Fermented Antler Improves Endurance during Exercise Performance by Increasing Mitochondrial Biogenesis and Muscle Strength in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Antler Samples
2.2. Screening of LAB
2.3. Identification of Fermented LAB
2.4. Cell Culture and Treatments
2.5. Preparation of RNA for RT-PCR
2.6. Animals, Diet, and Experimental Design
2.7. Treadmill Exercise Performance Test and Grip Strength Measurement
2.8. Swimming Endurance
2.9. Blood Analysis
2.10. Statistical Analyses
3. Results
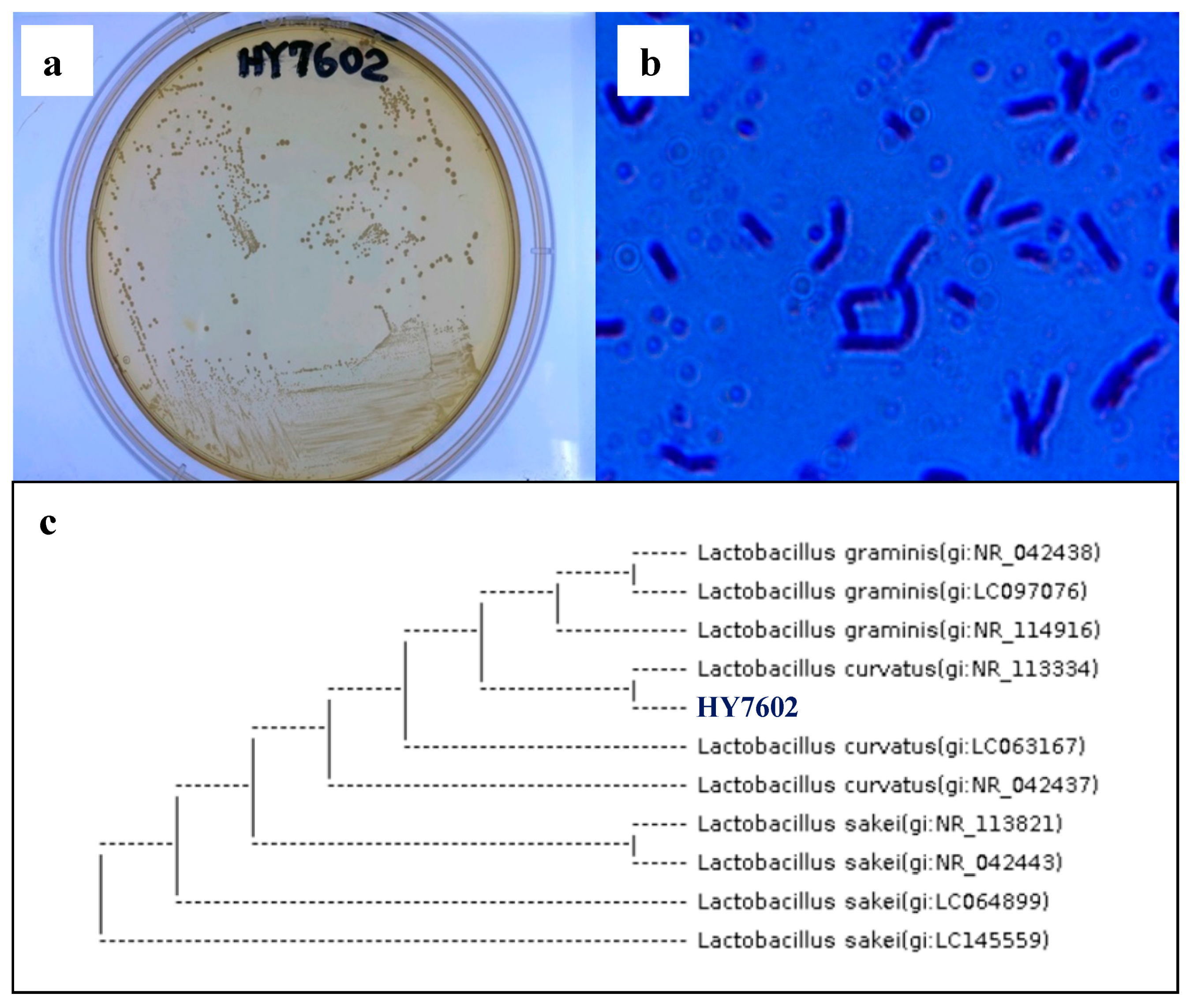
3.1. Screening of Antler-Fermenting LAB
3.2. Identification of Deer Antler-Fermenting LAB
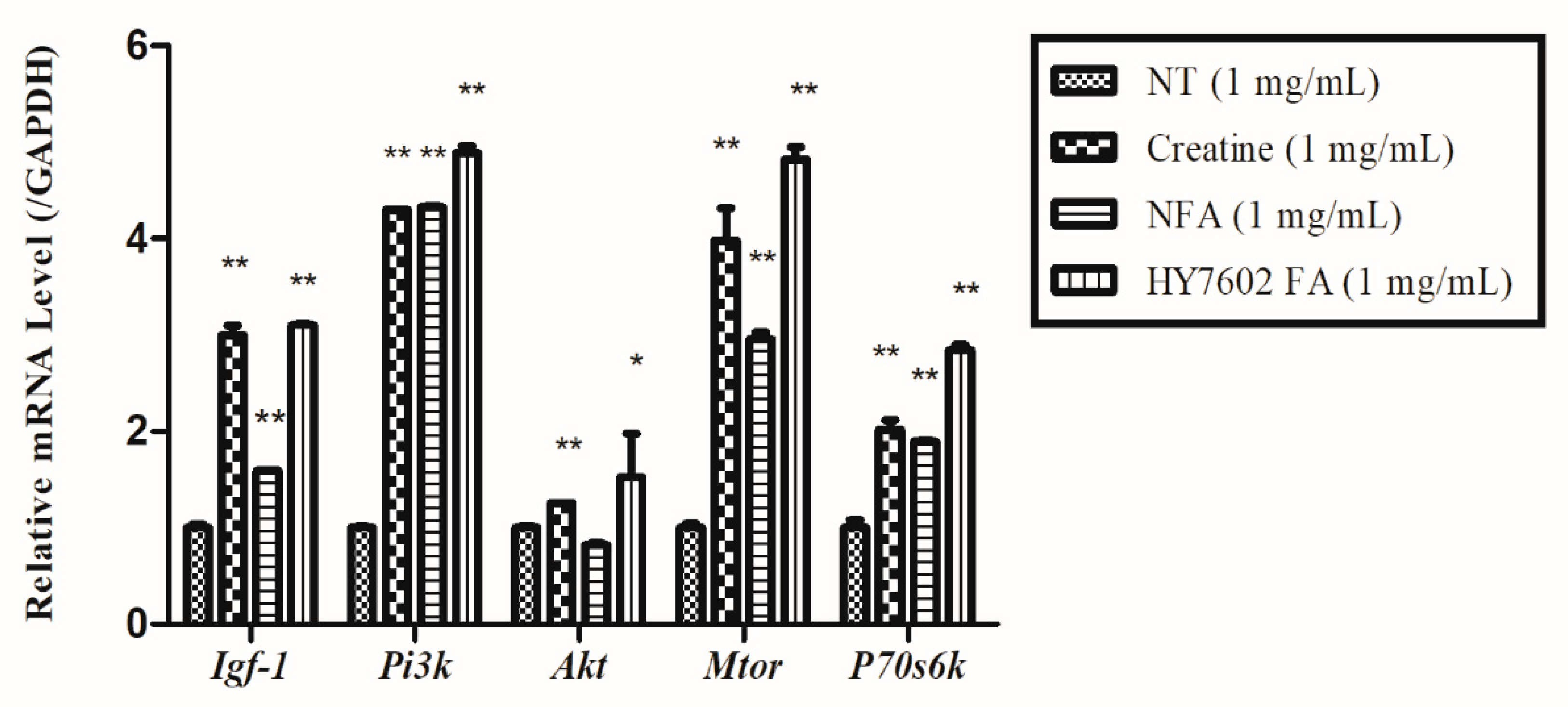
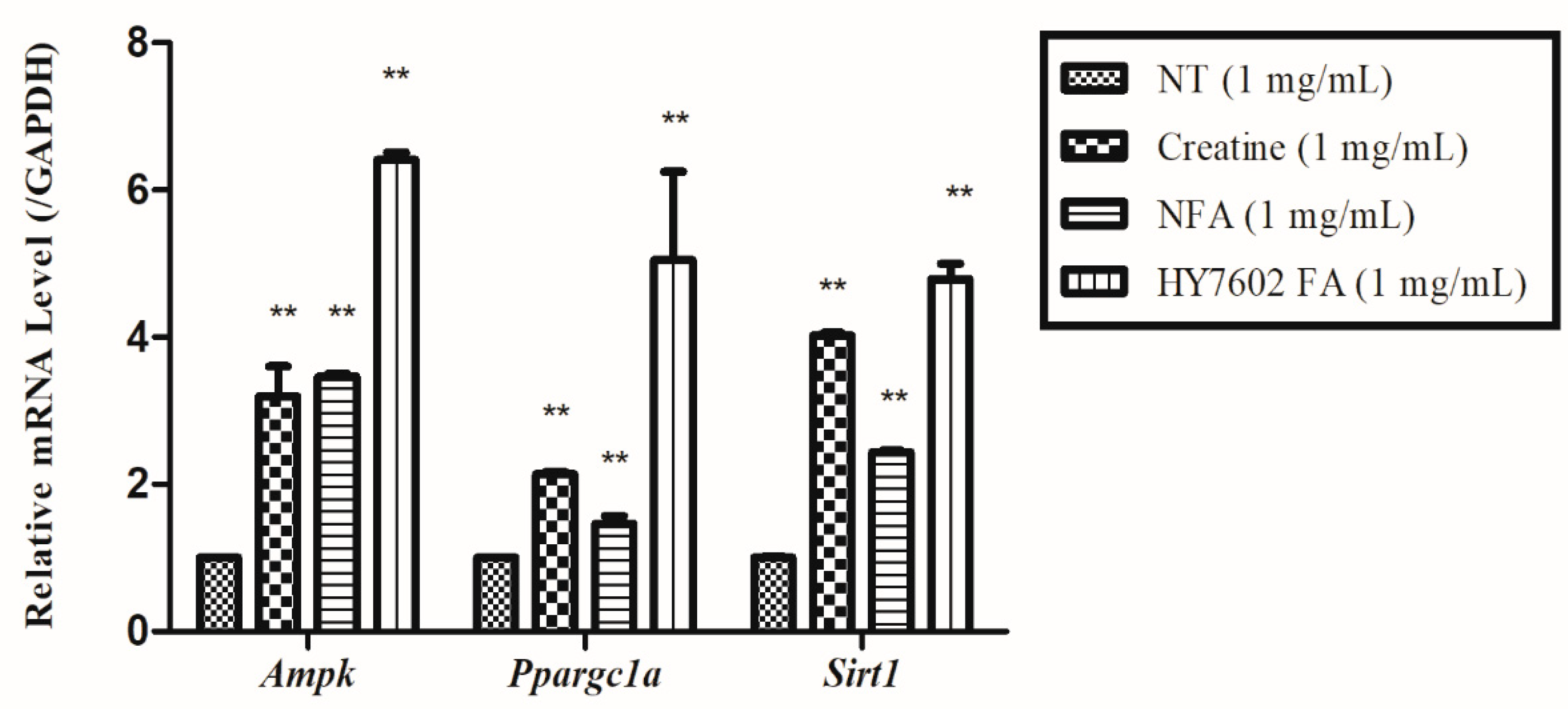
3.3. Effects of FA on Expression of Genes Related to Muscle Strength and Mitochondrial Energy Metabolism in C2C12 Cells
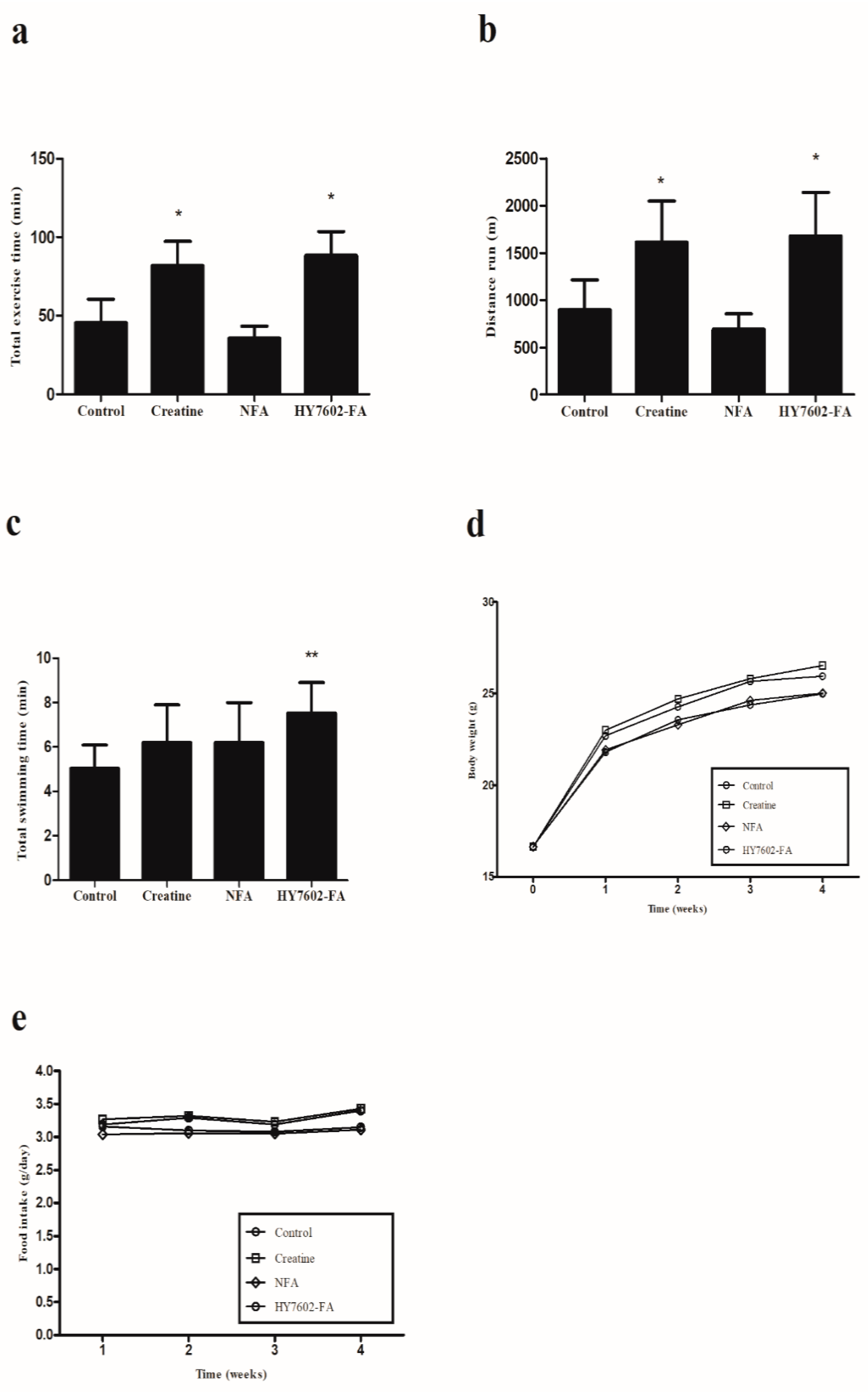
3.4. HY7602–FA Increases Exercise Endurance
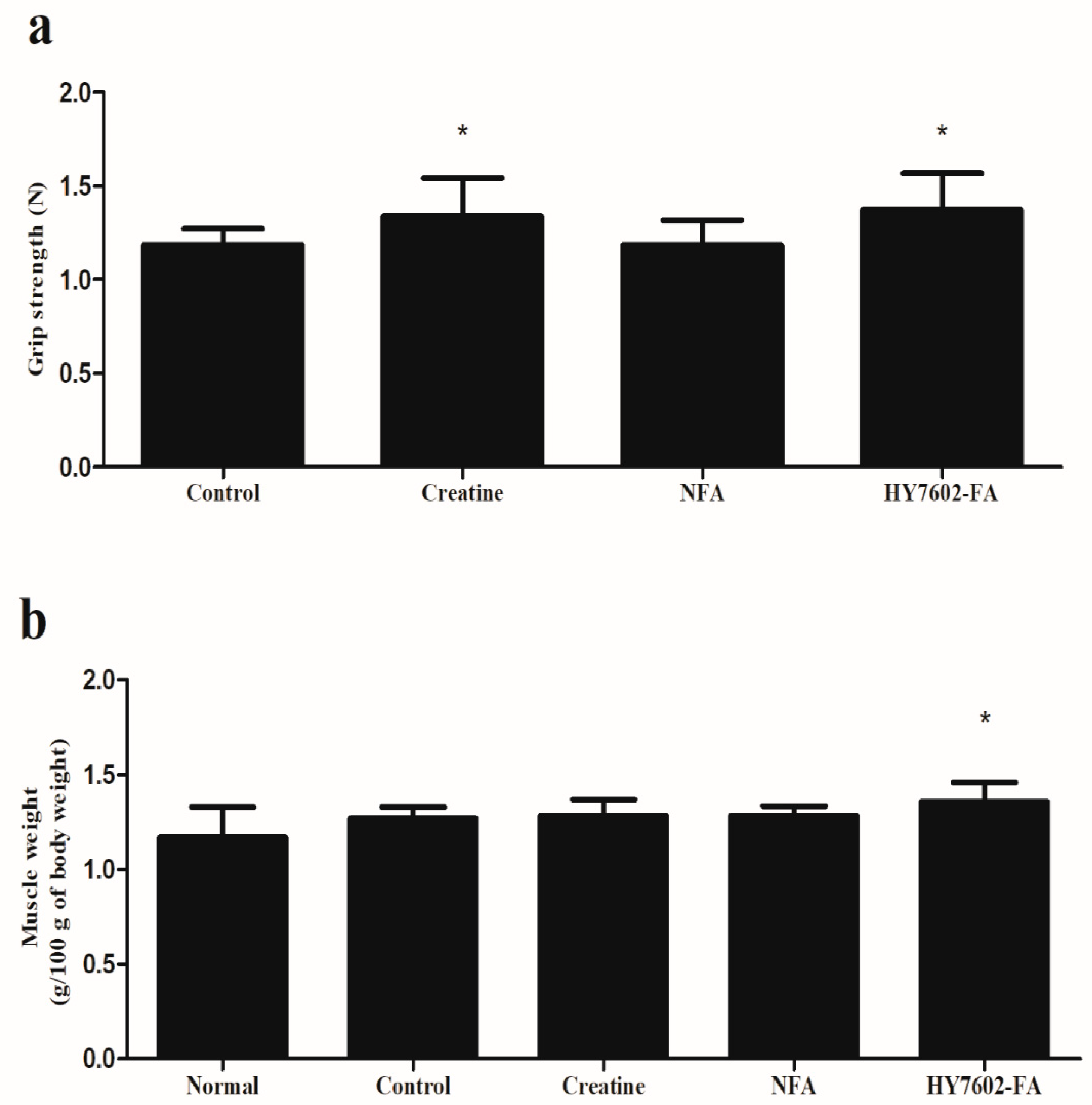
3.5. HY7602–FA Improves Muscle Hypertrophy and Strength
3.6. HY7602-FA Improves Biomarkers of Muscle Endurance, Energy Supply, and Muscle Fatigue after Exercise
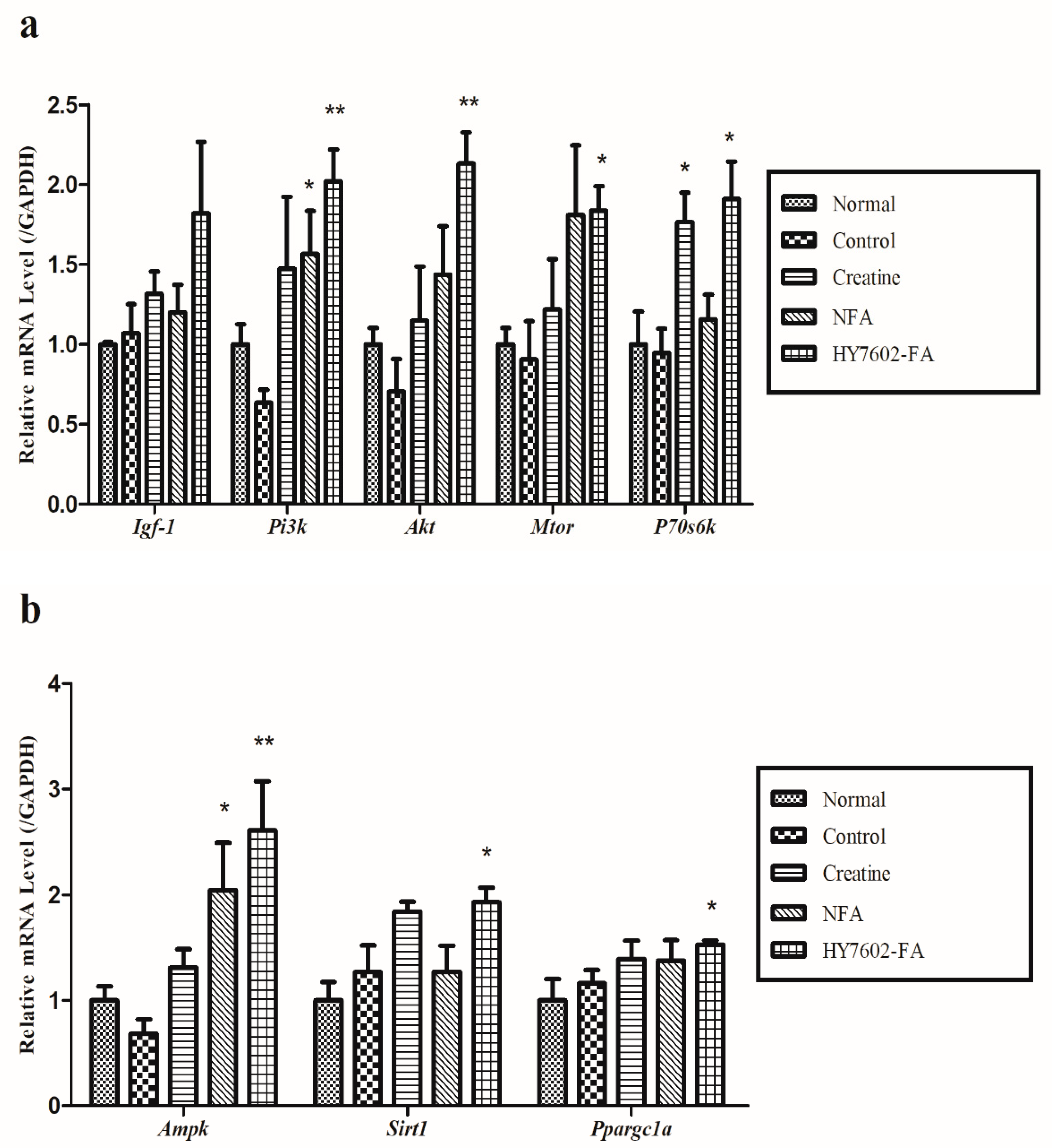
3.7. HY7602-FA Activates Muscle Protein Synthesis, Mitochondrial Biogenesis, and Exercise Signaling-Related mRNA Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hong, N.D.; Won, D.H.; Kim, N.J.; Chang, S.Y.; Youn, W.-G.; Kim, H.-S. Studies on the Analysis of Constituents of Deer Horn (I)-Assay of Trace Elements and TLC Pattern Analysis of Gangliosides. Korean Deer J. 1991, 14, 12–21. [Google Scholar]
- Ivankina, N.F.; Isay, S.V.; Busarova, N.G.; Mischenko, T.Y. Prostaglandin-like activity, fatty acid and phospholipid composition of sika deer (Cervus nippon) antlers at different growth stages. Comp. Biochem. Physiol. 1993, 106, 159–162. [Google Scholar] [CrossRef]
- Ha, H.; Yoon, S.-H. Analytical studies of constituents of antlers. J. Korean Soc. Food Sci. Nutr. 1996, 25, 279–282. [Google Scholar]
- Sunwoo, H.H.; Nakano, T.; Hudson, R.J.; Sim, J.S. Chemical composition of antlers from wapiti (Cervus elaphus). J. Agric. Food Chem. 1995, 43, 2846–2849. [Google Scholar] [CrossRef]
- Yoo, Y.S.; Kim, Y.S.; Jhon, G.-J.; Park, J. Separation of gangliosides using cyclodextrin in capillary zone electrophoresis. J. Chromatogr. A 1993, 652, 431–439. [Google Scholar] [PubMed]
- Pette, D.; Staron, R.S. Myosin isoforms, muscle fiber types, and transitions. Microsc. Res. Tech. 2000, 50, 500–509. [Google Scholar] [CrossRef]
- Zong, H.; Ren, J.M.; Young, L.H.; Pypaert, M.; Mu, J.; Birnbaum, M.J.; Shulman, G.I. AMP kinase is required for mitochondrial biogenesis in skeletal muscle in response to chronic energy deprivation. Proc. Natl. Acad. Sci. USA 2002, 99, 15983–15987. [Google Scholar] [CrossRef] [PubMed]
- Ticinesi, A.; Nouvenne, A.; Cerundolo, N.; Catania, P.; Prati, B.; Tana, C.; Meschi, T. Gut microbiota, muscle mass and function in aging: A focus on physical frailty and sarcopenia. Nutrients 2019, 11, 1633. [Google Scholar] [CrossRef]
- Sivamaruthi, B.S.; Kesika, P.; Suganthy, N.; Chaiyasut, C. A review on role of microbiome in obesity and antiobesity properties of probiotic supplements. Biomed Res. Int. 2019, 2019, 3291367. [Google Scholar] [CrossRef]
- Huang, W.-C.; Chen, Y.-M.; Kan, N.-W.; Ho, C.-S.; Wei, L.; Chan, C.-H.; Huang, H.-Y.; Huang, C.-C. Hypolipidemic effects and safety of Lactobacillus reuteri 263 in a hamster model of hyperlipidemia. Nutrients 2015, 7, 3767–3782. [Google Scholar] [CrossRef]
- Sichetti, M.; De Marco, S.; Pagiotti, R.; Traina, G.; Pietrella, D. Anti-inflammatory effect of multistrain probiotic formulation (L. rhamnosus, B. lactis, and B. longum). Nutrition 2018, 53, 95–102. [Google Scholar] [CrossRef]
- Pourramezan, Z.; Kermanshahi, R.K.; Oloomi, M.; Aliahmadi, A.; Rezadoost, H. In vitro study of antioxidant and antibacterial activities of Lactobacillus probiotic spp. Folia Microbiol. 2018, 63, 31–42. [Google Scholar] [CrossRef]
- Tiptiri-Kourpeti, A.; Spyridopoulou, K.; Santarmaki, V.; Aindelis, G.; Tompoulidou, E.; Lamprianidou, E.E.; Saxami, G.; Ypsilantis, P.; Lampri, E.S.; Simopoulos, C.; et al. Lactobacillus casei exerts anti-proliferative effects accompanied by apoptotic cell death and up-regulation of TRAIL in colon carcinoma cells. PLoS ONE 2016, 11, e0147960. [Google Scholar] [CrossRef]
- Oliveira, M.; Bosco, N.; Perruisseau, G.; Nicolas, J.; Segura-Roggero, I.; Duboux, S.; Briand, M.; Blum, S.; Benyacoub, J. Lactobacillus paracasei reduces intestinal inflammation in adoptive transfer mouse model of experimental colitis. Clin. Dev. Immunol. 2011, 2011, 807483. [Google Scholar] [CrossRef][Green Version]
- Chen, L.-H.; Chen, Y.-H.; Cheng, K.-C.; Chien, T.-Y.; Chan, C.-H.; Tsao, S.-P.; Huang, H.-Y. Antiobesity effect of Lactobacillus reuteri 263 associated with energy metabolism remodeling of white adipose tissue in high-energy-diet-fed rats. J. Nutr. Biochem. 2018, 54, 87–94. [Google Scholar] [CrossRef]
- Grosicki, G.J.; Fielding, R.A.; Lustgarten, M.S. Gut microbiota contribute to age-related changes in skeletal muscle size, composition, and function: Biological basis for a gut-muscle axis. Calcif. Tissue Int. 2018, 102, 433–442. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.E.; Bhattacharya, A.; Sataranatarajan, K.; Qaisar, R.; Sloane, L.; Rahman, M.M.; Kinter, M.; Van Remmen, H. The histone deacetylase inhibitor butyrate improves metabolism and reduces muscle atrophy during aging. Aging Cell 2015, 14, 957–970. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Milani, C.; De Giori, G.S.; Sesma, F.; Van Sinderen, D.; Ventura, M. Bacteria as vitamin suppliers to their host: A gut microbiota perspective. Curr. Opin. Biotechnol. 2013, 24, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.; Denman, S.E.; Hughes, R.J.; Geier, M.S.; Crowley, T.M.; Chen, H.; Haring, V.R.; Moore, R.J. Intestinal microbiota associated with differential feed conversion efficiency in chickens. Appl. Microbiol. Biotechnol. 2012, 96, 1361–1369. [Google Scholar] [CrossRef] [PubMed]
- Kunkel, S.D.; Suneja, M.; Ebert, S.M.; Bongers, K.S.; Fox, D.K.; Malmberg, S.E.; Alipour, F.; Shields, R.K.; Adams, C.M. mRNA expression signatures of human skeletal muscle atrophy identify a natural compound that increases muscle mass. Cell Metab. 2011, 13, 627–638. [Google Scholar] [CrossRef]
- Nader, G.A. Molecular determinants of skeletal muscle mass: Getting the “AKT” together. Int. J. Biochem. Cell Biol. 2005, 37, 1985–1996. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Park, E.D.; Suh, H.J.; Lee, S.H.; Kim, J.S.; Park, Y. Enhancement of exercise endurance capacity by fermented deer antler in BALB/c mice. Biosci. Biotechnol. Biochem. 2014, 78, 1716–1722. [Google Scholar] [CrossRef]
- Huang, W.-C.; Huang, C.-C.; Chuang, H.-L.; Chiu, C.-C.; Chen, W.-C.; Hsu, M.-C. Cornu cervi pantotrichum supplementation improves physiological adaptions during intensive endurance training. J. Vet. Med. Sci. 2017, 79, 674–682. [Google Scholar] [CrossRef]
- Kim, M.-K.; Jung, E.-Y.; Lee, H.-S.; Shin, K.-S.; Kim, Y.-K.; Ra, K.-S.; Park, C.-S.; Woo, M.-J.; Lee, S.-H.; Kim, J.-S.; et al. Isolation of strain for the preparation of the fermented antler and its physiological activities. J. Korean Soc. Food Sci. Nutr. 2009, 38, 1237–1242. [Google Scholar] [CrossRef]
- Wu, F.; Li, H.; Jin, L.; Li, X.; Ma, Y.; You, J.; Li, S.; Xu, Y. Deer antler base as a traditional Chinese medicine: A review of its traditional uses, chemistry and pharmacology. J. Ethnopharmacol. 2013, 145, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Suh, J.H.; Seo, H.W.; Han, B.C.; Kyung, J.S.; So, S.H. 13-Week repeated oral dose toxicity study on mixture of Korean red ginseng and deer antler extract in Sprague-Dawley rats. Toxicol. Res. 2019, 36, 1–12. [Google Scholar] [CrossRef]
- Kim, D.-H.; Han, S.-B.; Park, J.-S.; Han, M.-J. Fermentation of antler and its biological activity. Korean J. Pharmacogn. 1994, 25, 233–237. [Google Scholar]
- Chen, J.-C.; Hsiang, C.-Y.; Lin, Y.-C.; Ho, T.-Y. Deer Antler Extract Improves Fatigue Effect through Altering the Expression of Genes Related to Muscle Strength in Skeletal Muscle of Mice. Evid. Based Complement. Alternat. Med. 2014, 2014, 540580. [Google Scholar] [CrossRef] [PubMed]
- Han, G.-H.; Kwon, H.-T.; Son, H.-J.; Chai, J.-H.; Shan, Z.X.; Kim, H.-J.; Kim, C.-K. Change of mTOR, p70S6K, 4E-BP1, and AMPK protein expressions with a intensive endurance exercise in rats. Korean Soc. Sports Sci. 2011, 20, 1551–1561. [Google Scholar]
- Hornberger, T.A. Mechanotransduction and the regulation of mTORC1 signaling in skeletal muscle. Int. J. Biochem. Cell Biol. 2011, 43, 1267–1276. [Google Scholar] [CrossRef]
- Glass, D.J. Signalling pathways that mediate skeletal muscle hypertrophy and atrophy. Nat. Cell Biol. 2003, 5, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Handschin, C.; Spiegelman, B.M. Metabolic control through the PGC-1 family of transcription coactivators. Cell Metab. 2005, 1, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Gureev, A.P.; Shaforostova, E.A.; Popov, V.N. Regulation of mitochondrial biogenesis as a way for active longevity: Interaction between the Nrf2 and PGC-1α signaling pathways. Front Genet. 2019, 10, 435. [Google Scholar] [CrossRef] [PubMed]
- Kimura, Y.; Sumiyoshi, M. Effects of various Eleutherococcus senticosus cortex on swimming time, natural killer activity and corticosterone level in forced swimming stressed mice. J. Ethnopharmacol. 2004, 95, 447–453. [Google Scholar] [CrossRef]
- Cairns, S.P. Lactic acid and exercise performance: Culprit or friend? Sports Med. 2006, 36, 279–291. [Google Scholar] [CrossRef] [PubMed]
| Normal | Control | Creatine | NFA | HY7602-FA | |
|---|---|---|---|---|---|
| ALT (U/L) | 18.00 ± 2.7 | 19.57 ± 2.9 | 19.29 ± 2.0 | 16.38 ± 3.2 | 15.57 ± 4.6 * |
| LDH (U/L) | 376.14 ± 73.1 | 262.43 ± 101.5 | 273.00 ± 129.8 | 258.38 ± 117.9 | 145.71 ± 23.5 * |
| Creatine kinase (mg/dL) | 51.71 ± 21.9 | 132.71 ± 71.9 | 102.57 ± 39.6 | 72.25 ± 34.6 | 55.43 ± 22.9 * |
| Lactate (mg/dL) | 69.77 ± 5.6 | 78.13 ± 10.4 | 62.84 ± 16.4 | 61.23 ± 5.7 | 48.90 ± 13.7 * |
| Creatinine (mg/dL) | 0.32 ± 0.08 | 0.35 ± 0.1 | 0.44 ± 0.09 | 0.29 ± 0.04 | 0.26 ± 0.03 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, S.; Kim, S.-H.; Jeung, W.; Ra, J.; Heo, K.; Shim, J.-J.; Lee, J.-L. Fermented Antler Improves Endurance during Exercise Performance by Increasing Mitochondrial Biogenesis and Muscle Strength in Mice. Appl. Sci. 2021, 11, 5386. https://doi.org/10.3390/app11125386
Jung S, Kim S-H, Jeung W, Ra J, Heo K, Shim J-J, Lee J-L. Fermented Antler Improves Endurance during Exercise Performance by Increasing Mitochondrial Biogenesis and Muscle Strength in Mice. Applied Sciences. 2021; 11(12):5386. https://doi.org/10.3390/app11125386
Chicago/Turabian StyleJung, Seongeun, Sung-Hwan Kim, Woonhee Jeung, Jehyun Ra, Keon Heo, Jae-Jung Shim, and Jung-Lyoul Lee. 2021. "Fermented Antler Improves Endurance during Exercise Performance by Increasing Mitochondrial Biogenesis and Muscle Strength in Mice" Applied Sciences 11, no. 12: 5386. https://doi.org/10.3390/app11125386
APA StyleJung, S., Kim, S.-H., Jeung, W., Ra, J., Heo, K., Shim, J.-J., & Lee, J.-L. (2021). Fermented Antler Improves Endurance during Exercise Performance by Increasing Mitochondrial Biogenesis and Muscle Strength in Mice. Applied Sciences, 11(12), 5386. https://doi.org/10.3390/app11125386






