A Comprehensive Review of Stingless Bee Products: Phytochemical Composition and Beneficial Properties of Honey, Propolis, and Pollen
Abstract
:1. Introduction
2. Stingless Bee Honey
2.1. Antioxidant Properties
2.1.1. Phenolic and Flavonoid Compounds
2.1.2. Total Phenolic Content
2.1.3. Total Flavonoid Content
2.1.4. Antioxidant Capacity
2.2. Antimicrobial
| Study Population | Stingless Bee Species | Origin | Key Findings | Reference |
|---|---|---|---|---|
| Pseudomonas aeruginosa (ATCC 10145) and Streptococcus pyogenes (ATCC 19615) | Trigona sp. | Malaysia | The stingless bee honey used was able to inhibit the growth of two bacterial species: P. aeruginosa and Streptococcus pyogenes, at 25.2 ± 0.6 mm and 26.7 ± 1.0 mm, respectively. | [53] |
| Colletotrichum brevisporum | Trigona sp. | Malaysia | The results of the study showed that synthesis of CaO Nps was able to inhibit the fungus growth in as low as 15% concentration. It is stated that due to the size of Nps, better penetration, absorption, and migration into the fungi cell results in better antifungal action. | [62] |
| Bacillus subtilis ATCC 21332, Staphylococcus aureus ATCC 25923, P. aeruginosa ATCC 27853, and Escherichia coli ATCC 11775 | Heterotrigona itama | Malaysia | H. itama honey was able to inhibit growth of all the bacteria studied. The honey was more effective at inhibiting B. subtilis and S. aureus than P. aeruginosa and E. coli. The author stated it may be due to the outer membranes of E. coli and P. aeruginosa, which have greater resistance to the morphological changes caused by the honey. | [8] |
| S. aureus (ATCC 25923 and ATCC 33591) and E. coli (ATCC 25922 and ATCC 35218) | G. thoracica and H. itama | Malaysia | Greater antibacterial effect was observed in H. itama honey, of which the inhibition zones demonstrated were 0.8–1.3 cm, whereas Geniotrigona thoracica honey’s inhibition zone was 0.9–1.2 cm for the tested population. | [63] |
| Gram-positive bacteria; S. aureus (ATCC11632), B. subtilis (ATCC11774), and three Gram-negative bacteria; E. coli (ATCC10536), Serratia marcescens (ATCC13880), and Alcaligenes faecalis (ATCC15554) | H. itama, H. erythrogastra, Tetrigona apicalis, Lepidotrigona terminata, T. melanoleuca, T. bingami, G. thoracica, and Homotrigona fimbriata | Malaysia | Homotrigona fimbriata honey showed the highest antimicrobial activity, with inhibition of four of five tested bacteria species. However, H. erythrogastra did not inhibit any pathogen, though it had the lowest pH value of 1.83, and the study indicated little correlation of high acidity with high antimicrobial activity. | [47] |
| E. coli, Salmonella Thyphimurium, Klebsiella pneumonia, P. aeruginosa, Bacillus cereus, and S. aureus | H. itama | Malaysia | H. itama honey showed broad antimicrobial activity against pathogens. Specifically, it could inhibit the growth of B. cereus and S. thyphimurium. The antimicrobial activity of the honey was not just attributed to its physicochemical properties but also to isolates present, which were Bacillus strains. | [54] |
| S. aureus (ATCC) 25,923 and E. coli (ATCC 25,922), Haemophilus influenzae (ATCC 19, 418), and Streptococcus pyogenes (ATCC 19,615) | Melipona favosa (Fabricius, 1798) and Frieseomelitta nigra (Cresson, 1879) | Trinidad and Tobago | Both stingless bee honey samples showed that they could inhibit all of the pathogens and had greater bactericidal activities when compared to European honeybee honey and artificial honey (produced by in vitro assay). The minimum inhibitory concentrations (MIC) of 2–16% and minimum bactericidal concentrations (MBC) of 2–32% of the stingless bee honey were lower than those of European honeybee honey and artificial honey of 16–32%. | [64] |
| B. cereus TISTR 2372, P. aeruginosa TISTR 1287, S. aureus TISTR 1840, and Salmonella Typhimurium TISTR 1469 | Tetragonula laeviceps | Thailand | The stingless bee honey showed that it could inhibit all of the microorganism species’ growth rates successfully. The MIC and MBC value of the honey was in the range of 10–30% and 25–50%, respectively. The authors hypothesised that improving dehydration and carbohydrate elimination as well as isolation and extraction of phenolic and flavonoid compounds could provide better antimicrobial activity results. | [18] |
| E coli ATCC 25922, Klebsiella pneumoniae ATCC 4352, P aeruginosa ATCC 15442, and Gram-positive strains of Enterococcus faecalis ATCC 29212, S. aureus ATCC 25923, Streptococcus pneumoniae ATCC 11733, S chromogenes (LB03), and S. aureus (LB14) | Melipona eburnea (Friese, 1900), Melipona grandis (Guérin-Méneville, 1844), Melipona flavolineata (Friese, 1900), and Melipona seminigra (Friese, 1903) | Western Amazon | All of the stingless bee honey samples displayed antibacterial activity against all bacteria except E. coli. The MIC and MBC values of the tested honey were both in the range of 1.56–25%. | [55] |
| Candida albicans | Melipona beecheii | Mexico | The study showed that stingless bee honey could inhibit fungus growth at 35% concentration when tested using the agar dilution method. | [60] |
| S. aureus, E. coli, Klebsiella pneumonia, Methicillin-resistant Staphylococcus aureus (MRSA), P. aeruginosa, and Acinetobacter baumannii. | n.d. | India | The stingless bee honey alone could inhibit all of the pathogen species growth. The study demonstrated that the combination of honey, gelatine, and curcumin had better antibacterial activity than honey alone. | [56] |
| Colletotrichum brevisporum | Trigona sp. | Malaysia | Stingless bee honey at 15% concentration is the optimum in inhibiting and suppressing mycelial growth of the species C. brevisporum. | [59] |
| E. coli ATCC 25992, MRSA, B. subtilis CGMCC 1.2428), P. aeruginosa PAO1, C. albicans ATCC 10231, and Aspergillus terreus 01 | Tetragonisca angustula | Costa Rica | The study showed that Tetragonisca angustula honey strongly inhibited B. subtilis, S. aureus, and E. coli. Against P. aeruginosa, no inhibition activity occurred. The antimicrobial activity of honey was due to the presence of isolates identified as Streptomyces sp. | [57] |
| Bacillus cereus, S. aureus, Micrococcus luteus, E. coli, Enterobacter aerogenes, Alcaligenes faecalis, Aeromonas hydrophila, and Salmonella Typhimurium | H. itama | Malaysia | The study showed that Heterotrigona itama honey inhibited all of the bacterial growth. The antibacterial activity of the honey was attributed to the presence of various bacteria, such as Bacillus spp. | [65] |
| Gram-negative (Klebsiella pneumoniae, E. coli, Salmonella Typhimurium), Gram-positive (S. aureus, Listeria monocytogenes, Bacillus cereus), and fungus (C. albicans). | Melipona bicolor, Melipona quadrifasciata, Melipona marginata, and Scaptotrigona bipuncatata | Brazil | All of the stingless bee honey samples were able to inhibit all of the microorganisms. The study reported that the antimicrobial activity of stingless bee honey was twice as high as European honeybee honey when compared to previous reported findings of MIC. | [23] |
| Gram-positive bacteria were used, namely, S. aureus, S. intermedius B, S. xylosus, and Streptococcus alactolyticus, as well as Gram-negative bacteria, namely, Citrobacter koseri, E coli, Klebsiella pneumonia, P. aeruginosa, Salmonella enterica Serovar Choleraesuis, and Vibrio parahaemolyticus | G. thoracica and H. erythrogastra | Malaysia | The study showed that honey samples produced by Geniotrigona thoracica and Heterotrigona erythrogastra were able to inhibit the growth of all of the various bacterial species tested. By way of contrast, honey produced by Heterotrigona itama showed no inhibitory activity against K. pneumonia, S. enterica, and V. parahaemolyticus. | [30] |
| Gram-negative and Gram-positive bacteria: S. aureus, Bacillus cereus, E. coli, Salmonella Typhimurium, and P. aeruginosa | H. itama | Malaysia | All of the stingless bee honey samples showed great inhibitory activities against the pathogens, as the honey has a broad spectrum of antibacterial activity. The study showed that E. coli was the most sensitive pathogen to the stingless bee honey, which showed that the diameter of the inhibition zone ranged from 26.5 to 32.8 mm. | [58] |
| C. albicans and Aspergillus niger. | Trigona sp. | Malaysia | The study showed that stingless bee honey at 10% concentration could inhibit the growth of both fungus species. | [61] |
| Gram-positive and Gram-negative bacteria were used: S. aureus ATCC25923 and ATCC29213, S. epidermidis ATCC12228, Enterococcus faecalis ATCC29212, Enterococcus faecium ATCC6569, Streptococcus mutans UA159, Streptococcus pyogenes ATCC19615, E. coli ATCC25922 and ATCC8739, Salmonella enterica serovar Enteritidis ATCC13076, Klebsiella pneumoniae ATCC700603, and P. aeruginosa ATCC27853 and ATCC9027. | S. bipunctata and S. postica | Brazil | The study showed that both of the honey samples possess antimicrobial activity against bacteria, with the inhibition zone for Gram-positive strains in the range of 13.9–18.3 mm and Gram-negative strains in the range of 8.14–10.28 mm. It also showed that the combination of both honey samples has the potential for the development of new broad-spectrum antimicrobials that have the potential to prevent the emergence of resistant bacterial strains. | [66] |
2.3. Anticancer Potential of Stingless Bee Honey
3. Stingless Bee Propolis
3.1. Antioxidant Properties
3.2. Chemical Composition
3.3. Antimicrobial
4. Stingless Bee Pollen
5. Future Trends
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zulkhairi Amin, F.A.; Sabri, S.; Mohammad, S.M.; Ismail, M.; Chan, K.W.; Ismail, N.; Norhaizan, M.E.; Zawawi, N. Therapeutic Properties of Stingless Bee Honey in Comparison with European Bee Honey. Adv. Pharmacol. Sci. 2018, 2018, 6179596. [Google Scholar] [CrossRef] [PubMed]
- Mohd-Isa, W.N.; Nizam, A.; Ali, A. Image Segmentation of Meliponine Bee Using Faster R-CNN. In Proceedings of the 2019 Third World Conference on Smart Trends in Systems Security and Sustainablity (WorldS4), London, UK, 21 November 2019; pp. 235–238. [Google Scholar]
- Al-Hatamleh, M.A.I.; Boer, J.C.; Wilson, K.L.; Plebanski, M.; Mohamud, R.; Mustafa, M.Z. Antioxidant-Based Medicinal Properties of Stingless Bee Products: Recent Progress and Future Directions. Biomolecules 2020, 10, 923. [Google Scholar] [CrossRef] [PubMed]
- Ali, H.; Abu Bakar, M.F.; Majid, M.; Muhammad, N.; Lim, S.Y. In Vitro Anti-Diabetic Activity of Stingless Bee Honey from Different Botanical Origins. Food Res. 2020, 4, 1421–1426. [Google Scholar] [CrossRef]
- Shadan, A.F.; Mahat, N.A.; Wan Ibrahim, W.A.; Ariffin, Z.; Ismail, D. Provenance Establishment of Stingless Bee Honey Using Multi-Element Analysis in Combination with Chemometrics Techniques. J. Forensic Sci. 2018, 63, 80–85. [Google Scholar] [CrossRef]
- Majid, M.; Bakar, M.F.A.; Mian, Z. Determination of Xanthine Oxidase Inhibition in Stingless Bee Honey from Different Botanical Origin. In Proceedings of the IOP Conference Series: Earth and Environmental, Yinchuan, China, 19–21 September 2019; Volume 269. [Google Scholar]
- Nordin, A.; Sainik, N.Q.A.V.; Chowdhury, S.R.; Bin Saim, A.; Idrus, R.B.H. Physicochemical Properties of Stingless Bee Honey from around the Globe: A Comprehensive Review. J. Food Compos. Anal. 2018, 73, 91–102. [Google Scholar] [CrossRef]
- Syed Yaacob, S.N.; Wahab, R.A.; Huyop, F.; Lani, M.N.; Zin, N.M. Morphological Alterations in Gram-Positive and Gram-Negative Bacteria Exposed to Minimal Inhibitory and Bactericidal Concentration of Raw Malaysian Stingless Bee Honey. Biotechnol. Biotechnol. Equip. 2020, 34, 575–586. [Google Scholar] [CrossRef]
- Biluca, F.C.; Braghini, F.; Gonzaga, L.V.; Carolina, A.; Costa, O.; Fett, R. Physicochemical Profiles, Minerals and Bioactive Compounds of Stingless Bee Honey (Meliponinae). J. Food Compos. Anal. 2016, 50, 61–69. [Google Scholar] [CrossRef]
- Mustafa, M.Z.; Yaacob, N.S.; Sulaiman, S.A. Reinventing the Honey Industry: Opportunities of the Stingless Bee. Malays. J. Med. Sci. 2018, 25, 1–5. [Google Scholar] [CrossRef]
- Chuttong, B.; Chanbang, Y.; Sringarm, K.; Burgett, M. Physicochemical Profiles of Stingless Bee (Apidae: Meliponini) Honey from South East Asia (Thailand). Food Chem. 2016, 192, 149–155. [Google Scholar] [CrossRef]
- Lee, F. Malaysia’s Stingless Bees That Many Locals Don’t Know about Could Bring in RM3bil/Year. Vulcan Post. Available online: https://vulcanpost.com/742509/kelulut-stingless-bee-honey-farm-malaysia/ (accessed on 20 April 2021).
- Shamsudin, S.; Selamat, J.; Sanny, M.; Razak, S.-B.A.; Jambari, N.N.; Mian, Z.; Khatib, A. Influence of Origins and Bee Species on Physicochemical, Antioxidant Properties and Botanical Discrimination of Stingless Bee Honey. Int. J. Food Prop. 2019, 22, 238–263. [Google Scholar] [CrossRef] [Green Version]
- International Honey Commission. Harmonised Methods of the International Honey Commission; Swiss Bee Research Centre, FAM: Bern, Belgium, 2009; p. 63. [Google Scholar]
- Vit, P.; Medina, M.; Enríquez, M.E. Original Article Quality Standards for Medicinal Uses of Meliponinae Honey in Guatemala, Mexico and Venezuela. Bee World 2015, 85, 2–5. [Google Scholar] [CrossRef] [Green Version]
- MS 2683; Malaysian Standards Kelulut (Stingless Bee) Honey—Specification. Department of Standards Malaysia: Malaysia, 2017.
- Keng, C.B.; Haron, H.; Talib, R.A.; Subramaniam, P. Physical Properties, Antioxidant Content and Anti-Oxidative Activities of Malaysian Stingless Kelulut (Trigona spp.) Honey. J. Agric. Sci. 2017, 9, 32. [Google Scholar] [CrossRef] [Green Version]
- Khongkwanmueang, A.; Nuyu, A.; Straub, L.; Maitip, J. Physicochemical Profiles, Antioxidant and Antibacterial Capacity of Honey from Stingless Bee Tetragonula laeviceps Species Complex. E3S Web Conf. 2020, 141, 03007. [Google Scholar] [CrossRef] [Green Version]
- Sharin, S.N.; Sani, M.S.A.; Jaafar, M.A.; Yuswan, M.H.; Kassim, N.K.; Manaf, Y.N.; Wasoh, H.; Zaki, N.N.M.; Hashim, A.M. Discrimination of Malaysian Stingless Bee Honey from Different Entomological Origins Based on Physicochemical Properties and Volatile Compound Profiles Using Chemometrics and Machine Learning. Food Chem. 2021, 346, 128654. [Google Scholar] [CrossRef]
- Wong, P.; Ling, H.S.; Chung, K.C.; Yau, T.M.S.; Gindi, S.R.A. Chemical Analysis on the Honey of Heterotrigona itama and Tetrigona binghami from Sarawak, Malaysia. Sains Malays. 2019, 48, 1635–1642. [Google Scholar] [CrossRef]
- Ismail, W.I.W.; Hussin, N.N.; Mazlan, S.N.F.; Hussin, N.H.; Radzi, M.N.F.M. Physicochemical Analysis, Antioxidant and Anti Proliferation Activities of Honey, Propolis and Beebread Harvested from Stingless Bee. IOP Conf. Ser. Mater. Sci. Eng. 2018, 440, 012048. [Google Scholar] [CrossRef]
- Nascimento, A.; Marchini, L.; Carvalho, C.; Araújo, D.; Olinda, R.; Silveira, T. Physical-Chemical Parameters of Honey of Stingless Bee (Hymenoptera: Apidae). Am. Chem. Sci. J. 2015, 7, 139–149. [Google Scholar] [CrossRef]
- Ávila, S.; Lazzarotto, M.; Hornung, P.S.; Teixeira, G.L.; Ito, V.C.; Bellettini, M.B.; Beux, M.R.; Beta, T.; Ribani, R.H. Influence of Stingless Bee Genus (Scaptotrigona and Melipona) on the Mineral Content, Physicochemical and Microbiological Properties of Honey. J. Food Sci. Technol. 2019, 56, 4742–4748. [Google Scholar] [CrossRef]
- Missio, P.; Gauche, C.; Gonzaga, L.V.; Carolina, A.; Costa, O. Honey: Chemical Composition, Stability and Authenticity. Food Chem. 2016, 196, 309–323. [Google Scholar]
- Omar, S.; Enchang, F.K.; Nazri, M.U.I.A.; Ismail, M.M.; Ismail, W.I.W. Physicochemical Profiles of Honey Harvested from Four Major Species of Stingless Bee (Kelulut) in North East Peninsular of Malaysia. Malays. Appl. Biol. 2019, 48, 111–116. [Google Scholar]
- Abu Bakar, M.F.; Sanusi, S.B.; Abu Bakar, F.I.; Cong, O.J.; Mian, Z. Physicochemical and Antioxidant Potential of Raw Unprocessed Honey from Malaysian Stingless Bees. Pak. J. Nutr. 2017, 16, 888–894. [Google Scholar] [CrossRef] [Green Version]
- Jimenez, M.; Beristain, C.I.; Azuara, E.; Mendoza, M.R.; Pascual, L.A. Physicochemical and Antioxidant Properties of Honey from Scaptotrigona mexicana Bee. J. Apic. Res. 2016, 55, 151–160. [Google Scholar] [CrossRef]
- Agussalim; Agus, A.; Nurliyani; Umami, N.; Budisatria, I.G.S. Physicochemical Properties of Honey Produced by the Indonesian Stingless Bee: Tetragonula laeviceps. IOP Conf. Ser. Earth Environ. Sci. 2019, 387, 012084. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Tan, S.W.; Yusof, Y.A.; Chua, L.S. Classification of Honey from Its Bee Origin via Chemical Profiles and Mineral Content. Food Anal. Methods 2017, 10, 19–30. [Google Scholar] [CrossRef]
- Tuksitha, L.; Chen, Y.L.S.; Chen, Y.L.; Wong, K.Y.; Peng, C.C. Antioxidant and Antibacterial Capacity of Stingless Bee Honey from Borneo (Sarawak). J. Asia. Pac. Entomol. 2018, 21, 563–570. [Google Scholar] [CrossRef]
- Majid, M.; Ellulu, M.S.; Abu Bakar, M.F. Melissopalynological Study, Phenolic Compounds, and Antioxidant Properties of Heterotrigona itama Honey from Johor, Malaysia. Scientifica 2020, 2020, 2529592. [Google Scholar] [CrossRef] [PubMed]
- Ambriz-Pérez, D.L.; Leyva-López, N.; Gutierrez-Grijalva, E.P.; Heredia, J.B. Phenolic Compounds: Natural Alternative in Inflammation Treatment. A Review. Cogent Food Agric. 2016, 2, 1131412. [Google Scholar]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Cheng, M.Z.S.Z.; Ismail, M.; Chan, K.W.; Ooi, D.J.; Ismail, N.; Zawawi, N.; Mohd Esa, N. Comparison of Sugar Content, Mineral Elements and Antioxidant Properties of Heterotrigona itama Honey from Suburban and Forest in Malaysia. Malays. J. Med. Health Sci. 2019, 15, 104–112. [Google Scholar]
- Ávila, S.; Hornung, P.S.; Teixeira, G.L.; Malunga, L.N.; Apea-Bah, F.B.; Beux, M.R.; Beta, T.; Ribani, R.H. Bioactive Compounds and Biological Properties of Brazilian Stingless Bee Honey Have a Strong Relationship with the Pollen Floral Origin. Food Res. Int. 2019, 123, 1–10. [Google Scholar] [CrossRef]
- Biluca, F.C.; da Silva, B.; Caon, T.; Mohr, E.T.B.; Vieira, G.N.; Gonzaga, L.V.; Vitali, L.; Micke, G.; Fett, R.; Dalmarco, E.M.; et al. Investigation of Phenolic Compounds, Antioxidant and Anti-Inflammatory Activities in Stingless Bee Honey (Meliponinae). Food Res. Int. 2020, 129, 108756. [Google Scholar] [CrossRef]
- Alvarez-Suarez, M.; Giampieri, F.; Brenciani, A.; Mazzoni, L.; Gasparrini, M.; Gonz, A.M.; Morroni, G.; Simoni, S.; Forbes-hernandez, T.Y.; Giovanetti, E.; et al. Apis mellifera vs Melipona beecheii Cuban Poli Fl Oral Honeys: A Comparison Based on Their Physicochemical Parameters, Chemical Composition and Biological Properties. LWT 2018, 87, 272–279. [Google Scholar] [CrossRef]
- Biluca, F.C.; de Gois, J.S.; Schulz, M.; Braghini, F.; Gonzaga, L.V.; Maltez, H.F.; Rodrigues, E.; Vitali, L.; Micke, G.A.; Borges, D.L.G.; et al. Phenolic Compounds, Antioxidant Capacity and Bioaccessibility of Minerals of Stingless Bee Honey (Meliponinae). J. Food Compos. Anal. 2017, 63, 89–97. [Google Scholar] [CrossRef]
- Ismail, N.I.; Kadir, M.R.A.; Mahmood, N.H.; Singh, O.P.; Iqbal, N.; Zulkifli, R.M. Las Actividades de Pecoreo de Apini y Meliponini Influyen En El Contenido Fenólico de Diferentes Tipos de Miel de Malasia. J. Apic. Res. 2016, 55, 137–150. [Google Scholar] [CrossRef]
- Ranneh, Y.; Akim, A.M.; Hamid, H.A.; Khazaai, H.; Fadel, A.; Zakaria, Z.A.; Albujja, M.; Bakar, M.F.A. Honey and Its Nutritional and Anti-Inflammatory Value. BMC Complement. Med. Ther. 2021, 21, 30. [Google Scholar] [CrossRef]
- Maringgal, B.; Hashim, N.; Tawakkal, I.S.M.A.; Mohamed, M.T.M.; Shukor, N.I.A. Phytochemical Compositions and Antioxidant Activities of Malaysian Stingless Bee Honey. Pertanika J. Sci. Technol. 2019, 27 (Suppl. S1), 15–28. [Google Scholar]
- Ya’akob, H.; Norhisham, N.F.; Mohamed, M.; Sadek, N.; Endrini, S. Evaluation of Physicochemical Properties of Trigona sp. Stingless Bee Honey from Various Districts of Johor. J. Kejuruter. 2019, 2, 59–67. [Google Scholar]
- Ranneh, Y.; Ali, F.; Zarei, M.; Akim, A.M.; Hamid, H.A.; Khazaai, H. Malaysian Stingless Bee and Tualang Honeys: A Comparative Characterization of Total Antioxidant Capacity and Phenolic Profile Using Liquid Chromatography-Mass Spectrometry. LWT 2018, 89, 1–9. [Google Scholar] [CrossRef]
- Martinello, M.; Mutinelli, F. Antioxidant Activity in Bee Products: A Review. Antioxidants 2021, 10, 71. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Yusof, Y.A.; Tan, S.W.; Chua, L.S. Classification of Entomological Origin of Honey Based on Its Physicochemical and Antioxidant Properties. Int. J. Food Prop. 2018, 20, S2723–S2738. [Google Scholar] [CrossRef]
- Badrulhisham, N.S.R.; Ab Hamid, S.N.P.; Ismail, M.A.H.; Yong, Y.K.; Zakuan, N.M.; Harith, H.H.; Saidi, H.I.; Nurdin, A. Harvested Locations Influence the Total Phenolic Content, Antioxidant Levels, Cytotoxic, and Anti-Inflammatory Activities of Stingless Bee Honey. J. Asia-Pac. Entomol. 2020, 23, 950–956. [Google Scholar] [CrossRef]
- Rosli, F.N.; Hazemi, M.H.F.; Akbar, M.A.; Basir, S.; Kassim, H.; Bunawan, H. Stingless bee honey: Evaluating its antibacterial activity and bacterial diversity. Insects 2020, 11, 500. [Google Scholar] [CrossRef] [PubMed]
- Nweze, J.A.; Okafor, J.I.; Nweze, E.I.; Nweze, J.E. Evaluation of Physicochemical and Antioxidant Properties of Two Stingless Bee Honeys: A Comparison with Apis mellifera Honey from Nsukka, Nigeria. BMC Res. Notes 2017, 10, 4–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yaacob, M.; Rajab, N.F.; Shahar, S.; Sharif, R. Stingless Bee Honey and Its Potential Value: A Systematic Review. Food Res. 2018, 2, 124–133. [Google Scholar] [CrossRef]
- Jalil, M.A.A.; Kasmuri, A.R.; Hadi, H. Stingless Bee Honey, the Natural Wound Healer: A Review. Skin Pharmacol. Physiol. 2017, 30, 66–75. [Google Scholar] [CrossRef]
- Jibril, F.I.; Hilmi, A.B.M.; Salwani, I.; Lavaniya, M. Physicochemical Characteristics of Malaysian Stingless Bee Honey from Trigona Species. IIUM Med. J. Malays. 2018, 17, 186–191. [Google Scholar]
- Jibril, F.I.; Hilmi, A.B.M.; Manivannan, L. Isolation and Characterization of Polyphenols in Natural Honey for the Treatment of Human Diseases. Bull. Natl. Res. Cent. 2019, 43, 4. [Google Scholar] [CrossRef]
- Al-kafaween, M.A.; Hilmi, A.B.M.; Jaffar, N.; Hamid, A.N.; Zahri, M.K.; Jibril, F.I. Antibacterial and Antibiofilm Activities of Malaysian Trigona honey against Pseudomonas aeruginosa ATCC 10145 and Streptococcus pyogenes ATCC 19615. Jordan J. Biol. Sci. 2020, 13, 69–76. [Google Scholar]
- Aina, F.; Amin, Z.; Sabri, S.; Ismail, M.; Chan, K.W.; Ismail, N.; Esa, N.M.; Azmi, M.; Lila, M. Probiotic Properties of Bacillus Strains Isolated from Stingless Bee (Heterotrigona itama) Honey Collected across Malaysia. Int. J. Environ. Res. Public Health 2020, 17, 278. [Google Scholar]
- Domingos, S.C.B.; Clebis, V.H.; Nakazato, G.; de Oliveira, A.G.; Takayama Kobayashi, R.K.; Peruquetti, R.C.; Pereira, C.D.; Santa Rosa, M.T.; dos Santos Medeiros, L. Antibacterial Activity of Honeys from Amazonian Stingless Bees of Melipona spp. and Its Effects on Bacterial Cell Morphology. J. Sci. Food Agric. 2020, 101, 2072–2077. [Google Scholar] [CrossRef]
- Samraj, S.M.D.; Kirupha, S.D.; Elango, S.; Vadodaria, K. Fabrication of Nanofibrous Membrane Using Stingless Bee Honey and Curcumin for Wound Healing Applications. J. Drug Deliv. Sci. Technol. 2020, 63, 102271. [Google Scholar] [CrossRef]
- Cambronero-Heinrichs, J.C.; Matarrita-Carranza, B.; Murillo-Cruz, C.; Araya-Valverde, E.; Chavarría, M.; Pinto-Tomás, A.A. Phylogenetic Analyses of Antibiotic-Producing Streptomyces sp. Isolates Obtained from the Stingless-Bee Tetragonisca angustula (Apidae: Meliponini). Microbiology 2019, 165, 292–301. [Google Scholar] [CrossRef]
- Hasali, N.H.; Zamri, A.I.; Lani, M.N.; Mubarak, A.; Ahmad, F.; Chilek, T.Z.T. Physico-Chemical Analysis and Antibacterial Activity of Raw Honey of Stingless Bee Farmed in Coastal Areas in Kelantan and Terengganu. Malays. Appl. Biol. 2018, 47, 145–151. [Google Scholar]
- Maringgal, B.; Hashim, N.; Tawakkal, I.S.M.A.; Mohamed, M.T.M.; Hamzah, M.H.; Shukor, N.I.A. The Causal Agent of Anthracnose in Papaya Fruit and Control by Three Different Malaysian Stingless Bee Honeys, and the Chemical Profile. Sci. Hortic. 2019, 257, 108590. [Google Scholar] [CrossRef]
- Hau-Yama, N.E.; Magaña-Ortiz, D.; Oliva, A.I.; Ortiz-Vázquez, E. Antifungal Activity of Honey from Stingless Bee Melipona beecheii against Candida albicans. J. Apic. Res. 2020, 59, 12–18. [Google Scholar] [CrossRef]
- Hamid, Z.; Mohamad, I.; Harun, A.; Salim, R.; Sulaiman, S.A. Antifungal Effect of Three Local Malaysian Honeys on Selected Pathogenic Fungi of Otomycosis: An in Vitro Evaluation. J. Young Pharm. 2018, 10, 414–417. [Google Scholar] [CrossRef] [Green Version]
- Maringgal, B.; Hashim, N.; Tawakkal, I.S.M.A.; Hamzah, M.H.; Mohamed, M.T.M. Biosynthesis of CaO Nanoparticles Using Trigona sp. Honey: Physicochemical Characterization, Antifungal Activity, and Cytotoxicity Properties. J. Mater. Res. Technol. 2020, 9, 11756–11768. [Google Scholar] [CrossRef]
- Ng, W.-J.; Sit, N.-W.; Ooi, P.A.-C.; Ee, K.-Y.; Lim, T.-M. The Antibacterial Potential of Honeydew Honey Produced by Stingless Bee (Heterotrigona itama) against Antibiotic Resistant Bacteria. Antibiotics 2020, 9, 871. [Google Scholar] [CrossRef]
- Brown, E.; O’Brien, M.; Georges, K.; Suepaul, S. Physical Characteristics and Antimicrobial Properties of Apis mellifera, Frieseomelitta nigra and Melipona favosa Bee Honeys from Apiaries in Trinidad and Tobago. BMC Complement. Med. Ther. 2020, 20, 85. [Google Scholar] [CrossRef] [Green Version]
- Ngalimat, M.S.; Rahman, R.N.Z.R.A.; Yusof, M.T.; Syahir, A.; Sabri, S. Characterisation of Bacteria Isolated from the Stingless Bee, Heterotrigona itama, Honey, Bee Bread and Propolis. PeerJ 2019, 2019, 1–20. [Google Scholar] [CrossRef] [Green Version]
- Nishio, E.K.; Ribeiro, J.M.; Oliveira, A.G.; Andrade, C.G.T.J.; Proni, E.A.; Kobayashi, R.K.T.; Nakazato, G. Antibacterial Synergic Effect of Honey from Two Stingless Bees: Scaptotrigona bipunctata Lepeletier, 1836, and S. Postica Latreille, 1807. Sci. Rep. 2016, 6, 21641. [Google Scholar] [CrossRef] [PubMed]
- Nugitrangson, P.; Puthong, S.; Iempridee, T.; Pimtong, W.; Pornpakakul, S.; Chanchao, C. In Vitro and in Vivo Characterization of the Anticancer Activity of Thai Stingless Bee (Tetragonula laeviceps) Cerumen. Exp. Biol. Med. 2016, 241, 166–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, F.; Seerangan, P.; Mustafa, M.Z.; Osman, Z.F.; Abdullah, J.M.; Idris, Z. Anti-Cancer Properties of Heterotrigona itama sp. Honey via Induction of Apoptosis in Malignant Glioma Cells. Malays. J. Med. Sci. 2019, 26, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Wu, J.; Wang, J.; Huang, R. Tobacco and Oral Squamous Cell Carcinoma: A Review of Carcinogenic Pathways. Tob. Induc. Dis. 2019, 17, 29. [Google Scholar] [CrossRef]
- Mahmood, R.; Asif, J.A.; Shahidan, W.N.S. Stingless-Bee (Trigona itama) Honey Adversely Impacts the Growth of Oral Squamous Cell Carcinoma Cell Lines (HSC-2). Eur. J. Integr. Med. 2020, 37, 101162. [Google Scholar] [CrossRef]
- Al-Mahozi, S.; Salim, Z.; Malden, N.J.; Scully, C.; Lopes, V. Tobacco Habit-Associated Oral Disease and the Negative Effects on Surgical Outcomes. Dent. Update 2017, 44, 1065–1070. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment. JAMA 2019, 321, 316. [Google Scholar] [CrossRef] [Green Version]
- Chuntova, P.; Downey, K.M.; Hegde, B.; Almeida, N.D. Genetically Engineered T-Cells for Malignant Glioma: Overcoming the Barriers to Effective Immunotherapy. Front. Immunol. 2019, 9, 3062. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Oh, H.; Ko, J.; Seong, H.; Lee, Y.; Chan, S.; Young, D.; Baek, N. Bioorganic Chemistry Lanceoleins A—G, Hydroxychalcones, from the Flowers of Coreopsis lanceolata and Their Chemopreventive Effects against Human Colon Cancer Cells. Bioorg. Chem. 2019, 85, 274–281. [Google Scholar] [CrossRef]
- Zhao, Y.; Hu, X.; Wang, M. Function Chemopreventive effects of Some Popular Phytochemicals on Human Colon Cancer: A Review. Food Funct. 2018, 9, 4548–4568. [Google Scholar] [CrossRef]
- Yazan, L.S.; Muhamad Zali, M.F.S.; Ali, R.M.; Zainal, N.A.; Esa, N.; Sapuan, S.; Ong, Y.S.; Tor, Y.S.; Gopalsamy, B.; Voon, F.L.; et al. Chemopreventive Properties and Toxicity of Kelulut Honey in Sprague Dawley Rats Induced with Azoxymethane. BioMed Res. Int. 2016, 2016, 4036926. [Google Scholar]
- Didaras, N.A.; Karatasou, K.; Dimitriou, T.G.; Amoutzias, G.D.; Mossialos, D. Antimicrobial Activity of Bee-Collected Pollen and Beebread: State of the Art and Future Perspectives. Antibiotics 2020, 9, 811. [Google Scholar] [CrossRef]
- Lavinas, F.C.; Macedo, E.H.B.C.; Sá, G.B.L.; Amaral, A.C.F.; Silva, J.R.A.; Azevedo, M.M.B.; Vieira, B.A.; Domingos, T.F.S.; Vermelho, A.B.; Carneiro, C.S.; et al. Brazilian Stingless Bee Propolis and Geopropolis: Promising Sources of Biologically Active Compounds. Revista Brasileira de Farmacognosia 2018, 29, 389–399. [Google Scholar] [CrossRef]
- Ibrahim, N.; Niza, N.F.S.M.; Rodi, M.M.M.; Zakaria, A.J.; Ismail, Z.; Mohd, K.S. Chemical and Biological Analyses of Malaysian Stingless Bee Propolis Extracts. Malays. J. Anal. Sci. 2016, 20, 413–422. [Google Scholar] [CrossRef]
- Pazin, W.M.; Monaco, L.D.M.; Egea Soares, A.E.; Miguel, F.G.; Berretta, A.A.; Ito, A.S. Actividad Antioxidante de Tres Tipos de Propóleos de Abeja Sin Aguijón y Propóleos Verdes. J. Apic. Res. 2017, 56, 40–49. [Google Scholar] [CrossRef]
- Amalia, E.; Diantini, A.; Subarnas, A. Water-Soluble Propolis and Bee Pollen of Trigona spp. from South Sulawesi Indonesia Induce Apoptosis in the Human Breast Cancer MCF-7 Cell Line. Oncol. Lett. 2020, 20, 274. [Google Scholar] [CrossRef]
- Fikri, A.M.; Sulaeman, A.; Marliyati, S.A.; Fahrudin, M. Antioxidant Activity and Total Phenolic Content of Stingless Bee Propolis from Indonesia. J. Apic. Sci. 2019, 63, 139–147. [Google Scholar] [CrossRef] [Green Version]
- Akhir, R.A.M.; Bakar, M.F.A.; Sanusi, S.B. Antioxidant and Antimicrobial Activity of Stingless Bee Bread and Propolis Extracts. AIP Conf. Proc. 2017, 1891, 020090. [Google Scholar]
- Kasote, D.M.; Pawar, M.V.; Gundu, S.S.; Bhatia, R.; Nandre, V.S.; Jagtap, S.D.; Mahajan, S.G.; Kulkarni, M.V. Chemical Profiling, Antioxidant, and Antimicrobial Activities of Indian Stingless Bees Propolis Samples. J. Apic. Res. 2019, 58, 617–625. [Google Scholar] [CrossRef]
- Campos, J.F.; Das Santos, U.P.; Da Rocha, P.D.S.; Damião, M.J.; Balestieri, J.B.P.; Cardoso, C.A.L.; Paredes-Gamero, E.J.; Estevinho, L.M.; De Picoli Souza, K.; Dos Santos, E.L. Antimicrobial, Antioxidant, Anti-Inflammatory, and Cytotoxic Activities of Propolis from the Stingless Bee Tetragonisca fiebrigi (Jataí). Evid.-Based Complement. Altern. Med. 2015, 2015, 296186. [Google Scholar] [CrossRef] [Green Version]
- Surek, M.; Fachi, M.M.; de Fátima Cobre, A.; de Oliveira, F.F.; Pontarolo, R.; Crisma, A.R.; de Souza, W.M.; Felipe, K.B. Chemical Composition, Cytotoxicity, and Antibacterial Activity of Propolis from Africanized Honeybees and Three Different Meliponini Species. J. Ethnopharmacol. 2021, 269, 113662. [Google Scholar] [CrossRef] [PubMed]
- Hochheim, S.; Guedes, A.; Faccin-Galhardi, L.; Rechenchoski, D.Z.; Nozawa, C.; Linhares, R.E.; Filho, H.H.D.S.; Rau, M.; Siebert, D.A.; Micke, G.; et al. Determination of Phenolic Profile by HPLC–ESI-MS/MS, Antioxidant Activity, in Vitro Cytotoxicity and Anti-Herpetic Activity of Propolis from the Brazilian Native Bee Melipona quadrifasciata. Rev. Bras. Farmacogn. 2019, 29, 339–350. [Google Scholar] [CrossRef]
- Torres, A.R.; Sandjo, L.P.; Friedemann, M.T.; Tomazzoli, M.M.; Maraschin, M.; Mello, C.F.; Santos, A.R.S. Chemical Characterization, Antioxidant and Antimicrobial Activity of Propolis Obtained from Melipona quadrifasciata quadrifasciata and Tetragonisca angustula Stingless Bees. Braz. J. Med. Biol. Res. 2018, 51, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rubinho, M.P.; de Carvalho, P.L.N.; Reis, A.L.L.E.; de Alencar, S.M.; Ruiz, A.L.T.G.; de Carvalho, J.E.; Ikegaki, M. A Comprehensive Characterization of Polyphenols by LC-ESI–QTOF-MS from Melipona quadrifasciata anthidioides Geopropolis and Their Antibacterial, Antioxidant and Antiproliferative Effects. Nat. Prod. Res. 2020, 34, 3139–3144. [Google Scholar] [CrossRef]
- Mamoon, K.; Thammasit, P.; Iadnut, A.; Kitidee, K.; Anukool, U.; Tragoolpua, Y.; Tragoolpua, K. Unveiling the Properties of Thai Stingless Bee Propolis via Diminishing Cell Wall-Associated Cryptococcal Melanin and Enhancing the Fungicidal Activity of Macrophages. Antibiotics 2020, 9, 420. [Google Scholar] [CrossRef]
- de Sousa, J.M.B.; de Souza, E.L.; Marques, G.; de Toledo Benassi, M.; Gullón, B.; Pintado, M.M.; Magnani, M. Sugar Profile, Physicochemical and Sensory Aspects of Monofloral Honeys Produced by Different Stingless Bee Species in Brazilian Semi-Arid Region. LWT 2016, 65, 645–651. [Google Scholar] [CrossRef] [Green Version]
- Georgieva, K.; Popova, M.; Dimitrova, L.; Trusheva, B.; Thanh, L.N.; Lan Phuong, D.T.; Lien, N.T.P.; Najdenski, H.; Bankova, V. Phytochemical Analysis of Vietnamese Propolis Produced by the Stingless Bee Lisotrigona cacciae. PLoS ONE 2019, 14, e0216074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yam-Puc, A.; Santana-Hernández, A.A.; Yah-Nahuat, P.N.; Ramón-Sierra, J.M.; Cáceres-Farfán, M.R.; Borges-Argáez, R.L.; Ortiz-Vázquez, E. Pentacyclic Triterpenes and Other Constituents in Propolis Extract from Melipona beecheii Collected in Yucatan, México. Rev. Bras. Farmacogn. 2019, 29, 358–363. [Google Scholar] [CrossRef]
- Nazir, H.; Shahidan, W.N.S.; Ibrahim, H.A.; Ismail, T.N.N.T. Chemical Constituents of Malaysian Geniotrigona thoracica Propolis. Pertanika J. Trop. Agric. Sci. 2018, 41, 955–962. [Google Scholar]
- Chewchinda, S.; Vongsak, B. Development and Validation of a High-Performance Thin Layer Chromatography Method for the Simultaneous Quantitation of α- and γ-Mangostins in Thai Stingless Bee Propolis. Rev. Bras. Farmacogn. 2019, 29, 333–338. [Google Scholar] [CrossRef]
- Torres-González, A.; López-Rivera, P.; Duarte-Lisci, G.; López-Ramírez, Á.; Correa-Benítez, A.; Rivero-Cruz, J.F. Analysis of Volatile Components from Melipona Beecheii Geopropolis from Southeast Mexico by Headspace Solid-Phase Microextraction. Nat. Prod. Res. 2016, 30, 237–240. [Google Scholar] [CrossRef]
- De Souza, E.C.A.; Da Silva, E.J.G.; Cordeiro, H.K.C.; Filho, N.M.L.; Da Silva, F.M.A.; Dos Reis, D.L.S.; Porto, C.; Pilau, E.J.; Da Costa, L.A.M.A.; De Souza, A.D.L.; et al. Chemical Compositions and Antioxidant and Antimicrobial Activities of Propolis Produced by Frieseomelitta longipes and Apis mellifera Bees. Quim. Nova 2018, 41, 485–491. [Google Scholar] [CrossRef]
- De Los Reyes, M.M.; Oyong, G.G.; Ebajo, V.D.; Shen, C.-C.; Ragasa, C.Y. Cytotoxic Prenylflavanones from Philippine Stingless Bee (Tetragonula biroi friese) Nests. Asian J. Chem. 2018, 30, 613–619. [Google Scholar] [CrossRef]
- Abdullah, N.A.; Ja’afar, F.; Yasin, H.M.; Taha, H.; Petalcorin, M.I.R.; Mamit, M.H.; Kusrini, E.; Usman, A. Physicochemical Analyses, Antioxidant, Antibacterial, and Toxicity of Propolis Particles Produced by Stingless Bee Heterotrigona itama Found in Brunei Darussalam. Heliyon 2019, 5, e02476. [Google Scholar] [CrossRef] [Green Version]
- Akhir, R.A.M.; Bakar, M.F.A.; Sanusi, S.B. Antioxidant and Antimicrobial Potential of Stingless Bee (Heterotrigona itama) by-Products. J. Adv. Res. Fluid Mech. Therm. Sci. 2018, 42, 72–79. [Google Scholar]
- Shehu, A.; Ismail, S.; Rohin, M.A.K.; Harun, A.; Aziz, A.A.; Haque, M. Antifungal Properties of Malaysian Tualang Honey and Stingless Bee Propolis against Candida albicans and Cryptococcus neoformans. J. Appl. Pharm. Sci. 2016, 6, 044–050. [Google Scholar] [CrossRef] [Green Version]
- Dutra, R.P.; Bezerra, J.L.; da Silva, M.C.P.; Batista, M.C.A.; Patrício, F.J.B.; Nascimento, F.R.F.; Ribeiro, M.N.S.; Guerra, R.N.M. Antileishmanial Activity and Chemical Composition from Brazilian Geopropolis Produced by Stingless Bee Melipona fasciculata. Rev. Bras. Farmacogn. 2019, 29, 287–293. [Google Scholar] [CrossRef]
- Abdullah, N.A.; Zullkiflee, N.; Zaini, S.N.Z.; Taha, H.; Hashim, F.; Usman, A. Phytochemicals, Mineral Contents, Antioxidants, and Antimicrobial Activities of Propolis Produced by Brunei Stingless Bees Geniotrigona thoracica, Heterotrigona itama, and Tetrigona binghami. Saudi J. Biol. Sci. 2020, 27, 2902–2911. [Google Scholar] [CrossRef]
- Dos Santos, H.F.; Campos, J.F.; Dos Santos, C.M.; Balestieri, J.B.P.; Silva, D.B.; Carollo, C.A.; Souza, K.D.P.; Estevinho, L.M.; Dos Santos, E.L. Chemical Profile and Antioxidant, Anti-Inflammatory, Antimutagenic and Antimicrobial Activities of Geopropolis from the Stingless Bee Melipona orbignyi. Int. J. Mol. Sci. 2017, 18, 953. [Google Scholar] [CrossRef]
- Utispan, K.; Chitkul, B.; Monthanapisut, P.; Meesuk, L.; Pugdee, K.; Koontongkaew, S. Propolis Extracted from the Stingless Bee Trigona sirindhornae Inhibited S. Mutans Activity in Vitro. Oral Health Prev. Dent. 2017, 15, 279–284. [Google Scholar]
- Omar, W.A.W.; Azhar, N.A.; Fadzilah, N.H.; Kamal, N.N.S.N.M. Bee Pollen Extract of Malaysian Stingless Bee Enhances the Effect of Cisplatin on Breast Cancer Cell Lines. Asian Pac. J. Trop. Biomed. 2016, 6, 265–269. [Google Scholar] [CrossRef] [Green Version]
- Harif Fadzilah, N.; Jaapar, M.F.; Jajuli, R.; Wan Omar, W.A. Contenido Total Fenólico y Flavonoide, y Actividad Antioxidante En Extractos Etanólicos de Polen de Tres Especies Diferentes de Abeja Malasia Sin Aguijón. J. Apic. Res. 2017, 56, 130–135. [Google Scholar] [CrossRef]
- Mohammad, S.M.; Mahmud-Ab-Rashid, N.-K.; Zawawi, N. Stingless Bee-Collected Pollen (Bee Bread): Chemical and Microbiology Properties and Health Benefits. Molecules 2021, 26, 957. [Google Scholar] [CrossRef]
- Nurdianah, H.F.; Firdaus, A.A.; Azam, O.E.; Adnan, W.W. Antioxidant Activity of Bee Pollen Ethanolic Extracts from Malaysian Stingless Bee Measured Using DPPH-HPLC Assay. Int. Food Res. J. 2016, 23, 403–405. [Google Scholar]
- Jorge, A.; Lopes, O.; Vasconcelos, C.C.; Assis, F.; Pereira, N.; Helena, R.; Silva, M.; Felipe, P.; Queiroz, S.; Fernandes, C.V.; et al. Anti-Inflammatory and Antinociceptive Activity of Pollen Extract Collected by Stingless Bee Melipona fasciculata. Int. J. Mol. Sci. 2019, 20, 4512. [Google Scholar] [CrossRef] [Green Version]
- Wan Omar, W.A.; Yahaya, N.; Ghaffar, Z.A.; Fadzilah, N.H. Gc-Ms Analysis of Chemical Constituents in Ethanolic Bee Pollen Extracts from Three Species of Malaysian Stingless Bee. J. Apic. Sci. 2018, 62, 275–284. [Google Scholar]
- Kieliszek, M.; Piwowarek, K.; Kot, A.M.; Błażejak, S.; Chlebowska-Śmigiel, A.; Wolska, I. Pollen and Bee Bread as New Health-Oriented Products: A Review. Trends Food Sci. Technol. 2018, 71, 170–180. [Google Scholar] [CrossRef]
- Mohammad, S.M.; Mahmud-Ab-Rashid, N.K.; Zawawi, N. Probiotic Properties of Bacteria Isolated from Bee Bread of Stingless Bee Heterotrigona itama. J. Apic. Res. 2020, 60, 172–187. [Google Scholar] [CrossRef]
- Belina-Aldemita, M.D.; Schreiner, M.; D’Amico, S. Characterization of Phenolic Compounds and Antioxidative Potential of Pot-Pollen Produced by Stingless Bees (Tetragonula biroi friese) from the Philippines. J. Food Biochem. 2020, 44, e13102. [Google Scholar] [CrossRef]
- Cenet, M.; Bozdogan, A.; Sezer, G.; Acar, L.; Ulukanli, Z. Antimicrobial Activities, Pollen Diversity and Physicochemical Properties of Natural Honey from Southeastern Anatolia of Turkey. Adv. Life Sci. 2017, 4, 47–54. [Google Scholar]
- Radev, Z. Variety in Protein Content of Pollen from 50 Plants from Bulgaria. Bee World 2018, 95, 81–83. [Google Scholar] [CrossRef]
- Carneiro, A.L.B.; Gomes, A.A.; da Silva, L.A.; Alves, L.B.; da Silva, E.C.; da Silva Pinto, A.C.; Tadei, W.P.; Pohlit, A.M.; Simas Teixeira, M.F.; Gomes, C.C.; et al. Antimicrobial and Larvicidal Activities of Stingless Bee Pollen from Maues, Amazonas, Brazil. Bee World 2019, 96, 98–103. [Google Scholar] [CrossRef]
- Bárbara, M.; Machado, C.; Sodré, G.; Dias, L.; Estevinho, L.; de Carvalho, C. Microbiological Assessment, Nutritional Characterization and Phenolic Compounds of Bee Pollen from Mellipona mandacaia smith, 1983. Molecules 2015, 20, 12525–12544. [Google Scholar] [CrossRef] [Green Version]



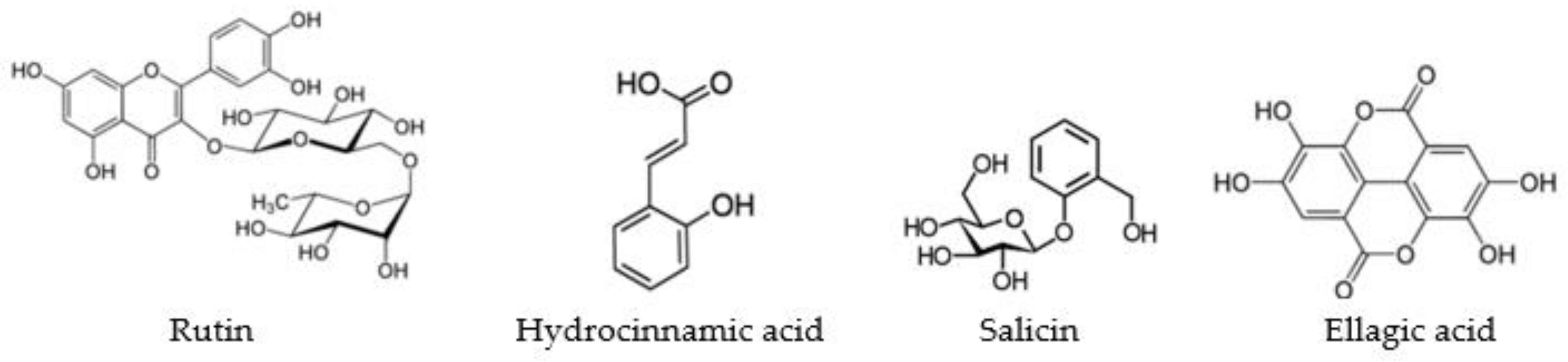
| Parameter | Standards | ||
|---|---|---|---|
| International Honey Commission (European Honeybee Honey) | Vit et al. [15] (Stingless Bee Honey) | Department of Malaysian Standards (Stingless Bee Honey) | |
| Moisture (g/100 g) | ≤20 | ≤30 | ≤35 |
| Sum of fructose and glucose (g/100 g) | ≥60 | ≥50 | ≥85 |
| Sucrose (g/100 g) | ≤5 | ≤6 | ≤7.5 |
| Maltose (g/100 g) | - | - | ≤9.5 |
| Free acidity (meq/100 g) | ≤50 | ≤85 | - |
| Ash content (g/100 g) | ≤0.5 | ≤0.5 | ≤1.0 |
| Electrical conductivity (mS/cm) | ≤0.8 | - | - |
| Hydroxymethylfurfural (HMF) content (mg/kg) | ≤40 | ≤40 | ≤30 |
| Diastase activity (DN) | ≥8 | ≥3 | - |
| pH | - | - | 2.5 to 3.8 |
| Phenolic compounds | - | - | Present |
| Phenolic Compounds | Stingless Bee Species/Reference | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Heterotrigona itama | Scaptotrigona bipuncatata | Trigona hypogea | Tetragonisca angustula | Tetragona clavipes | Melipona marginata | Melipona quadriasciata | Melipona bicolor | Melipona beecheii (Bennet, 1831) | Melipona mondury | Melipona scutellaris | Melipona rufiventris mondory | |
| Chlorogenic acid | [31] | [36] | [38] | [38] | ||||||||
| Coumaric acid | [37] | |||||||||||
| p-coumaric acid | [31,39] | [35,36,38] | [36] | [36,38] | [36,38] | [36,38] | [35,36,38] | [35,38] | [38] | [38] | ||
| Salicylic acid | [36,38] | [36] | [36,38] | [36,38] | [36,38] | [38] | [38] | [38] | [38] | |||
| Protocatechuic acid | [31] | [36,38] | [36,38] | [36,38] | [36,38] | [38] | [38] | |||||
| Ferulic acid | [36] | [36] | [36] | [36] | ||||||||
| Mandelic acid | [36,38] | [36] | ||||||||||
| Rosmarenic acid | [36] | [36,38] | [38] | [38] | ||||||||
| Vanillic acid | [36] | [36] | [36] | [36,38] | [38] | |||||||
| Caffeic acid | [39] | [36] | [36,38] | [38] | [36,38] | [38] | [38] | |||||
| Ellagic acid | [39] | [35] | [35] | [35] | [35] | |||||||
| Dihydrocaffeic acid | [37] | |||||||||||
| Sinapic acid | [38] | |||||||||||
| TRANS ferulic acid | [39] | [38] | [38] | [38] | [38] | [38] | ||||||
| Syringic acid | [39] | [38] | [38] | [38] | [38] | [38] | ||||||
| 4-(hydroxy-methyl) benzoic acid | [38] | [38] | ||||||||||
| 4-aminobenzoic acid | [36] | |||||||||||
| Benzoic acid | [39] | |||||||||||
| Trans-cinnamic acid | [39] | |||||||||||
| Epicatechin | [31] | |||||||||||
| Rutin | [31] | |||||||||||
| Rutin hydrate | [39] | |||||||||||
| Catechin | [31] | |||||||||||
| Naringenin | [39] | [36,38] | [36] | [36,38] | [36,38] | [38] | [38] | [38] | [38] | [38] | ||
| Aromadendrin | [36] | [36] | [36,38] | [36,38] | [36,38] | [36,38] | [38] | [38] | [38] | |||
| Taxifolin | [36,38] | [36] | [36,38] | [36,38] | [36,38] | [38] | [38] | [38] | [38] | [38] | ||
| Isoquercetin | [36,38] | [36] | [38] | [38] | ||||||||
| Vanilin | [38] | |||||||||||
| Quercetin | [39] | [35,36] | [36,38] | [35,36] | [35,36,38] | [35] | ||||||
| Syringaldehyde | [36] | [36] | [38] | [36,38] | ||||||||
| Carnosol | [38] | [36] | [36,38] | [38] | ||||||||
| Scopoletin | [36] | [36] | ||||||||||
| Eriodictol | [38] | [36] | [36,38] | [36,38] | [36,38] | [38] | ||||||
| Umbelliferone | [36,38] | |||||||||||
| Hesperitin | [39] | [35] | [35] | [35] | [35] | |||||||
| C-pentosyl-c-hexosyl-apigenin isomer | [37] | |||||||||||
| Quercetin deoxyhexosyl hexoside | [37] | |||||||||||
| Apigenin trihexoside | [37] | |||||||||||
| Kaempferol deoxyhexosyl hexoside | [37] | |||||||||||
| Kaempferol | [39] | [37] | ||||||||||
| Isorhamnetin deoxyhexosyl hexoside | [37] | |||||||||||
| Isorhamnetin | [37] | |||||||||||
| Luteolin | [39] | [37] | ||||||||||
| Bis-methylated quercetin | [37] | |||||||||||
| Apigenin | [38] | [38] | [38] | [37] | [38] | [38] | ||||||
| Methyl luteolin | [37] | |||||||||||
| Methyl quercetin | [37] | |||||||||||
| Hispidulin | [38] | |||||||||||
| Chrysin | [39] | [38] | ||||||||||
| Mirecetrin | [38] | [38] | ||||||||||
| Sinapaldehyde | [38] | |||||||||||
| Stingless Bee | Study | Findings | Reference |
|---|---|---|---|
| Heterotrigona itama | Oral squamous cell carcinoma (OSCC) | The study showed that Heterotrigona itama honey could inhibit cancerous cells. The stingless bee honey needed to inhibit 50% of cell growth was only less than 1% of the dose. | [70] |
| Trigona sp. | Breast | The study demonstrated the potential use of stingless bee honey in treating breast cancer. The author compared three different samples of stingless bee honey that were collected across Malaysia, and the results showed that the ideal honey sample was that which had the greatest cytotoxic activity towards ER- and PR-positive cells compared to triple-negative breast cancer cells. | [46] |
| Heterotrigona itama | Malignant glioma | The study displayed high anticancer activities of stingless bee honey, which can inhibit cell proliferation and prevent malignant glioma in cell lines. | [68] |
| Trigona sp. | Colon | The study reported potential chemopreventive properties of stingless bee honey against colon cancer cells. | [76] |
| Chemical Compounds | Stingless Bee Species/Reference | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Scaptotrigona bipuncatata | Melipona quadrifasciata quadrifasciata (Lepeletier, 1836) | Plebeia remota | Melipona quadrifasciata anthidioides | Tetragonula laeviceps | Tetrigona melanoleuca | Trigona sp. | Melipona beecheii | Lisotrigona cacciae | Friesomelitta longipes | Tetragonula biroi | Tetragonisca angustula | Tetragonula fuscobaleata | Geniotrigona thoracica | Melipona fasciculata | Tetragonisca fiebrigi | |
| p-coumaric acid | [86] | [86,87,88] | [80,89] | [84] | [85] | |||||||||||
| p-coumaric hexoside acid | [89] | |||||||||||||||
| Ferulic acid | [86] | [84] | ||||||||||||||
| Isoferulic acid | [86] | |||||||||||||||
| Drupanin (3-prenyl-4- hydroxycinnamic acid) | [86] | |||||||||||||||
| Oleic acid | [86] | |||||||||||||||
| Stearic acid | [86] | |||||||||||||||
| Ellagic acid | [86] | [86] | [89] | |||||||||||||
| Cinnamic acid | [84] | [85] | ||||||||||||||
| Hydrocinnamic acid | [85] | |||||||||||||||
| Gallic acid | [86,88] | [89] | [90] | [90] | [84] | [88] | [91] | |||||||||
| Palmitic acid | [86] | |||||||||||||||
| Anacardic acid | [86] | [92] | ||||||||||||||
| Junicedric acid | [86] | [86] | ||||||||||||||
| Mangiferonic acid | [86] | |||||||||||||||
| Isomangiferolic acid | [86] | |||||||||||||||
| Trans-communic acid | [86] | |||||||||||||||
| Caffeic acid | [84] | |||||||||||||||
| Pimaric acid | [86] | |||||||||||||||
| Arachidonic acid | [86] | |||||||||||||||
| Benzoic acid | [85] | |||||||||||||||
| Agathic acid | [86] | |||||||||||||||
| Cupressic acid | [86] | [86] | ||||||||||||||
| Isocupressic acid | [86] | [86] | ||||||||||||||
| Kaurenoic acid | [85] | |||||||||||||||
| 15-acetoxy-cupressic acid | [86] | |||||||||||||||
| 4-methoxybenzoic acid | [85] | |||||||||||||||
| Hydrocinnamic acid ethyl ester | [85] | |||||||||||||||
| 3-phenyl-p-coumaric acid | [85] | |||||||||||||||
| 4-hydroxy-3(e)-(4-hydroxy-3- methyl-2-butenyl)-5-prenyl cinnamic acid | [86] | |||||||||||||||
| 3-hydroxy-2,2-dimethyl-8-prenyl- 2 h-1-benzopyran-6-propenoic acid | [86] | |||||||||||||||
| Eicosapentaenoic acid | [86] | |||||||||||||||
| Dicaffeoylquinic acid isomer | [86] | |||||||||||||||
| Vicenin-2, e)-3-{4-hydroxy-3-[(e)-4-(2,3- dihydrocinnamoyloxy)-3-methyl-2- butenyl]-5-prenyl-phenyl}-2- propenoic acid | [86] | |||||||||||||||
| Isoliquiritigenin | [86] | |||||||||||||||
| Formononetin | [86] | |||||||||||||||
| Biochanin a | [86] | |||||||||||||||
| Kaempferol methyl ether | [86] | |||||||||||||||
| Dihydrokaempferide | [86] | |||||||||||||||
| Retusin 8-methyl ether, | [86] | |||||||||||||||
| Artepillin c | [86] | |||||||||||||||
| Artepillin c derivative | [86] | |||||||||||||||
| Naringenin | [86,87] | [89] | [84] | |||||||||||||
| Methyl-naringenin | [89] | |||||||||||||||
| Aromadendrin | [86,87] | [89] | ||||||||||||||
| Methyl-aromadendrin | [89] | |||||||||||||||
| Isosakuranetin | [86] | |||||||||||||||
| Aromadendrin methyl ether | [86] | |||||||||||||||
| Sugiol | [86] | [86] | ||||||||||||||
| Cinnamoyl-coumaroyl-hexoside | [86] | |||||||||||||||
| Dicoumaroyl-hexoside | [86] | |||||||||||||||
| Digalloyl-cinnamoyl-hexoside | [86] | |||||||||||||||
| Cinnamoyl-coumaroyl-galloyl-hexoside | [86] | |||||||||||||||
| Dicoumaroyl-galloyl-hexoside | [86] | |||||||||||||||
| Betuletol | [86] | |||||||||||||||
| Totarol | [86] | |||||||||||||||
| O-coumaroyl o-galloyl hexoside | [89] | |||||||||||||||
| Di-o-galloyl o-cinnamoyl hexoside | [89] | |||||||||||||||
| O-cinnamoyl o-galloyl hexoside | [89] | |||||||||||||||
| O-galloyl hexoside | [90] | |||||||||||||||
| O-cinnamoyl o-coumaroyl hexoside | [89] | |||||||||||||||
| Luteolin-methyl-ether | [89] | |||||||||||||||
| Quercetin-3-methyl-ether | [89] | |||||||||||||||
| Pinocembrin | [87] | [90] | ||||||||||||||
| Quercetin | [88] | [90] | [84] | |||||||||||||
| Kaempferol | [84] | |||||||||||||||
| Phenethyl caffeate | [84] | |||||||||||||||
| Pentacyclic triterpens | [93] | |||||||||||||||
| Catechin | [87] | |||||||||||||||
| Epicatechin | [87] | |||||||||||||||
| Alkylresorcynols | [92] | [94] | ||||||||||||||
| Triterpenes | [92] | [94] | ||||||||||||||
| Homoisoflavanes | [92] | [91] | ||||||||||||||
| Prenylated xantones | [92] | |||||||||||||||
| 7,4′-dihydroxy-5-methoxyhomoisoflavane | [92] | |||||||||||||||
| 10,11-dihydroxydracaenone C | [92] | |||||||||||||||
| 3-geranyloxy-1,7-dihydroxyxanthone | [92] | |||||||||||||||
| 7-geranyloxy-1,3-dihydroxyxanthone | [92] | |||||||||||||||
| 2,6,8-trihydroxy-5-geranyl-7-prenylxanthone | [92] | |||||||||||||||
| A-mangostin | [92] | [95] | ||||||||||||||
| Γ-mangostin | [95] | |||||||||||||||
| Garcinone b | [92] | |||||||||||||||
| Cycloartenone | [92] | |||||||||||||||
| Lupeol | [92] | |||||||||||||||
| Monoterpenes | [96] | [97] | ||||||||||||||
| Sesquiterpenes | [97] | |||||||||||||||
| Prenylated benzophenones | [97] | |||||||||||||||
| Glyasperin a | [98] | |||||||||||||||
| Propolin e | [98] | |||||||||||||||
| Propolin a | [98] | |||||||||||||||
| Vanillin | [88] | |||||||||||||||
| Styrene | [96] | |||||||||||||||
| Benzaldehyde | [96] | |||||||||||||||
| Cinnamyl caffeate | [85] | |||||||||||||||
| Benzyl caffeate | [85] | |||||||||||||||
| Study Population | Stingless Bee Species | Origin | Key Findings | Reference |
|---|---|---|---|---|
| Escherichia coli ATCC 25922, E. coli (ATCC 35218), Klebsiella pneumoniae (ATCC 13883), K. pneumoniae (ATCC 700603, Pseudomonas aeruginosa (ATCC 27853), Enterococcus faecalis (ATCC 29212), E. faecalis (ATCC 51299, methicillin-sensitive Staphylococcus aureus (MSSA) ATCC 6538), and methicillin-resistant Staphylococcus aureus (MRSA, ATCC 33591) | Scaptotrigona bipunctata and Melipona quadrifasciata | Brazil | Extract from Melipona quadrifasciata geopropolis inhibited most of the growth of the sample microorganisms except for E. coli, K. pneumoniae, and P. aeruginosa. However, S. bipunctata extract did not show any inhibition. The antimicrobial activity of the extracts was attributed to the presence of diterpene compounds, gallic acid, and totarol. | [86] |
| Staphylococcus aureus ATCC-29213 and Bacillus subtilis ATCC-11774) and two Gram-negative bacterial strains (E. coli ATCC-11775 and P. aeruginosa ATCC-27853) | Geniotrigona thoracica, Heterotrigona itama, and Tetrigona binghami | Brunei | Extracts from both geopropolis inhibited all of the growth of all the microorganisms. Furthermore, in comparison of both geopropolis extracts to the control antibiotic samples, rifampicin and streptomycin, the geopropolis extracts showed weaker microorganism inhibition of 7.0–13.0 mm, whereas that of the antibiotics was 12.4–14.8 mm. | [103] |
| Leishmania amazonensis | Melipona fasciculata | Brazil | Extract from the geopropolis inhibited the protozoan growth and effectively reduced infection of murine macrophages. The anti-Leishmania activity of the extracts was likely attributed to the presence of gallic acid and ellagic acid. | [102] |
| S. aureus ATCC-29213 and B. subtilis ATCC-11774, E. coli ATCC-11775, and P. aeruginosa ATCC-27853 | Heterotrigona itama | Brunei | Geopropolis extracts inhibited the growth of all the microorganism species, most of which was stronger than the control antibiotic samples. The inhibition zones of geopropolis extracts were in the range of 7.3–17.0 mm, whereas the control antibiotic inhibition zones were in the range of 4.0–18.3 mm. Better inhibition zones were observed only for E. coli. | [99] |
| S. aureus ATCC 9144 and Bacillus subtilis ATCC 6633, E. coli ATCC 8739, P. aeruginosa ATCC 9027, and Candida albicans ATCC 10231 | Trigona sp. | India | Extract from the geopropolis inhibited all of the growth of all the microorganism species. Candida albicans was the most sensitive (MIC = 0.5 to 8 mg/mL), whereas the least sensitive was E. coli (MIC = 20 to 40 mg/mL). However, the study showed no correlation of antimicrobial activity with phenolics and flavonoid contents. | [84] |
| B. cereus, S. aureus, Micrococcus luteus, E. coli, Enterobacter aerogenes, Alcaligenes faecalis, Aeromonas hydrophila, and Salmonella Typhimurium | Heterotrigona itama | Malaysia | This study showed the extract of beneficial bacteria from the geopropolis, and Bacillus spp. Could inhibit all of the evaluated microorganisms. It is known that Bacillus isolates are commonly found to eliminate unfavourable miroorganisms that could cause destruction of the bee colony. | [64] |
| S. aureus ATCC 25923, MRSA (clinic isolate), E. faecalis ATCC 29212, E. coli ATCC 25922, and K. pneumoniae ATCC 23883 | Melipona quadrifasciata and Tetragonisca angustula | Brazil | Extract from both geopropolis extracts inhibited all the microorganism species’ growth, and M. quadrifasciata showed stronger antimicrobial activity by showing lower MIC values (5–7 mg/mL). | [88] |
| S. aureus ATCC 6538™, S. aureus ESA 175, S. aureus ESA 159, ATCC 43300™, E. faecalis ESA 201, E. faecalis ESA 361, E. coli ATCC 29998™, E. coli ESA 37, E. coli ESA 54, P. aeruginosa ATCC 15442, P. aeruginosa ESA 22, P. aeruginosa ESA 23, Cryptococcus neoformans ATCC 32264, C. neoformans ESA 211, C. neoformans ESA 105, C. albicans ATCC 10231™, C. albicans ESA 100, and C. albicans ESA 97 | Melipona orbignyi (Guérin-Méneville, 1844) | Brazil | Geopropolis extracts inhibited all the microorganism species. In addition, it showed bactericidal and fungicidal activity against all of the evaluated microorganisms. The inhibition observed was in the sequence of S. aureus > E. faecalis > E. coli > P. aeruginosa > C. neoformans > C. albicans, with the MBC value ranging from 8.5 mg/mL for S. aureus to 36.1 mg/mL for C. albicans. | [104] |
| B. subtilis, S. aureus, E. coli, and Salmonella | Heterotrigona itama | Malaysia | Geopropolis extracts inhibited all the microorganism species. Additionally, the geopropolis extract using ethanol showed higher antimicrobial activity than extracts using hexane. Besides the method of extraction, osmotic effect, pH level, presence of the hydrogen peroxide, and phytochemicals likely affected the antimicrobial activity. | [83] |
| Streptococcus mutans | Trigona sirindhornae | Thailand | Extract of the propolis significantly inhibited bacterial growth. The inhibition value of extracts was 43.5 μg/mL. | [105] |
| C. albicans and C. neoformans | Geniotrigona thoracica | Malaysia | Extract from the geopropolis efficiently inhibited, with an MIC value of 1.56 mg/mL for both of the fungal species. The antifungal activity may be attributed to its phenolic and flavonoids compounds. | [101] |
| S. aureus ATCC 43300, S. aureus ESA 654, S. epidermidis ATCC 12228, S. epidermidis ESA 675, Enterococcus faecalis ATCC 43300, E. faecalis ESA 553, K. pneumonia ATCC 4352, K. pneumoniae ESA 154, P. aeruginosa ATCC 15442, P. aeruginosa ESA 22, Proteus mirabilis ATCC 43300, P. mirabilis ESA 37, C. glabrata ATCC 90030, C. glabrata ESA 123, C. albicans ATCC 90028, and C. albicans ESA 345. | Tetragonisca fiebrigi | Brazil | Extract from the geopropolis inhibited all the microorganism species. The inhibition was observed in the sequence of S. aureus > S. epidermidis > E. faecalis > P. mirabilis > K. pneumonia > P. aeruginosa, with the MIC value ranging from 1.5 mg/mL for S. aureus to 15.5 mg/mL for P. aeruginosa. | [85] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rozman, A.S.; Hashim, N.; Maringgal, B.; Abdan, K. A Comprehensive Review of Stingless Bee Products: Phytochemical Composition and Beneficial Properties of Honey, Propolis, and Pollen. Appl. Sci. 2022, 12, 6370. https://doi.org/10.3390/app12136370
Rozman AS, Hashim N, Maringgal B, Abdan K. A Comprehensive Review of Stingless Bee Products: Phytochemical Composition and Beneficial Properties of Honey, Propolis, and Pollen. Applied Sciences. 2022; 12(13):6370. https://doi.org/10.3390/app12136370
Chicago/Turabian StyleRozman, Azri Shahir, Norhashila Hashim, Bernard Maringgal, and Khalina Abdan. 2022. "A Comprehensive Review of Stingless Bee Products: Phytochemical Composition and Beneficial Properties of Honey, Propolis, and Pollen" Applied Sciences 12, no. 13: 6370. https://doi.org/10.3390/app12136370
APA StyleRozman, A. S., Hashim, N., Maringgal, B., & Abdan, K. (2022). A Comprehensive Review of Stingless Bee Products: Phytochemical Composition and Beneficial Properties of Honey, Propolis, and Pollen. Applied Sciences, 12(13), 6370. https://doi.org/10.3390/app12136370







