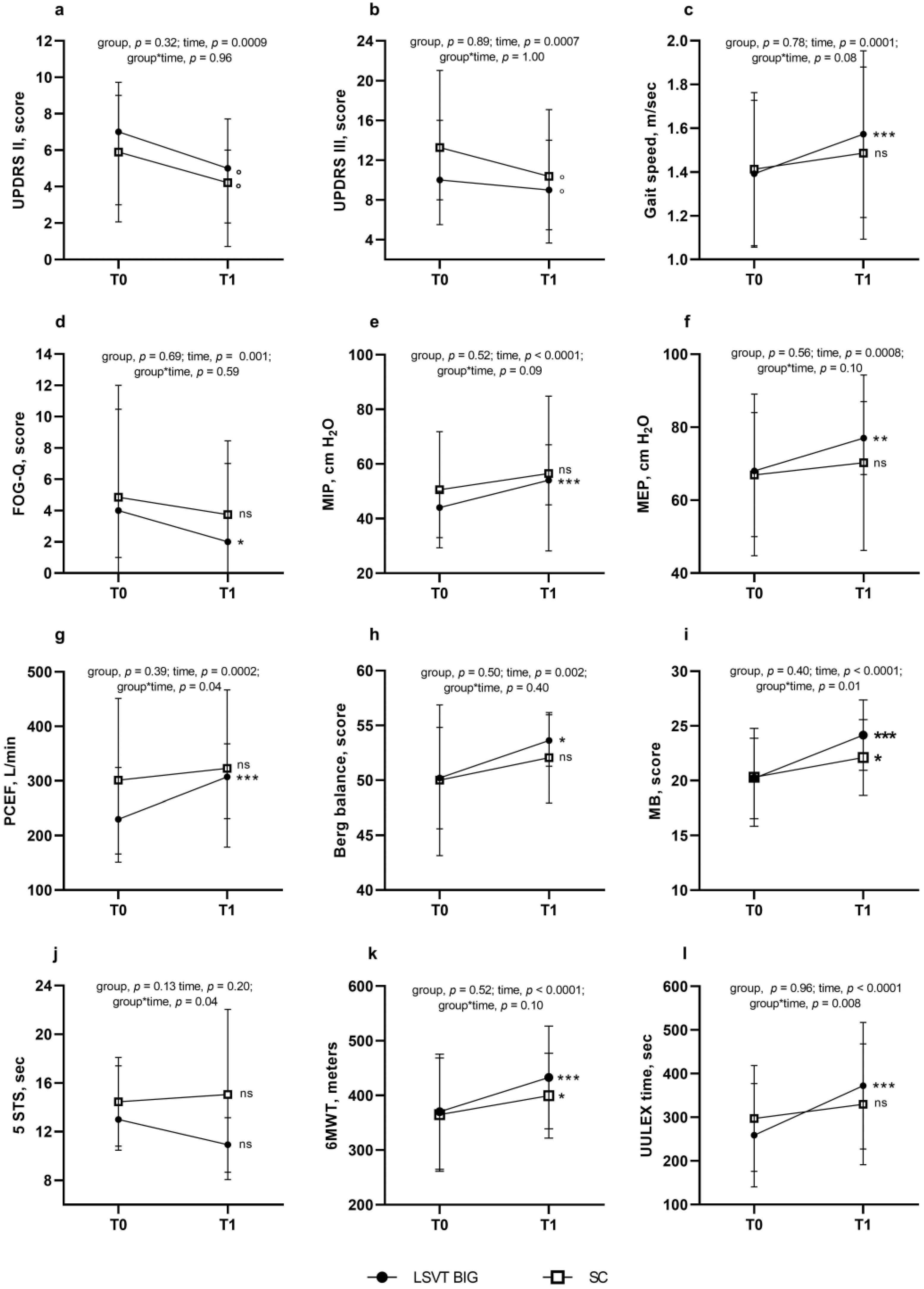
3.3. Secondary Outcomes: Pre-to-Post Changes Between Outcomes
3.4. Correlations
4. Discussion
4.1. Primary Outcome
4.2. Secondary Outcomes
4.3. Clinical Implication
4.4. Limitations
4.5. Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | Body Mass Index |
| 5-STS | 5 repetitions Sit-to-Stand |
| FITT | Frequency, Intensity, Time, Type |
| FOG-Q | Freezing of Gait Questionnaire |
| HY | Hoehn and Yahr |
| LSVT BIG | Lee Silverman Voice Treatment BIG |
| LTOT | Long-term oxygen therapy |
| MB | Mini-BESTest |
| MIP | Maximal inspiratory pressure |
| MCID | Minimal Clinical Important Difference |
| MEP | Maximal expiratory pressure |
| MMSE | Mini mental state Examination |
| PCEF | Peak Cough Expiratory Flow |
| PD | Parkinson’s disease |
| PDQ39 | Parkinson’s Disease Quality of Life Questionnaire-39 scale |
| PFS | Parkinson Fatigue scale (polytomous method score) |
| 6MWT | 6 min walking test |
| SatO2 | Oxygen saturation |
| SI | Summary Index |
| SC | Structured care |
| TUG | Timed Up and Go test; |
| TUG cog | Timed Up and Go dual task (cognitive involvement) |
| UPDRS | Unified Parkinson’s Disease Rating Scale |
| UULEX | Unsupported Upper Limb Exercise test |
References
- Simon, D.K.; Tanner, C.M.; Brundin, P. Parkinson Disease Epidemiology, Pathology, Genetics, and Pathophysiology. Clin. Geriatr. Med. 2020, 36, 1–12. [Google Scholar] [CrossRef]
- Tysnes, O.-B.; Storstein, A. Epidemiology of Parkinson’s disease. J. Neural Transm. 2017, 124, 901–905. [Google Scholar] [CrossRef]
- Ray, S.; Agarwal, P. Depression and Anxiety in Parkinson Disease. Clin. Geriatr. Med. 2020, 36, 93–104. [Google Scholar] [CrossRef] [PubMed]
- Alberts, J.L.; Rosenfeldt, A.B. The Universal Prescription for Parkinson’s Disease: Exercise. J. Park. Dis. 2020, 10, S21–S27. [Google Scholar] [CrossRef] [PubMed]
- Keus, S.H.; Bloem, B.R.; Hendriks, E.J.; Bredero-Cohen, A.B.; Munneke, M.; Practice Recommendations Development Group. Evidence-based analysis of physical therapy in Parkinson’s disease with recommendations for practice and research. Mov. Disord. 2007, 22, 451–600. [Google Scholar] [CrossRef] [PubMed]
- Suchowersky, O.; Gronseth, G.; Perlmutter, J.; Reich, S.; Zesiewicz, T.; Weiner, W.J.; Quality Standards Subcommittee of the American Academy of Neurology. Practice Parameter: Neuroprotective strategies and alternative therapies for Parkinson disease (an evidence-based review): Report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology 2006, 66, 976–982. [Google Scholar] [CrossRef]
- Ernst, M.; Folkerts, A.-K.; Gollan, R.; Lieker, E.; Caro-Valenzuela, J.; Adams, A.; Cryns, N.; Monsef, I.; Dresen, A.; Roheger, M.; et al. Physical exercise for people with Parkinson’s disease: A systematic review and network meta-analysis. Cochrane Database Syst. Rev. 2023, 2023, CD013856. [Google Scholar] [CrossRef]
- Abbruzzese, G.; Marchese, R.; Avanzino, L.; Pelosin, E. Rehabilitation for Parkinson’s disease: Current outlook and future challenges. Park. Relat. Disord. 2016, 22 (Suppl. S1), S60–S64. [Google Scholar] [CrossRef]
- Lim, I.; van Wegen, E.; de Goede, C.; Deutekom, M.; Nieuwboer, A.; Willems, A.; Jones, D.; Rochester, L.; Kwakkel, G. Effects of external rhythmical cueing on gait in patients with Parkinson’s disease: A systematic review. Clin. Rehabil. 2005, 19, 695–713. [Google Scholar] [CrossRef]
- Nieuwboer, A.; Kwakkel, G.; Rochester, L.; Jones, D.; van Wegen, E.; Willems, A.M.; Chavret, F.; Hetherington, V.; Baker, K.; Lim, I. Cueing training in the home improves gait-related mobility in Parkinson’s disease: The RESCUE trial. J. Neurol. Neurosurg. Psychiatry 2007, 78, 134–140. [Google Scholar] [CrossRef]
- Platz, T.; Brown, R.G.; Marsden, C.D. Training improves the speed of aimed movements in Parkinson’s disease. Brain 1998, 121, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Jöbges, M.; Heuschkel, G.; Pretzel, C.; Illhardt, C.; Renner, C.; Hummelsheim, H. Repetitive training of compensatory steps: A therapeutic approach for postural instability in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2004, 75, 1682–1687. [Google Scholar] [CrossRef] [PubMed]
- Van Eijkeren, F.J.; Reijmers, R.S.; Kleinveld, M.J.; Minten, A.; ter Bruggen, J.P.; Bloem, B.R. Nordic walking improves mobility in Parkinson’s disease. Mov. Disord. 2008, 23, 2239–2243. [Google Scholar] [CrossRef] [PubMed]
- Ashburn, A.; Fazakarley, L.; Ballinger, C.; Pickering, R.; McLellan, L.D.; Fitton, C. A randomised controlled trial of a home based exercise programme to reduce the risk of falling among people with Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2007, 78, 678–684. [Google Scholar] [CrossRef]
- Dibble, L.E.; Hale, T.F.; Marcus, R.L.; Droge, J.; Gerber, J.P.; LaStayo, P.C. High-intensity resistance training amplifies muscle hypertrophy and functional gains in persons with Parkinson’s disease. Mov. Disord. 2006, 21, 1444–1452. [Google Scholar] [CrossRef]
- Farley, B.G.; Koshland, G.F. Training BIG to move faster: The application of the speed-amplitude relation as a rehabilitation strategy for people with Parkinson’s disease. Exp. Brain Res. 2005, 167, 462–467. [Google Scholar] [CrossRef]
- Ebersbach, G.; Ebersbach, A.; Edler, D.; Kaufhold, O.; Kusch, M.; Kupsch, A.; Wissel, J. Comparing exercise in Parkinson’s disease–The Berlin LSVTBIG® BIG study. Mov. Disord. 2010, 25, 1902–1908. [Google Scholar] [CrossRef]
- McDonnell, M.N.; Rischbieth, B.; Schammer, T.T.; Seaforth, C.; Shaw, A.J.; Phillips, A.C. Lee Silverman Voice Treatment (LSVT)-BIG to improve motor function in people with Parkinson’s disease: A systematic review and meta-analysis. Clin. Rehabil. 2018, 32, 607–618. [Google Scholar] [CrossRef]
- Schaible, F.; Maier, F.; Buchwitz, T.M.; Schwartz, F.; Hoock, M.; Schönau, E.; Libuda, M.; Hordt, A.; van Eimeren, T.; Timmermann, L.; et al. Effects of Lee Silverman Voice Treatment BIG and conventional physiotherapy on non-motor and motor symptoms in Parkinson’s disease: A randomized controlled study comparing three exercise models. Ther. Adv. Neurol. Disord. 2021, 14, 1756286420986744. [Google Scholar] [CrossRef]
- Aytutuldu, G.K.; Huseyinsinoglu, B.E.; Sakalli, N.K.; Sen, A.; Yeldan, I. LSVT® BIG versus progressive mobility training through synchronous telerehabilitation in Parkinson’s disease: A randomized controlled trial. Neurol. Sci. 2024, 45, 3163–3172. [Google Scholar] [CrossRef]
- Martinez-Martin, P.; Skorvanek, M.; Rojo-Abuin, J.M.; Gregova, Z.; Stebbins, G.; Goetz, C.G.; Members of the QUALPD Study Group. Validation study of the Hoehn And Yahr scale included in the MDS-UPDRS. Mov. Disord. 2018, 33, 651–652. [Google Scholar] [CrossRef]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.A. Psychophysical bases of perceived exertion. Med. Sci. Sports Exerc. 1982, 14, 377–381. [Google Scholar] [CrossRef]
- Maltais, F.; Decramer, M.; Casaburi, R.; Barreiro, E.; Burelle, Y.; Debigaré, R.; Dekhuijzen, P.N.R.; Franssen, F.; Gayan-Ramirez, G.; Gea, J.; et al. ATS/ERS Ad Hoc Committee on Limb Muscle Dysfunction in COPD. An official American Thoracic Society/European Respiratory Society statement: Update on limb muscle dysfunction in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2014, 189, e15–e62. [Google Scholar] [CrossRef] [PubMed]
- Antonini, A.; Abbruzzese, G.; Strambi, L.F.; Tilley, B.; Huang, J.; Stebbins, G.T.; Goetz, C.G.; Barone, P.; MDS-UPDRS Italian Validation Study Group; di Poggio, M.B.; et al. Validation of the Italian version of the Movement Disorder Society—Unified Parkinson’s Disease Rating Scale. Neurol. Sci. 2013, 34, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Watson, M.J. Refining the ten-meter walking test for use with neurologically impaired people. Physiotherapy 2002, 88, 386–397. [Google Scholar] [CrossRef]
- Giladi, N.; Tal, J.; Azulay, T.; Rascol, O.; Brooks, D.J.; Melamed, E.; Oertel, W.; Poewe, W.H.; Stocchi, F.; Tolosa, E. Validation of the freezing of gait questionnaire in patients with Parkinson’s disease. Mov. Disord. 2009, 24, 655–661. [Google Scholar] [CrossRef]
- Franchignoni, F.; Horak, F.; Godi, M.; Nardone, A.; Giordano, A. Using psychometric techniques to improve the Balance Evaluation Systems Test: The mini-BESTest. J. Rehabil. Med. 2010, 42, 323–331. [Google Scholar] [CrossRef]
- Zampieri, C.; Salarian, A.; Carlson-Kuhta, P.; Aminian, K.; Nutt, J.G.; Horak, F.B. The instrumented timed up and go test: Potential outcome measure for disease modifying therapies in Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2010, 81, 171–176. [Google Scholar] [CrossRef]
- Berg, K.O.; Wood-Dauphinee, S.L.; Williams, J.I.; Gayton, D. Measuring balance in the elderly: Preliminary development of an instrument. Physiother. Can. 1989, 41, 304–311. [Google Scholar] [CrossRef]
- Bohannon, R.W. Reference values for the five-repetition sit-to-stand test: A descriptive meta-analysis of data from elders. Percept. Mot. Skills 2006, 103, 215–222. [Google Scholar] [CrossRef]
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: Guidelines for the 6-minute walk test. Am. J. Respir. Crit. Care Med. 2002, 166, 111–117. [Google Scholar] [CrossRef]
- Takahashi, C.D.; Nemet, D.; Rose-Gottron, C.M.; Larson, J.K.; Cooper, D.M.; Reinkensmeyer, D.J. Effect of muscle fatigue on internal model formation and retention during reaching with the arm. J. Appl. Physiol. 2006, 100, 695–706. [Google Scholar] [CrossRef] [PubMed]
- Enright, P.L.; Sherrill, D.L. Reference equations for the six-minute walk in healthy adults. Am. J. Respir. Crit. Care Med. 1998, 158 Pt 1, 1384–1387. [Google Scholar] [CrossRef] [PubMed]
- Standardized lung function testing. Report working party. Bull. Eur. Physiopathol. Respir. 1983, 19, 1–95.
- Kulnik, S.T.; MacBean, V.; Birring, S.S.; Moxham, J.; Rafferty, G.F.; Kalra, L. Accuracy of portable devices in measuring peak cough flow. Physiol. Meas. 2015, 36, 243–257. [Google Scholar] [CrossRef]
- Brown, R.; Dittner, A.; Findley, L.; Wessely, S. The Parkinson fatigue scale. Park. Relat. Disord. 2005, 11, 49–55. [Google Scholar] [CrossRef]
- Jenkinson, C.; Fitzpatrick, R.; Peto, V.; Greenhall, R.; Hyman, N. The Parkinson’s Disease Questionnaire (PDQ-39): Development and validation of a Parkinson’s disease summary index score. Age Ageing 1997, 26, 353–357. [Google Scholar] [CrossRef]
- Millage, B.; Vesey, E.; Finkelstein, M.; Anheluk, M. Effect on Gait Speed, Balance, Motor Symptom Rating, and Quality of Life in Those with Stage I Parkinson’s Disease Utilizing LSVT BIG. Rehabil. Res. Pract. 2017, 2017, 9871070. [Google Scholar] [CrossRef]
- Iwakura, M.; Okura, K.; Kubota, M.; Sugawara, K.; Kawagoshi, A.; Takahashi, H.; Shioya, T. Estimation of minimal clinically important difference for quadriceps and inspiratory muscle strength in older outpatients with chronic obstructive pulmonary disease: A prospective cohort study. Phys. Ther. Res. 2020, 24, 35–42. [Google Scholar] [CrossRef]
- Bohannon, R.W.; Crouch, R. Minimal clinically important difference for change in 6-minute walk test distance of adults with pathology: A systematic review. J. Eval. Clin. Pract. 2017, 23, 377–381. [Google Scholar] [CrossRef]
- Godi, M.; Arcolin, I.; Giardini, M.; Corna, S.; Schieppati, M. Responsiveness and minimal clinically important difference of the Mini-BESTest in patients with Parkinson’s disease. Gait Posture 2020, 80, 14–19. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing, v3.1.3; R Foundation for Statistical Computing: Vienna, Austria, 2015. Available online: http://www.R-project.org (accessed on 27 July 2025).
- Zieliński, G. Effect Size Guidelines for Individual and Group Differences in Physiotherapy. Arch. Phys. Med. Rehabil. 2025, 19, S0003-9993(25)00717-8. [Google Scholar] [CrossRef] [PubMed]
- Fırat, Y.E.; Turgay, T.; Soğan, S.S.; Karadeniz, P.G. Effects of LSVT-BIG via telerehabilitation on non-motor and motor symptoms and quality of life in Parkinson’s disease. Acta Neurol. Belg. 2023, 123, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Vitacca, M.; Olivares, A.; Comini, L.; Vezzadini, G.; Langella, A.; Luisa, A.; Petrolati, A.; Frigo, G.; Paneroni, M. Exercise Intolerance and Oxygen Desaturation in Patients with Parkinson’s Disease: Triggers for Respiratory Rehabilitation? Int. J. Environ. Res. Public Health 2021, 18, 12298. [Google Scholar] [CrossRef]
- Eldemir, S.; Eldemir, K.; Saygili, F.; Ozkul, C.; Yilmaz, R.; Akbostancı, M.C.; Guclu-Gunduz, A. The effects of standard and modified LSVT BIG therapy protocols on balance and gait in Parkinson’s disease: A randomized controlled trial. Brain Behav. 2024, 14, e3458. [Google Scholar] [CrossRef]
- Ebersbach, G.; Grust, U.; Ebersbach, A.; Wegner, B.; Gandor, F.; Kühn, A.A. Amplitude-oriented exercise in Parkinson’s disease: A randomized study comparing LSVT-BIG and a short training protocol. J. Neural Transm. 2015, 122, 253–256. [Google Scholar] [CrossRef]
- Mehdizadeh, M.; Fereshtehnejad, S.-M.; Landers, M.R.; Sharabiani, P.T.A.; Shati, M.; Mortazavi, S.S.; Habibi, S.A.H.; Mansoori, K.; Meimandi, M.; Nodehi, Z.; et al. Responsiveness of the mini-balance evaluation systems test, dynamic gait index, Berg balance scale, and performance-oriented mobility assessment in parkinson’s disease. Sci. Rep. 2025, 15, 21126. [Google Scholar] [CrossRef]
- Walsh, S.F.; Balster, C.; Chandler, A.; Brown, J.; Boehler, M.; O’rEar, S. LSVT BIG and long-term retention of functional gains in individuals with Parkinson’s disease. Physiother. Theory Pract. 2022, 38, 629–636. [Google Scholar] [CrossRef]
- Zhang, F.; Zhong, Y.; Qin, Z.; Li, X.; Wang, W. Effect of Muscle Training on Dyspnea in Patients with Chronic Obstructive Pulmonary Disease: A Meta-Analysis of Randomized Controlled Trials. Medicine 2021, 100, e24930. [Google Scholar] [CrossRef]
- Janaudis-Ferreira, T.; Hill, K.; Goldstein, R.S.; Robles-Ribeiro, P.; Beauchamp, M.K.; Dolmage, T.E.; Wadell, K.; Brooks, D. Resistance arm training in patients with COPD: A Randomized Controlled Trial. Chest 2011, 139, 151–158. [Google Scholar] [CrossRef]
- Couser, J.I.; Martinez, F.J.; Celli, B.R. Respiratory Response and Ventilatory Muscle Recruitment During Arm Elevation in Normal Subjects. Chest 1992, 101, 336–340. [Google Scholar] [CrossRef]
- Templeman, L.; Roberts, F. Effectiveness of Expiratory Muscle Strength Training on Expiratory Strength, Pulmonary Function and Cough in the Adult Population: A Systematic Review. Physiotherapy 2020, 106, 43–51. [Google Scholar] [CrossRef]
- Emirza, C.; Aslan, G.K.; Kilinc, A.A.; Cokugras, H. Effect of Expiratory Muscle Training on Peak Cough Flow in Children and Adolescents with Cystic Fibrosis: A Randomized Controlled Trial. Pediatr. Pulmonol. 2021, 56, 939–947. [Google Scholar] [CrossRef] [PubMed]
- Fontana, G.A.; Pantaleo, T.; Lavorini, F.; Benvenuti, F.; Gangemi, S. Defective motor control of coughing in Parkinson’s disease. Am. J. Respir. Crit. Care Med. 1998, 158, 458–464. [Google Scholar] [CrossRef] [PubMed]
- Van de Wetering-van Dongen, V.A.; Kalf, J.G.; van der Wees, P.J.; Bloem, B.R.; Nijkrake, M.J. The Effects of Respiratory Training in Parkinson’s Disease: A Systematic Review. J. Park. Dis. 2020, 10, 1315–1333. [Google Scholar] [CrossRef] [PubMed]
- Reyes, A.; Castillo, A.; Castillo, J.; Cornejo, I. The effects of respiratory muscle training on peak cough flow in patients with Parkinson’s disease: A randomized controlled study. Clin. Rehabil. 2018, 32, 1317–1327. [Google Scholar] [CrossRef]
- Reyes, A.; Castillo, A.; Castillo, J. Effects of Expiratory Muscle Training and Air Stacking on Peak Cough Flow in Individuals with Parkinson’s Disease. Lung 2020, 198, 207–211. [Google Scholar] [CrossRef]
- Cools, C.I.; Kotz, S.A.; Bloem, B.R.; Duits, A.A.; de Vries, N.M. Motivation Matters: Elucidating Factors Driving Exercise in People with Parkinson’s Disease. Phys. Ther. 2025, 105, pzaf048. [Google Scholar] [CrossRef]
| All (n = 38) | LSVT BIG (n = 19) | SC (n = 19) | p | |
|---|---|---|---|---|
| Age, years | 72 (68–76) | 71 (70–76) | 73 (67–76) | 0.2125 |
| Male, n (%) | 25 (66%) | 11 (58%) | 14 (74%) | 0.4950 |
| BMI, Kg/m2 | 26.7 (4.4) | 26.6 (5.0) | 26.8 (3.8) | 0.6827 |
| Disease duration PD, years | 5 (3–8) | 7 (4–9) | 4 (2–8) | 0.2125 |
| Comorbidities | ||||
| COPD yes, n (%) | 8 (8) | 2 (11) | 1 (5) | 1.0000 |
| CHF yes, n (%) | 7 (18) | 4 (21) | 3 (16) | 1.0000 |
| Diabetes yes, n (%) | 7 (18) | 4 (21) | 3 (16) | 1.0000 |
| Hypertension yes, n (%) | 20 (53) | 11 (58) | 9 (47) | 0.7459 |
| Others yes, n (%) | 10 (26) | 4 (21) | 6 (32) | 0.7141 |
| Levodopa, yes n (%) | 36 (95) | 19 (100) | 17 (89) | 0.4865 |
| Dopamine agonist, yes n (%) | 23 (61) | 13 (68) | 10 (53) | 0.5076 |
| IMAO B, yes n (%) | 18 (47) | 9 (47) | 9 (47) | 1.0000 |
| MMSE, score | 28.4 (26.0–30.0) | 28.3 (26.2–29.9) | 29.0 (25.9–30.0) | 0.6749 |
| HY, score | 2.0 (2.0–2.5) | 2.0 (2.0–2.5) | 2.0 (2.0–2.5) | 0.8586 |
| UPDRSII, score | 6 (3–9) | 7 (4–9) | 5 (3–9) | 0.4112 |
| UPDRSIII, score | 11 (8–16) | 10 (8–16) | 11 (9–17) | 0.9767 |
| Gait speed #, m/s | 1.3 (1.2–1.6) | 1.3 (1.2–1.7) | 1.3 (1.2–1.6) | 0.9767 |
| FOG-Q, score | 3.5 (0.3- 9.8) | 4.0 (1.5–10.5) | 2.0 (0.0–9.0) | 0.4169 |
| MIP, cm H2O | 47 (18) | 43 (15) | 51 (21) | 0.2119 |
| MIP, % pred | 52 (19) | 50 (16) | 54 (21) | 0.4831 |
| MEP, cm H2O | 68 (22) | 68 (22) | 67 (22) | 0.8829 |
| MEP, % pred | 40 (12) | 42 (13) | 38 (11) | 0.3282 |
| PCEF, L/min | 274 (130) | 246 (104) | 301 (150) | 0.1955 |
| Berg Balance, score | 52.0 (49.0–54.0) | 50.0 (48.0–53.5) | 53.0 (50.0–54.0) | 0.5184 |
| MB, score | 20.3 (4.0) | 20.2 (3.7) | 20.3 (4.5) | 0.9373 |
| MB-TUG, s | 9.3 (7.6–10.4) | 9.2 (7.4–10.4) | 8.3 (7.8–10.5) | 0.3606 |
| MB-TUG cog, s | 11.7 (3.9) | 11.5 (3.6) | 11.9 (4.2) | 0.7604 |
| 5 STS, s | 14.2 (3.8) | 13.9 (4.0) | 14.4 (3.7) | 0.6842 |
| 6MWT, meters | 367 (103) | 370 (106) | 365 (104) | 0.8776 |
| 6MWT, % pred | 82 (68–92) | 83 (69–90) | 79 (66–92) | 0.8491 |
| 6MWT, SatO2 mean, % | 97 (97–98) | 97 (96–98) | 97 (96–98) | 0.7901 |
| 6MWT, Exercise DES, n (%) | 2 (5.3) | 1 (5.3) | 1 (5.3) | 1.0000 |
| 6MWT, Borg Fatigue pre, score | 1 (0–1) | 0 (0–1) | 1 (0–1.5) | 0.8289 |
| 6MWT, Borg Fatigue post, score | 4.5 (3–6) | 4 (2.5–6) | 5 (3–7) | 0.2305 |
| UULEX time, s | 278 (120) | 259 (118) | 297 (121) | 0.3295 |
| UULEX level, score | 5 (2) | 5 (2) | 5 (2) | 0.3843 |
| UULEX, SatO2 mean, % | 97 (96–98) | 97 (96–98) | 97 (97–98) | 0.7010 |
| UULEX Exercise DES, n (%) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1.0000 |
| PFS, score | 2.4 (0.8) | 2.4 (0.7) | 2.4 (0.8) | 0.8949 |
| PDQ39-SI, score% | 19.5 (12.3– 26.8) | 19.8 (17.0–24.7) | 17.9 (10.3–31.4) | 0.5395 |
| Variables | LSVT BIG (n = 19) | SC (n = 19) | p | Effect Size (CI95%) |
|---|---|---|---|---|
| UPDRSII, score | −2.0 (−3.0; −0.5) | −1.0 (−3.5; 0.0) | 0.7240 | −0.059 (−0.372; 0.266) |
| UPDRSIII, score | −2.9 (5.2) | −2.9 (4.4) | 1.000 | 0 (−0.636; 0.636) |
| Gait speed #, m/s | 0.15 (0.03; 0.29) | 0.02 (−0.04; 0.12) | 0.0295 * | 0.355 (0.040; 0.606) |
| FOG-Q, score | −1.0 (−3.0; 0.0) | 0.0 (−3.0; 0.0) | 0.3571 | −0.149 (−0.448; 0.179) |
| MIP, cm H2O | 11 (5; 17) | 2 (−2; 11) | 0.0877 ° | 0.358 (0.043; 0.608) |
| MEP, cm H2O | 9 (3; 15) | 2 (−3; 13) | 0.1561 | 0.232 (−0.095; 0.514) |
| PCEF, L/min | 48 (17; 79) | 17 (−7; 43) | 0.0343 * | 0.346 (0.029; 0.599) |
| Berg Balance, score | 0.0 (−4.2; 1.0) | −1.3 (−5.1; 5.4) | 0.0933 ° | 0.270 (−0.054; 0.543) |
| MB, score | 3.9 (2.4) | 1.8 (2.7) | 0.0129 * | 0.822 (0.153; 1.480) |
| MB-TUG, s | −1.5 (−2.1; −0.6) | −0.9 (−1.3: −0.2) | 0.1217 | −0.253 (−0.530; 0.072) |
| MB-TUG cog, s | −1.6 (−3.7 −0.4) | −0.9 (−1.8; 0.0) | 0.1025 | −0.268 (−0.541; 0.057) |
| 5 STS, s | −2.0 (−3.1; −0.9) | −0.8 (−2.4; 0.9) | 0.0876 ° | −0.279 (−0.550; 0.044) |
| 6MWT, meters | 60 (35; 83) | 5 (−3; 54) | 0.0113 * | 0.412 (0.106; 0.647) |
| 6MWT, SatO2 mean, % | 0.0 (0.0; 0.5) | 0.0 (−1.0; 0.5) | 0.1428 | 0.212 (−0.112; 0.501) |
| UULEX level, score | 1.0 (1.0; 2.5) | 0.0 (0.0; 1.0) | 0.0002 ** | 0.561 (0.294; 0.747) |
| UULEX time, s | 71 (52; 163) | 9 (−2; 47) | 0.0004 ** | 0.573 (0.310; 0.755) |
| UULEX, SatO2 mean, % | 1.0 (0.8) | 0.3 (1.2) | 0.0481 * | 0.686 (0.027; 1.337) |
| PFS, score | 0.0 (0.7) | −0.3 (0.6) | 0.0836 ° | 0.460 (−0.188; 1.102) |
| PDQ39 SI, score% | 0.0 (−4.2; 1.0) | −1.3 (−5.1; 5.4) | 0.8839 | −0.026 (−0.296; 0.343) |
| Variables | LSVT BIG (n = 19) | SC (n = 19) | OR | 95% CI | p |
|---|---|---|---|---|---|
| Delta T1-T0 UPDRS III ≤−2.5 points, n (%) | 9 (47%) | 9 (47%) | 1.00 | 0.23; 4.28 | 0.3269 |
| Delta T1-T0 Gait speed # ≥0.16 m/s, n (%) | 9 (50%) | 3 (16%) | 4.60 | 0.87; 32.89 | 0.0789 ° |
| Delta T1-T0 MIP ≥17.2 cmH2O, n (%) | 5 (26%) | 4 (21%) | 1.33 | 0.23; 8.18 | 1.0000 |
| Delta T1-T0 MB ≥4 points, n (%) | 12 (63%) | 3 (16%) | 8.54 | 1.63; 62.54 | 0.0069 ** |
| Delta T1-T0 6MWD ≥30.5 m, n (%) | 14 (74%) | 8 (42%) | 3.71 | 0.82; 19.17 | 0.0991 ° |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Estatico, F.; Olivares, A.; Comini, L.; Paneroni, M.; Vitacca, M.; Tavolazzi, F.; Maffi, G.; Forlani, C.; Vezzadini, G. In-Hospital LSVT BIG Training Versus Structured Rehabilitation Treatment in Parkinson’s Disease: Feasibility and Primary Evaluation on Functional and Respiratory Outcomes. Appl. Sci. 2025, 15, 10611. https://doi.org/10.3390/app151910611
Estatico F, Olivares A, Comini L, Paneroni M, Vitacca M, Tavolazzi F, Maffi G, Forlani C, Vezzadini G. In-Hospital LSVT BIG Training Versus Structured Rehabilitation Treatment in Parkinson’s Disease: Feasibility and Primary Evaluation on Functional and Respiratory Outcomes. Applied Sciences. 2025; 15(19):10611. https://doi.org/10.3390/app151910611
Chicago/Turabian StyleEstatico, Francesco, Adriana Olivares, Laura Comini, Mara Paneroni, Michele Vitacca, Francesca Tavolazzi, Giovanna Maffi, Chiara Forlani, and Giuliana Vezzadini. 2025. "In-Hospital LSVT BIG Training Versus Structured Rehabilitation Treatment in Parkinson’s Disease: Feasibility and Primary Evaluation on Functional and Respiratory Outcomes" Applied Sciences 15, no. 19: 10611. https://doi.org/10.3390/app151910611
APA StyleEstatico, F., Olivares, A., Comini, L., Paneroni, M., Vitacca, M., Tavolazzi, F., Maffi, G., Forlani, C., & Vezzadini, G. (2025). In-Hospital LSVT BIG Training Versus Structured Rehabilitation Treatment in Parkinson’s Disease: Feasibility and Primary Evaluation on Functional and Respiratory Outcomes. Applied Sciences, 15(19), 10611. https://doi.org/10.3390/app151910611







