Exploring the Ruminal Microbial Community Associated with Fat Deposition in Lambs
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Sampling
2.2. Body Mass Index Calculation and Subgroup Analysis
2.3. Extraction of Microbial DNA and Bacterial 16S rRNA Gene PCR Amplification
2.4. Sequence Processing, OTU Clustering, and OTU Filtering
2.5. Statistical Analysis
3. Results
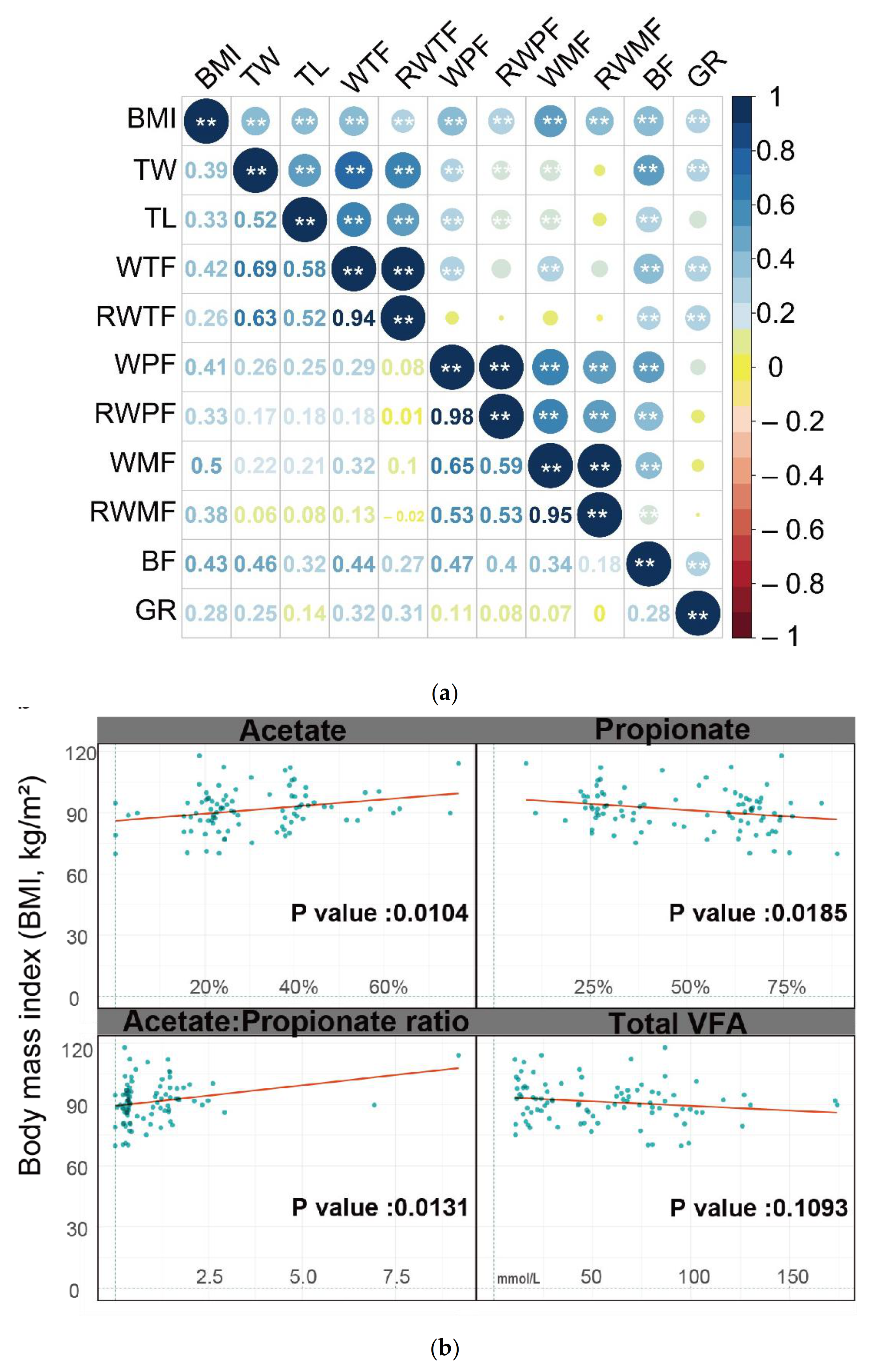
3.1. Phenotypic Parameters and Correlations between Traits
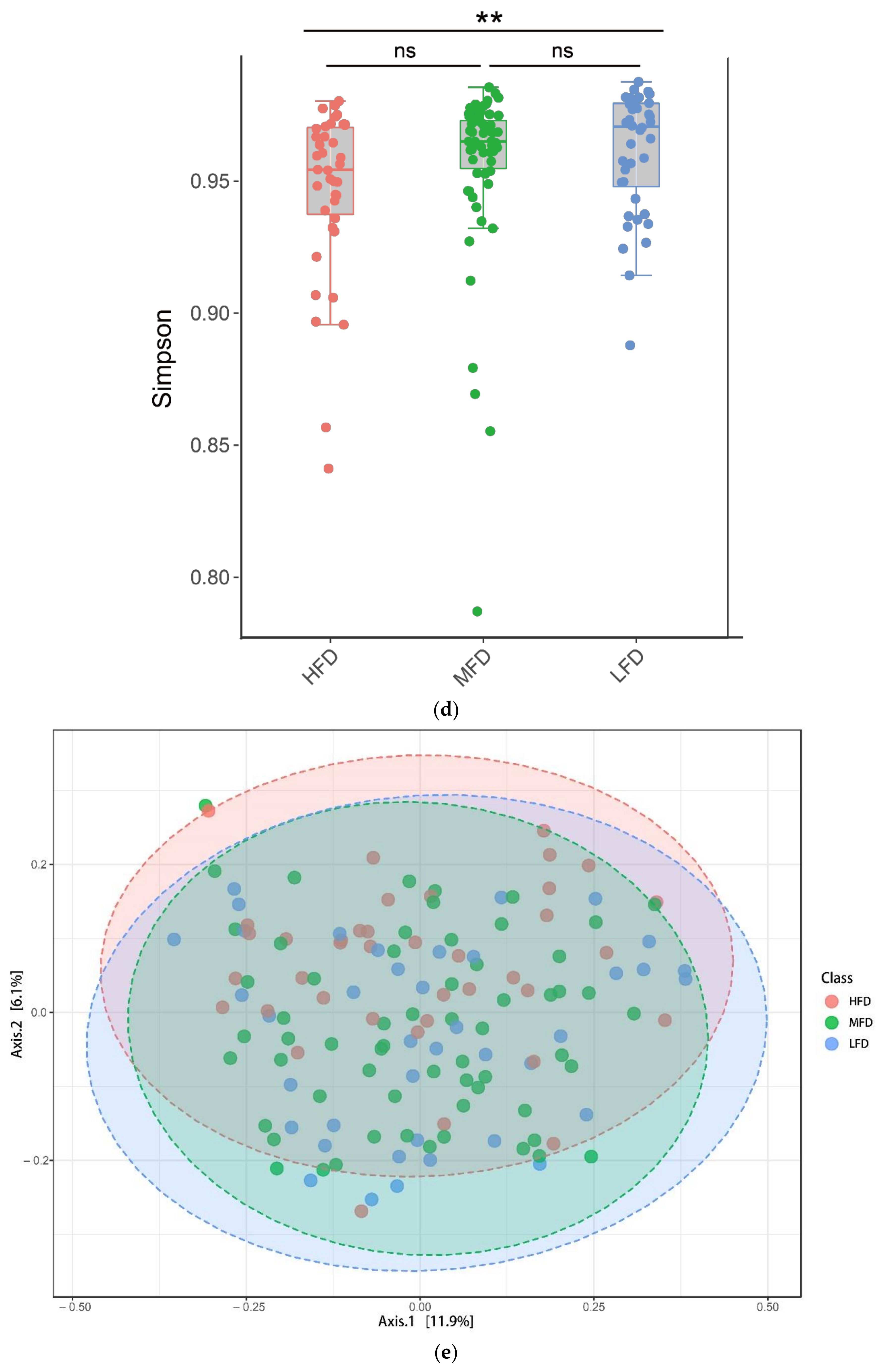
3.2. Comparing the Rumen Microbial Community Structures among Sheep with Distinct Levels of Fat Deposition
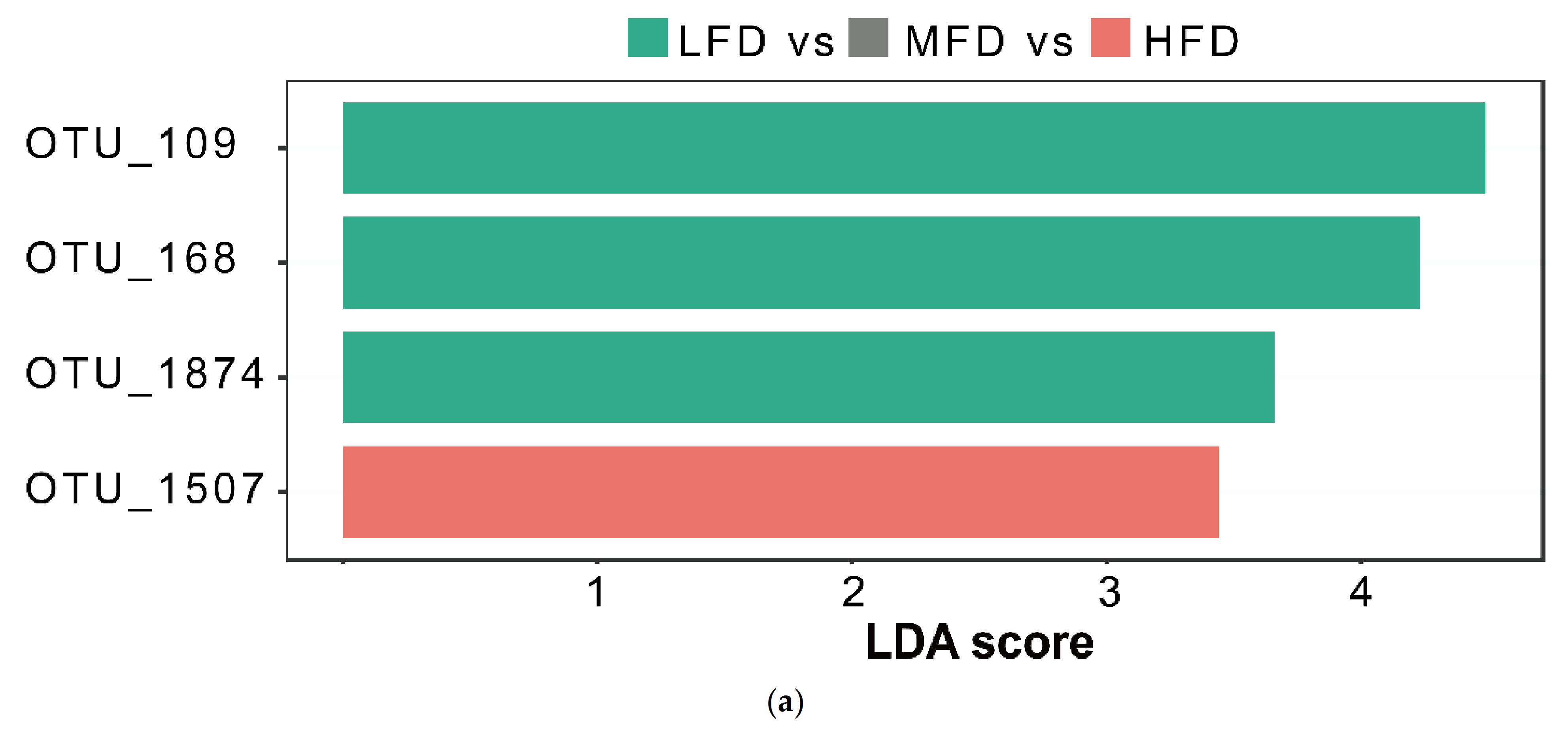
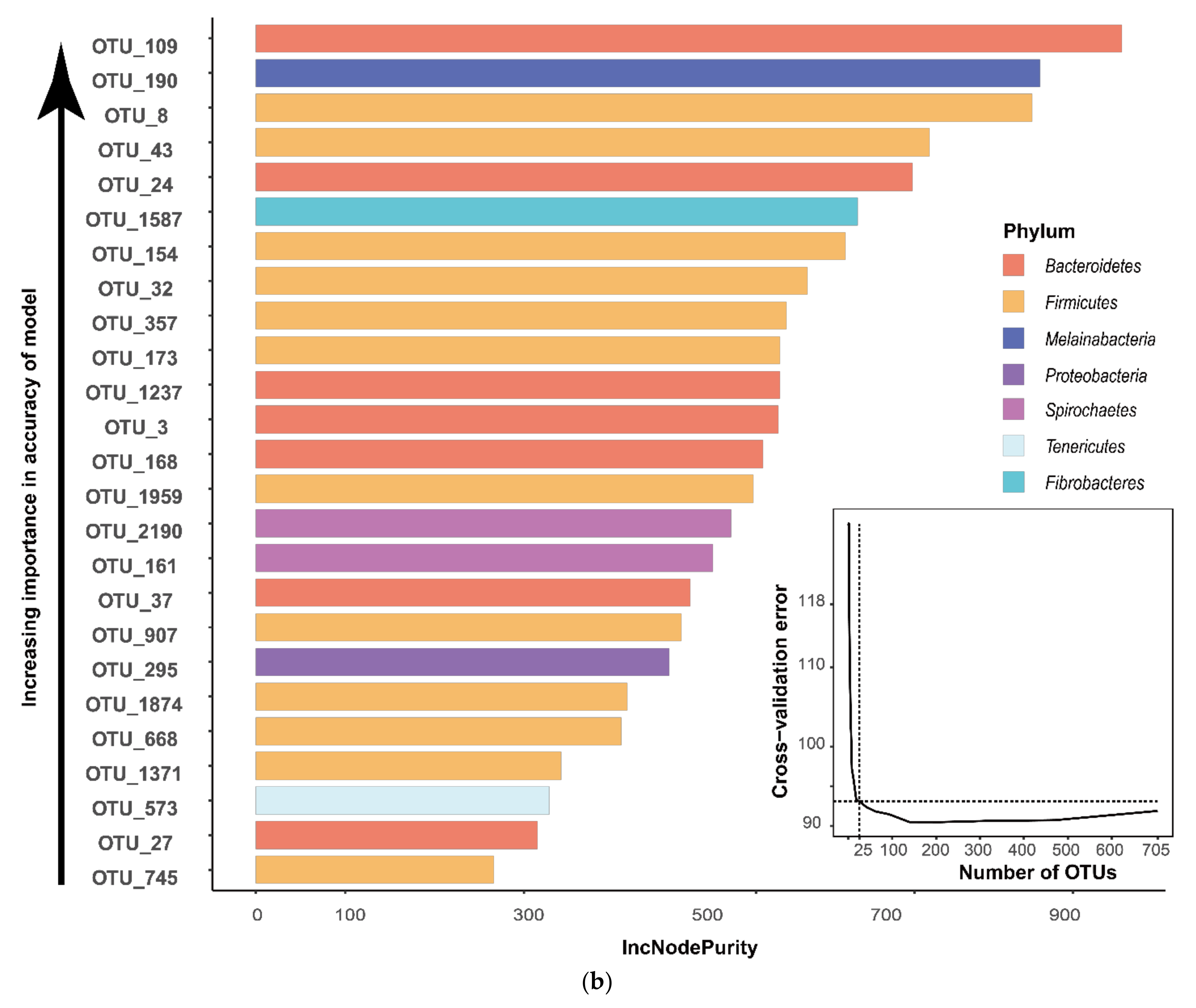
3.3. Analysis of Differential Rumen Microbiota
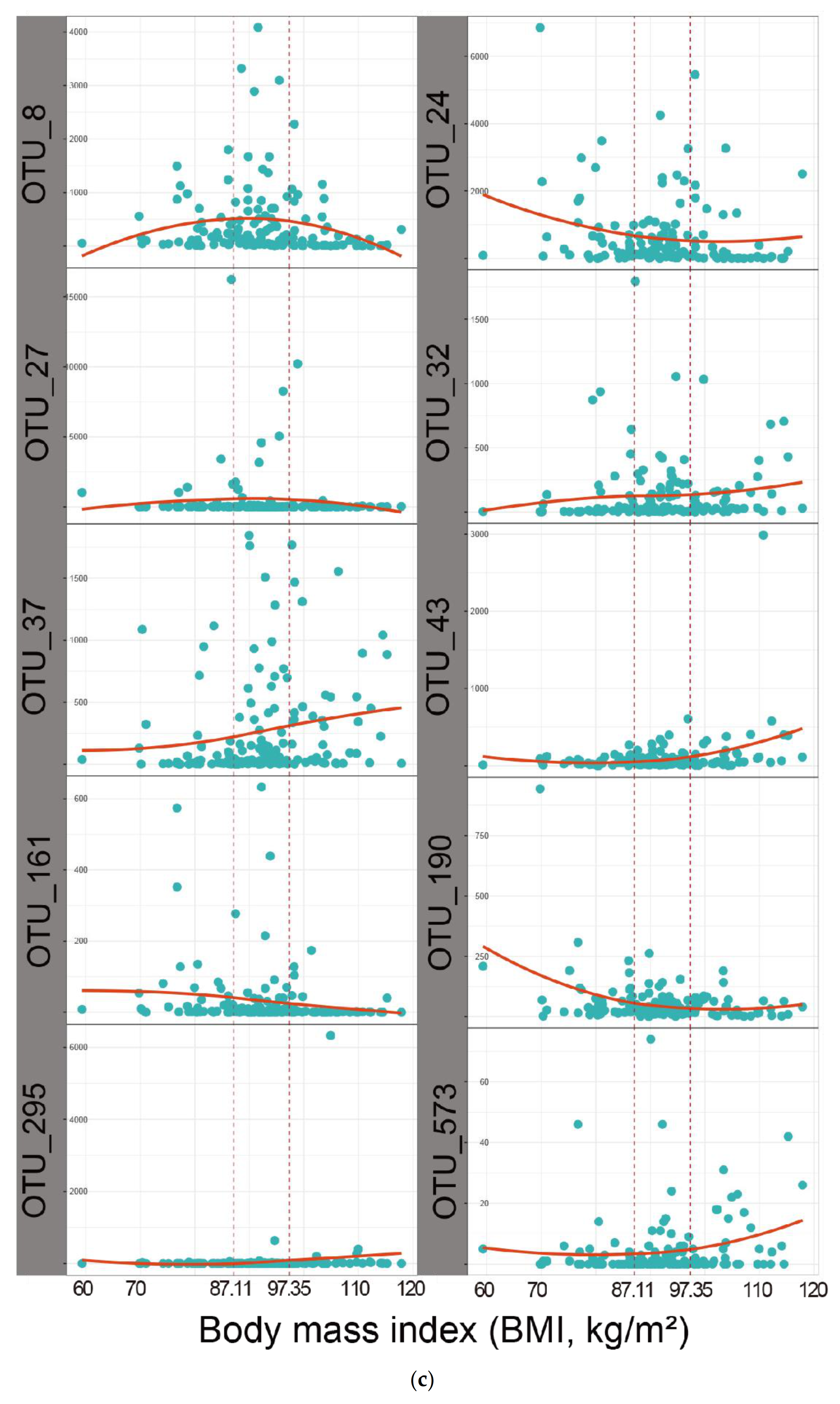
3.4. Correlation between VFA and Significant Biomarkers of Rumen Digesta
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
Appendix A.1. Supplementary Methods: Volatile Fatty Acid Analysis
| Ingredient Composition (% as fed) | Chemical Composition | ||
|---|---|---|---|
| Corn | 32.5 | Dry matter (DM) [%] | 88.78 |
| Corn germ meal | 18 | Crude protein (CP) [%] | 13.09 |
| Corn stalks | 12 | Digestible energy [MJ/kg] | 11.11 |
| Corn hulls | 11.2 | Neutral detergent fiber (NDF) [%] | 27.08 |
| Corn cob | 8 | Acid detergent fiber (ADF) [%] | 13.99 |
| Soybean meal | 5 | Crude fiber (CF) [%] | 9.78 |
| Cotton meal | 5 | ||
| Molasses | 3.3 | ||
| Bentonite | 1.5 | ||
| Baking soda | 1 | ||
| Stone powder | 0.8 | ||
| Expanded Urea | 0.5 | ||
| Premix | 0.5 | ||
| Regression Coefficients(β) | Std. Error | p Value | |
|---|---|---|---|
| Acetate | 17.57 | 6.71 | 0.0104 |
| Propionate | −12.05 | 5.02 | 0.0185 |
| Isobutyrate | 134.57 | 78.1 | 0.0884 |
| Butyrate | 7.74 | 11.88 | 0.5164 |
| Isovalerate | 13.74 | 27.67 | 0.6208 |
| Valerate | −64.9 | 96.26 | 0.502 |
| Total VFA | −0.04 | 0.03 | 0.1093 |
| Acetate:Propionate ratio | 2.02 | 0.8 | 0.0131 |
| OTUs | Phylum | Class | Order | Family | Genus | Species |
|---|---|---|---|---|---|---|
| OTU_1237 | Bacteroidetes | Bacteroidia | Bacteroidales | Prevotellaceae | unidentified_Prevotellaceae | rumen_bacterium_R-9 |
| OTU_27 | Bacteroidetes | Bacteroidia | Bacteroidales | Muribaculaceae | ||
| OTU_3 | Bacteroidetes | Bacteroidia | Bacteroidales | Prevotellaceae | ||
| OTU_109 | Bacteroidetes | Bacteroidia | Bacteroidales | Prevotellaceae | ||
| OTU_168 | Bacteroidetes | Bacteroidia | Bacteroidales | Prevotellaceae | ||
| OTU_24 | Bacteroidetes | Bacteroidia | Bacteroidales | Rikenellaceae | ||
| OTU_37 | Bacteroidetes | Bacteroidia | Bacteroidales | |||
| OTU_1587 | Fibrobacteres | Fibrobacteria | Fibrobacterales | Fibrobacteraceae | Fibrobacter | bacterium_NC3008 |
| OTU_43 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | Oribacterium | |
| OTU_173 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | unidentified_Lachnospiraceae | bacterium_AD3010 |
| OTU_907 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | unidentified_Lachnospiraceae | Lachnospiraceae_bacterium_NK4A179 |
| OTU_1959 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | unidentified_Lachnospiraceae | bacterium_enrichment_culture_clone_R1-3 |
| OTU_154 | Firmicutes | Negativicutes | Selenomonadales | Veillonellaceae | unidentified_Veillonellaceae | Selenomonas_ruminantium |
| OTU_668 | Firmicutes | Negativicutes | Selenomonadales | Veillonellaceae | unidentified_Veillonellaceae | rumen_bacterium_RC-11 |
| OTU_8 | Firmicutes | Clostridia | Clostridiales | Christensenellaceae | ||
| OTU_357 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | ||
| OTU_1371 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | ||
| OTU_1874 | Firmicutes | Clostridia | Clostridiales | Lachnospiraceae | ||
| OTU_32 | Firmicutes | Clostridia | Clostridiales | Ruminococcaceae | ||
| OTU_745 | Firmicutes | Negativicutes | Selenomonadales | Veillonellaceae | g__unidentified_Veillonellaceae | |
| OTU_190 | Melainabacteria | |||||
| OTU_295 | Proteobacteria | Gammaproteobacteria | Aeromonadales | Succinivibrionaceae | Succinivibrio | |
| OTU_2190 | Spirochaetes | Spirochaetia | Spirochaetales | Spirochaetaceae | unidentified_Spirochaetaceae | Treponema_bryantii |
| OTU_161 | Spirochaetes | Spirochaetia | Spirochaetales | Spirochaetaceae | ||
| OTU_573 | Tenericutes | Mollicutes |
| Sample Size (Randomly Selected) | Number of Replications | Correlation Coefficient (r) | p Value | |||
|---|---|---|---|---|---|---|
| Mean | Max | Min | Sd | |||
| 20.00 | 120.00 | 0.23 | 0.38 | 0.17 | 0.05 | <0.001 |
| 30.00 | 110.00 | 0.20 | 0.30 | 0.15 | 0.04 | <0.001 |
| 50.00 | 90.00 | 0.15 | 0.24 | 0.10 | 0.03 | <0.001 |
| 70.00 | 70.00 | 0.13 | 0.22 | 0.09 | 0.03 | <0.001 |
| 80.00 | 60.00 | 0.12 | 0.19 | 0.08 | 0.03 | <0.001 |
| 90.00 | 50.00 | 0.12 | 0.16 | 0.08 | 0.02 | <0.001 |
| 100.00 | 40.00 | 0.11 | 0.15 | 0.07 | 0.02 | <0.001 |
| 110.00 | 30.00 | 0.10 | 0.15 | 0.07 | 0.02 | <0.001 |
| 120.00 | 20.00 | 0.10 | 0.16 | 0.07 | 0.02 | <0.001 |
| 130.00 | 10.00 | 0.10 | 0.12 | 0.09 | 0.01 | <0.001 |
| 140.00 | 1.00 | 0.10 | -- | -- | -- | <0.001 |

Appendix A.2. Appendix A References
- Erwin, E.S.; Marco, G.J.; Emery, E.M. Volatile fatty acid analyses of blood and rumen fluid by gas chromatography. J. Dairy Sci. 1961, 44, 1768–1771.
- AOAC. Official Methods of Analysis, 15th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 1990.
- Van, Soest, P.J.; Robertson, J.B.; Lewis, B.A. Methods for dietary fiber, neutral detergent fiber, and nonstarch polysaccharides in relation to animal nutrition. J. Dairy Sci. 1991, 74, 3583–3597.
References
- Xu, S.-S.; Ren, X.; Yang, G.-L.; Xie, X.-L.; Zhao, Y.-X.; Zhang, M.; Shen, Z.-Q.; Ren, Y.-L.; Gao, L.; Shen, M.; et al. Genome-wide association analysis identifies the genetic basis of fat deposition in the tails of sheep (Ovis aries). Anim. Genet. 2017, 48, 560–569. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, H.; Hou, Y.; Sahana, G.; Gao, H.; Zhu, C.; Du, L.; Zhao, F.; Wang, L. A Transcriptomic Study of the Tail Fat Deposition in Two Types of Hulun Buir Sheep According to Tail Size and Sex. Animals 2019, 9, 655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Xu, M.; Wang, X.; Yang, J.; Gao, L.; Zhang, Y.; Huang, X.; Han, M.; Gao, R.; Gan, S. Comparative Proteome Analysis Reveals Lipid Metabolism-Related Protein Networks in Response to Rump Fat Mobilization. Int. J. Mol. Sci. 2018, 19, 2556. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, X.; Wang, W.; Mo, F.; La, Y.; Li, C.; Li, F. Association of residual feed intake with growth and slaughtering performance, blood metabolism, and body composition in growing lambs. Sci. Rep. 2017, 7, 12681. [Google Scholar] [CrossRef] [Green Version]
- Domínguez, R.; Pateiro, M.; Gagaoua, M.; Barba, F.J.; Zhang, W.; Lorenzo, J.M. A Comprehensive Review on Lipid Oxidation in Meat and Meat Products. Antioxidants 2019, 8, 429. [Google Scholar] [CrossRef] [Green Version]
- Fan, Q.; Wanapat, M.; Yan, T.; Hou, F. Altitude influences microbial diversity and herbage fermentation in the rumen of yaks. BMC Microbiol. 2020, 20, 370. [Google Scholar] [CrossRef]
- Clemmons, B.A.; Martino, C.; Schneider, L.; Lefler, J.; Embree, M.M.; Myer, P.R. Temporal Stability of the Ruminal Bacterial Communities in Beef Steers. Sci. Rep. 2019, 9, 9522. [Google Scholar] [CrossRef]
- Li, F.; Hitch, T.C.A.; Chen, Y.; Creevey, C.; Guan, L.L. Comparative metagenomic and metatranscriptomic analyses reveal the breed effect on the rumen microbiome and its associations with feed efficiency in beef cattle. Microbiome 2019, 7, 6. [Google Scholar] [CrossRef] [Green Version]
- Islam, M.; Lee, S.-S. Advanced estimation and mitigation strategies: A cumulative approach to enteric methane abatement from ruminants. J. Anim. Sci. Technol. 2019, 61, 122–137. [Google Scholar] [CrossRef] [Green Version]
- Erwin, S.; Marco, G.; Emery, A.E. Volatile fatty acid analysis of blood and rumen fluid by gas chromatography. J. Dairy Sci. 1961, 44, 1768–1771. [Google Scholar] [CrossRef]
- Gootwine, E. Meta-analysis of morphometric parameters of late-gestation fetal sheep developed under natural and artificial constraints. J. Anim. Sci. 2012, 91, 111–119. [Google Scholar] [CrossRef]
- Nieto, C.R.; Mantey, A.; Makela, B.; Byrem, T.; Ehrhardt, R.; Veiga-Lopez, A. Shearing during late pregnancy increases size at birth but does not alter placental endocrine responses in sheep. Anim. Int. J. Anim. Biosci. 2020, 14, 799–806. [Google Scholar] [CrossRef]
- Keys, A.; Fidanza, F.; Karvonen, M.J.; Kimura, N.; Taylor, H.L. Indices of relative weight and obesity. J. Chronic Dis. 1972, 25, 329–343. [Google Scholar] [CrossRef]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-Source, Platform-Independent, Community-Supported Software for Describing and Comparing Microbial Communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [Green Version]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef] [Green Version]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naïve Bayesian Classifier for Rapid Assignment of rRNA Sequences into the New Bacterial Taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [Green Version]
- Batool, M.; Ali, S.B.; Jaan, A.; Khalid, K.; Ali, S.A.; Kamal, K.; Raja, A.A.; Gul, F.; Nasir, A. Initial Sequencing and Characterization of the Gastrointestinal and Oral Microbiota in Urban Pakistani Adults. Front. Cell. Infect. Microbiol. 2020, 10, 409. [Google Scholar] [CrossRef]
- Paranjape, K.; Bédard, É.; Shetty, D.; Hu, M.; Choon, F.C.P.; Prévost, M.; Faucher, S.P. Unravelling the importance of the eukaryotic and bacterial communities and their relationship with Legionella spp. ecology in cooling towers: A complex network. Microbiome 2020, 8, 157. [Google Scholar] [CrossRef]
- Revelle, W. Psych: Procedures for Personality and Psychological Research (1.9.12.). 2019. Available online: https://CRAN.R-project.org/package=psych (accessed on 14 December 2021).
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Chong, J.; Liu, P.; Zhou, G.; Xia, J. Using Microbiome Analyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat. Protoc. 2020, 15, 799–821. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’Hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, H.; Wagner, H.H. Vegan: Community Ecology Package. 2015. Available online: https://mran.microsoft.com/snapshot/2015-11-17/web/packages/vegan/vegan.pdf (accessed on 14 December 2021).
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef] [Green Version]
- Yousefi, A.R.; Kohram, H.; Shahneh, A.Z.; Nik-Khah, A.; Campbell, A.W. Comparison of the meat quality and fatty acid composition of traditional fat-tailed (Chall) and tailed (Zel) Iranian sheep breeds. Meat Sci. 2012, 92, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Akaboshi, N.; Inoue, Y.; Kamomae, H.; Kaneda, Y. Fasting-induced suppression of pulsatile luteinizing hormone secretion is related to body energy status in ovariectomized goats. Anim. Reprod. Sci. 2002, 72, 185–196. [Google Scholar] [CrossRef]
- Blasco, A.; Nagy, I.; Hernández, P. Genetics of growth, carcass and meat quality in rabbits. Meat Sci. 2018, 145, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Clark, J.; Klusmeyer, T.; Cameron, M. Microbial Protein Synthesis and Flows of Nitrogen Fractions to the Duodenum of Dairy Cows. J. Dairy Sci. 1992, 75, 2304–2323. [Google Scholar] [CrossRef]
- Baird, G.D.; Lomax, A.M.; Symonds, H.W.; Shaw, S.R. Net hepatic and splanchnic metabolism of lactate, pyruvate and propionate in dairy cows in vivo in relation to lactation and nutrient supply. Biochem. J. 1980, 186, 47–57. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.N.; Song, J.; Kim, E.J.; Chang, J.; Kim, C.-H.; Seo, S.; Chang, M.B.; Bae, G.-S. Effects of short-term fasting on in vivo rumen microbiota and in vitro rumen fermentation characteristics. Asian-Australas. J. Anim. Sci. 2019, 32, 776–782. [Google Scholar] [CrossRef] [Green Version]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate Improves Insulin Sensitivity and Increases Energy Expenditure in Mice. Diabetes 2009, 58, 1509–1517. [Google Scholar] [CrossRef] [Green Version]
- Xiong, Y.; Pang, J.; Lv, L.; Wu, Y.; Li, N.; Huang, S.; Feng, Z.; Ren, Y.; Wang, J. Effects of Maternal Supplementation with Rare Earth Elements during Late Gestation and Lactation on Performances, Health, and Fecal Microbiota of the Sows and Their Offspring. Animals 2019, 9, 738. [Google Scholar] [CrossRef] [Green Version]
- Ilario, F.; Valentina, P.; Roberto, G.; Adriana, Z.; Filomena, L.; Clara, M.; Ilaria, G.; Rosalba, R.; Angelo, R.; Giorgio, G. Changes in the gut microbiota composition during pregnancy in patients with gestational diabetes mellitus (GDM). Sci. Rep. 2018, 8, 12216. [Google Scholar]
- De Nardi, R.; Marchesini, G.; Li, S.; Khafipour, E.; Plaizier, K.J.C.; Gianesella, M.; Ricci, R.; Andrighetto, I.; Segato, S. Metagenomic analysis of rumen microbial population in dairy heifers fed a high grain diet supplemented with dicarboxylic acids or polyphenols. BMC Vet. Res. 2016, 12, 29. [Google Scholar] [CrossRef]
- Jami, E.; Mizrahi, I. Composition and Similarity of Bovine Rumen Microbiota across Individual Animals. PLoS ONE 2012, 7, e33306. [Google Scholar] [CrossRef] [Green Version]
- Di Rienzi, S.; Sharon, I.; Wrighton, K.C.; Koren, O.; Hug, A.L.; Thomas, B.C.; Goodrich, J.K.; Bell, J.; Spector, T.D.; Banfield, J.F.; et al. The human gut and groundwater harbor non-photosynthetic bacteria belonging to a new candidate phylum sibling to Cyanobacteria. eLife 2013, 2, e01102. [Google Scholar] [CrossRef]
- Dumke, R.; Rust, M.; Glaunsinger, T. MgpB Types among Mycoplasma genitalium Strains from Men Who Have Sex with Men in Berlin, Germany, 2016–2018. Pathogens 2019, 9, 12. [Google Scholar] [CrossRef] [Green Version]
- Min, B.R.; Castleberry, L.; Allen, H.; Parker, D.; Waldrip, H.; Brauer, D.; Willis, W. Associative effects of wet distiller’s grains plus solubles and tannin-rich peanut skin supplementation on in vitro rumen fermentation, greenhouse gas emissions, and microbial changes. J. Anim. Sci. 2019, 97, 4668–4681. [Google Scholar] [CrossRef]
- Bekele, A.Z.; Koike, S.; Kobayashi, Y. Phylogenetic diversity and dietary association of rumen Treponema revealed using group-specific 16S rRNA gene-based analysis. FEMS Microbiol. Lett. 2011, 316, 51–60. [Google Scholar] [CrossRef]


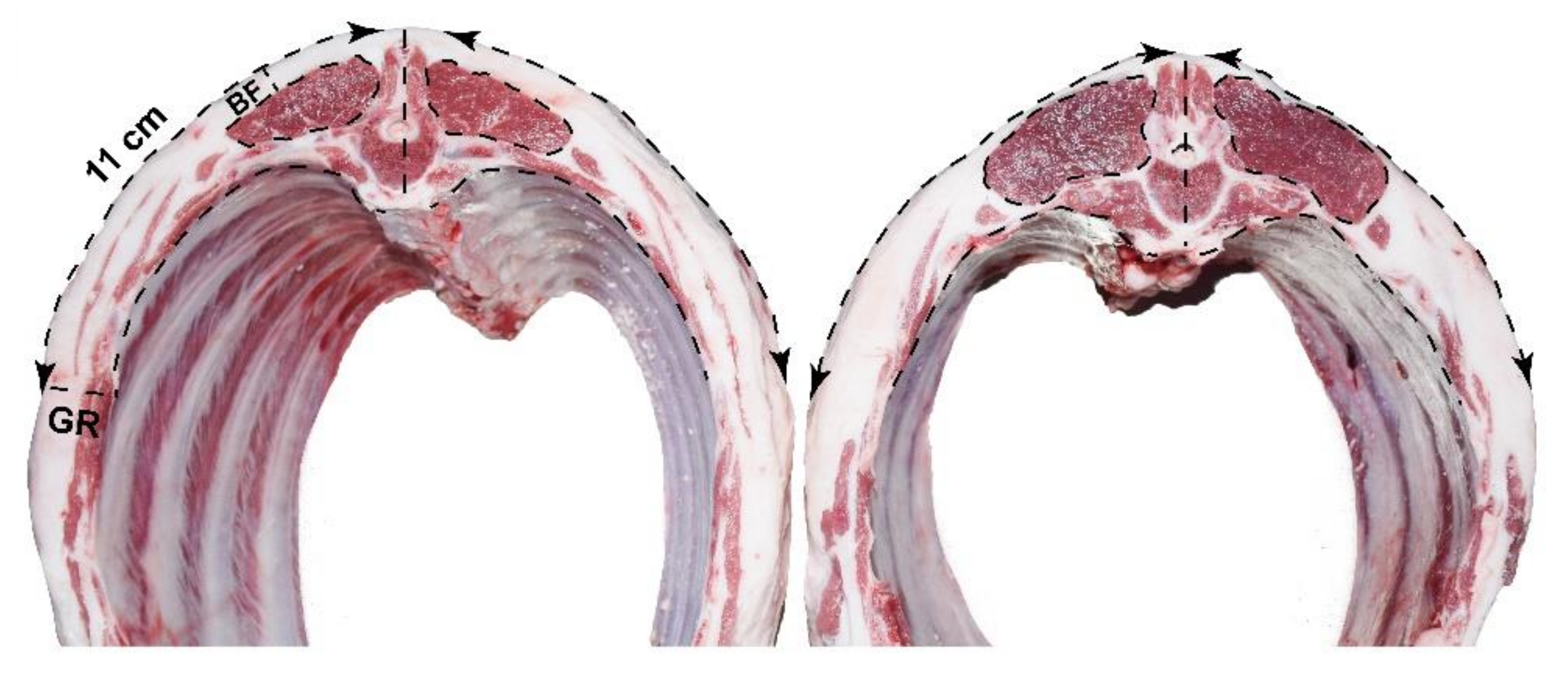
| Fat Deposit Traits | n | Mean | SD | CV 3 (%) |
|---|---|---|---|---|
| BMI 1 180 d, kg/(m2) | 141 | 92.23 | 10.25 | 11.11 |
| Tail width 180 d, cm | 141 | 18.50 | 2.23 | 12.08 |
| Tail length 180 d, cm | 141 | 19.31 | 2.83 | 14.67 |
| The weight of tail fat, kg | 141 | 1.30 | 0.43 | 33.22 |
| The relative weight of tail fat, % | 141 | 2.87 | 0.77 | 26.82 |
| The weight of perirenal fat, kg | 141 | 0.57 | 0.28 | 49.70 |
| The relative weight of perirenal fat, % | 141 | 1.23 | 0.52 | 42.70 |
| The weight of mesenteric fat, kg | 141 | 1.08 | 0.36 | 33.71 |
| The relative weight of mesenteric fat, % | 141 | 2.37 | 0.65 | 27.52 |
| Thickness of backfat, mm | 141 | 23.99 | 5.77 | 24.04 |
| Rib thickness (GR value) 2, mm | 141 | 15.61 | 4.35 | 27.85 |
| Trait | Groups | SE | p Value | ||
|---|---|---|---|---|---|
| LFD | MFD | HFD | |||
| No. of animals | 40 | 62 | 39 | ||
| Fat Deposit Traits | |||||
| BMI 1 180 d (BMI), kg/m2 | 80.42 C | 92.04 B | 104.64 A | 0.863 | <0.001 |
| Tail width 180 d (TW), cm | 17.78 b | 18.31 b | 19.54 a | 0.188 | 0.001 |
| Tail length 180 d (TL), cm | 18.00 b | 19.60 a | 20.21 a | 0.239 | 0.001 |
| The weight of tail fat (WTF), kg | 1.09 c | 1.31 b | 1.51 a | 0.036 | <0.001 |
| The relative weight of tail fat (RWTF), % | 2.63 b | 2.89 a,b | 3.08 a | 0.065 | 0.028 |
| The weight of perirenal fat (WPF), kg | 0.41 b | 0.58 a | 0.69 a | 0.024 | <0.001 |
| The relative weight of perirenal fat (RWPF), % | 0.98 b | 1.28 a | 1.41 a | 0.044 | <0.001 |
| The weight of mesenteric fat (WMF), kg | 0.83 c | 1.08 b | 1.33 a | 0.031 | <0.001 |
| The relative weight of mesenteric fat (RWMF), % | 1.98 c | 2.41 b | 2.72 a | 0.055 | <0.001 |
| Thickness of backfat (BF), mm | 21.55 b | 23.73 b | 26.90 a | 0.486 | <0.001 |
| Rib thickness (GR value) 2, mm | 14.15 b | 15.94 a | 16.59 a | 0.366 | 0.032 |
| Growth Performance | |||||
| BW 80 d, kg | 16.75 c | 18.95 b | 21.99 a | 0.32 | <0.001 |
| BW 180 d, kg | 41.44 c | 45.74 b | 50.69 a | 0.507 | <0.001 |
| Live weight before slaughter, kg | 40.88 c | 44.86 b | 49.01 a | 0.472 | <0.001 |
| Body Length 180 d, cm | 71.65 b | 70.40 a,b | 69.62 a | 0.307 | 0.042 |
| ADFI 3 80 d–180 d, kg/d | 1.43 c | 1.53 b | 1.67 a | 0.017 | <0.001 |
| ADG 4 80 d–180 d, kg/d | 0.25 c | 0.27 b | 0.29 a | 0.003 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhang, X.; Li, F.; Li, C.; Zhang, D.; Li, X.; Zhao, Y.; Wang, W. Exploring the Ruminal Microbial Community Associated with Fat Deposition in Lambs. Animals 2021, 11, 3584. https://doi.org/10.3390/ani11123584
Zhang Y, Zhang X, Li F, Li C, Zhang D, Li X, Zhao Y, Wang W. Exploring the Ruminal Microbial Community Associated with Fat Deposition in Lambs. Animals. 2021; 11(12):3584. https://doi.org/10.3390/ani11123584
Chicago/Turabian StyleZhang, Yukun, Xiaoxue Zhang, Fadi Li, Chong Li, Deyin Zhang, Xiaolong Li, Yuan Zhao, and Weimin Wang. 2021. "Exploring the Ruminal Microbial Community Associated with Fat Deposition in Lambs" Animals 11, no. 12: 3584. https://doi.org/10.3390/ani11123584
APA StyleZhang, Y., Zhang, X., Li, F., Li, C., Zhang, D., Li, X., Zhao, Y., & Wang, W. (2021). Exploring the Ruminal Microbial Community Associated with Fat Deposition in Lambs. Animals, 11(12), 3584. https://doi.org/10.3390/ani11123584






