Anti-Inflammatory and Antioxidant Properties of Ethanol Extracts of Raw, Blanched, Steamed, and Sous-Vide Cooked Okra (Abelmoschus esculentus L.) in LPS or H2O2-Treated RAW264.7 Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.1.1. Blanching
2.1.2. Steaming
2.1.3. Sous-Vide
2.1.4. Preparation of Okra Extract
2.1.5. Chemicals and Antibodies
2.2. RAW264.7 Cell Culture
2.3. Cell Viability Assay
2.4. Nitric Oxide (NO) Assay
2.5. Preparation of Protein Samples
2.6. Western Blot Analysis
2.7. Reverse-Transcription-Polymerase Chain Reaction (RT-PCR) Analysis
2.8. Reactive Oxygen Species (ROS) Assay
2.9. Statistical Analysis
3. Results and Discussion
3.1. RAW264.7 Cell Viability
3.2. Nitric Oxide (NO) Production
3.3. COX-2 and iNOS in Protein Level
3.4. iNOS, TNF-α, IL-6 and IL-1βin mRNA Level
3.5. Reactive Oxygen Species (ROS)
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2008, 454, 428–435. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.W.; Whisnant, R.; Nathan, C. Promoter of the mouse gene encoding calcium-independent nitric oxide synthase confers inducibility by interferon gamma and bacterial lipopolysaccharide. J. Exp. Med. 1993, 177, 1779–1784. [Google Scholar] [CrossRef] [PubMed]
- Nathan, C. Nitric oxide as a secretory product of mammalian cells. FASEB J. 1992, 6, 3051–3064. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.U.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Habtamu, F.G.; Negussie, R.; Gulelat, D.H.; Woldegiorgis, A.Z.; Fekadu, B. Nutritional quality and health benefits of okra (Abelmoschus esculentus): A review. Pak. J. Food Sci. 2015, 25, 16–25. [Google Scholar]
- Kumar, S.; Dagnoko, S.; Haougui, A.; Ratnadass, A.; Pasternak, N.; Kouame, C. Okra(Abelmoschus spp.) in West and Cen-tral Africa: Potential and progress on its improvement. Afr. J. Agric. Res. 2010, 5, 3590–3598. [Google Scholar]
- Kang, X.U.; Jin-Hua, D.U. Effects of drying methods on antioxidant capacities of okrafruit. Food Ferment Ind. 2016, 42, 120–125. [Google Scholar]
- Gemede, H.F.; Haki, G.D.; Beyene, F.; Woldegiorgis, A.Z.; Rakshit, S.K. Proximate, mineral, and antinutrient compositions of indigenous Okra ( Abelmoschus esculentus ) pod accessions: Implications for mineral bioavailability. Food Sci. Nutr. 2016, 4, 223–233. [Google Scholar] [CrossRef]
- Li, H.; Xie, L.; Ma, Y.; Zhang, M.; Zhao, Y.; Zhao, X. Effects of drying methods on drying characteristics, physicochemical properties and antioxidant capacity of okra. LWT 2019, 101, 630–638. [Google Scholar] [CrossRef]
- Islam, M.T. Phytochemical information and pharmacological activities of Okra (Abelmoschus esculentus): A literature-based review. Phytother. Res. 2019, 33, 72–80. [Google Scholar] [CrossRef]
- Petropoulos, S.; Fernandes, Â.; Barros, L.; Ferreira, I.C. Chemical composition, nutritional value and antioxidant properties of Mediterranean okra genotypes in relation to harvest stage. Food Chem. 2018, 242, 466–474. [Google Scholar] [CrossRef]
- Mishra, N.; Kumar, D.; Rizvi, S.I. Protective Effect of Abelmoschus esculentus Against Alloxan-induced Diabetes in Wistar Strain Rats. J. Diet. Suppl. 2016, 13, 1–13. [Google Scholar] [CrossRef]
- Chen, H.; Jiao, H.; Cheng, Y.; Xu, K.; Jia, X.; Shi, Q.; Guo, S.; Wang, M.; Du, L.; Wang, F. In Vitro and In Vivo Immunomodulatory Activity of Okra (Abelmoschus esculentus L.) Polysaccharides. J. Med. Food 2016, 19, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Mairuae, N.; Cheepsunthorn, P.; Cheepsunthorn, C.L.; Tongjaroenbuangam, W. Okra (Abelmoschus esculentus Linn) inhibits lipopolysaccharide-induced inflammatory mediators in BV2 microglial cells. Trop. J. Pharm. Res. 2017, 16, 1285. [Google Scholar] [CrossRef]
- Chen, Y.; Zhou, R.; He, L.; Wang, F.; Yang, X.; Teng, L.; Li, C.; Liao, S.; Zhu, Y.; Yang, Y.; et al. Okra polysaccharide-2 plays a vital role on the activation of RAW264.7 cells by TLR2/4-mediated signal transduction pathways. Int. Immunopharmacol. 2020, 86, 106708. [Google Scholar] [CrossRef] [PubMed]
- Shahidi, F.; Yeo, J. Bioactivities of Phenolics by Focusing on Suppression of Chronic Diseases: A Review. Int. J. Mol. Sci. 2018, 19, 1573. [Google Scholar] [CrossRef]
- Eo, H.J.; Kim, D.S.; Kang, Y.; Kim, K.; Park, Y.; Park, G.H. Antioxidant and immunoregulatory effects of Korean Rhamnaceae. J. Plant Biotechnol. 2020, 47, 254–259. [Google Scholar] [CrossRef]
- Zhu, F.; Du, B.; Xu, B. Anti-inflammatory effects of phytochemicals from fruits, vegetables, and food legumes: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1260–1270. [Google Scholar] [CrossRef]
- Shin, J.; Joo, N. Inhibitory Effects on Human Breast Cancer Cells Migration of Small Black Bean according to the Cooking Methods. Korean J. Food Nutr. 2017, 30, 728–734. [Google Scholar] [CrossRef]
- Kim, D.-S.; Joo, N. Nutritional composition of Sacha inchi (Plukenetia Volubilis L.) as affected by different cooking methods. Int. J. Food Prop. 2019, 22, 1235–1241. [Google Scholar] [CrossRef]
- Lafarga, T.; Bobo, G.; Viñas, I.; Zudaire, L.; Simó, J.; Aguiló-Aguayo, I. Steaming and sous-vide: Effects on antioxidant activity, vitamin C, and total phenolic content of Brassica vegetables. Int. J. Gastron. Food Sci. 2018, 13, 134–139. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Newstead, S.L.; Gates, A.J.; Hartley, M.G.; Rowland, C.A.; Williamson, E.D.; Lukaszewski, R.A. Control of IntracellularFrancisella tularensisby Different Cell Types and the Role of Nitric Oxide. J. Immunol. Res. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Mustafa, I.; Chin, N.L.; Fakurazi, S.; Palanisamy, A. Comparison of Phytochemicals, Antioxidant and Anti-Inflammatory Properties of Sun-, Oven- and Freeze-Dried Ginger Extracts. Foods 2019, 8, 456. [Google Scholar] [CrossRef] [PubMed]
- Han, A.-R.; Kil, Y.-S.; Hong, M.J.; Park, J.; Park, H.H.; Jin, C.H.; Nam, J.-W.; Kim, J.-B. Anti-Inflammatory Flavonolignans from Triticum aestivum Linn. Hull. Appl. Sci. 2020, 10, 8656. [Google Scholar] [CrossRef]
- Papi, S.; Ahmadizar, F.; Hasanvand, A. The role of nitric oxide in inflammation and oxidative stress. Immunopathol. Persa 2019, 5, e08. [Google Scholar] [CrossRef]
- Cheon, J.M.; Kim, H.S.; Choi, E.O.; Kwon, D.H.; Choi, Y.H.; Kim, B.W.; Hwang, H.J. Anti-inflammatory Activities of an Ethanol Extract of Sargassum macrocarpum in Lipopolysaccharide (LPS)-stimulated RAW 264.7 Macrophages. J. Life Sci. 2017, 27, 1437–1444. [Google Scholar] [CrossRef]
- Ryu, J.H.; An, J.H.; Woo, Y.K.; Cho, H.J. The anti-inflammation effects of ACC extracts on the LPS-induced Raw 264.7 cell. J. Korea Acad. Ind. Coop. Soc. 2017, 18, 503–511. [Google Scholar] [CrossRef]
- Jeong, D.H.; Kang, B.K.; Kim, K.B.W.R.; Kim, M.J.; Ahn, D.H. Anti-inflammatory Activity of Sargassum micracanthum Water Extract. J. Appl. Biol. Chem. 2014, 57, 227–234. [Google Scholar] [CrossRef]
- Cho, E.J.; Lee, J.H.; Sung, N.Y.; Byun, E.H. Anti-inflammatory effects of Annona muricata leaf ethanol extracts. J. Korean Soc. Food Sci. Nutr. 2017, 46, 681–687. [Google Scholar] [CrossRef]
- Byun, Y.H.; Lee, M.H.; Kim, S.S.; Kim, H.; Chang, H.K.; Lee, T.H.; Jang, M.H.; Shin, M.C.; Shin, M.S.; Kim, C.J. Treadmill running promotes functional recovery and decreases brain-derived neurotrophic factor mRNA expression following sciatic crushed nerve injury in rats. J. Sports Med. Phys. Fit. 2005, 45, 222–228. [Google Scholar]
- Shim, J.H. Anti-inflammatory Effect of Brazilin in RAW264.7 Cells. Asian J. Beauty Cosmetol. 2019, 17, 107–115. [Google Scholar] [CrossRef]
- Schieber, M.; Chandel, N.S. ROS Function in Redox Signaling and Oxidative Stress. Curr. Biol. 2014, 24, R453–R462. [Google Scholar] [CrossRef] [PubMed]
- Matta, F.V.; Xiong, J.; Lila, M.A.; Ward, N.I.; Felipe-Sotelo, M.; Esposito, D. Chemical Composition and Bioactive Properties of Commercial and Non-Commercial Purple and White Açaí Berries. Foods 2020, 9, 1481. [Google Scholar] [CrossRef] [PubMed]
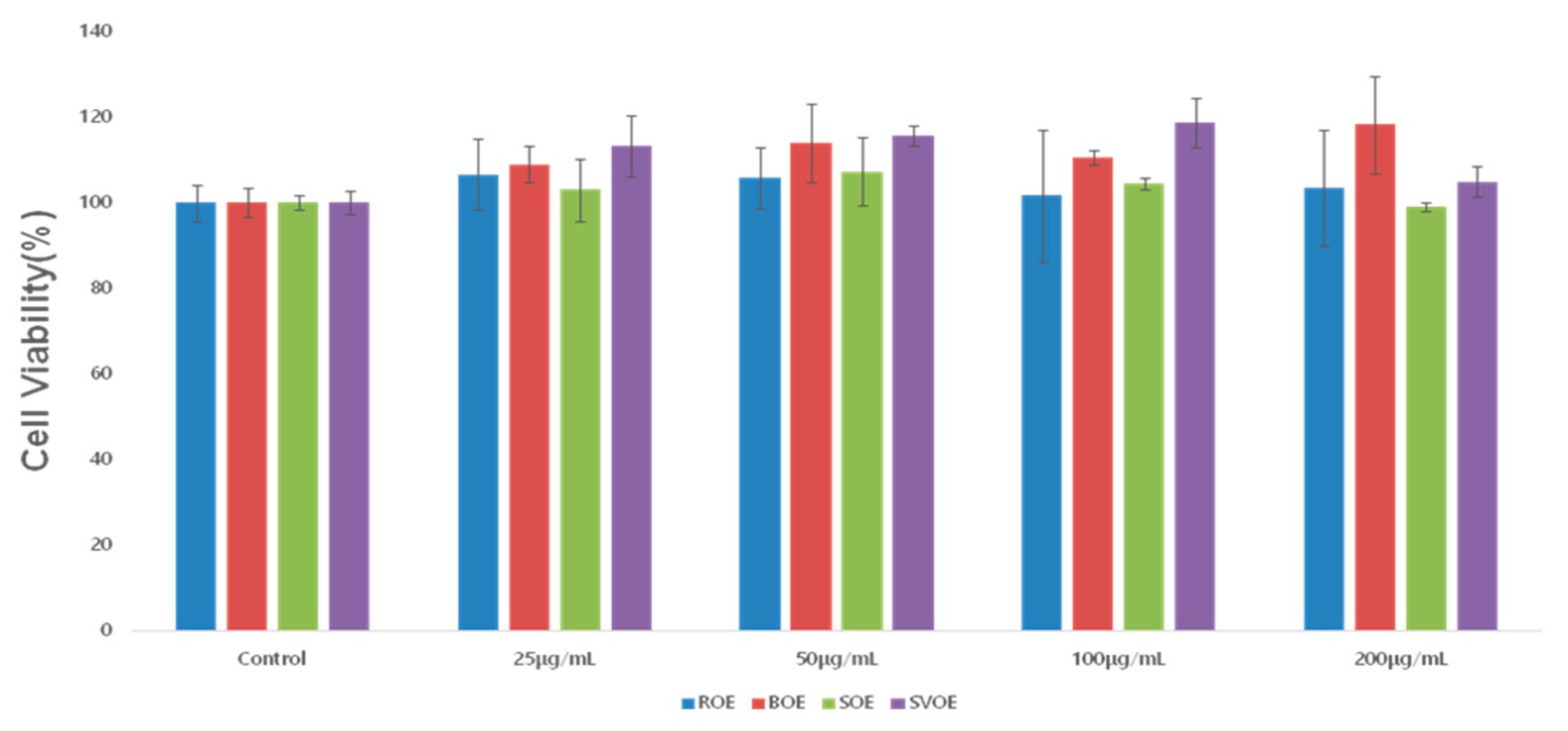
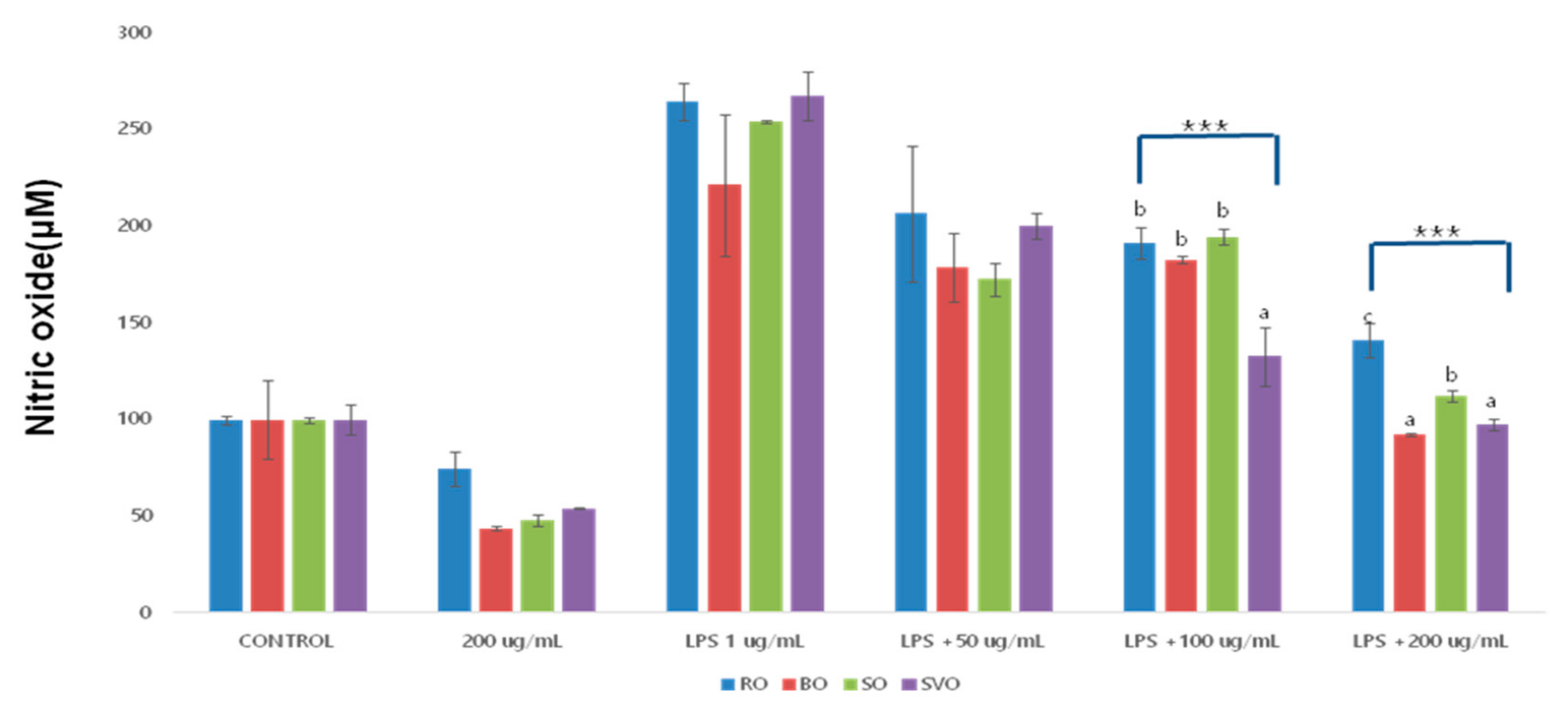


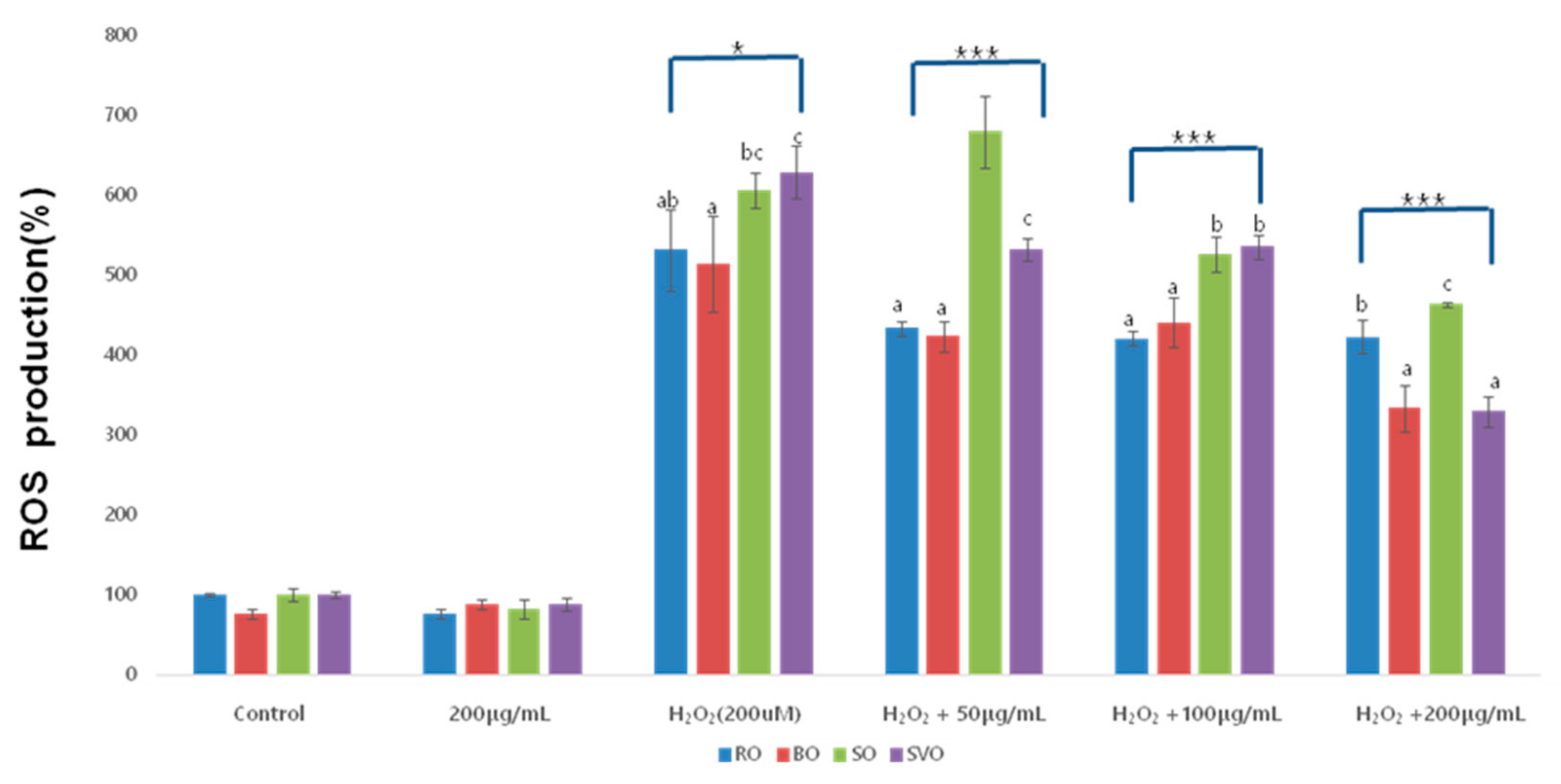
| Okra Extract (μg/mL) | ROE | BOE | SOE | SVOE | F-Value (p) |
|---|---|---|---|---|---|
| 0 | 100 ± 4.28 | 100 ± 3.31 | 100 ± 1.62 | 100 ± 2.63 | 0 |
| (−1) | |||||
| 25 | 106.55 ± 8.27 aA | 108.980 ± 4.33 aA | 103.09 ± 7.26 aA | 113.3 ± 7.1 aAB | 1.156 |
| (−0.385) | |||||
| 50 | 105.76 ± 7.07 aA | 113.85 ± 9.20 aA | 107.25 ± 7.89 aA | 115.55 ± 2.38 aB | 1.374 |
| (−0.319) | |||||
| 100 | 101.72 ± 15.39 aA | 110.58 ± 1.81 abA | 104.44 ± 1.35 abA | 118.68 ± 5.60 bB | 2.488 |
| (−0.135) | |||||
| 200 | 103.46 ± 13.44 abA | 118.30 ± 11.32 bA | 99.01 ± 1.13 aA | 104.91 ± 3.50 abA | 2.572 |
| (−0.127) | |||||
| F-value (p) | 2.02 | 2.82 | 1.39 | 8.54 | |
| (−0.932) | (−0.84) | (−0.31) | (0.003 *) |
| Okra Extract (μg/mL) | ROE | BOE | SOE | SVOE | F-Value (p) |
|---|---|---|---|---|---|
| Control | 100.00 ± 2.16 | 100.00 ± 20.55 | 100.00 ± 1.40 | 100.0.490 ± 8 | |
| 200 | 74.86 ± 8.80 | 43.78 ± 1.02 | 47.85 ± 3.04 | 54.26 ± 0.10 | |
| LPS + 1 | 264.67 ± 9.57 bC | 221.36 ± 36.7 aC | 254.06 ± 0.52 abD | 267.52 ± 12.82 bD | 3.47 |
| (−0.47) | |||||
| LPS + 50 | 206.7 ± 35.3 bB | 179.0 ± 17.88 abB | 172.55 ± 8.18 aB | 200.43 ± 6.83 abC | 3.611 |
| (−0.58) | |||||
| LPS + 100 | 191.49 ± 8.37 bB | 182.85 ± 1.53 bB | 194.63 ± 3.81 bC | 132.71 ± 15.26 aB | 43.17 *** |
| (0.000) | |||||
| LPS + 200 | 141.34 ± 8.80 cA | 92.43 ± 1.02 aA | 112.51 ± 3.04 bA | 97.56 ± 3.23 aA | 68.83 *** |
| (0.000) | |||||
| F-value (p) | 35.88 ** | 21.24 *** | 451.217 *** | 149.592 *** | |
| (0.000) | (0.000) | (0.000) | (0.000) |
| Okra Extract (μg/mL) | ROE | BOE | SOE | SVOE | F-Value (p) |
|---|---|---|---|---|---|
| Control | 100 ± 2.00 | 76.17 ± 5.64 | 100.00 ± 8.83 | 100 ± 4.56 | |
| 200 μg/mL | 76.17 ± 5.52 | 88.07 ± 5.20 | 81.3 ± 12.08 | 88.02 ± 8.40 | |
| H2O2 (200 uM) | 531.38 ± 50.87 abA | 514.25 ± 60.19 aC | 605.85 ± 21.83 bcC | 628.35 ± 32.93 cC | 5.680 * |
| (0.016) | |||||
| H2O2 + 50 | 433.61 ± 8.98 aB | 423.36 ± 19.30 aB | 679.68 ± 44.7 bD | 531.30 ± 14.04 cB | 80.059 *** |
| (0.000) | |||||
| H2O2 + 100 | 420.81 ± 9.65 aB | 440.67 ± 31.06 aB | 526.33 ± 22.52 bB | 535.43 ± 14.72 bB | 32.174 *** |
| (0.000) | |||||
| H2O2 + 200 | 423.00 ± 20.84 bB | 333.35 ± 29.36 aA | 463.77 ± 2.82 cA | 329.29 ± 19.79 aA | 35.408 *** |
| (0.000) | |||||
| F-value (p) | 44.23 *** | 11.39 *** | 35.43 *** | 280.56 *** | |
| (0.000) | (0.003) | (0.000) | (0.000) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, T.; Joo, N. Anti-Inflammatory and Antioxidant Properties of Ethanol Extracts of Raw, Blanched, Steamed, and Sous-Vide Cooked Okra (Abelmoschus esculentus L.) in LPS or H2O2-Treated RAW264.7 Cells. Appl. Sci. 2021, 11, 2432. https://doi.org/10.3390/app11052432
Lee T, Joo N. Anti-Inflammatory and Antioxidant Properties of Ethanol Extracts of Raw, Blanched, Steamed, and Sous-Vide Cooked Okra (Abelmoschus esculentus L.) in LPS or H2O2-Treated RAW264.7 Cells. Applied Sciences. 2021; 11(5):2432. https://doi.org/10.3390/app11052432
Chicago/Turabian StyleLee, Taenam, and Nami Joo. 2021. "Anti-Inflammatory and Antioxidant Properties of Ethanol Extracts of Raw, Blanched, Steamed, and Sous-Vide Cooked Okra (Abelmoschus esculentus L.) in LPS or H2O2-Treated RAW264.7 Cells" Applied Sciences 11, no. 5: 2432. https://doi.org/10.3390/app11052432
APA StyleLee, T., & Joo, N. (2021). Anti-Inflammatory and Antioxidant Properties of Ethanol Extracts of Raw, Blanched, Steamed, and Sous-Vide Cooked Okra (Abelmoschus esculentus L.) in LPS or H2O2-Treated RAW264.7 Cells. Applied Sciences, 11(5), 2432. https://doi.org/10.3390/app11052432





