Biological Processes Highlighted in Saccharomyces cerevisiae during the Sparkling Wines Elaboration
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganism, Cultivation Conditions, and Sampling
2.2. Viability
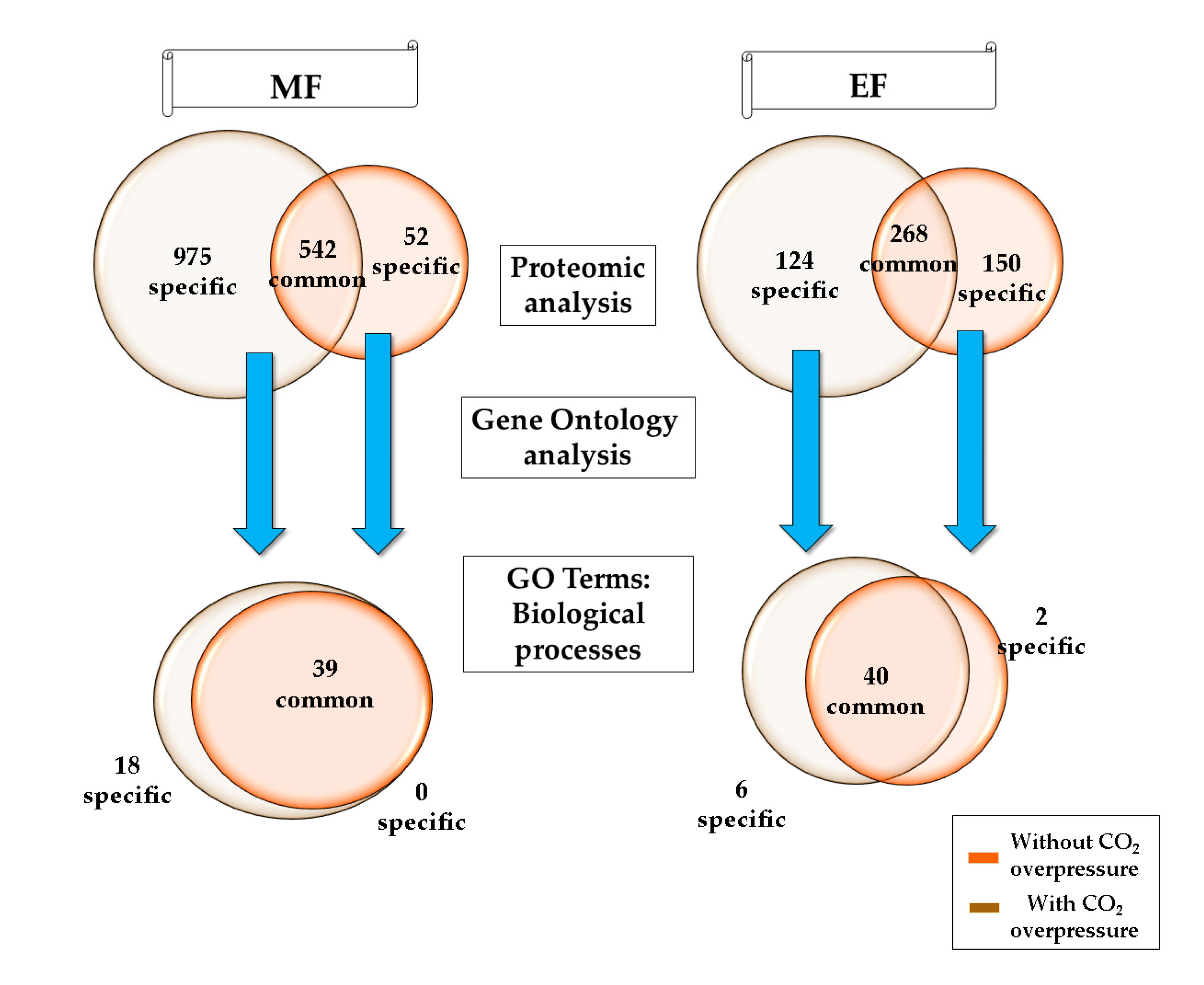
2.3. Proteomic Analysis
2.4. Biological Processes Analysis
3. Results and Discussion
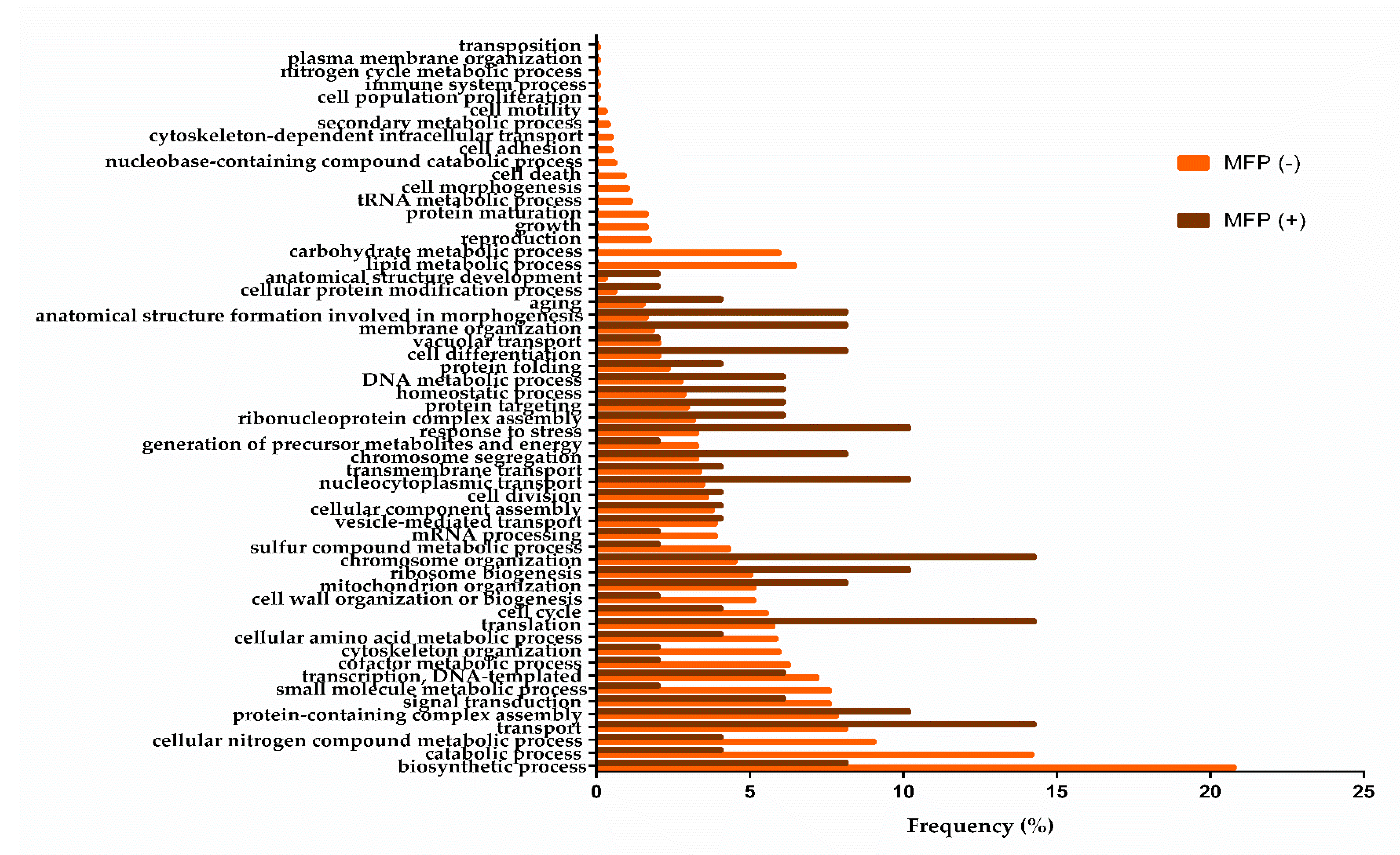
3.1. Biological Processes during the Middle of the Second Fermentation
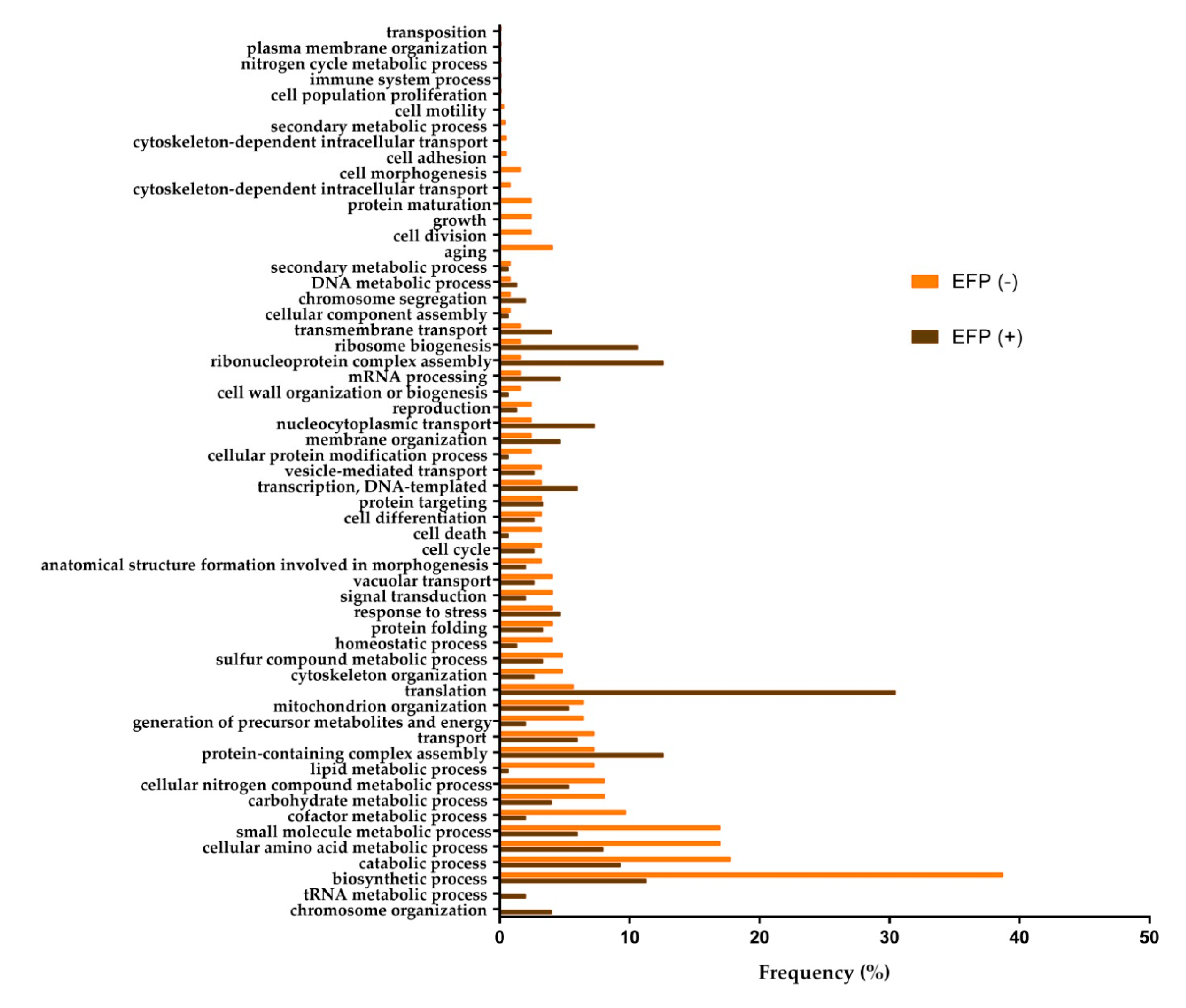
3.2. Biological Processes at the End of the Second Fermentation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- González-Ramos, D.; González, R. Genetic Determinants of the release of mannoproteins of enological interest by Saccharomyces cerevisiae. J. Agric. Food Chem. 2006, 54, 9411–9416. [Google Scholar] [CrossRef] [PubMed]
- Charpentier, C.; Feuillat, M. Yeast autolysis. In Wine Microbiology and Biotechnology; Fleet, G., Ed.; Harwood Academic Publishers: London, UK, 1993; pp. 225–242. [Google Scholar]
- Martínez-Rodríguez, A.J.; Polo, M.C.; Carrascosa, A.V. Structural and ultrastructural changes in yeast cells during autolysis in a model wine system and in sparkling wines. Int. J. Food Microbiol. 2001, 71, 45–51. [Google Scholar] [CrossRef]
- Troton, D.; Charpentier, M.; Robillard, B.; Calvayrac, R.; Duteurtre, B. Evolution of the lipid contents of Champagne wine during the second fermentation of Saccharomyces cerevisiae. Am. J. Enol. Viticult. 1989, 40, 175–182. [Google Scholar]
- Pozo-Bayón, M.A.; Martín-Álvarez, P.J.; Moreno-Arribas, M.V.; Andújar-Ortiz, I.; Pueyo, E. Impact of using Trepat and Monastrell red grape varieties on the volatile and nitrogen composition during the manufacture of rosé Cava sparkling wines. LWT-Food Sci. Technol. 2010, 43, 1526–1532. [Google Scholar] [CrossRef] [Green Version]
- Torrens, J.; Urpí, P.; Riu-Aumatell, M.; Vichi, S.; López-Tamames, E.; Buxaderas, S. Different commercial yeast strains affecting the volatile and sensory profile of cava base wine. Int. J. Food Microbiol. 2008, 124, 48–57. [Google Scholar] [CrossRef]
- Pozo-Bayón, M.A.; Martínez-Rodríguez, A.; Pueyo, E.; Moreno-Arribas, M.V. Chemical and biochemical features involved in sparkling wine production: From a traditional to an improved winemaking technology. Trends. Food Sci. Technol. 2009, 20, 289–299. [Google Scholar] [CrossRef]
- Torresi, S.; Frangipane, M.T.; Anelli, G. Biotechnologies in sparkling wine production. Interesting approaches for quality improvement: A review. Food Chem. 2011, 129, 1232–1241. [Google Scholar]
- Morales, M.L.; Ochoa, M.; Valdivia, M.; Ubeda, C.; Romero-Sanchez, S.; Ibeas, J.I.; Valero, E. Volatile metabolites produced by different flor yeast strains during wine biological ageing. Food Res. Int. 2020, 128, 108771. [Google Scholar] [CrossRef]
- Martínez-García, R.; García-Martínez, T.; Puig-Pujol, A.; Mauricio, J.C.; Moreno, J. Changes in sparkling wine aroma during the second fermentation under CO2 pressure in sealed bottle. Food Chem. 2017, 237, 1030–1040. [Google Scholar] [CrossRef]
- Alexandre, H.; Guilloux-Benatier, M. Yeast autolysis in sparkling wine—A review. Aust. J. Grape Wine Res. 2006, 12, 119–127. [Google Scholar] [CrossRef]
- Kemp, B.; Alexandre, H.; Robillard, B.; Marchal, R. Effect of production phase on bottle-fermented sparkling wine quality. J. Agr. Food Chem. 2015, 63, 19–38. [Google Scholar] [CrossRef] [PubMed]
- Martí-Raga, M.; Martín, V.; Gil, M.; Sancho, M.; Zamora, F.; Mas, A.; Beltran, G. Contribution of yeast and base wine supplementation to sparkling wine composition. J. Sci. Food Agric. 2016, 96, 4962–4972. [Google Scholar] [CrossRef] [PubMed]
- Penacho, V.; Valero, E.; González, R. Transcription profiling of sparkling wine second fermentation. Int. J. Food Microbiol. 2012, 153, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Porras-Agüera, J.A.; Román-Camacho, J.J.; Moreno-García, J.; Mauricio, J.C.; Moreno, J.; García-Martínez, T. Effect of endogenous CO2 overpressure on the yeast “stressome” during the “prise de mousse” of sparkling wine. Food Microbiol. 2020, 89, 103431. [Google Scholar] [CrossRef]
- Bartra, E.; Casado, M.; Carro, D.; Campama, C.; Piña, B. Differential expression of thiamine biosynthetic genes in yeast strains with high and low production of hydrogen sulfide during wine fermentation. J. Appl. Microbiol. 2010, 109, 272–281. [Google Scholar] [CrossRef]
- Bradford, M.M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Moreno-García, J.; García-Martínez, T.; Moreno, J.; Mauricio, J.C. Proteins Involved in Flor Yeast Carbon Metabolism under Biofilm Formation Conditions. Food Microbiol. 2015, 46, 25–33. [Google Scholar] [CrossRef]
- Dasari, S.; Chambers, M.C.; Slebos, R.J.; Zimmerman, L.J.; Ham, A.J.L.; Tabb, D.L. TagRecon: High-throughput mutation identification through sequence tagging. J. Proteome Res. 2010, 9, 1716–1726. [Google Scholar] [CrossRef] [Green Version]
- Denny, P.; Feuermann, M.; Hill, D.P.; Lovering, R.C.; Plun-Favreau, H.; Roncaglia, P. Exploring autophagy with gene ontology. Autophagy 2018, 14, 419–436. [Google Scholar] [CrossRef]
- Matallana, E.; Aranda, A. Biotechnological impact of stress response on wine yeast. Lett. Appl. Microbiol. 2017, 64, 103–110. [Google Scholar] [CrossRef] [Green Version]
- Hazel, J.R.; Williams, E.E. The role of alterations in membrane lipid composition in enabling physiological adaptation of organisms to their physical environment. Prog. Lipid Res. 1990, 29, 167–227. [Google Scholar] [CrossRef]
- Valero, E.; Millan, M.C.; Mauricio, J.C.; Ortega, J.M. Effect of grape skin maceration on sterol, phospholipid, and fatty acid contents of Saccharomyces cerevisiae during alcoholic fermentation. Am. J. Enol. Vitic. 1998, 49, 119–124. [Google Scholar]
- Tesnière, C. Importance and role of lipids in wine yeast fermentation. Appl. Microbiol. Biotechnol. 2019, 103, 8293–8300. [Google Scholar] [CrossRef] [PubMed]
- Francois, J.; Parrou, J.L. Reserve carbohydrates metabolism in the yeast Saccharomyces cerevisiae. FEMS Microbiol. Rev. 2001, 25, 125–145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suomalainen, H.; Lehtonen, M. The production of aroma compounds by yeast. J. Inst. Brew. 1979, 85, 149–156. [Google Scholar] [CrossRef]
- Kwast, K.E.; Burke, P.V.; Poyton, R.O. Oxygen sensing and the transcriptional regulation of oxygen-responsive genes in yeast. J. Exp. Biol. 1998, 201, 1177–1195. [Google Scholar]
- Aguilera, A.; Benítez, T. Role of mitochondria in ethanol tolerance of Saccharomyces cerevisiae. Arch. Microbiol. 1985, 142, 389–392. [Google Scholar] [CrossRef]
- Winderickx, J.; Holsbeeks, I.; Lagatie, O.; Giots, F.; Thevelein, J.; Winde, H. From feast to famine; adaptation to nutrient availability in yeast. In Yeast Stress Responses; Hohmann, S., Mager, W.H., Eds.; Springer: Berlin/Heidelberg, Germany, 2003; pp. 305–386. [Google Scholar]
- De Virgilio, C. The essence of yeast quiescence. FEMS Microbiol. Rev. 2012, 36, 306–339. [Google Scholar] [CrossRef] [Green Version]
- González-Jiménez, M.C.; Moreno-García, J.; García-Martínez, T.; Moreno, J.J.; Puig-Pujol, A.; Capdevilla, F.; Mauricio, J.C. Differential analysis of proteins involved in ester metabolism in two Saccharomyces cerevisiae strains during the second fermentation in sparkling wine elaboration. Microorganisms 2020, 8, 403. [Google Scholar] [CrossRef] [Green Version]
- Verduyn, C.; Postma, E.; Scheffers, W.; van Dijken, J. Energetics of S. cerevisiae in anaerobic glucose-limited chemostat cultures. J. Gen. Microbiol. 1990, 136, 405–412. [Google Scholar]
- Kutyna, D.R.; Varela, C.; Henschke, P.A.; Chambers, P.J.; Stanley, G.A. Microbiological approaches to lowering ethanol concentration in wine. Trends. Food Sci. Technol. 2010, 21, 293–302. [Google Scholar] [CrossRef]
- Peeters, K.; Van Leemputte, F.; Fischer, B.; Bonini, B.M.; Quezada, H.; Tsytlonok, M.; Haesen, D.; Vanthienen, W.; Bernardes, N.; Bravo Gonzalez-Blas, C.; et al. Fructose-1,6-bisphosphate couples glycolytic flux to activation of Ras. Nat. Commun. 2017, 8, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Peláez-Soto, A.; Roig, P.; Martínez-Culebras, P.V.; Fernández-Espinar, M.T.; Gil, J.V. Proteomic analysis of Saccharomyces cerevisiae response to oxidative stress mediated by cocoa polyphenols extract. Molecules 2020, 25, 452. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jing, H.; Liu, H.; Zhang, L.; Gao, J.; Song, H.; Tan, X. Ethanol induces autophagy regulated by mitochondrial ROS in Saccharomyces cerevisiae. J. Microbiol. Biotechnol. 2018, 28, 1982–1991. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cebollero, E.; Gonzalez, R. Induction of autophagy by second-fermentation yeasts during elaboration of sparkling wines. Appl. Environ. Microbiol. 2006, 72, 4121–4127. [Google Scholar] [CrossRef] [Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Jiménez, M.d.C.; García-Martínez, T.; Puig-Pujol, A.; Capdevila, F.; Moreno-García, J.; Moreno, J.; Mauricio, J.C. Biological Processes Highlighted in Saccharomyces cerevisiae during the Sparkling Wines Elaboration. Microorganisms 2020, 8, 1216. https://doi.org/10.3390/microorganisms8081216
González-Jiménez MdC, García-Martínez T, Puig-Pujol A, Capdevila F, Moreno-García J, Moreno J, Mauricio JC. Biological Processes Highlighted in Saccharomyces cerevisiae during the Sparkling Wines Elaboration. Microorganisms. 2020; 8(8):1216. https://doi.org/10.3390/microorganisms8081216
Chicago/Turabian StyleGonzález-Jiménez, María del Carmen, Teresa García-Martínez, Anna Puig-Pujol, Fina Capdevila, Jaime Moreno-García, Juan Moreno, and Juan Carlos Mauricio. 2020. "Biological Processes Highlighted in Saccharomyces cerevisiae during the Sparkling Wines Elaboration" Microorganisms 8, no. 8: 1216. https://doi.org/10.3390/microorganisms8081216







