The Effect of Light on Antioxidant Properties and Metabolic Profile of Chia Microgreens
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Light Treatment
2.2. Determination of Chlorophyll and Carotenoid Content
2.3. Total Soluble Polyphenols and Protein Content
2.4. Estimation of Total Antioxidant Capacity
2.4.1. DPPH Scavenging Activity
2.4.2. Ferric Reducing Antioxidant Power Assay (FRAP)
2.5. Ascorbic Acid Content
2.6. Determination of Crystalline Cellulose Content
2.7. Fourier Transform Infrared Spectroscopy (FTIR)
2.8. Statistical Analyses
3. Results and Discussion
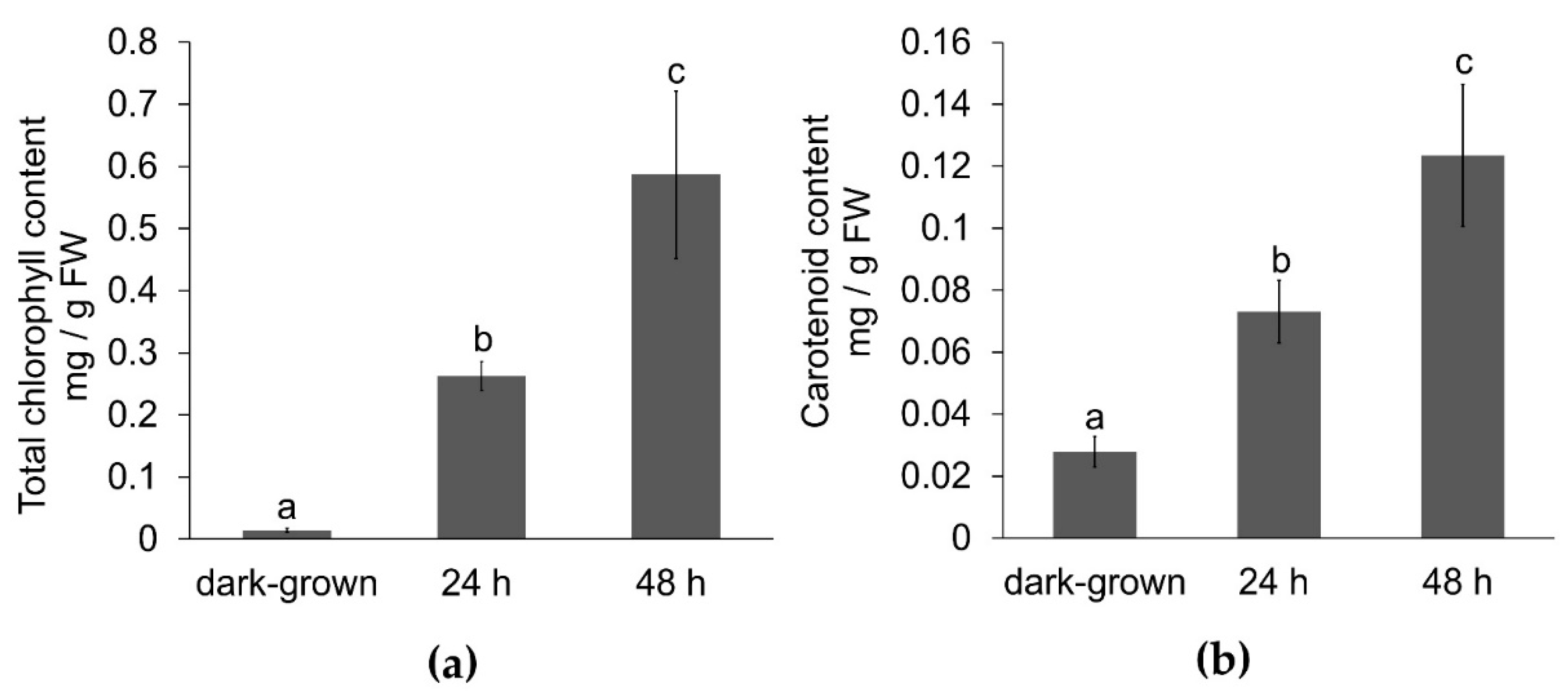
3.1. Effect of Illumination on Carotenoid and Chlorophyll Content
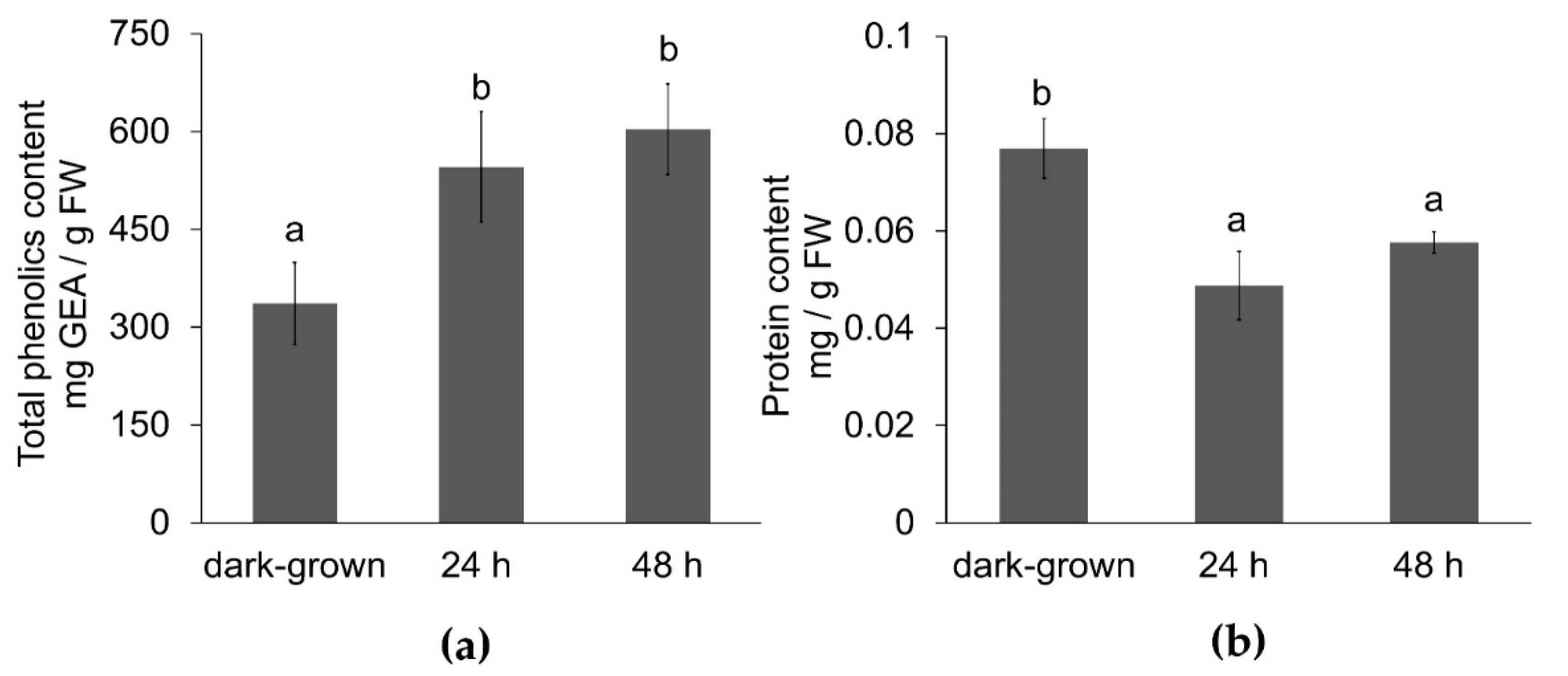
3.2. Effect of Illumination Total Phenolic and Protein Content
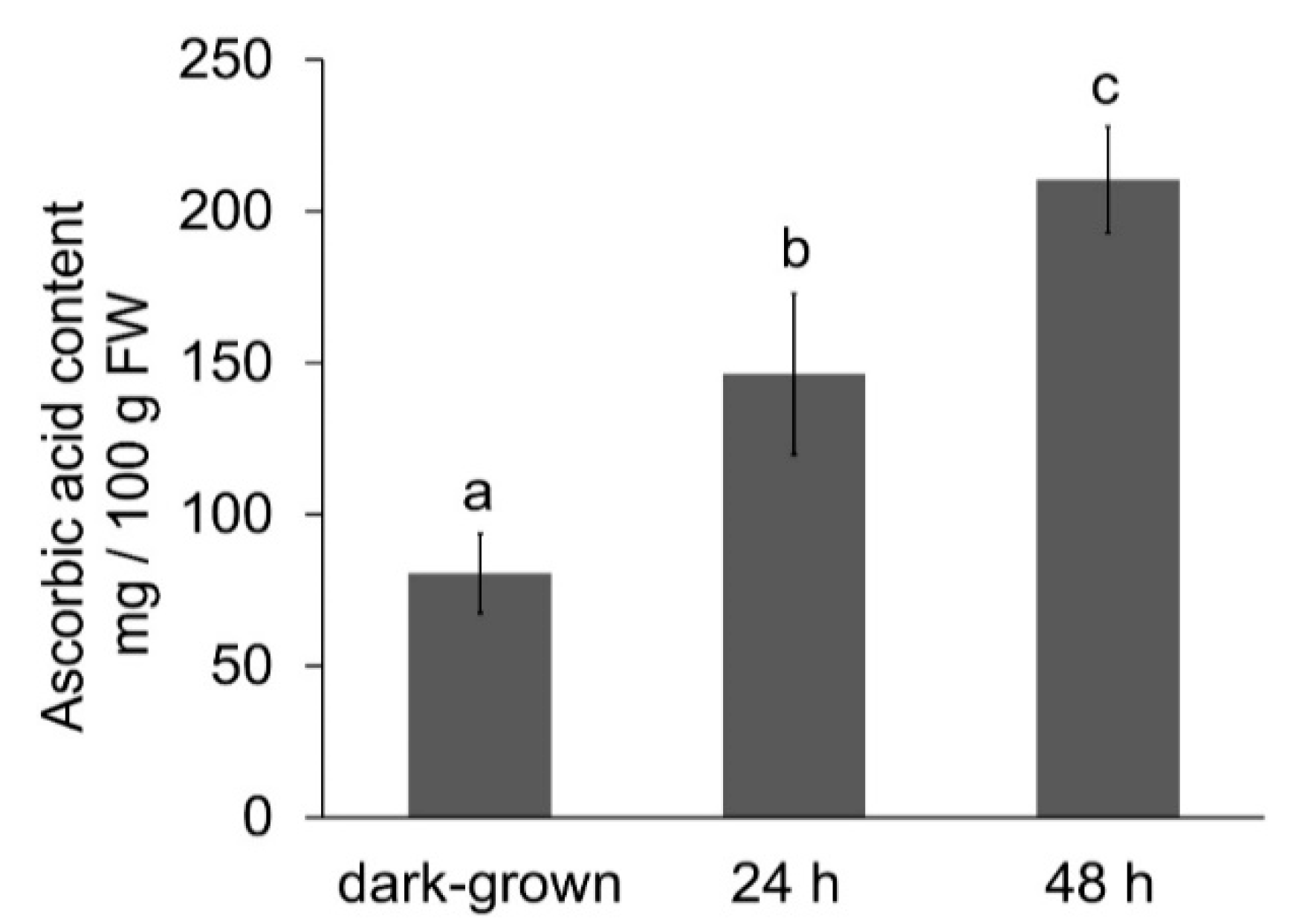
3.3. Effect of Illumination on Ascorbic Acid Content
3.4. Crystalline Cellulose Content
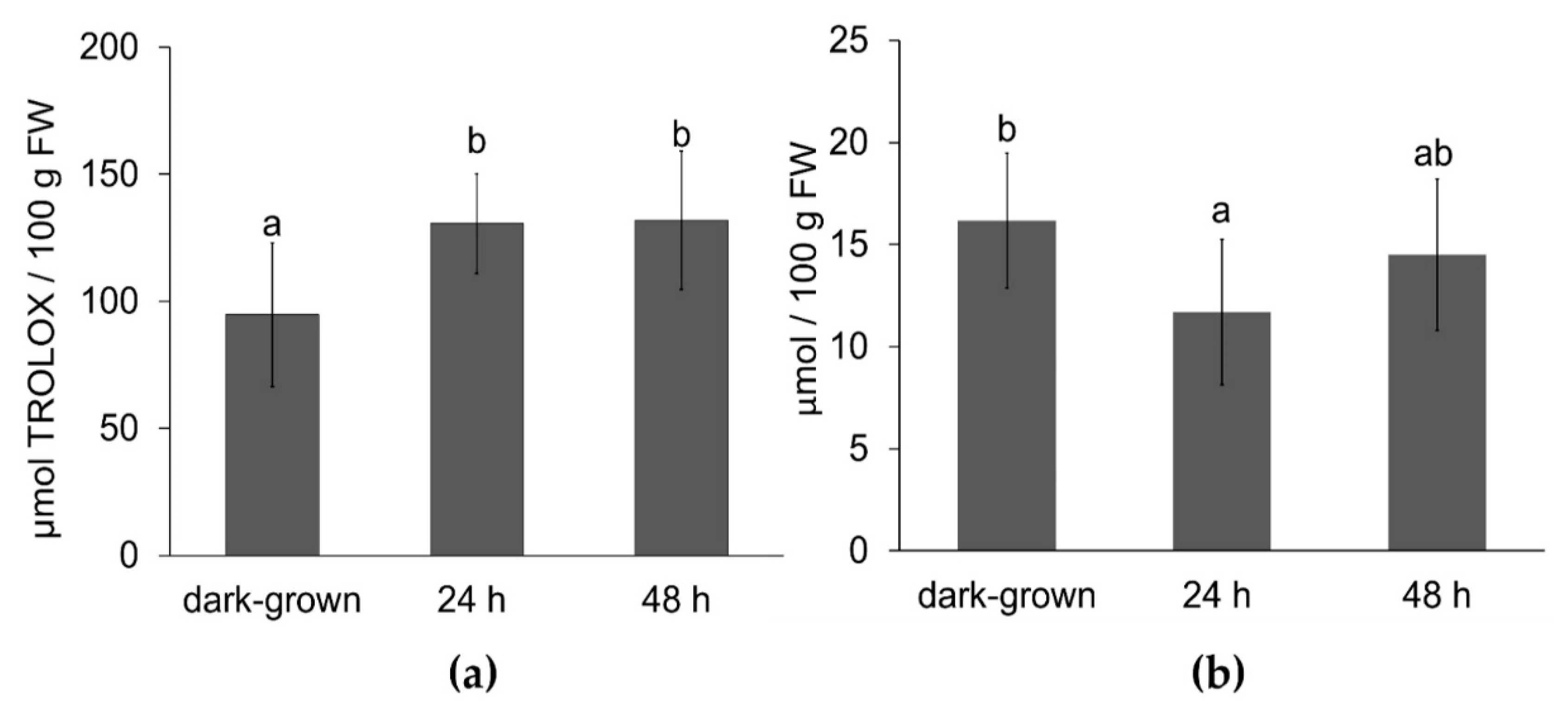
3.5. Effect of Illumination on Antioxidant Capacity
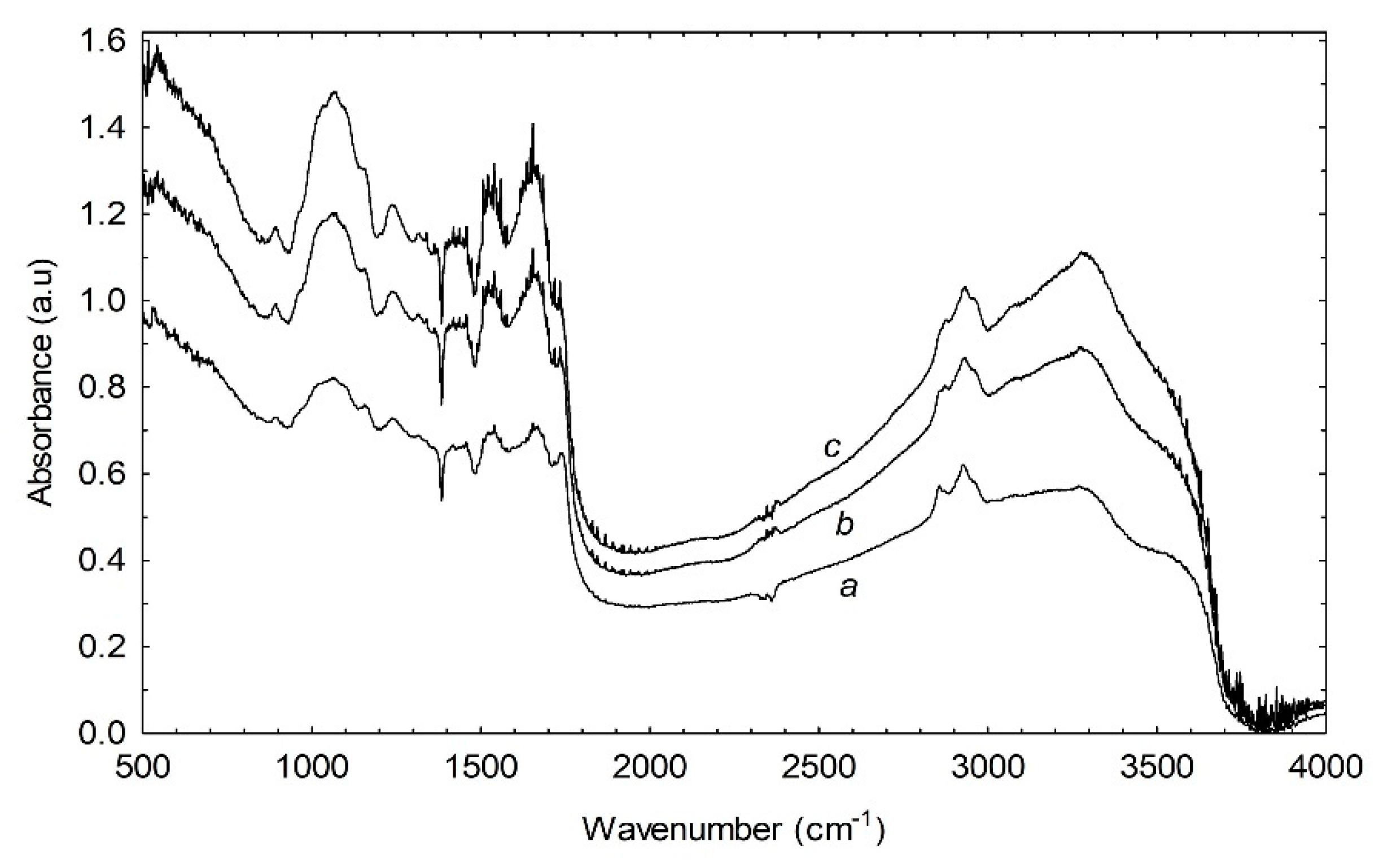
3.6. Effect of Illumination on FTIR Spectra
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cahill, J.P. Ethnobotany of Chia, Salvia hispanica L. (Lamiaceae). Econ. Bot. 2003, 57, 604–618. [Google Scholar] [CrossRef]
- Borneo, R.; Aguirre, A.; León, A.E. Chia (Salvia hispanica L) Gel Can Be Used as Egg or Oil Replacer in Cake Formulations. J. Am. Diet. Assoc. 2010, 110, 946–949. [Google Scholar] [CrossRef] [PubMed]
- da Silva Marineli, R.; Moraes, É.A.; Lenquiste, S.A.; Godoy, A.T.; Eberlin, M.N.; Maróstica, M.R., Jr. Chemical characterization and antioxidant potential of Chilean chia seeds and oil (Salvia hispanica L.). LWT Food Sci. Technol. 2014, 59, 1304–1310. [Google Scholar] [CrossRef]
- del Carmen Beltran-Orozco, M.; Martinez-Olguin, A.; del Carmen Robles-Ramirez, M. Changes in the Nutritional Composition and Antioxidant Capacity of Chia Seeds (Salvia hispanica L.) During Germination Process. Food Sci. Biotechnol. 2020, 29, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Favela, M.A.; Gutiérrez-Dorado, R.; Cuevas-Rodríguez, E.; Canizález-Román, A.; León-Sicairos, C.D.R.; Milán-Carrillo, J.; Reyes-Moreno, C. Improvement of Chia Seeds with Antioxidant Activity, GABA, Essential Amino Acids, and Dietary Fiber by Controlled Germination Bioprocess. Plant Foods Hum. Nutr. 2017, 72, 345–352. [Google Scholar] [CrossRef]
- Benincasa, P.; Falcinelli, B.; Lutts, S.; Stagnari, F.; Galieni, A. Sprouted Grains: A Comprehensive Review. Nutrients 2019, 11, 421. [Google Scholar] [CrossRef]
- Gan, R.-Y.; Lui, W.-Y.; Wu, K.; Chan, C.-L.; Dai, S.-H.; Sui, Z.-Q.; Corke, H. Bioactive compounds and bioactivities of germinated edible seeds and sprouts: An updated review. Trends Food Sci. Technol. 2017, 59, 1–14. [Google Scholar] [CrossRef]
- Wang, H.; Gui, M.; Tian, X.; Xin, X.; Li, J. Effects of UV-B on vitamin C, phenolics, flavonoids and their related enzyme activities in mung bean sprouts (Vigna radiata). Int. J. Food Sci. Technol. 2017, 52, 827–833. [Google Scholar] [CrossRef]
- Samuolienė, G.; Urbonavičiūtė, A.; Brazaitytė, A.; Šabajevienė, G.; Sakalauskaitė, J.; Duchovskis, P. The impact of LED illumination on antioxidant properties of sprouted seeds. Open Life Sci. 2011, 6, 68–74. [Google Scholar] [CrossRef]
- Chen, C.-C.; Huang, M.Y.; Lin, K.H.; Wong, S.L.; Huang, W.D.; Yang, C.M. Effects of Light Quality on the Growth, Development and Metabolism of Rice Seedlings (Oryza sativa L.). Res. J. Biotechnol. 2014, 9, 15–24. [Google Scholar]
- Lu, Y.; Guo, X. The Effect of Light in Vitamin C Metabolism Regulation and Accumulation in Mung Bean (Vigna radiata) Germination. Plant Foods Hum. Nutr. 2020, 75, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Ntagkas, N.; Woltering, E.J.; Marcelis, L.F. Light regulates ascorbate in plants: An integrated view on physiology and biochemistry. Environ. Exp. Bot. 2018, 147, 271–280. [Google Scholar] [CrossRef]
- Bailly, C. Active oxygen species and antioxidants in seed biology. Seed Sci. Res. 2004, 14, 93–107. [Google Scholar] [CrossRef]
- Sun, T.; Li, L. Toward the ‘golden’ era: The status in uncovering the regulatory control of carotenoid accumulation in plants. Plant Sci. 2020, 290, 110331. [Google Scholar] [CrossRef]
- Rodríguez-Villalón, A.; Gas, E.; Rodríguez-Concepción, M. Colors in the Dark: A Model for the Regulation of Carotenoid Biosynthesis in Etioplasts. Plant Signal. Behav. 2009, 4, 965–967. [Google Scholar] [CrossRef]
- Kyriacou, M.C.; De Pascale, S.; Kyratzis, A.; Rouphael, Y. Microgreens as a Component of Space Life Support Systems: A Cornucopia of Functional Food. Front. Plant Sci. 2017, 8, 1587. [Google Scholar] [CrossRef]
- Baseri, M.K.; Baker, S. Identification of Cellular Components of Medicinal Plants Using Ftir. Rom. J. Biophys. 2011, 21, 277–284. [Google Scholar]
- Carrión-Prieto, P.; Martín-Ramos, P.; Hernández-Navarro, S.; Silva-Castro, I.; Silva, M.R.; Martín-Gil, J. Vibrational Analysis and Thermal Behavior of Salvia hispanica, Nigella sativa and Papaver somniferum Seeds. Pharmacogn. J. 2017, 9, 157–162. [Google Scholar] [CrossRef]
- Vargas, O.L.T.; Salcedo, A.J.G.; Calderón, H.A. Physical-chemical characterization of quinoa (Chenopodium quinoa Willd.), amaranth (Amaranthus caudatus L.), and chia (Salvia hispanica L.) flours and seeds. Acta Agron. 2018, 67, 215–222. [Google Scholar] [CrossRef]
- Darwish, A.; Khalifa, R.E.; El Sohaimy, S.A. Functional Properties of Chia Seed Mucilage Supplemented In Low Fat Yoghurt. Alex. Sci. Exch. J. 2018, 39, 450–459. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. [34] Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Bessey, O.A.; Lowry, O.H.; Brock, M.J. A method for the rapid determination of alkaline phosphates with five cubic millimeters of serum. J. Biol. Chem. 1946, 164, 321–329. [Google Scholar]
- Updegraff, D.M. Semimicro determination of cellulose inbiological materials. Anal. Biochem. 1969, 32, 420–424. [Google Scholar] [CrossRef]
- Foster, C.E.; Martin, T.M.; Pauly, M. Comprehensive Compositional Analysis of Plant Cell Walls (Lignocellulosic Biomass) Part II: Carbohydrates. JoVE (J. Vis. Exp.) 2010, 37, e1837. [Google Scholar] [CrossRef]
- de Paiva, E.P.; Torres, S.B.; Sá, F.V.; Nogueira, N.W.; Freitas, R.M.; Leite, M.D. Light regime and temperature on seed germination in Salvia hispanica L. Acta Sci. Agron. 2016, 38, 513–519. [Google Scholar] [CrossRef]
- Solymosi, K.; Mysliwa-Kurdziel, B. Chlorophylls and their Derivatives Used in Food Industry and Medicine. Mini Rev. Med. Chem. 2017, 17, 1194–1222. [Google Scholar] [CrossRef]
- Zepka, L.Q.; Jacob-Lopes, E.; Roca, M. Catabolism and bioactive properties of chlorophylls. Curr. Opin. Food Sci. 2019, 26, 94–100. [Google Scholar] [CrossRef]
- Zheng, H.; You, Y.; Hua, M.; Wu, P.; Liu, Y.; Chen, Z.; Zhang, L.; Wei, H.; Li, Y.; Luo, M. Chlorophyllin Modulates Gut Microbiota and Inhibits Intestinal Inflammation to Ameliorate Hepatic Fibrosis in Mice. Front. Physiol. 2018, 9, 1671. [Google Scholar] [CrossRef]
- Honarvar, N.M.; Saedisomeolia, A.; Abdolahi, M.; Shayeganrad, A.; Sangsari, G.T.; Rad, B.H.; Muench, G. Molecular Anti-inflammatory Mechanisms of Retinoids and Carotenoids in Alzheimer’s Disease: A Review of Current Evidence. J. Mol. Neurosci. 2016, 61, 289–304. [Google Scholar] [CrossRef] [PubMed]
- Elvira-Torales, L.I.; García-Alonso, F.J.; Periago, M.J. Nutritional Importance of Carotenoids and Their Effect on Liver Health: A Review. Antioxidants 2019, 8, 229. [Google Scholar] [CrossRef] [PubMed]
- Bahonar, A.; Saadatnia, M.; Khorvash, F.; Maracy, M.; Khosravi, M. Carotenoids as potential antioxidant agents in stroke prevention: A systematic review. Int. J. Prev. Med. 2017, 8. [Google Scholar] [CrossRef]
- Edge, R.; Truscott, T.G. Singlet Oxygen and Free Radical Reactions of Retinoids and Carotenoids—A Review. Antioxidants 2018, 7, 5. [Google Scholar] [CrossRef] [PubMed]
- Ilieva, I.; Ivanova, T.; Naydenov, Y.; Dandolov, I.; Stefanov, D. Plant experiments with light-emitting diode module in Svet space greenhouse. Adv. Space Res. 2010, 46, 840–845. [Google Scholar] [CrossRef]
- Walters, R.G.; Shephard, F.; Rogers, J.J.; Rolfe, S.A.; Horton, P. Identification of Mutants of Arabidopsis Defective in Acclimation of Photosynthesis to the Light Environment1. Plant Physiol. 2003, 131, 472–481. [Google Scholar] [CrossRef]
- Weiguo, F.; Pingping, L.; Yanyou, W.; Jianjian, T. Effects of different light intensities on anti-oxidative enzyme activity, quality and biomass in lettuce. Hortic. Sci. 2012, 39, 129–134. [Google Scholar] [CrossRef]
- Mitrović, J.S.; Nikolić, N.; Karabegović, I.; Lazić, M.; Stojanović, G. Characterization of free and insoluble-bound phenolics of chia (Salvia hispanica L.) seeds. Nat. Prod. Res. 2020, 1–5. [Google Scholar] [CrossRef]
- Paśko, P.; Barton, H.J.; Zagrodzki, P.; Gorinstein, S.; Fołta, M.; Zachwieja, Z. Anthocyanins, total polyphenols and antioxidant activity in amaranth and quinoa seeds and sprouts during their growth. Food Chem. 2009, 115, 994–998. [Google Scholar] [CrossRef]
- Guzmán-Ortiz, F.A.; Martín-Martínez, E.S.; Valverde, M.E.; Rodríguez-Aza, Y.; Berríos, J.D.J.; Mora-Escobedo, R. Profile Analysis and Correlation across Phenolic Compounds, Isoflavones and Antioxidant Capacity during Germination of Soybeans (Glycine max L.). CyTA J. Food 2017, 15, 516–524. [Google Scholar] [CrossRef]
- Wu, Z.; Song, L.; Feng, S.; Liu, Y.; He, G.; Yioe, Y.; Liu, S.; Huang, D.-J. Germination Dramatically Increases Isoflavonoid Content and Diversity in Chickpea (Cicer arietinum L.) Seeds. J. Agric. Food Chem. 2012, 60, 8606–8615. [Google Scholar] [CrossRef]
- Xu, M.; Jin, Z.; Ohm, J.B.; Schwarz, P.; Rao, J.; Chen, B. Improvement of the Antioxidative Activity of Soluble Phenolic Compounds in Chickpea by Germination. J. Agric. Food Chem. 2018, 66, 6179–6187. [Google Scholar] [CrossRef]
- Mastropasqua, L.; Dipierro, N.; Paciolla, C. Effects of Darkness and Light Spectra on Nutrients and Pigments in Radish, Soybean, Mung Bean and Pumpkin Sprouts. Antioxidants 2020, 9, 558. [Google Scholar] [CrossRef] [PubMed]
- Smirnoff, N. Ascorbic Acid Metabolism and Functions: A Comparison of Plants and Mammals. Free Radic. Boil. Med. 2018, 122, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Dowdle, J.; Ishikawa, T.; Gatzek, S.; Rolinski, S.; Smirnoff, N. Two Genes in Arabidopsis Thaliana Encoding Gdp-L-Galactose Phosphorylase Are Required for Ascorbate Biosynthesis and Seedling Viability. Plant J. 2007, 52, 673–689. [Google Scholar] [CrossRef]
- Talla, S.; Riazunnisa, K.; Padmavathi, L.; Sunil, B.; Rajsheel, P.; Raghavendra, A. Ascorbic Acid Is a Key Participant During the Interactions between Chloroplasts and Mitochondria to Optimize Photosynthesis and Protect against Photoinhibition. J. Biosci. 2011, 36, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Reyes-Caudillo, E.; Tecante, A.; Valdivia-López, M. Dietary fibre content and antioxidant activity of phenolic compounds present in Mexican chia (Salvia hispanica L.) seeds. Food Chem. 2008, 107, 656–663. [Google Scholar] [CrossRef]
- Nelson, K.; Stojanovska, L.; Vasiljevic, T.; Mathai, M. Germinated grains: A superior whole grain functional food? Can. J. Physiol. Pharmacol. 2013, 91, 429–441. [Google Scholar] [CrossRef]
- Martín-Cabrejas, M.A.; Ariza, N.; Esteban, R.M.; Mollá, E.; Waldron, K.; López-Andréu, F.J. Effect of Germination on the Carbohydrate Composition of the Dietary Fiber of Peas (Pisum sativum L.). J. Agric. Food Chem. 2003, 51, 1254–1259. [Google Scholar] [CrossRef] [PubMed]
- Perales-Sánchez, J.X.K.; Reyes-Moreno, C.; Gómez-Favela, M.A.; Milán-Carrillo, J.; Cuevas-Rodríguez, E.; Valdez-Ortiz, A.; Gutiérrez-Dorado, R. Increasing the Antioxidant Activity, Total Phenolic and Flavonoid Contents by Optimizing the Germination Conditions of Amaranth Seeds. Plant Foods Hum. Nutr. 2014, 69, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Tiansawang, K.; Luangpituksa, P.; Varanyanond, W.; Hansawasdi, C. Gaba (Γ-Aminobutyric Acid) Production, Antioxidant Activity in Some Germinated Dietary Seeds and the Effect of Cooking on Their Gaba Content. Food Sci. Technol. 2016, 36, 313–321. [Google Scholar] [CrossRef]
- Mølmann, J.A.B.; Johansen, T.J. Sprout Growth Inhibition and Photomorphogenic Development of Potato Seed Tubers (Solanum tuberosum L.) Under Different LED Light Colours. Potato Res. 2020, 63, 199–215. [Google Scholar]
- Dong, J.; Terzaghi, W.B.; Deng, X.W.; Chen, H. Multiple Photomorphogenic Repressors Work in Concert to Regulate Arabidopsis Seedling Development. Plant Signal. Behav. 2015, 10, e1011934. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pająk, P.; Socha, R.; Broniek, J.; Królikowska, K.; Fortuna, T. Antioxidant Properties, Phenolic and Mineral Composition of Germinated Chia, Golden Flax, Evening Primrose, Phacelia and Fenugreek. Food Chem. 2019, 275, 69–76. [Google Scholar] [CrossRef]
- Pellegrini, M.; Lucas-González, R.; Sayas-Barberá, E.; Fernández-López, J.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Bioaccessibility of Phenolic Compounds and Antioxidant Capacity of Chia (Salvia hispanica L.) Seeds. Plant Foods Hum. Nutr. 2017, 73, 47–53. [Google Scholar] [CrossRef]
| Dark-Grown | 24 h | 48 h |
|---|---|---|
| 0.656 a | 0.657 a | 0.317 b |
| (±0.08) | (±0.09) | (±0.02) |
| Peak No. | Assignments | Dark-Grown Wavenumber (cm−1) | 24 h Wavenumber (cm−1) | 48 h Wavenumber (cm−1) |
|---|---|---|---|---|
| I | not assigned | 889.9 | 890 | 889 |
| II | Polysaccharides | 1062 | 1062 | 1063 |
| III | Amide III region | 1238 | 1239 | 1239 |
| IV | Amide III region | 1457 | 1457 | 1457 |
| V | Amide II region | 1540 | 1540 | 1540 |
| VI | Amide I region | 1647 | 1647 | 1647 |
| VII | Fat content | 2855 | disappeared | disappeared |
| VIII | Fat content | 2926 | 2927 | 2925 |
| IX | Protein content | 3269 | 3270 | 3271 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mlinarić, S.; Gvozdić, V.; Vuković, A.; Varga, M.; Vlašiček, I.; Cesar, V.; Begović, L. The Effect of Light on Antioxidant Properties and Metabolic Profile of Chia Microgreens. Appl. Sci. 2020, 10, 5731. https://doi.org/10.3390/app10175731
Mlinarić S, Gvozdić V, Vuković A, Varga M, Vlašiček I, Cesar V, Begović L. The Effect of Light on Antioxidant Properties and Metabolic Profile of Chia Microgreens. Applied Sciences. 2020; 10(17):5731. https://doi.org/10.3390/app10175731
Chicago/Turabian StyleMlinarić, Selma, Vlatka Gvozdić, Ana Vuković, Martina Varga, Ivan Vlašiček, Vera Cesar, and Lidija Begović. 2020. "The Effect of Light on Antioxidant Properties and Metabolic Profile of Chia Microgreens" Applied Sciences 10, no. 17: 5731. https://doi.org/10.3390/app10175731
APA StyleMlinarić, S., Gvozdić, V., Vuković, A., Varga, M., Vlašiček, I., Cesar, V., & Begović, L. (2020). The Effect of Light on Antioxidant Properties and Metabolic Profile of Chia Microgreens. Applied Sciences, 10(17), 5731. https://doi.org/10.3390/app10175731





