Oxidation of Florfenicol and Oxolinic Acid in Seawater by Ozonation
Abstract
:Article Highlights
- It is necessary to prevent antibiotics used in marine aquaculture from entering the oceans and harming the aquatic environment.
- In seawater ozonation, ozone reacts rapidly with bromide ion to produce bromine. The primary oxidants of seawater ozonation are trace ozone and stable bromine.
- Ozone and bromine were recommended for FF and OA removal in seawater.
1. Introduction
2. Materials and Methods
2.1. Materials
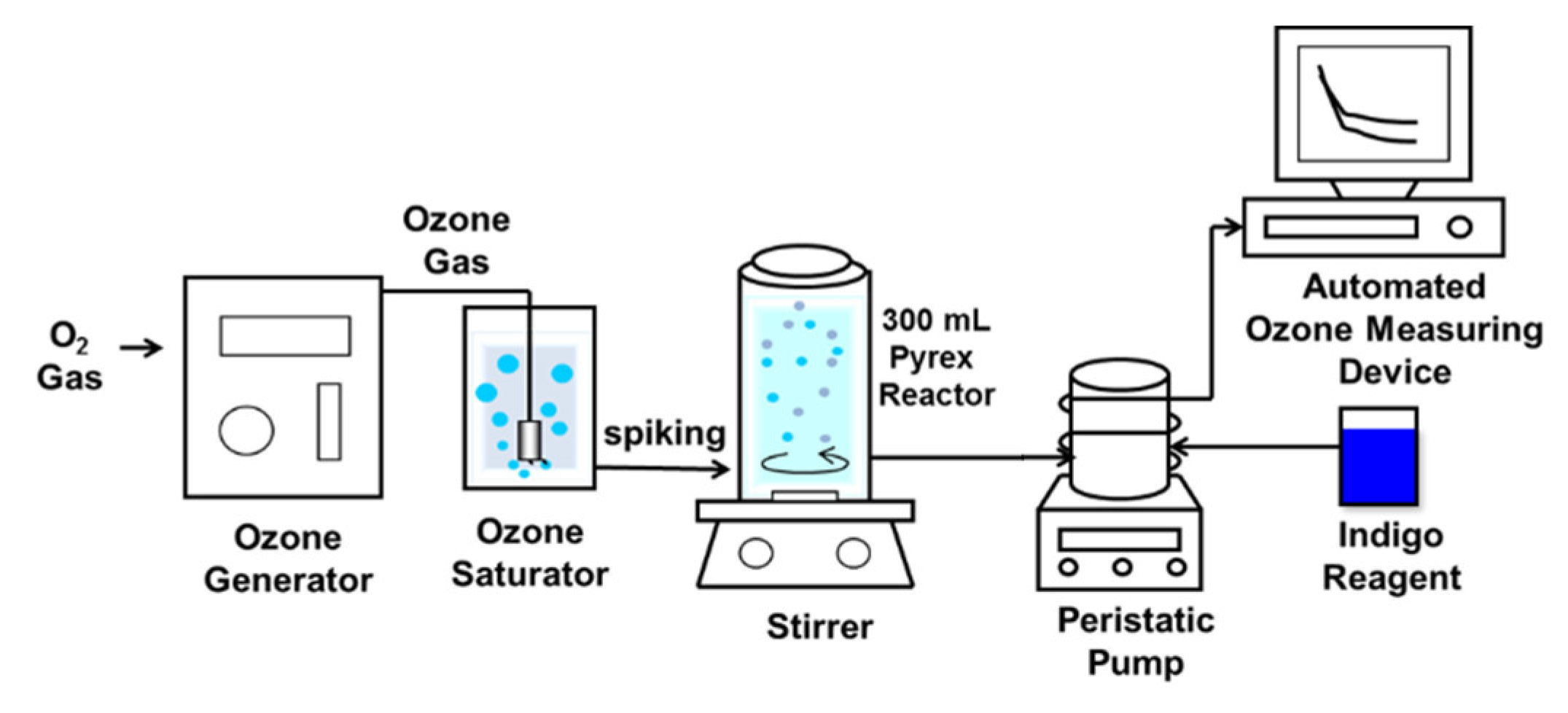
2.2. Experimental Procedure
2.2.1. Reaction Rate with Ozone
2.2.2. Reaction Rate with Bromine
2.3. Analytical Method
2.4. Removal of Antibiotics from Bromide Containing Water
3. Results and Discussion
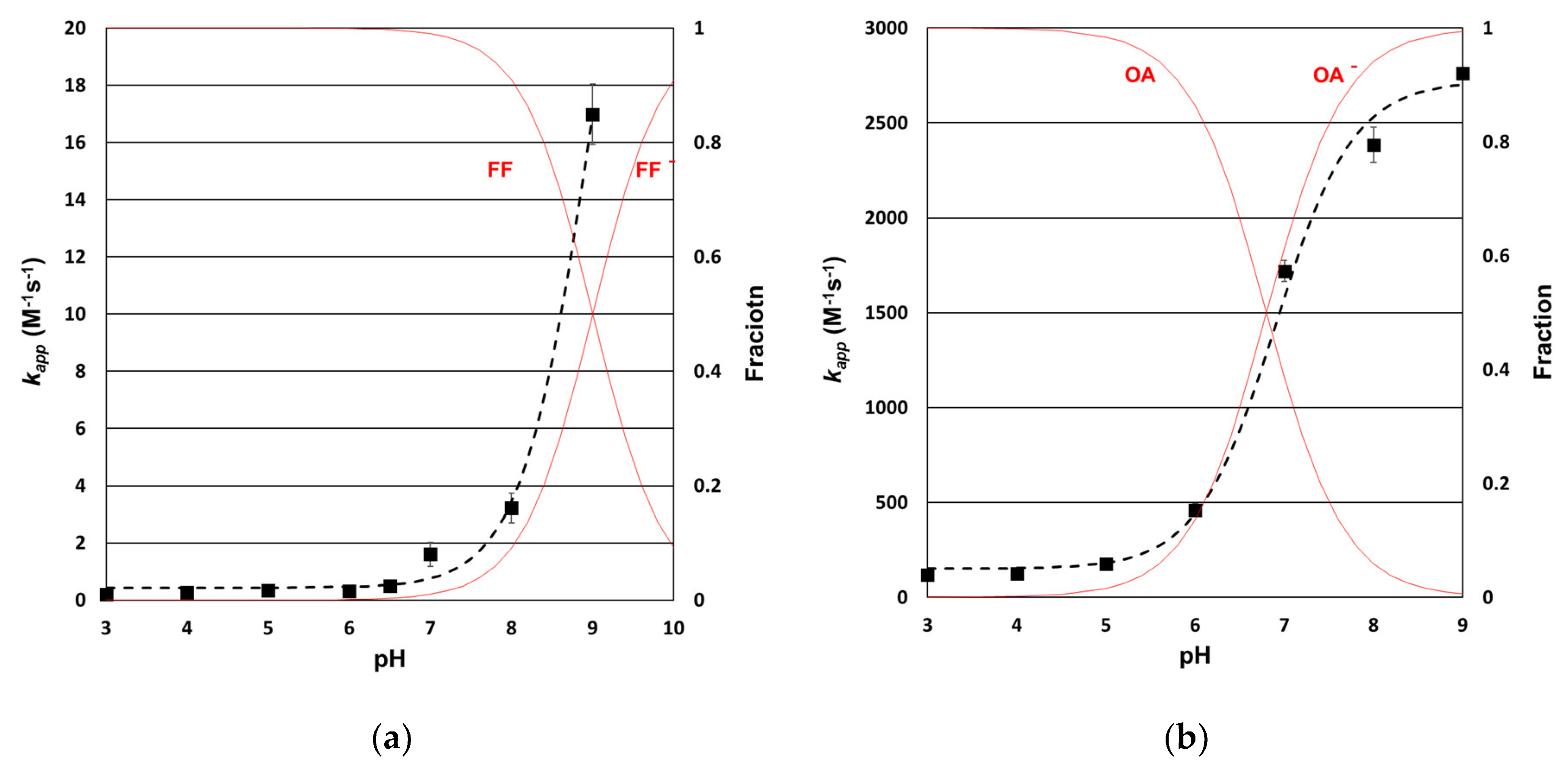
3.1. Determination of Ozone Rate Constants for the Selected Antibiotics
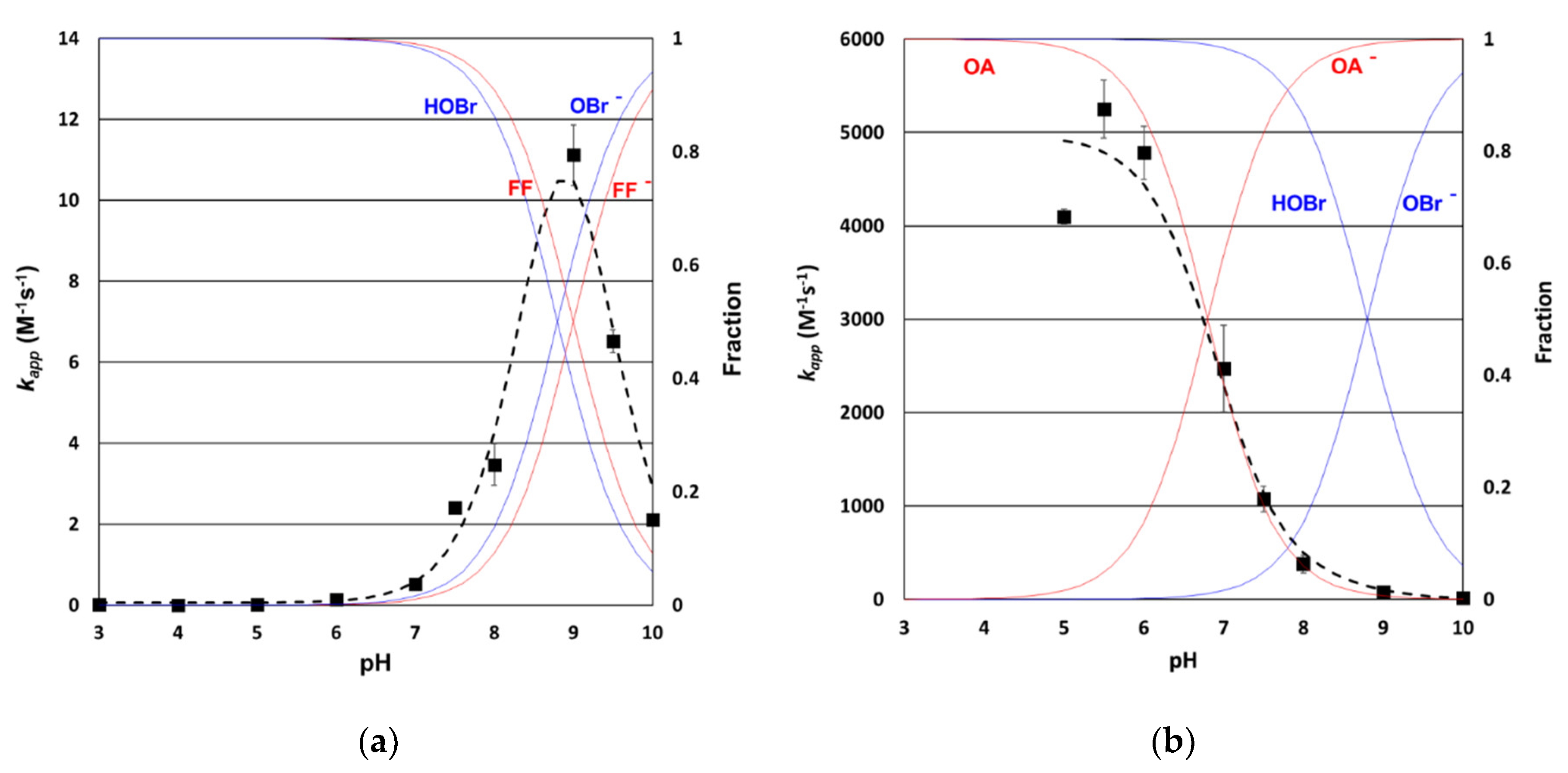
3.2. Determination of Bromine Rate Constants for the Selected Antibiotics
3.3. Prediction of Antibiotics Removal Efficiency in Absence of Br—In Synthetic Water
3.3.1. Formation of Bromine in Seawater Ozonation
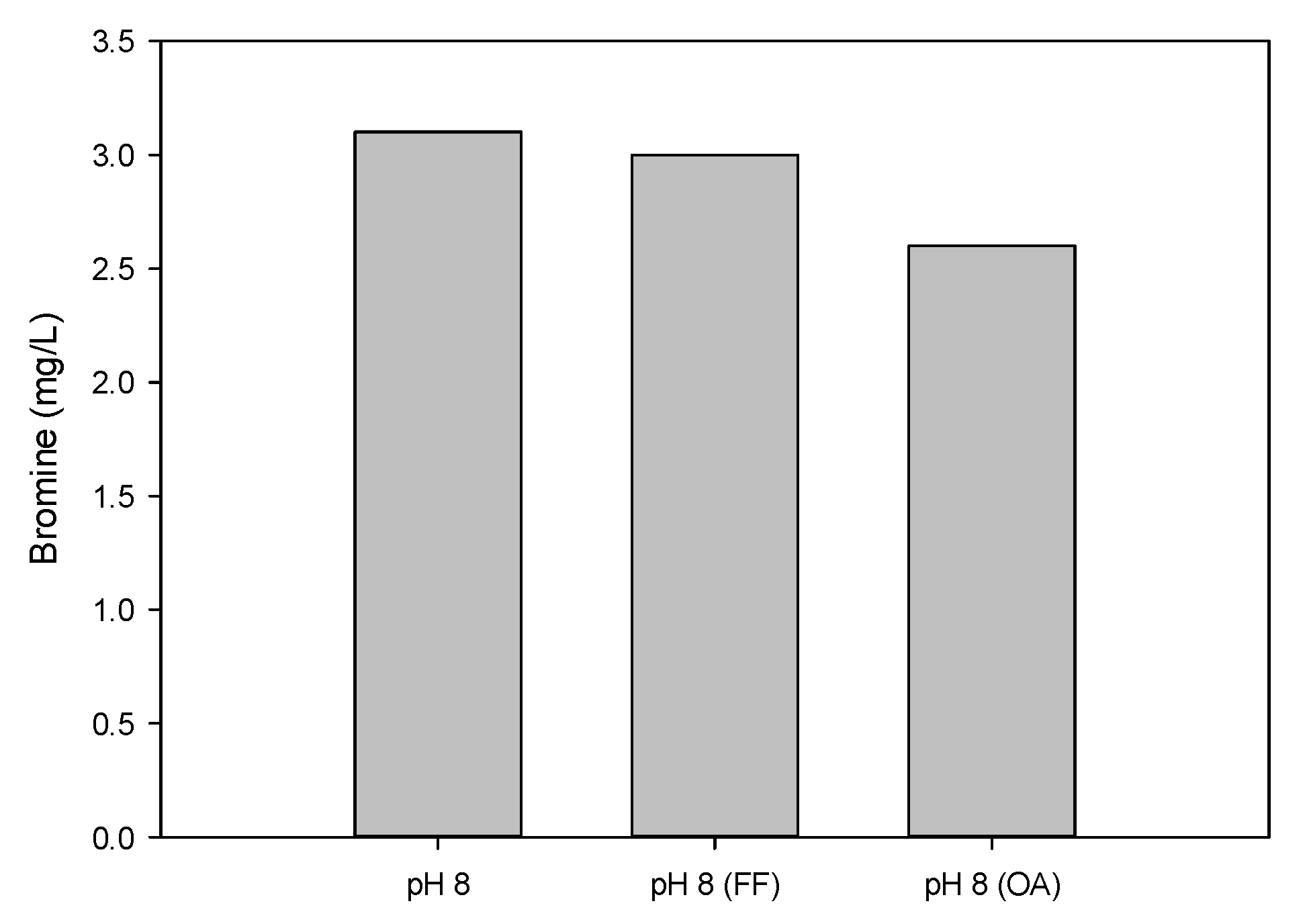
3.3.2. Removal of FF and OA by Ozonation and Bromination
3.4. Removal of FF and OA by Ozonation in Water Containing Br−
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gao, Y.Q.; Gao, N.Y.; Deng, Y.; Yin, D.Q.; Zhang, Y.S. Degradation of florfenicol in water by UV/Na2S2O8 process. Environ. Sci. Pollut. 2015, 22, 8693–8701. [Google Scholar] [CrossRef] [PubMed]
- Norambuena, L.; Gras, N.; Contreras, S. Development and validation of a method for the simultaneous extraction and separate measurement of oxytetracycline, florfenicol, oxolinic acid and flumequine from marine sediments. Mar. Pollut. Bull. 2013, 73, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.-B.; Huang, D.-R.; Zhong, M.; Liu, P.; Dong, J.-D. Pharmacokinetics of florfenicol and behaviour of its metabolite florfenicol amine in orange-spotted grouper (Epinephelus coioides) after oral administration. J. Fish. Diseas. 2016, 39, 833–843. [Google Scholar] [CrossRef] [PubMed]
- Christensen, A.M.; Ingerslev, F.; Baun, A. Ecotoxicity of mixtures of antibiotics used in aquacultures. Environ. Toxicol Chem. 2006, 25, 2208–2215. [Google Scholar] [CrossRef]
- Sapkota, A.; Sapkota, A.R.; Kucharski, M.; Burke, J.; McKenzie, S.; Walker, P.; Lawrence, R. Aquaculture practices and potential human health risks: Current knowledge and future priorities. Environ. Int. 2008, 34, 1215–1226. [Google Scholar] [CrossRef]
- Palominos, R.A.; Mora, A.; Mondaca, M.A.; Pérez-Moya, M.; Mansilla, H.D. Oxolinic acid photo-oxidation using immobilized TiO2. J. Hazard. Mater. 2008, 158, 460–464. [Google Scholar] [CrossRef]
- Rigos, G.; Nengas, I.; Alexis, M.; Troisi, G.M. Potential drug (oxytetracycline and oxolinic acid) pollution from Mediterranean sparid fish farms. Aquat. Toxicol. 2004, 69, 281–288. [Google Scholar] [CrossRef]
- Lai, H.-T.; Chien, Y.-H.; Lin, J.-S. Long-Term transformation of oxolinic acid in water from an eel pond. Aquaculture 2008, 275, 96–101. [Google Scholar] [CrossRef]
- Sutherland, T.F.; Levings, C.D.; Elliott, C.C.; Hesse, W.W. Effect of a ballast water treatment system on survivorship of natural populations of marine plankton. Mar. Ecol. Prog Ser. 2001, 210, 139–148. [Google Scholar] [CrossRef]
- Waite, T.D.; Kazumi, J.; Lane, P.V.Z.; Farmer, L.L.; Smith, S.G.; Smith, S.L.; Hitchcock, G.; Cap, T.R. Removal of natural populations of marine plankton by a large-Scale ballast water treatment system. Mar. Ecol. Prog Ser. 2003, 258, 51–63. [Google Scholar] [CrossRef]
- Tang, Z.J.; Butkus, M.A.; Xie, Y.F.F. Crumb rubber filtration: A potential technology for ballast water treatment. Mar. Environ. Res. 2006, 61, 410–423. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.J.; Yoon, Y.; Pyo, T.S.; Lee, S.T.; Shin, K.; Kang, J.W. Evaluation of disinfection efficacy and chemical formation using MPUV ballast water treatment system (GloEn-Patrol (TM)). Environ. Technol. 2012, 33, 1953–1961. [Google Scholar] [CrossRef] [PubMed]
- Summerfelt, S.T.; Hochheimer, J.N. Review of ozone processes and applications as an oxidizing agent in aquaculture. Prog Fish. Cult. 1997, 59, 94–105. [Google Scholar] [CrossRef]
- Powell, A.; Chingombe, P.; Lupatsch, I.; Shields, R.J.; Lloyd, R. The effect of ozone on water quality and survival of turbot (Psetta maxima) maintained in a recirculating aquaculture system. Aquacult Eng. 2015, 64, 20–24. [Google Scholar] [CrossRef]
- Perrins, J.C.; Cooper, W.J.; van Leeuwen, J.H.; Herwig, R.P. Ozonation of seawater from different locations: Formation and decay of total residual oxidant - implications for ballast water treatment. Mar. Pollut Bull. 2006, 52, 1023–1033. [Google Scholar] [CrossRef]
- Jung, Y.; Yoon, Y.; Hong, E.; Kwon, M.; Kang, J.W. Inactivation characteristics of ozone and electrolysis process for ballast water treatment using B. subtilis spores as a probe. Mar. Pollut Bull. 2013, 72, 71–79. [Google Scholar] [CrossRef]
- Heeb, M.B.; Criquet, J.; Zimmermann-Steffens, S.G.; von Gunten, U. Oxidative treatment of bromide-containing waters: Formation of bromine and its reactions with inorganic and organic compounds—A critical review. Water Res. 2014, 48, 1–28. [Google Scholar] [CrossRef]
- Pinkernell, U.; Nowack, B.; Gallard, H.; von Gunten, U. Methods for the photometric determination of reactive bromine and chlorine species with ABTS. Water Res. 2000, 34, 4343–4350. [Google Scholar] [CrossRef]
- Buxton, G.V.; Bydder, M.; Arthur salmon, G. Reactivity of chlorine atoms in aqueous solution Part 1 The equilibrium ClMNsbd+Cl−Cl2−. J. Chem. Soc. Faraday Trans. 1998, 94, 653–657. [Google Scholar] [CrossRef]
- Barder, H.; Hoigné, J. Determination of ozone in water by the indigo method. Water Res. 1981, 15, 449–456. [Google Scholar] [CrossRef]
- Jung, Y.; Hong, E.; Kwon, M.; Kang, J.-W. A kinetic study of ozone decay and bromine formation in saltwater ozonation: Effect of O3 dose, salinity, pH, and Temperature. Chem. Eng. J. 2017, 31, 30–38. [Google Scholar] [CrossRef]



| Florfenicol (FF) | Classes | A Fluorinated Derivative of Thiamphenicol |
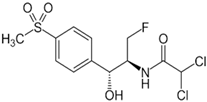 | gram-negative and gram-positive bacteria | |
| M.F. | C12H14Cl2FNO4S | |
| M.W. | 358.21 g/mol | |
| pKa | 9.0 (reference) | |
| Oxolinic Acid (OA) | Classes | Quinolone |
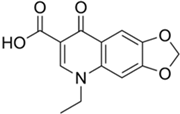 | gram-negative and gram-positive bacteria | |
| M.F. | C13H11NO5 | |
| M.W. | 261.23 g/mol | |
| pKa | 6.8 (reference) | |
| Florfenicol | ||
|---|---|---|
| [Br2]0 (mM) | PH | Kapp |
| 5 | 3 | 4.5 10−3 |
| 5 | 7 | 5.2 10−1 |
| 3 | 8 | 3.8 |
| 5 | 8 | 3.7 |
| Pharmaceutical | pKa | Ozone | Bromine |
|---|---|---|---|
| Kapp | Kapp | ||
| Florfenicol (FF) | 9.0 | 3.2 | 3.5 |
| Oxolinic acid (OA) | 6.8 | 2.4 103 | 4.0 102 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kye, H.; Oh, H.; Jung, Y.; Kwon, M.; Yoon, Y.; Kang, J.-W.; Hwang, T.-M. Oxidation of Florfenicol and Oxolinic Acid in Seawater by Ozonation. Appl. Sci. 2020, 10, 4944. https://doi.org/10.3390/app10144944
Kye H, Oh H, Jung Y, Kwon M, Yoon Y, Kang J-W, Hwang T-M. Oxidation of Florfenicol and Oxolinic Acid in Seawater by Ozonation. Applied Sciences. 2020; 10(14):4944. https://doi.org/10.3390/app10144944
Chicago/Turabian StyleKye, Homin, Heegun Oh, Youmi Jung, Minhwan Kwon, Yeojoon Yoon, Joon-Wun Kang, and Tae-Mun Hwang. 2020. "Oxidation of Florfenicol and Oxolinic Acid in Seawater by Ozonation" Applied Sciences 10, no. 14: 4944. https://doi.org/10.3390/app10144944
APA StyleKye, H., Oh, H., Jung, Y., Kwon, M., Yoon, Y., Kang, J.-W., & Hwang, T.-M. (2020). Oxidation of Florfenicol and Oxolinic Acid in Seawater by Ozonation. Applied Sciences, 10(14), 4944. https://doi.org/10.3390/app10144944







