Effects of Dietary Energy on Growth Performance, Rumen Fermentation and Bacterial Community, and Meat Quality of Holstein-Friesians Bulls Slaughtered at Different Ages
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal and Experimental Design
2.2. Samples Collection
2.3. Chemical Analysis
2.4. Rumen Fermentation Parameters
2.5. Bacteria DNA Extraction and 16S rRNA Pyrosequencing
2.6. Meat Quality
2.7. Statistical Analysis
3. Results
3.1. Growth Performance
3.2. Rumen Fermentation Parameters
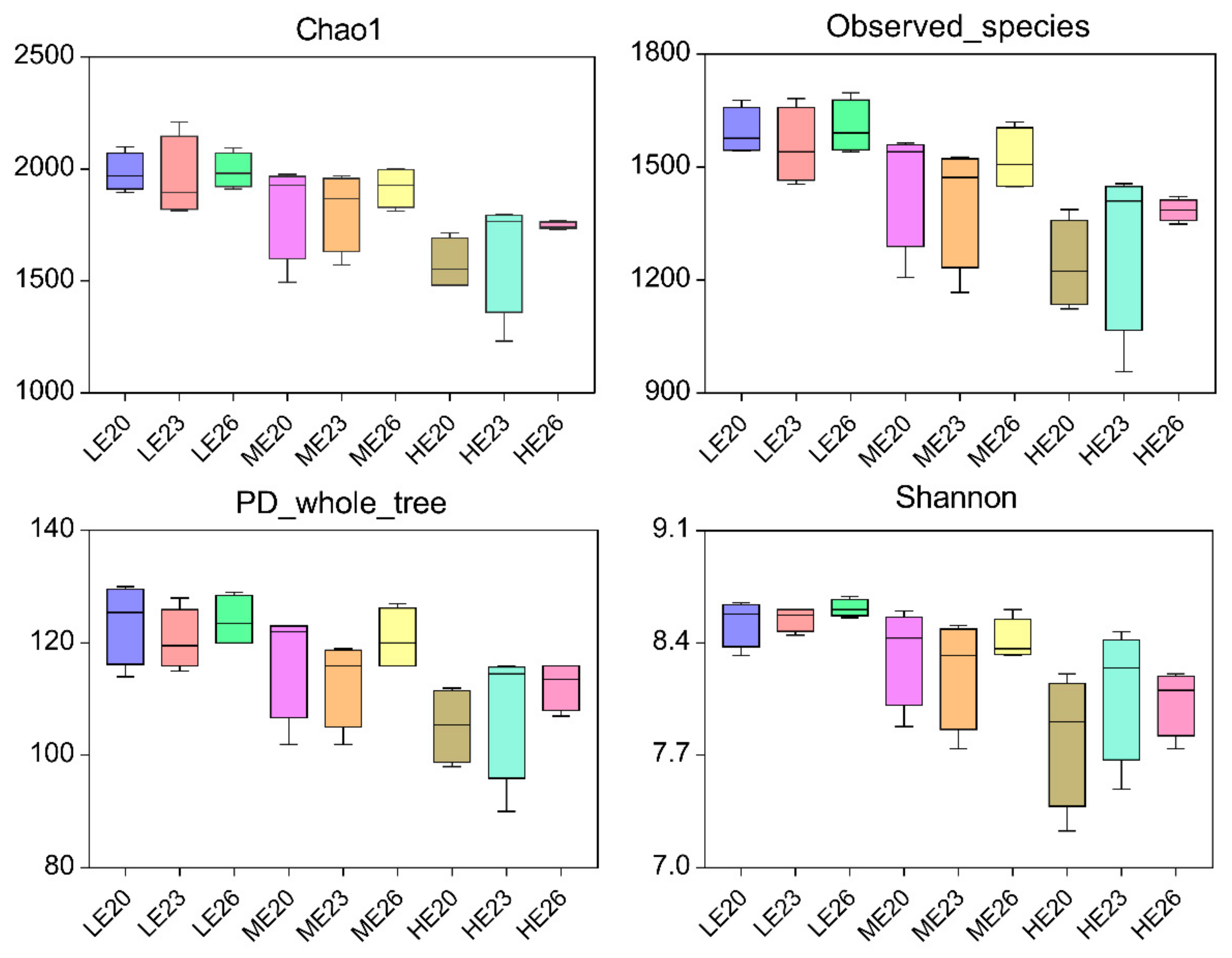
3.3. Sequencing Depth, Richness and Diversity
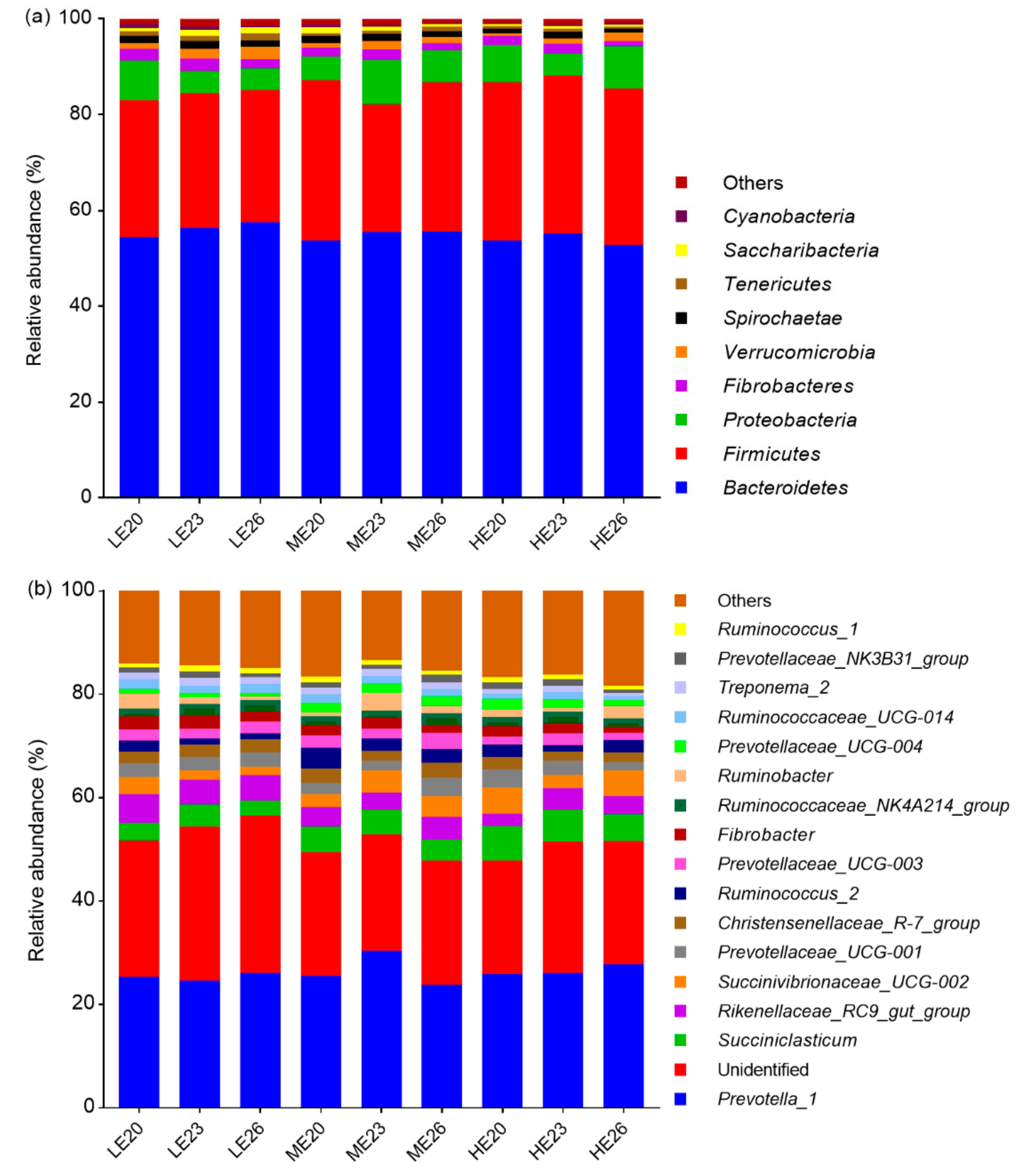
3.4. Taxonomic Profiles
3.5. Meat Quality
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Robbins, K.; Jensen, J.; Ryan, K.J.; Homco-Ryan, C.; McKeith, F.K.; Brewer, M.S. Consumer attitudes towards beef and acceptability of enhanced beef. Meat Sci. 2003, 65, 721–729. [Google Scholar] [CrossRef]
- Miller, M.F.; Carr, M.A.; Ramsey, C.B.; Crockett, K.L.; Hoover, L.C. Consumer thresholds for establishing the value of beef tenderness. J. Anim. Sci. 2001, 79, 3062–3068. [Google Scholar] [CrossRef] [PubMed]
- Platter, W.J.; Tatum, J.D.; Belk, K.E.; Koontz, S.R.; Chapman, P.L.; Smith, G.C. Effects of marbling and shear force on consumers’ willingness to pay for beef strip loin steaks. J. Anim. Sci. 2005, 83, 890–899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Emerson, M.R.; Woerner, D.R.; Belk, K.E.; Tatum, J.D. Effectiveness of USDA instrument-based marbling measurements for categorizing beef carcasses according to differences in longissimus muscle sensory attributes. J. Anim. Sci. 2013, 91, 1024–1034. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mezgebo, G.B.; Monahan, F.J.; McGee, M.; O’Riordan, E.G.; Richardson, I.R.; Brunton, N.P.; Moloney, A.P. Fatty acid, volatile and sensory characteristics of beef as affected by grass silage or pasture in the bovine diet. Food Chem. 2017, 235, 86–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunt, M.R.; Legako, J.F.; Dinh, T.T.; Garmyn, A.J.; O’Quinn, T.G.; Corbin, C.H.; Rathmann, R.J.; Brooks, J.C.; Miller, M.F. Assessment of volatile compounds, neutral and polar lipid fatty acids of four beef muscles from USDA Choice and Select graded carcasses and their relationships with consumer palatability scores and intramuscular fat content. Meat Sci. 2016, 116, 91–101. [Google Scholar] [CrossRef]
- Park, S.J.; Beak, S.H.; Jung, D.J.S.; Kim, S.Y.; Jeong, I.H.; Piao, M.Y.; Kang, H.J.; Fassah, D.M.; Na, S.W.; Yoo, S.P.; et al. Genetic, management, and nutritional factors affecting intramuscular fat deposition in beef cattle—A review. Asian-Australas. J. Anim. Sci. 2018, 31, 1043–1061. [Google Scholar] [CrossRef] [Green Version]
- Warren, H.E.; Scollan, N.D.; Nute, G.R.; Hughes, S.I.; Wood, J.D.; Richardson, R.I. Effects of breed and a concentrate or grass silage diet on beef quality in cattle of 3 ages. II: Meat stability and flavour. Meat Sci. 2008, 78, 270–278. [Google Scholar] [CrossRef]
- Nousiainen, J.; Rinne, M.; Huhtanen, P. A meta-analysis of feed digestion in dairy cows. 1. The effects of forage and concentrate factors on total diet digestibility. J. Dairy Sci. 2009, 92, 5019–5030. [Google Scholar] [CrossRef] [Green Version]
- Muir, P.; Deaker, J.; Bown, M. Effects of forage- and grain- based feeding systems on beef quality: A review. N. Z. J. Agric. Res. 1998, 41, 623–635. [Google Scholar] [CrossRef]
- Nogalski, Z.; Wielgosz-Groth, Z.; Purwin, C.; Nogalska, A.; Sobczuk-Szul, M.; Winarski, R.; Pogorzelska, P. The effect of slaughter weight and fattening intensity on changes in carcass fatness in young Holstein-Friesian bulls. Ital. J. Anim. Sci. 2016, 13, 2824. [Google Scholar] [CrossRef] [Green Version]
- Choi, N.J.; Enser, M.; Wood, J.D.; Scollan, N.D. Effect of breed on the deposition in beef muscle and adipose tissue of dietary n-3 polyunsaturated fatty acids. Anim. Sci. 2000, 71, 509–519. [Google Scholar] [CrossRef]
- Kirkland, R.M.; Patterson, D.C.; Keady, T.W.; Moss, B.W.; Steen, R.W. Beef production potential of Norwegian Red and Holstein-Friesian bulls slaughtered at two ages. Animal 2007, 1, 1506–1514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- NRC. Nutrient Requirements of Beef Cattle, 8th ed.; The National Academies Press: Washington, DC, USA, 2016. [Google Scholar]
- AOAC. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 1990. [Google Scholar]
- Van Soest, P.v.; Robertson, J.; Lewis, B. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597. [Google Scholar] [CrossRef]
- Wang, H.; He, Y.; Li, H.; Wu, F.; Qiu, Q.; Niu, W.; Gao, Z.; Su, H.; Cao, B. Rumen fermentation, intramuscular fat fatty acid profiles and related rumen bacterial populations of Holstein bulls fed diets with different energy levels. Appl. Microbiol. Biotechnol. 2019, 103, 4931–4942. [Google Scholar] [CrossRef]
- Weatherburn, M. Phenol-hypochlorite reaction for determination of ammonia. Anal. Chem. 1967, 39, 971–974. [Google Scholar] [CrossRef]
- Arelovich, H.M.; Abney, C.S.; Vizcarra, J.A.; Galyean, M.L. Effects of dietary neutral detergent fiber on intakes of dry matter and net energy by dairy and beef cattle: Analysis of published data. Prof. Anim. Sci. 2008, 24, 375–383. [Google Scholar] [CrossRef]
- Arthur, P.F.; Archer, J.A.; Johnston, D.J.; Herd, R.M.; Richardson, E.C.; Parnell, P.F. Genetic and phenotypic variance and covariance components for feed intake, feed efficiency, and other postweaning traits in Angus cattle. J. Anim. Sci. 2001, 79, 2805–2811. [Google Scholar] [CrossRef] [Green Version]
- Jerez-Timaure, N.; Huerta-Leidenz, N. Effects of breed type and supplementation during grazing on carcass traits and meat quality of bulls fattened on improved savannah. Livest. Sci. 2009, 121, 219–226. [Google Scholar] [CrossRef]
- Allen, M.S. Relationship between fermentation acid production in the rumen and the requirement for physically effective fiber. J. Dairy Sci. 1997, 80, 1447–1462. [Google Scholar] [CrossRef]
- Hoover, W.; Stokes, S. Balancing carbohydrates and proteins for optimum rumen microbial yield. J. Dairy Sci. 1991, 74, 3630–3644. [Google Scholar] [CrossRef]
- Clark, J.; Klusmeyer, T.; Cameron, M. Microbial protein synthesis and flows of nitrogen fractions to the duodenum of dairy cows. J. Dairy Sci. 1992, 75, 2304–2323. [Google Scholar] [CrossRef]
- Dijkstra, J. Production and absorption of volatile fatty acids in the rumen. Livest. Prod. Sci. 1994, 39, 61–69. [Google Scholar] [CrossRef]
- Bugaut, M. Occurrence, absorption and metabolism of short chain fatty acids in the digestive tract of mammals. Comp. Biochem. Physiol. B. 1987, 86, 439–472. [Google Scholar] [CrossRef]
- Shabat, S.K.B.; Sasson, G.; Doron-Faigenboim, A.; Durman, T.; Yaacoby, S.; Miller, M.E.B.; White, B.A.; Shterzer, N.; Mizrahi, I. Specific microbiome-dependent mechanisms underlie the energy harvest efficiency of ruminants. ISME J. 2016, 10, 2958. [Google Scholar] [CrossRef] [Green Version]
- Petri, R.M.; Forster, R.J.; Yang, W.; McKinnon, J.J.; McAllister, T.A. Characterization of rumen bacterial diversity and fermentation parameters in concentrate fed cattle with and without forage. J. Appl. Microbiol. 2012, 112, 1152–1162. [Google Scholar] [CrossRef]
- Pan, X.; Xue, F.; Nan, X.; Tang, Z.; Wang, K.; Beckers, Y.; Jiang, L.; Xiong, B. Illumina sequencing approach to characterize thiamine metabolism related bacteria and the impacts of thiamine supplementation on ruminal microbiota in dairy cows fed high-grain diets. Front. Microbiol. 2017, 8, 1818. [Google Scholar] [CrossRef]
- Wetzels, S.; Mann, E.; Metzler-Zebeli, B.; Wagner, M.; Klevenhusen, F.; Zebeli, Q.; Schmitz-Esser, S. Pyrosequencing reveals shifts in the bacterial epimural community relative to dietary concentrate amount in goats. J. Dairy Sci. 2015, 98, 5572–5587. [Google Scholar] [CrossRef] [Green Version]
- Opdahl, L.J.; Gonda, M.G.; St-Pierre, B. Identification of uncultured bacterial species from Firmicutes, Bacteroidetes and CANDIDATUS Saccharibacteria as candidate cellulose utilizers from the rumen of beef cows. Microorganisms 2018, 6, 17. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.H.; Chen, Z.D.; Zhou, S.; Song, X.Z.; Ouyang, K.H.; Pan, K.; Xu, L.J.; Liu, C.J.; Qu, M.R. Effects of daidzein on performance, serum metabolites, nutrient digestibility, and fecal bacterial community in bull calves. Anim. Feed Sci. Technol. 2017, 225, 87–96. [Google Scholar] [CrossRef]
- McGovern, E.; Kenny, D.A.; McCabe, M.S.; Fitzsimons, C.; McGee, M.; Kelly, A.K.; Waters, S.M. 16S rRNA sequencing reveals relationship between potent cellulolytic genera and feed efficiency in the rumen of bulls. Front. Microbiol. 2018, 9, 1842. [Google Scholar] [CrossRef] [PubMed]
- Carberry, C.A.; Kenny, D.A.; Han, S.; McCabe, M.S.; Waters, S.M. Effect of phenotypic residual feed intake and dietary forage content on the rumen microbial community of beef cattle. Appl. Environ. Microbiol. 2012, 78, 4949–4958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Gylswyk, N. Succiniclasticum ruminis gen. nov., sp. nov., a ruminal bacterium converting succinate to propionate as the sole energy-yielding mechanism. Int. J. Syst. Evolut. Microbiol. 1995, 45, 297–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pitta, D.W.; Pinchak, W.E.; Indugu, N.; Vecchiarelli, B.; Sinha, R.; Fulford, J.D. Metagenomic analysis of the rumen microbiome of steers with wheat-induced frothy bloat. Front. Microbiol. 2016, 7, 689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagaraja, T.; Titgemeyer, E. Ruminal acidosis in beef cattle: The current microbiological and nutritional outlook. J. Dairy Sci. 2007, 90, E17–E38. [Google Scholar] [CrossRef] [Green Version]
- Fernando, S.C.; Purvis, H.T., 2nd; Najar, F.Z.; Sukharnikov, L.O.; Krehbiel, C.R.; Nagaraja, T.G.; Roe, B.A.; Desilva, U. Rumen microbial population dynamics during adaptation to a high-grain diet. Appl. Environ. Microbiol. 2010, 76, 7482–7490. [Google Scholar] [CrossRef] [Green Version]
- Krause, D.O.; Denman, S.E.; Mackie, R.I.; Morrison, M.; Rae, A.L.; Attwood, G.T.; McSweeney, C.S. Opportunities to improve fiber degradation in the rumen: Microbiology, ecology, and genomics. FEMS Microbiol. Rev. 2003, 27, 663–693. [Google Scholar] [CrossRef] [Green Version]
- Zened, A.; Combes, S.; Cauquil, L.; Mariette, J.; Klopp, C.; Bouchez, O.; Troegeler-Meynadier, A.; Enjalbert, F. Microbial ecology of the rumen evaluated by 454 GS FLX pyrosequencing is affected by starch and oil supplementation of diets. FEMS Microbiol. Ecol. 2013, 83, 504–514. [Google Scholar] [CrossRef] [Green Version]
- Lawrence, G.; Buchin, S.; Achilleos, C.; Berodier, F.; Septier, C.; Courcoux, P.; Salles, C. In vivo sodium release and saltiness perception in solid lipoprotein matrices. 1. Effect of composition and texture. J. Agric. Food Chem. 2012, 60, 5287–5298. [Google Scholar] [CrossRef] [Green Version]
- Smith, S.B.; Crouse, J.D. Relative contributions of acetate, lactate and glucose to lipogenesis in bovine intramuscular and subcutaneous adipose tissue. J. Nutr. 1984, 114, 792–800. [Google Scholar] [CrossRef]
- Frank, D.; Ball, A.; Hughes, J.; Krishnamurthy, R.; Piyasiri, U.; Stark, J.; Watkins, P.; Warner, R. Sensory and flavor chemistry characteristics of Australian beef: Influence of intramuscular fat, feed, and breed. J. Agric. Food Chem. 2016, 64, 4299–4311. [Google Scholar] [CrossRef]
- Kim, C.J.; Lee, E.S. Effects of quality grade on the chemical, physical and sensory characteristics of Hanwoo (Korean native cattle) beef. Meat Sci. 2003, 63, 397–405. [Google Scholar] [CrossRef]
- Kemp, C.M.; Sensky, P.L.; Bardsley, R.G.; Buttery, P.J.; Parr, T. Tenderness–An enzymatic view. Meat Sci. 2010, 84, 248–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Modzelewska-Kapitula, M.; Nogalski, Z. The influence of diet on collagen content and quality attributes of infraspinatus muscle from Holstein-Friesian young bulls. Meat Sci. 2016, 117, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Oksbjerg, N.; Gondret, F.; Vestergaard, M. Basic principles of muscle development and growth in meat-producing mammals as affected by the insulin-like growth factor (IGF) system. Domest. Anim. Endocrinol. 2004, 27, 219–240. [Google Scholar] [CrossRef]
| Item | Diet 1 | ||
|---|---|---|---|
| LE | ME | HE | |
| Ingredient, % | |||
| Corn grain | 24.8 | 40.7 | 57.6 |
| DDGS 2 | 4.00 | 4.50 | 5.00 |
| Extruded soybean | 3.04 | 3.25 | 3.50 |
| Soybean meal | 3.08 | 2.00 | 0.81 |
| Corn silage | 49.9 | 36.5 | 23.0 |
| Wheat straw | 8.10 | 6.80 | 5.40 |
| Peanut hay | 6.00 | 4.70 | 2.60 |
| Mineral-vitamin premix 3 | 0.36 | 0.52 | 0.69 |
| NaHCO3 | 0.36 | 0.52 | 0.69 |
| NaCl | 0.36 | 0.52 | 0.69 |
| Nutrient level | |||
| Metabolizable energy (MJ/kg) | 10.12 | 10.90 | 11.68 |
| OM (%) | 92.64 | 93.26 | 93.99 |
| CP (%) | 10.61 | 10.62 | 10.59 |
| NDF (%) | 38.39 | 31.71 | 24.59 |
| EE (%) | 3.89 | 4.01 | 4.14 |
| Item 1 | LE | ME | HE | SEM | p-Value 2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 23 | 26 | 20 | 23 | 26 | 20 | 23 | 26 | Diet | Age | Diet × Age | ||
| DMI 3 (kg/d) | 8.42 ab | 9.30 c | 9.97 d | 8.18 a | 8.83 b | 10.2 de | 8.72 b | 10.5 e | 12.0 f | 0.16 | <0.001 | <0.001 | <0.001 |
| ADG (kg/d) | 0.95 a | 0.93 a | 1.10 b | 0.92 a | 1.00 ab | 1.28 c | 1.00 ab | 1.28 c | 1.50 d | 0.04 | <0.001 | <0.001 | <0.001 |
| FCR | 8.71 bcd | 9.56 d | 9.49 d | 9.02 cd | 8.83 bcd | 8.07 ab | 8.91 cd | 8.33 abc | 7.89 a | 0.25 | 0.002 | 0.096 | 0.012 |
| Body weight (kg) | 580 a | 661 b | 799 d | 575 a | 672 b | 838 d | 583 a | 723 c | 898 e | 14.3 | 0.001 | 0.001 | 0.048 |
| HCW (kg) | 308 a | 354 b | 460 e | 311 a | 380 c | 484 e | 320 a | 410 d | 522 f | 14.9 | <0.001 | <0.001 | 0.056 |
| Dressing percentage (%) | 53.05 a | 53.50 ab | 57.62 c | 54.04 ab | 56.48 c | 57.74 c | 54.80 b | 56.69 c | 58.18 c | 0.39 | 0.002 | <0.001 | 0.099 |
| Item 1 | LE | ME | HE | SEM | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 23 | 26 | 20 | 23 | 26 | 20 | 23 | 26 | Diet | Age | Diet × Age | ||
| pH | 6.73 | 6.74 | 6.89 | 6.67 | 6.71 | 6.83 | 6.85 | 6.70 | 6.69 | 0.08 | 0.687 | 0.378 | 0.231 |
| NH3-N (mg/dL) | 4.50 | 4.47 | 4.41 | 4.64 | 5.28 | 4.72 | 5.53 | 4.93 | 5.38 | 0.71 | 0.381 | 0.994 | 0.927 |
| VFA 2 (mmol/L) | |||||||||||||
| Acetate | 60.2 | 60.7 | 61.1 | 57.7 | 56.8 | 58.4 | 54.0 | 54.2 | 51.3 | 1.19 | <0.001 | 0.929 | 0.340 |
| Propionate | 13.6 | 13.7 | 13.0 | 16.3 | 16.1 | 15.5 | 18.2 | 18.0 | 17.2 | 0.67 | <0.001 | 0.294 | 0.999 |
| Isobutyrate | 0.61 | 0.59 | 0.59 | 0.58 | 0.60 | 0.57 | 0.65 | 0.61 | 0.60 | 0.04 | 0.475 | 0.698 | 0.966 |
| Butyrate | 7.15 | 6.91 | 7.37 | 6.82 | 7.22 | 7.47 | 8.32 | 7.19 | 7.91 | 0.49 | 0.194 | 0.485 | 0.635 |
| Isovalerate | 1.62 | 1.39 | 1.56 | 1.45 | 1.76 | 1.53 | 1.55 | 1.64 | 1.77 | 0.15 | 0.579 | 0.793 | 0.412 |
| Valerate | 0.63 | 0.67 | 0.84 | 0.68 | 0.71 | 0.81 | 0.80 | 0.87 | 0.86 | 0.06 | 0.019 | 0.031 | 0.637 |
| Total VFA | 83.8 | 83.9 | 84.5 | 83.5 | 83.2 | 84.3 | 83.5 | 82.6 | 79.7 | 1.33 | 0.133 | 0.767 | 0.373 |
| A/P | 4.42 | 4.44 | 4.73 | 3.56 | 3.57 | 3.79 | 2.98 | 3.02 | 3.00 | 0.18 | <0.001 | 0.398 | 0.899 |
| Item 1 | LE | ME | HE | SEM | p-Value 2 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 20 | 23 | 26 | 20 | 23 | 26 | 20 | 23 | 26 | Diet | Age | Diet × Age | ||
| pH | 5.59 | 5.69 | 5.55 | 5.65 | 5.56 | 5.61 | 5.66 | 5.65 | 5.71 | 0.06 | 0.430 | 0.987 | 0.478 |
| DM 3 (%) | 26.6 ab | 26.1 a | 26.5 ab | 26.7 ab | 27.1 ab | 28.0 b | 26.7 ab | 27.0 ab | 31.1 c | 0.45 | 0.001 | <0.001 | 0.002 |
| Cooking loss (%) | 31.2 | 31.5 | 31.3 | 29.5 | 29.3 | 29.4 | 30.1 | 30.5 | 28.7 | 2.03 | 0.533 | 0.936 | 0.993 |
| Pressing loss (%) | 66.6 | 66.7 | 66.3 | 66.2 | 67.9 | 68.7 | 67.9 | 68.5 | 68.7 | 1.65 | 0.460 | 0.772 | 0.962 |
| Drip loss (%) | 5.87 | 5.43 | 5.22 | 5.74 | 5.23 | 4.99 | 5.60 | 4.89 | 4.61 | 0.53 | 0.562 | 0.208 | 0.998 |
| WBSF (N) | 57.5 | 50.8 | 49.9 | 59.5 | 56.2 | 49.8 | 61.5 | 52.3 | 55.1 | 4.45 | 0.657 | 0.124 | 0.909 |
| IMF (%) | 4.43 a | 5.13 ab | 5.44 ab | 4.54 a | 5.55 ab | 7.17 c | 4.87 ab | 6.32 bc | 9.24 d | 0.51 | 0.001 | <0.001 | 0.052 |
| CP (%) | 18.9 | 19.3 | 19.5 | 19.1 | 19.5 | 18.7 | 18.7 | 19.0 | 19.5 | 0.28 | 0.793 | 0.300 | 0.333 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Li, H.; Wu, F.; Qiu, X.; Yu, Z.; Niu, W.; He, Y.; Su, H.; Cao, B. Effects of Dietary Energy on Growth Performance, Rumen Fermentation and Bacterial Community, and Meat Quality of Holstein-Friesians Bulls Slaughtered at Different Ages. Animals 2019, 9, 1123. https://doi.org/10.3390/ani9121123
Wang H, Li H, Wu F, Qiu X, Yu Z, Niu W, He Y, Su H, Cao B. Effects of Dietary Energy on Growth Performance, Rumen Fermentation and Bacterial Community, and Meat Quality of Holstein-Friesians Bulls Slaughtered at Different Ages. Animals. 2019; 9(12):1123. https://doi.org/10.3390/ani9121123
Chicago/Turabian StyleWang, Haibo, Hang Li, Fei Wu, Xinjun Qiu, Zhantao Yu, Wenjing Niu, Yang He, Huawei Su, and Binghai Cao. 2019. "Effects of Dietary Energy on Growth Performance, Rumen Fermentation and Bacterial Community, and Meat Quality of Holstein-Friesians Bulls Slaughtered at Different Ages" Animals 9, no. 12: 1123. https://doi.org/10.3390/ani9121123
APA StyleWang, H., Li, H., Wu, F., Qiu, X., Yu, Z., Niu, W., He, Y., Su, H., & Cao, B. (2019). Effects of Dietary Energy on Growth Performance, Rumen Fermentation and Bacterial Community, and Meat Quality of Holstein-Friesians Bulls Slaughtered at Different Ages. Animals, 9(12), 1123. https://doi.org/10.3390/ani9121123







