The Educational Efficacy of Humane Teaching Methods: A Systematic Review of the Evidence
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
3. Results
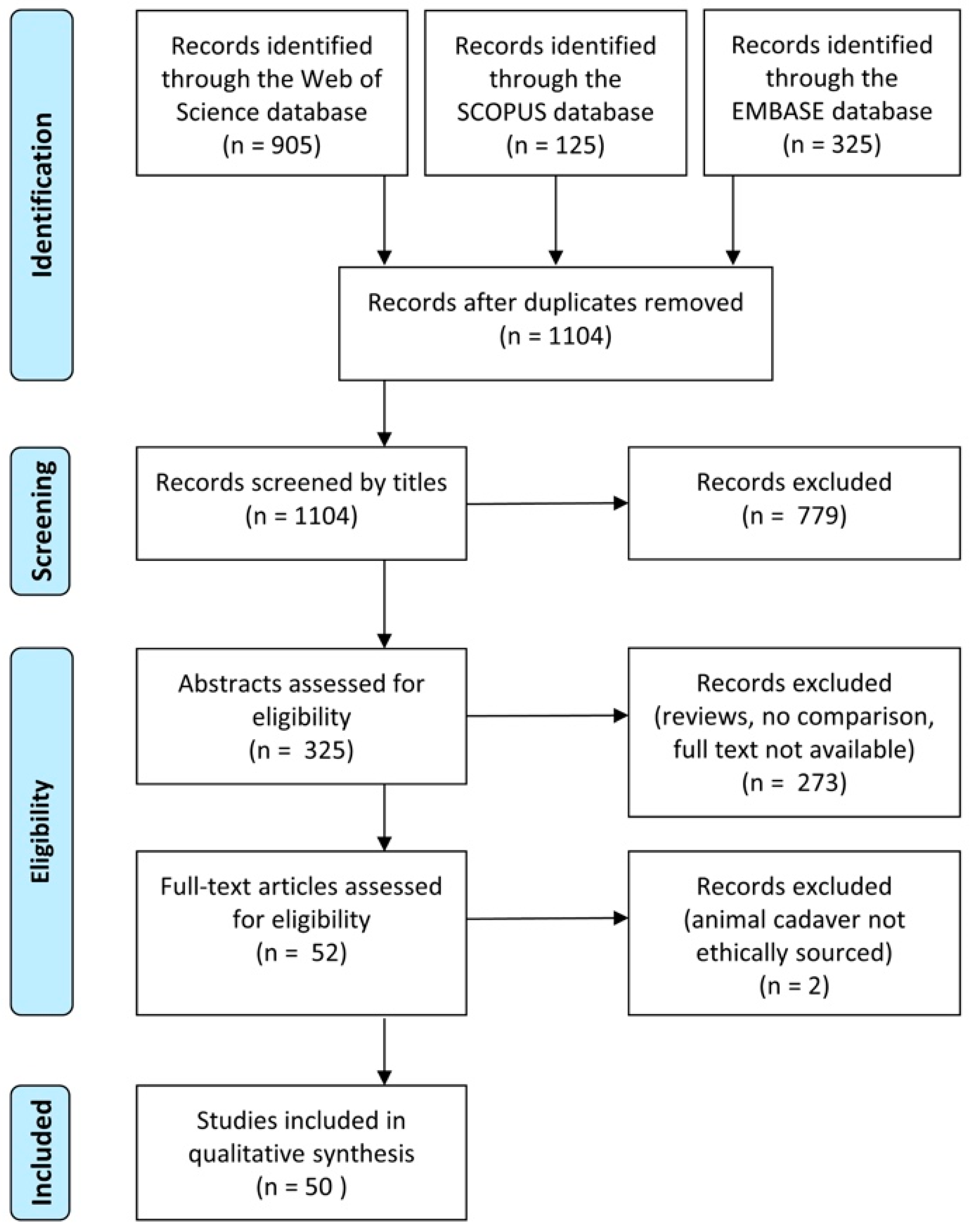
3.1. Search Results and Study Selection
3.2. Computer and Virtual Reality Simulation
3.3. Models, Mannequins, and Simulators
3.4. Other Humane Teaching Methods
4. Discussion
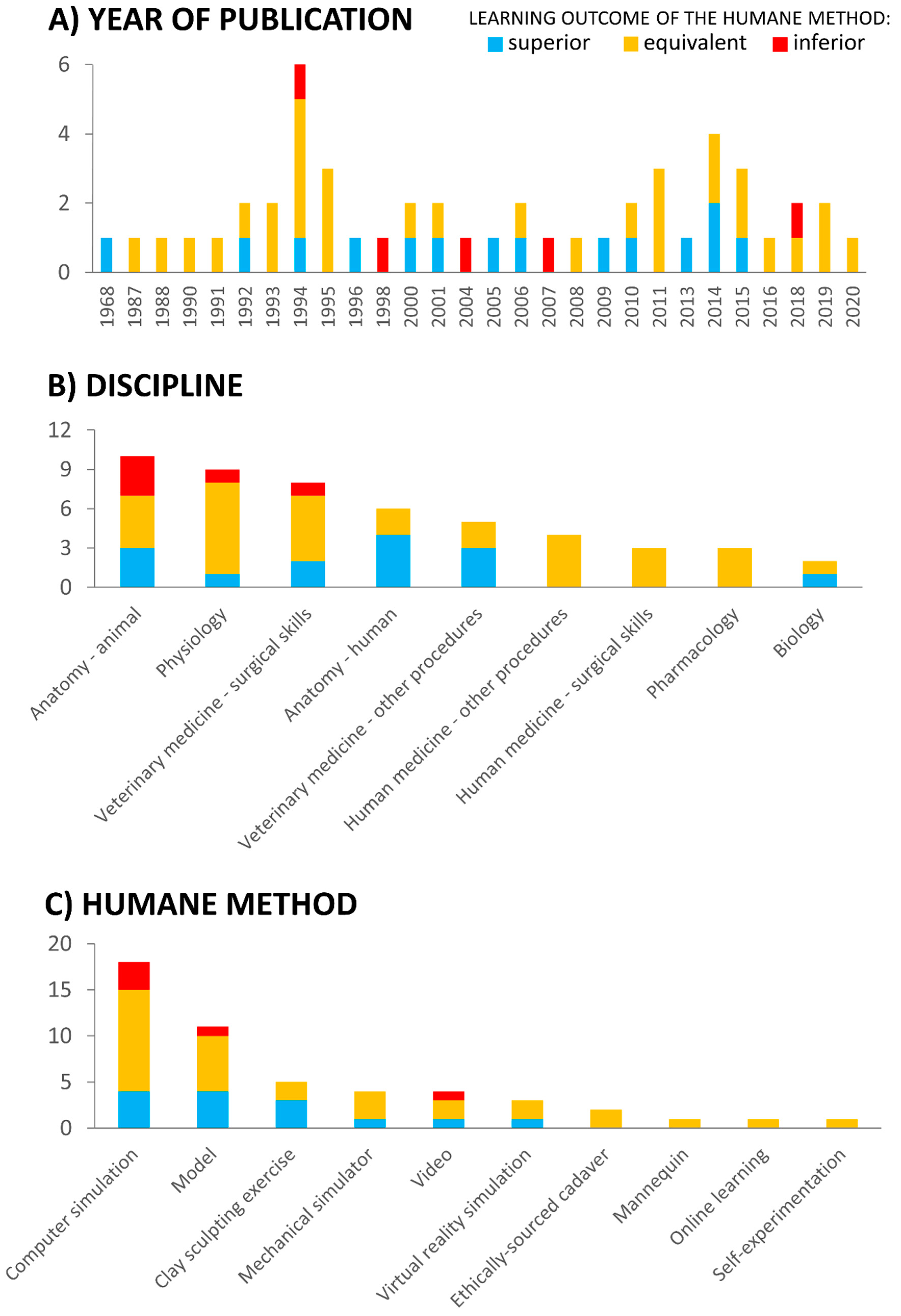
4.1. Teaching Efficacy of Humane Methods
4.2. Trends within Disciplines
4.3. Pedagogical Factors Affecting Teaching Efficacy
4.4. Other Advantages of Humane Teaching Methods
4.5. Other Types of Humane Teaching Methods
“[clinical and surgical] rotations are more likely to expose students to a higher volume of commonly-encountered conditions … Resultant benefits include greater exposure to the clinical histories, examinations, and presenting signs of cases more directly relevant to new graduates, and to the diagnostic workups and post-operative management of such cases. Surgical participation is normally conducted under close individual supervision, as distinct from the group supervision normally provided during veterinary school surgical laboratories.”
4.6. Transitioning toward Humane Teaching Methods
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Balcombe, J. The Use of Animals in Higher Education: Problems, Alternatives, and Recommendations; The Humane Society Press: Washington, DC, USA, 2000. [Google Scholar]
- Pawlowski, J.B.; Feinstein, D.M.; Gala, S.G. Developments in the transition from animal use to simulation-based biomedical education. Simul. Healthc. 2018, 13, 420–426. [Google Scholar] [CrossRef]
- Freire, R.; Nicol, C.J. A bibliometric analysis of past and emergent trends in animal welfare science. Anim. Welf. 2019, 28, 465–485. [Google Scholar] [CrossRef]
- Crettaz von Roten, F. European attitudes towards animal research: Overview and consequences for science. Sci. Technol. Soc. 2009, 14, 349–364. [Google Scholar] [CrossRef]
- Funk, C.; Rainie, L. Attitudes and beliefs on science and technology topics. In Public and Scientists’ Views on Science and Society; Funk, C., Rainie, L., Eds.; Pew Research Center: Washington, DC, USA, 2015. [Google Scholar]
- Knight, A. The effectiveness of humane teaching methods in veterinary education. ALTEX 2007, 24, 91–109. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K. The use of microcomputer simulations in undergraduate neurophysiology experiments. ATLA 1987, 14, 134–140. [Google Scholar] [CrossRef]
- Leathard, H.L.; Dewhurst, D.G. Comparison of the cost-effectiveness of a computer-assisted learning program with a tutored demonstration to teach intestinal motility to medical students. ALT-J 1995, 3, 118–125. [Google Scholar] [CrossRef]
- Botden, S.M.B.I.; Christie, L.; Goossens, R.; Jakimowicz, J.J. Training for laparoscopic Nissen fundoplication with a newly designed model: A replacement for animal tissue models? Surg. Endosc. 2010, 24, 3134–3140. [Google Scholar] [CrossRef]
- Fawver, A.L.; Branch, C.E.; Trentham, L.; Robertson, B.; Beckett, S. A comparison of interactive videodisc instruction with live animal laboratories. Adv. Physiol. Educ. 1990, 259, S11. [Google Scholar] [CrossRef]
- Leonard, W.H. A comparison of student performance following instruction by interactive videodisc versus conventional laboratory. J. Res. Sci. Teach. 1992, 29, 93–102. [Google Scholar] [CrossRef]
- Rubeis, G.; Steger, F. Is live-tissue training ethically justified? An evidence-based ethical analysis. ATLA 2018, 46, 65–71. [Google Scholar] [CrossRef]
- Shurtleff, R.S. The attitudes of today’s veterinary students on animal welfare issues. Calif. Vet. 1983, 37, 29–30. [Google Scholar]
- Heim, A. Desensitization and health. AV 1986, 94, 12–14. [Google Scholar]
- Herzog, H.A.; Vore, T.L.; New, J.C., Jr. Conversations with veterinary students: Attitudes, ethics, and animals. Anthrozoös 1989, 2, 181–188. [Google Scholar] [CrossRef]
- Shapiro, K.J. The pedagogy of learning and unlearning empathy. Phenomenol. Pedagog. 1990, 8, 43–48. [Google Scholar] [CrossRef]
- Capaldo, T. The psychological effects on students of using animals in ways that they see as ethically, morally or religiously wrong. ATLA 2004, 32, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Gelberg, S.; Gelberg, H. Stress management interventions for veterinary students. J. Vet. Med Educ. 2005, 32, 173–181. [Google Scholar] [CrossRef]
- European Commission. 2019 Report on the Statistics on the Use of Animals for Scientific Purposes in the Member States of the European Union in 2015–2017; EC: Brussels, Belgium, 2020. [Google Scholar]
- Moore, R. Why I support dissection in science education. J. Appl. Anim. Welf. Sci. 2001, 4, 135–138. [Google Scholar] [CrossRef]
- Hedlund, C.S.; Hosgood, G.; Naugler, S. Surgical education: Attitudes toward animal use in teaching surgery at Louisiana State University. J. Vet. Med Educ. 2002, 29, 50–55. [Google Scholar] [CrossRef]
- Zemanova, M.A.; Bastos, L.; Knight, A.; Lybæk, S. Use of animals in education and training: Non-technical summaries reveal a reluctance to implement alternatives. ALTEX 2021. submitted. [Google Scholar]
- Patronek, G.J.; Rauch, A. Systematic review of comparative studies examining alternatives to the harmful use of animals in biomedical education. J. Am. Vet. Med. Assoc. 2007, 230, 37–43. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Bramer, W.M.; Rethlefsen, M.L.; Kleijnen, J.; Franco, O.H. Optimal database combinations for literature searches in systematic reviews: A prospective exploratory study. Syst. Rev. 2017, 6, 245. [Google Scholar] [CrossRef] [PubMed]
- Abutarbush, S.M.; Naylor, J.M.; Parchoma, G.; D’Eon, M.; Petrie, L.; Carruthers, T. Evaluation of traditional instruction versus a self-learning computer module in teaching veterinary students how to pass a nasogastric tube in the horse. J. Vet. Med. Educ. 2006, 33, 447–454. [Google Scholar] [CrossRef] [PubMed]
- Andreatta, P.B.; Klotz, J.J.; Dooley-Hash, S.L.; Hauptman, J.G.; Biddinger, B.; House, J.B.; Ahern, K. Performance-based comparison of neonatal intubation training outcomes. Adv. Neonatal Care 2015, 15, 56–64. [Google Scholar] [CrossRef]
- Carpenter, L.G.; Piermattei, D.L.; Salman, M.D.; Orton, E.C.; Nelson, A.W.; Smeak, D.D.; Jennings, P.B., Jr.; Taylor, R.A. A comparison of surgical training with live anesthetized dogs and cadavers. Vet. Surg. 1991, 20, 373–378. [Google Scholar] [CrossRef]
- Cronholm, J.-O.H. Dick Martenson, Tomas. Student attitudes towards laboratory exercises in medical biochemistry. Med. Teach. 2000, 22, 30–33. [Google Scholar] [CrossRef]
- Cross, T.R.; Cross, V.E. Scalpel or mouse? A statistical comparison of real and virtual frog dissections. Am. Biol. Teach. 2004, 66, 408–411. [Google Scholar] [CrossRef]
- Davy, R.B.; Hamel, P.E.; Su, Y.; Berry, C.R.; Conner, B.J. Evaluation of two training methods for teaching the abdominal focused assessment with sonography for trauma technique (A-FAST) to first- and second-year veterinary students. J. Vet. Med. Educ. 2019, 46, 258–263. [Google Scholar] [CrossRef]
- DeHoff, M.E.; Clark, K.L.; Meganathan, K. Learning outcomes and student-perceived value of clay modeling and cat dissection in undergraduate human anatomy and physiology. Adv. Physiol. Educ. 2011, 35, 68–75. [Google Scholar] [CrossRef]
- Dewhurst, D.G.; Brown, G.J.; Meehan, A.S. Microcomputer simulations of laboratory experiments in physiology. ATLA 1988, 15, 280–289. [Google Scholar] [CrossRef]
- Dewhurst, D.; Meehan, A. Evaluation of the use of computer-simulations of experiments in teaching undergraduate students. Br. J. Pharmacol. 1993, 108, P238. [Google Scholar]
- Dewhurst, D.G.; Hardcastle, J.; Hardcastle, P.T.; Stuart, E. Comparison of a computer simulation program and a traditional laboratory practical class for teaching the principles of intestinal absorption. Adv. Physiol. Educ. 1994, 267, S95. [Google Scholar] [CrossRef] [PubMed]
- Downie, R.; Meadows, J. Experience with a dissection opt-out scheme in university level biology. J. Biol. Educ. 1995, 29, 187–194. [Google Scholar] [CrossRef]
- Durand, M.d.T.; Restini, C.B.A.; Wolff, A.C.D.; Faria, M., Jr.; Couto, L.B.; Bestetti, R.B. Students’ perception of animal or virtual laboratory in physiology practical classes in PBL medical hybrid curriculum. Adv. Physiol. Educ. 2019, 43, 451–457. [Google Scholar] [CrossRef]
- Eichel, J.-C.; Korb, W.; Schlenker, A.; Bausch, G.; Brehm, W.; Delling, U. Evaluation of a training model to teach veterinary students a technique for injecting the jugular vein in horses. J. Vet. Med. Educ. 2013, 40, 288–295. [Google Scholar] [CrossRef]
- Fančovičova, J.; Prokop, P. The effects of 3D plastic models of animals and cadaveric dissection on students’ perceptions of the internal organs of animals. J. Balt. Sci. Educ. 2014, 13, 767–775. [Google Scholar]
- Fowler, H.S.; Brosius, E.J. A research study on the values gained from dissection of animals in secondary school biology. Sci. Educ. 1968, 52, 55–57. [Google Scholar] [CrossRef]
- González Guevara, M.C.; Losada Camacho, M.; Garavito Cárdenas, G. Pharmacy students opinion about the implementation of computer based alternatives on experimental pharmacology learning-teaching process. Rev. Fac. Med. 2008, 56, 43–53. [Google Scholar]
- Greenfield, C.L.; Johnson, A.L.; Smith, C.W.; Marretta, S.M.; Farmer, J.A.; Klippert, L. Integrating alternative models into the existing surgical curriculum. J. Vet. Med. Educ. 1994, 21, 23–24. [Google Scholar]
- Greenfield, C.L.; Johnson, A.L.; Schaeffer, D.J.; Hungerford, L.L. Comparison of surgical skills of veterinary students trained using models or live animals. J. Am. Vet. Med. Assoc. 1995, 206, 1840–1845. [Google Scholar]
- Griffon, D.J.; Cronin, P.; Kirby, B.; Cottrell, D.F. Evaluation of a hemostasis model for teaching ovariohysterectomy in veterinary surgery. Vet. Surg. 2000, 29, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.; Andrew, B. Randomized objective comparison of live tissue training versus simulators for emergency procedures. Am. Surg. 2011, 77, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Hall, A.B.; Riojas, R.; Sharon, D. Comparison of self-efficacy and its improvement after artificial simulator or live animal model emergency procedure training. Mil. Med. 2014, 179, 320–323. [Google Scholar] [CrossRef] [PubMed]
- Haspel, C.; Motoike, H.K.; Lenchner, E. The implementation of clay modeling and rat dissection into the human anatomy and physiology curriculum of a large urban community college. Anat. Sci. Educ. 2014, 7, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.; Tillo, N.; Kirlum, H.-J.; Till, H. Comparison of different training models for laparoscopic surgery in neonates and small infants. Surg. Endosc. Other Interv. Tech. 2006, 20, 641–644. [Google Scholar] [CrossRef] [PubMed]
- Hughes, I.E. Do computer simulations of laboratory practicals meet learning needs? Trends Pharmacol. Sci. 2001, 22, 71–74. [Google Scholar] [CrossRef]
- Iverson, K.; Riojas, R.; Sharon, D.; Hall, A.B. Objective comparison of animal training versus artificial simulation for initial cricothyroidotomy training. Am. Surg. 2015, 81, 515–518. [Google Scholar] [CrossRef]
- Kinzie, M.B.; Strauss, R.; Foss, J. The effects of an interactive dissection simulation on the performance and achievement of high school biology students. J. Res. Sci. Teach. 1993, 30, 989–1000. [Google Scholar] [CrossRef]
- Lalley, J.P.; Piotrowski, P.S.; Battaglia, B.; Brophy, K.; Chugh, K. A comparison of V-Frog© to physical frog dissection. Int. J. Environ. Sci. Educ. 2010, 5, 189–200. [Google Scholar]
- Lombardi, S.A.; Hicks, R.E.; Thompson, K.V.; Marbach-Ad, G. Are all hands-on activities equally effective? Effect of using plastic models, organ dissections, and virtual dissections on student learning and perceptions. Adv. Physiol. Educ. 2014, 38, 80–86. [Google Scholar] [CrossRef]
- Matthews, D. Comparison of MacPig to fetal pig dissection in college biology. Am. Biol. Teach. 1998, 60, 228–229. [Google Scholar] [CrossRef]
- McCool, K.E.; Bissett, S.A.; Hill, T.L.; Degernes, L.A.; Hawkins, E.C. Evaluation of a human virtual-reality endoscopy trainer for teaching early endoscopy skills to veterinarians. J. Vet. Med. Educ. 2020, 47, 106–116. [Google Scholar] [CrossRef] [PubMed]
- More, D.; Ralph, C.L. A test of effectiveness of courseware in a college biology class. J. Educ. Technol. Syst. 1992, 21, 79–84. [Google Scholar] [CrossRef]
- Motoike, H.K.; O’Kane, R.L.; Lenchner, E.; Haspel, C. Clay modeling as a method to learn human muscles: A community college study. Anat. Sci. Educ. 2009, 2, 19–23. [Google Scholar] [CrossRef]
- Mouraviev, V.; Klein, M.; Schommer, E.; Thiel, D.D.; Samavedi, S.; Kumar, A.; Leveillee, R.J.; Thomas, R.; Pow-Sang, J.M.; Su, L.M.; et al. Urology residents experience comparable workload profiles when performing live porcine nephrectomies and robotic surgery virtual reality training modules. J. Robot. Surg. 2016, 10, 49–56. [Google Scholar] [CrossRef]
- Nagel, C.; Ille, N.; Aurich, J.; Aurich, C. Teaching of diagnostic skills in equine gynecology: Simulator-based training versus schooling on live horses. Theriogenology 2015, 84, 1088–1095. [Google Scholar] [CrossRef]
- Olsen, D.; Bauer, M.S.; Seim, H.B.; Salman, M.D. Evaluation of a hemostasis model for teaching basic surgical skills. Vet. Surg. 1996, 25, 49–58. [Google Scholar] [CrossRef]
- Pavletic, M.M.; Schwartz, A.; Berg, J.; Knapp, D. An assessment of the outcome of the alternative medical and surgical laboratory program at Tufts University. J. Am. Vet. Med. Assoc. 1994, 205, 97–100. [Google Scholar]
- Predavec, M. Evaluation of E-Rat, a computer-based rat dissection, in terms of student learning outcomes. J. Biol. Educ. 2001, 35, 75–80. [Google Scholar] [CrossRef]
- Samsel, R.W.; Schmidt, G.A.; Hall, J.B.; Wood, L.; Shroff, S.G.; Schumacker, P.T. Cardiovascular physiology teaching: Computer simulations vs. animal demonstrations. Adv. Physiol. Educ. 1994, 266, S36. [Google Scholar] [CrossRef]
- Smeak, D.D.; Hill, L.N.; Beck, M.L.; Shaffer, C.A.; Birchard, S.J. Evaluation of an autotutorial-simulator program for instruction of hollow organ closure. Vet. Surg. 1994, 23, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Strauss, R.T.; Kinzie, M.B. Student achievement & attitudes in a pilot study comparing an interactive videodisc simulation to conventional dissection. Am. Biol. Teach. 1994, 56, 398–402. [Google Scholar]
- Theoret, C.L.; Carmel, É.-N.; Bernier, S. Why dissection videos should not replace cadaver prosections in the gross veterinary anatomy curriculum: Results from a comparative study. J. Vet. Med. Educ. 2007, 34, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Liu, C.Y.; Ma, T. Evaluation of a virtual neurophysiology laboratory as a new pedagogical tool for medical undergraduate students in China. Adv. Physiol. Educ. 2018, 42, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Waters, J.R.; Van Meter, P.; Perrotti, W.; Drogo, S.; Cyr, R.J. Cat dissection vs. sculpting human structures in clay: An analysis of two approaches to undergraduate human anatomy laboratory education. Adv. Physiol. Educ. 2005, 29, 27–34. [Google Scholar] [CrossRef][Green Version]
- Waters, J.R.; Van Meter, P.; Perrotti, W.; Drogo, S.; Cyr, R.J. Human clay models versus cat dissection: How the similarity between the classroom and the exam affects student performance. Adv. Physiol. Educ. 2011, 35, 227–236. [Google Scholar] [CrossRef]
- Winder, C.B.; LeBlanc, S.J.; Haley, D.B.; Lissemore, K.D.; Godkin, M.A.; Duffield, T.F. Comparison of online, hands-on, and a combined approach for teaching cautery disbudding technique to dairy producers. J. Dairy Sci. 2018, 101, 840–849. [Google Scholar] [CrossRef]
- Ritskes-Hoitinga, M.; van Luijk, J. How can systematic reviews teach us more about the implementation of the 3Rs and animal welfare? Animals 2019, 9, 1163. [Google Scholar] [CrossRef]
- Valliyate, M.; Robinson, N.G.; Goodman, J.R. Current concepts in simulation and other alternatives for veterinary education: A review. Vet. Med. 2012, 57, 325–337. [Google Scholar] [CrossRef]
- Badash, I.; Burtt, K.; Solorzano, C.A.; Carey, J.N. Innovations in surgery simulation: A review of past, current and future techniques. Ann. Transl. Med. 2016, 4, 453. [Google Scholar] [CrossRef]
- Simkin, D.J.; Greene, J.A.; Jung, J.; Sacks, B.C.; Fessler, H.E. The death of animals in medical school. N. Engl. J. Med. 2017, 376, 713–715. [Google Scholar] [CrossRef] [PubMed]
- Zevin, B.; Aggarwal, R.; Grantcharov, T.P. Surgical simulation in 2013: Why is it still not the standard in surgical training? J. Am. Coll. Surg. 2014, 218, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Knight, A. The Costs and Benefits of Animal Experiments; Palgrave Macmillan: London, UK, 2011; p. 254. [Google Scholar]
- Martinsen, S. Training the animal doctor: Caring as a clinical skill. Altern. Anim. Test. Exp. 2007, 12, 155–159. [Google Scholar]
- Paul, E.S.; Podberscek, A.L. Veterinary education and students’ attitudes towards animal welfare. Vet. Rec. 2000, 146, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Hellyer, P.W.; Frederick, C.; Lacy, M.; Salman, M.D.; Wagner, A.E. Attitudes of veterinary medical students, house officers, clinical faculty, and staff toward pain management in animals. J. Am. Vet. Med. Assoc. 1999, 214, 238–244. [Google Scholar]
- Self, D.J.; Schrader, D.E.; Baldwin, J.D.C.; Root, S.K.; Wolinsky, F.D.; Shadduck, J.A. Study of the influence of veterinary medical education on the moral development of veterinary students. J. Am. Vet. Med. Assoc. 1991, 198, 782–787. [Google Scholar]
- Arluke, A.; Hafferty, F. From apprehension to fascination with ’’dog lab’’: The use of absolutions by medical students. J. Contemp. Ethnogr. 1996, 25, 201–225. [Google Scholar] [CrossRef]
- Sobani, Z.A.; Ahmed, N.; Bhamani, A.; Sharif, H.; Beg, M.A. Desensitisation of medical students towards the dying. Med. Educ. 2010, 44, 1152. [Google Scholar] [CrossRef]
- Pereira, G.D.; Dieguez, J.; Demirbas, Y.S.; Menache, A. Alternatives to animal use in veterinary education: A growing debate. Ank. Univ. Vet. Fak Derg. 2017, 64, 235–239. [Google Scholar]
- Kumar, A.M.; Murtaugh, R.; Brown, D.; Ballas, T.; Clancy, E.; Patronek, G. Client donation program for acquiring dogs and cats to teach veterinary gross anatomy. J. Vet. Med. Educ. 2001, 28, 73–77. [Google Scholar] [CrossRef]
- Fearon, R. Cadaver donation programme boosts US veterinary education. Vet. Times 2005, 35, 8–9. [Google Scholar]
- Knight, A. The potential of humane teaching methods within veterinary and other biomedical education. ALTEX Proc. 2012, 1, 365–375. [Google Scholar]
- Baillie, S.; Crossan, A.; Reid, S.; Brewster, S. Preliminary development and evaluation of a bovine rectal palpation simulator for training veterinary students. Cattle Pract. 2003, 11, 101–106. [Google Scholar]
- Baillie, S.; Crossan, A.; Brewster, S.A.; Mellor, D.; Reid, S. Validation of a bovine rectal palpation simulator for training veterinary students. Stud. Health Technol. Inform. 2005, 111, 33–36. [Google Scholar]
- Baillie, S.; Mellor, D.J.; Brewster, S.A.; Reid, S.W.J. Integrating a bovine rectal palpation simulator into an undergraduate veterinary curriculum. J. Vet. Med. Educ. 2005, 32, 79–85. [Google Scholar] [CrossRef]
- Bastos, J.C.F.; Rangel, A.M.; Paixão, R.L.; Rego, S. Implicações éticas do uso de animais no processo de ensino-aprendizagem nas faculdades de medicina do Rio de Janeiro e Niterói. Rev. Bras. Educ. Médica 2002, 26, 162–170. [Google Scholar]
- Balcombe, J. Student/teacher conflict regarding animal dissection. Am. Biol. Teach. 1997, 59, 22–25. [Google Scholar] [CrossRef]
- King, L.A.; Ross, C.L.; Stephens, M.L.; Rowan, A.N. Biology teachers’ attitudes to dissection and alternatives. ATLA 2004, 32, 475–484. [Google Scholar] [CrossRef]
- van der Valk, J.; Dewhurst, D.; Hughes, I.; Atkinson, J.; Balcombe, J.; Braun, H.; Gabrielson, K.; Gruber, F.; Miles, J.; Nab, J.; et al. Alternatives to the use of animals in higher education—The report and recommendations of ECVAM Workshop 33. ATLA 1999, 27, 39–52. [Google Scholar]
- Bones, V.C.; Clemente, H.C.; Weary, D.M.; Molento, C.F.M. Perceived barriers to the adoption of alternatives to laboratory animal use for rabies diagnosis. ATLA 2014, 42, 171–179. [Google Scholar] [CrossRef]
- Grigg, E.; Hart, L.A.; Moffett, J. Comparison of the effects of clay modeling & cat cadaver dissection on high school students’ outcomes & attitudes in a human anatomy course. Am. Biol. Teach. 2020, 82, 596–605. [Google Scholar]
| Study | Country | Discipline | Level | Species | Humane Method | Students: Humane Method (Total) | Assessment of Learning Outcome | Learning Outcome of the Humane method |
|---|---|---|---|---|---|---|---|---|
| Abutarbush et al., 2006 [26] | Canada | Veterinary medicine—other procedures | Tertiary | Horse | Computer simulation | 27 (52) | Exam; task performance | Superior |
| Andreatta et al., 2015 [27] | USA | Human medicine—other procedures | Tertiary | Cat | Mannequin | 167 (294) | Exam; task performance | Equivalent |
| Botden et al., 2010 [9] | Nether-lands | Human medicine—surgical skills | Tertiary | Pig | Model | 20 (20) | Task performance | Equivalent |
| Carpenter et al., 1991 [28] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Ethically sourced cadaver | 12 (24) | Task performance | Equivalent |
| Clarke 1987 [7] | UK | Physiology | Tertiary | Frog | Computer simulation | 15 (28) | Exam | Equivalent |
| Cronholm 2000 [29] | Sweden | Physiology | Tertiary | Rat | Self-experimentation | 94 (133) | Exam | Equivalent |
| Cross and Cross 2004 [30] | USA | Animal anatomy | Secondary | Frog | Computer simulation | 38 (74) | Exam | Inferior |
| Davy et al., 2019 [31] | USA | Veterinary medicine—other procedures | Tertiary | Dog | Video | 19 (38) | Task performance | Equivalent |
| DeHoff et al., 2011 [32] | USA | Human anatomy | Tertiary | Cat | Clay sculpting exercise | 88 (193) | Exam | Equivalent |
| Dewhurst et al., 1988 [33] | UK | Physiology | Tertiary | Frog | Computer simulation | 66 (112) | Exam | Equivalent |
| Dewhurst and Meehan 1993 [34] | UK | Pharmacology | Tertiary | NA | Computer simulation | NA (65) | Exam | Equivalent |
| Dewhurst et al., 1994 [35] | USA | Physiology | Tertiary | Rat | Computer simulation | 6 (14) | Exam | Equivalent |
| Downie and Meadows 1995 [36] | UK | Animal anatomy | Tertiary | Rat | Model | 308 (2913) | Exam | Equivalent |
| Durand et al., 2019 [37] | Brazil | Physiology | Tertiary | Rat | Video | 108 (350) | Exam | Equivalent |
| Eichel et al., 2013 [38] | Germ-any | Veterinary medicine—other procedures | Tertiary | Horse | Model | 12 (24) | Task performance | Superior |
| Fančovičova and Prokop 2014 [39] | Slovakia | Animal anatomy | Tertiary | Fish, rat | Model | 15 (46) | Exam | Equivalent |
| Fawver et al., 1990 [10] | USA | Physiology | Tertiary | Dog | Computer simulation | 18–24 (85) | Exam | Equivalent |
| Fowler and Brosius 1968 [40] | USA | Animal anatomy | Secondary | Earthworm, crayfish, frog, fish | Video | NA (156) | Exam | Superior |
| González Guevara et al., 2008 [41] | Colom-bia | Pharmacology | Tertiary | Guinea-pig | Computer simulation | 73 | Perceived clarity of information | Equivalent |
| Greenfield et al., 1994 [42] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Model | 36 (36) | Task performance | Equivalent |
| Greenfield et al., 1995 [43] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Model | 18 (36) | Task performance | Equivalent |
| Griffon et al., 2000 [44] | UK | Veterinary medicine—surgical skills | Tertiary | Dog | Model | 20 (40) | Task performance | Superior |
| Hall and Andrew 2011 [45] | USA | Human medicine—other procedures | Tertiary | Pig | Mechanical simulator | 12 (24) | Task performance | Equivalent |
| Hall et al., 2014 [46] | USA | Human medicine—other procedures | Tertiary | Pig | Mechanical simulator | 23 (101) | Perceived self-efficacy | Equivalent |
| Haspel et al., 2014 [47] | USA | Human anatomy | Tertiary | Cat, rat | Clay sculpting exercise | 304 (747) | Exam | Superior |
| Heinrich et al., 2006 [48] | Germ-any | Human medicine—surgical skills | Tertiary | Rabbit | Model | 6 (12) | Task performance | Equivalent |
| Hughes 2001 [49] | UK | Pharmacology | Tertiary | Guinea-pig | Computer simulation | 66 (112) | Exam | Equivalent |
| Iverson et al., 2015 [50] | USA | Human medicine—other procedures | Tertiary | Pig | Mechanical simulator | 33 (69) | Task performance | Equivalent |
| Kinzie et al., 1993 [51] | USA | Animal anatomy | Secondary | Frog | Computer simulation | 15 (61) | Exam | Equivalent |
| Lalley et al., 2010 [52] | USA | Animal anatomy | Secondary | Frog | Virtual reality simulation | 51 (102) | Exam | Superior |
| Leathard and Dewhurst 1995 [8] | UK | Physiology | Tertiary | Rat | Computer simulation | 70–80 (156) | Exam | Equivalent |
| Leonard 1992 [11] | USA | Biology | Tertiary | Frog, mouse | Computer simulation | 70 (142) | Exam | Equivalent |
| Lombardi 2014 [53] | USA | Human anatomy | Tertiary | Sheep heart | Model | 16 (29) | Exam | Superior |
| Matthews 1998 [54] | USA | Animal anatomy | Secondary | Pig | Computer simulation | 12 (20) | Exam | Inferior |
| McCool et al., 2020 [55] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Virtual reality simulation | 6 (12) | Task performance | Equivalent |
| More and Ralph 1992 [56] | USA | Biology | Tertiary | NA | Computer simulation | 93 (184) | Exam | Superior |
| Motoike et al., 2009 [57] | USA | Human anatomy | Tertiary | Cat | Clay sculpting exercise | NA (181) | Exam | Superior |
| Mouraviev et al., 2016 [58] | USA | Human medicine—surgical skills | Tertiary | Pig | Virtual reality simulation | 10–11 (21) | Task performance | Equivalent |
| Nagel et al., 2015 [59] | Austria | Veterinary medicine—other procedures | Tertiary | Horse | Mechanical simulator | 8 (25) | Task performance | Superior |
| Olsen et al., 1996 [60] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Model | 20 (40) | Task performance | Superior |
| Pavletic et al., 1994 [61] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Ethically sourced cadaver | 12 (48) | Skills assessment by employers | Equivalent |
| Predavec 2001 [62] | Austra-lia | Animal anatomy | Tertiary | Rat | Computer simulation | 233 (401) | Exam | Superior |
| Samsel et al., 1994 [63] | USA | Physiology | Tertiary | Dog | Computer simulation | 110 | Perceived usefulness of the teaching | Superior |
| Smeak et al., 1994 [64] | USA | Veterinary medicine—surgical skills | Tertiary | Dog | Model | 20 (40) | Task performance | Inferior |
| Strauss and Kinzie 1994 [65] | USA | Animal anatomy | Secondary | Frog | Computer simulation | 9 (20) | Exam | Equivalent |
| Theoret et al., 2007 [66] | Canada | Animal anatomy | Tertiary | Cow | Video | 37 (75) | Exam | Inferior |
| Wang et al., 2018 [67] | China | Physiology | Tertiary | Frog | Computer simulation | 18 (23) | Exam | Inferior |
| Waters et al., 2005 [68] | USA | Human anatomy | Tertiary | Cat | Clay sculpting exercise | 60 (136) | Exam | Superior |
| Waters et al., 2011 [69] | USA | Human anatomy | Tertiary | Cat | Clay sculpting exercise | 75 (196) | Exam | Equivalent |
| Winder et al., 2018 [70] | Canada | Veterinary medicine—other procedures | Tertiary | Cow | Online learning | 23 (43) | Task performance | Equivalent |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zemanova, M.A.; Knight, A. The Educational Efficacy of Humane Teaching Methods: A Systematic Review of the Evidence. Animals 2021, 11, 114. https://doi.org/10.3390/ani11010114
Zemanova MA, Knight A. The Educational Efficacy of Humane Teaching Methods: A Systematic Review of the Evidence. Animals. 2021; 11(1):114. https://doi.org/10.3390/ani11010114
Chicago/Turabian StyleZemanova, Miriam A., and Andrew Knight. 2021. "The Educational Efficacy of Humane Teaching Methods: A Systematic Review of the Evidence" Animals 11, no. 1: 114. https://doi.org/10.3390/ani11010114
APA StyleZemanova, M. A., & Knight, A. (2021). The Educational Efficacy of Humane Teaching Methods: A Systematic Review of the Evidence. Animals, 11(1), 114. https://doi.org/10.3390/ani11010114






