1. Introduction
Stroke is a major cause of death and disability worldwide, affecting up to one in five people in some developed countries and nearly one in two in certain developing areas [
1]. Epidemiological evidence indicates that stroke is the second leading cause of death and the third leading cause of disability worldwide [
2]. Post-stroke disability is documented in up to 80% of patients, usually manifesting as hemiparesis, sensory deficits, compromised balance, and gait disturbances [
3,
4]. Among the musculoskeletal issues linked to hemiplegia, shoulder pain is widespread and usually develops within the first few weeks to six months after a stroke, affecting 34% to 84% of patients [
5,
6]. The leading causes include rotator cuff injuries, glenohumeral subluxation, muscular weakness or spasticity, and neurological problems [
7]. Prolonged muscle weakness in the shoulder girdle often leads to persistent pain and glenohumeral subluxation in 17–81% of patients with hemiplegia following a stroke [
8,
9]. Shoulder subluxation is characterized by an increased space between the humerus and the acromion, altering the mechanics and alignment of the joint [
10]. It may present with symptoms and complications such as pain, ligament tears, muscle tendinitis, and axillary nerve injury, among others [
8,
11].
For the prevention of shoulder subluxation and associated pain after stroke, various interventions have been utilized. Specifically, functional electrical stimulation (FES) and neuromuscular electrical stimulation (NMES) have shown significant reductions in subluxation, particularly in the acute and subacute phases, although their impact on functional recovery remains unclear [
12,
13]. Kinesiology taping has demonstrated statistically significant benefits compared to sham taping, reducing sub-luxation distance, improving range of motion, and decreasing pain [
14]. Repetitive peripheral magnetic stimulation (rPMS) has been associated with a smaller increase in the acromiohumeral interval compared to controls [
15], while electroacupuncture has been found to reduce pain without affecting subluxation [
16]. In addition, robot-assisted therapy may improve stability, muscle strength, and pain reduction through repetitive and controlled movements, although available data are limited and larger-scale studies are needed [
17]. Finally, shoulder orthoses represent a useful supportive intervention, as they stabilize the glenohumeral joint and reduce subluxation distance while applied, counteracting the effects of gravity and preventing further soft tissue injury; however, their effects do not appear to persist in the long term, and their role is primarily preventive [
12,
13,
18]. Despite these benefits, the literature also reports potential drawbacks, such as restricted range of motion, discomfort, exacerbation of spasticity, skin irritation, and possible muscle weakness or adhesive capsulitis [
19,
20,
21], which should be taken into account when selecting the most appropriate orthosis. Further details regarding the advantages and disadvantages of utilizing shoulder orthoses in stroke patients are provided in the
Appendix A.
Shoulder subluxation, apart from musculoskeletal impairments, can adversely affect posture, balance, and gait symmetry [
22,
23,
24]. Although the primary purpose of shoulder orthoses is to support and align the shoulder girdle, preventing subluxation and reducing pain [
5,
13,
25], some research suggests that shoulder orthoses may also enhance static and dynamic balance, as well as improve gait performance [
2,
19,
24,
26].
The relationship between shoulder stability and functional mobility can be partially elucidated by the interconnected myofascial and motor pathways that link the trunk, pelvis, and extremities, facilitating coordinated movement during gait [
27,
28]. In hemiplegic patients, diminished upper limb mobility has been associated with decreased pelvic and trunk rotation, thereby disrupting the normal gait pattern [
29,
30]. Moreover, impaired proprioception, asymmetrical weight distribution, displacement of the center of gravity, and trunk instability increase the risk of falls and impede functional independence [
31,
32,
33,
34].
The international literature encompasses research investigations evaluating the effects of shoulder orthoses on balance, gait parameters, and energy efficiency. Specifically, statistically significant improvements in static and dynamic balance [
22,
35] were reported in patients with hemiplegia following a stroke during orthosis use. In contrast, studies by Sohn et al. [
36] and Van Bladel et al. [
37] found no significant differences in static and dynamic balance between conditions with and without an orthosis. Regarding gait parameters, some studies have reported slight increases in gait speed with the use of orthoses [
38,
39], while others such as observed minimal or no significant differences [
24,
37,
40]. Young-In et al. [
40] documented increases in stride and step lengths, especially on the affected limb. Hesse et al. [
19] observed enhancements in stance phase duration and gait symmetry. Yavuzer et al. [
24] reported increased step length and improved stability during the use of orthoses. Conversely, Van Bladel et al. [
37] and Hesse et al. [
19] did not identify significant alterations in variables such as stance time, swing phase, double support phase, and swing symmetry ratio. At the kinematic level, Yavuzer et al. [
24] documented reductions in pelvic excursion across all planes and increases in peak vertical ground reaction force during orthosis use, thereby indicating enhanced postural control and loading on the affected limb. Furthermore, Hwang et al. [
26] observed improvements in pelvic rotation and knee joint positioning during the stance phase, although some of these findings did not attain statistical significance. Han et al. [
38] demonstrated reductions in heart rate, oxygen consumption, and oxygen cost when the orthosis was worn. In contrast, Jeong et al. [
39] found no significant changes in most conditions, except for one scenario where a small but statistically significant reduction in oxygen consumption and cost was observed. Finally, findings from systematic reviews and meta-analyses suggest that although shoulder orthoses may enhance gait velocity, their impact on balance remains uncertain or inconsistent [
41,
42].
Most studies have focused on basic motor parameters, such as straight-line walking [
19,
24,
37,
38,
39,
40], often neglecting functional daily tasks like sit-to-stand transfers, directional changes, or obstacle avoidance. Additionally, few have specifically examined the subacute phase of stroke rehabilitation, [
19,
24,
35,
36] a vital period when patients are at a higher risk of falls and functional decline. This study aimed to examine the impact of a humeral cuff-type shoulder orthosis on functional mobility and dynamic balance in patients with hemiplegia during the subacute rehabilitation phase. It specifically investigated whether the use of the orthosis can improve functional activity performance and contribute to fall prevention. The primary hypothesis posited that the utilization of a shoulder orthosis on the hemiplegic arm would yield a positive influence on mobility and balance among subacute post-stroke patients. Additionally, it was hypothesized that functional mobility and dynamic balance would be related to scores on the Functional Ambulation Category (FAC) scale and the Brunnstrom recovery stage for the upper limb.
3. Results
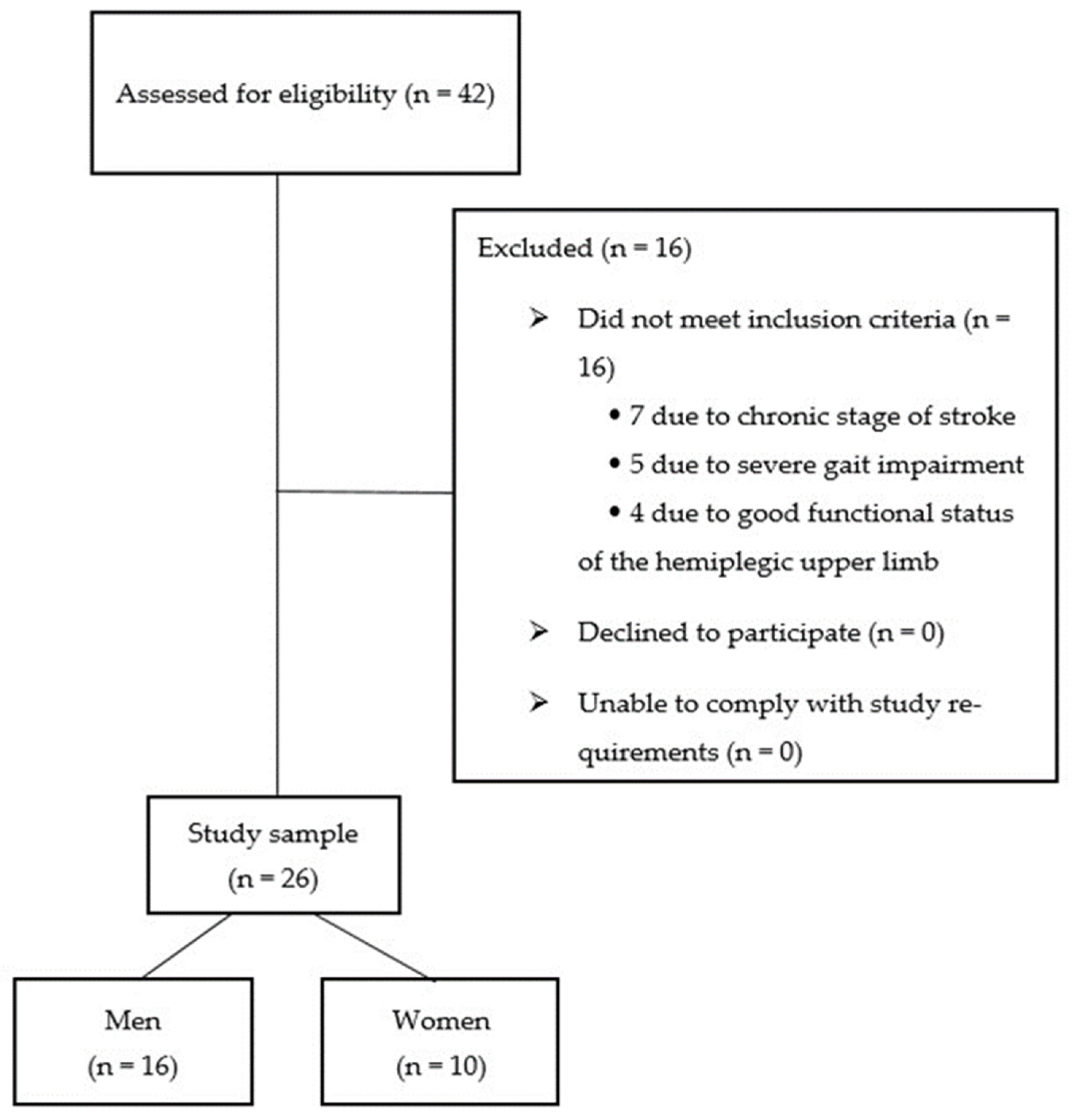
The inclusion of participants to the study, according to the set inclusion and exclusion criteria, is shown in
Figure 2.
The participants’ demographic, stroke type and their functional level characteristics are detailed in
Table 1.
The results of the normality tests for the three functional test variables (TUG, FRT, mFSST), analyzed with and without a splint are shown in
Table 2. The tests used the Shapiro–Wilk method, which is more suitable for small samples (n < 50). The non-normal distribution of TUG and mFSST justifies the use of non-parametric methods (Wilcoxon Signed Ranks Test), whereas the FRT’s normality permitted a parametric paired t-test t-test).
We verified that there were no significant differences (p < 0.05) for the outcome measures of the study (TUG, FRT, and MFSST) both before and after the application of the brace, neither between male and female participants nor for the hemiplegia side with the independent samples t-test (FRT) or the Mann–Whitney U test (TUG and MFSST).
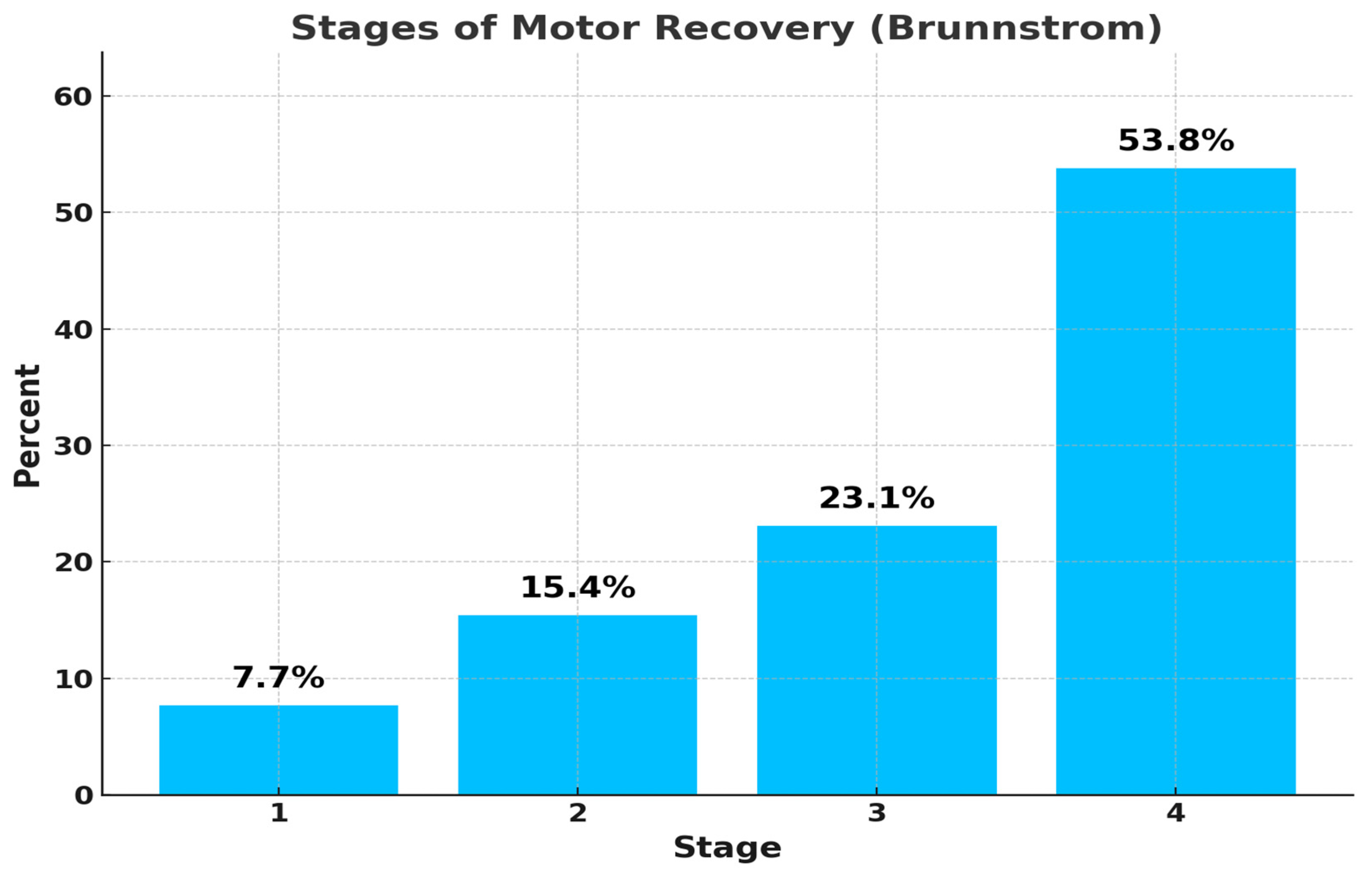
Motor recovery of the upper limb, evaluated through the Brunnstrom method, showed an average score of 3.23 (SD = 0.99), reflecting partial motor control recovery, with study stages ranging from 1 to 4. Most participants (53.8%) reached stage 4, followed by 23.1% in stage 3, with smaller groups in stages 2 (15.4%) and 1 (7.7%) (
Figure 3).
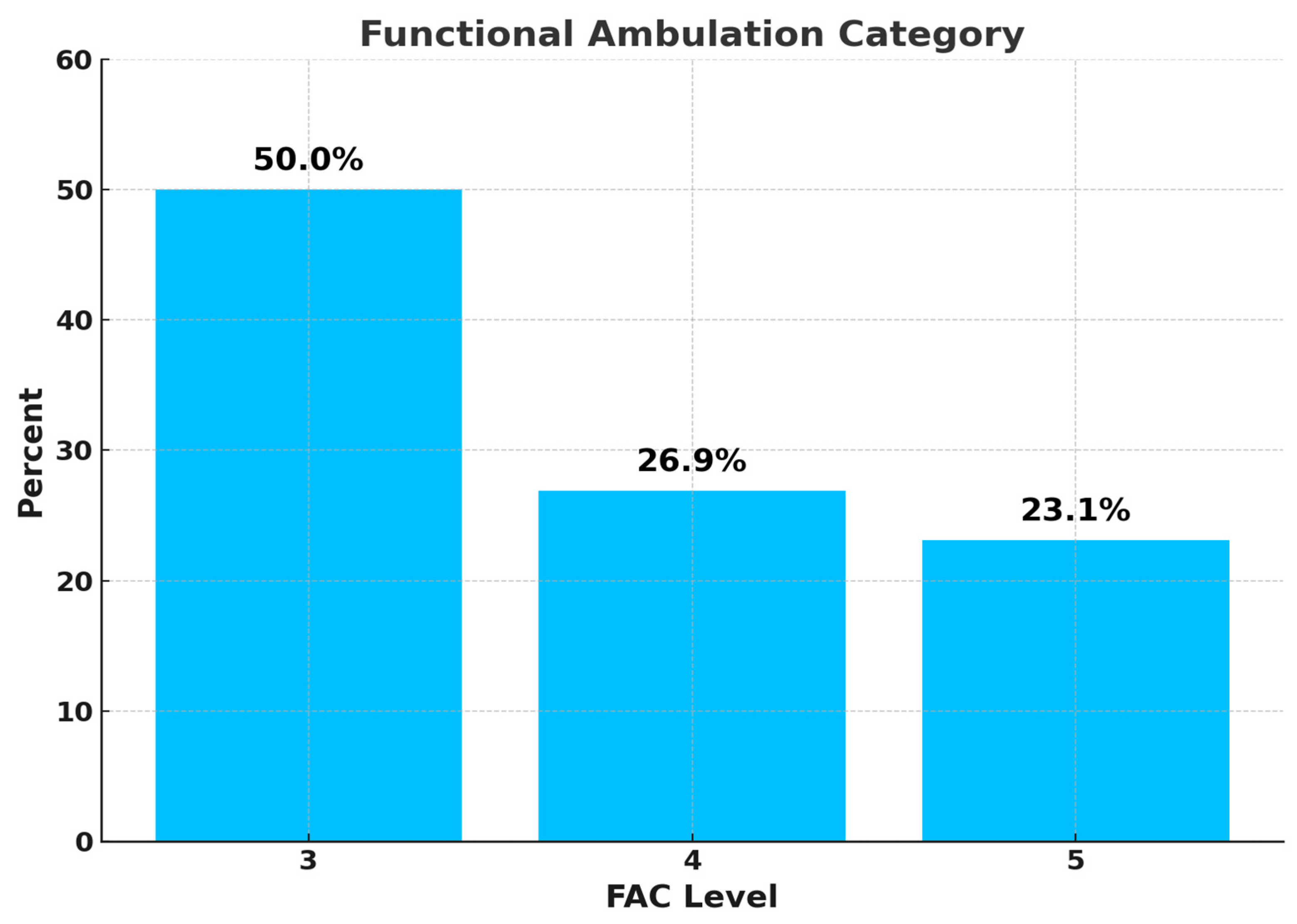
The participants had an average FAC score of 3.73 (SD = 0.83), with range 3–5 across study categories. This suggests most could walk independently on level ground, with some also able to navigate more challenging terrains such as stairs or uneven surfaces. Half of the participants (50%) were classified as FAC 3, meaning they required supervision to walk. About 26.9% were classified as FAC 4, indicating they could walk independently only on level surfaces. The remaining 23.1% fell into FAC 5, demonstrating independence across all environments, as shown in
Figure 4.
Functional Reach Test
The dependent samples t-test showed a statistically significant difference in the Functional Reach Test between using and not using the splint (
p = 0.049, t = 2.070), with a moderate effect size of d = 0.406 (
Table 3 and
Figure 5).
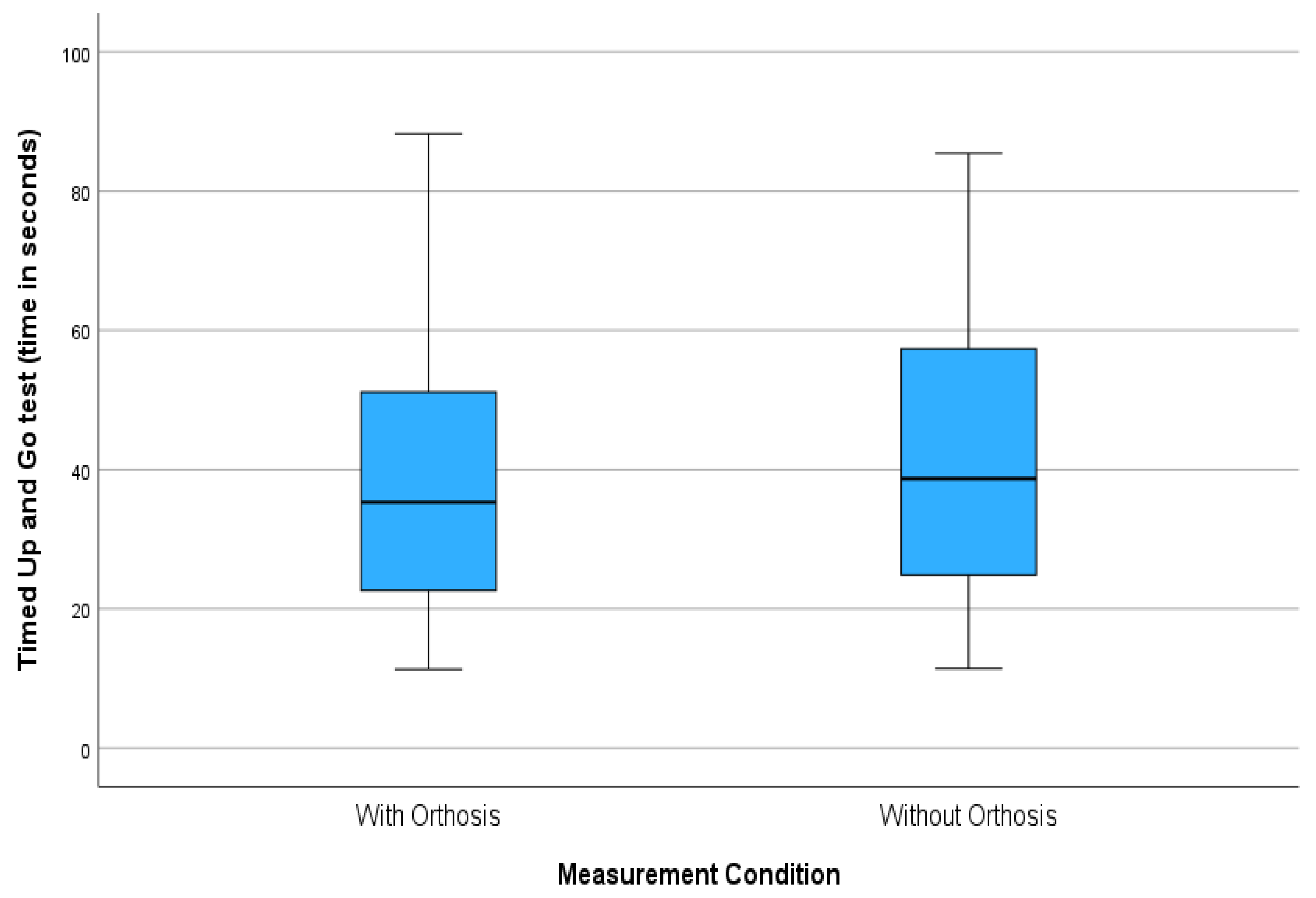
Timed Up and Go
The Wilcoxon Signed Ranks Test, a non-parametric method, was employed to compare participants’ performance on the TUG test with and without an upper limb splint. The analysis revealed a statistically significant difference in results when the splint was used [
31], with a
p-value of 0.023 and a z-score of −2.273. The effect size was d = 0.446, suggesting a moderate impact. It was noted that the median time was shorter when wearing the splint, Md = 35.32 s, compared to Md = 38.70 s without it. The difference in median times was −3.38 s, as shown in
Table 4 and
Figure 6.
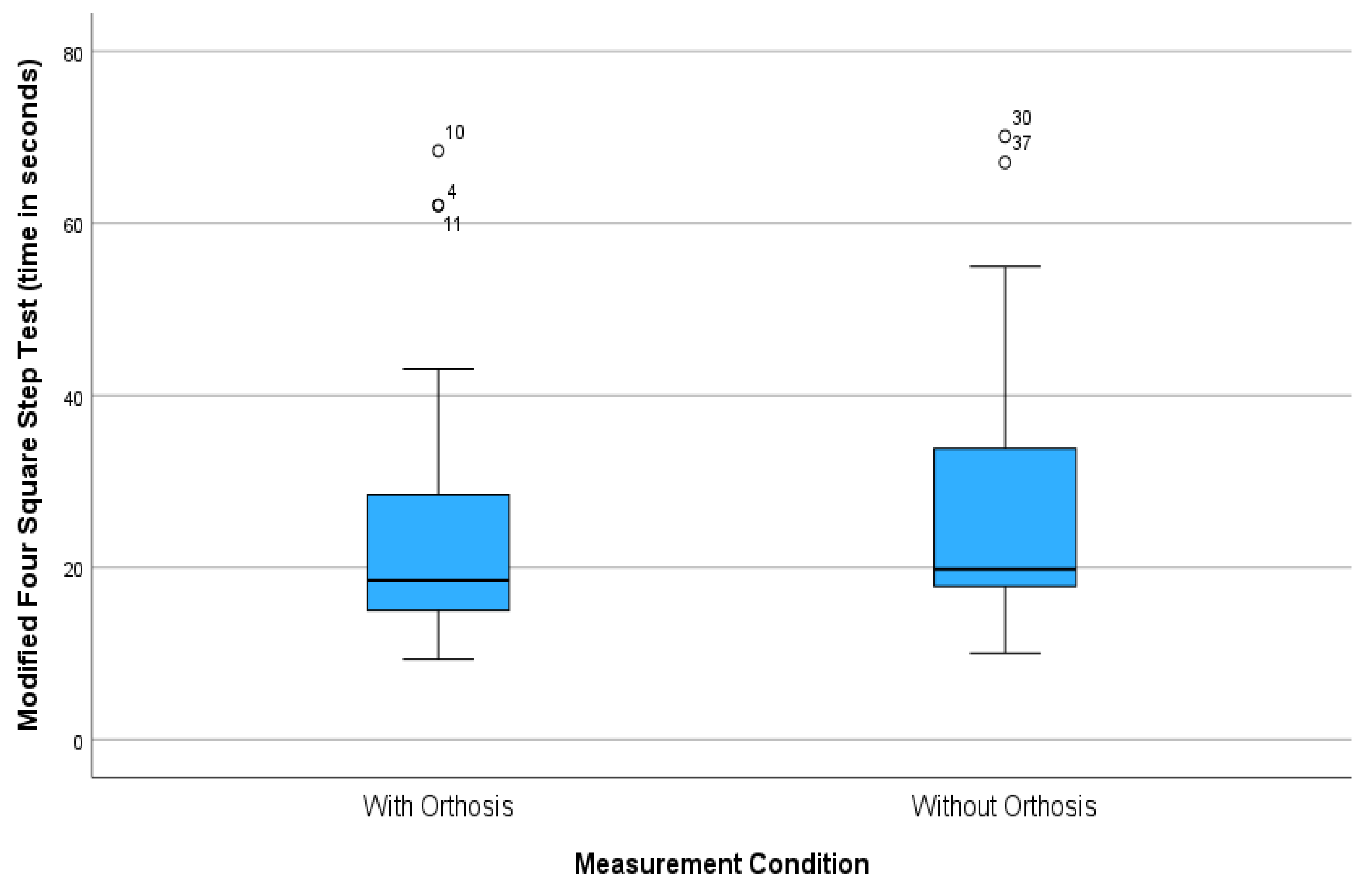
Modified Four Square Step Test
The Wilcoxon Signed Ranks Test showed a statistically significant difference between using and not using the splint in the Modified Four-Square Step Test (
p = 0.004, z = −2.883). The effect size was 0.565, representing a moderate intervention impact (
Table 4 and
Figure 7).
The FAC scale demonstrated strong negative correlations with performance on both the TUG and mFSST tests, whether with or without a splint (ρ > −0.807,
p < 0.001). This indicates that higher FAC scores, reflecting better functional walking ability, are linked to quicker and more effective motor performance in these assessments. Additionally, there was a positive correlation with the FRT (ρ = 0.603,
p = 0.001 with a splint and ρ = 0.500,
p = 0.009 without a splint), suggesting that patients who are more independent in walking also tend to have better forward displacement capabilities (
Table 5).
The analysis of the correlation between test performance and the stages of motor recovery of the upper limb according to Brunnstrom shows strong negative correlations with TUG both with (ρ = −0.592,
p = 0.001) and without a splint (ρ = −0.575,
p = 0.002), as well as with mFSST with (ρ = −0.632,
p <0.001) and without a splint (ρ = −0.665,
p < 0.001). These findings confirm that higher motor control stages are linked to better functional mobility and balance. Additionally, Brunnstrom’s correlation with FRT was positive and statistically significant, with (ρ = 0.556,
p = 0.003) with a splint and (ρ = 392,
p = 0.047) without a splint, supporting the idea that upper limb stability influences forward balance. (
Table 6).
4. Discussion
The present study examined the impact of shoulder orthosis utilization on functional mobility and dynamic balance in patients with hemiplegia during the subacute post-stroke phase. Twenty-six patients participated, with a mean age of 68.2 years, an upper limb Brunnstrom motor recovery stage of 3.23, and a FAC score of 3.73. The findings demonstrated a statistically significant improvement in both functional mobility and dynamic balance following the application of the orthosis, with a moderate effect size. However, the difference in the mean score for the FRT without orthosis and with orthosis conditions was 0.67 cm, indicating that it is not clinically meaningful. Similarly, the difference in the median for the mFSST was 1.39 s, suggesting a lack of clinical significance. Conversely, the difference in the median for the TUG test was 3.38 s, which may imply a clinically meaningful outcome. Additionally, significant correlations were observed between Brunnstrom motor recovery stages and FAC scores with performance in the functional assessments both with and without the orthosis-except for the FRT without orthosis.
In this study, the use of a shoulder orthosis significantly enhanced participants’ performance in dynamic balance, as measured by the FRT and mFSST. The application of the arm splint provided participants with a sense of security, reduced uncontrolled arm movements, and served as a reminder of the affected limb, thereby acting as a feedback mechanism that supported posture correction. These results are consistent with those of Acar et al. [
22], who examined the effect of shoulder orthosis on static and dynamic balance in 26 subacute stroke patients at Brunnstrom stage 3, similar to the present study. Similarly, Şahin et al. [
35] found a positive impact of shoulder orthosis on static balance in 23 stroke patients at Brunnstrom stages 1–4.
In contrast, the findings of Sohn et al. [
36] and Van Bladel et al. [
37] do not support the present results, as these researchers reported no significant effect of orthosis use on balance. This discrepancy may be attributed to differences in rehabilitation stage and the types of orthoses used, as well as the small sample size (10 participants) in the study by Van Bladel et al. [
37]. Specifically, Sohn et al. [
36] and Van Bladel et al. [
37] reported mean post-stroke durations of 2.12 and 6.08 months, respectively, while in the present study the mean duration was 4.62 months. Furthermore, Sohn et al. [
36] used Bobath and simple arm slings, while Van Bladel et al. [
37] applied Actimove sling and Shoulderlift devices. These orthoses differ from the shoulder-hugging orthosis used in the present study in terms of functional and structural characteristics, including the degree of shoulder stabilization, forearm support, trunk attachment, and allowance for upper limb movement. More specifically, simple arm splints may incorporate contralateral or bilateral shoulder straps to support the forearm, thereby ensuring optimal correction of subluxation and preventing the flaccid arm from striking the body during activities. However, immobilizing the arm in flexion, adduction, and internal rotation can lead to an increase in flexor tone, promote synergistic movement patterns, and impede arm swing during ambulation. [
36,
55]. The most frequently utilized Bobath sling comprises foam rolls positioned in the axillae on both sides, secured with shoulder straps that neither induce flexor posturing nor restrict arm movement. However, it offers inadequate correction of subluxation and may additionally result in lateral displacement of the humeral head, stressing the soft tissues [
36,
56]. A humeral cuff-type shoulder orthosis, such as the Reh4mat AM-SOB utilized in the present study, elevates the humeral head from above, stabilizes the shoulder joint, and offers the most effective overall reduction in subluxation, while preventing synergy patterns [
55]. Notably, the shoulder-hugging orthosis allows the patient to maintain balance during weight-shifting in ambulation without significantly restricting upper limb mobility during functional activities [
54,
56]. The findings of this study also contrast with the systematic review and meta-analysis by Lin et al. [
41], who found no significant differences in balance between stroke patients using or not using an orthosis. However, the authors noted a clear trend favoring better performance among orthosis users.
The current results also revealed a statistically significant improvement in functional mobility, as measured by the TUG test, following the use of the shoulder-hugging orthosis. This supports the hypothesis that stabilization and alignment of the shoulder girdle may contribute to improved motor performance in stroke patients. The TUG test evaluates functional mobility involving sit-to-stand and stand-to-sit transitions, walking, and directional changes, which all require sufficient balance [
49,
57]. A review of the literature revealed no prior studies evaluating the impact of shoulder orthosis on functional tests such as the TUG in post-stroke patients. However, several studies have highlighted the positive effects of shoulder orthoses on various aspects of functional mobility, including gait speed, endurance, and balance [
22,
24,
35,
38,
39].
During the performance of functional activities, the upper limb in stroke patients typically adopts a relaxed and downward position due to hemiplegia or hemiparesis [
33], resulting in increased resistance torque and displacement of the center of mass toward the affected side. Consequently, the mechanical load on trunk and lower limb muscles increases as they attempt to compensate for asymmetric loading. As a result, postural stability is reduced and energy demands for executing simple movements increase [
31,
32]. The use of a shoulder orthosis helps support the weight of the upper limb and may improve scapular and arm alignment, bringing the upper limb closer to the body’s center of mass [
40,
58]. Additionally, by stabilizing the upper limb in a more optimal position, the body’s postural and balance systems can function more symmetrically, reducing the need for increased trunk muscle activation [
19,
24]. Therefore, patients with limited muscle strength may be able to perform equivalent motor tasks with reduced muscular effort, improved balance, functional ability, and endurance [
39,
40].
The present study solely evaluated the immediate effects of an arm sling on functional mobility and dynamic balance. Extending the duration of the intervention may afford patients an opportunity to mitigate discomfort caused by sling application and to adapt their balance and gait, which may have changed abruptly under the current experimental conditions [
42]. Specifically, since patients often have limited awareness of the hemiplegic side, arm slings could serve as a feedback mechanism, reminding patients of their affected limb and facilitating postural adjustments [
22,
24]. Furthermore, an arm sling that permits functional movement might reduce asymmetry in arm swing, thereby increasing weight-bearing on the weaker leg during ambulation. Nevertheless, incorrect usage of the arm sling or selection of a sling that restricts arm movement could lead to the development of soft-tissue contractures, promote undesired synergistic flexion patterns, diminish activation of shoulder muscles, and potentially result in the non-use of the affected arm [
25,
36,
55].
While shoulder orthoses may offer essential advantages in mitigating subluxation, enhancing shoulder girdle alignment, and supporting functional performance, the literature also documents several disadvantages and adverse effects [
13]. According to Hesse et al. [
19], some patients and therapists have observed that orthosis use may restrict shoulder range of motion, worsen elbow flexor spasticity, or induce skin irritation and discomfort, particularly during extended wear. Furthermore, fewer than 50% of participants in their randomized controlled trial reported pain alleviation, indicating that analgesic benefits are not universally experienced. Similarly, the systematic review by Nadler and Pauls [
20] underscores potential risks such as adhesive capsulitis, muscular weakness resulting from diminished activation, and cutaneous irritation due to prolonged contact with materials. These issues, coupled with possible challenges in fitting and adjusting the device, can impede patient acceptance and adherence. Van Bladel et al. [
21] additionally observed that inadequate fit or poor ventilation in certain orthotic designs may lead to increased perspiration and odor, thereby negatively impacting comfort and tolerability. Moreover, some models may be difficult to don or remove without assistance, thereby reducing the patient’s independence. Additional details regarding the comparison of the benefits and drawbacks of employing shoulder orthoses in stroke patients are presented in the
Appendix A.
Overall, shoulder orthoses offer notable clinical benefits, but selecting the appropriate device requires evaluating more than just its therapeutic effectiveness. Comfort, tolerability, and the risk of secondary issues must also be considered. Furthermore, choosing a shoulder orthosis should be personalized, taking into account factors such as the stage of stroke recovery, the mobility of the hemiplegic arm, overall functional ability, and balancing the advantages with potential drawbacks of each option [
20,
41].
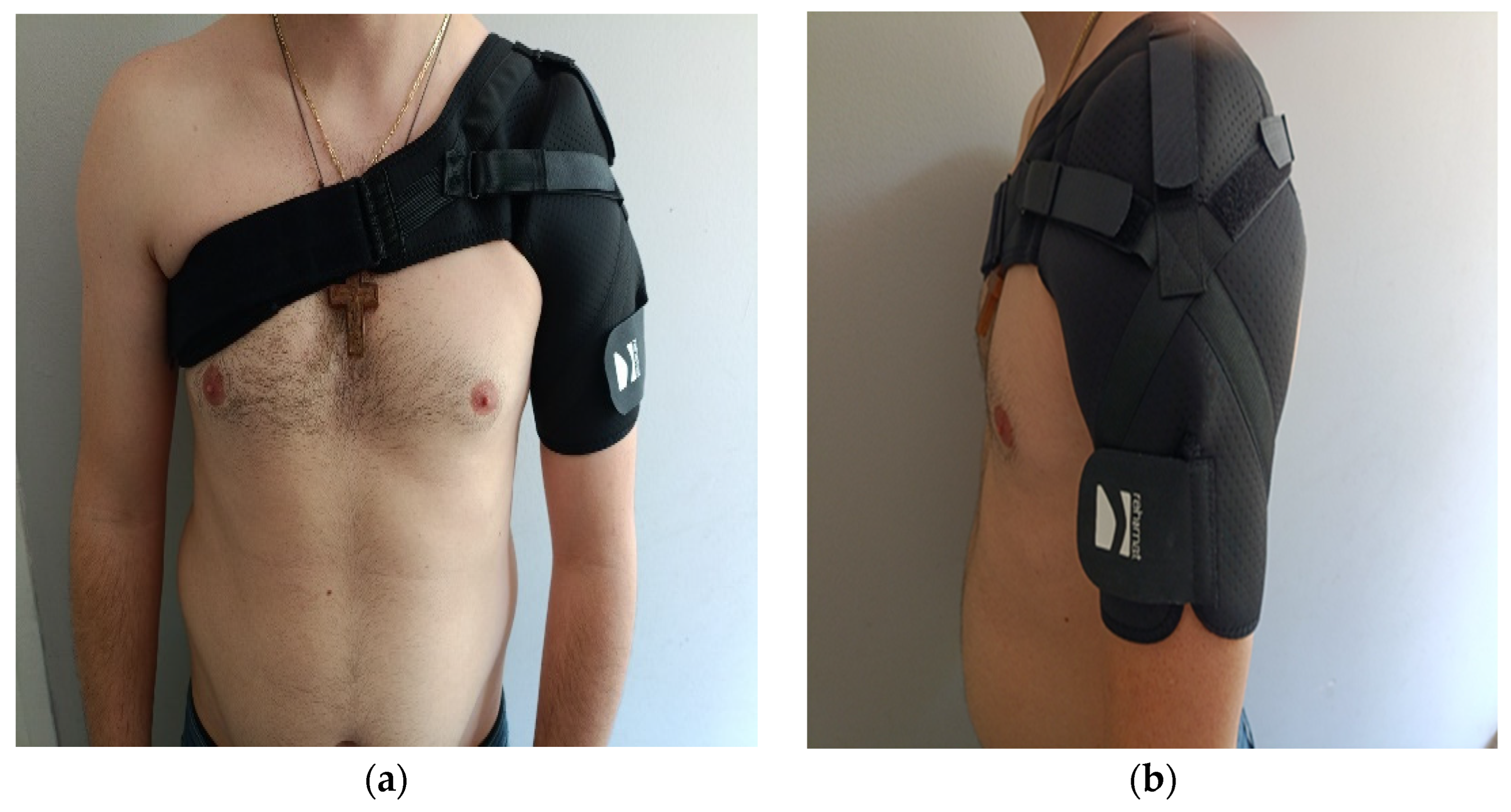
The practical implementation of shoulder orthosis use is also influenced by cost and availability considerations. In Greece, the National Organization For Health Care Services (EOPYY) covers part of the cost, provided there is a medical prescription from a specialist physician. The Reh4mat AM-SOB orthosis used in the present study is available on the Greek market at affordable cost, while both more affordable models with basic features and more expensive models with specialized designs and additional functions are also available. Access is generally easier in large urban centers; however, in remote or island regions, ordering from other parts of the country may be necessary, which increases the cost and delays delivery. Moreover, the inability to try the product before purchase may negatively affect its suitability and patient satisfaction.
The findings of this study also demonstrated strong negative correlations between upper limb Brunnstrom recovery stage, FAC levels, and completion times for the TUG and mFSST tests, as well as a moderate positive correlation with FRT performance. Specifically, higher FAC scores and Brunnstrom stages were associated with shorter completion times on the TUG and mFSST, indicating better functional mobility and balance. Similarly, higher FAC and Brunnstrom scores were associated with greater forward reach distance in the FRT, indicating improved balance. These results align with the existing literature, which suggests that progress in upper limb motor recovery and improved ambulation (as measured by FAC) are related to better dynamic balance and functional mobility in stroke patients [
41,
42,
46].
According to Khan et al. [
59] male patients following a stroke are less likely to experience balance impairments compared to females. Similarly, Vincent-Onabajo et al. [
60] stated that older age and female gender increase the risk of balance impairment among stroke patients. Additionally, female patients often face poorer treatment outcomes, decreased quality of life, and greater disabilities both before and after a stroke compared to male patients [
61]. They also more frequently require assistance with activities of daily living and ambulation and are more likely to reside in nursing homes after their stroke [
62]. Moreover, patients with left hemiparesis exhibited less body weight on the paretic side and greater postural instability compared to those with right hemiparesis, Khan et al. [
59]. This suggests that the affected hemisphere and disrupted cerebral networks lead to different postural deficits in patients with right or left hemiparesis [
63]. However, in the present study, no significant differences were observed in the outcome measures (TUG, FRT, and MFSST) either before or after the application of the brace, neither between male and female participants nor regarding the side of hemiplegia. Assessing functional status through measures like the Functional Independence Measure [
64] and Fugl-Meyer Assessment (FMA) [
65], which were not used in this study, would offer more comprehensive insights into participants’ abilities that could impact the current results. In our study, participants could walk with or without assistance, scoring at least 3 on the FAC scale, indicating a range from dependent to independent walking.
This study includes certain limitations that should be considered when interpreting the results. The sample was restricted to patients in the subacute rehabilitation phase, with specific motor function criteria (FAC ≥ 3 and Brunnstrom stage ≤ 4). The evaluation process was not blinded, as both participants and the assessor were aware of the testing condition (with or without orthosis). Specifically, whether or not an arm sling was used with the same individuals was clearly noticeable and could not be hidden from participants or evaluators, potentially introducing bias. An additional limitation is the heterogeneity in assistive device use among participants (7 walked unaided, 18 used a cane, and 1 used a walker). A further limitation of the present study is the use of the Brunnstrom motor recovery scale, which, although simple and easy to apply, is relatively general and less detailed compared to more contemporary and specialized tools such as the Fugl-Meyer Assessment (FMA) [
65] or the Action Research Arm Test (ARAT) [
66]. In addition, the lack of a specialized assessment of individual shoulder muscle strength or stability (e.g., manual muscle testing, electromyography) may have reduced the precision of the motor function evaluation. Finally, this study only examined the immediate effects of orthosis use, without evaluating its long-term impact or potential patient adaptation over time.
Future research should focus on comparing various types of orthoses, analyzing their effects according to the recovery phase (acute, subacute, chronic), evaluating their impact on pain, and exploring their roles across different levels of patient functionality. Future research should also incorporate more specialized and detailed motor assessment tools, such as the Fugl-Meyer Assessment (FMA), the Action Research Arm Test (ARAT), or targeted evaluations of individual shoulder muscle strength and stability, to enable a more comprehensive and accurate evaluation of motor function. Furthermore, studies with larger sample sizes and long-term follow-up are recommended to assess the prolonged effects of continuous orthosis use. Lastly, examining the impact of orthoses on daily functional activities may offer additional insights into the ecological validity of this intervention.