Mutation and Recombination Rates Vary Across Bacterial Chromosome
Abstract
1. Introduction
2. Mutational Processes Vary Across the Bacterial Genome
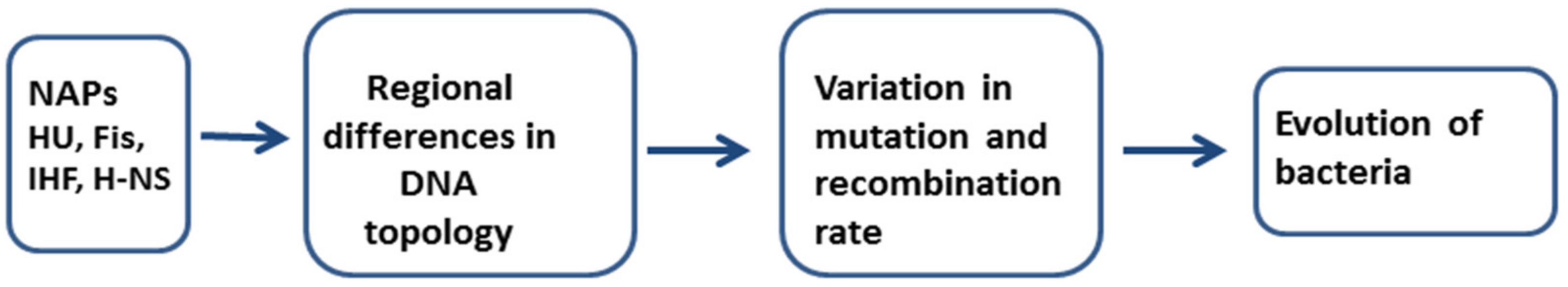
3. A Role of NAPs in Mutagenesis and Recombination Processes
4. Concluding Remarks
Funding
Conflicts of Interest
References
- Pomerantz, R.T.; O’Donnell, M. What happens when replication and transcription complexes collide? Cell Cycle. 2010, 9, 2537–2543. [Google Scholar] [CrossRef] [PubMed]
- Srivatsan, A.; Tehranchi, A.; MacAlpine, D.M.; Wang, J.D. Co-orientation of replication and transcription preserves genome integrity. PLoS Genet. 2010, 6, e1000810. [Google Scholar] [CrossRef] [PubMed]
- Frank, A.C.; Lobry, J.R. Asymmetric substitution patterns: A review of possible underlying mutational or selective mechanisms. Gene 1999, 238, 65–77. [Google Scholar] [CrossRef]
- Fijalkowska, I.J.; Jonczyk, P.; Tkaczyk, M.M.; Bialoskorska, M.; Schaaper, R.M. Unequal fidelity of leading strand and lagging strand DNA replication on the Escherichia coli chromosome. Proc. Natl. Acad. Sci. USA 1998, 95, 10020–10025. [Google Scholar] [CrossRef]
- Gawel, D.; Jonczyk, P.; Bialoskorska, M.; Schaaper, R.M.; Fijalkowska, I.J. Asymmetry of frameshift mutagenesis during leading and lagging-strand replication in Escherichia coli. Mutat. Res. 2002, 501, 129–136. [Google Scholar] [CrossRef]
- Wright, B.E. Stress-directed adaptive mutations and evolution. Mol. Microbiol. 2004, 52, 643–650. [Google Scholar] [CrossRef]
- Reimers, J.M.; Schmidt, K.H.; Longacre, A.; Reschke, D.K.; Wright, B.E. Increased transcription rates correlate with increased reversion rates in leuB and argH Escherichia coli auxotrophs. Microbiology 2004, 150, 1457–1466. [Google Scholar] [CrossRef]
- Beletskii, A.; Bhagwat, A.S. Transcription-induced mutations: Increase in C to T mutations in the nontranscribed strand during transcription in Escherichia coli. Proc. Natl. Acad. Sci. USA 1996, 93, 13919–13924. [Google Scholar] [CrossRef]
- Paul, S.; Million-Weaver, S.; Chattopadhyay, S.; Sokurenko, E.; Merrikh, H. Accelerated gene evolution through replication-transcription conflicts. Nature 2013, 495, 512–515. [Google Scholar] [CrossRef]
- Juurik, T.; Ilves, H.; Teras, R.; Ilmjarv, T.; Tavita, K.; Ukkivi, K.; Kivisaar, M. Mutation frequency and spectrum of mutations vary at different chromosomal positions of Pseudomonas putida. PLoS ONE 2012, 7, e48511. [Google Scholar] [CrossRef]
- Valens, M.; Penaud, S.; Rossignol, M.; Cornet, F.; Boccard, F. Macrodomain organization of the Escherichia coli chromosome. EMBO J. 2004, 23, 4330–4341. [Google Scholar] [CrossRef] [PubMed]
- Lioy, V.S.; Cournac, A.; Marbouty, M.; Duigou, S.; Mozziconacci, J.; Espeli, O.; Koszul, R. Multiscale structuring of the E. coli chromosome by nucleoid-associated and condensin proteins. Cell 2018, 172, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Champoux, J.J. DNA topoisomerases: Structure, function, and mechanism. Annu. Rev. Biochem. 2001, 70, 369–413. [Google Scholar] [CrossRef] [PubMed]
- Dorman, C.J.; Deighan, P. Regulation of gene expression by histone-like proteins in bacteria. Curr. Opin. Genet. Dev. 2003, 13, 179–184. [Google Scholar] [CrossRef]
- Ali Azam, T.; Iwata, A.; Nishimura, A.; Ueda, S.; Ishihama, A. Growth phase-dependent variation in protein composition of the Escherichia coli nucleoid. J. Bacteriol. 1999, 181, 6361–6370. [Google Scholar]
- Balke, V.L.; Gralla, J.D. Changes in the linking number of supercoiled DNA accompany growth transitions in Escherichia coli. J. Bacteriol. 1987, 169, 4499–4506. [Google Scholar] [CrossRef]
- Travers, A.; Muskhelishvili, G. DNA supercoiling—A global transcriptional regulator for enterobacterial growth? Nat. Rev. Microbiol. 2005, 3, 157–169. [Google Scholar] [CrossRef]
- Dame, R.T. The role of nucleoid-associated proteins in the organization and compaction of bacterial chromatin. Mol. Microbiol. 2005, 56, 858–870. [Google Scholar] [CrossRef]
- Amzallag, G.N. Adaptive changes in bacteria: A consequence of nonlinear transitions in chromosome topology? J. Theor. Biol. 2004, 229, 361–369. [Google Scholar] [CrossRef]
- Karpinets, T.; Greenwood, D.; Pogribny, I.; Samatova, N. Bacterial stationary-state mutagenesis and Mammalian tumorigenesis as stress-induced cellular adaptations and the role of epigenetics. Curr. Genom. 2006, 7, 481–496. [Google Scholar] [CrossRef][Green Version]
- Flores-Rios, R.; Quatrini, R.; Loyola, A. Endogenous and foreign nucleoid-associated proteins of bacteria: Occurrence, interactions and effects on mobile genetic elements and host’s biology. Comput. Struct. Biotechnol. J. 2019, 17, 746–756. [Google Scholar] [CrossRef] [PubMed]
- Hanawalt, P.C. Subpathways of nucleotide excision repair and their regulation. Oncogene 2002, 21, 8949–8956. [Google Scholar] [CrossRef] [PubMed]
- Rimsky, S.; Travers, A. Pervasive regulation of nucleoid structure and function by nucleoid-associated proteins. Curr. Opin. Microbiol. 2011, 14, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Dame, R.T.; Kalmykowa, O.J.; Grainger, D.C. Chromosomal macrodomains and associated proteins: Implications for DNA organization and replication in gram negative bacteria. PLoS Genet. 2011, 7, e1002123. [Google Scholar] [CrossRef]
- Dame, R.T.; Tark-Dame, M. Bacterial chromatin: Converging views at different scales. Curr. Opin. Cell Biol. 2016, 40, 60–65. [Google Scholar] [CrossRef]
- Ishihama, A. The nucleoid: An overview. EcoSal Plus 2009, 2, 1–45. [Google Scholar] [CrossRef]
- Browning, D.F.; Grainger, D.C.; Busby, S.J. Effects of nucleoid-associated proteins on bacterial chromosome structure and gene expression. Curr. Opin. Microbiol. 2010, 13, 773–780. [Google Scholar] [CrossRef]
- Lynch, M.; Ackerman, M.S.; Gout, J.F.; Long, H.; Sung, W.; Thomas, W.K.; Foster, P.L. Genetic drift, selection and the evolution of the mutation rate. Nat. Rev. Genet. 2016, 17, 704–714. [Google Scholar] [CrossRef]
- Maki, H. Origins of spontaneous mutations: Specificity and directionality of base-substitution, frameshift, and sequence-substitution mutageneses. Annu. Rev. Genet. 2002, 36, 279–303. [Google Scholar] [CrossRef]
- Benzer, S. On the topography of the genetic fine structure. Proc. Natl. Acad. Sci. USA 1961, 47, 403–415. [Google Scholar] [CrossRef]
- Sharp, P.M.; Shields, D.C.; Wolfe, K.H.; Li, W.H. Chromosomal location and evolutionary rate variation in enterobacterial genes. Science 1989, 246, 808–810. [Google Scholar] [CrossRef] [PubMed]
- Mira, A.; Ochman, H. Gene location and bacterial sequence divergence. Mol. Biol. Evol. 2002, 19, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Hudson, R.E.; Bergthorsson, U.; Roth, J.R.; Ochman, H. Effect of chromosome location on bacterial mutation rates. Mol. Biol. Evol. 2002, 19, 85–92. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Martina, M.A.; Correa, E.M.; Argarana, C.E.; Barra, J.L. Escherichia coli frameshift mutation rate depends on the chromosomal context but not on the GATC content near the mutation site. PLoS ONE 2012, 7, e33701. [Google Scholar] [CrossRef]
- Halligan, D.L.; Keightley, P.D. Spontaneous mutation accumulation studies in evolutionary genetics. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 151–172. [Google Scholar] [CrossRef]
- Schroeder, J.W.; Yeesin, P.; Simmons, L.A.; Wang, J.D. Sources of spontaneous mutagenesis in bacteria. Crit. Rev. Biochem. Mol. Biol. 2018, 53, 29–48. [Google Scholar] [CrossRef]
- Foster, P.L.; Hanson, A.J.; Lee, H.; Popodi, E.M.; Tang, H.X. On the mutational topology of the bacterial genome. G3 Genes Genomes Genet. 2013, 3, 399–407. [Google Scholar] [CrossRef]
- Lee, H.; Popodi, E.; Tang, H.; Foster, P.L. Rate and molecular spectrum of spontaneous mutations in the bacterium Escherichia coli as determined by whole-genome sequencing. Proc. Natl. Acad. Sci. USA 2012, 109, E2774–E2783. [Google Scholar] [CrossRef]
- Katju, V.; Bergthorsson, U. Old trade, new tricks: Insights into the spontaneous mutation process from the partnering of classical mutation accumulation experiments with high-throughput genomic approaches. Genome Biol. Evol. 2019, 11, 136–165. [Google Scholar] [CrossRef]
- Lind, P.A.; Andersson, D.I. Whole-genome mutational biases in bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 17878–17883. [Google Scholar] [CrossRef]
- Drake, J.W. A constant rate of spontaneous mutation in DNA-based microbes. Proc. Natl. Acad. Sci. USA 1991, 88, 7160–7164. [Google Scholar] [CrossRef] [PubMed]
- Foster, P.L.; Lee, H.; Popodi, E.; Townes, J.P.; Tang, H. Determinants of spontaneous mutation in the bacterium Escherichia coli as revealed by whole-genome sequencing. Proc. Natl. Acad. Sci. USA 2015, 112, E5990–E5999. [Google Scholar] [CrossRef] [PubMed]
- Niccum, B.A.; Lee, H.; MohammedIsmail, W.; Tang, H.; Foster, P.L. The symmetrical wave pattern of base-pair substitution rates across the Escherichia coli chromosome has multiple causes. mBio 2019, 10, e01226-19. [Google Scholar] [CrossRef] [PubMed]
- Long, H.A.; Sung, W.; Miller, S.F.; Ackerman, M.S.; Doak, T.G.; Lynch, M. Mutation rate, spectrum, topology, and context-dependency in the DNA mismatch repair-deficient Pseudomonas fluorescens ATCC948. Genome Biol. Evol. 2015, 7, 262–271. [Google Scholar] [CrossRef]
- Dettman, J.R.; Sztepanacz, J.L.; Kassen, R. The properties of spontaneous mutations in the opportunistic pathogen Pseudomonas aeruginosa. BMC Genom. 2016, 17, 27. [Google Scholar] [CrossRef]
- Dillon, M.M.; Sung, W.; Sebra, R.; Lynch, M.; Cooper, V.S. Genome-wide biases in the rate and molecular spectrum of spontaneous mutations in Vibrio cholerae and Vibrio fischeri. Mol. Biol. Evol. 2017, 34, 93–109. [Google Scholar] [CrossRef]
- Wei, W.; Xiong, L.; Ye, Y.N.; Du, M.Z.; Gao, Y.Z.; Zhang, K.Y.; Kan, B. Mutation landscape of base substitutions, duplications, and deletions in the representative current cholera pandemic strain. Genome Biol. Evol. 2018, 10, 2072–2085. [Google Scholar] [CrossRef]
- Dillon, M.M.; Sung, W.; Lynch, M.; Cooper, V.S. Periodic variation of mutation rates in bacterial genomes associated with replication timing. mBio 2018, 9, e01371-18. [Google Scholar] [CrossRef]
- Val, M.E.; Soler-Bistue, A.; Bland, M.J.; Mazel, D. Management of multipartite genomes: The Vibrio cholerae model. Curr. Opin. Microbiol. 2014, 22, 120–126. [Google Scholar] [CrossRef]
- Rasmussen, T.; Jensen, R.B.; Skovgaard, O. The two chromosomes of Vibrio cholerae are initiated at different time points in the cell cycle. EMBO J. 2007, 26, 3124–3131. [Google Scholar] [CrossRef]
- Baek, J.H.; Chattoraj, D.K. Chromosome I controls chromosome II replication in Vibrio cholerae. PLoS Genet. 2014, 10, e1004184. [Google Scholar] [CrossRef] [PubMed]
- Gon, S.; Camara, J.E.; Klungsoyr, H.K.; Crooke, E.; Skarstad, K.; Beckwith, J. A novel regulatory mechanism couples deoxyribonucleotide synthesis and DNA replication in Escherichia coli. EMBO J. 2006, 25, 1137–1147. [Google Scholar] [CrossRef] [PubMed]
- Foster, P.L.; Niccum, B.A.; Popodi, E.; Townes, J.P.; Lee, H.; MohammedIsmail, W.; Tang, H. Determinants of base-pair substitution patterns revealed by whole-genome sequencing of DNA mismatch repair defective Escherichia coli. Genetics 2018, 209, 1029–1042. [Google Scholar] [CrossRef] [PubMed]
- Warnecke, T.; Supek, F.; Lehner, B. Nucleoid-associated proteins affect mutation dynamics in E. coli in a growth phase-specific manner. PLoS Comput. Biol. 2012, 8, e1002846. [Google Scholar] [CrossRef] [PubMed]
- Mercier, R.; Petit, M.A.; Schbath, S.; Robin, S.; El Karoui, M.; Boccard, F.; Espéli, O. The MatP/matS site-specific system organizes the terminus region of the E. coli chromosome into a macrodomain. Cell 2008, 135, 475–485. [Google Scholar] [CrossRef] [PubMed]
- Nolivos, S.; Upton, A.L.; Badrinarayanan, A.; Muller, J.; Zawadzka, K.; Wiktor, J.; Sherratt, D. MatP regulates the coordinated action of topoisomerase IV and MukBEF in chromosome segregation. Nat. Commun. 2016, 7, 10466. [Google Scholar] [CrossRef]
- Van Noort, J.; Verbrugge, S.; Goosen, N.; Dekker, C.; Dame, R.T. Dual architectural roles of HU: Formation of flexible hinges and rigid filaments. Proc. Natl. Acad. Sci. USA 2004, 101, 6969–6974. [Google Scholar] [CrossRef]
- Stojkova, P.; Spidlova, P.; Stulik, J. Nucleoid-associated protein HU: A lilliputian in gene regulation of bacterial virulence. Front. Cell. Infect. Microbiol. 2019, 9, 159. [Google Scholar] [CrossRef]
- Claret, L.; Rouviere-Yaniv, J. Variation in HU composition during growth of Escherichia coli: The heterodimer is required for long term survival. J. Mol. Biol. 1997, 273, 93–104. [Google Scholar] [CrossRef]
- Boubrik, F.; Rouviere-Yaniv, J. Increased sensitivity to gamma irradiation in bacteria lacking protein, HU. Proc. Natl. Acad. Sci. USA 1995, 92, 3958–3962. [Google Scholar] [CrossRef]
- Bonnefoy, E.; Takahashi, M.; Yaniv, J.R. DNA-binding parameters of the HU protein of Escherichia coli to cruciform DNA. J. Mol. Biol. 1994, 242, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Castaing, B.; Zelwer, C.; Laval, J.; Boiteux, S. HU protein of Escherichia coli binds specifically to DNA that contains single-strand breaks or gaps. J. Biol. Chem. 1995, 270, 10291–10296. [Google Scholar] [CrossRef]
- Pontiggia, A.; Negri, A.; Beltrame, M.; Bianchi, M.E. Protein HU binds specifically to kinked DNA. Mol. Microbiol. 1993, 7, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Waters, R. Escherichia coli strains lacking protein HU are UV sensitive due to a role for HU in homologous recombination. J. Bacteriol. 1998, 180, 3750–3756. [Google Scholar] [PubMed]
- Kamashev, D.; Rouviere-Yaniv, J. The histone-like protein HU binds specifically to DNA recombination and repair intermediates. EMBO J. 2000, 19, 6527–6535. [Google Scholar] [CrossRef]
- Foster, P.L. Mechanisms of stationary phase mutation: A decade of adaptive mutation. Annu. Rev. Genet. 1999, 33, 57–88. [Google Scholar] [CrossRef]
- Rosenberg, S.M. Evolving responsively: Adaptive mutation. Nat. Rev. Genet. 2001, 2, 504–515. [Google Scholar] [CrossRef]
- Harris, R.S.; Longerich, S.; Rosenberg, S.M. Recombination in adaptive mutation. Science 1994, 264, 258–260. [Google Scholar] [CrossRef]
- Foster, P.L.; Trimarchi, J.M. adaptive reversion of an episomal frameshift mutation in Escherichia coli requires conjugal functions but not actual conjugation. Proc. Natl. Acad. Sci. USA 1995, 92, 5487–5490. [Google Scholar] [CrossRef]
- Foster, P.L.; Trimarchi, J.M.; Maurer, R.A. Two enzymes, both of which process recombination intermediates, have opposite effects on adaptive mutation in Escherichia coli. Genetics 1996, 142, 25–37. [Google Scholar]
- Bull, H.J.; Lombardo, M.J.; Rosenberg, S.M. Stationary-phase mutation in the bacterial chromosome: Recombination protein and DNA polymerase IV dependence. Proc. Natl. Acad. Sci. USA 2001, 98, 8334–8341. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, G.J.; Lee, P.L.; Lombardo, M.J.; Hastings, P.J.; Rosenberg, S.M. SOS mutator DNA polymerase IV functions in adaptive mutation and not adaptive amplification. Mol. Cell 2001, 7, 571–579. [Google Scholar] [CrossRef]
- Foster, P.L. Adaptive mutation in Escherichia coli. Cold Spring Harb. Symp. Quant. Biol. 2000, 65, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.B.; Foster, P.L. The Escherichia coli histone-like protein HU has a role in stationary phase adaptive mutation. Genetics 2007, 177, 723–735. [Google Scholar] [CrossRef]
- Moore, J.M.; Magnan, D.; Mojica, A.K.; Nunez, M.A.; Bates, D.; Rosenberg, S.M.; Hastings, P.J. Roles of nucleoid-associated proteins in stress-induced mutagenic break repair in starving Escherichia coli. Genetics 2015, 201, 1349–1362. [Google Scholar] [CrossRef]
- Al Mamun, A.A.; Lombardo, M.J.; Shee, C.; Lisewski, A.M.; Gonzalez, C.; Lin, D.; Lichtarge, O. Identity and function of a large gene network underlying mutagenic repair of DNA breaks. Science 2012, 338, 1344–1348. [Google Scholar] [CrossRef]
- Moore, J.M.; Wimberly, H.; Thornton, P.C.; Rosenberg, S.M.; Hastings, P.J. Gross chromosomal rearrangement mediated by DNA replication in stressed cells: Evidence from Escherichia coli. Ann. N. Y. Acad. Sci. 2012, 1267, 103–109. [Google Scholar] [CrossRef][Green Version]
- Karas, V.O.; Westerlaken, I.; Meyer, A.S. The DNA-binding protein from starved cells (Dps) utilizes dual functions to defend cells against multiple stresses. J. Bacteriol. 2015, 197, 3206–3215. [Google Scholar] [CrossRef]
- Janissen, R.; Arens, M.M.A.; Vtyurina, N.N.; Rivai, Z.; Sunday, N.D.; Eslami-Mossallam, B.; Dekker, N.H. Global DNA compaction in stationary-phase bacteria does not affect transcription. Cell 2018, 174, 1188–1199. [Google Scholar] [CrossRef]
- Micka, B.; Marahiel, M.A. The DNA-binding protein HBsu is essential for normal growth and development in Bacillus subtilis. Biochimie 1992, 74, 641–650. [Google Scholar] [CrossRef]
- Lopez-Torrejon, G.; Martinez-Jimenez, M.I.; Ayora, S. Role of LrpC from Bacillus subtilis in DNA transactions during DNA repair and recombination. Nucleic Acids Res. 2006, 34, 120–129. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fernandez, S.; Rojo, F.; Alonso, J.C. The Bacillus subtilis chromatin-associated protein Hbsu is involved in DNA repair and recombination. Mol. Microbiol. 1997, 23, 1169–1179. [Google Scholar] [CrossRef] [PubMed]
- Tavita, K.; Mikkel, K.; Tark-Dame, M.; Jerabek, H.; Teras, R.; Sidorenko, J.; Kivisaar, M. Homologous recombination is facilitated in starving populations of Pseudomonas putida by phenol stress and affected by chromosomal location of the recombination target. Mutat. Res. 2012, 737, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Sidorenko, J.; Ukkivi, K.; Kivisaar, M. NER enzymes maintain genome integrity and suppress homologous recombination in the absence of exogenously induced DNA damage in Pseudomonas putida. DNA Repair 2015, 25, 15–26. [Google Scholar] [CrossRef]
- Nash, H.A.; Robertson, C.A. Purification and properties of the Escherichia coli protein factor required for lambda integrative recombination. J. Biol. Chem. 1981, 256, 9246–9253. [Google Scholar]
- Miller, H.I.; Friedman, D.I. An E. coli gene product required for lambda site-specific recombination. Cell 1980, 20, 711–719. [Google Scholar] [CrossRef]
- Dillon, S.C.; Dorman, C.J. Bacterial nucleoid-associated proteins, nucleoid structure and gene expression. Nat. Rev. Microbiol. 2010, 8, 185–195. [Google Scholar] [CrossRef]
- Nunez, J.K.; Bai, L.; Harrington, L.B.; Hinder, T.L.; Doudna, J.A. CRISPR immunological memory requires a host factor for specificity. Mol. Cell 2016, 62, 824–833. [Google Scholar] [CrossRef]
- Mikkel, K.; Tagel, M.; Ukkivi, K.; Ilves, H.; Kivisaar, M. Integration host factor IHF facilitates homologous recombination and mutagenic processes in Pseudomonas putida. DNA Repair 2019, 85, 102745. [Google Scholar] [CrossRef]
- Darmon, E.; Leach, D.R. Bacterial genome instability. Microbiol. Mol. Biol. Rev. 2014, 78, 1–39. [Google Scholar] [CrossRef]
- Rocha, E.P. The replication-related organization of bacterial genomes. Microbiology 2004, 150, 1609–1627. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, P.H.; Touchon, M.; Cury, J.; Rocha, E.P.C. The chromosomal organization of horizontal gene transfer in bacteria. Nat. Commun. 2017, 8, 841. [Google Scholar] [CrossRef] [PubMed]
- Touchon, M.; Rocha, E.P. Coevolution of the organization and structure of prokaryotic genomes. Cold Spring Harb. Perspect. Biol. 2016, 8, a018168. [Google Scholar] [CrossRef] [PubMed]
- Kopejtka, K.; Lin, Y.; Jakubovicova, M.; Koblizek, M.; Tomasch, J. Clustered core- and pan-genome content on Rhodobacteraceae chromosomes. Genome Biol. Evol. 2019, 11, 2208–2217. [Google Scholar] [CrossRef]
- Garcia-Russell, N.; Harmon, T.G.; Le, T.Q.; Amaladas, N.H.; Mathewson, R.D.; Segall, A.M. Unequal access of chromosomal regions to each other in Salmonella: Probing chromosome structure with phage lambda integrase-mediated long-range rearrangements. Mol. Microbiol. 2004, 52, 329–344. [Google Scholar] [CrossRef]
- Krogh, T.J.; Moller-Jensen, J.; Kaleta, C. Impact of chromosomal architecture on the function and evolution of bacterial genomes. Front. Microbiol. 2018, 9, 2019. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kivisaar, M. Mutation and Recombination Rates Vary Across Bacterial Chromosome. Microorganisms 2020, 8, 25. https://doi.org/10.3390/microorganisms8010025
Kivisaar M. Mutation and Recombination Rates Vary Across Bacterial Chromosome. Microorganisms. 2020; 8(1):25. https://doi.org/10.3390/microorganisms8010025
Chicago/Turabian StyleKivisaar, Maia. 2020. "Mutation and Recombination Rates Vary Across Bacterial Chromosome" Microorganisms 8, no. 1: 25. https://doi.org/10.3390/microorganisms8010025
APA StyleKivisaar, M. (2020). Mutation and Recombination Rates Vary Across Bacterial Chromosome. Microorganisms, 8(1), 25. https://doi.org/10.3390/microorganisms8010025



