Lactobacillus salivarius Subspecies salicinius SA-03 is a New Probiotic Capable of Enhancing Exercise Performance and Decreasing Fatigue
Abstract
1. Introduction
2. Materials and Methods
2.1. Lactobacillus salivarius subsp. salicinius (SA-03) Preparation
2.2. Animals and Experimental Design
2.3. Swimming Exercise Endurance Test
2.4. Forelimb Grip Strength
2.5. Determination of Fatigue-Associated Biochemical Variables
2.6. Resting Biochemical Profiles at the End of the Study
2.7. Body Composition, Glycogen Content, and Histopathology
2.8. Bacterial DNA Extraction and 16S rRNA Sequencing
2.9. Statistical Analysis
3. Results
3.1. Effect of SA-03 Supplementation on Body Weight, Body Composition, and Food and Water Intake
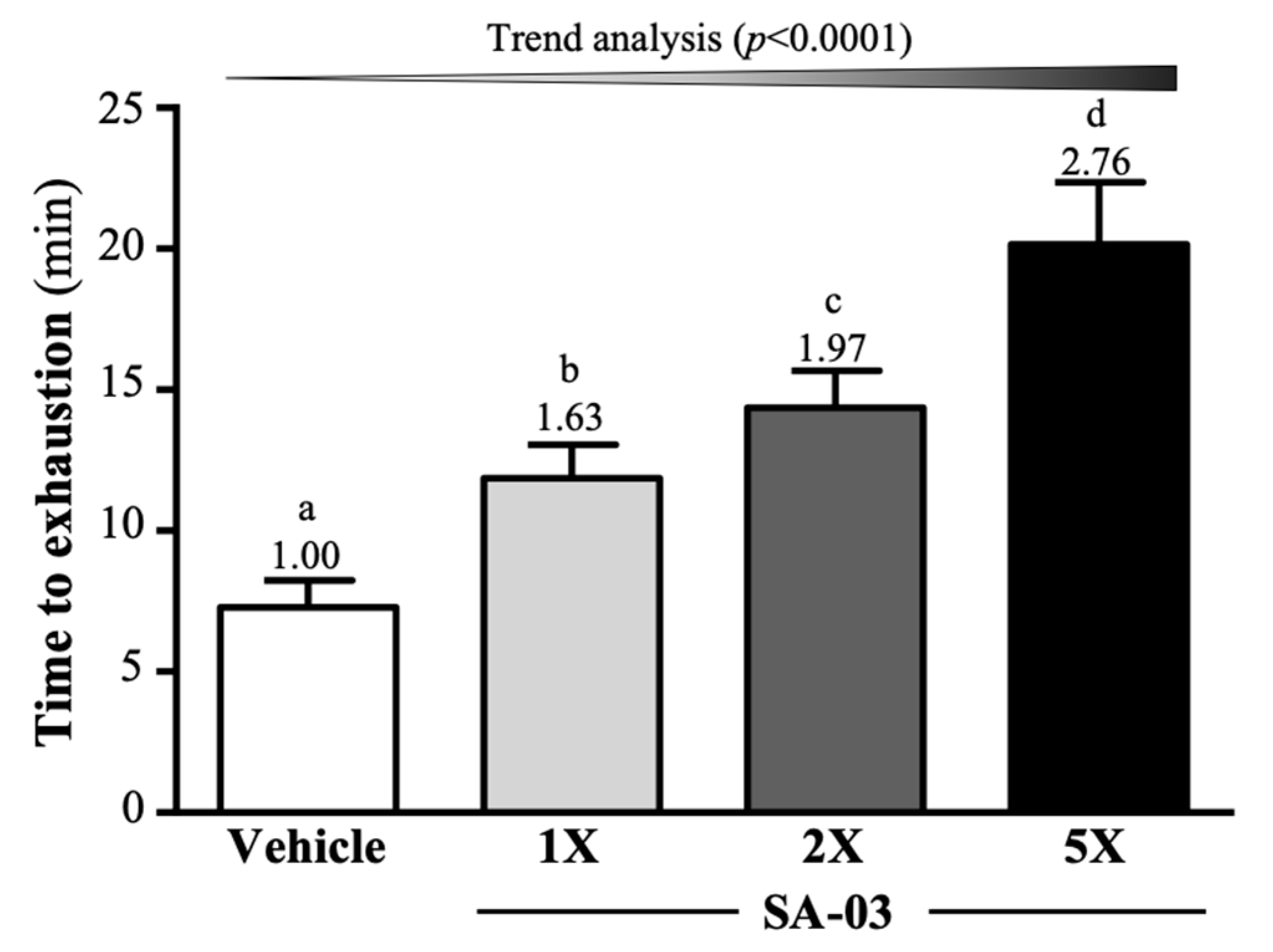
3.2. Effect of SA-03 Supplementation on Endurance Capacity
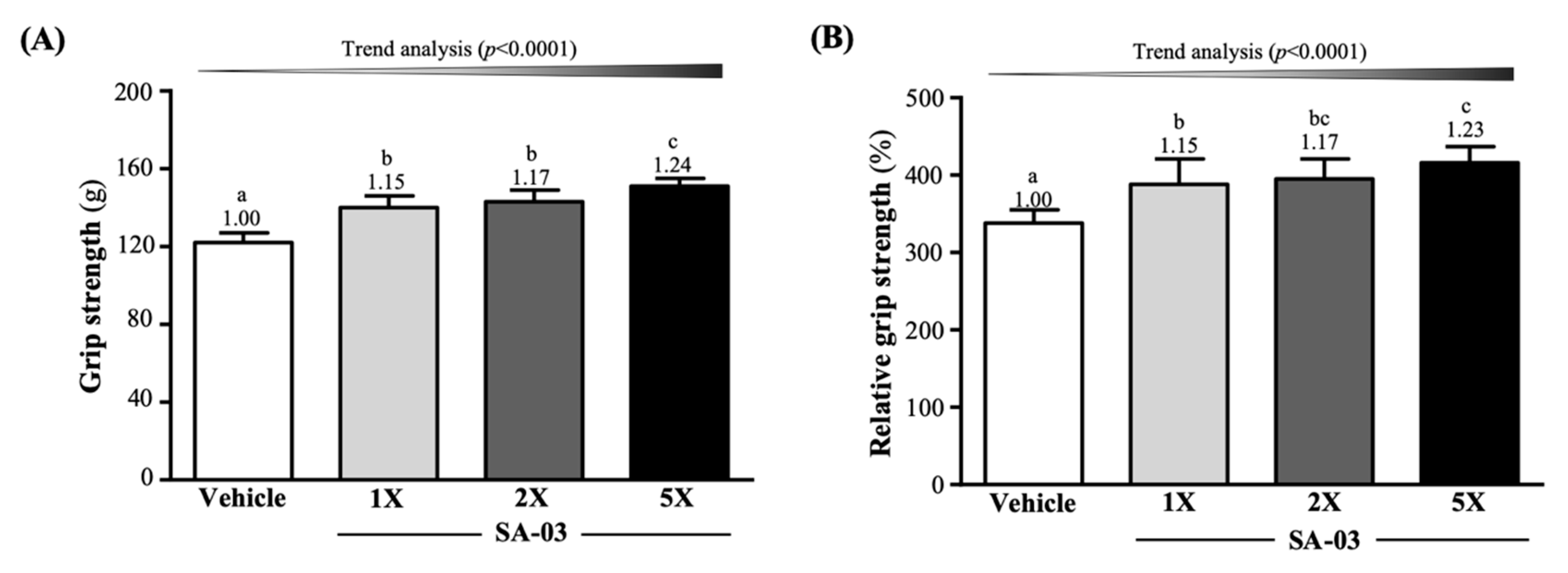
3.3. Effect of SA-03 Supplementation on Grip Strength
3.4. Effect of SA-03 Supplementation on Serum Lactate Levels after the 10-Min Swim Test
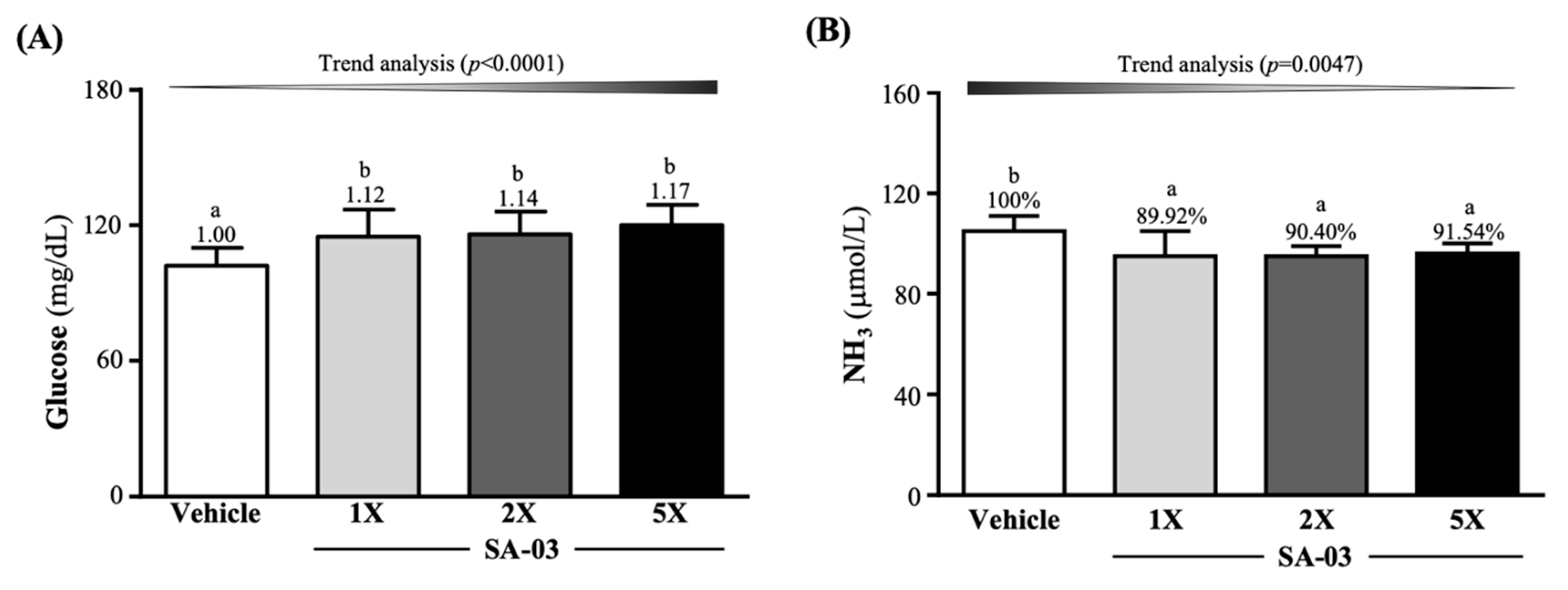
3.5. Effect of SA-03 Supplementation on Serum Glucose and Ammonia Levels after the 10 Min Swim Test
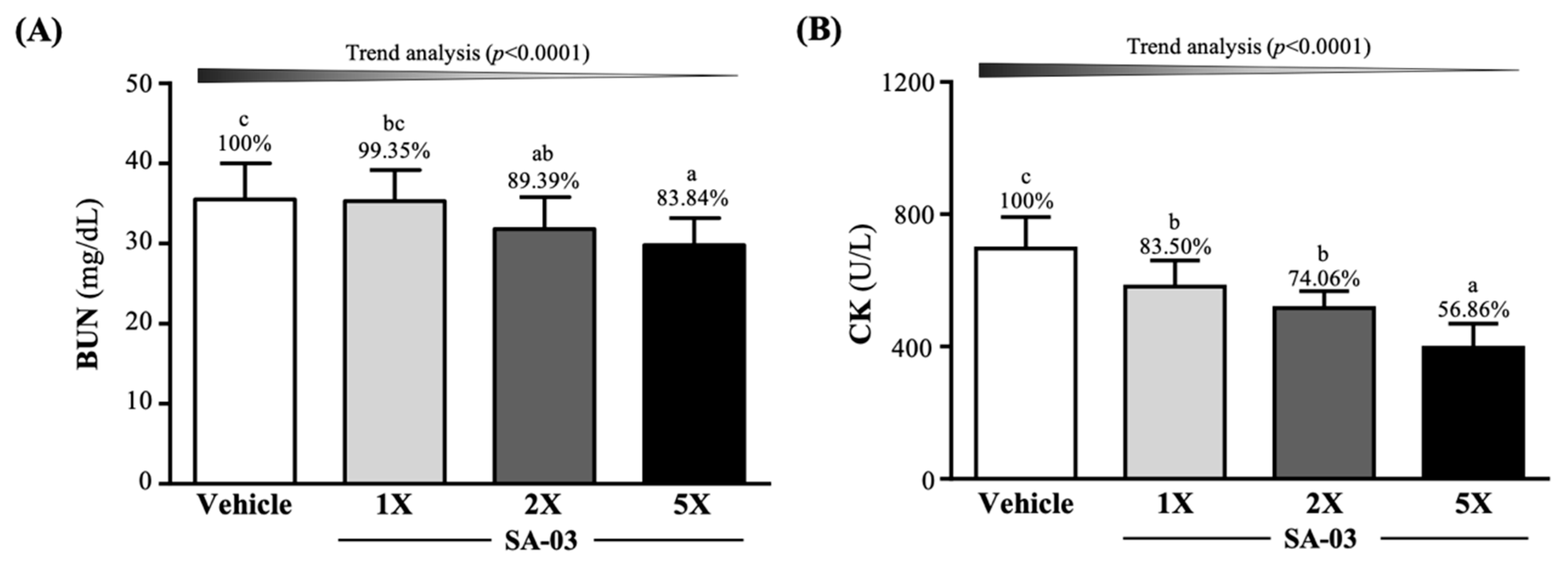
3.6. Effect of SA-03 Supplementation on Serum BUN and CK Levels after 90 Min Swimming and 60 Min Rest
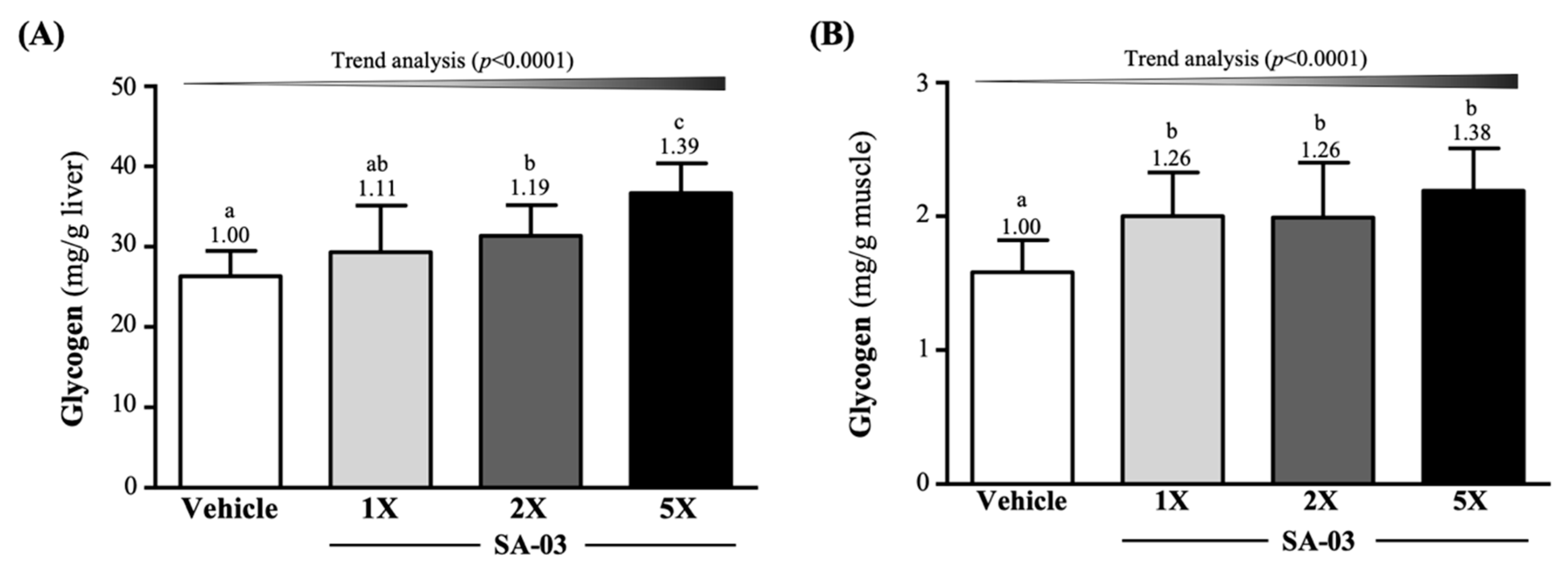
3.7. Effect of SA-03 Supplementation on Liver and Muscle Glycogen Levels
3.8. Effect of SA-03 Supplementation on Biochemical Profiles
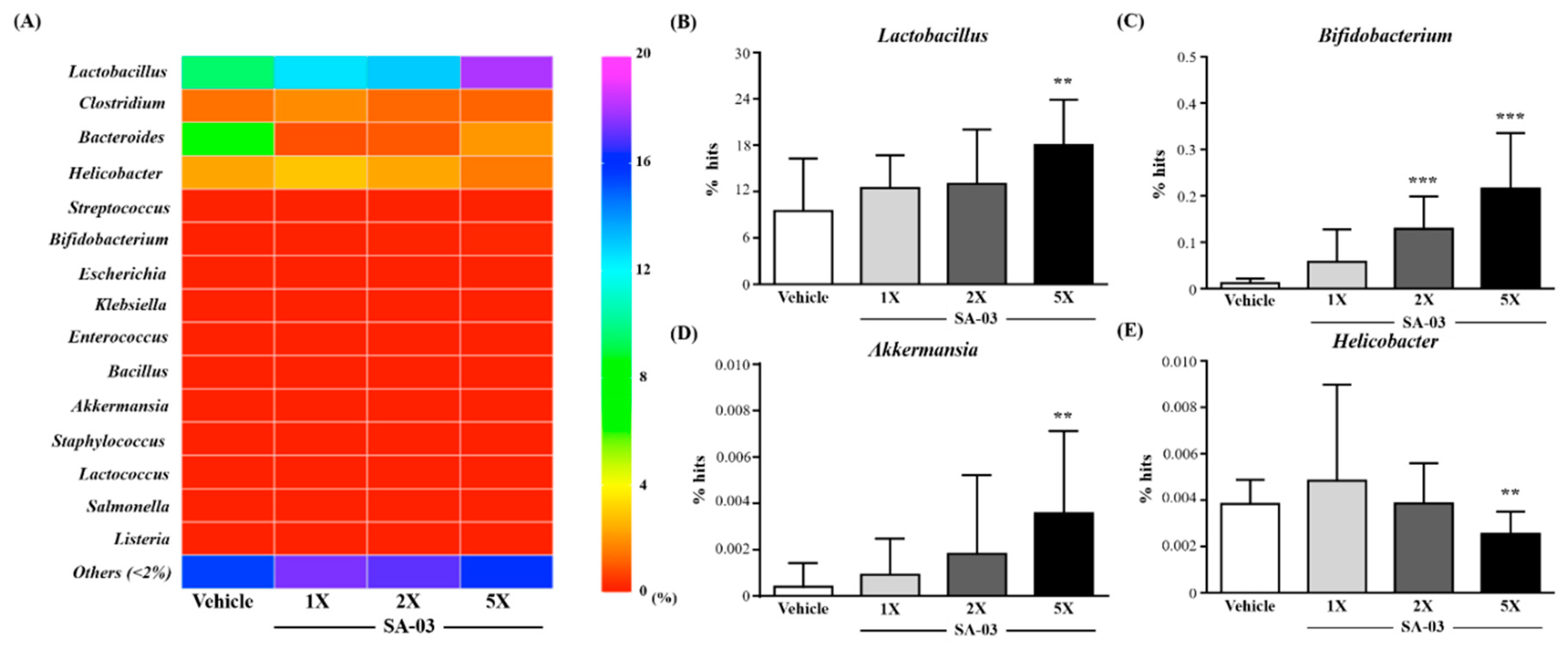
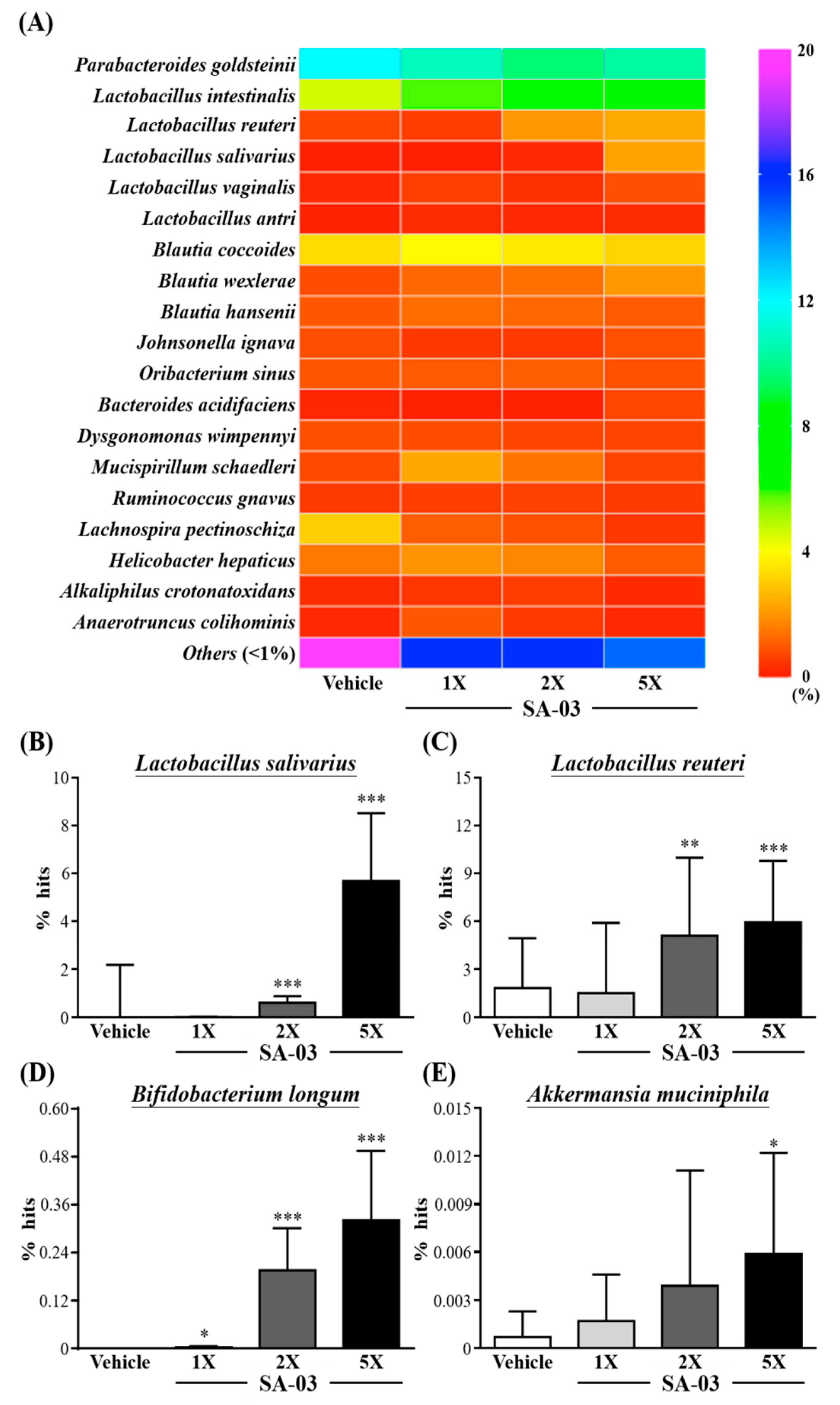
3.9. Effect of SA-03 Supplementation on the Gut Microbiota
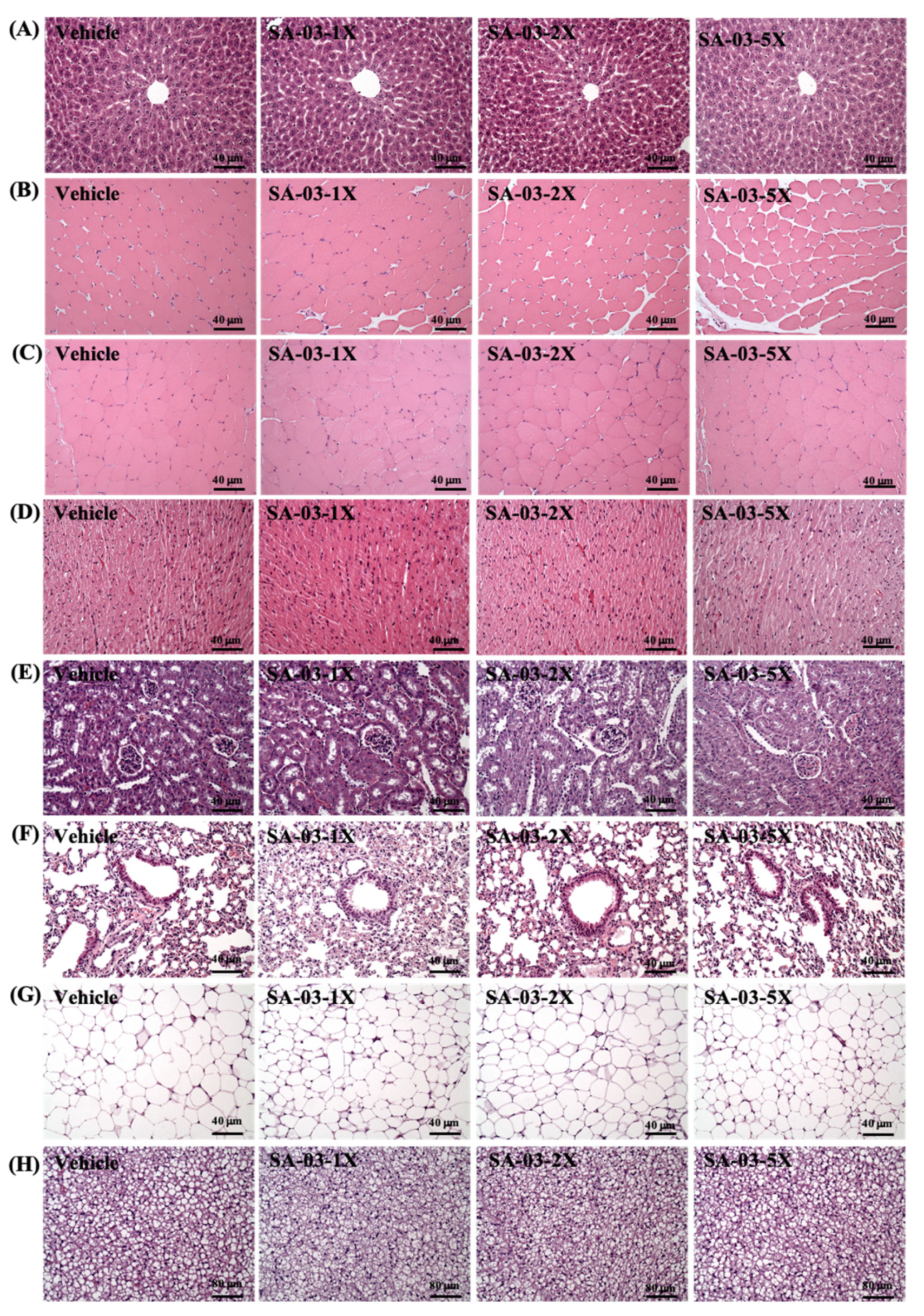
3.10. Effect of SA-03 Supplementation on Tissue Histology
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Davis, J.M. Central and peripheral factors in fatigue. J. Sports Sci. 1995, 13, S49–S53. [Google Scholar] [CrossRef] [PubMed]
- Zając, A.; Chalimoniuk, M.; Gołaś, A.; Lngfort, J.; Maszczyk, A. Central and peripheral fatigue during resistance exercise—A critical review. J. Hum. Kinet. 2015, 49, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.J.; Qin, Z.; Wang, P.Y.; Sun, Y.; Liu, X. Muscle fatigue: general understanding and treatment. Exp Mol. Med. 2017, 49, e384. [Google Scholar] [CrossRef] [PubMed]
- Mach, N.; Fuster-Botella, D. Endurance exercise and gut microbiota: A review. J. Sport Health Sci. 2017, 6, 179–197. [Google Scholar] [CrossRef] [PubMed]
- Leite, G.S.F.; Resende, A.S.; West, N.P.; Lancha, A.H., Jr. Probiotics and sports: A new magic bullet? Nutrition 2019, 60, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization of the United Nations; World Health Organization. Guidelines for the Evaluation of Probiotics in Food. In Proceedings of the Joint FAO/WHO Working Group Report on Drafting Guidelines for the Evaluation of Probiotics in Food, London, ON, Canada, 30 April–1 May 2002. [Google Scholar]
- Sivamaruthi, B.S.; Kesika, P.; Suganthy, N.; Chaiyasut, C. A Review on Role of Microbiome in Obesity and Antiobesity Properties of Probiotic Supplements. Biomed. Res. Int. 2019, 2019, 3291367. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Chen, Y.M.; Kan, N.W.; Ho, C.S.; Wei, L.; Chan, C.H.; Huang, C.C. Hypolipidemic Effects and Safety of Lactobacillus Reuteri 263 in a Hamster Model of Hyperlipidemia. Nutrients 2015, 7, 3767–3782. [Google Scholar] [CrossRef]
- Sichetti, M.; De Marco, S.; Pagiotti, R.; Traina, G.; Pietrella, D. Anti-inflammatory effect of multistrain probiotic formulation (L. rhamnosus, B. lactis, and B. longum). Nutrition 2018, 53, 95–102. [Google Scholar] [CrossRef]
- Kang, M.S.; Lim, H.S.; Oh, J.S.; Lim, Y.J.; Wuertz-Kozak, K.; Harro, J.M.; Shirtliff, M.E.; Achermann, Y. Antimicrobial activity of Lactobacillus salivarius and Lactobacillus fermentum against Staphylococcus aureus. Pathog. Dis. 2017, 75, 2. [Google Scholar] [CrossRef]
- Pourramezan, Z.; Kasra Kermanshahi, R.; Oloomi, M.; Aliahmadi, A.; Rezadoost, H. In vitro study of antioxidant and antibacterial activities of Lactobacillus probiotic spp. Folia Microbiol. (Praha) 2018, 63, 31–42. [Google Scholar] [CrossRef]
- Tiptiri-Kourpeti, A.; Spyridopoulou, K.; Santarmaki, V.; Aindelis, G.; Tompoulidou, E.; Lamprianidou, E.E.; Saxami, G.; Ypsilantis, P.; Lampri, E.S.; Simopoulos, C.; et al. Lactobacillus casei exerts anti-proliferative effects accompanied by apoptotic cell death and up-regulation of TRAIL in colon carcinoma cells. PLoS ONE 2016, 11, e0147960. [Google Scholar] [CrossRef] [PubMed]
- Nagpal, R.; Wang, S.; Ahmadi, S.; Hayes, J.; Gagliano, J.; Subashchandrabose, S.; Kitzman, D.W.; Becton, T.; Read, R.; Yadav, H. Human-origin probiotic cocktail increases short-chain fatty acid production via modulation of mice and human gut microbiome. Sci. Rep. 2018, 8, 12649. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.; Mach, N. The Crosstalk between the Gut Microbiota and Mitochondria during Exercise. Front. Physiol. 2017, 8, 319. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.M.; Wei, L.; Chiu, Y.S.; Hsu, Y.J.; Tsai, T.Y.; Wang, M.F.; Huang, C.C. Lactobacillus plantarum TWK10 Supplementation Improves Exercise Performance and Increases Muscle Mass in Mice. Nutrients 2016, 8, 205. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.C.; Hsu, Y.J.; Chuang, H.L.; Hsieh, P.S.; Ho, H.H.; Chen, W.L.; Chiu, Y.S.; Huang, C.C. In Vivo Ergogenic Properties of the Bifidobacterium longum OLP-01 Isolated from a Weightlifting Gold Medalist. Nutrients 2019, 11, 2003. [Google Scholar] [CrossRef]
- Rogosa, M.; Wiseman, R.F.; Mitchell, J.A.; Disraely, M.N.; Beaman, A.J. Species differentiation of oral lactobacilli from man including descriptions of Lactobacillus salivarius nov spec and Lactobacillus cellobiosus nov spec. J. Bacteriol. 1953, 65, 681–699. [Google Scholar] [CrossRef]
- Claesson, M.J.; Li, Y.; Leahy, S.; Canchaya, C.; van Pijkeren, J.P.; Cerdeño-Tárraga, A.M.; Parkhill, J.; Flynn, S.; O’Sullivan, G.C.; Collins, J.K.; et al. Multireplicon genome architecture of Lactobacillus salivarius. Proc. Natl. Acad. Sci. USA 2006, 103, 6718–6723. [Google Scholar] [CrossRef]
- Raftis, E.J.; Salvetti, E.; Torriani, S.; Felis, G.E.; O’Toole, P.W. Genomic diversity of Lactobacillus salivarius. J. Bacteriol. 2011, 77, 954–965. [Google Scholar]
- Neville, B.A.; O’Toole, P.W. Probiotic properties of Lactobacillus salivarius and closely related Lactobacillus species. Future Microbiol. 2010, 5, 759–774. [Google Scholar] [CrossRef]
- Axelrod, C.L.; Brennan, C.J.; Cresci, G.; Paul, D.; Hull, M.; Fealy, C.E.; Kirwan, J.P. UCC118 supplementation reduces exercise-induced gastrointestinal permeability and remodels the gut microbiome in healthy humans. Physiol. Rep. 2019, 7, e14276. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.E.; Huang, W.C.; Liao, C.C.; Chang, Y.K.; Kan, N.W.; Huang, C.C. Resveratrol protects against physical fatigue and improves exercise performance in mice. Molecules 2013, 18, 4689–4702. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Huang, H.Y.; Hsu, Y.J.; Su, W.H.; Shen, S.Y.; Lee, M.C.; Lin, C.L.; Huang, C.C. The Effects of Thiamine Tetrahydrofurfuryl Disulfide on Physiological Adaption and Exercise Performance Improvement. Nutrients 2018, 10, 851. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.W.; Hsu, Y.J.; Lee, M.C.; Li, H.S.; Yeo, P.C.W.; Lim, A.L.; Huang, C.C. In Vitro and In Vivo Functional Characterization of Essence of Chicken as An Ergogenic Aid. Nutrients. Nutrients 2018, 10, 1943. [Google Scholar] [CrossRef] [PubMed]
- Hsu, Y.J.; Huang, W.C.; Lin, J.S.; Chen, Y.M.; Ho, S.T.; Huang, C.C.; Tung, Y.T. Kefir Supplementation Modifies Gut Microbiota Composition, Reduces Physical Fatigue, and Improves Exercise Performance in Mice. Nutrients 2018, 10, 862. [Google Scholar] [CrossRef] [PubMed]
- Jäger, R.; Mohr, A.E.; Carpenter, K.C.; Kerksick, C.M.; Purpura, M.; Moussa, A.; Townsend, J.R.; Lamprecht, M.; West, N.P.; Black, K.; et al. International Society of Sports Nutrition Position Stand: Probiotics. J. Int. Soc. Sports Nutr. 2019, 16, 62. [Google Scholar] [CrossRef]
- Barton, W.; Penney, N.C.; Cronin, O.; Garcia-Perez, I.; Molloy, M.G.; Holmes, E.; Shanahan, F.; Cotter, P.D.; O’Sullivan, O. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 2018, 67, 625–633. [Google Scholar] [CrossRef]
- Petersen, L.M.; Bautista, E.J.; Nguyen, H.; Hanson, B.M.; Chen, L.; Lek, S.H.; Sodergren, E.; Weinstock, G.M. Community characteristics of the gut microbiomes of competitive cyclists. Microbiome 2017, 5, 98. [Google Scholar] [CrossRef]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef]
- Coqueiro, A.Y.; de Oliveira Garcia, A.B.; Rogero, M.M.; Tirapegui, J. Probiotic supplementation in sports and physical exercise: Does it present any ergogenic effect? Nutr. Health. 2017, 23, 239–249. [Google Scholar] [CrossRef]
- Bachmann, C. Mechanisms of hyperammonemia. Clin. Chem. Lab. Med. 2002, 40, 653–662. [Google Scholar] [CrossRef] [PubMed]
- Norenberg, M.D.; Rao, K.V.; Jayakumar, A.R. Mechanisms of ammonia-induced astrocyte swelling. Metab. Brain Dis. 2005, 20, 303–318. [Google Scholar] [CrossRef] [PubMed]
- Bongaerts, G.; Severijnen, R.; Timmerman, H. Effect of antibiotics, prebiotics and probiotics in treatment for hepatic encephalopathy. Med. Hypotheses 2005, 64, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lkhagva, E.; Chung, H.J.; Kim, H.J.; Hong, S.T. The Pharmabiotic Approach to Treat Hyperammonemia. Nutrients 2018, 10, 140. [Google Scholar] [CrossRef] [PubMed]
- Milajerdi, A.; Mousavi, S.M.; Sadeghi, A.; Salari-Moghaddam, A.; Parohan, M.; Larijani, B.; Esmaillzadeh, A. The effect of probiotics on inflammatory biomarkers: A meta-analysis of randomized clinical trials. Eur. J. Nutr. 2020, 59, 633–649. [Google Scholar] [CrossRef]
- Leonel, A.J.; Alvarez-Leite, J.I. Butyrate: implications for intestinal function. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 474–479. [Google Scholar] [CrossRef]
- Den Besten, G.; Gerding, A.; van Dijk, T.H.; Ciapaite, J.; Bleeker, A.; van Eunen, K.; Havinga, R.; Groen, A.K.; Reijngoud, D.J.; Bakker, B.M. Protection against the Metabolic Syndrome by Guar Gum-Derived Short-Chain Fatty Acids Depends on Peroxisome Proliferator-Activated Receptor γ and Glucagon-Like Peptide-1. PLoS ONE 2015, 10, e0136364. [Google Scholar] [CrossRef]
- Walsh, M.E.; Bhattacharya, A.; Sataranatarajan, K.; Qaisar, R.; Sloane, L.; Rahman, M.M.; Kinter, M.; Van Remmen, H. The histone deacetylase inhibitor butyrate improves metabolism and reduces muscle atrophy during aging. Aging Cell 2015, 14, 957–970. [Google Scholar] [CrossRef]
- Soares, A.D.N.; Wanner, S.P.; Morais, E.S.S.; Hudson, A.S.R.; Martins, F.S.; Cardoso, V.N. Supplementation with Saccharomyces boulardii Increases the Maximal Oxygen Consumption and Maximal Aerobic Speed Attained by Rats Subjected to an Incremental-Speed Exercise. Nutrients 2019, 11, 2352. [Google Scholar] [CrossRef]
- Huang, W.C.; Lee, M.C.; Lee, C.C.; Ng, K.S.; Hsu, Y.J.; Tsai, T.Y.; Young, S.L.; Lin, J.S.; Huang, C.C. Effect of Lactobacillus plantarum TWK10 on Exercise Physiological Adaptation, Performance, and Body Composition in Healthy Humans. Nutrients 2019, 11, 2836. [Google Scholar] [CrossRef]
- Al-Obaidi, S.; Al-Sayegh, N.; Nadar, M. Smoking impact on grip strength and fatigue resistance: Implications for exercise and hand therapy practice. J. Phys. Act. Health 2014, 11, 1025–1031. [Google Scholar] [PubMed]
- Goh, Y.J.; Klaenhammer, T.R. Insights into glycogen metabolism in Lactobacillus acidophilus: Impact on carbohydrate metabolism, stress tolerance and gut retention. Microb. Cell Fact. 2014, 13, 94. [Google Scholar] [CrossRef] [PubMed]
- Hearris, M.A.; Hammond, K.M.; Fell, J.M.; Morton, J.P. Regulation of Muscle Glycogen Metabolism during Exercise: Implications for Endurance Performance and Training Adaptations. Nutrients 2018, 10, 298. [Google Scholar] [CrossRef] [PubMed]
- Samuel, B.S.; Shaito, A.; Motoike, T.; Rey, F.E.; Backhed, F.; Manchester, J.K.; Hammer, R.E.; Williams, S.C.; Crowley, J.; Yanagisawa, M.; et al. Effects of the gut microbiota on host adiposity are modulated by the short-chain fatty-acid binding G protein-coupled receptor, Gpr41. Proc. Natl. Acad. Sci. USA 2008, 105, 16767–16772. [Google Scholar] [CrossRef] [PubMed]
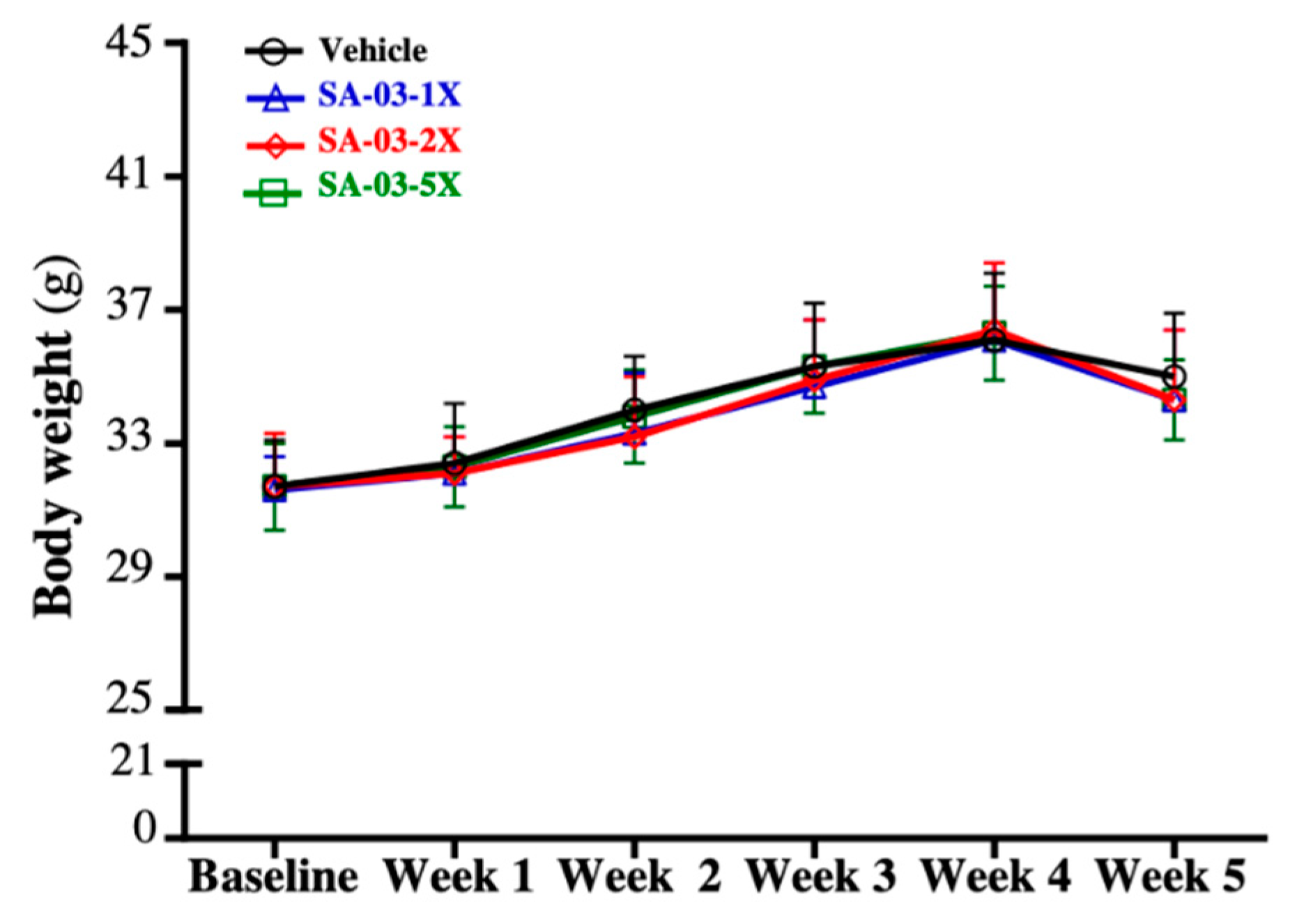
| Characteristic | Vehicle (PBS) | SA-03-1X | SA-03-2X | SA-03-5X | Trend Analysis |
|---|---|---|---|---|---|
| Initial BW (g) | 31.7 ± 1.4 | 31.6 ± 1.0 | 31.7 ± 1.6 | 31.7 ± 1.3 | 0.8686 |
| Final BW (g) | 36.8 ± 2.2 | 36.4 ± 2.1 | 36.1 ± 2.3 | 35.9 ± 1.7 | 0.6070 |
| Water intake (mL/mouse/day) | 7.6 ± 0.5 | 7.3 ± 0.9 | 7.2 ± 0.9 | 7.4 ± 1.0 | 0.1138 |
| Food intake (g/mouse/day) | 8.8 ± 1.7 | 9.2 ± 1.2 | 9.0 ± 1.4 | 8.9 ± 1.9 | 0.6364 |
| Liver (g) | 1.86 ± 0.19 | 1.83 ± 0.22 | 1.89 ± 0.25 | 1.82 ± 0.11 | 0.7169 |
| Muscle (g) | 0.37 ± 0.03 | 0.37 ± 0.03 | 0.36 ± 0.04 | 0.37 ± 0.03 | 0.4901 |
| Quadriceps (g) | 0.48 ± 0.05 | 0.49 ± 0.05 | 0.47 ± 0.04 | 0.49 ± 0.04 | 0.8718 |
| Kidney (g) | 0.65 ± 0.09 | 0.66 ± 0.06 | 0.62 ± 0.03 | 0.62 ± 0.06 | 0.2541 |
| Heart (g) | 0.18 ± 0.03 | 0.18 ± 0.02 | 0.18 ± 0.02 | 0.18 ± 0.04 | 0.2773 |
| Lung (g) | 0.23 ± 0.03 | 0.23 ± 0.03 | 0.23 ± 0.03 | 0.23 ± 0.04 | 0.1584 |
| EFP (g) | 0.32 ± 0.10 | 0.31 ± 0.07 | 0.31 ± 0.07 | 0.31 ± 0.06 | 0.7057 |
| BAT (g) | 0.09 ± 0.02 | 0.08 ± 0.03 | 0.09 ± 0.02 | 0.09 ± 0.02 | 0.4066 |
| Cecum (g) | 0.85 ± 0.07 | 0.88 ± 0.14 | 0.83 ± 0.13 | 0.88 ± 0.10 | 0.8411 |
| * Relative liver weight (%) | 5.03 ± 0.25 | 5.04 ± 0.74 | 5.23 ± 0.61 | 5.07 ± 0.17 | 0.5428 |
| Relative muscle weight (%) | 1.01 ± 0.09 | 1.01 ± 0.04 | 1.01 ± 0.10 | 1.02 ± 0.07 | 0.9060 |
| Relative quadriceps weight (%) | 1.31 ± 0.14 | 1.34 ± 0.11 | 1.30 ± 0.10 | 1.35 ± 0.07 | 0.5903 |
| Relative kidney weight (%) | 1.76 ± 0.20 | 1.82 ± 0.11 | 1.73 ± 0.12 | 1.72 ± 0.18 | 0.4407 |
| Relative heart weight (%) | 0.50 ± 0.08 | 0.50 ± 0.04 | 0.51 ± 0.09 | 0.51 ± 0.12 | 0.4467 |
| Relative lung weight (%) | 0.63 ± 0.08 | 0.63 ± 0.08 | 0.63 ± 0.07 | 0.64 ± 0.10 | 0.1420 |
| Relative EFP weight (%) | 0.87 ± 0.24 | 0.84 ± 0.18 | 0.87 ± 0.23 | 0.86 ± 0.17 | 0.6941 |
| Relative BAT weight (%) | 0.25 ± 0.06 | 0.22 ± 0.08 | 0.25 ± 0.05 | 0.26 ± 0.06 | 0.3942 |
| Relative cecum weight (%) | 2.31 ± 0.19 | 2.43 ± 0.34 | 2.29 ± 0.38 | 2.46 ± 0.33 | 0.3665 |
| Time Point | Vehicle | SA-03-1X | SA-03-2X | SA-03-5X | Trend Analysis |
|---|---|---|---|---|---|
| Lactate (mmol/L) | |||||
| Before swimming (A) | 3.41 ± 0.69 | 3.34 ± 0.56 | 3.30 ± 0.85 | 3.50 ± 0.37 | 0.6087 |
| After swimming (B) | 8.73 ± 0.65 d | 7.53 ± 0.90 c | 6.65 ± 0.85 b | 5.78 ± 0.64 a | <0.0001 |
| After 20 min resting (C) | 6.84 ± 0.48 c | 5.85 ± 1.08 b | 5.40 ± 0.71 b | 4.53 ± 0.89 a | <0.0001 |
| Rate of lactate production and clearance | |||||
| Production rate = B/A | 2.64 ± 0.47 c | 2.30 ± 0.41 b,c | 2.13 ± 0.57 b | 1.68±0.30 a | <0.0001 |
| Clearance rate = (B-C)/B | 0.21 ± 0.07 | 0.22 ± 0.10 | 0.19 ± 0.08 | 0.22 ± 0.11 | 0.9901 |
| Parameter | Vehicle | SA-03-1X | SA-03-2X | SA-03-5X | Trend Analysis |
|---|---|---|---|---|---|
| AST (U/L) | 67 ± 9 | 69 ± 6 | 67 ± 9 | 67 ± 9 | 0.7926 |
| ALT (U/L) | 36 ± 8 | 34 ± 9 | 34 ± 8 | 30 ± 6 | 0.0367 |
| CK (U/L) | 162 ± 30 | 165 ± 24 | 159 ± 16 | 162 ± 22 | 0.3890 |
| GLU (mg/dL) | 186 ± 14 | 186 ± 11 | 187 ± 14 | 187 ± 15 | 0.8841 |
| CREA (mg/dL) | 0.41 ± 0.02 | 0.42 ± 0.03 | 0.42 ± 0.02 | 0.42 ± 0.03 | 0.3146 |
| BUN (mg/dL) | 21.4 ± 3.0 | 21.4 ± 2.0 | 21.1 ± 1.9 | 21.3 ± 1.8 | 0.9156 |
| UA (mg/dL) | 1.7 ± 0.2 | 1.7 ± 0.2 | 1.7 ± 0.2 | 1.7 ± 0.3 | 0.9649 |
| TC (mg/dL) | 147 ± 22 | 147 ± 20 | 147 ± 16 | 147 ± 16 | 0.9617 |
| TG (mg/dL) | 156 ± 22 | 156 ± 19 | 156 ± 21 | 154 ± 21 | 0.7516 |
| ALB (g/dL) | 3.0 ± 0.1 | 3.0 ± 0.3 | 3.0 ± 0.3 | 3.0 ± 0.2 | 0.9723 |
| TP (g/dL) | 5.6 ± 0.3 | 5.7 ± 0.4 | 5.7 ± 0.3 | 5.6 ± 0.3 | 0.9616 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, M.-C.; Hsu, Y.-J.; Ho, H.-H.; Hsieh, S.-H.; Kuo, Y.-W.; Sung, H.-C.; Huang, C.-C. Lactobacillus salivarius Subspecies salicinius SA-03 is a New Probiotic Capable of Enhancing Exercise Performance and Decreasing Fatigue. Microorganisms 2020, 8, 545. https://doi.org/10.3390/microorganisms8040545
Lee M-C, Hsu Y-J, Ho H-H, Hsieh S-H, Kuo Y-W, Sung H-C, Huang C-C. Lactobacillus salivarius Subspecies salicinius SA-03 is a New Probiotic Capable of Enhancing Exercise Performance and Decreasing Fatigue. Microorganisms. 2020; 8(4):545. https://doi.org/10.3390/microorganisms8040545
Chicago/Turabian StyleLee, Mon-Chien, Yi-Ju Hsu, Hsieh-Hsun Ho, Shih-Hung Hsieh, Yi-Wei Kuo, Hsin-Ching Sung, and Chi-Chang Huang. 2020. "Lactobacillus salivarius Subspecies salicinius SA-03 is a New Probiotic Capable of Enhancing Exercise Performance and Decreasing Fatigue" Microorganisms 8, no. 4: 545. https://doi.org/10.3390/microorganisms8040545
APA StyleLee, M.-C., Hsu, Y.-J., Ho, H.-H., Hsieh, S.-H., Kuo, Y.-W., Sung, H.-C., & Huang, C.-C. (2020). Lactobacillus salivarius Subspecies salicinius SA-03 is a New Probiotic Capable of Enhancing Exercise Performance and Decreasing Fatigue. Microorganisms, 8(4), 545. https://doi.org/10.3390/microorganisms8040545







