Proteomics of Brucella: Technologies and Their Applications for Basic Research and Medical Microbiology
Abstract
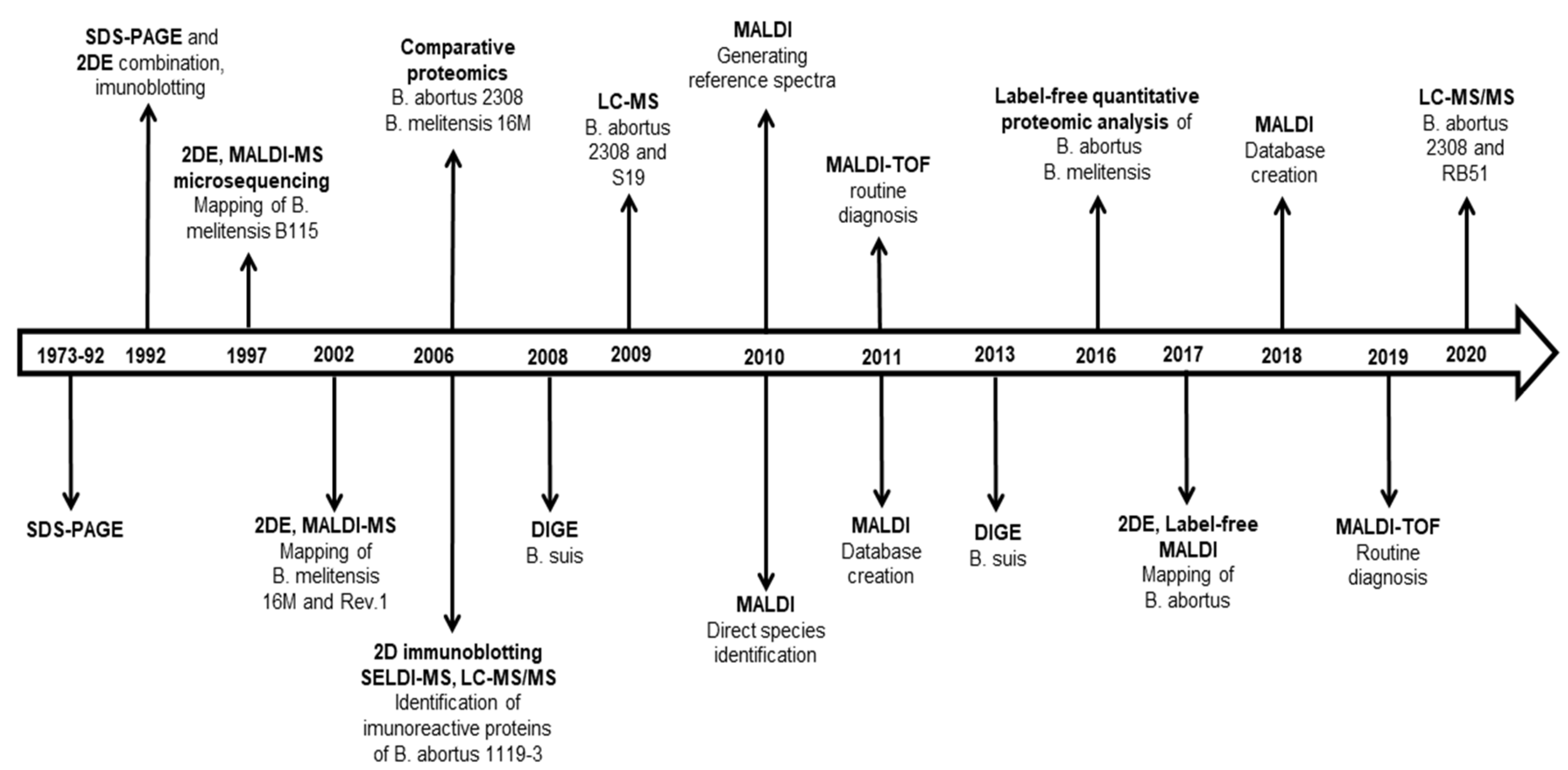
1. Introduction
2. Rapid Species Identification of Brucella Using Matrix-Assisted Laser Desorption Ionization—Time of Flight Mass Spectrometry (MALDI—TOF MS)
3. Gel-Based Quantitative Brucella Proteomics Analysis
4. Mass Spectrometry Based Quantitative Brucella Proteomics
5. Serological Proteome Analysis (SERPA)
6. Summary and Perspective
Author Contributions
Funding
Conflicts of Interest
References
- Atluri, V.L.; Xavier, M.N.; de Jong, M.F.; den Hartigh, A.B.; Tsolis, R.M. Interactions of the human pathogenic Brucella species with their hosts. Annu. Rev. Microbiol. 2011, 65, 523–541. [Google Scholar] [CrossRef]
- Godfroid, J.; Scholz, H.C.; Barbier, T.; Nicolas, C.; Wattiau, P.; Fretin, D.; Whatmore, A.M.; Cloeckaert, A.; Blasco, J.M.; Moriyon, I.; et al. Brucellosis at the animal/ecosystem/human interface at the beginning of the 21st century. Prev. Vet. Med. 2011, 102, 118–131. [Google Scholar] [CrossRef]
- Foster, G.; Osterman, B.; Godfroid, J.; Jacques, I.; Cloeckaert, A. Brucella ceti sp. nov. and Brucella pinnipedialis sp. nov. for Brucella strains with cetaceans and seals as their preferred hosts. Int. J. Syst. Evol. Microbiol. 2007, 57, 2688–2693. [Google Scholar]
- Scholz, H.; Nockler, K.; Gollner, C. Brucella inopinata sp. nov., isolated from a breast implant infection. Int. J. Syst. Evol. Microbiol. 2010, 60, 801–808. [Google Scholar] [CrossRef]
- Scholz, H.; Hubalek, Z.; Sedlácek, I.; Vergnaud, G.; Tomaso, H.; Al Dahouk, S.; Melzer, F.; Kämpfer, P.; Neubauer, H.; Cloeckaert, A.; et al. Brucella microti sp. nov.isolated from the common vole Microtus Arvalis. Int. J. Syst. Evol. Microbiol. 2008, 58, 375–382. [Google Scholar] [CrossRef]
- Whatmore, A.; Davison, N.; Cloeckaert, A.; Al Dahouk, S.; Zygmunt, M.; Brew, S.; Perrett, L.; Koylass, M.; Vergnaud, G.; Quance, C.; et al. Brucella papionis sp. nov. isolated from baboons (Papio spp.). Int. J. Syst. Evol. Microbiol. 2014, 64, 4120–4128. [Google Scholar] [CrossRef]
- Scholz, H.C.; Revilla-Fernandez, S.; Al Dahouk, S.; Hammerl, J.A.; Zygmunt, M.S.; Cloeckaert, A.; Koylass, M.; Whatmore, A.M.; Blom, J.; Vergnaud, G.; et al. Brucella vulpis sp. nov., isolated from mandibular lymph nodes of red foxes (Vulpes vulpes). Int. J. Syst. Evol. Microbiol. 2016, 66, 2090–2098. [Google Scholar] [CrossRef]
- Martirosyan, A.; Moreno, E.; Gorvel, J.-P. An evolutionary strategy for a stealthy intracellular Brucella pathogen. Immunol. Rev. 2011, 240, 211–234. [Google Scholar] [CrossRef]
- Halling, S.M.; Peterson-Burch, B.D.; Bricker, B.J.; Zuerner, R.L.; Qing, Z.; Li, L.L.; Kapur, V.; Alt, D.P.; Olsen, S.C. Completion of the genome sequence of Brucella abortus and comparison to the highly similar genomes of Brucella melitensis and Brucella suis. J. Bacteriol. 2005, 187, 2715–2726. [Google Scholar] [CrossRef]
- Wang, Y.; Ke, Y.; Wang, Z.; Yuan, X.; Qiu, Y.; Zhen, Q.; Xu, J.; Li, T.; Wang, D.; Huang, L.; et al. Genome sequences of three live attenuated vaccine strains of Brucella species and implications for pathogenesis and differential diagnosis. J. Bacteriol. 2012, 194, 6012–6013. [Google Scholar] [CrossRef]
- Chain, P.S.; Comerci, D.J.; Tolmasky, M.E.; Larimer, F.W.; Malfatti, S.A.; Vergez, L.M.; Aguero, F.; Land, M.L.; Ugalde, R.A.; Garcia, E. Whole-genome analyses of speciation events in pathogenic Brucellae. Infect. Immun. 2005, 73, 8353–8361. [Google Scholar] [CrossRef]
- Murugaiyan, J.; Eravci, M.; Weise, C.; Roesler, U.; Melzer, F.; Sprague, L.; Neubauer, H.; Wareth, G. Pan-proteomic analysis and elucidation of protein abundance among the closely related Brucella species, Brucella abortus and Brucella melitensis. Biomolecules 2020, in press. [Google Scholar]
- Pascual, D.W.; Yang, X.; Wang, H.; Goodwin, Z.; Hoffman, C.; Clapp, B. Alternative strategies for vaccination to brucellosis. Emerg. Microbes Infect. 2018, 20, 599–605. [Google Scholar] [CrossRef]
- Hou, H.; Liu, X.; Peng, Q. The advances in brucellosis vaccines. Vaccine 2019, 37, 3981–3988. [Google Scholar] [CrossRef]
- Wareth, G.; Eravci, M.; Weise, C.; Roesler, U.; Melzer, F.; Sprague, L.D.; Neubauer, H.; Murugaiyan, J. Comprehensive identification of immunodominant proteins of Brucella abortus and Brucella melitensis using antibodies in the sera from naturally infected hosts. Int. J. Mol. Sci. 2016, 17. [Google Scholar] [CrossRef]
- Lee, J.J.; Simborio, H.L.; Reyes, A.W.; Kim, D.G.; Hop, H.T.; Min, W.; Her, M.; Jung, S.C.; Yoo, H.S.; Kim, S. Immunoproteomic identification of immunodominant antigens independent of the time of infection in Brucella abortus 2308-challenged cattle. Vet. Res. 2015, 46, 17. [Google Scholar] [CrossRef][Green Version]
- Brotz-Oesterhelt, H.; Bandow, J.E.; Labischinski, H. Bacterial proteomics and its role in antibacterial drug discovery. Mass Spectrom. Rev. 2005, 24, 549–565. [Google Scholar] [CrossRef]
- Schmidt, F.; Volker, U. Proteome analysis of host-pathogen interactions: Investigation of pathogen responses to the host cell environment. Proteomics 2011, 11, 3203–3211. [Google Scholar] [CrossRef]
- Poetsch, A.; Marchesini, M.I. Proteomics of Brucella. Proteomes 2020, 8, 8. [Google Scholar] [CrossRef]
- Alton, G.G.; Jones, L.M.; Angus, R.D.; Verger, J.M. Techniques for the Brucellosis Laboratory; Institut Tional de la Recherche Agronomique: Paris, France, 1988; pp. 17–62. [Google Scholar]
- Yagupsky, P.; Morata, P.; Colmenero, J.D. Laboratory diagnosis of human brucellosis. Clin. Microbiol. Rev. 2019, 33. [Google Scholar] [CrossRef]
- Dadar, M.; Shahali, Y.; Wareth, G. Molecular diagnosis of acute and chronic brucellosis in humans. In Microbial Technology for the Welfare of Society; Arora, P.K., Ed.; Springer: Singapore, 2019; pp. 223–245. [Google Scholar] [CrossRef]
- Bricker, B.J.; Halling, S.M. Differentiation of Brucella abortus bv. 1, 2, and 4, Brucella melitensis, Brucella ovis, and Brucella suis bv. 1 by PCR. J. Clin. Microbiol. 1994, 32, 2660–2666. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Goni, I.; Garcia-Yoldi, D.; Marin, C.M.; de Miguel, M.J.; Barquero-Calvo, E.; Guzman-Verri, C.; Albert, D.; Garin-Bastuji, B. New Bruce-ladder multiplex PCR assay for the biovar typing of Brucella suis and the discrimination of Brucella suis and Brucella canis. Vet. Microbiol. 2011, 154, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Al Dahouk, S.; Fleche, P.L.; Nockler, K.; Jacques, I.; Grayon, M.; Scholz, H.C.; Tomaso, H.; Vergnaud, G.; Neubauer, H. Evaluation of Brucella MLVA typing for human brucellosis. J. Microbiol. Methods 2007, 69, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Sacchini, L.; Wahab, T.; Di Giannatale, E.; Zilli, K.; Abass, A.; Garofolo, G.; Janowicz, A. Whole genome sequencing for tracing geographical origin of imported cases of human brucellosis in Sweden. Microorganisms 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Lusk Pfefer, T.S.; Timme, R.; Kase, J.A. Identification of Brucella genus and eight Brucella species by Luminex bead-based suspension array. Food Microbiol. 2018, 70, 113–119. [Google Scholar] [CrossRef]
- Lasch, P.; Wahab, T.; Weil, S.; Palyi, B.; Tomaso, H.; Zange, S.; Kiland Granerud, B.; Drevinek, M.; Kokotovic, B.; Wittwer, M.; et al. Identification of highly pathogenic microorganisms by matrix-assisted laser desorption ionization-time of flight mass spectrometry: Results of an interlaboratory ring trial. J. Clin. Microbiol. 2015, 53, 2632–2640. [Google Scholar] [CrossRef]
- Bardon, J.; Stromerova, N. Identification of zoonotic bacterial pathogens by the MALDI TOF MS method. Klin. Mikrobiol. Infekc. Lek. 2015, 21, 46–50. [Google Scholar]
- Drevinek, M.; Dresler, J.; Klimentova, J.; Pisa, L.; Hubalek, M. Evaluation of sample preparation methods for MALDI-TOF MS identification of highly dangerous bacteria. Lett. Appl. Microbiol. 2012, 55, 40–46. [Google Scholar] [CrossRef]
- Karger, A.; Melzer, F.; Timke, M.; Bettin, B.; Kostrzewa, M.; Nockler, K.; Hohmann, A.; Tomaso, H.; Neubauer, H.; Al Dahouk, S. Interlaboratory comparison of intact-cell matrix-assisted laser desorption ionization-time of flight mass spectrometry results for identification and differentiation of Brucella spp. J. Clin. Microbiol. 2013, 51, 3123–3126. [Google Scholar] [CrossRef]
- Lista, F.; Reubsaet, F.A.; De Santis, R.; Parchen, R.R.; de Jong, A.L.; Kieboom, J.; van der Laaken, A.L.; Voskamp-Visser, I.A.; Fillo, S.; Jansen, H.J.; et al. Reliable identification at the species level of Brucella isolates with MALDI-TOF-MS. BMC Microbiol. 2011, 11, 267. [Google Scholar] [CrossRef]
- Bardon, J.; Kohnova, I.; Prokes, Z.; Skalka, P.; Bzdil, J. Direct detection of the brucellosis-causing pathogen—A case report. Klin. Mikrobiol. Infekc. Lek. 2011, 17, 50–54. [Google Scholar] [PubMed]
- Ferreira, L.; Vega Castano, S.; Sanchez-Juanes, F.; Gonzalez-Cabrero, S.; Menegotto, F.; Orduna-Domingo, A.; Gonzalez-Buitrago, J.M.; Munoz-Bellido, J.L. Identification of Brucella by MALDI-TOF mass spectrometry. Fast and reliable identification from agar plates and blood cultures. PLoS ONE 2010, 5, e14235. [Google Scholar] [CrossRef] [PubMed]
- Nyvang Hartmeyer, G.; Kvistholm Jensen, A.; Bocher, S.; Damkjaer Bartels, M.; Pedersen, M.; Engell Clausen, M.; Abdul-Redha, R.; Dargis, R.; Schouenborg, P.; Hojlyng, N.; et al. Mass spectrometry: Pneumococcal meningitis verified and Brucella species identified in less than half an hour. Scand. J. Infect. Dis. 2010, 42, 716–718. [Google Scholar] [CrossRef]
- Murugaiyan, J.; Roesler, U. MALDI-TOF MS profiling-advances in species identification of pests, parasites, and vectors. Front. Cell. Infect. Microbiol. 2017, 7, 184. [Google Scholar] [CrossRef]
- Suarez, S.; Ferroni, A.; Lotz, A.; Jolley, K.A.; Guerin, P.; Leto, J.; Dauphin, B.; Jamet, A.; Maiden, M.C.; Nassif, X.; et al. Ribosomal proteins as biomarkers for bacterial identification by mass spectrometry in the clinical microbiology laboratory. J. Microbiol. Methods 2013, 94, 390–396. [Google Scholar] [CrossRef]
- Sali, M.; De Maio, F.; Tarantino, M.; Garofolo, G.; Tittarelli, M.; Sacchini, L.; Zilli, K.; Pasquali, P.; Petrucci, P.; Marianelli, C.; et al. Rapid and safe one-step extraction method for the identification of Brucella strains at genus and species level by MALDI-TOF mass spectrometry. PLoS ONE 2018, 13, e0197864. [Google Scholar] [CrossRef]
- Mesureur, J.; Arend, S.; Celliere, B.; Courault, P.; Cotte-Pattat, P.J.; Totty, H.; Deol, P.; Mick, V.; Girard, V.; Touchberry, J.; et al. A MALDI-TOF MS database with broad genus coverage for species-level identification of Brucella. PLoS Negl. Trop. Dis. 2018, 12, e0006874. [Google Scholar] [CrossRef]
- Berendsen, E.M.; Levin, E.; Braakman, R.; Prodan, A.; van Leeuwen, H.C.; Paauw, A. Untargeted accurate identification of highly pathogenic bacteria directly from blood culture flasks. Int. J. Med. Microbiol. Ijmm 2020, 310, 151376. [Google Scholar] [CrossRef]
- Khan, A.U.; Shell, W.S.; Melzer, F.; Sayour, A.E.; Ramadan, E.S.; Elschner, M.C.; Moawad, A.A.; Roesler, U.; Neubauer, H.; El-Adawy, H. Identification, genotyping and antimicrobial susceptibility testing of Brucella spp. isolated from livestock in Egypt. Microorganisms 2019, 7. [Google Scholar] [CrossRef]
- Choudhary, P.; Shringi, B.N.; Choudhary, O.P.; Kashyap, S.K. MALDI-TOF mass spectrometry based identification of Brucella abortus in bovines of Western Rajasthan. Intern. J. Appl. Res. Vet. Med. 2019, 17, 108–120. [Google Scholar]
- Purvis, T.J.; Krouse, D.; Miller, D.; Livengood, J.; Thirumalapura, N.R.; Tewari, D. Detection of Brucella canis infection in dogs by blood culture and bacterial identification using matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J. Vet. Diagn. Investig. 2017, 29, 586–588. [Google Scholar] [CrossRef]
- Ulshina, D.V.; Kovalev, D.A.; Zhirov, A.M.; Zharinova, N.V.; Khudoleev, A.A.; Kogotkova, O.I.; Efremenko, V.I.; Evchenko, N.I.; Kulichenko, A.N. Features of mass-spectrometric protein profiles of strains of brucellosis causative agent during preparation of culture on various nutrient media. Zh Mikrobiol. Epidemiol. Immunobiol. 2016, 1, 29–34. [Google Scholar] [CrossRef]
- Mesureur, J.; Ranaldi, S.; Monnin, V.; Girard, V.; Arend, S.; Welker, M.; O’Callaghan, D.; Lavigne, J.P.; Keriel, A. A simple and safe protocol for preparing Brucella samples for matrix-assisted laser desorption ionization-time of flight mass spectrometry analysis. J. Clin. Microbiol. 2016, 54, 449–452. [Google Scholar] [CrossRef]
- Cunningham, S.A.; Patel, R. Importance of using Bruker’s security-relevant library for Biotyper identification of Burkholderia pseudomallei, Brucella species, and Fr. Tularensis. J. Clin. Microbiol. 2013, 51, 1639–1640. [Google Scholar] [CrossRef]
- Morris, J. The use of polyacrylamide gel electrophoresis in taxonomy of Brucella. J. Gen. Microbiol. 1973, 76, 231–237. [Google Scholar] [CrossRef][Green Version]
- Verstreate, D.; Winter, A.J. Comparison of sodium dodecyl sulfate-polyacrylamide gel electrophoresis profiles and antigenic relatedness among outer membrane proteins of 49 Brucella abortus strains. Infect. Immun. 1984, 46, 182–187. [Google Scholar] [CrossRef]
- Verstreate, D.R.; Creasy, M.T.; Caveney, N.T.; Baldwin, C.L.; Blab, M.W.; Winter, A.J. Outer membrane proteins of Brucella abortus: Isolation and characterization. Infect. Immun. 1982, 35, 979–989. [Google Scholar] [CrossRef]
- Santos, J.; Verstreate, D.; Perera, V.; Winter, A.J. Outer membrane proteins from rough strains of four Brucella species. Infect. Immun. 1984, 46, 188–194. [Google Scholar] [CrossRef]
- Gamazo, C.; Winter, A.J.; Moriyón, I.; Riezu-Boj, J.; Blasco, J.; Díaz, R. Comparative analyses of proteins extracted by hot saline or released spontaneously into outer membrane blebs from field strains of Brucella ovis and Brucella melitensis. Infect. Immun. 1989, 57, 1419–1426. [Google Scholar] [CrossRef]
- Brooks-Worrell, B.; Splitter, G. Sodium dodecyl sulfate- and salt-extracted antigens from various Brucella species induce proliferation of bovine lymphocytes. Infect. Immun. 1992, 60, 2136–2138. [Google Scholar] [CrossRef]
- Hill, R.; Cook, D. Protein profiles of Brucella suis and Brucella abortus in isoelectric focusing and sodium dodecyl sulphate-polyacrylamide gel electrophoresis. Vet. Microbiol. 1994, 39, 25–32. [Google Scholar] [CrossRef]
- Tabatabai, L.; Deyoe, B. Characterization of salt-extractable protein antigens from Brucella abortus by crossed immunoelectrophoresis and isoelectricfocusing. Vet. Microbiol. 1984, 9, 549–560. [Google Scholar] [CrossRef]
- Gorg, A.; Weiss, W.; Dunn, M.J. Current two-dimensional electrophoresis technology for proteomics. Proteomics 2004, 4, 3665–3685. [Google Scholar] [CrossRef] [PubMed]
- Teixeira-Gomes, A.P.; Cloeckaert, A.; Bezard, G.; Dubray, G.; Zygmunt, M.S. Mapping and identification of Brucella melitensis proteins by two-dimensional electrophoresis and microsequencing. Electrophoresis 1997, 18, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Teixeira-Gomes, A.P.; Cloeckaert, A.; Bezard, G.; Bowden, R.A.; Dubray, G.; Zygmunt, M.S. Identification and characterization of Brucella ovis immunogenic proteins using two-dimensional electrophoresis and immunoblotting. Electrophoresis 1997, 18, 1491–1497. [Google Scholar] [CrossRef]
- Lin, J.; Ficht, T.A. Protein synthesis in Brucella abortus induced during macrophage infection. Infect. Immun. 1995, 63, 1409–1414. [Google Scholar] [CrossRef]
- Sowa, B.A.; Kelly, K.A.; Ficht, T.A.; Adams, L.G. Virulence associated proteins of Brucella abortus identified by paired two-dimensional gel electrophoretic comparisons of virulent, vaccine and LPS deficient strains. Appl. Theor. Electrophor. 1992, 3, 33–40. [Google Scholar]
- Rafie-Kolpin, M.; Essenberg, R.C.; Wyckoff, J.H., 3rd. Identification and comparison of macrophage-induced proteins and proteins induced under various stress conditions in Brucella abortus. Infect. Immun. 1996, 64, 5274–5283. [Google Scholar] [CrossRef]
- Wagner, M.A.; Eschenbrenner, M.; Horn, T.A.; Kraycer, J.A.; Mujer, C.V.; Hagius, S.; Elzer, P.; DelVecchio, V.G. Global analysis of the Brucella melitensis proteome: Identification of proteins expressed in laboratory-grown culture. Proteomics 2002, 2, 1047–1060. [Google Scholar] [CrossRef]
- Mujer, C.V.; Wagner, M.A.; Eschenbrenner, M.; Horn, T.; Kraycer, J.A.; Redkar, R.; Hagius, S.; Elzer, P.; Delvecchio, V.G. Global analysis of Brucella melitensis proteomes. Ann. N. Y. Acad. Sci. 2002, 969, 97–101. [Google Scholar] [CrossRef]
- Eschenbrenner, M.; Wagner, M.A.; Horn, T.A.; Kraycer, J.A.; Mujer, C.V.; Hagius, S.; Elzer, P.; DelVecchio, V.G. Comparative proteome analysis of Brucella melitensis vaccine strain Rev 1 and a virulent strain, 16M. J. Bacteriol. 2002, 184, 4962–4970. [Google Scholar] [CrossRef] [PubMed]
- Eschenbrenner, M.; Horn, T.A.; Wagner, M.A.; Mujer, C.V.; Miller-Scandle, T.L.; DelVecchio, V.G. Comparative proteome analysis of laboratory grown Brucella abortus 2308 and Brucella melitensis 16M. J. Proteome Res. 2006, 5, 1731–1740. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Lim, J.J.; Kim, D.G.; Simborio, H.L.; Kim, D.H.; Reyes, A.W.; Min, W.; Lee, H.J.; Kim, D.H.; Chang, H.H.; et al. Characterization of culture supernatant proteins from Brucella abortus and its protection effects against murine brucellosis. Comp. Immunol. Microbiol. Infect. Dis. 2014, 37, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Al Dahouk, S.; Jubier-Maurin, V.; Scholz, H.C.; Tomaso, H.; Karges, W.; Neubauer, H.; Kohler, S. Quantitative analysis of the intramacrophagic Brucella suis proteome reveals metabolic adaptation to late stage of cellular infection. Proteomics 2008, 8, 3862–3870. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Cervantes, V.; Flores-Mejia, R.; Moreno-Lafont, M.C.; Lanz-Mendoza, H.; Tello-Lopez, A.T.; Castillo-Vera, J.; Pando-Robles, V.; Hurtado-Sil, G.; Gonzalez-Gonzalez, E.; Rodriguez-Cortes, O.; et al. Comparative proteome analysis of Brucella abortus 2308 and its virB type IV secretion system mutant reveals new T4SS-related candidate proteins. J. Proteom. 2011, 74, 2959–2971. [Google Scholar] [CrossRef]
- Al Dahouk, S.; Jubier-Maurin, V.; Neubauer, H.; Kohler, S. Quantitative analysis of the Brucella suis proteome reveals metabolic adaptation to long-term nutrient starvation. BMC Microbiol. 2013, 13, 199. [Google Scholar] [CrossRef]
- Wu, Y.; Jin, Y.; Pan, W.; Ye, C.; Sun, X.; Sun, Y.; Hu, B.; Zhou, J. Comparative proteomics analysis of host cells infected with Brucella abortus A19. Electrophoresis 2014, 35, 1130–1143. [Google Scholar] [CrossRef]
- Fu, Y.; Wang, Z.; Lu, B.; Zhao, S.; Zhang, Y.; Zhao, Z.; Zhang, C.; Li, J.; Zhou, B.; Guo, Z.; et al. Immune response and differentially expressed proteins in the lung tissue of BALB/c mice challenged by aerosolized Brucella melitensis 5. J. Int. Med. Res. 2018, 46, 4740–4752. [Google Scholar] [CrossRef]
- Lamontagne, J.; Forest, A.; Marazzo, E.; Denis, F.; Butler, H.; Michaud, J.F.; Boucher, L.; Pedro, I.; Villeneuve, A.; Sitnikov, D.; et al. Intracellular adaptation of Brucella abortus. J. Proteome Res. 2009, 8, 1594–1609. [Google Scholar] [CrossRef]
- Zai, X.; Yang, Q.; Yin, Y.; Li, R.; Qian, M.; Zhao, T.; Li, Y.; Zhang, J.; Fu, L.; Xu, J.; et al. Relative quantitative proteomic analysis of Brucella abortus reveals metabolic adaptation to multiple environmental stresses. Front. Microbiol. 2017, 8, 2347. [Google Scholar] [CrossRef]
- Zai, X.; Yang, Q.; Liu, K.; Li, R.; Qian, M.; Zhao, T.; Li, Y.; Yin, Y.; Dong, D.; Fu, L.; et al. A comprehensive proteogenomic study of the human Brucella vaccine strain 104 M. BMC Genom. 2017, 18, 402. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Tian, M.; Hu, H.; Yin, Y.; Guan, X.; Ding, C.; Wang, S.; Yu, S. Lable-free based comparative proteomic analysis of secretory proteins of rough Brucella mutants. J. Proteom. 2019, 195, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Araiza-Villanueva, M.; Avila-Calderon, E.D.; Flores-Romo, L.; Calderon-Amador, J.; Sriranganathan, N.; Qublan, H.A.; Witonsky, S.; Aguilera-Arreola, M.G.; Ruiz-Palma, M.D.S.; Ruiz, E.A.; et al. Proteomic analysis of membrane blebs of Brucella abortus 2308 and RB51 and their evaluation as an acellular vaccine. Front. Microbiol. 2019, 10, 2714. [Google Scholar] [CrossRef] [PubMed]
- Wattam, A.R.; Williams, K.P.; Snyder, E.E.; Almeida, N.F., Jr.; Shukla, M.; Dickerman, A.W.; Crasta, O.R.; Kenyon, R.; Lu, J.; Shallom, J.M.; et al. Analysis of ten Brucella genomes reveals evidence for horizontal gene transfer despite a preferred intracellular lifestyle. J. Bacteriol. 2009, 191, 3569–3579. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; Leng, D.; Burk, C.; Nakajima-Sasaki, R.; Kayala, M.A.; Atluri, V.L.; Pablo, J.; Unal, B.; Ficht, T.A.; Gotuzzo, E.; et al. Large scale immune profiling of infected humans and goats reveals differential recognition of Brucella melitensis antigens. PLoS Negl. Trop. Dis. 2010, 4, e673. [Google Scholar] [CrossRef] [PubMed]
- Wareth, G.; Melzer, F.; Weise, C.; Neubauer, H.; Roesler, U.; Murugaiyan, J. Proteomics-based identification of immunodominant proteins of Brucellae using sera from infected hosts points towards enhanced pathogen survival during the infection. Biochem. Biophys. Res. Commun. 2015, 456, 202–206. [Google Scholar] [CrossRef]
- Al Dahouk, S.; Nöckler, K.; Scholz, H.C.; Tomaso, H.; Bogumil, R.; Neubauer, H. Immunoproteomic characterization of Brucella abortus 1119-3 preparations used for the serodiagnosis of Brucella infections. J. Immunol. Methods 2006, 309, 34–47. [Google Scholar] [CrossRef]
- Connolly, J.P.; Comerci, D.; Alefantis, T.G.; Walz, A.; Quan, M.; Chafin, R.; Grewal, P.; Mujer, C.V.; Ugalde, R.A.; DelVecchio, V.G. Proteomic analysis of Brucella abortus cell envelope and identification of immunogenic candidate proteins for vaccine development. Proteomics 2006, 6, 3767–3780. [Google Scholar] [CrossRef]
- Ko, K.Y.; Kim, J.W.; Her, M.; Kang, S.I.; Jung, S.C.; Cho, D.H.; Kim, J.Y. Immunogenic proteins of Brucella abortus to minimize cross reactions in brucellosis diagnosis. Vet. Microbiol. 2012, 156, 374–380. [Google Scholar] [CrossRef]
- Yang, Y.; Wang, L.; Yin, J.; Wang, X.; Cheng, S.; Lang, X.; Wang, X.; Qu, H.; Sun, C.; Wang, J.; et al. Immunoproteomic analysis of Brucella melitensis and identification of a new immunogenic candidate protein for the development of brucellosis subunit vaccine. Mol. Immunol. 2011, 49, 175–184. [Google Scholar] [CrossRef]
- Zhao, Z.; Yan, F.; Ji, W.; Luo, D.; Liu, X.; Xing, L.; Duan, Y.; Yang, P.; Shi, X.; Lu, Z.; et al. Identification of immunoreactive proteins of Brucella melitensis by immunoproteomics. Sci. China Life Sci. 2011, 54, 880–887. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Simborio, H.L.; Reyes, A.W.; Kim, D.G.; Hop, H.T.; Min, W.; Her, M.; Jung, S.C.; Yoo, H.S.; Kim, S. Proteomic analyses of the time course responses of mice infected with Brucella abortus 544 reveal immunogenic antigens. Fems Microbiol. Lett. 2014, 357, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Dorneles, E.M.; Sriranganathan, N.; Lage, A.P. Recent advances in Brucella abortus vaccines. Vet. Res. 2015, 46, 76. [Google Scholar] [CrossRef] [PubMed]
- Huy, T.X.N.; Bernardo Reyes, A.W.; Vu, S.H.; Arayan, L.T.; Hop, H.T.; Min, W.; Lee, H.J.; Lee, J.H.; Kim, S. Immunogenicity and protective response induced by recombinant Brucella abortus proteins Adk, SecB and combination of these two recombinant proteins against a virulent strain B. abortus 544 infection in BALB/c mice. Microb. Pathog. 2020, 143, 104137. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Song, M.; Yin, Y.; Lian, Z.; Li, Z.; Hu, H.; Guan, X.; Cai, Y.; Ding, C.; Wang, S.; et al. Characterization of the main immunogenic proteins in Brucella infection for their application in diagnosis of brucellosis. Comp. Immunol. Microbiol. Infect. Dis. 2020, 70, 101462. [Google Scholar] [CrossRef] [PubMed]

| No. | Year | Brucella spp. Used in Each Study | No. of Strains | Aim of the Study | Study |
|---|---|---|---|---|---|
| 1 | 2020 | B. abortus, B. melitensis, B. suis | 3, 4 and 3 | Diagnosis | [40] |
| 2 | 2019 | B. abortus, B. melitensis | 29 | Diagnosis | [41] |
| 3 | 2019 | B. abortus | 5 | Diagnosis | [42] |
| 4 | 2018 | B. abortus, B. melitensis, B. suis, B. canis, B. canis, B. ovis, B. neotomae | 75 | Database creation | [38] |
| 5 | 2018 | B. abortus, B. melitensis, B. suis, B. canis, B. ceti, B. inopinata, B. microti, B. ovis, B. pinnipedialis, and B. neotomae, B. papionis | 84 | Database creation | [39] |
| 6 | 2017 | B. canis | 38 | Diagnosis | [43] |
| 7 | 2016 | B. melitensis Rev-1 and B. abortus 19BA | 2 | Comparative analysis | [44] |
| 8 | 2016 | B. abortus, B. melitensis, B. suis, B. canis, B. neotomae, B. inopinata, B. microti, B. ceti, B. ovis, B. pinnipedialis, and B. papionis | 15 reference strains | Protocol for sample preparation | [45] |
| 9 | 2015 | B. canis | 1 | Case diagnosis | [28] |
| 10 | 2015 | B. melitensis and B. suis | 19 | Diagnosis | [29] |
| 11 | 2013 | B. abortus, B. melitensis, B. suis, B. canis, B. ceti, B. inopinata, B. microti, B. ovis, B. pinnipedialis, and B. neotomae | 104 field and 33 reference strains | Interlaboratory comparison | [31] |
| 12 | 2013 | B. melitensis and B. suis | 6 and 3 | Diagnosis | [46] |
| 13 | 2012 | B. melitensis | 1 | Sample preparation | [30] |
| 14 | 2011 | B. abortus, B. melitensis, B. suis, B. canis, B. ceti, B. neotomae, B. ovis and B. pinnipedialis | 170 | Diagnosis, Database creation | [32] |
| 15 | 2011 | B. melitensis | 1 | Case diagnosis | [33] |
| 16 | 2010 | B. abortus, B. melitensis, B. suis, B. canis, B. ceti, B. pinnipedialis | 131 | Generating reference spectra | [34] |
| 17 | 2010 | B. melitensis | 1 | Case diagnosis | [35] |
| No. | Brucella spp. Used | Source of Anti-Sera Used | No. of Immunoreactive Proteins Identified | Technique Applied | Year | Ref. |
|---|---|---|---|---|---|---|
| 1 | B. abortusB. melitensis clinical strains | Cattle, buffaloes, sheep and goats | 25 proteins of B. abortus and 20 proteins of B. melitensis | 2D immunoblotting, MALDI—TOF MS | 2016 | [15] |
| 2 | B. abortusB. melitensis clinical strains | Cattle, buffaloes, sheep and goats | Eight proteins of B. abortus and ten proteins of B. melitensis | SDS-PAGE Western blotting, MALDI—TOF MS | 2015 | [78] |
| 3 | B. abortus 544 | B. abortus-infected cattle | 134, 110 and 106 proteins were recognized at 3, 7 and 10 weeks of infection, and 55 common antigens | 2D immunoblotting, MALDI—TOF MS | 2015 | [16] |
| 4 | B. abortus 544 | Experimentally infected mice | 13, 24 and 55 immunoreactive proteins were detected at 10, 30 and 60 days post-infection, respectively. MALDI identified 17 of the 67 proteins | 2D immunoblotting, MALDI—TOF MS | 2014 | [84] |
| 5 | B. abortus 1119-3 | Experimentally infected cattle with B. abortus 2308 | 18 immunogenic insoluble proteins showed immunoreactivity against positive sera | 2D immunoblotting, MALDI—TOF MS | 2012 | [81] |
| 6 | B. melitensis 16 M and Rev.1 | Pooled human and goat antisera | 23, 33 and 11 proteins reacted with human, goat and both sera, respectively. | 2D immunoblotting, LC—MS | 2011 | [82] |
| 7 | B. melitensis M5 vaccine strain | Pool of 15 bovine anti-Brucella-positive sera | 88 immunoreactive protein spots assigned to 61 proteins. 12 are immunogenic, 8 are virulence-related proteins | 2D immunoblotting, MALDI—TOF MS | 2011 | [83] |
| 8 | B. abortus 1119-3 | Hyper-immune antisera from rabbits | 17 immunoreactive out of 383 spots assigned to 6 proteins | 2D immunoblotting, MALDI—TOF MS and SELDI-MS | 2006 | [79] |
| 9 | B. abortus 2308 | Bovines and infected human patients with B. suis infection | 42 and 23 immunogenic spots identified against human and bovine sera, respectively | 2D immunoblotting, MALDI—TOF MS and LC—MS. | 2006 | [80] |
| 10 | B. ovis | Ram naturally infected with B. ovis | 82 reactive protein spots were assigned to 21 proteins | 2D immunoblotting and protein microsequencing | 1997 | [57] |
| 11 | B. melitensis B115 | Monoclonal antibodies (MAbs) | 25 protein spots from 595 protein spots separated by 2DE reacted | 2D immunoblotting, N-terminal microsequencing | 1997 | [56] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wareth, G.; Pletz, M.W.; Neubauer, H.; Murugaiyan, J. Proteomics of Brucella: Technologies and Their Applications for Basic Research and Medical Microbiology. Microorganisms 2020, 8, 766. https://doi.org/10.3390/microorganisms8050766
Wareth G, Pletz MW, Neubauer H, Murugaiyan J. Proteomics of Brucella: Technologies and Their Applications for Basic Research and Medical Microbiology. Microorganisms. 2020; 8(5):766. https://doi.org/10.3390/microorganisms8050766
Chicago/Turabian StyleWareth, Gamal, Mathias W. Pletz, Heinrich Neubauer, and Jayaseelan Murugaiyan. 2020. "Proteomics of Brucella: Technologies and Their Applications for Basic Research and Medical Microbiology" Microorganisms 8, no. 5: 766. https://doi.org/10.3390/microorganisms8050766
APA StyleWareth, G., Pletz, M. W., Neubauer, H., & Murugaiyan, J. (2020). Proteomics of Brucella: Technologies and Their Applications for Basic Research and Medical Microbiology. Microorganisms, 8(5), 766. https://doi.org/10.3390/microorganisms8050766







