Nascent Rice Husk as an Adsorbent for Removing Cationic Dyes from Textile Wastewater
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Adsorbate Solution
2.2. Thermal Treatment and Characterization of Rice Husk
2.3. Batch Adsorption Experiments
3. Results and Discussion
3.1. Impact of Thermal Treatment on the Adsorption Capacity of Rice Husk
3.2. Kinetic Studies
3.3. Adsorption Isotherm
3.4. Thermodynamics
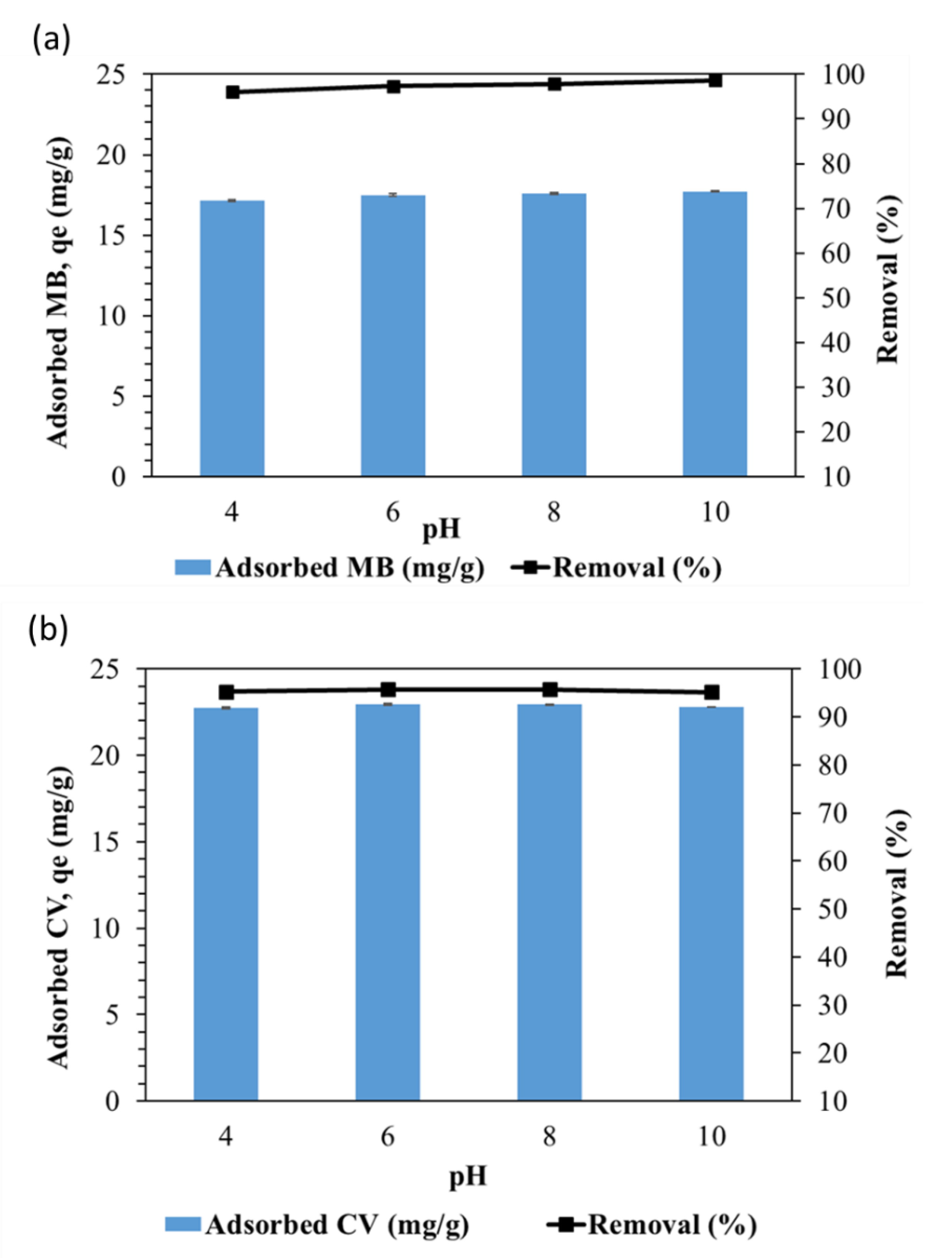
3.5. Influence of solution pH
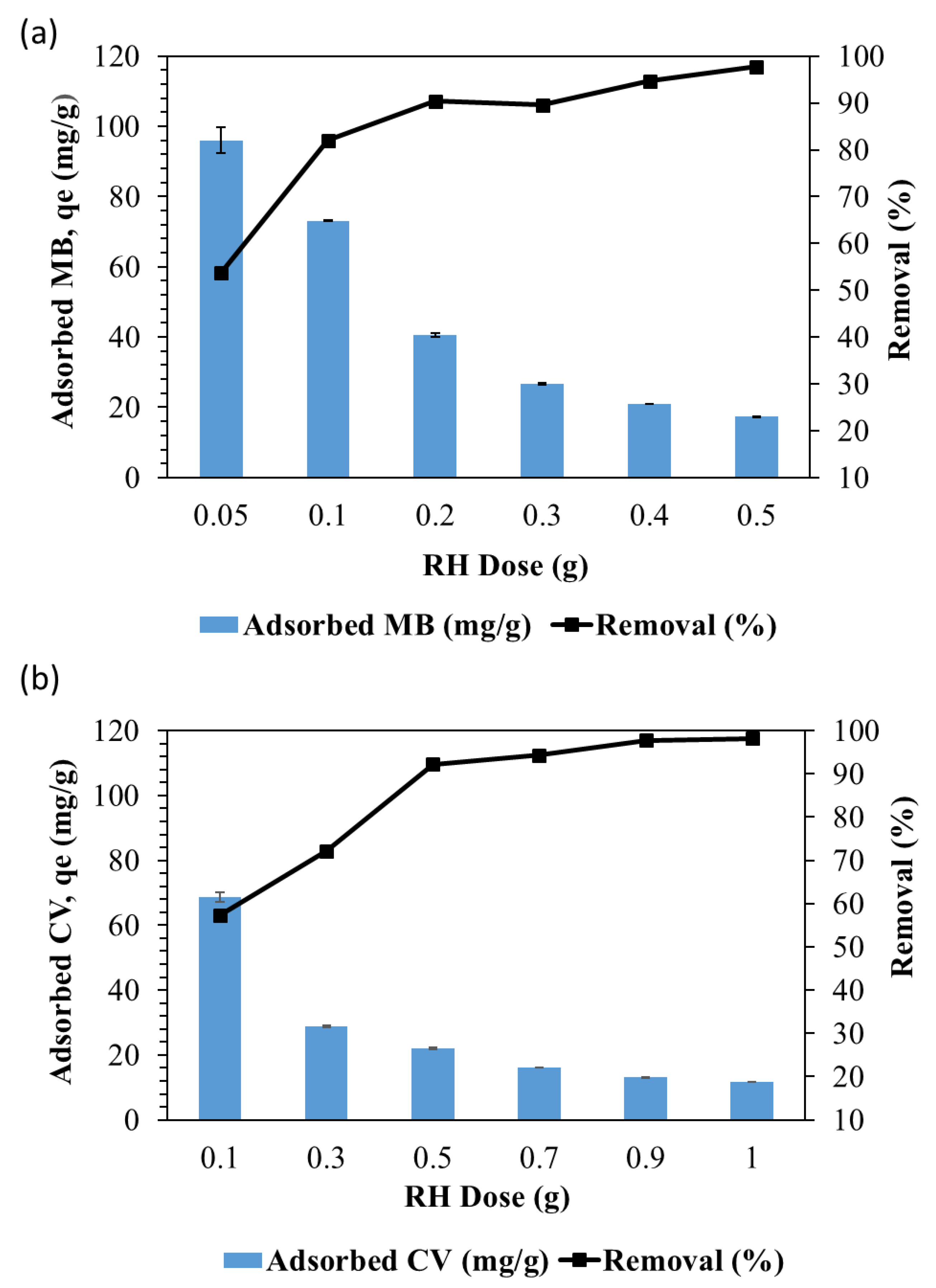
3.6. Effect of Adsorbent Dosage
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bağda, E. The feasibility of using Rosa canina galls as an effective new biosorbent for removal of methylene blue and crystal violet. Desalin. Water Treat. 2012, 43, 63–75. [Google Scholar] [CrossRef]
- Alizadeh, N.; Shariati, S.; Besharati, N. Adsorption of crystal violet and methylene blue on azolla and fig leaves modified with magnetite iron oxide nanoparticles. Int. J. Environ. Health Res. 2017, 11, 197–206. [Google Scholar] [CrossRef]
- Levin, R.L.; Degrange, M.A.; Bruno, G.F.; Del Mazo, C.D.; Taborda, D.J.; Griotti, J.J.; Boullon, F.J. Methylene blue reduces mortality and morbidity in vasoplegic patients after cardiac surgery. Ann. Thorac. Surg. 2004, 77, 496–499. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, L.; Gao, C.; Ma, J.; Ma, X.; Han, R. Adsorption of Copper Ions and Methylene Blue in a Single and Binary System on Wheat Straw. J. Chem. Eng. Data 2009, 54, 3229–3234. [Google Scholar] [CrossRef]
- Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. Adsorption of methylene blue on low-cost adsorbents: A review. J. Hazard. Mater. 2010, 177, 70–80. [Google Scholar] [CrossRef]
- Ahmad, R. Studies on adsorption of crystal violet dye from aqueous solution onto coniferous pinus bark powder (CPBP). J. Hazard. Mater. 2009, 171, 767–773. [Google Scholar] [CrossRef]
- Chen, C.C.; Liao, H.J.; Cheng, C.Y.; Yen, C.Y.; Chung, Y.C. Biodegradation of crystal violet by Pseudomonas putida. Biotechnol. Lett. 2007, 29, 391–396. [Google Scholar] [CrossRef]
- Kumar, R.; Ahmad, R. Biosorption of hazardous crystal violet dye from aqueous solution onto treated ginger waste (TGW). Desalination 2011, 265, 112–118. [Google Scholar] [CrossRef]
- Santhi, T.; Manonmani, S.; Smitha, T. Removal of malachite green from aqueous solution by activated carbon prepared from the epicarp of Ricinus communis by adsorption. J. Hazard. Mater. 2010, 179, 178–186. [Google Scholar] [CrossRef]
- Shanmugam, S.; Rajasekaran, P.; Thanikal, J.V. Synthetic dye decolourization, textile dye and paper industrial effluent treatment using white rot fungi Lentines edodes. Desalin. Water Treat. 2009, 4, 143–147. [Google Scholar] [CrossRef]
- Ciardelli, G.; Ranieri, N. The treatment and reuse of wastewater in the textile industry by means of ozonation and electroflocculation. Water Res. 2001, 35, 567–572. [Google Scholar] [CrossRef]
- Jirankova, H.; Mrazek, J.; Dolecek, P.; Cakl, J. Organic dye removal by combined adsorption—membrane separation process. Desalin. Water Treat. 2010, 20, 96–101. [Google Scholar] [CrossRef]
- Ghosh, M.; Liu, J.; Chuang, S.S.; Jana, S.C. Fabrication of hierarchical V2O5 nanorods on TiO2 nanofibers and their enhanced photocatalytic activity under visible light. Chem. Cat. Chem. 2018, 10, 3305–3318. [Google Scholar]
- Miklos, D.B.; Remy, C.; Jekel, M.; Linden, K.G.; Drewes, J.E.; Hübner, U. Evaluation of advanced oxidation processes for water and wastewater treatment–A critical review. Water Res. 2018, 139, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Kyriakopoulos, J.; Kordouli, E.; Bourikas, K.; Kordulis, C.; Lycourghiotis, A. Decolorization of Orange-G aqueous solutions over C60/MCM-41 photocatalysts. Appl. Sci. 2019, 9, 1958. [Google Scholar] [CrossRef]
- Wang, A.; Qu, J.; Liu, H.; Ge, J. Degradation of azo dye Acid Red 14 in aqueous solution by electrokinetic and electrooxidation process. Chemosphere 2004, 55, 1189–1196. [Google Scholar] [CrossRef]
- Yang, Z.; Yang, H.; Jiang, Z.; Cai, T.; Li, H.; Li, H.; Li, A.; Cheng, R. Flocculation of both anionic and cationic dyes in aqueous solutions by the amphoteric grafting flocculant carboxymethyl chitosan-graft-polyacrylamide. J. Hazard. Mater. 2013, 254, 36–45. [Google Scholar] [CrossRef]
- Liang, C.Z.; Sun, S.P.; Li, F.Y.; Ong, Y.K.; Chung, T.S. Treatment of highly concentrated wastewater containing multiple synthetic dyes by a combined process of coagulation/flocculation and nanofiltration. J. Membr. Sci. 2014, 469, 306–315. [Google Scholar] [CrossRef]
- Cheruiyot, G.K.; Wanyonyi, W.C.; Kiplimo, J.J.; Maina, E.N. Adsorption of toxic crystal violet dye using coffee husks: Equilibrium, kinetics and thermodynamics study. Sci. Afr. 2019, 5, e00116. [Google Scholar] [CrossRef]
- Al-Bastaki, N. Removal of methyl orange dye and Na2SO4 salt from synthetic waste water using reverse osmosis. Chem. Eng. Process 2004, 43, 1561–1567. [Google Scholar] [CrossRef]
- Oei, B.C.; Ibrahim, S.; Wang, S.; Ang, H.M. Surfactant modified barley straw for removal of acid and reactive dyes from aqueous solution. Bioresour. Technol. 2009, 100, 4292–4295. [Google Scholar] [CrossRef] [PubMed]
- Taştan, B.E.; Ertuğrul, S.; Dönmez, G. Effective bioremoval of reactive dye and heavy metals by Aspergillus versicolor. Bioresour. Technol. 2010, 101, 870–876. [Google Scholar] [CrossRef] [PubMed]
- Gomez, V.; Larrechi, M.; Callao, M. Kinetic and adsorption study of acid dye removal using activated carbon. Chemosphere 2007, 69, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Malik, P. Dye removal from wastewater using activated carbon developed from sawdust: Adsorption equilibrium and kinetics. J. Hazard. Mater. 2004, 113, 81–88. [Google Scholar] [CrossRef]
- Gupta, V.K.; Sharma, S. Removal of zinc from aqueous solutions using bagasse fly ash− a low cost adsorbent. Ind. Eng. Chem. Res. 2003, 42, 6619–6624. [Google Scholar] [CrossRef]
- Gupta, V. Application of low-cost adsorbents for dye removal–A review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef]
- Song, J.; Zou, W.; Bian, Y.; Su, F.; Han, R. Adsorption characteristics of methylene blue by peanut husk in batch and column modes. Desalination 2011, 265, 119–125. [Google Scholar] [CrossRef]
- Wang, Z.; Han, P.; Jiao, Y.; Ma, D.; Dou, C.; Han, R. Adsorption of congo red using ethylenediamine modified wheat straw. Desalin. Water Treat. 2011, 30, 195–206. [Google Scholar] [CrossRef]
- Franca, A.S.; Oliveira, L.S.; Saldanha, S.A.; Santos, P.I.; Salum, S.S. Malachite green adsorption by mango (Mangifera indica L.) seed husks: Kinetic, equilibrium and thermodynamic studies. Desalin. Water Treat. 2010, 19, 241–248. [Google Scholar] [CrossRef]
- Aydin, H.; Baysal, G.; Bulut, Y. Utilization of walnut shells (Juglans regia) as an adsorbent for the removal of acid dyes. Desalin. Water Treat. 2009, 2, 141–150. [Google Scholar] [CrossRef]
- Khattri, S.; Singh, M. Removal of malachite green from dye wastewater using neem sawdust by adsorption. J. Hazard. Mater. 2009, 167, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Garg, V.K.; Amita, M.; Kumar, R.; Gupta, R. Basic dye (methylene blue) removal from simulated wastewater by adsorption using Indian Rosewood sawdust: A timber industry waste. Dyes Pigm. 2004, 63, 243–250. [Google Scholar] [CrossRef]
- Mane, R.; Bhusari, V. Removal of colour (dyes) from textile effluent by adsorption using orange and banana peel. Int. J. Eng. Res. Appl. 2012, 2, 1997–2004. [Google Scholar]
- Foo, K.; Hameed, B. Preparation of activated carbon from date stones by microwave induced chemical activation: Application for methylene blue adsorption. Chem. Eng. J. 2011, 170, 338–341. [Google Scholar] [CrossRef]
- Kumar, P.S.; Ramalingam, S.; Senthamarai, C.; Niranjanaa, M.; Vijayalakshmi, P.; Sivanesan, S. Adsorption of dye from aqueous solution by cashew nut shell: Studies on equilibrium isotherm, kinetics and thermodynamics of interactions. Desalination 2010, 261, 52–60. [Google Scholar] [CrossRef]
- Juang, R.S.; Wu, F.C.; Tseng, R.L. Mechanism of adsorption of dyes and phenols from water using activated carbons prepared from plum kernels. J. Colloid Interface Sci. 2000, 227, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Ranjan, M.R. Heavy metal removal from wastewater using low cost adsorbents. J. Bioremediat. Biodegrad. 2015, 6, 1–5. [Google Scholar] [CrossRef]
- Adegoke, K.A.; Bello, O.S. Dye sequestration using agricultural wastes as adsorbents. Water Resour. Ind. 2015, 12, 8–24. [Google Scholar] [CrossRef]
- Savova, D.; Apak, E.; Ekinci, E.; Yardim, F.; Petrov, N.; Budinova, T.; Razvigorova, M.; Minkova, V. Biomass conversion to carbon adsorbents and gas. Biomass Bioenergy 2001, 21, 133–142. [Google Scholar] [CrossRef]
- Li, Y.; Ding, X.; Guo, Y.; Rong, C.; Wang, L.; Qu, Y.; Ma, X.; Wang, Z. A new method of comprehensive utilization of rice husk. J. Hazard. Mater. 2011, 186, 2151–2156. [Google Scholar] [CrossRef]
- Tong, K.T.; Vinai, R.; Soutsos, M.N. Use of Vietnamese rice husk ash for the production of sodium silicate as the activator for alkali-activated binders. J. Clean. Prod. 2018, 201, 272–286. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. Food balance sheets; FAO Communication: Rome, Italy, 2018. [Google Scholar]
- Chowdhury, S.; Saha, P.D. Artificial neural network (ANN) modeling of adsorption of methylene blue by NaOH-modified rice husk in a fixed-bed column system. Environ. Sci. Pollut. Res. 2013, 20, 1050–1058. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Chowdhury, S.; Saha, P.D. Adsorption of crystal violet from aqueous solution onto NaOH-modified rice husk. Carbohydr. Polym. 2011, 86, 1533–1541. [Google Scholar] [CrossRef]
- Chuah, T.G.; Jumasiah, A.; Azni, I.; Katayon, S.; Choong, S.T. Rice husk as a potentially low-cost biosorbent for heavy metal and dye removal: An overview. Desalination 2005, 175, 305–316. [Google Scholar] [CrossRef]
- Ahmad, W.; Yang, Z.; Khan, J.; Jing, W.; Jiang, F.; Chu, L.; Liu, N.; Li, L.; Gao, Y. Extraction of nano-silicon with activated carbons simultaneously from rice husk and their synergistic catalytic effect in counter electrodes of dye-sensitized solar cells. Sci. Rep. 2016, 6, 1–11. [Google Scholar] [CrossRef]
- Yadav, S.; Asthana, A.; Chakraborty, R.; Jain, B.; Singh, A.K.; Carabineiro, S.A.; Susan, M.; Hassan, A.B. Cationic Dye Removal Using Novel Magnetic/Activated Charcoal/β-Cyclodextrin/Alginate Polymer Nanocomposite. J. Nanomater. 2020, 10, 170. [Google Scholar] [CrossRef]
- Doble, M.; Kumar, A. Biotreatment of Industrial Effluents; Elsevier: Waltham, MA, USA, 24 February 2005. [Google Scholar]
- El-Sayed, G.O. Removal of methylene blue and crystal violet from aqueous solutions by palm kernel fiber. Desalination 2011, 272, 225–232. [Google Scholar] [CrossRef]
- Hong, S.H.; Lyonga, F.N.; Kang, J.K.; Seo, E.J.; Lee, C.G.; Jeong, S.; Hong, S.G.; Park, S.J. Synthesis of Fe-impregnated biochar from food waste for Selenium (Ⅵ)removal from aqueous solution through adsorption: Process optimization and assessment. Chemosphere 2020, 252, 126475. [Google Scholar] [CrossRef]
- Lee, J.I.; Hong, S.H.; Lee, C.G.; Park, S.J. Experimental and model study for fluoride removal by thermally activated sepiolite. Chemosphere 2020, 241, 125094. [Google Scholar] [CrossRef]
- Wang, X.S.; Lin, H.Q. Adsorption of basic dyes by dried waste sludge: Kinetic, equilibrium and desorption studies. Desalin. Water Treat. 2011, 29, 10–19. [Google Scholar] [CrossRef][Green Version]
- Della, V.P.; Kühn, I.; Hotza, D. Rice husk ash as an alternate source for active silica production. Mater. Lett. 2002, 57, 818–821. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Pramada, P. Rice husk ash as an adsorbent for methylene blue—Effect of ashing temperature. Adsorption 2006, 12, 27. [Google Scholar] [CrossRef]
- Chen, Y.; Zhu, Y.; Wang, Z.; Li, Y.; Wang, L.; Ding, L.; Gao, X.; Ma, Y.; Guo, Y. Application studies of activated carbon derived from rice husks produced by chemical-thermal process—A review. Adv. Colloid Interface Sci. 2011, 163, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Deiana, C.; Granados, D.; Venturini, R.; Amaya, A.; Sergio, M.; Tancredi, N. Activated carbons obtained from rice husk: Influence of leaching on textural parameters. Ind. Eng. Chem. Res. 2008, 47, 4754–4757. [Google Scholar] [CrossRef]
- Sakin Omer, O.; Hussein, M.A.; Hussein, B.H.M.; Mgaidi, A. Adsorption thermodynamics of cationic dyes (methylene blue and crystal violet) to a natural clay mineral from aqueous solution between 293.15 and 323.15 K. Arab. J. Chem. 2018, 11, 615–623. [Google Scholar] [CrossRef]
- Cazetta, A.L.; Vargas, A.M.M.; Nogami, E.M.; Kunita, M.H.; Guilherme, M.R.; Martins, A.C.; Silva, T.L.; Moraes, J.C.G.; Almeida, V.C. NaOH-activated carbon of high surface area produced from coconut shell: Kinetics and equilibrium studies from the methylene blue adsorption. Chem. Eng. J. 2011, 174, 117–125. [Google Scholar] [CrossRef]
- Ho, Y.S.; Mckay, G. The kinetics of sorption of basic dyes from aqueous solution by sphagnum moss peat. Can. J. Chem. Eng. 1998, 76, 822–827. [Google Scholar] [CrossRef]
- Tharaneedhar, V.; Kumar, P.S.; Saravanan, A.; Ravikumar, C.; Jaikumar, V. Prediction and interpretation of adsorption parameters for the sequestration of methylene blue dye from aqueous solution using microwave assisted corncob activated carbon. Sustain. Mater. Technol. 2017, 11, 1–11. [Google Scholar] [CrossRef]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Cheung, W.; Szeto, Y.; McKay, G. Intraparticle diffusion processes during acid dye adsorption onto chitosan. Bioresour. Technol. 2007, 98, 2897–2904. [Google Scholar] [CrossRef]
- Ramírez-Rodríguez, T.; De Landa Castillo-Alvarado, F. Application of the intra-particle diffusion model for activated carbon fibers in an aqueous medium. Mater. Res. Soc. Symp. Proc. 2012, 1373. [Google Scholar] [CrossRef]
- Wu, F.C.; Tseng, R.L.; Juang, R.S. Initial behavior of intraparticle diffusion model used in the description of adsorption kinetics. Chem. Eng. J. 2009, 153, 1–8. [Google Scholar] [CrossRef]
- Wang, X.S.; Liu, X.; Wen, L.; Zhou, Y.; Jiang, Y.; Li, Z. Comparison of Basic Dye Crystal Violet Removal from Aqueous Solution by Low-Cost Biosorbents. Sep. Sci. Technol. 2008, 43, 3712–3731. [Google Scholar] [CrossRef]
- Hu, L.; Guang, C.; Liu, Y.; Su, Z.; Gong, S.; Yao, Y.; Wang, Y. Adsorption behavior of dyes from an aqueous solution onto composite magnetic lignin adsorbent. Chemosphere 2020, 246, 125757. [Google Scholar] [CrossRef]
- Jayasantha Kumari, H.; Krishnamoorthy, P.; Arumugam, T.K.; Radhakrishnan, S.; Vasudevan, D. An efficient removal of crystal violet dye from waste water by adsorption onto TLAC/Chitosan composite: A novel low cost adsorbent. Int. J. Biol. Macromol. 2017, 96, 324–333. [Google Scholar] [CrossRef]
- Duraisamy, R.; Kiruthiga, P.; Hirpaye, B.; Berekute, A. Adsorption of azure B dye on rice husk activated carbon: Equilibrium, kinetic and thermodynamic studies. Int. J. Water Res. 2015, 5, 18–28. [Google Scholar]
- Mushtaq, M.; Bhatti, H.N.; Iqbal, M.; Noreen, S. Eriobotrya japonica seed biocomposite efficiency for copper adsorption: Isotherms, kinetics, thermodynamic and desorption studies. J. Environ. Manag. 2016, 176, 21–33. [Google Scholar] [CrossRef]
- Atmani, F.; Bensmaili, A.; Mezenner, N. Synthetic textile effluent removal by skin almonds waste. J. Environ. Sci. Technol. 2009, 2, 153–169. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Sharma, P.; Kaur, H.; Sharma, M.; Sahore, V. A review on applicability of naturally available adsorbents for the removal of hazardous dyes from aqueous waste. Environ. Monit. Assess. 2011, 183, 151–195. [Google Scholar] [CrossRef]
- Chowdhury, S.; Mishra, R.; Saha, P.; Kushwaha, P. Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically modified rice husk. Desalination 2011, 265, 159–168. [Google Scholar] [CrossRef]
- Hameed, B.H. Evaluation of papaya seeds as a novel non-conventional low-cost adsorbent for removal of methylene blue. J. Hazard. Mater. 2009, 162, 939–944. [Google Scholar] [CrossRef] [PubMed]
- Bello, O.S.; Adeogun, I.A.; Ajaelu, J.C.; Fehintola, E.O. Adsorption of methylene blue onto activated carbon derived from periwinkle shells: Kinetics and equilibrium studies. J. Chem. Ecol. 2008, 24, 285–295. [Google Scholar] [CrossRef]
- Ahmed, M.J.; Dhedan, S.K. Equilibrium isotherms and kinetics modeling of methylene blue adsorption on agricultural wastes-based activated carbons. Fluid Phase Equilib. 2012, 317, 9–14. [Google Scholar] [CrossRef]
- Liu, Q.-S.; Zheng, T.; Li, N.; Wang, P.; Abulikemu, G. Modification of bamboo-based activated carbon using microwave radiation and its effects on the adsorption of methylene blue. Appl. Surf. Sci. 2010, 256, 3309–3315. [Google Scholar] [CrossRef]
- Chang, M.-Y.; Juang, R.-S. Adsorption of tannic acid, humic acid, and dyes from water using the composite of chitosan and activated clay. J. Colloid Interface Sci. 2004, 278, 18–25. [Google Scholar] [CrossRef]
- Geçgel, Ü.; Özcan, G.; Gürpınar, G.Ç. Removal of methylene blue from aqueous solution by activated carbon prepared from pea shells (Pisum sativum). J. Chem. 2012, 2013, 614083. [Google Scholar] [CrossRef]
- Boumehdi Toumi, L.; Hamdi, L.; Salem, Z.; Allia, K. Batch adsorption of methylene blue from aqueous solutions by untreated Alfa grass. Desalin. Water Treat. 2015, 53, 806–817. [Google Scholar] [CrossRef]
- Hameed, B.H.; El-Khaiary, M.I. Sorption kinetics and isotherm studies of a cationic dye using agricultural waste: Broad bean peels. J. Hazard. Mater. 2008, 154, 639–648. [Google Scholar] [CrossRef]
- Hameed, B.; Ahmad, A. Batch adsorption of methylene blue from aqueous solution by garlic peel, an agricultural waste biomass. J. Hazard. Mater. 2009, 164, 870–875. [Google Scholar] [CrossRef]
- Han, X.; Niu, X.; Ma, X. Adsorption characteristics of methylene blue on poplar leaf in batch mode: Equilibrium, kinetics and thermodynamics. Korean J. Chem. Eng. 2012, 29, 494–502. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Ang, H. Equilibrium, kinetics, and thermodynamics of methylene blue adsorption by pine tree leaves. Water Air Soil Pollut. 2012, 223, 5267–5282. [Google Scholar] [CrossRef]
- Auta, M.; Hameed, B.H. Modified mesoporous clay adsorbent for adsorption isotherm and kinetics of methylene blue. Chem. Eng. J. 2012, 198, 219–227. [Google Scholar] [CrossRef]
- Wang, X.S.; Zhou, Y.; Jiang, Y.; Sun, C. The removal of basic dyes from aqueous solutions using agricultural by-products. J. Hazard. Mater. 2008, 157, 374–385. [Google Scholar] [CrossRef]
- Ertaş, M.; Acemioğlu, B.; Alma, M.H.; Usta, M. Removal of methylene blue from aqueous solution using cotton stalk, cotton waste and cotton dust. J. Hazard. Mater. 2010, 183, 421–427. [Google Scholar] [CrossRef]
- Monárrez-Cordero, B.E.; Sáenz-Trevizo, A.; Bautista-Carrillo, L.M.; Silva-Vidaurri, L.G.; Miki-Yoshida, M.; Amézaga-Madrid, P. Simultaneous and fast removal of As3+, As5+, Cd2+, Cu2+, Pb2+ and F− from water with composite Fe-Ti oxides nanoparticles. J. Alloys Compd. 2018, 757, 150–160. [Google Scholar] [CrossRef]
- Navarathna, C.M.; Karunanayake, A.G.; Gunatilake, S.R.; Pittman, C.U.; Perez, F.; Mohan, D.; Mlsna, T. Removal of Arsenic(III) from water using magnetite precipitated onto Douglas fir biochar. J. Environ. Manag. 2019, 250, 109429. [Google Scholar] [CrossRef]
- Hong, S.; Wen, C.; He, J.; Gan, F.; Ho, Y.-S. Adsorption thermodynamics of methylene blue onto bentonite. J. Hazard. Mater. 2009, 167, 630–633. [Google Scholar] [CrossRef]
- Nandi, B.; Goswami, A.; Purkait, M. Removal of cationic dyes from aqueous solutions by kaolin: Kinetic and equilibrium studies. Appl. Clay Sci. 2009, 42, 583–590. [Google Scholar] [CrossRef]
- Deng, S.; Niu, L.; Bei, Y.; Wang, B.; Huang, J.; Yu, G. Adsorption of perfluorinated compounds on aminated rice husk prepared by atom transfer radical polymerization. Chemosphere 2013, 91, 124–130. [Google Scholar] [CrossRef]
- Shittu, I.; Edathil, A.A.; Alsaeedi, A.; Al-Asheh, S.; Polychronopoulou, K.; Banat, F. Development of novel surfactant functionalized porous graphitic carbon as an efficient adsorbent for the removal of methylene blue dye from aqueous solutions. J. Water Process. Eng. 2019, 28, 69–81. [Google Scholar] [CrossRef]


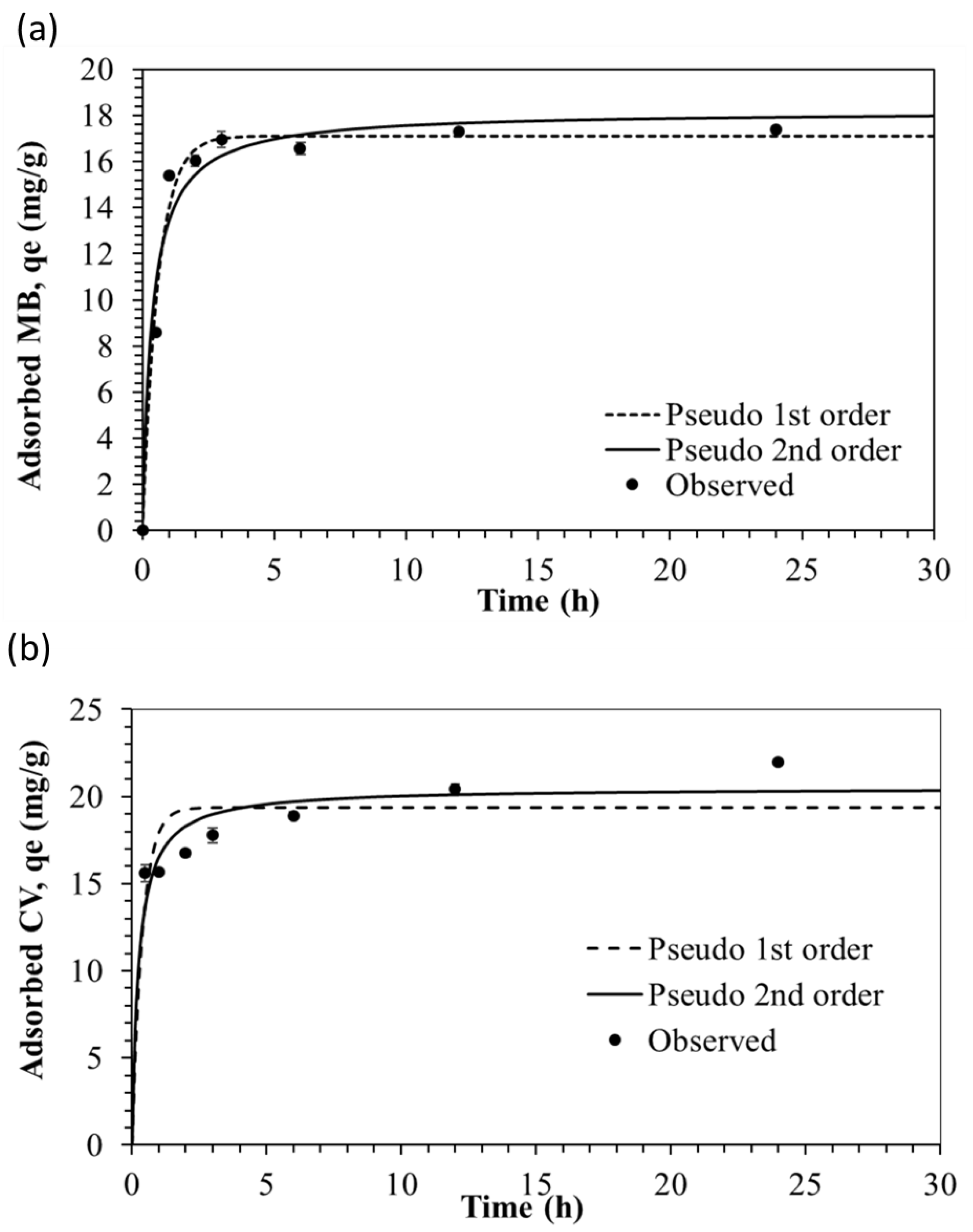
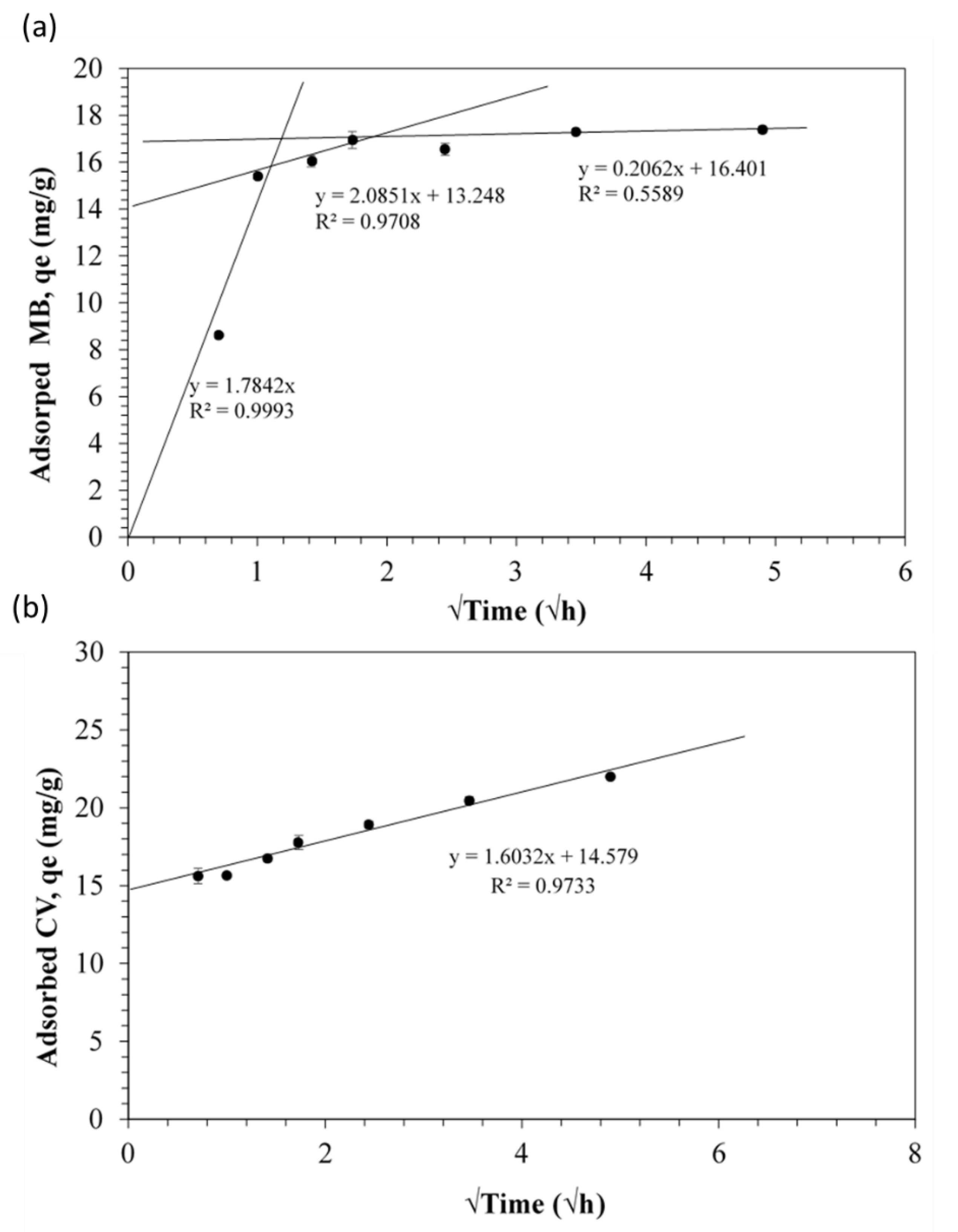

| Adsorbate | Pseudo-First-Order Kinetics | Pseudo-Second-Order Kinetics | ||||
|---|---|---|---|---|---|---|
| qe (mg/g) | k1 (1/h) | R2 | qe (mg/g) | k2 (g/mg/h) | R2 | |
| MB | 17.1038 | 1.6835 | 0.9848 | 18.1906 | 0.1537 | 0.9641 |
| CV | 18.9546 | 2.8671 | 0.9185 | 20.2015 | 0.2275 | 0.9649 |
| Adsorbate | Langmuir Isotherm | Freundlich Isotherm | ||||
|---|---|---|---|---|---|---|
| Qm (mg/g) | KL (L/mg) | R2 | KF | 1/n | R2 | |
| MB | 25.4645 | 0.3068 | 0.9868 | 7.3348 | 0.3155 | 0.9062 |
| CV | 24.4781 | 0.2493 | 0.9785 | 7.4143 | 0.2773 | 0.9107 |
| Adsorbent | Adsorption Capacity (mg/g) | Particle Size | pH | Temperature (°C) | Reference |
|---|---|---|---|---|---|
| Papaya seeds | 555.56 | 125−250 µm | 4.00 | 30 | [74] |
| Periwinkle shells | 500 | 150 μm | 7.00 | 25 | [75] |
| Date stones | 398.19 | 250 μm | 7.00 | 30 | [76] |
| Bamboo activated carbon | 286 | 1–2 mm | 3.70 | 25 | [77] |
| Chitosan beads | 262 | 4.42 mm | NA | 30 | [78] |
| Pea shells | 246.91 | 212 µm | NA | 25 | [79] |
| Alfa grass | 200 | 500–650 µm | 12.0 | 20 | [80] |
| Broad bean peels | 192.7 | 350−400 μm | 5.00 | 30 | [81] |
| Garlic peels | 142.86 | NA | 6.00 | 50 | [82] |
| Natural poplar leaf | 135.35 | 0.425−0.850 mm | 7.00 | 20 | [83] |
| Pine-tree leaf biomass | 126.58 | <350 μm | 9.20 | 30 | [84] |
| Modified clay-ball | 100 | 1−2 mm | NA | 50 | [85] |
| Palm kernel fiber | 95.4 | 50–80 μm | 7.20 | 25 | [49] |
| Peanut husk | 72.13 | 0.425−0.85 mm | 7.00 | 20 | [27] |
| Wheat bran | 54.79 | 150−250 µm | 6.00 | 20 | [86] |
| Rice husk | 25.46 | 0.075−1.16 mm | 7.00 | 25 | This study |
| Rice bran | 20.29 | 150−250 µm | 6.00 | 20 | [86] |
| Adsorbate | Temperature (K) | ∆H° (kJ/mol) | ∆S° (J/mol K) | ∆G° (kJ/mol) |
|---|---|---|---|---|
| MB | 288 | 28.50 | 0.103 | −1.1115 |
| 298 | −1.9774 | |||
| 308 | −3.1703 | |||
| CV | 288 | 151.53 | 0.535 | −2.7018 |
| 298 | −7.6901 | |||
| 308 | −13.4146 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quansah, J.O.; Hlaing, T.; Lyonga, F.N.; Kyi, P.P.; Hong, S.-H.; Lee, C.-G.; Park, S.-J. Nascent Rice Husk as an Adsorbent for Removing Cationic Dyes from Textile Wastewater. Appl. Sci. 2020, 10, 3437. https://doi.org/10.3390/app10103437
Quansah JO, Hlaing T, Lyonga FN, Kyi PP, Hong S-H, Lee C-G, Park S-J. Nascent Rice Husk as an Adsorbent for Removing Cationic Dyes from Textile Wastewater. Applied Sciences. 2020; 10(10):3437. https://doi.org/10.3390/app10103437
Chicago/Turabian StyleQuansah, Jude Ofei, Thandar Hlaing, Fritz Ndumbe Lyonga, Phyo Phyo Kyi, Seung-Hee Hong, Chang-Gu Lee, and Seong-Jik Park. 2020. "Nascent Rice Husk as an Adsorbent for Removing Cationic Dyes from Textile Wastewater" Applied Sciences 10, no. 10: 3437. https://doi.org/10.3390/app10103437
APA StyleQuansah, J. O., Hlaing, T., Lyonga, F. N., Kyi, P. P., Hong, S.-H., Lee, C.-G., & Park, S.-J. (2020). Nascent Rice Husk as an Adsorbent for Removing Cationic Dyes from Textile Wastewater. Applied Sciences, 10(10), 3437. https://doi.org/10.3390/app10103437








