Production of Monacolin K in Monascus pilosus: Comparison between Industrial Strains and Analysis of Its Gene Clusters
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains and Culture Conditions
2.2. Preparation of the Spore Suspensions for Monascus spp. Strains
2.3. Classification and Identification of Monascus spp. Strains
2.3.1. Colonial Morphologies of Monascus spp. Strains
2.3.2. Microscopic Morphologies of Monascus spp. Strains
2.3.3. Molecular Identification of Monascus spp. Strains
2.4. Solid-State and Liquid-State Cultures of Monacus spp. Strains
2.4.1. Culture Media
2.4.2. Preparation of Seed Liquid
2.4.3. Solid-State and Liquid-State Cultures
2.5. MK and CIT Analysis
2.5.1. MK and CIT Extraction of Solid-State Samples
2.5.2. MK and CIT Extraction of Liquid-State Samples
2.5.3. MK and CIT Detection
2.5.4. Statistical Analyses
2.6. Genomic DNA Extraction
2.7. DNA Sequencing and Assembly
2.8. Annotation and Analysis of Monascus spp. Genomes
2.9. Data Deposition
3. Results
3.1. Classification and Identification
3.1.1. Morphological Identification of Monascus spp.
3.1.2. Molecular Identification of Monascus spp. Strains
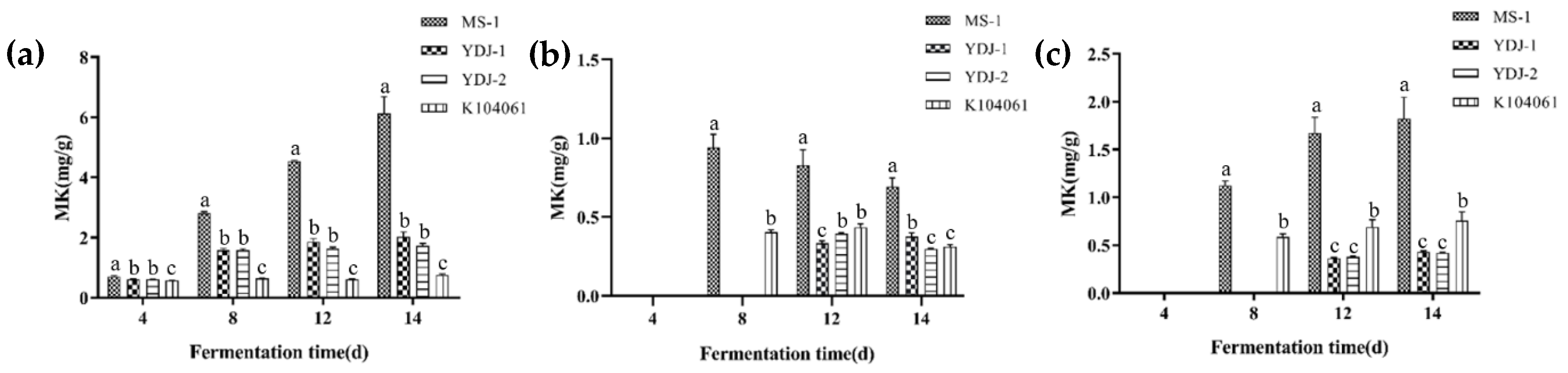
3.2. MK and CIT Production in Solid-State and Liquid-State Cultures
3.3. Genome Sequencing and Prediction of SM Gene Clusters
3.4. Comparison of MK BGCs
3.4.1. Function Re-Prediction of the Genes in the LOV and MK BGCs Reported Previously
3.4.2. Comparison of MK BGCs
3.4.3. Multiple sequence alignment of LOV and MK BGCs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chen, W.P.; He, Y.; Zhou, Y.X.; Shao, Y.C.; Feng, Y.L.; Li, M.; Chen, F.S. Edible filamentous fungi from the species Monascus: Early traditional fermentations, modern molecular biology, and future genomics. Compr. Rev. Food Sci. Food Saf. 2015, 14, 555–567. [Google Scholar] [CrossRef]
- Shi, Y.C.; Pan, T.M. Beneficial effects of Monascus purpureus NTU 568-fermented products: A review. Appl. Microbiol. Biotechnol. 2011, 90, 1207–1217. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Li, Y.; Ye, Q.; Li, J.; Hua, Y.; Ju, D.; Zhang, D.; Cooper, R.; Chang, M. Constituents of red yeast rice, a traditional chinese food and medicine. J. Agric. Food Chem. 2000, 48, 5220–5225. [Google Scholar] [CrossRef]
- Hong, S.H.; Lee, I.; Kim, S.J.; Imm, J.Y. Improved functionality of soft soybean curd containing Monascus fermented soybean ethanol extract. Food Sci. Biotechnol. 2012, 21, 701–707. [Google Scholar] [CrossRef]
- Liu, M.T.; Li, J.J.; Shang, X.Y.; Li, S.; Li, L.L.; Luan, N.; Jin, Z.L. Structure elucidation and complete NMR spectral assignment of an unusual aromatic monacolin analog from Monascus purpureus-fermented rice. Magn. Reason. Chem. 2011, 49, 129–131. [Google Scholar] [CrossRef]
- Cheng, M.J.; Chen, J.J.; Wu, M.D.; Yang, P.S.; Yuan, G.F. Isolation and structure determination of one new metabolite isolated from the red fermented rice of Monascus purpureus. Nat. Prod. Res. 2010, 24, 979–988. [Google Scholar] [CrossRef]
- Knecht, A.; Cramer, B.; Humpf, H.U. New Monascus metabolites: Structure elucidation and toxicological properties studied with immortalized human kidney epithelial cells. Mol. Nutr. Food Res. 2006, 50, 314–321. [Google Scholar] [CrossRef]
- Wild, D.; Toth, G.; Humpf, H.U. New Monascus metabolite isolated from red yeast rice (angkak, red koji). J. Agric. Food Chem. 2002, 50, 3999–4002. [Google Scholar] [CrossRef]
- Blanc, P.J.; Goma, G.; Loret, M.O. Production of citrinin by various species of Monascus. Biotechnol. Lett. 1995, 17, 291–294. [Google Scholar] [CrossRef]
- Feng, Y.L.; Chen, W.P.; Chen, F.S. A Monascus pilosus MS-1 strain with high-yield monacolin K but no citrinin. Food Sci. Biotechnol. 2016, 25, 1115–1122. [Google Scholar] [CrossRef]
- Hsu, Y.W.; Hsu, L.C.; Liang, Y.H.; Kuo, Y.H.; Pan, T.M. New bioactive orange pigments with yellow fluorescence from Monascus-fermented dioscorea. J. Agric. Food Chem. 2011, 59, 4512–4518. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.J.; Wu, M.D.; Chen, Y.L.; Chen, I.S.; Su, Y.S.; Yuan, G.F. Chemical constituents of red yeast rice fermented with the fungus Monascus pilosus. Chem. Nat. Compd. 2013, 49, 249–252. [Google Scholar] [CrossRef]
- Lian, X.; Liu, L.; Dong, S.; Wu, H.; Zhao, J.; Han, Y. Two new Monascus red pigments produced by Shandong Zhonghui Food Company in China. Eur. Food Res. Technol. 2015, 240, 719–724. [Google Scholar] [CrossRef]
- Feng, Y.L.; Shao, Y.C.; Chen, F.S. Monascus pigments. Appl. Microbiol. Biotechnol. 2012, 96, 1421–1440. [Google Scholar] [CrossRef] [PubMed]
- Tsukahara, M.; Shinzato, N.; Tamaki, Y.; Namihira, T.; Matsui, T. Red yeast rice fermentation by selected Monascus sp. with deep-red color, lovastatin production but no citrinin, and effect of temperature-shift cultivation on lovastatin production. Appl. Biochem. Biotechnol. 2009, 158, 476–482. [Google Scholar] [CrossRef]
- Liu, Z.X. Molecular Mechanism of Monacolin K and Pigment Production by Monascus Based on Transcriptomics. Master’s Thesis, Shihezi University, Shihezi, China, 2019. [Google Scholar]
- Brakhage, A. Regulation of fungal secondary metabolism. Nat. Rev. Microbiol. 2013, 11, 21–32. [Google Scholar] [CrossRef]
- Keller, N.P. Fungal secondary metabolism: Regulation, function and drug discovery. Nat. Rev. Microbiol. 2019, 17, 167–180. [Google Scholar] [CrossRef]
- Keller, N.P.; Turner, G.; Bennett, J.W. Fungal secondary metabolism—from biochemistry to genomics. Nat. Rev. Microbiol. 2005, 3, 937–947. [Google Scholar] [CrossRef]
- Hertweck, C. The biosynthetic logic of polyketide diversity. Angew. Chem. Int. Ed. Engl. 2009, 48, 4688–4716. [Google Scholar] [CrossRef]
- Du, L.; Lou, L. PKS and NRPS release mechanisms. Nat. Prod. Rep. 2010, 27, 255–278. [Google Scholar] [CrossRef] [PubMed]
- Crawford, J.M.; Townsend, C.A. New insights into the formation of fungal aromatic polyketides. Nat. Rev. Microbiol. 2010, 8, 879–889. [Google Scholar] [CrossRef] [PubMed]
- Brakhage, A.A.; Schroeckh, V. Fungal secondary metabolites—strategies to activate silent gene clusters. Fungal Genet. Biol. 2011, 48, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.J. Polyketides, proteins and genes in fungi: Programmed nano-machines begin to reveal their secrets. Org. Biomol. Chem. 2007, 5, 2010–2026. [Google Scholar] [CrossRef]
- Williams, G.J. Engineering polyketide synthases and nonribosomal peptide synthetases. Curr. Opin. Struct. Biol. 2013, 23, 603–612. [Google Scholar] [CrossRef] [PubMed]
- Hur, G.H.; Vickery, C.R.; Burkart, M.D. Explorations of catalytic domains in non-ribosomal peptide synthetase enzymology. Nat. Prod. Rep. 2012, 29, 1074. [Google Scholar] [CrossRef] [PubMed]
- Strieker, M.; Tanović, A.; Marahiel, M.A. Nonribosomal peptide synthetases: Structures and dynamics. Curr. Opin. Struct. Biol. 2010, 20, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Boettger, D.; Hertweck, C. Molecular diversity sculpted by fungal PKS-NRPS hybrids. Chem. Biol. Chem. 2012, 14, 28–42. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, B.; Du, X.; Li, P.; Liang, B.; Cheng, X.; Du, L.; Di, H.; Wang, L.; Wang, S. Complete genome sequence and transcriptomics analyses reveal pigment biosynthesis and regulatory mechanisms in an industrial strain, Monascus purpureus YY-1. Sci. Rep. 2015, 5, 8331. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, X.; Liu, X.; Li, X.; Zhang, C.; Li, W.; Suna, X.; Wanga, W.; Sunac, B. Discovery and development of a novel short-chain fatty acid ester synthetic biocatalyst under aqueous phase from Monascus purpureus isolated from Baijiu. Food Chem. 2020, 338, 128025. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, T.; Tsukahara, M.; Katayama, N.; Yaoi, K.; Aburatani, S.; Ohdan, K.; Fujimori, K.E. Whole-genome sequence of Monascus purpureus GB-01, an industrial strain for food colorant production. Microbiol. Resour. Announce. 2019, 8, e00196-e19. [Google Scholar] [CrossRef]
- Weusthuis, R.A.; Mars, A.E.; Springer, J.; Wolbert, E.J.H.; van der Wal, H.; de Vrije, T.G.; Levissonb, M.; Leprinceb, A.; Moers, A.P.H.A. Monascus ruber as cell factory for lactic acid production at low pH. Metab. Eng. 2017, 42, 66–73. [Google Scholar] [CrossRef]
- Feng, Y.L. Identification and Fermentation Characteristics of Monascus pilosus MS-1. Ph.D. Thesis, Huazhong Agricultural University, Wuhan, China, 2014. [Google Scholar]
- Xie, Y.M. Study on cAMP and dbcAMP Regulating Development and Monacolin K Production of Monascus spp. Master’s Thesis, Huazhong Agricultural University, Wuhan, China, 2019. [Google Scholar]
- Hawksworth, D.L.; Pitt, J.I. A new taxonomy for Monascus species based on cultural and microscopical characters. Aust. J. Bot. 1983, 31, 51. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Saitou, N.; Nei, M. The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol. Biol. Evol. 1987, 4, 406–425. [Google Scholar] [PubMed]
- Felsenstein, J. Confidence limits on phylogenies: An approach using the bootstrap. Evolution 1985, 39, 783–791. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Van Burik, J.A.; Schreckhise, R.W.; White, T.C.; Bowden, R.A.; Myerson, D. Comparison of six extraction techniques for isolation of DNA from filamentous fungi. Med. Mycol. 1998, 36, 299–303. [Google Scholar] [CrossRef]
- Andrews, S. FASTQC. A Quality Control Tool for High throughput Sequence Data. 2010. Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 15 August 2019).
- Ardui, S.; Ameur, A.; Vermeesch, J.R.; Hestand, M.S. Single molecule real-time (SMRT) sequencing comes of age: Applications and utilities for medical diagnostics. Nucleic Acids Res. 2018, 46, 2159–2168. [Google Scholar] [CrossRef] [PubMed]
- Reiner, J.; Pisani, L.; Qiao, W.; Singh, R.; Yang, Y.; Shi, L.; Scott, A.S.; Cohen, N.; Babu, A.; Edelmann, L. Cytogenomic identification and long-read single molecule real-time (SMRT) sequencing of aBardet–Biedl Syndrome 9(BBS9) deletion. NPJ Genom. Med. 2018, 3, 1–5. [Google Scholar] [CrossRef]
- Stanke, M.; Diekhans, M.; Baertsch, R.; Haussler, D. Using native and syntenically mapped cDNA alignments to improve de novo gene finding. Bioinformatics 2008, 24, 637–644. [Google Scholar] [CrossRef]
- Blin, K.; Shaw, S.; Steinke, K.; Villebro, R.; Ziemert, N.; Lee, S.Y.; Weber, T. AntiSMASH 5.0: Updates to the secondary metabolite genome mining pipeline. Nucleic Acids Res. 2019, 47, W81–W87. [Google Scholar] [CrossRef]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Thierer, T.; Ashton, B.; Meintjes, P.; Drummond, A. Geneious Basic: An integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.Q.; Guo, F. Morphology and Taxonomy of Monascus; China Light Industry Press: Beijing, China, 2003. (In Chinese) [Google Scholar]
- Kennedy, J. Modulation of polyketide synthase activity by accessory proteins during lovastatin biosynthesis. Science 1999, 284, 1368–1372. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Chooi, Y.-H.; Choi, J.W.; Li, S.; Vederas, J.C.; Da Silva, N.A.; Tang, Y. LovG: The thioesterase required for dihydromonacolin L release and lovastatin nonaketide synthase turnover in lovastatin biosynthesis. Angew. Chem. Int. Ed. 2013, 52, 6472–6475. [Google Scholar] [CrossRef]
- Chen, Y.P.; Tseng, C.P.; Liaw, L.L.; Wang, C.L.; Chen, I.C.; Wu, W.J.; Yuan, G.F. Cloning and characterization of monacolin K biosynthetic gene cluster from Monascus pilosus. J. Agric. Food Chem. 2008, 56, 5639–5646. [Google Scholar] [CrossRef] [PubMed]
- Endo, A. Monacolin K, a new hypocholesterolemic agent produced by a Monascus species. J. Antibiot. 1979, 32, 852–854. [Google Scholar] [CrossRef] [PubMed]
- Alberts, A.W.; Chen, J.; Kuron, G.; Hunt, V.; Huff, J.; Hoffman, C.; Springer, J.; Hirshfield, J.; Hoogsteen, K.; Liesch, J. Mevinolin: A highly potent competitive inhibitor of hydroxymethylglu-taryl-coenzyme a reductase and a cholesterol-lowering agent. Proc. Natl. Acad. Sci. USA 1980, 77, 3957–3961. [Google Scholar] [CrossRef]
- Manzoni, M.; Rollini, M. Biosynthesis and biotechnological production of statins by filamentous fungi and application of these cholesterol-lowering drugs. Appl. Microbiol. Biotechnol. 2002, 58, 555–564. [Google Scholar] [PubMed]
- Studer, R.A.; Dessailly, B.H.; Orengo, C.A. Residue mutations and their impact on protein structure and function: Detecting beneficial and pathogenic changes. Biochem. J. 2013, 449, 581–594. [Google Scholar] [CrossRef]




| MA | CYA | PDA | G25N | |
|---|---|---|---|---|
| Colonial Morphologies | The colonial diameters reached 40–48 mm. Their obverse and reverse surfaces were white and light orange to orange-red, respectively. Their mycelia were compact and dense. | The colonial diameters reached 50–65 mm. Their shapes were irregular. Their obverse and reverse surfaces were white and red to dark red with radial folds. Their mycelia were sparse and short. | The colonial diameters reached 40–55 mm. Their reverse surfaces were light orange or orange-red and the edges were light yellow to golden yellow. Their middle parts were raised, and their mycelia were dense and fluffy. | The colonial diameters reached 11–25 mm. Their obverse and reverse surfaces were white and colorless. |
| Microscopic Morphologies | Cleistothecia arose singly at the tips of stalk-like hyphae and walls were hyaline or pale brown. Conidia were hyaline and borne laterally on pedicels and terminally on hyphae, arising singly or occasionally in short chains, obpyriform to globose. | No cleistothecium; conidia were transparent or brown and obpyriform to globose. | Cleistothecia were globose and arose singly from distinct stalk-like hyphae. Conidia were spherical or upside-down pear-shaped with colorless or brown colors. | No cleistothecium; conidia were spherical, transparent, and colorless. |
| Genome Features | MS-1 | YDJ-1 | YDJ-2 | K104061 |
|---|---|---|---|---|
| Genome length (Mb) | 26.21 | 26.15 | 26.16 | 26.14 |
| GC content (%) | 48.89 | 48.90 | 48.89 | 48.87 |
| Gene amount (#) | 7771 | 7687 | 7718 | 7634 |
| Gene total length (Mb) | 13.26 | 13.04 | 13.13 | 13.09 |
| Gene average length (bp) | 1707 | 1696 | 1701 | 1715 |
| Gene length/Genome (%) | 50.60 | 49.86 | 50.18 | 50.08 |
| lov/mk* | Function (Previous Study) | Function (Re-Predicted in This Study) |
|---|---|---|
| lovA/mkC | Cytochrome P450 monooxygenase | Cytochrome P450 monooxygenase |
| lovB/mkA | LOV nonaketide synthase | LOV nonaketide synthase |
| lovC/mkE | Enoyl reductase/Dehydrogenase | Enoyl reductase/Dehydrogenase |
| lovD/mkF | Transesterase | Transesterase |
| lovE/mkH | Regulatory protein | Regulatory protein |
| lovF/mkB | LOV diketide synthase | LOV diketide synthase |
| lovG/mkD | Thioesterase/Oxidoreductase | α/β hydrolase |
| ORF1/- | Esterase | Esterase |
| ORF2/- | Unknown | Transporters |
| ORF8/mkG | HMG-CoA reductase | HMG-CoA reductase |
| ORF10/mkI | Unknown/Efflux pump | Transporters |
| ORF12/- | Unknown | Unknown |
| ORF13/- | Regulatory protein | Regulatory protein |
| ORF14/- | Unknown | Mitochondrial carrier protein |
| ORF15/- | Unknown | Dehydratase |
| ORF16/- | Unknown | Transporters |
| ORF17/- | Cytochrome P450 monooxygenase | Cytochrome P450 monooxygenase |
| ORF18/- | Unknown | Glycosyl hydrolase |
| lov/mk Genes | M. pilosus | M. ruber | |||||
|---|---|---|---|---|---|---|---|
| MS-1 | YDJ-1 | YDJ-2 | K104061 | FWB13 | CBS127566 | NRRL1597 | |
| lovB/mkA | 76.75/99.93 | 76.75/99.93 | 76.75/99.93 | 76.75/99.93 | 76.75/99.93 | 76.65/99.80 | 76.81/100 |
| lovF/mkB | 72.23/99.96 | 72.57/99.96 | 72.61/100 | 72.23/99.96 | 71.48/98.44 | 71.52/98.44 | - |
| lovA/mkC | 67.14/88.73 92.11/100 | -/- 84.57/97.91 | 67.14/88.73 92.11/100 | 67.14/88.73 92.11/100 | 67.14/88.73 92.11/100 | 67.14/88.73 92.11/100 | 67.61/88.73 mkC1 92.11/100 mkC2 |
| lovG/mkD | 67.16/99.62 | 66.79/100 | 66.79/100 | 66.79/100 | 64.18/100 | 64.18/96.58 | 64.18/100 |
| lovC/mkE | 78.01/93.02 | 78.01/93.02 | 77.75/92.76 | 78.01/93.02 | 78.59/94.17 | 78.59/94.17 | 78.59/94.17 |
| lovD/mkF | 87.41/97.28 | 88.17/100 | 87.93/96.25 | 87.41/97.28 | 86.90/97.02 | 86.90/97.02 | 86.90/97.02 |
| ORF8/mkG | 71.98/100 | 71.72/99.62 | 71.98/100 | 71.98/100 | 71.98/100 | 71.98/100 | 71.98/100 |
| lovE/mkH | 55.65/100 | 55.65/100 | 55.65/100 | 55.65/100 | 55.65/100 | 55.86/99.77 | 55.44/99.32 |
| ORF10/mkI | 79.26/100 | 79.26/100 | 79.26/100 | 79.26/100 | 78.31/97.24 | 79.75/96.82 | 78.31/97.24 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, W.; Shao, Y.; Chen, F. Production of Monacolin K in Monascus pilosus: Comparison between Industrial Strains and Analysis of Its Gene Clusters. Microorganisms 2021, 9, 747. https://doi.org/10.3390/microorganisms9040747
Dai W, Shao Y, Chen F. Production of Monacolin K in Monascus pilosus: Comparison between Industrial Strains and Analysis of Its Gene Clusters. Microorganisms. 2021; 9(4):747. https://doi.org/10.3390/microorganisms9040747
Chicago/Turabian StyleDai, Weihua, Yanchun Shao, and Fusheng Chen. 2021. "Production of Monacolin K in Monascus pilosus: Comparison between Industrial Strains and Analysis of Its Gene Clusters" Microorganisms 9, no. 4: 747. https://doi.org/10.3390/microorganisms9040747
APA StyleDai, W., Shao, Y., & Chen, F. (2021). Production of Monacolin K in Monascus pilosus: Comparison between Industrial Strains and Analysis of Its Gene Clusters. Microorganisms, 9(4), 747. https://doi.org/10.3390/microorganisms9040747







