Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition
Abstract
1. Introduction
2. Probiotics
3. Modern Probiosis
4. Prebiotics, Functional Food, Bioactive Ingredients and Nutraceuticals
4.1. Prebiotic Compounds
4.2. Cereals and Their Derivatives as Prebiotics
4.2.1. Gastrointestinal Tract
4.2.2. Skin
4.2.3. Urogenital Tract
4.2.4. Metabolic Diseases
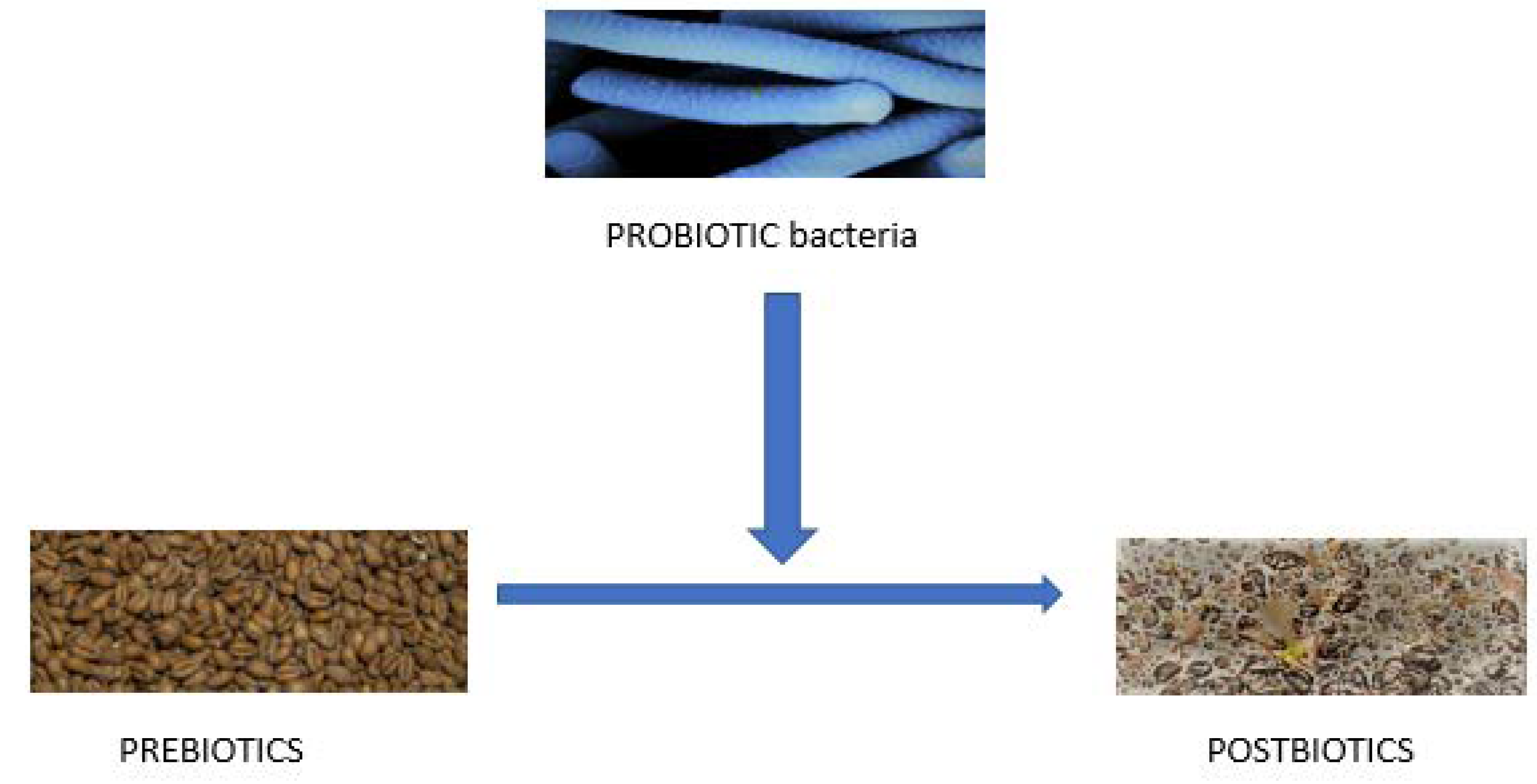
5. New-Quality—Processing of Foodstuff by Probiotic Bacteria In Vitro
6. Postbiotics
6.1. Better Taste—An Extension to the “Beneficial” Action of Probiotics
6.2. Probiotics in the Grain Preservation
7. The Recognition and Uptake of Prebiotics among Consumers
8. Limitations of Functional Food
In Summary
Author Contributions
Funding
Conflicts of Interest
References
- Tomasik, P.J.; Tomasik, P. Prebiotics and probiotics. Cereal Chem. 2003, 80, 113–117. [Google Scholar] [CrossRef]
- Yeo, S.K.; Ewe, J.-A.; Sau-Chan Tham, C.; Liong, M.-Z. Carriers of probiotic organisms. In Probiotics: Biology, Genetics, and Health Aspects; Book Series: Microbiology Monographs; Springer: Berlin, Germany, 2011; Volume 21, pp. 191–220. [Google Scholar]
- Saxelin, M. Probiotic formulations and applications, the current probiotics market and changes in the marketplace: A European perspectives. Clin. Infect. Dis. 2008, 46 (Suppl. 2), S76–S79. [Google Scholar] [CrossRef]
- Bansal, S.; Mangal, M.; Sharma, S.K.; Gupta, R.K. Non-dairy based probiotics: A healthy treat for intestine. Crit. Rev. Food Sci. Nutr. 2015, 56, 1856–1867. [Google Scholar] [CrossRef]
- Shori, A.B. Influence of food matrix on the viability of probiotic bacteria: A review based on dairy and non-dairy beverages. Food Biosci. 2016, 13, 1–8. [Google Scholar] [CrossRef]
- Enujiugha, V.N.; Badejo, A.A. Probiotic potentials of cereal-based beverages. Crit. Rev. Food Sci. Nutr. 2017, 57, 790–804. [Google Scholar] [CrossRef] [PubMed]
- Greppi, A.; Saubade, F.; Botta, C.; Humblot, C.; Guyot, J.P.; Cocolin, L. Potential probiotic Pichia kudriavzevii strains and their ability to enhance folate content of traditional cereal-based African fermented food. Food Microbiol. 2017, 62, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Salmeron, I. Fermented cereal beverages: From probiotic, prebiotic and symbiotic towards nanoscience designed healthy drinks. Lett. Appl. Microbiol. 2017, 65, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Hassani, A.; Procopio, S.; Becker, T. Influence of malting and lactic acid fermentation on functional bioactive components in cereal-based raw materials: A review paper. Int. J. Food Sci. Technol. 2016, 51, 14–22. [Google Scholar] [CrossRef]
- Lamsal, B.P.; Faubion, J.M. The beneficial use of cereal and cereal compounds in probiotic foods. Food Rev. Int. 2009, 25, 103–114. [Google Scholar] [CrossRef]
- Furtado Martins, E.M.; Ramos, A.M.; Lago Vanzela, E.S.; Stringheta, P.C.; de Oliveira Pinto, C.L.; Martins, J.M. Products of vegetable origin: A new alternative for the consumption of probiotic bacteria. Food Res. Int. 2013, 51, 764–770. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N. Probiotic fermentation of plant based products: Possibilities and opportunities. Crit. Rev. Food Sci. Nutr. 2012, 52, 183–199. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H. Probiotics and prebiotics in pediatrics: Where are we now? Turk. J. Pediatr. 2007, 49, 231–244. [Google Scholar] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Assadi, H.; Naeimi, B.; Gharibi, S.; Khosravi, A.; Dobaradaran, S.; Taherkhani, R.; Tajbakhsh, S. Detection of Acinetobacter spp. in blood cultures by an improved fluorescent in situ hybridization assay. Pol. J. Microbiol. 2018, 67, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Païssé, S.; Valle, C.; Servant, F.; Courtney, M.; Burcelin, R.; Amar, J.; Lelouvier, B. Comprehensive description of blood microbiome from healthy donors assessed by 16S targeted metagenomic sequencing. Transfusion 2016, 56, 1138–1147. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, J.M. The origin of human milk bacteria: Is there a bacterial entero-mammary pathway during late pregnancy and lactation? Adv. Nutr. 2014, 5, 779–784. [Google Scholar] [CrossRef] [PubMed]
- Jost, T.; Lacroix, C.; Braegger, C.; Chassard, C. Impact of human milk bacteria and oligosaccharides on neonatal gut microbiota establishment and gut health. Nutr. Rev. 2015, 73, 426–437. [Google Scholar] [CrossRef]
- Biagi, E.; Quercia, S.; Aceti, A.; Beghetti, I.; Rampelli, S.; Turroni, S.; Faldella, G.; Candela, M.; Brigidi, P.; Corvaglia, L. The bacterial ecosystem of mother’s milk and infant’s mouth and gut. Front. Microbiol. 2017, 8, 1214–1224. [Google Scholar] [CrossRef]
- Stinson, L.F.; Payne, M.S.; Keelan, J.A. Planting the seed: Origins, composition, and postnatal health significance of the fetal gastrointestinal microbiota. Crit. Revs. Microbiol. 2017, 43, 352–369. [Google Scholar] [CrossRef]
- Gokavi, S.; Zhang, L.W.; Huang, M.-K.; Xin Zhao, X.; Guo, M. Oat-based synbiotic beverage fermented by Lactobacillus plantarum, Lactobacillus paracasei ssp. casei, and Lactobacillus acidophilus. J. Food Sci. 2005, 70, M216–M223. [Google Scholar] [CrossRef]
- Coda, R.; Rizzello, C.G.; Trani, A.; Gobbetti, M. Manufacture and characterization of functional emmer beverages fermented by selected lactic acid bacteria. Food Microbiol. 2011, 28, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Pelikanova, J.; Liptakova, D.; Valik, L. Suitability of lactic acid bacteria for fermentation of maize and amaranth. J. Food Nutr. Res. 2015, 54, 354–364. [Google Scholar]
- Gao, F.; Cai, S.; Nout, R.M.J.; Wang, Y.; Xia, Y.; Li, Y.; Ji, B. Production of oat-based synbiotic beverage by two-stage fermentation with Rhizopus oryzae and Lactobacillus acidophilus. J. Food Agric. Environ. 2012, 10, 175–179. [Google Scholar]
- Fusco, V.; Quero, G.M.; Cho, G.-S.; Kabisch, J.; Meske, D.; Neve, H.; Bockelmann, W.; Franz, C.M. The genus Weissella: Taxonomy, ecology and biotechnological potential. Front. Microbiol. 2015, 6, 155. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, E.B.M.; Skov, L.; Thyssen, J.P.; Jensen, P. Role of the gut microbiota in atopic dermatitis: A systematic review. Acta Dermato-Venereol. 2019, 99, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.; Fabersani, E.; Marquez, A.; Gauffin-Cano, P. Adipose tissue inflammation and metabolic syndrome. The proactive role of probiotics. Eur. J. Nutr. 2019, 58, 27–43. [Google Scholar] [CrossRef]
- Nowak, A.; Paliwoda, A.; Blasiak, J. Anti-proliferative, pro-apoptotic and anti-oxidative activity of Lactobacillus and Bifidobacterium strains: A review of mechanisms and therapeutic perspectives. Crit. Revs. Food Sci. Nutr. 2019, 59, 3456–3467. [Google Scholar] [CrossRef]
- Chorell, E.; Karlsson Videhult, F.; Hernell, O.; Antti, H.; West, C.E. Impact of probiotic feeding during weaning on the serum lipid profile and plasma metabolome in infants. Br. J. Nutr. 2013, 110, 116–126. [Google Scholar] [CrossRef]
- West, C.E.; Hammarstrom, M.-L.; Hernell, O. Probiotics in primary prevention of allergic disease – follow-up at 8–9 years of age. Allergy 2013, 68, 1015–1020. [Google Scholar] [CrossRef]
- Hasslöf, P.; West, C.E.; Karlsson Videhult, F.; Brandelius, C.; Stecksén-Blicks, C. Early intervention with probiotic Lactobacillus paracasei F19 has no long-term effect on caries experience. Caries Res. 2013, 47, 559–565. [Google Scholar] [CrossRef]
- Benhadou, F.; Mintoff, D.; Schnebert, B.; Thio, H.B. Psoriasis and microbiota: A systematic review. Diseases 2018, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Deidda, F.; Amoruso, A.; Nicola, S.; Graziano, T.; Pane, M.; Mogna, L. New approach in acne therapy: A specific bacteriocin activity and a targeted anti IL-8 property in Just 1 probiotic strain, the L. salivarius LS03. J. Clin. Gastroenterol. 2018, 52, S78–S81. [Google Scholar] [CrossRef] [PubMed]
- Nwanodi, O. Skin protective nutraceuticals: The current evidence in brief. Healthcare (Basel) 2018, 6, 40. [Google Scholar] [CrossRef] [PubMed]
- Rajpoot, M.; Sharma, A.K.; Sharma, A.; Gupta, G.K. Understanding the microbiome: Emerging biomarkers for exploiting the microbiota for personalized medicine against cancer. Semin. Cancer Biol. 2018, 52(pt.1), 1–8. [Google Scholar] [CrossRef]
- FAO. Technical Meeting on Prebiotics; FAO: Rome, Italy, 2008; pp. 1–12. [Google Scholar]
- Gibson, G.R.; Hutkins, R.W.; Sanders, M.E.; Prescott, S.L.; Reimer, R.A.; Salminen, S.J.; Scott, K.; Stanton, C.; Swanson, K.S.; Cani, P.D.; et al. Expert consensus document: The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of prebiotics. Nat. Revs. Gastroenterol. Hepatol. 2017, 14, 491–502. [Google Scholar] [CrossRef]
- Das, A.; Raychaudhuri, U.; Chakraborty, R. Cereal based functional food of Indian subcontinent. A review. J. Food Sci. Technol. Mysore 2012, 49, 665–672. [Google Scholar] [CrossRef]
- Reque, P.M.; Pinilla, C.M.B.; Gautério, G.V.; Kalil, S.J.; Brandelli, A. Xylooligosaccharides production from wheat middlings bioprocessed with Bacillus subtilis. Food Res. Int. 2019, 126, 108673. [Google Scholar] [CrossRef]
- Sharma, M.; Devi, M. Probiotics: A comprehensive approach towards health foods. Crit. Rev. Food Sci. Nutr. 2014, 54, 537–552. [Google Scholar] [CrossRef]
- Barczynska, R.; Slizewska, K.; Litwin, M.; Szalecki, M.; Kapusniak, J. Effect of dietary fiber preparations made from maize starch and the growth and activity of selected bacteria from the Formicutes, Bacteroidetes and Actinobacteria phyla in fecal samples from obese children. Acta Biochim. Polon. 2015, 63, 261–266. [Google Scholar]
- Barczynska, R.; Kapusniak, J.; Litwin, M.; Slizewska, K.; Szalecki, M. Dextrins from maize starch as substances activating the growth of Bacteroidetes and Actinobacteria simultaneously inhibiting the growth of Firmicutes resposnisble for the occurrence of obesity. Plant Foods Hum. Nutr. 2016, 71, 190–196. [Google Scholar] [CrossRef]
- Barczynska, R.; Jurgonski, A.; Slizewska, K.; Juskiewicz, J.; Kapusniak, J. Cornstarch dextrin changes intestinal microbiota and iys metabolic activity in rats fed a basal and high-fat diet. Br. Food J. 2019, 121, 2219–2232. [Google Scholar] [CrossRef]
- Slizewska, K.; Libudzisz, Z.; Barczynska, R.; Kapusniak, J.; Zdunczyk, Z.; Juskiewicz, J. Dietary resistant dextrins positvey modulate fecal and cecal microbiota composition in young rats. Acta Biochim. Polon. 2015, 62, 677–681. [Google Scholar] [CrossRef] [PubMed]
- Barczynska, R.; Jurgonski, A.; Slizewska, K.; Juskiewicz, J.; Kapusniak, J. Effect of potato dextrin on the composition and metabolism of the gut microbiota in rats fed standard and high-fat diets. J. Funct. Food 2017, 34, 398–407. [Google Scholar] [CrossRef]
- Franz, C.M.A.P.; Hucha, M.; Mathara, J.M.; Abriouel, H.; Benomar, N.; Reid, G.; Galvez, A.; Holzapfel, W.H. African fermented foods and probiotics. Int. J. Food Microbiol. 2014, 190, 84–96. [Google Scholar] [CrossRef] [PubMed]
- Kalui, C.M.; Mathara, J.M.; Kutima, P.M. Probiotic potential of spontaneously fermented cereal based foods —A review. Afr. J. Biotechnol. 2010, 9, 2490–2498. [Google Scholar]
- Gupt, M.; Bajaj, B.K. Selection criteria for probiotics and potential of cereal based food products as novel probiotic-carriers. Curr. Nutr. Food Sci. 2016, 12, 157–174. [Google Scholar] [CrossRef]
- Ouwehand, A.C.; Kurvinen, T.; Rissanen, P. Use of a probiotic Bifidobacterium in a dry food matrix, an in vivo study. Int. J. Food Microbiol. 2004, 95, 103–106. [Google Scholar] [CrossRef]
- Bernat, N.; Chafer, M.; Gonzalez-Martınez, C.; Rodrıguez-Garcıa, J.; Chiralt, A. Optimization of oat milk formulation to obtain fermented derivatives by using probiotic Lactobacillus reuteri microorganisms. Food Sci. Technol. Int. 2014, 20, 145–157. [Google Scholar]
- Arena, M.P.; Caggianiello, G.; Fiocco, D.; Russo, P.; Torelli, M.; Spano, G.; Capozz, V. Barley β-glucans-containing food enhances probiotic performances of beneficial bacteria. Int. J. Mol. Sci. 2014, 15, 3025–3039. [Google Scholar] [CrossRef]
- Shamir, R.; Makhoul, I.R.; Etzioni, A.; Shehadeh, N. Evaluation of diet containing probiotics and zinc for the treatment of mild diarrheal illness in children younger than one year of age. J. Am. Coll. Nutr. 2005, 24, 370–375. [Google Scholar] [CrossRef]
- Ogunremi, O.R.; Sanni, A.I.; Agrawal, R. Probiotic potentials of yeasts isolated from some cereal-based Nigerian traditional fermented food products. J. Appl. Microbiol. 2015, 119, 797–808. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Bian, Z.; Xu, B. Skin health promotion effects of natural beta-glucan derived from cereals and microorganisms: A review. Phytother. Res. 2014, 28, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Manosroi, J.; Khositsuntiwong, N.; Manosroi, A. Biological activities of fructooligosaccharide (FOS)-containing Coix lachryma-jobi Linn. extract. J. Food Sci. Technol. 2014, 51, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Tester, R.; Al-Ghazzewi, F.H. Intrinsic and extrinsic carbohydrates in the vagina: A short review on vaginal glycogen. Int. J. Biol. Macromol. 2018, 112, 203–206. [Google Scholar] [CrossRef] [PubMed]
- De Alberti, D.; Russo, R.; Terruzzi, F.; Nobile, V.; Ouwehand, A.C. Lactobacilli vaginal colonisation after oral consumption of Respecta(®) complex: A randomised controlled pilot study. Arch. Gynecol. Obstetrics 2015, 29, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Johnson, D.W.; Morrison, M.; Pascoe, E.M.; Coombes, J.S.; Forbes, J.M.; Szeto, C.C.; McWhinney, B.C.; Ungerer, J.P.; Campbell, K.L. Synbiotics easing renal failure by improving gut microbiology (Synergy): A randomized trial. Clin. J. Am. Soc. Nephrol. 2016, 11, 223–231. [Google Scholar] [CrossRef]
- Maretti, C.; Cavallini, G. The association of a probiotic with a prebiotic (Flortec, Bracco) to improve the quality/quantity of spermatozoa in infertile patients with idiopathic oligoasthenoteratospermia: A pilot study. Andrology 2017, 5, 439–444. [Google Scholar] [CrossRef]
- Tuohy, K.M.; Fava, F.; Viola, R. ‘The way to a man’s heart is through his gut microbiota’—dietary pro- and prebiotics for the management of cardiovascular risk. Proc. Nutr. Soc. 2014, 73, 172–185. [Google Scholar] [CrossRef]
- Fallucca, F.; Porrata, C.; Fallucca, S.; Pianesi, M. Influence of diet on gut microbiota, inflammation and type 2 diabetes mellitus. First experience with macrobiotic Ma-Pi 2 diet. Diabet. Metabol. Res. Revs. 2014, 30 (Suppl. 1), 48–54. [Google Scholar] [CrossRef]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef]
- Shaikh Uzma, A.; Deshpande, H.W.; Kulkarni, D.B. A review on probiotic beverages prepared using vegetables. Int. J. Chem. Stud. 2018, 6(5), 61–65. [Google Scholar]
- Aguilar-Toala, J.; Garcia-Valera, R.; Garcia, H.S.; Mata-Haro, V.; Cordova-Gonzalez, A.F.; Hernandez-Mendoza, A. Postbiotics: An evolving term within the functional foods field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
- Arora, S.; Jood, S.; Khetarpaul, N. Effect of germination and probiotic fermentation on nutrient profile of pearl millet based food blends. Br. Food J. 2011, 113, 470–481. [Google Scholar] [CrossRef]
- Capozzi, V.; Russo, P.; Dueñas, M.T.; López, P.; Spano, G. Lactic acid bacteria producing B-group vitamins: A great potential for functional cereals products. Appl. Microbiol. Biotechnol. 2012, 96, 1383–1394. [Google Scholar] [CrossRef]
- Pessione, E.; Cirrincione, S. Bioactive molecules released in food by lactic acid bacteria: Encrypted peptides and biogenic amines. Front. Microbiol. 2016, 7, 876–895. [Google Scholar]
- Ayyash, M.; Johnson, S.K.; Liu, S.Q.; Mesmari, N.; Dahmani, S.; Al Dhaheri, A.S.; Kizhakkayil, J. In vitro investigation of bioactivities of solid-state fermented lupin, quinoa and wheat using Lactobacillus spp. Food Chem. 2019, 275, 50–58. [Google Scholar] [CrossRef]
- Sanz-Penell, J.M.; Frontela, C.; Ros, G.; Martinez, C.; Monedero, V.; Haros, M. Application of bifidobacterial phytases in infant cereals: Effect on phytate contents and mineral dialyzability. J. Agric. Food Chem. 2012, 60, 11787–11792. [Google Scholar] [CrossRef]
- Sarno, M.; Lania, G.; Cuomo, M.; Passannanti, F.; Budelli, A.; Fasano, F.; Troncone, R.; Auricchio, S.; Barone, M.V.; Nigro, R.; et al. Lactobacillus paracasei CBA L74 interferes with gliadin peptides entrance in Caco-2 cells. Int. J. Food Sci. Nutr. 2014, 65, 953–959. [Google Scholar] [CrossRef]
- Wegh, C.A.M.; Geerlings, S.Y.; Knol, J.; Roeselers, G.; Belzer, C. Postbiotics and their potential applications in early life nutrition and beyond. Int. J. Mol. Sci. 2019, 20, 4673. [Google Scholar] [CrossRef]
- Salari, M.; Razavi, S.H.; Gharibzahedi, S.M.T. Characterising the synbiotic beverages based on barley and malt flours fermented by Lactobacillus delbrueckii and paracasei strains. Qual. Assur. Saf. Crops Foods 2015, 7, 355–361. [Google Scholar] [CrossRef]
- Bailey, R. Functional Foods in Japan: Foshu (“foods for Specified Health Uses”) and “foods with Nutrient Function Claims”. In Regulation of Functional Foods and Nutraceuticals: A Global Perspective; Hasler, C.M., Ed.; Blackwell Publishing: Hoboken, NJ, USA, 2005; pp. 247–261. ISBN 9780813811772. [Google Scholar]
- Saito, M. Role of FOSHU (food for specified health uses) for healthier life. Yakugaku Zasshi 2007, 127, 407–446. [Google Scholar] [CrossRef] [PubMed][Green Version]
- China Dictionary.net. Available online: https://nortonsafe.search.ask.com/web?q=china%20dictionary&hp=0&year=2015&installstatus=updated&schemaver=1.0.0.0&os=windows&geo=us&schemacat=sbu_w&ssdcat=321&dsp=0&3in1=0&showuninstallsurvey=1&locale=pl_us&machinelocation=191&version=22.16.2.22&templatecat=sbu_w_ns_cto&sw=0&vendortesteligible=no&tb=0&osvers=6.3&vendor=iac&vendorsrc=firefox&olpchannel=configure_to_order&installsource=nag&source=nag&o=APN12174&prt=ngc&ver=3.3.0.4&tpr=111&chn=3000&guid=f36725de-2708-4191-858d-1f2391ffb9af&doi=2018-11-28 (accessed on 29 July 2018).
- Salmeron, I.; Rozada, R.; Thomas, K.; Ortega-Rivas, E.; Pandiella, S.S. Sensory characteristics and volatile composition of a cereal beverage fermented with Bifidobacterium breve NCIMB 702257. Food Sci. Technol. Int. 2014, 20, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Ogunremi, O.R.; Agrawal, R.; Sanni, A.I. Development of cereal-based functional food using cereal mix substrate fermented with probiotic strain—Pichia kudriavzevii OG32. Food Sci. Nutr. 2015, 3, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Reddy, K.R.N.; Raghavender, C.R.; Reddy, B.N.; Salleh, B. Biological control of Aspergillus flavus growth and subsequent aflatoxin B1 production in sorghum grains. Afr. J. Biotechnol. 2010, 9, 4247–4250. [Google Scholar]
- Quattrini, M.; Bernardi, C.; Stuknytė, M.; Masotti, F.; Passera, A.; Ricci, G.; Vallone, L.; De Noni, I.; Brasca, M.; Fortina, M.G. Functional characterization of Lactobacillus plantarum ITEM 17215: A potential biocontrol agent of fungi with plant growth promoting traits, able to enhance the nutritional value of cereal products. Food Res. Int. 2018, 106, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Wacoo, A.P.; Mukisa, I.M.; Meeme, R.; Byakika, S.; Wendiro, D.; Sybesma, W.; Kort, R. Probiotic enrichment and reduction of aflatoxins in a traditional African maize-based fermented food. Nutrients 2019, 11, 265. [Google Scholar] [CrossRef]
- da Silva, J.F.; Peluzio, J.M.; Prado, G.; Madeira, J.E.; Silva, M.O.; de Morais, P.B.; Rosa, C.A.; Pimenta, R.S.; Nicoli, J.R. Use of probiotics to control aflatoxin production in peanut grains. Sci. World J. 2015, 2015, 959138. [Google Scholar] [CrossRef]
- Kraus, A. Development of functional food with the participation of the consumer. Motivators for consumption of functional products. Int. J. Consum. Stud. 2015, 39, 2–11. [Google Scholar] [CrossRef]
- Kraus, A.; Annunziata, A.; Vecchio, R. Sociodemographic factors differentiating the consumer and the motivations for functional food consumption. J. Am. Coll. Nutr. 2017, 36, 116–126. [Google Scholar] [CrossRef]
- Betz, M.; Uzueta, A.; Rasmussen, H.; Gregoire, M.; Vanderwall, C.; Witowich, G. Knowledge, use and perceptions of probiotics and prebiotics in hospitalised patients. Nutr. Diet. 2015, 72, 261–266. [Google Scholar] [CrossRef]
- Ozen, A.E.; Bibiloni, M.; Pons, A.; Tur, J.A. Sociodemographic and lifestyle determinants of functional food consumption in an adult population of the Balearic Islands. Ann. Nutr. Metabol. 2013, 63, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Zmora, N.; Zilberman-Schapira, G.; Suez, J.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Kotler, E.; Zur, M.; Regev-Lehavi, D.; Brik, R.B.; et al. Personalized gut mucosal colonization resistance to empiric probiotics is associated with unique host and microbiome features. Cell 2018, 174, 1388–1405.e21. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Zmora, N.; Zilberman-Schapira, G.; Mor, U.; Dori-Bachash, M.; Bashiardes, S.; Zur, M.; Regev-Lehavi, D.; Ben-Zeev Brik, R.; Federici, S.; et al. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell 2018, 174, 1406–1423.e16. [Google Scholar]
- Goetze, O.; Fruehauf, H.; Pohl, D.; Giarrè, M.; Rochat, F.; Ornstein, K.; Menne, D.; Fried, M.; Thumshirn, M. Effect of a prebiotic mixture on intestinal comfort and general wellbeing in health. Brit. J. Nutr. 2008, 100, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.S.C.; Rehman, A.; Yu, S.; Andino, N.M. Brain fogginess, gas and bloating: A link between SIBO, probiotics and metabolic acidosis. Clin. Translat. Gastroenterol. 2018, 9, 162. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, P.; Chen, M.; Luo, Y.; Prabhakar, M.; Zheng, H.; He, Y.; Qi, Q.; Long, H.; Zhang, Y.; et al. Fructooligosaccharide (FOS) and galactooligosaccharide (GOS) increase Bifidobacterium but reduce butyrate producing bacteria with adverse glycemic metabolism in healthy young population. Sci. Repts. 2017, 7, 11789. [Google Scholar] [CrossRef]
- Skorka, A.; Piescik-Lech, M.; Kołodziej, M.; Szajewska, H. Infant formulae supplemented with prebiotics: Are they better than unsupplemented formulae? An updated systematic review. Brit. J. Nutr. 2018, 119, 810–825. [Google Scholar] [CrossRef]
- François, I.E.; Lescroart, O.; Veraverbeke, W.S.; Windey, K.; Verbeke, K.; Broekaert, W.F. Tolerance and the effect of high doses of wheat bran extract, containing arabinoxylan-oligosaccharides, and oligofructose on fecal output: A double-blind, randomized, placebo-controlled, cross-over trial. J. Nutr. Sci. 2014, 3, e49. [Google Scholar] [CrossRef]
- Hoving, L.R.; de Vries, M.R.; de Jong, R.C.M.; Katirae, i.S.; Pronk, A.; Quax, P.H.A.; van Harmelen, V.; Willems van Dijk, K. The prebiotic inulin aggravates accelerated atherosclerosis in hypercholesterolemic APOE*3-Leiden mice. Nutrients 2018, 10, 172. [Google Scholar] [CrossRef]
- Lin, F.; Wu, H.; Zeng, M.; Yu, G.; Dong, G.; Yang, H. Probiotic/prebiotic correction for adverse effects of iron fortification on intestinal resistance to Salmonella infection in weaning mice. Food Funct. 2018, 9, 1070–1078. [Google Scholar] [CrossRef]
| Dish | Fermented Source | Country | Fermentig Species |
|---|---|---|---|
| Beverages | |||
| Non-alcoholic | |||
| Amazake | rice | Japan | Aspergillus spp. |
| Boza | various cereals | Balkan countries | Leuconostoc spp., Lactobascillus spp. Saccharomyces spp. |
| Bhaati jaanr | rice | Himalaya countries | Saccharomycopsis fibuligera, Rhizopus spp. |
| Bushera | sorghum, millet | Central Africa | Lactobacillus spp. Streptococcus spp. |
| Haria | rice | India | Bifidobacterium, Saccharomyces spp. |
| Koko | pearl millet | Ghana | Weissella confusa, Lactobacillus fermentum |
| Kvass | rye bread, rye and barley malt/flour | Russia, Ukraine | Lactobacillus casei, Lactobacillus mesenteroides, Saccharomyces spp. |
| Mahewu | maize | Central Africa | Lactate producing bacteria |
| Malta | barley, hops | Denmark, Germany, Caribbean | Saccharomyces spp. |
| Mangisi | millet | Zimbabwe | yeast and mould |
| Podpiwek | grain coffee, hops | Poland, Lithuania | Sacharomuyces cerevisia |
| Pozol | maize | Mexico | Lactococcusus lactis, Lactobacilli alimentarium, casei and delbruekii, Enterococcus, Lactol |
| Supermalt | malt | Worldwide | Saccharomyces spp. |
| Tongwa | maize | Central Africa | Lactobacilli, Saccharomyces spp. |
| Low-alcoholic beverages | |||
| Chhang | rice, barley, millet | Tybet | Lactobacillus spp., Saccharomyces spp. |
| Malt beer | barley malt syrup, hops | Worldwide | yeast |
| Average-alcoholic beverages | |||
| Malt beer | barley malt syrup, hops | Worldwide | yeast |
| Malt liquor | Worldwide | Saccharomyces spp. | |
| High-alcoholic beverages | |||
| Malt whisky | malted barley | Saccharomyces spp. | |
| Sake | rice | Japan | Aspergillus oryzae + yeast |
| Spiritus (vodka) | rye, barley, maize | Worldwide | Saccharomyces spp. |
| Tesgüino | Sprouted corn | Mexico | yeast |
| Fermented food a | |||
| Appam | rice batter + coconut milk | India, Sri Lanka | |
| Bread | cereal flours | Worldwide | |
| Brem | rice | Indonesia | |
| Chakuli pitha | rice and black gram | India | |
| Dhokla | rice | India | |
| Dosa | rice | India | |
| Enduri pita | rice and black gram | India | |
| Injera | a sourdough-risen flatbread made out of teff flour | Ethiopia | |
| Kenkey | White maize grits | Africa | |
| Khanom chin | rice noodles | Thailand | |
| Mageu | porridge | South Africa | |
| Ogi | cereal pudding | Nigeria | |
| Pesaha appam | Fermented bread | India | |
| Puto | Rice cake | Philippines | |
| Sowan | starch remaining on the inner husks of oats after milling | Scotland | |
| Tapai | Rice or starch | Southeast Asia | |
| Zhur | Rye flour | Poland | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomasik, P.; Tomasik, P. Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition. Appl. Sci. 2020, 10, 1470. https://doi.org/10.3390/app10041470
Tomasik P, Tomasik P. Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition. Applied Sciences. 2020; 10(4):1470. https://doi.org/10.3390/app10041470
Chicago/Turabian StyleTomasik, Przemysław, and Piotr Tomasik. 2020. "Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition" Applied Sciences 10, no. 4: 1470. https://doi.org/10.3390/app10041470
APA StyleTomasik, P., & Tomasik, P. (2020). Probiotics, Non-Dairy Prebiotics and Postbiotics in Nutrition. Applied Sciences, 10(4), 1470. https://doi.org/10.3390/app10041470






