The Functional Properties of Lactobacillus casei HY2782 Are Affected by the Fermentation Time
Abstract
1. Introduction
2. Materials and Methods
2.1. Probiotic Strains
2.2. Preparation of the Inoculum
2.3. Preparation of Fermented Milk
2.4. pH Determination
2.5. Titratable Acidity
2.6. Microbial Viability
2.7. Viability of Probiotic Bacteria during In Vitro Digestion
2.8. Propagation and Maintenance of Cell Lines
2.9. Adhesion Assay
2.10. mRNA Expression Analyses
2.11. Statistical Analyses
3. Results
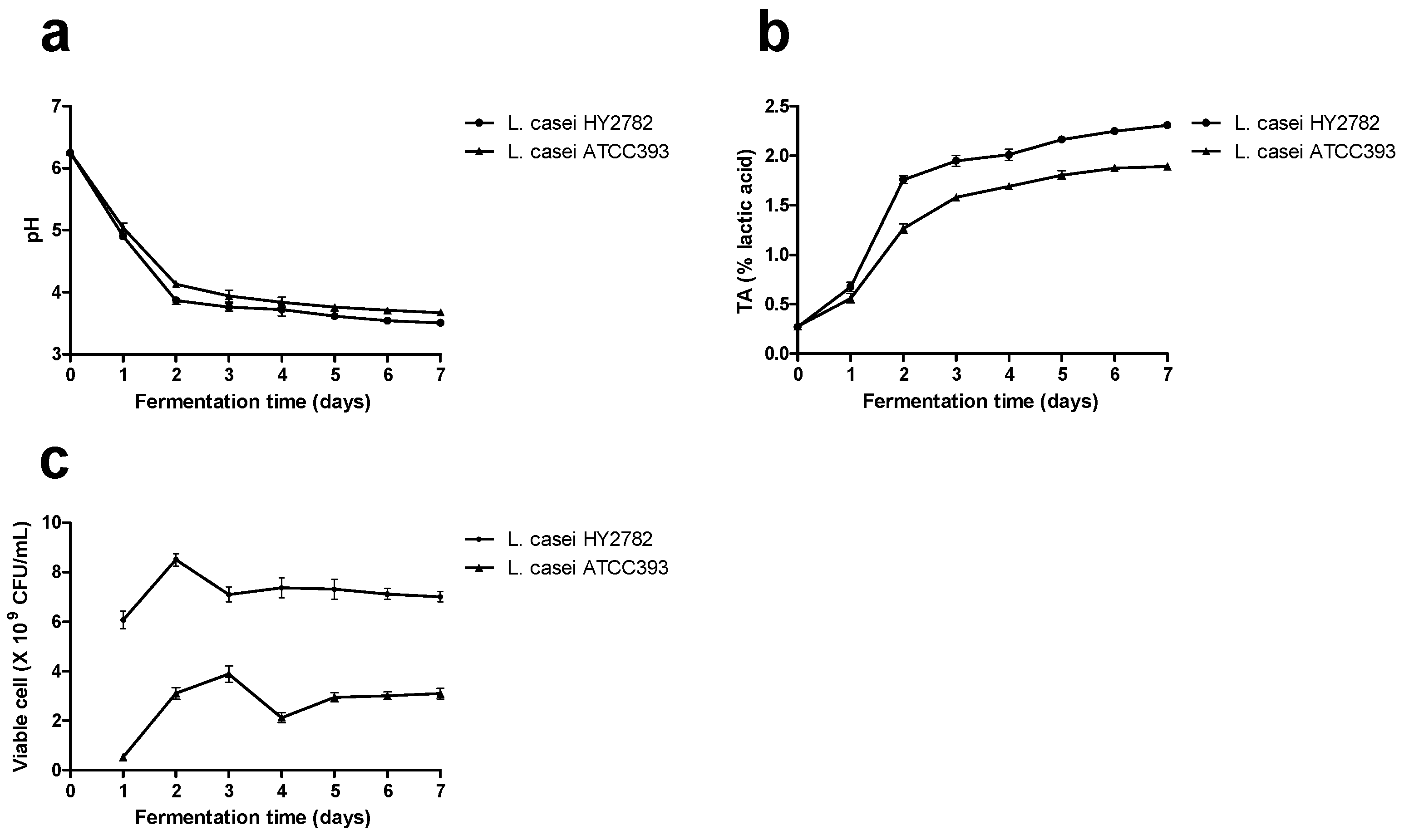
3.1. Acidification Capability and Growth Performance
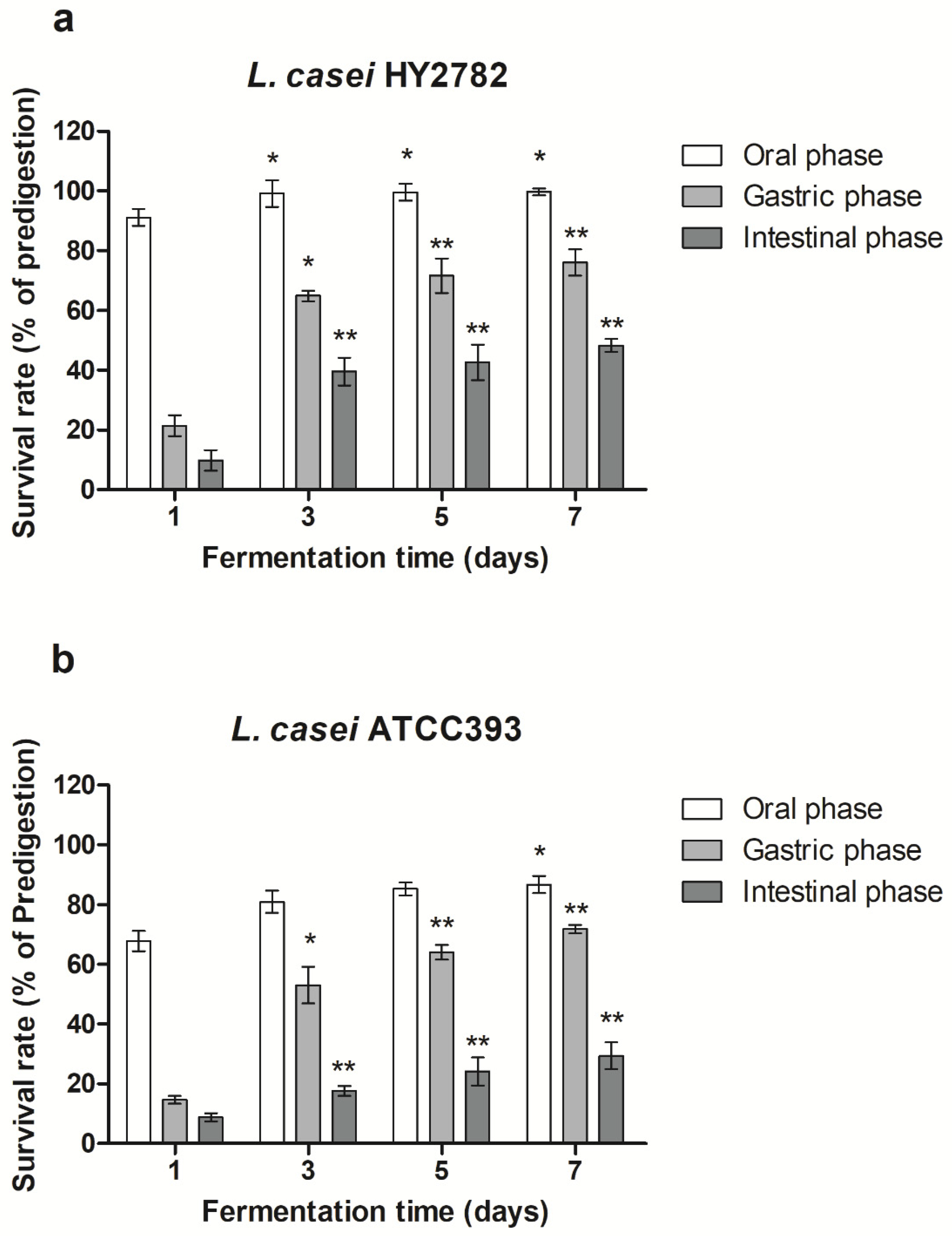
3.2. Bacterial Survival Rate under Simulated Gastrointestinal Digestion
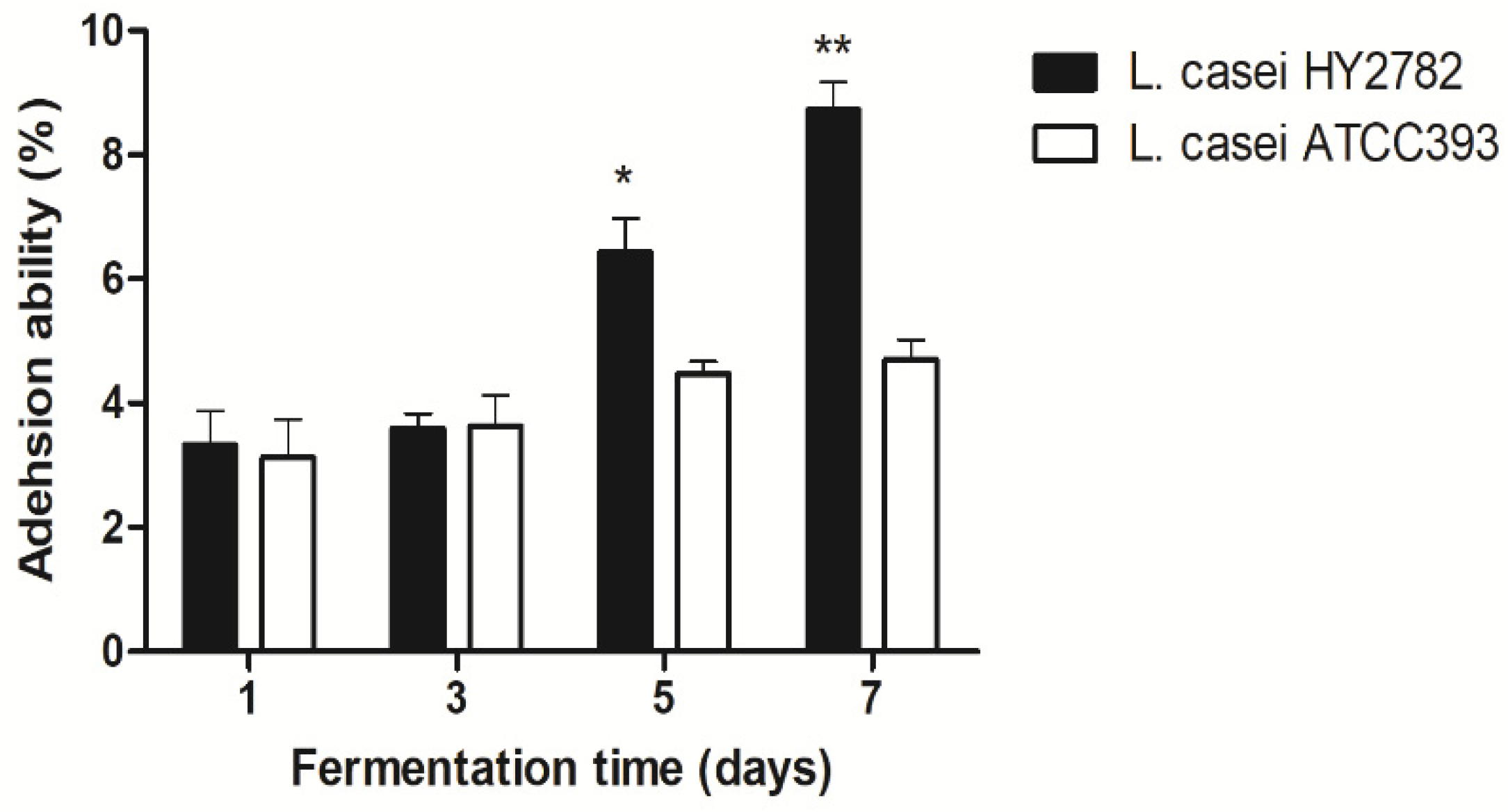
3.3. Intestinal Adhesion Ability on HT-29 Cells
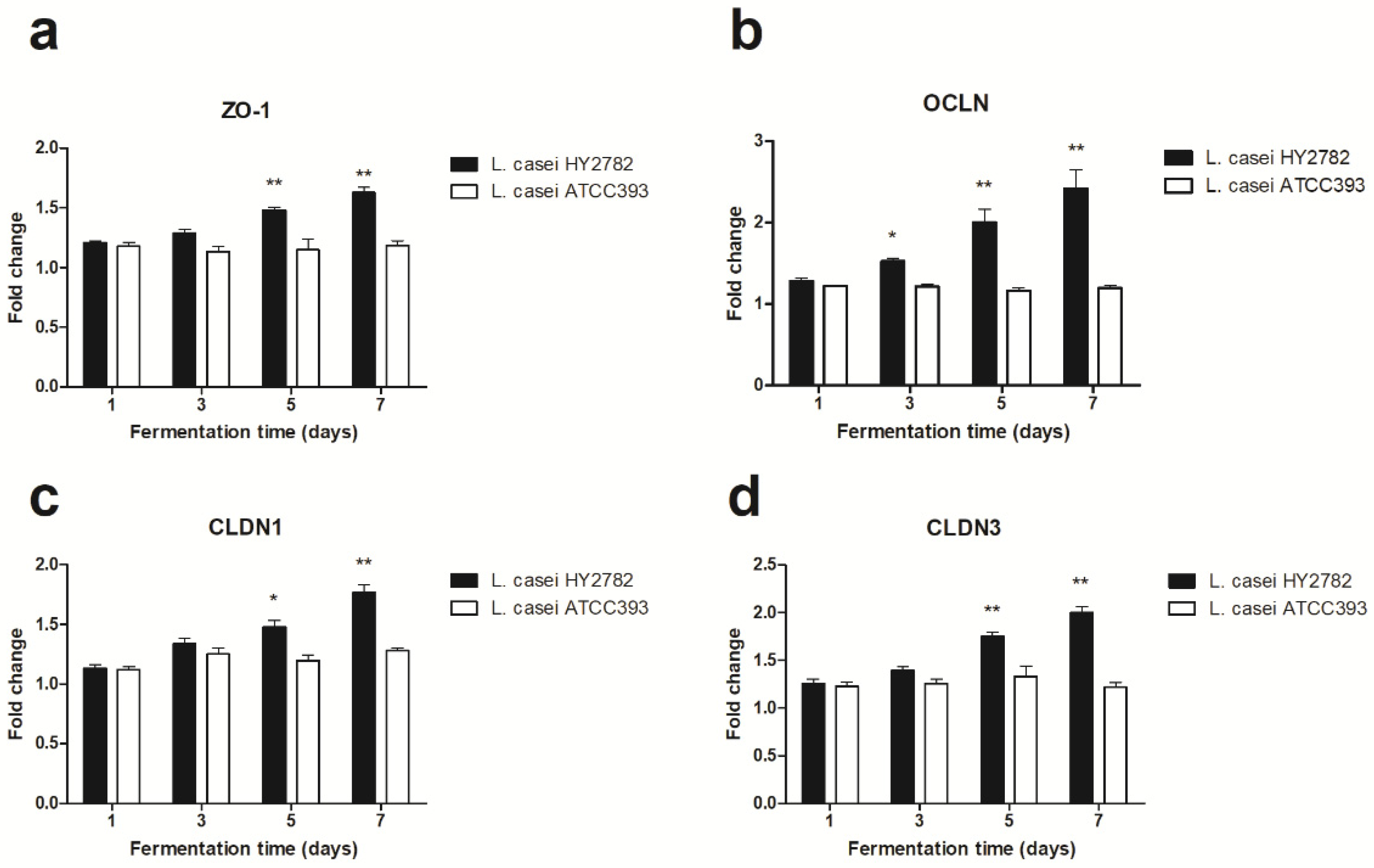
3.4. Tight-Junction-Related mRNA Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert Consensus Document. The International Scientific Association for Probiotics and Prebiotics Consensus Statement on the Scope and Appropriate Use of the Term Probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- David, L.A.; Maurice, C.F.; Carmody, R.N.; Gootenberg, D.B.; Button, J.E.; Wolfe, B.E.; Ling, A.V.; Devlin, A.S.; Varma, Y.; Fischbach, M.A.; et al. Diet Rapidly and Reproducibly Alters the Human Gut Microbiome. Nature 2014, 505, 559–563. [Google Scholar] [CrossRef]
- Derrien, M.; van Hylckama Vlieg, J.E. Fate, Activity, and Impact of Ingested Bacteria within the Human Gut Microbiota. Trends Microbiol. 2015, 23, 354–366. [Google Scholar] [CrossRef]
- Ungaro, F.; Rubbino, F.; Danese, S.; D’Alessio, S. Actors and Factors in the Resolution of Intestinal Inflammation: Lipid Mediators as a New Approach to Therapy in Inflammatory Bowel Diseases. Front. Immunol. 2017, 8, 1331. [Google Scholar] [CrossRef]
- Parolin, C.; Marangoni, A.; Laghi, L.; Foschi, C.; Ñahui Palomino, R.A.; Calonghi, N.; Cevenini, R.; Vitali, B. Isolation of Vaginal Lactobacilli and Characterization of Anti-Candida Activity. PLoS ONE 2015, 10, e0131220. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-M.; Tsai, C.-L.; Wee, W.-C.; Yan, T.-R. Isolation and functional study of potentially probiotic. Afr. J. Microbiol. Res. 2013, 7, 683–691. [Google Scholar]
- Akabanda, F.; Owusu-Kwarteng, J.; Tano-Debrah, K.; Parkouda, C.; Jespersen, L. The Use of Lactic Acid Bacteria Starter Culture in the Production of Nunu, a Spontaneously Fermented Milk Product in Ghana. Int. J. Food Sci. 2014, 2014, 721067. [Google Scholar] [CrossRef] [PubMed]
- Ao, X.; Zhang, X.; Zhang, X.; Shi, L.; Zhao, K.; Yu, J.; Dong, L.; Cao, Y.; Cai, Y. Identification of Lactic Acid Bacteria in Traditional Fermented Yak Milk and Evaluation of Their Application in Fermented Milk Products. J. Dairy Sci. 2012, 95, 1073–1084. [Google Scholar] [CrossRef]
- Bao, Q.; Song, Y.; Xu, H.; Yu, J.; Zhang, W.; Menghe, B.; Zhang, H.; Sun, Z. Multilocus Sequence Typing of Lactobacillus casei Isolates from Naturally Fermented Foods in China and Mongolia. J. Dairy Sci. 2016, 99, 5202–5213. [Google Scholar] [CrossRef]
- Aryana, K.J.; Olson, D.W. A 100-Year Review: Yogurt and Other Cultured Dairy Products. J. Dairy Sci. 2017, 100, 9987–10013. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.M.; Ross, R.P.; Fitzgerald, G.F.; Caplice, N.M.; Stanton, C. Sugar-Coated: Exopolysaccharide Producing Lactic Acid Bacteria for Food and Human Health Applications. Food Funct. 2015, 6, 679–693. [Google Scholar] [CrossRef]
- Ouwehand, A.C.; Kirjavainen, P.V.; Shortt, C.; Salminen, S. Probiotics: Mechanisms and Established Effects. Int. Dairy J. 1999, 9, 43–52. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Cummings, J.H. Probiotics, Infection and Immunity. Curr. Opin. Infect. Dis. 2002, 15, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Lebeer, S.; Vanderleyden, J.; De Keersmaecker, S.C. Genes and Molecules of Lactobacilli Supporting Probiotic Action. Microbiol. Mol. Biol. Rev. 2008, 72, 728–764. [Google Scholar] [CrossRef] [PubMed]
- Karczewski, J.; Troost, F.J.; Konings, I.; Dekker, J.; Kleerebezem, M.; Brummer, R.J.; Wells, J.M. Regulation of Human Epithelial Tight Junction Proteins by Lactobacillus plantarum In Vivo and Protective Effects on the Epithelial Barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G851–G859. [Google Scholar] [CrossRef] [PubMed]
- Lourens-Hattingh, A.; Viljoen, B.C. Yogurt as Probiotic Carrier Food. Int. Dairy J. 2001, 11, 1–17. [Google Scholar] [CrossRef]
- Jost, R. Milk and dariy products. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH Verlag GmbH & Co. KgaA: Weinheim, Germany, 2007; p. 300. [Google Scholar]
- Park, I.J.; Lee, J.H.; Kye, B.H.; Oh, H.K.; Cho, Y.B.; Kim, Y.T.; Kim, J.Y.; Sung, N.Y.; Kang, S.B.; Seo, J.M.; et al. Effects of Probiotics on the Symptoms and Surgical ouTComes after Anterior REsection of Colon Cancer (POSTCARE): A Randomized, Double-Blind, Placebo-Controlled Trial. J. Clin. Med. 2020, 9, 2181. [Google Scholar] [CrossRef]
- Kim, J.Y.; Lee, S.Y.; Jung, S.H.; Kim, M.R.; Choi, I.D.; Lee, J.L.; Sim, J.H.; Pan, C.H.; Kang, K. Protective Effect of Lactobacillus casei HY2782 against Particulate Matter Toxicity in Human Intestinal CCD-18Co Cells and Caenorhabditis elegans. Biotechnol. Lett. 2020, 42, 519–528. [Google Scholar] [CrossRef]
- Nam, W.; Kim, H.; Bae, C.; Kim, J.; Nam, B.; Lee, Y.; Kim, J.; Park, S.; Lee, J.; Sim, J. Lactobacillus HY2782 and Bifidobacterium HY8002 Decrease Airway Hyperresponsiveness Induced by Chronic PM2.5 Inhalation in Mice. J. Med. Food 2020, 23, 575–583. [Google Scholar]
- Østlie, H.M.; Helland, M.H.; Narvhus, J.A. Growth and Metabolism of Selected Strains of Probiotic Bacteria in Milk. Int. J. Food Microbiol. 2003, 87, 17–27. [Google Scholar] [CrossRef]
- Jung, S.; Lee, J.H. Characterization of Transcriptional Response of Lactobacillus plantarum under Acidic Conditions Provides Insight into Bacterial Adaptation in Fermentative Environments. Sci. Rep. 2020, 10, 19203. [Google Scholar] [CrossRef]
- Hill, D.; Sugrue, I.; Tobin, C.; Hill, C.; Stanton, C.; Ross, R.P. The Lactobacillus casei Group: History and Health Related Applications. Front. Microbiol. 2018, 9, 2107. [Google Scholar] [CrossRef] [PubMed]
- Bang, M.; Yong, C.C.; Ko, H.J.; Choi, I.G.; Oh, S. Transcriptional Response and Enhanced Intestinal Adhesion Ability of Lactobacillus rhamnosus GG after Acid Stress. J. Microbiol. Biotechnol. 2018, 28, 1604–1613. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, B.; Bressollier, P.; Urdaci, M.C. Exported Proteins in Probiotic Bacteria: Adhesion to Intestinal Surfaces, Host Immunomodulation and Molecular Cross-Talking with the Host. FEMS. Immunol. Med. Microbiol. 2008, 54, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Michael, M.; Phebus, R.K.; Schmidt, K.A. Impact of a Plant Extract on the Viability of Lactobacillus delbrueckii ssp. bulgaricus and Streptococcus thermophilus in Nonfat Yogurt. Int. Dairy J. 2010, 20, 665–672. [Google Scholar]
- Panda, S.H.; Ray, R.C. Lactic Acid Fermentation of Beta-Carotene Rich Sweet Potato (Ipomoea batatas L.) into Lacto-Juice. Plant Foods Hum. Nutr. 2007, 62, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A Standardised Static In Vitro Digestion Method Suitable for Food—An International Consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, C.N.; Rosenfeldt Nielsen, V.; Hayford, A.E.; Møller, P.L.; Michaelsen, K.F.; Paerregaard, A.; Sandström, B.; Tvede, M.; Jakobsen, M. Screening of Probiotic Activities of Forty-Seven Strains of Lactobacillus spp. by In Vitro Techniques and Evaluation of the Colonization Ability of Five Selected Strains in Humans. Appl. Environ. Microbiol. 1999, 65, 4949–4956. [Google Scholar] [CrossRef]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Saarela, M.; Mogensen, G.; Fondén, R.; Mättö, J.; Mattila-Sandholm, T. Probiotic Bacteria: Safety, Functional and Technological Properties. J. Biotechnol. 2000, 84, 197–215. [Google Scholar] [CrossRef]
- Colum Dunne, L.O.; Mahony, L.M.; Thornton, G.; Morrissey, D.; O’Halloran, S.; Feeney, M.; Flynn, G.F.S.; Daly, C.; Kiely, B.; O’Gerald, C.; et al. In Vitro Selection Criteria for Probiotic Bacteria of Human Origin Correlation with In Vivo Findings. Am. J. Clin. Nutr. 2001, 73, 386S–392S. [Google Scholar] [CrossRef]
- Faghfoori, Z.; Pourghassem Gargari, B.; Saber Gharamaleki, A.; Bagherpour, H.; Yari Khosroushahi, A. Cellular and Molecular Mechanisms of Probiotics Effects on Colorectal Cancer. J. Funct. Foods 2015, 18, 463–472. [Google Scholar] [CrossRef]
- Stanton, C.; Ross, R.P.; Fitzgerald, G.F.; Van Sinderen, D. Fermented Functional Foods Based on Probiotics and Their Biogenic Metabolites. Curr. Opin. Biotechnol. 2005, 16, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Ewaschuk, J.B.; Diaz, H.; Meddings, L.; Diederichs, B.; Dmytrash, A.; Backer, J.; Looijer-van Langen, M.; Madsen, K.L. Secreted Bioactive Factors from Bifidobacterium infantis Enhance Epithelial Cell Barrier Function. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 295, G1025–G1034. [Google Scholar] [CrossRef]
- Ruas-Madiedo, P.; Hugenholtz, J.; Zoon, P. An Overview of the Functionality of Exopolysaccharides Produced by Lactic Acid Bacteria. Int. Dairy J. 2002, 12, 163–171. [Google Scholar] [CrossRef]
- Lee, J.H.; O’Sullivan, D.J. Genomic Insights into Bifidobacteria. Microbiol. Mol. Biol. Rev. 2010, 74, 378–416. [Google Scholar] [CrossRef] [PubMed]
- Sidira, M.; Kourkoutas, Y.; Kanellaki, M.; Charalampopoulos, D. In vitro study on the cell adhesion ability of immobilized lactobacilli on natural supports. Food Res. Int. 2015, 76, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Ren, Q.; Tang, Y.; Zhang, L.; Xu, Y.; Liu, N.; Ren, H. Exopolysaccharide Produced by Lactobacillus casei Promotes the Differentiation of CD4(+) T Cells into Th17 Cells in BALB/C Mouse Peyer’s Patches In Vivo and In Vitro. J. Agric. Food Chem. 2020, 68, 2664–2672. [Google Scholar] [CrossRef]
- Mozzi, F.; de Giori, G.S.; Oliver, G.; de Valdez, G.F. Exopolysaccharide Production by Lactobacillus Casei under Controlled pH. Biotechnol. Lett. 1996, 18, 435–439. [Google Scholar] [CrossRef]
- Mozzi, F.; Gerbino, E.; Font de Valdez, G.; Torino, M.I. Functionality of Exopolysaccharides Produced by Lactic Acid Bacteria in an In Vitro Gastric System. J. Appl. Microbiol. 2009, 107, 56–64. [Google Scholar] [CrossRef]
- Degeest, B.; Janssens, B.; De Vuyst, L. Exopolysaccharide (EPS) biosynthesis by Lactobacillus sakei 0-1: Production kinetics, enzyme activities and EPS yields. J. Appl. Microbiol. 2001, 91, 470–477. [Google Scholar] [CrossRef]
- Eun, C.S.; Kim, Y.S.; Han, D.S.; Choi, J.H.; Lee, A.R.; Park, Y.K. Lactobacillus casei Prevents Impaired Barrier Function in Intestinal Epithelial Cells. APMIS 2011, 119, 49–56. [Google Scholar] [CrossRef]
- Putt, K.K.; Pei, R.; White, H.M.; Bolling, B.W. Yogurt Inhibits Intestinal Barrier Dysfunction in Caco-2 Cells by Increasing Tight Junctions. Food Funct. 2017, 8, 406–414. [Google Scholar] [CrossRef]
- Tang, C.; Lu, Z. Health Promoting Activities of Probiotics. J. Food Biochem. 2019, 43, e12944. [Google Scholar] [CrossRef] [PubMed]
- Thompson, W.; Longstreth, G.; Drossman, D.; Heaton, K.; Irvine, E.; Müller-Lissner, S. Functional Bowel Disorders and Functional Abdominal Pain. Gut 1999, 45, 43–47. [Google Scholar] [CrossRef]
- Camilleri, M.; Lasch, K.; Zhou, W. Irritable Bowel Syndrome: Methods, Mechanisms, and Pathophysiology. The Confluence of Increased Permeability, Inflammation, and Pain in Irritable Bowel Syndrome. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 303, G775–G785. [Google Scholar]
- Gecse, K.; Róka, R.; Séra, T.; Rosztóczy, A.; Annaházi, A.; Izbéki, F.; Nagy, F.; Molnár, T.; Szepes, Z.; Pávics, L.; et al. Leaky Gut in Patients with Diarrhea-Predominant Irritable Bowel Syndrome and Inactive Ulcerative Colitis. Digestion 2012, 85, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Ji, Y.R.; Lee, S.; Choi, M.J.; Cho, Y. Microencapsulation of Probiotic Lactobacillus acidophilus KBL409 by Extrusion Technology to Enhance Survival under Simulated Intestinal and Freeze-Drying Conditions. J. Microbiol. Biotechnol. 2019, 29, 721–730. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Toalá, J.E.; Garcia-Varela, R.; Garcia, H.S.; Mata-Haro, V.; González-Córdova, A.F.; Vallejo-Cordoba, B.; Hernández-Mendoza, A. Postbiotics: An Evolving Term within the Functional Foods Field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, S.H.; Hong, D.K.; Bang, S.-J.; Heo, K.; Sim, J.-J.; Lee, J.-L. The Functional Properties of Lactobacillus casei HY2782 Are Affected by the Fermentation Time. Appl. Sci. 2021, 11, 2481. https://doi.org/10.3390/app11062481
Jung SH, Hong DK, Bang S-J, Heo K, Sim J-J, Lee J-L. The Functional Properties of Lactobacillus casei HY2782 Are Affected by the Fermentation Time. Applied Sciences. 2021; 11(6):2481. https://doi.org/10.3390/app11062481
Chicago/Turabian StyleJung, Seung Hee, Dong Ki Hong, So-Jung Bang, Keon Heo, Jae-Jung Sim, and Jung-Lyoul Lee. 2021. "The Functional Properties of Lactobacillus casei HY2782 Are Affected by the Fermentation Time" Applied Sciences 11, no. 6: 2481. https://doi.org/10.3390/app11062481
APA StyleJung, S. H., Hong, D. K., Bang, S.-J., Heo, K., Sim, J.-J., & Lee, J.-L. (2021). The Functional Properties of Lactobacillus casei HY2782 Are Affected by the Fermentation Time. Applied Sciences, 11(6), 2481. https://doi.org/10.3390/app11062481





