Physiology of Methylotrophs Living in the Phyllosphere
Abstract
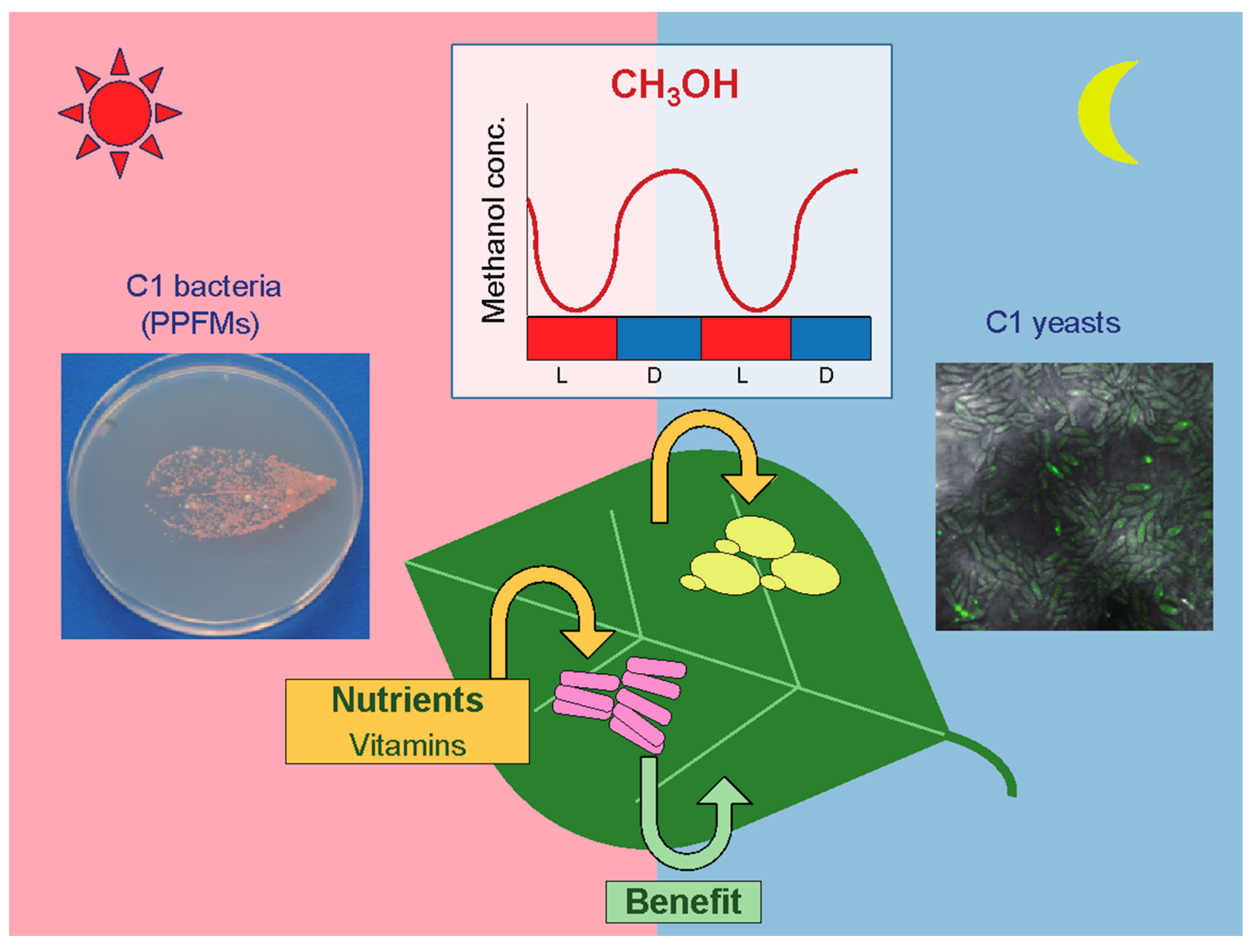
1. Introduction
2. Physiology of Methylotrophic Yeasts in the Phyllosphere
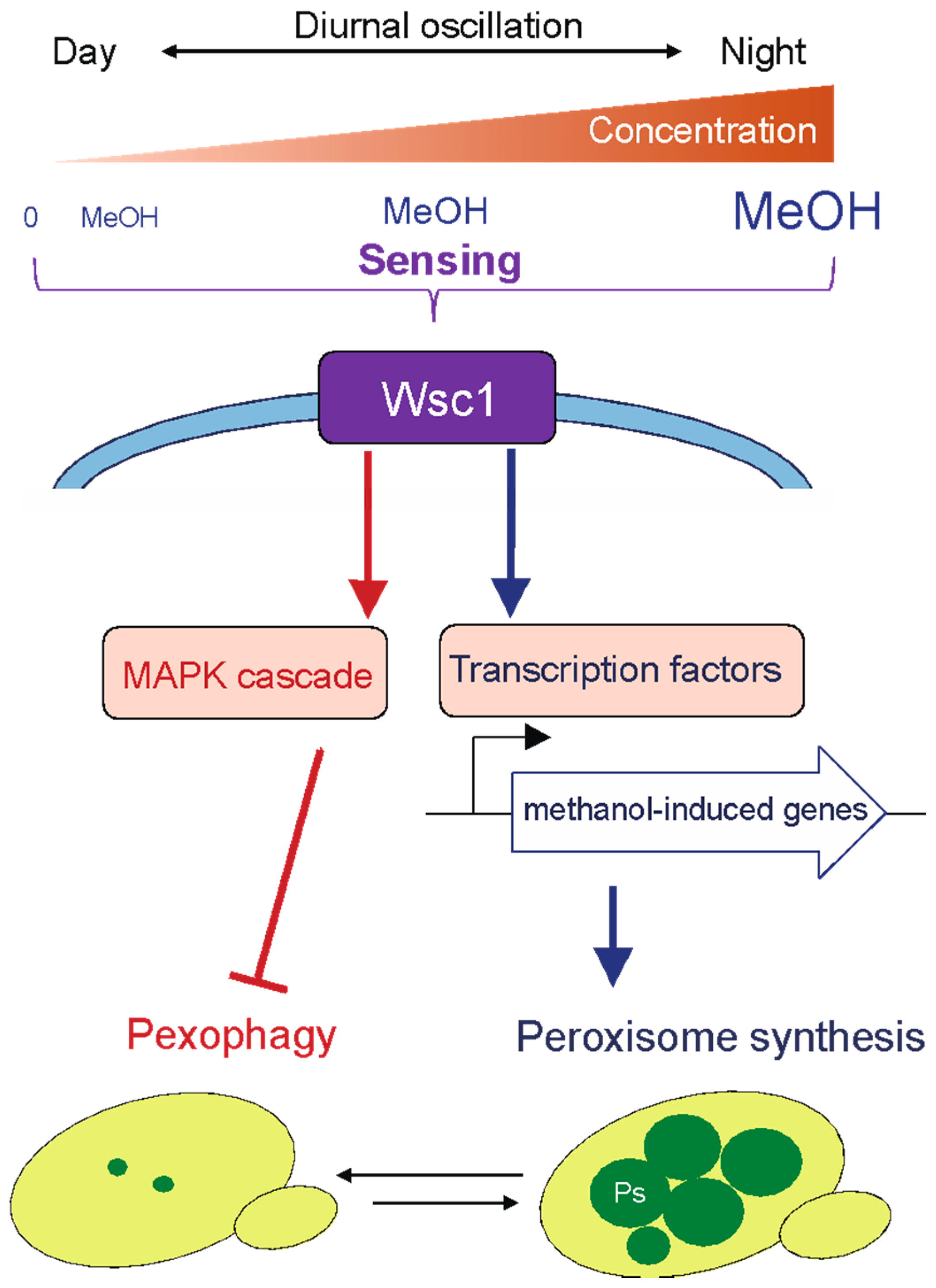
2.1. Proliferation of Methylotrophic Yeasts on Plant Leaves Where Methanol Concentrations Fluctuate Diurnally
2.2. Molecular and Cellular Mechanisms of Adaptation to the Phyllosphere Environment in Methylotrophic Yeasts
3. Ubiquitous Colonization of PPFMs on the Surface of Plants and Species-Level Specific Interactions
3.1. Distribution of Methane- and Methanol-Utilizing Bacteria in the Phyllosphere
3.2. Species Level Specificity between PPFMs and Plants
3.3. Pantothenate Auxotrophy of Methylobacterium sp. OR01 and Fitness Advantage in the Phyllosphere Environment
4. Survival Strategy to Adapt to Various Environmental Stresses in the Phyllosphere
4.1. General Stress Response Regulator PhyR in Methylotrophs
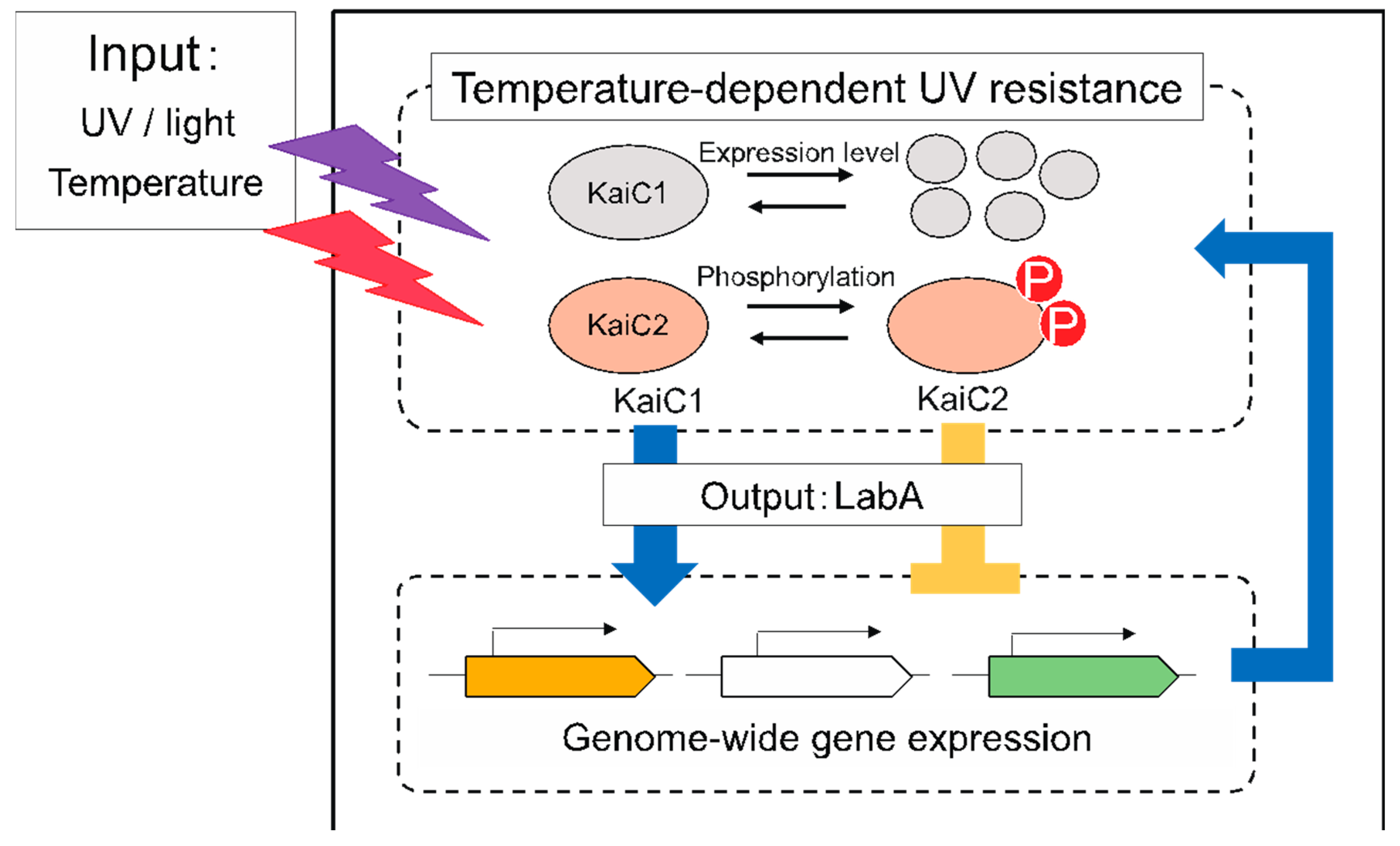
4.2. Role of KaiC Family Proteins in M. Extorquens AM1
5. Improvement of Crop Yield by PPFMs
6. Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fall, R.; Benson, A.A. Leaf methanol—The simplest natural product from plants. Trends Plant Sci. 1996, 1, 296–301. [Google Scholar] [CrossRef]
- Dorokhov, Y.L.; Sheshukova, E.V.; Komarova, T.V. Methanol in plant life. Front. Plant Sci. 2018, 9, 1623. [Google Scholar] [CrossRef] [PubMed]
- Galbally, I.E.; Kirstine, W. The production of methanol by flowering plants and the global cycle of methanol. J. Atmos. Chem. 2002, 43, 195–229. [Google Scholar] [CrossRef]
- Henrot, A.J.; Stanelle, T.; Schroder, S.; Siegenthaler, C.; Taraborrelli, D.; Schultz, M.G. Implementation of the MEGAN (v2.1) biogenic emission model in the ECHAM6-HAMMOZ chemistry climate model. Geosci. Model. Dev. 2017, 10, 903–926. [Google Scholar] [CrossRef]
- Nemecek-Marshall, M.; Macdonald, R.C.; Franzen, F.J.; Wojciechowski, C.L.; Fall, R. Methanol emission from leaves: Enzymatic detection of gas-phase methanol and relation of methanol fluxes to stomatal conductance and leaf development. Plant Physiol. 1995, 108, 1359–1368. [Google Scholar] [CrossRef]
- Kawaguchi, K.; Yurimoto, H.; Oku, M.; Sakai, Y. Yeast methylotrophy and autophagy in a methanol-oscillating environment on growing Arabidopsis thaliana leaves. PLoS ONE 2011, 6, e25257. [Google Scholar] [CrossRef]
- Yurimoto, H.; Sakai, Y. Methylotrophic yeasts: Current understanding of their C1-metabolism and its regulation by sensing methanol for survival on plant leaves. Curr. Issues Mol. Biol. 2019, 33, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Lindow, S.E.; Brandl, M.T. Microbiology of the phyllosphere. Appl. Environ. Microbiol. 2003, 69, 1875–1883. [Google Scholar] [CrossRef]
- Vorholt, J.A. Microbial life in the phyllosphere. Nat. Rev. Microbiol. 2012, 10, 828–840. [Google Scholar] [CrossRef]
- Trivedi, P.; Leach, J.E.; Tringe, S.G.; Sa, T.M.; Singh, B.K. Plant-microbiome interactions: From community assembly to plant health. Nat. Rev. Microbiol. 2020, 18, 607–621. [Google Scholar] [CrossRef]
- Knief, C.; Ramette, A.; Frances, L.; Alonso-Blanco, C.; Vorholt, J.A. Site and plant species are important determinants of the Methylobacterium community composition in the plant phyllosphere. ISME J. 2010, 4, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Knief, C.; Delmotte, N.; Chaffron, S.; Stark, M.; Innerebner, G.; Wassmann, R.; von Mering, C.; Vorholt, J.A. Metaproteogenomic analysis of microbial communities in the phyllosphere and rhizosphere of rice. ISME J. 2012, 6, 1378–1390. [Google Scholar] [CrossRef]
- Green, P.N.; Ardley, J.K. Review of the genus Methylobacterium and closely related organisms: A proposal that some Methylobacterium species be reclassified into a new genus, Methylorubrum gen. nov. Int. J. Syst. Evol. Microbiol. 2018, 68, 2727–2748. [Google Scholar] [CrossRef]
- Dourado, M.N.; Camargo Neves, A.A.; Santos, D.S.; Araujo, W.L. Biotechnological and agronomic potential of endophytic pink-pigmented methylotrophic Methylobacterium spp. BioMed Res. Int. 2015, 2015, 909016. [Google Scholar] [CrossRef]
- Kumar, M.; Tomar, R.S.; Lade, H.; Paul, D. Methylotrophic bacteria in sustainable agriculture. World J. Microbiol. Biotechnol. 2016, 32, 120. [Google Scholar] [CrossRef] [PubMed]
- Yurimoto, H. Molecular basis of methanol-inducible gene expression and its application in the methylotrophic yeast Candida boidinii. Biosci. Biotechnol. Biochem. 2009, 73, 793–800. [Google Scholar] [CrossRef] [PubMed]
- Yurimoto, H.; Oku, M.; Sakai, Y. Yeast methylotrophy: Metabolism, gene regulation and peroxisome homeostasis. Int. J. Microbiol. 2011, 2011, 101298. [Google Scholar] [CrossRef]
- Baghban, R.; Farajnia, S.; Rajabibazl, M.; Ghasemi, Y.; Mafi, A.; Hoseinpoor, R.; Rahbarnia, L.; Aria, M. Yeast expression systems: Overview and recent advances. Mol. Biotechnol. 2019, 61, 365–384. [Google Scholar] [CrossRef]
- Karbalaei, M.; Rezaee, S.A.; Farsiani, H. Pichia pastoris: A highly successful expression system for optimal synthesis of heterologous proteins. J. Cell. Physiol. 2020, 235, 5867–5881. [Google Scholar] [CrossRef]
- Aulakh, M.S.; Wassmann, R.; Rennenberg, H.; Fink, S. Pattern and amount of aerenchyma relate to variable methane transport capacity of different rice cultivars. Plant Biol. 2000, 2, 182–194. [Google Scholar] [CrossRef]
- Bruhn, D.; Moller, I.M.; Mikkelsen, T.N.; Ambus, P. Terrestrial plant methane production and emission. Physiol. Plant 2012, 144, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, H.; Sato, I.; Sakakibara, M.; Yurimoto, H.; Sakai, Y. Distribution of methanotrophs in the phyllosphere. Biosci. Biotechnol. Biochem. 2012, 76, 1580–1583. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Iguchi, H.; Yurimoto, H.; Murakami, A.; Sakai, Y. Aquatic plant surface as a niche for methanotrophs. Front. Microbiol. 2014, 5, 30. [Google Scholar] [CrossRef]
- Stepniewska, Z.; Goraj, W.; Kuzniar, A.; Lopacka, N.; Malysza, M. Enrichment culture and identification of endophytic methanotrophs isolated from peatland plants. Folia Microbiol. 2017, 62, 381–391. [Google Scholar] [CrossRef][Green Version]
- Frindte, K.; Maarastawi, S.A.; Lipski, A.; Hamacher, J.; Knief, C. Characterization of the first rice paddy cluster I isolate, Methyloterricola oryzae gen. nov., sp nov and amended description of Methylomagnum ishizawai. Int. J. Syst. Evol. Microbiol. 2017, 67, 4507–4514. [Google Scholar] [CrossRef]
- Peng, X.J.; Xie, J.; Li, W.Z.; Xie, H.W.; Cai, Y.H.; Ding, X. Comparison of wild rice (Oryza longistaminata) tissues identifies rhizome-specific bacterial and archaeal endophytic microbiomes communities and network structures. PLoS ONE 2021, 16, e0246687. [Google Scholar] [CrossRef]
- Sy, A.; Timmers, A.C.; Knief, C.; Vorholt, J.A. Methylotrophic metabolism is advantageous for Methylobacterium extorquens during colonization of Medicago truncatula under competitive conditions. Appl. Environ. Microbiol. 2005, 71, 7245–7252. [Google Scholar] [CrossRef]
- Schmidt, S.; Christen, P.; Kiefer, P.; Vorholt, J.A. Functional investigation of methanol dehydrogenase-like protein XoxF in Methylobacterium extorquens AM1. Microbiology 2010, 156, 2575–2586. [Google Scholar] [CrossRef]
- Shiraishi, K.; Oku, M.; Kawaguchi, K.; Uchida, D.; Yurimoto, H.; Sakai, Y. Yeast nitrogen utilization in the phyllosphere during plant lifespan under regulation of autophagy. Sci. Rep. 2015, 5, 9719. [Google Scholar] [CrossRef]
- Ohsawa, S.; Yurimoto, H.; Sakai, Y. Novel function of Wsc proteins as a methanol-sensing machinery in the yeast Pichia pastoris. Mol. Microbiol. 2017, 104, 349–363. [Google Scholar] [CrossRef]
- Ohsawa, S.; Inoue, K.; Isoda, T.; Oku, M.; Yurimoto, H.; Sakai, Y. Methanol sensor Wsc1 and MAP kinase suppress degradation of methanol-induced peroxisomes in methylotrophic yeast. J. Cell Sci. 2021, in press. [Google Scholar] [CrossRef]
- Yang, C.H.; Crowley, D.E.; Borneman, J.; Keen, N.T. Microbial phyllosphere populations are more complex than previously realized. Proc. Natl. Acad. Sci. USA 2001, 98, 3889–3894. [Google Scholar] [CrossRef]
- Delmotte, N.; Knief, C.; Chaffron, S.; Innerebner, G.; Roschitzki, B.; Schlapbach, R.; von Mering, C.; Vorholt, J.A. Community proteogenomics reveals insights into the physiology of phyllosphere bacteria. Proc. Natl. Acad. Sci. USA 2009, 106, 16428–16433. [Google Scholar] [CrossRef]
- Finkel, O.M.; Burch, A.Y.; Lindow, S.E.; Post, A.F.; Belkin, S. Geographical location determines the population structure in phyllosphere microbial communities of a salt-excreting desert tree. Appl. Environ. Microbiol. 2011, 77, 7647–7655. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, H.; Umeda, R.; Taga, H.; Oyama, T.; Yurimoto, H.; Sakai, Y. Community composition and methane oxidation activity of methanotrophs associated with duckweeds in a fresh water lake. J. Biosci. Bioeng. 2019, 128, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, M.; Yurimoto, H.; Yoshida, N.; Iguchi, H.; Sakai, Y. Distribution of pink-pigmented facultative methylotrophs on leaves of vegetables. Biosci. Biotechnol. Biochem. 2012, 76, 578–580. [Google Scholar] [CrossRef]
- Mizuno, M.; Yurimoto, H.; Iguchi, H.; Tani, A.; Sakai, Y. Dominant colonization and inheritance of Methylobacterium sp. strain OR01 on perilla plants. Biosci. Biotechnol. Biochem. 2013, 77, 1533–1538. [Google Scholar] [CrossRef]
- Yoshida, Y.; Iguchi, H.; Sakai, Y.; Yurimoto, H. Pantothenate auxotrophy of Methylobacterium spp. isolated from living plants. Biosci. Biotechnol. Biochem. 2019, 83, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Gourion, B.; Rossignol, M.; Vorholt, J.A. A proteomic study of Methylobacterium extorquens reveals a response regulator essential for epiphytic growth. Proc. Natl. Acad. Sci. USA 2006, 103, 13186–13191. [Google Scholar] [CrossRef]
- Gourion, B.; Francez-Charlot, A.; Vorholt, J.A. PhyR is involved in the general stress response of Methylobacterium extorquens AM1. J. Bacteriol. 2008, 190, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Gourion, B.; Sulser, S.; Frunzke, J.; Francez-Charlot, A.; Stiefel, P.; Pessi, G.; Vorholt, J.A.; Fischer, H.M. The PhyR-sEcfG signalling cascade is involved in stress response and symbiotic efficiency in Bradyrhizobium japonicum. Mol. Microbiol. 2009, 73, 291–305. [Google Scholar] [CrossRef] [PubMed]
- Francez-Charlot, A.; Kaczmarczyk, A.; Fischer, H.M.; Vorholt, J.A. The general stress response in Alphaproteobacteria. Trends Microbiol. 2015, 23, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Iguchi, H.; Sato, I.; Yurimoto, H.; Sakai, Y. Stress resistance and C1 metabolism involved in plant colonization of a methanotroph Methylosinus sp. B4S. Arch. Microbiol. 2013, 195, 717–726. [Google Scholar] [CrossRef]
- Iguchi, H.; Yoshida, Y.; Fujisawa, K.; Taga, H.; Yurimoto, H.; Oyama, T.; Sakai, Y. KaiC family proteins integratively control temperature-dependent UV resistance in Methylobacterium extorquens AM1. Environ. Microbiol. Rep. 2018, 10, 634–643. [Google Scholar] [CrossRef]
- Johnson, C.H.; Mori, T.; Xu, Y. A cyanobacterial circadian clockwork. Curr. Biol. 2008, 18, R816–R825. [Google Scholar] [CrossRef]
- Johnson, C.H.; Zhao, C.; Xu, Y.; Mori, T. Timing the day: What makes bacterial clocks tick? Nat. Rev. Microbiol. 2017, 15, 232–242. [Google Scholar] [CrossRef]
- Schrader, J.; Schilling, M.; Holtmann, D.; Sell, D.; Villela, M.; Marx, A.; Vorholt, J.A. Methanol-based industrial biotechnology: Current status and future perspectives of methylotrophic bacteria. Trends Biotechnol. 2009, 27, 107–115. [Google Scholar] [CrossRef]
- Abanda-Nkpwatt, D.; Musch, M.; Tschiersch, J.; Boettner, M.; Schwab, W. Molecular interaction between Methylobacterium extorquens and seedlings: Growth promotion, methanol consumption, and localization of the methanol emission site. J. Exp. Bot. 2006, 57, 4025–4032. [Google Scholar] [CrossRef]
- Ryu, J.; Madhaiyan, M.; Poonguzhali, S.; Yim, W.; Indiragandhi, P.; Kim, K.; Anandham, R.; Yun, J.; Hoon Kim, K.; Sa, T. Plant growth substances produced by Methylobacterium spp. and their effect on tomato (Lycopersicon esculentum L.) and red pepper (Capsicum annuum L.) growth. J. Microbiol. Biotechnol. 2006, 16, 1622–1628. [Google Scholar]
- Madhaiyan, M.; Poonguzhali, S.; Sundaram, S.P.; Sa, T. A new insight into foliar applied methanol influencing phylloplane methylotrophic dynamics and growth promotion of cotton (Gossypium hirsutum L.) and sugarcane (Saccharum officinarum L.). Environ. Exp. Bot. 2006, 57, 168–176. [Google Scholar] [CrossRef]
- Meena, K.K.; Kumar, M.; Kalyuzhnaya, M.G.; Yandigeri, M.S.; Singh, D.P.; Saxena, A.K.; Arora, D.K. Epiphytic pink-pigmented methylotrophic bacteria enhance germination and seedling growth of wheat (Triticum aestivum) by producing phytohormone. Antonie Leeuwenhoek 2012, 101, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Yurimoto, H.; Iguchi, H.; Di Thien, D.T.; Tani, A.; Okumoto, Y.; Ota, A.; Yamauchi, T.; Akashi, T.; Sakai, Y. Methanol bioeconomy: Promotion of rice crop yield in paddy fields with microbial cells prepared from natural gas-derived C1 compound. Microb. Biotechnol. 2021, in press. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yurimoto, H.; Shiraishi, K.; Sakai, Y. Physiology of Methylotrophs Living in the Phyllosphere. Microorganisms 2021, 9, 809. https://doi.org/10.3390/microorganisms9040809
Yurimoto H, Shiraishi K, Sakai Y. Physiology of Methylotrophs Living in the Phyllosphere. Microorganisms. 2021; 9(4):809. https://doi.org/10.3390/microorganisms9040809
Chicago/Turabian StyleYurimoto, Hiroya, Kosuke Shiraishi, and Yasuyoshi Sakai. 2021. "Physiology of Methylotrophs Living in the Phyllosphere" Microorganisms 9, no. 4: 809. https://doi.org/10.3390/microorganisms9040809
APA StyleYurimoto, H., Shiraishi, K., & Sakai, Y. (2021). Physiology of Methylotrophs Living in the Phyllosphere. Microorganisms, 9(4), 809. https://doi.org/10.3390/microorganisms9040809





