Ovariectomy Impairs Socio-Cognitive Functions in Dogs
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Experimental Procedure
2.3. Data Analysis
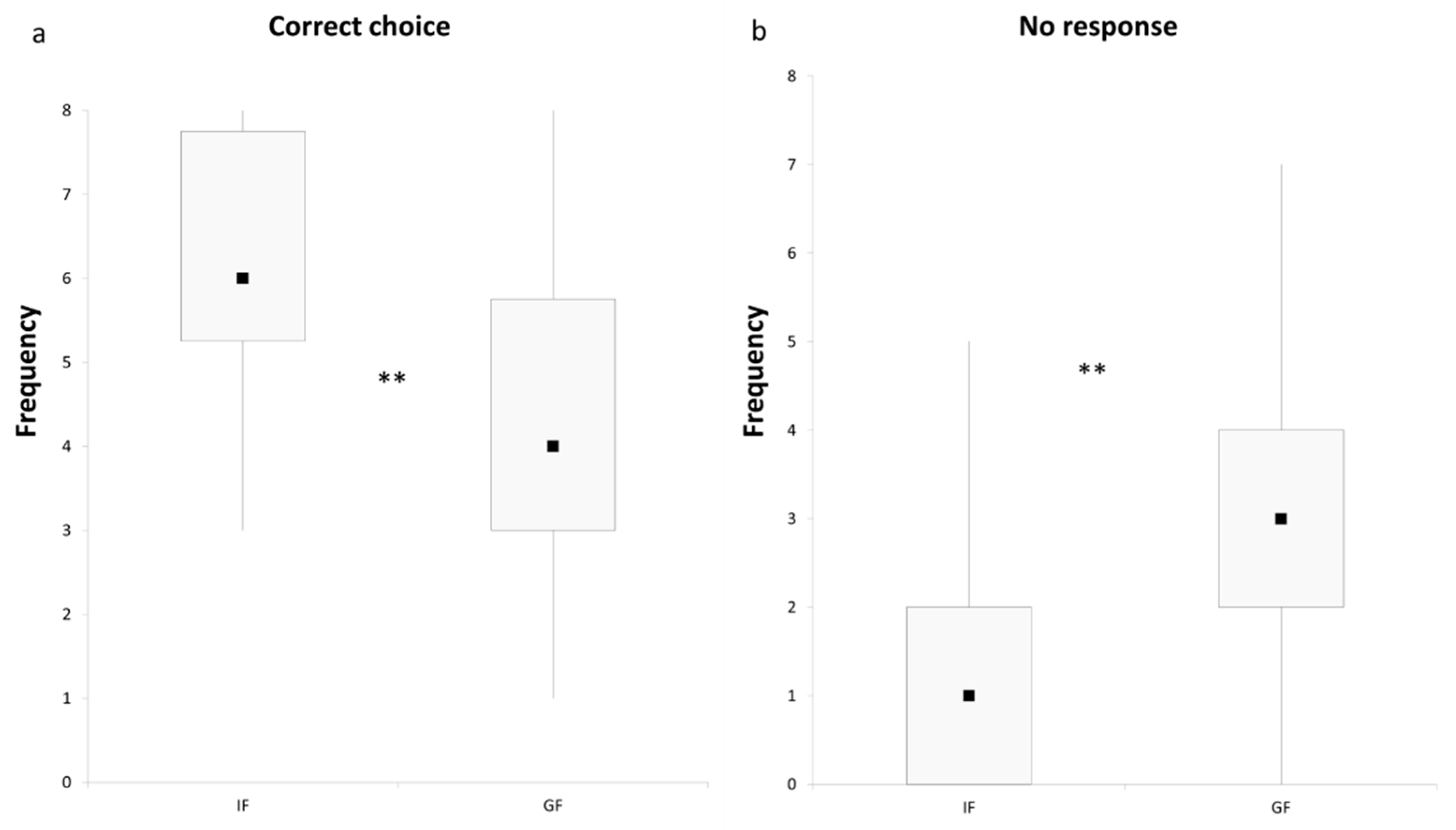
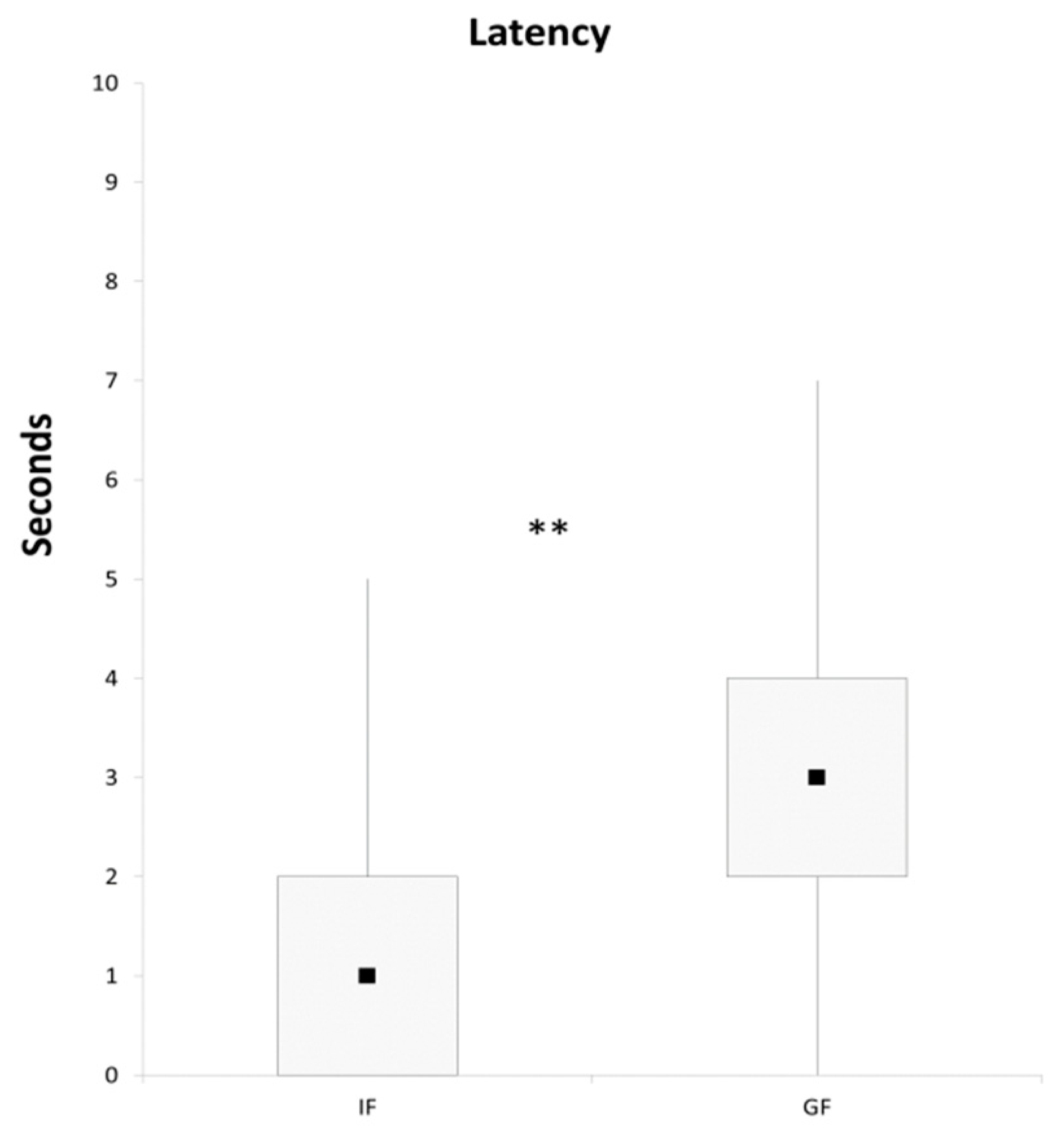
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Frick, K.M.; Kim, J.; Koss, W.A. Estradiol and hippocampal memory in female and male rodents. Curr. Opin. Behav. Sci. 2018, 23, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Scandurra, A.; Marinelli, L.; Lõoke, M.; D’Aniello, B.; Mongillo, P. The effect of age, sex and gonadectomy on dogs’ use of spatial navigation strategies. Appl. Anim. Behav. Sci. 2018, 205, 89–97. [Google Scholar] [CrossRef]
- Mongillo, P.; Scandurra, A.; D’Aniello, B.; Marinelli, L. Effect of sex and gonadectomy on dogs’ spatial performance. Appl. Anim. Behav. Sci. 2017, 191, 84–89. [Google Scholar] [CrossRef]
- Mongillo, P.; Pitteri, E.; Candaten, M.; Marinelli, L. Can attention be taught? Interspecific attention by dogs (Canis familiaris) performing obedience tasks. Appl. Anim. Behav. Sci. 2016, 182, 30–37. [Google Scholar] [CrossRef]
- D’Aniello, B.; Scandurra, A.; Alterisio, A.; Valsecchi, P.; Prato-Previde, E. The importance of gestural communication: A study of human-dog communication using incongruent information. Anim. Cogn. 2016, 19, 1231–1235. [Google Scholar] [CrossRef]
- Scandurra, A.; Alterisio, A.; Marinelli, L.; Mongillo, P.; Semin, G.R.; D’Aniello, B. Effectiveness of verbal and gestural signals and familiarity with signal-senders on the performance of working dogs. Appl. Anim. Behav. Sci. 2017, 191, 78–83. [Google Scholar] [CrossRef]
- Scandurra, A.; Alterisio, A.; Aria, M.; Vernese, R.; D’Aniello, B. Should I fetch one or the other? A study on dogs on the object choice in the bimodal contrasting paradigm. Anim. Cogn. 2018, 21, 119–126. [Google Scholar] [CrossRef]
- Hare, B.; Brown, M.; Williamson, C.; Tomasello, M. The domestication of social cognition in dogs. Science 2002, 298, 1634–1636. [Google Scholar] [CrossRef]
- Wynne, C.D.L.; Udell, M.A.R.; Lord, K.A. Ontogeny’s impact on human-dog communication. Anim. Behav. 2008, 76, e1–e4. [Google Scholar] [CrossRef]
- D’Aniello, B.; Alterisio, A.; Scandurra, A.; Petremolo, E.; Iommelli, M.R.; Aria, M. What’s the point? Golden and Labrador retrievers living in kennels do not understand human pointing gestures. Anim. Cogn. 2017, 20, 777–787. [Google Scholar] [CrossRef]
- D’Aniello, B.; Scandurra, A. Ontogenetic effects on gazing behaviour: A case study of kennel dogs (Labrador Retrievers) in the impossible task paradigm. Anim. Cogn. 2016, 19, 565–570. [Google Scholar] [CrossRef] [PubMed]
- Svartberg, K. Breed-typical behaviour in dogs—Historical remnants or recent constructs? Appl. Anim. Behav. Sci. 2006, 96, 293–313. [Google Scholar] [CrossRef]
- Starling, M.J.; Branson, N.; Thomson, P.C.; McGreevy, P.D. Age, sex and reproductive status affect boldness in dogs. Vet. J. 2013, 197, 868–872. [Google Scholar] [CrossRef] [PubMed]
- Arden, R.; Adams, M.J. A general intelligence factor in dogs. Intelligence 2016, 55, 79–85. [Google Scholar] [CrossRef]
- Beatty, W.W. Gonadal hormones and sex differences in non-reproductive behaviors in rodents: Organizational and activational influences. Horm. Behav. 1979, 12, 112–163. [Google Scholar] [CrossRef]
- Nene, A.S.; Pazare, P.A. A study of auditory reaction time in different phases of the normal menstrual cycle. Indian J. Physiol. Pharmacol. 2010, 54, 386–390. [Google Scholar]
- Niruba, R.; Maruthy, K.N. Assessment of auditory and visual reaction time in type 2 diabetics-A case control study. Al Ameen J. Med. Sci. 2011, 4, 274–279. [Google Scholar]
- Nikam, L.H.; Gadkari, J.V. Effect of age, gender and body mass index on visual and auditory reaction times in Indian population. Indian J. Physiol. Pharmacol. 2012, 56, 94–99. [Google Scholar]
- Frank, L.A.; Rohrbach, B.W.; Bailey, E.M.; West, J.R.; Oliver, J.W. Steroid hormone concentration profiles in healthy intact and neutered dogs before and after cosyntropin administration. Domest. Anim. Endocrinol. 2003, 24, 43–57. [Google Scholar] [CrossRef]
- Martin, R.B.; Butcher, R.L.; Sherwood, L.L.; Buckendahl, P.; Boyd, R.D.; Farris, D.; Sharkey, N.; Dannucci, G. Effects of ovariectomy in beagle dogs. Bone 1987, 8, 23–31. [Google Scholar] [CrossRef]
- Miller, W.L. Steroidogenesis: Unanswered questions. Trends Endocrinol. Metabol. 2017, 28, 771–793. [Google Scholar] [CrossRef] [PubMed]
- De Lisa, E.; Paolucci, M.; Di Cosmo, A. Conservative nature of oestradiol signalling pathways in the brain lobes of Octopus vulgaris involved in reproduction, learning and motor coordination. J. Neuroendocrinol. 2011, 24, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Caruso, D.; Pesaresi, M.; Maschi, O.; Giatti, S.; Garcia-Segura, L.M.; Melcandi, R.C. Effect of short-and long-term gonadectomy on neuroactive steroid levels in the central and peripheral nervous system of male and female rats. J. Neuroendocrinol. 2010, 22, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Kato, A.; Hojo, Y.; Higo, S.; Komatsuzaki, Y.; Murakami, G.; Yoshino, H.; Uebayashi, M.; Kawato, S. Female hippocampal estrogens have a significant correlation with cyclic fluctuation of hippocampal spines. Front. Neural Circuits 2013, 7, 149. [Google Scholar] [CrossRef] [PubMed]
- Luine, V. Neuroendocrinology of memory and cognition. In Handbook of Neurochemistry and Molecular Neurobiology; Behavioral Neurochemistry and Neuroendocrinology; Lajtha, A., Jeffrey, D.B., Eds.; Springer: Berlin, Germany, 2006; pp. 775–800. [Google Scholar]
- Fazio, E.; Medica, P.; Cravana, C.; Pupillo, A.; Ferlazzo, A. Effects of ovariohysterectomy in dogs and cats on adrenocortical, haematological and behavioural parameters. Acta Sci. Vet. 2015, 43, 1339. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scandurra, A.; Alterisio, A.; Di Cosmo, A.; D’Ambrosio, A.; D’Aniello, B. Ovariectomy Impairs Socio-Cognitive Functions in Dogs. Animals 2019, 9, 58. https://doi.org/10.3390/ani9020058
Scandurra A, Alterisio A, Di Cosmo A, D’Ambrosio A, D’Aniello B. Ovariectomy Impairs Socio-Cognitive Functions in Dogs. Animals. 2019; 9(2):58. https://doi.org/10.3390/ani9020058
Chicago/Turabian StyleScandurra, Anna, Alessandra Alterisio, Anna Di Cosmo, Antonio D’Ambrosio, and Biagio D’Aniello. 2019. "Ovariectomy Impairs Socio-Cognitive Functions in Dogs" Animals 9, no. 2: 58. https://doi.org/10.3390/ani9020058
APA StyleScandurra, A., Alterisio, A., Di Cosmo, A., D’Ambrosio, A., & D’Aniello, B. (2019). Ovariectomy Impairs Socio-Cognitive Functions in Dogs. Animals, 9(2), 58. https://doi.org/10.3390/ani9020058






