Precipitation Alleviates Adverse Effects of Nitrogen and Phosphorus Enrichment on Soil Microbial Co-Occurrence Network Complexity and Stability in Karst Shrubland
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description and Experimental Design
2.2. Soil Sampling
2.3. Soil Physico-Chemical Properties Analysis
2.4. Gene Extraction, Replication, and Sequencing
2.5. Co-Occurrence Network Analysis
2.6. Statistical Analysis
3. Results
3.1. Soil Physico-Chemical Properties
3.2. Soil Microbial Abundance and Diversity
3.3. Soil Microbial Community Composition
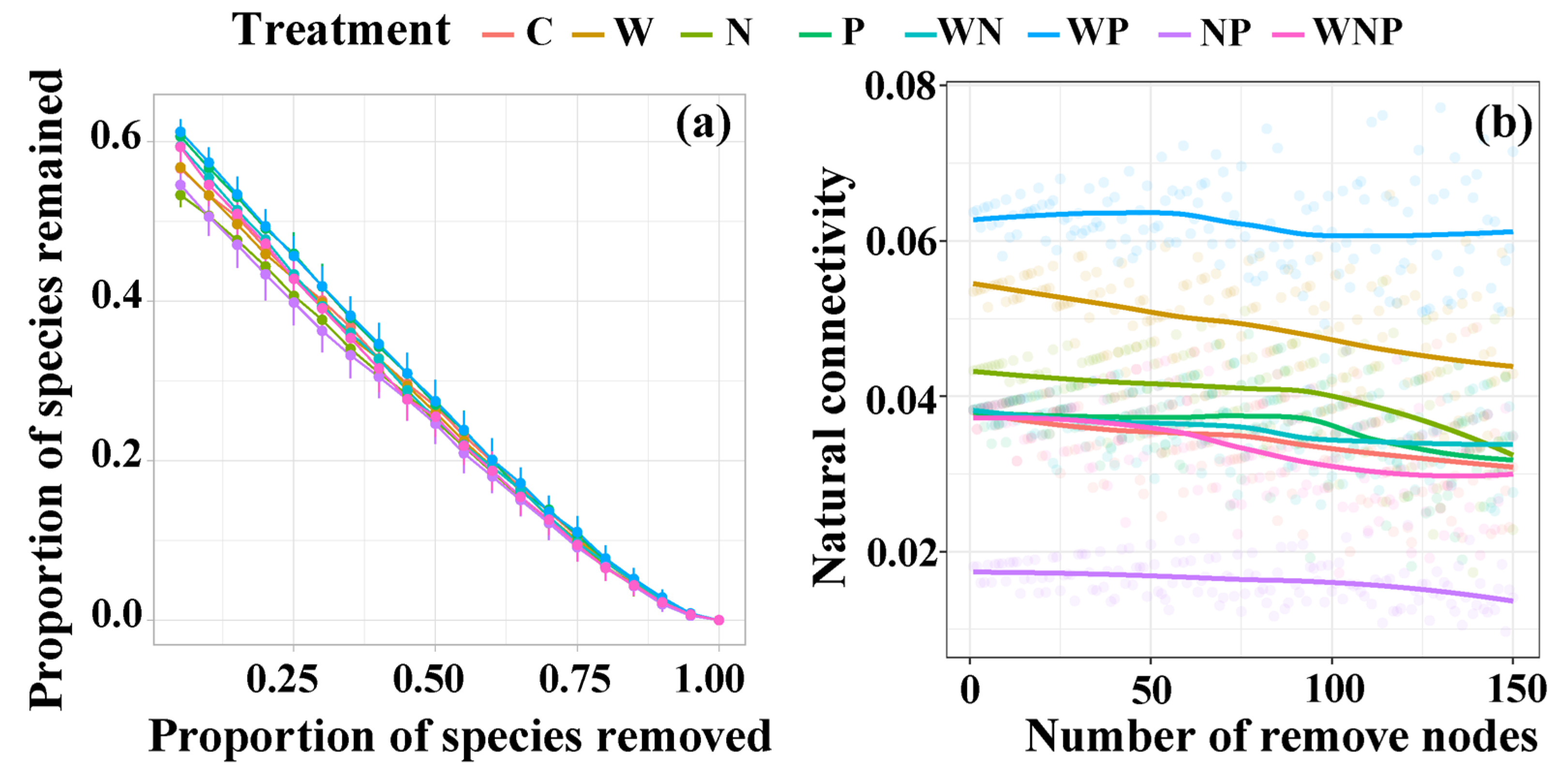
3.4. Soil Microbial Co-Occurrence Network
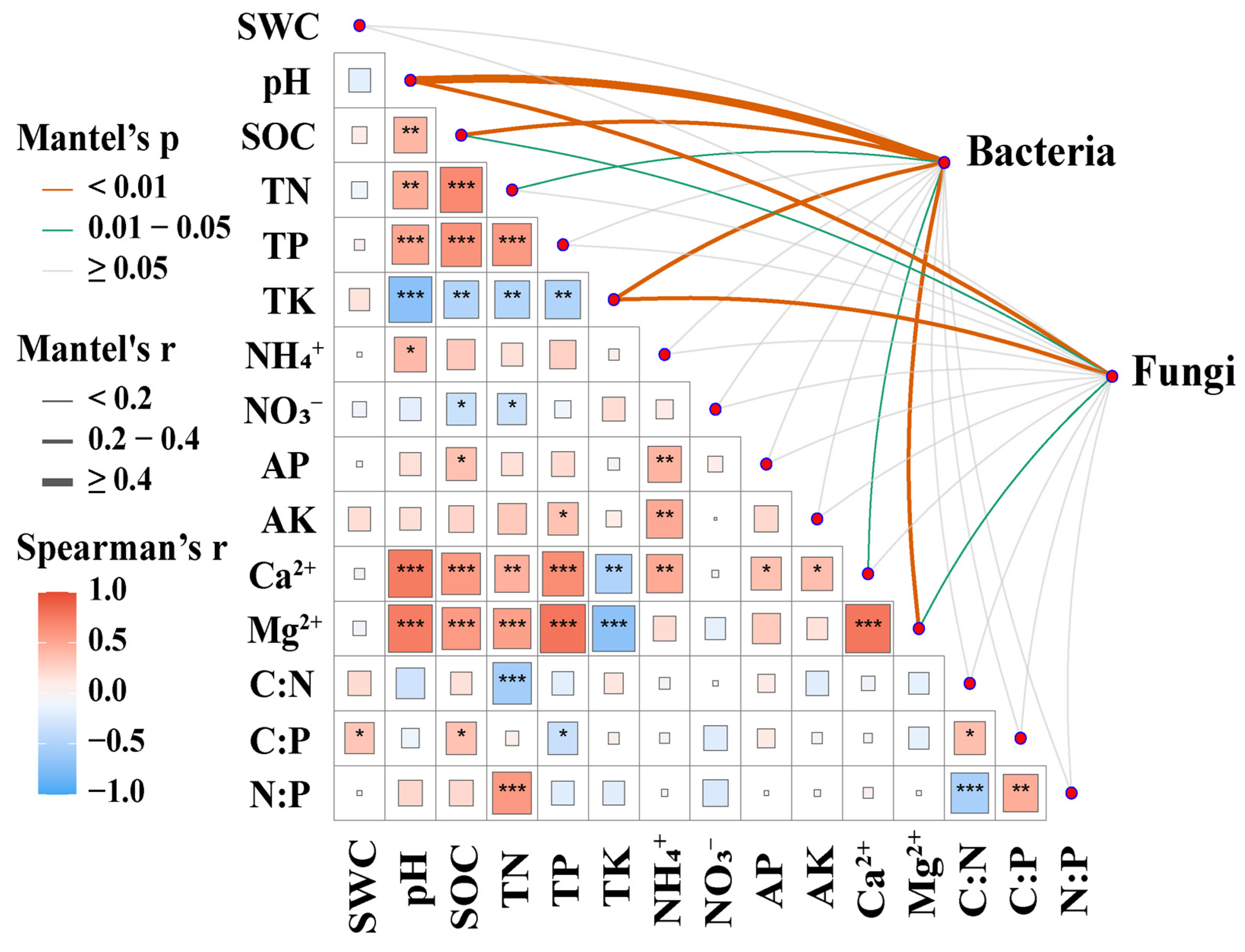
3.5. Factors Affecting Soil Microbial Communities
4. Discussion
4.1. Soil Microbial Abundance Was Sensitive to Water Addition
4.2. Opposing Roles of Water and Nutrients in Microbial Network Complexity and Stability
4.3. Research Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wagg, C.; Bender, S.F.; Widmer, F.; van der Heijden, M.G.A. Soil biodiversity and soil community composition determine ecosystem multifunctionality. Proc. Natl. Acad. Sci. USA 2014, 111, 5266–5270. [Google Scholar] [CrossRef]
- Wu, L.; Chen, H.; Chen, D.; Wang, S.; Wu, Y.; Wang, B.; Liu, S.; Yue, L.; Yu, J.; Bai, Y. Soil biota diversity and plant diversity both contributed to ecosystem stability in grasslands. Ecol. Lett. 2023, 26, 858–868. [Google Scholar] [CrossRef] [PubMed]
- Wagg, C.; Schlaeppi, K.; Banerjee, S.; Kuramae, E.E.; van der Heijden, M.G.A. Fungal-bacterial diversity and microbiome complexity predict ecosystem functioning. Nat. Commun. 2019, 10, 4841. [Google Scholar] [CrossRef]
- Zhou, Z.; Wang, C.; Luo, Y. Meta-analysis of the impacts of global change factors on soil microbial diversity and functionality. Nat. Commun. 2020, 11, 3072. [Google Scholar] [CrossRef]
- Yang, Y.; Li, T.; Wang, Y.; Cheng, H.; Chang, S.X.; Liang, C.; An, S. Negative effects of multiple global change factors on soil microbial diversity. Soil Biol. Biochem. 2021, 156, 108229. [Google Scholar] [CrossRef]
- Yang, A.; Song, B.; Zhang, W.; Zhang, T.; Li, X.; Wang, H.; Zhu, D.; Zhao, J.; Fu, S. Chronic enhanced nitrogen deposition and elevated precipitation jointly benefit soil microbial community in a temperate forest. Soil Biol. Biochem. 2024, 193, 109397. [Google Scholar] [CrossRef]
- Chen, C.; Xiao, W. The global positive effect of phosphorus addition on soil microbial biomass. Soil Biol. Biochem. 2023, 176, 108882. [Google Scholar] [CrossRef]
- Wang, H.; Liu, S.; Zhang, X.; Mao, Q.; Li, X.; You, Y.; Wang, J.; Zheng, M.; Zhang, W.; Lu, X.; et al. Nitrogen addition reduces soil bacterial richness, while phosphorus addition alters community composition in an old-growth N-rich tropical forest in southern China. Soil Biol. Biochem. 2018, 127, 22–30. [Google Scholar] [CrossRef]
- Wang, K.; Zhang, C.; Chen, H.; Yue, Y.; Zhang, W.; Zhang, M.; Qi, X.; Fu, Z. Karst landscapes of China: Patterns, ecosystem processes and services. Landsc. Ecol. 2019, 34, 2743–2763. [Google Scholar] [CrossRef]
- Jiang, Z.; Lian, Y.; Qin, X. Rocky desertification in Southwest China: Impacts, causes, and restoration. Earth-Sci. Rev. 2014, 132, 1–12. [Google Scholar] [CrossRef]
- Tong, X.; Brandt, M.; Yue, Y.; Horion, S.; Wang, K.; Keersmaecker, W.D.; Tian, F.; Schurgers, G.; Xiao, X.; Luo, Y.; et al. Increased vegetation growth and carbon stock in China karst via ecological engineering. Nat. Sustain. 2018, 1, 44–50. [Google Scholar] [CrossRef]
- Chen, H.; Li, D.; Mao, Q.; Xiao, K.; Wang, K. Resource limitation of soil microbes in karst ecosystems. Sci. Total Environ. 2019, 650, 241–248. [Google Scholar] [CrossRef]
- Chen, H.; Li, D.; Xiao, K.; Wang, K. Soil microbial processes and resource limitation in karst and non-karst forests. Funct. Ecol. 2018, 32, 1400–1409. [Google Scholar] [CrossRef]
- Wang, L.; Yue, Y.; Cui, J.; Liu, H.; Shi, L.; Liang, B.; Li, Q.; Wang, K. Precipitation sensitivity of vegetation growth in southern China depends on geological settings. J. Hydrol. 2024, 643, 131916. [Google Scholar] [CrossRef]
- Liu, M.; Xu, X.; Sun, A.Y.; Wang, K.; Liu, W.; Zhang, X. Is southwestern China experiencing more frequent precipitation extremes? Environ. Res. Lett. 2014, 9, 064002. [Google Scholar] [CrossRef]
- Manzoni, S.; Schaeffer, S.M.; Katul, G.; Porporato, A.; Schimel, J.P. A theoretical analysis of microbial eco-physiological and diffusion limitations to carbon cycling in drying soils. Soil Biol. Biochem. 2014, 73, 69–83. [Google Scholar] [CrossRef]
- Sierra, C.A.; Malghani, S.; Loescher, H.W. Interactions among temperature, moisture, and oxygen concentrations in controlling decomposition rates in a boreal forest soil. Biogeosciences 2017, 14, 703–710. [Google Scholar] [CrossRef]
- de Vries, F.T.; Griffiths, R.I.; Bailey, M.; Craig, H.; Girlanda, M.; Gweon, H.S.; Hallin, S.; Kaisermann, A.; Keith, A.M.; Kretzschmar, M.; et al. Soil bacterial networks are less stable under drought than fungal networks. Nat. Commun. 2018, 9, 3033. [Google Scholar] [CrossRef] [PubMed]
- Morrison, E.W.; Frey, S.D.; Sadowsky, J.J.; van Diepen, L.T.A.; Thomas, W.K.; Pringle, A. Chronic nitrogen additions fundamentally restructure the soil fungal community in a temperate forest. Fungal Ecol. 2016, 23, 48–57. [Google Scholar] [CrossRef]
- Wang, C.; Feng, X.; Guo, P.; Han, G.; Tian, X. Response of degradative enzymes to N fertilization during litter decomposition in a subtropical forest through a microcosm experiment. Ecol. Res. 2010, 25, 1121–1128. [Google Scholar] [CrossRef]
- Kangi, E.; Ridgeway, J.R.; DeHetre, H.D.; Brzostek, E.R. Nitrogen induced soil carbon gains are resistant to loss after the cessation of excess nitrogen inputs. Soil Biol. Biochem. 2025, 200, 109640. [Google Scholar] [CrossRef]
- Ying, J.; Li, X.; Wang, N.; Lan, Z.; He, J.; Bai, Y. Contrasting effects of nitrogen forms and soil pH on ammonia oxidizing microorganisms and their responses to long-term nitrogen fertilization in a typical steppe ecosystem. Soil Biol. Biochem. 2017, 107, 10–18. [Google Scholar] [CrossRef]
- Lu, M.; Yang, Y.; Luo, Y.; Fang, C.; Zhou, X.; Chen, J.; Yang, X.; Li, B. Responses of ecosystem nitrogen cycle to nitrogen addition: A meta-analysis. New Phytol. 2011, 189, 1040–1050. [Google Scholar] [CrossRef] [PubMed]
- Hou, E.; Wen, D.; Jiang, L.; Luo, X.; Kuang, Y.; Lu, X.; Chen, C.; Allen, K.T.; He, X.; Huang, X.; et al. Latitudinal patterns of terrestrial phosphorus limitation over the globe. Ecol. Lett. 2021, 24, 1420–1431. [Google Scholar] [CrossRef]
- Liu, L.; Gundersen, P.; Zhang, W.; Zhang, T.; Chen, H.; Mo, J. Effects of nitrogen and phosphorus additions on soil microbial biomass and community structure in two reforested tropical forests. Sci. Rep. 2015, 5, 14378. [Google Scholar] [CrossRef]
- Xiao, H.; Wang, B.; Lu, S.; Chen, D.; Wu, Y.; Zhu, Y.; Hu, S.; Bai, Y. Soil acidification reduces the effects of short-term nutrient enrichment on plant and soil biota and their interactions in grasslands. Glob. Change Biol. 2020, 26, 4626–4637. [Google Scholar] [CrossRef]
- Yue, K.; Fornara, D.A.; Yang, W.; Peng, Y.; Peng, C.; Liu, Z.; Wu, F. Influence of multiple global change drivers on terrestrial carbon storage: Additive effects are common. Ecol. Lett. 2017, 20, 663–672. [Google Scholar] [CrossRef]
- Luo, Y.; Jiang, L.; Niu, S.; Zhou, X. Nonlinear responses of land ecosystems to variation in precipitation. New Phytol. 2017, 214, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; He, K.; Zhang, Q.; Han, M.; Zhu, B. Changes in plant inputs alter soil carbon and microbial communities in forest ecosystems. Glob. Change Biol. 2022, 28, 3426–3440. [Google Scholar] [CrossRef]
- Li, Y.; Niu, S.; Yu, G. Aggravated phosphorus limitation on biomass production under increasing nitrogen loading: A meta-analysis. Glob. Change Biol. 2016, 22, 934–943. [Google Scholar] [CrossRef]
- Long, X.; Li, J.; Liao, X.; Wang, J.; Zhang, W.; Wang, K.; Zhao, J. Stable Soil Biota Network Enhances Soil Multifunctionality in Agroecosystems. Glob. Change Biol. 2025, 31, e70041. [Google Scholar] [CrossRef]
- Yuan, M.M.; Guo, X.; Wu, L.; Zhang, Y.; Xiao, N.; Ning, D.; Shi, Z.; Zhou, X.; Wu, L.; Yang, Y.; et al. Climate warming enhances microbial network complexity and stability. Nat. Clim. Change 2021, 11, 343–348. [Google Scholar] [CrossRef]
- Chen, Q.-L.; Hu, H.-W.; Sun, A.-Q.; Zhu, Y.-G.; He, J.-Z. Aridity decreases soil protistan network complexity and stability. Soil Biol. Biochem. 2022, 166, 108575. [Google Scholar] [CrossRef]
- Wang, C.; Shi, Z.; Li, A.; Geng, T.; Liu, L.; Liu, W. Long-term nitrogen input reduces soil bacterial network complexity by shifts in life history strategy in temperate grassland. iMeta 2024, 3, e194. [Google Scholar] [CrossRef]
- Li, J.; Zhao, J.; Liao, X.; Yi, Q.; Zhang, W.; Lin, H.; Liu, K.; Peng, P.; Wang, K. Long-term returning agricultural residues increases soil microbe-nematode network complexity and ecosystem multifunctionality. Geoderma 2023, 430, 116340. [Google Scholar] [CrossRef]
- Hood-Nowotny, R.; Umana, N.H.-N.; Inselbacher, E.; Oswald- Lachouani, P.; Wanek, W. Alternative Methods for Measuring Inorganic, Organic, and Total Dissolved Nitrogen in Soil. Soil Sci. Soc. Am. J. 2010, 74, 1018–1027. [Google Scholar] [CrossRef]
- Nelson, D.W.; Sommers, L. Total carbon, organic carbon and organic matter. In Methods of Soil Analysis Part 2. Chemical and Microbial Properties; American Society of Agronomy, Inc.: Madison, WI, USA; Soil Science Society of America, Inc.: Madison, WI, USA, 1982; pp. 408–411. [Google Scholar]
- Bremner, J.M. Determination of nitrogen in soil by the Kjeldahl method. J. Agric. Sci. 1960, 55, 11–33. [Google Scholar] [CrossRef]
- Olsen, S.R. Estimation of Available Phosphorus in Soils by Extraction with Sodium Bicarbonate; United States Department of Agriculture: Washington, DC, USA, 1954. [Google Scholar]
- Beegle, D.; Denning, J. Recommended Chemical Soil Test Procedures for the North Central Region. In North Central Regional Research Publication; Missouri Agricultural Experiment Station: Columbia, MO, USA, 1998; pp. 21–30. [Google Scholar]
- Hendershot, W.H.; Lalande, H.; Carter, M.R. Ion exchange and exchangeable cations. In Soil Sampling and Methods of Analysis; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Zhou, Y.; Liu, D.; Li, F.; Dong, Y.; Jin, Z.; Liao, Y.; Li, X.; Peng, S.; Delgado-Baquerizo, M.; Li, X. Superiority of native soil core microbiomes in supporting plant growth. Nat. Commun. 2024, 15, 6599. [Google Scholar] [CrossRef]
- Lekberg, Y.; Arnillas, C.A.; Borer, E.T.; Bullington, L.S.; Fierer, N.; Kennedy, P.G.; Leff, J.W.; Luis, A.D.; Seabloom, E.W.; Henning, J.A. Nitrogen and phosphorus fertilization consistently favor pathogenic over mutualistic fungi in grassland soils. Nat. Commun. 2021, 12, 3484. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, S.; Li, R.; Zhang, J.; Liu, Y.; Lv, L.; Zhu, H.; Wu, W.; Li, W. Plant cultivars imprint the rhizosphere bacterial community composition and association networks. Soil Biol. Biochem. 2017, 109, 145–155. [Google Scholar] [CrossRef]
- Magoc, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhou, Y.; Chen, Y.; Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinformatics 2018, 34, i884–i890. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Wen, T.; Xie, P.; Yang, S.; Niu, G.; Liu, X.; Ding, Z.; Xue, C.; Liu, Y.-X.; Shen, Q.; Yuan, J. ggClusterNet: An R package for microbiome network analysis and modularity-based multiple network layouts. iMeta 2022, 1, e32. [Google Scholar] [CrossRef]
- Li, Y.; Chen, Z.; Wagg, C.; Castellano, M.J.; Zhang, N.; Ding, W. Soil organic carbon loss decreases biodiversity but stimulates multitrophic interactions that promote belowground metabolism. Glob. Change Biol. 2023, 30, e17101. [Google Scholar] [CrossRef] [PubMed]
- Jiao, S.; Yang, Y.; Xu, Y.; Zhang, J.; Lu, Y. Balance between community assembly processes mediates species coexistence in agricultural soil microbiomes across eastern China. ISME J. 2020, 14, 202–216. [Google Scholar] [CrossRef]
- Wu, M.-H.; Chen, S.-Y.; Chen, J.-W.; Xue, K.; Chen, S.-L.; Wang, X.-M.; Chen, T.; Kang, S.-C.; Rui, J.-P.; Thies, J.E.; et al. Reduced microbial stability in the active layer is associated with carbon loss under alpine permafrost degradation. Proc. Natl. Acad. Sci. USA 2021, 118, e2025321118. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, B.; Simpson, G.; Solymos, P.; Stevens, H.; Wagner, H. Vegan: Community Ecology Package, R Package Version 2.2-1; The R Foundation for Statistical Computing: Vienna, Austria, 2015.
- Sunagawa, S.; Coelho, L.P.; Chaffron, S.; Kultima, J.R.; Labadie, K.; Salazar, G.; Djahanschiri, B.; Zeller, G.; Mende, D.R.; Alberti, A.; et al. Structure and function of the global ocean microbiome. Science 2015, 348, 1261359. [Google Scholar] [CrossRef]
- Huang, H. linkET: Everything is Linkable. R Package Version 0.0.3. 2021. Available online: https://github.com/Hy4m/linkET (accessed on 8 October 2024).
- Hu, P.; Xiao, J.; Zhang, W.; Xiao, L.; Yang, R.; Xiao, D.; Zhao, J.; Wang, K. Response of soil microbial communities to natural and managed vegetation restoration in a subtropical karst region. Catena 2020, 195, 104849. [Google Scholar] [CrossRef]
- Umair, M.; Sun, N.; Du, H.; Hui, N.; Altaf, M.; Du, B.; Yin, S.; Liu, C. Bacterial Communities Are More Sensitive to Water Addition Than Fungal Communities Due to Higher Soil K and Na in a Degraded Karst Ecosystem of Southwestern China. Front. Microbiol. 2020, 11, 562546. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, H.Y.H.; Chen, C.; Peng, S. Water availability regulates negative effects of species mixture on soil microbial biomass in boreal forests. Soil Biol. Biochem. 2019, 139, 107634. [Google Scholar] [CrossRef]
- Meier, I.C.; Finzi, A.C.; Phillips, R.P. Root exudates increase N availability by stimulating microbial turnover of fast-cycling N pools. Soil Biol. Biochem. 2017, 106, 119–128. [Google Scholar] [CrossRef]
- Zheng, L.; Wu, S.; Lu, L.; Li, T.; Liu, Z.; Li, X.; Li, H. Unraveling the interaction effects of soil temperature and moisture on soil nematode community: A laboratory study. Eur. J. Soil Biol. 2023, 118, 103537. [Google Scholar] [CrossRef]
- Song, M.; Li, X.; Jing, S.; Lei, L.; Wang, J.; Wan, S. Responses of soil nematodes to water and nitrogen additions in an old-field grassland. Appl. Soil Ecol. 2016, 102, 53–60. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, J.; Ren, Z.; Hu, Z.; Tian, S.; Fan, W.; Chen, X.; Griffiths, B.S.; Hu, F.; Liu, M. Root traits mediate functional guilds of soil nematodes in an ex-arable field. Soil Biol. Biochem. 2020, 151, 108038. [Google Scholar] [CrossRef]
- Fellbaum, C.R.; Gachomo, E.W.; Beesetty, Y.; Choudhari, S.; Strahan, G.D.; Pfeffer, P.E.; Kiers, E.T.; Bücking, H. Carbon availability triggers fungal nitrogen uptake and transport in arbuscular mycorrhizal symbiosis. Proc. Natl. Acad. Sci. USA 2012, 109, 2666–2671. [Google Scholar] [CrossRef]
- Xiong, B.-J.; Kleinsteuber, S.; Sträuber, H.; Dusny, C.; Harms, H.; Wick Lukas, Y. Impact of Fungal Hyphae on Growth and Dispersal of Obligate Anaerobic Bacteria in Aerated Habitats. mBio 2022, 13, e00769-00722. [Google Scholar] [CrossRef] [PubMed]
- Forczek, S.T.; Bukovská, P.; Püschel, D.; Janoušková, M.; Blažková, A.; Jansa, J. Drought rearranges preferential carbon allocation to arbuscular mycorrhizal community members co-inhabiting roots of Medicago truncatula. Environ. Exp. Bot. 2022, 199, 104897. [Google Scholar] [CrossRef]
- Wang, R.; Cavagnaro, T.R.; Jiang, Y.; Keitel, C.; Dijkstra, F.A. Carbon allocation to the rhizosphere is affected by drought and nitrogen addition. J. Ecol. 2021, 109, 3699–3709. [Google Scholar] [CrossRef]
- Zhao, A.; Liu, L.; Chen, B.; Fu, W.; Xie, W.; Xu, T.; Zhang, W.; Ye, Q.; Feng, H.; Fu, S. Soil fungal community is more sensitive to nitrogen deposition than increased rainfall in a mixed deciduous forest of China. Soil Ecol. Lett. 2020, 2, 20–32. [Google Scholar] [CrossRef]
- Ma, S.; Chen, X.; Su, H.; Xing, A.; Chen, G.; Zhu, J.; Zhu, B.; Fang, J. Phosphorus addition decreases soil fungal richness and alters fungal guilds in two tropical forests. Soil Biol. Biochem. 2022, 175, 108836. [Google Scholar] [CrossRef]
- Wang, Q.-C.; Jin, S.-S.; Liu, S.; Song, G.; Duan, C.; Lü, P.-P.; Maitra, P.; Xie, L.; Lin, Y.; Hu, H.-W.; et al. Phosphorus amendment alters soil arbuscular mycorrhizal fungal functional guild compositions in a subtropical forest. J. Soils Sediments 2023, 23, 2700–2711. [Google Scholar] [CrossRef]
- Shi, X.; Hu, H.-W.; Wang, J.; He, J.-Z.; Zheng, C.; Wan, X.; Huang, Z. Niche separation of comammox Nitrospira and canonical ammonia oxidizers in an acidic subtropical forest soil under long-term nitrogen deposition. Soil Biol. Biochem. 2018, 126, 114–122. [Google Scholar] [CrossRef]
- Li, C.; Hu, H.-W.; Chen, Q.-L.; Chen, D.; He, J.-Z. Niche differentiation of clade A comammox Nitrospira and canonical ammonia oxidizers in selected forest soils. Soil Biol. Biochem. 2020, 149, 107925. [Google Scholar] [CrossRef]
- Yu, Y.; Chen, L.; Duan, W. Responses of bacterial and fungal communities to short-term nitrogen and phosphorus additions in temperate forest soil aggregates in northeastern China. Appl. Soil Ecol. 2024, 197, 105341. [Google Scholar] [CrossRef]
- Yan, Y.; Sun, X.; Sun, F.; Zhao, Y.; Sun, W.; Guo, J.; Zhang, T. Sensitivity of soil fungal and bacterial community compositions to nitrogen and phosphorus additions in a temperate meadow. Plant Soil 2022, 471, 477–490. [Google Scholar] [CrossRef]
- Fierer, N.; Lauber, C.L.; Ramirez, K.S.; Zaneveld, J.; Bradford, M.A.; Knight, R. Comparative metagenomic, phylogenetic and physiological analyses of soil microbial communities across nitrogen gradients. ISME J. 2012, 6, 1007–1017. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, H.; Zheng, C.; Wu, X.; Zhao, Y.; Li, X.; Liu, H.; Dong, L.; Lu, Z.; Zhou, J.; et al. Bacteria life-history strategies and the linkage of soil C-N-P stoichiometry to microbial resource limitation differed in karst and non-karst plantation forests in southwest China. Catena 2023, 231, 107341. [Google Scholar] [CrossRef]
- Xiao, D.; He, X.; Zhang, W.; Hu, P.; Sun, M.; Wang, K. Comparison of bacterial and fungal diversity and network connectivity in karst and non-karst forests in southwest China. Sci. Total Environ. 2022, 822, 153179. [Google Scholar] [CrossRef]
- Xiao, D.; Gai, S.; He, X.; Zhang, W.; Hu, P.; Soromotin, A.V.; Kuzyakov, Y.; Wang, K. Habitat heterogeneity drives arbuscular mycorrhizal fungi and shrub communities in karst ecosystems. Catena 2023, 233, 107513. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, H.; Chen, W.; Xue, H.; Liu, H.; Wang, J.; Mao, S.; Liu, G.; Xue, S. The combined nitrogen and phosphorus fertilizer application reduced soil multifunctionality in Qinghai-Tibet plateau grasslands, China. Eur. J. Soil Biol. 2024, 123, 103684. [Google Scholar] [CrossRef]
- Shi, Y.; Samhita, S.; Loeppmann, S.; Zimmermann, I.; Dippold, M.A.; Spielvogel, S. Redundancy of microbial P mobilization in beech forest soils with contrasting P stock: A microbial dilution experiment. Appl. Soil Ecol. 2025, 206, 105824. [Google Scholar] [CrossRef]
- Peng, T.; Wang, S.-j. Effects of land use, land cover and rainfall regimes on the surface runoff and soil loss on karst slopes in southwest China. Catena 2012, 90, 53–62. [Google Scholar] [CrossRef]
- Liu, X.; Fu, Z.; Zhang, W.; Xiao, S.; Chen, H.; Wang, K. Soluble carbon loss through multiple runoff components in the shallow subsurface of a karst hillslope: Impact of critical zone structure and land use. Catena 2023, 222, 106868. [Google Scholar] [CrossRef]
- Tibbett, M.; Gil-Martínez, M.; Fraser, T.; Green, I.D.; Duddigan, S.; De Oliveira, V.H.; Raulund-Rasmussen, K.; Sizmur, T.; Diaz, A. Long-term acidification of pH neutral grasslands affects soil biodiversity, fertility and function in a heathland restoration. Catena 2019, 180, 401–415. [Google Scholar] [CrossRef]
- Li, J.; Zhao, J.; Liao, X.; Wang, W.; Long, X.; Liu, Y.; Xiao, J.; Zhang, W.; Wang, K. Nitrogen deposition exhibits limited influence on soil nematode energy fluxes and soil carbon and nitrogen mineralization in a typical karst ecosystem. Soil Ecol. Lett. 2025, 7, 250298. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, H.; Tan, X.; Fu, S.; Liu, D.; Shen, W. Increased precipitation alters the effects of nitrogen deposition on soil bacterial and fungal communities in a temperate forest. Sci. Total Environ. 2024, 916, 170017. [Google Scholar] [CrossRef] [PubMed]
- Hestrin, R.; Kan, M.; Lafler, M.; Wollard, J.; Kimbrel, J.A.; Ray, P.; Blazewicz, S.J.; Stuart, R.; Craven, K.; Firestone, M.; et al. Plant-associated fungi support bacterial resilience following water limitation. ISME J. 2022, 16, 2752–2762. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, F.A.; He, M.; Johansen, M.P.; Harrison, J.J.; Keitel, C. Plant and microbial uptake of nitrogen and phosphorus affected by drought using 15N and 32P tracers. Soil Biol. Biochem. 2015, 82, 135–142. [Google Scholar] [CrossRef]
- Wu, J.; Qi, L.; Huang, T.; Wang, J.; Sun, Q. A short period of revegetation and fertilization increased the nutrients, enzyme activities, and bacterial community diversity in backfill soils. Appl. Soil Ecol. 2023, 189, 104959. [Google Scholar] [CrossRef]
- Chen, L.; Sun, J.; Baoyin, T. Effects of artificial restoration on vertical distribution of soil carbon storage following revegetation in a semiarid grassland of North China. Land Degrad. Dev. 2023, 34, 805–814. [Google Scholar] [CrossRef]
- Plett, K.L.; Snijders, F.; Castañeda-Gómez, L.; Wong-Bajracharya, J.W.H.; Anderson, I.C.; Carrillo, Y.; Plett, J.M. Nitrogen fertilization differentially affects the symbiotic capacity of two co-occurring ectomycorrhizal species. Environ. Microbiol. 2022, 24, 309–323. [Google Scholar] [CrossRef]
- Keyhani, A.B.; He, W.; Teng, M.; Yan, Z.; Ma, Z.; Xu, J.; Fayaz, M.; Zhou, C.; Wei, P.; Wang, P. Effect of mineral fertilizers on microorganisms community characteristic during leaf litter decomposition under Pinus massoniana in a subtropical forest. Appl. Soil Ecol. 2024, 199, 105421. [Google Scholar] [CrossRef]
- Addison, S.L.; Smaill, S.J.; Garrett, L.G.; Wakelin, S.A. Fertiliser use has multi-decadal effects on microbial diversity and functionality of forest soils. Appl. Soil Ecol. 2021, 163, 103964. [Google Scholar] [CrossRef]
- Gu, S.; Zhou, X.; Yu, H.; Yan, H.; Wang, Y.; Liu, Y.; Wang, Z.; Feng, K.; Du, X.; Lu, G.; et al. Microbial and chemical fertilizers for restoring degraded alpine grassland. Biol. Fertil. Soils 2023, 59, 911–926. [Google Scholar] [CrossRef]
- Yan, T.; Xue, J.; Zhou, Z.; Wu, Y. Biochar-based fertilizer amendments improve the soil microbial community structure in a karst mountainous area. Sci. Total Environ. 2021, 794, 148757. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhao, W.; Zhou, Z.; Huang, G.; Wang, X.; Han, Q.; Liu, G. The Application of Mixed Organic and Inorganic Fertilizers Drives Soil Nutrient and Bacterial Community Changes in Teak Plantations. Microorganisms 2022, 10, 958. [Google Scholar] [CrossRef]
- Xu, G.; Xiao, J.; Oliver, D.M.; Yang, Z.; Xiong, K.; Zhao, Z.; Zheng, L.; Fan, H.; Zhang, F. Spatio-temporal characteristics and determinants of anthropogenic nitrogen and phosphorus inputs in an ecologically fragile karst basin: Environmental responses and management strategies. Ecol. Indic. 2021, 133, 108453. [Google Scholar] [CrossRef]
- Carpenter, S.R. Eutrophication of aquatic ecosystems: Bistability and soil phosphorus. Proc. Natl. Acad. Sci. USA 2005, 102, 10002–10005. [Google Scholar] [CrossRef]
- Brooks, B.W.; Lazorchak, J.M.; Howard, M.D.A.; Johnson, M.V.V.; Morton, S.L.; Perkins, D.A.K.; Reavie, E.D.; Scott, G.I.; Smith, S.A.; Steevens, J.A. Are harmful algal blooms becoming the greatest inland water quality threat to public health and aquatic ecosystems? Environ. Toxicol. Chem. 2016, 35, 6–13. [Google Scholar] [CrossRef]
- Cardinale, B.J.; Duffy, J.E.; Gonzalez, A.; Hooper, D.U.; Perrings, C.; Venail, P.; Narwani, A.; Mace, G.M.; Tilman, D.; Wardle, D.A.; et al. Biodiversity loss and its impact on humanity. Nature 2012, 486, 59–67. [Google Scholar] [CrossRef]
- Zhao, C.; Miao, Y.; Yu, C.; Zhu, L.; Wang, F.; Jiang, L.; Hui, D.; Wan, S. Soil microbial community composition and respiration along an experimental precipitation gradient in a semiarid steppe. Sci. Rep. 2016, 6, 24317. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Qiu, Y.; Zhao, Y.; Wang, S.; Deng, J.; Chen, M.; Xu, X.; Wang, H.; Bai, T.; He, T.; et al. Moderate precipitation reduction enhances nitrogen cycling and soil nitrous oxide emissions in a semi-arid grassland. Glob. Change Biol. 2023, 29, 3114–3129. [Google Scholar] [CrossRef] [PubMed]
- Conley, D.J.; Paerl, H.W.; Howarth, R.W.; Boesch, D.F.; Seitzinger, S.P.; Havens, K.E.; Lancelot, C.; Likens, G.E. Controlling Eutrophication: Nitrogen and Phosphorus. Science 2009, 323, 1014–1015. [Google Scholar] [CrossRef] [PubMed]
- Schindler, D.W.; Carpenter, S.R.; Chapra, S.C.; Hecky, R.E.; Orihel, D.M. Reducing Phosphorus to Curb Lake Eutrophication is a Success. Environ. Sci. Technol. 2016, 50, 8923–8929. [Google Scholar] [CrossRef]





| Variables | Treatments | |||||||
|---|---|---|---|---|---|---|---|---|
| C | W | N | P | WN | WP | NP | WNP | |
| SWC | 46.6 ± 2.5 | 53.3 ± 1.3 | 47.0 ± 1.0 | 50.9 ± 3.2 | 50.4 ± 3.1 | 51.5 ± 0.9 | 49.1 ± 1.4 | 53.2 ± 1.4 |
| pH | 7.5 ± 0.2 | 7.3 ± 0.2 | 7.4 ± 0.2 | 7.5 ± 0.2 | 7.1 ± 0.2 | 7.3 ± 0.2 | 7.1 ± 0.2 | 7.0 ± 0.1 |
| SOC | 53.4 ± 5.1 | 55.6 ± 4.9 | 48.1 ± 3.0 | 56.1 ± 6.0 | 50.0 ± 5.6 | 51.1 ± 5.5 | 50.4 ± 1.2 | 55.6 ± 3.9 |
| TN | 4.9 ± 0.3 | 5.1 ± 0.7 | 6.2 ± 1.8 | 5.6 ± 0.5 | 4.7 ± 0.5 | 4.5 ± 0.4 | 3.8 ± 0.4 | 4.4 ± 0.4 |
| TP | 1.9 ± 0.0 | 1.8 ± 0.2 | 1.6 ± 0.2 | 2.2 ± 0.2 | 1.9 ± 0.3 | 1.8 ± 0.2 | 1.8 ± 0.1 | 1.8 ± 0.1 |
| TK | 4.0 ± 0.6 | 4.4 ± 0.5 | 4.4 ± 0.4 | 4.0 ± 0.4 | 4.6 ± 0.4 | 4.6 ± 0.5 | 4.3 ± 0.4 | 5.0 ± 0.5 |
| C:N | 11.0 ± 1.1 bc | 11.2 ± 0.7 ab | 9.1 ± 1.2 c | 10.0 ± 0.7 c | 10.6 ± 0.6 bc | 11.5 ± 0.5 abc | 14.0 ± 1.6 a | 13.1 ± 1.3 ab |
| C:P | 28.8 ± 2.8 | 31.8 ± 1.3 | 30.1 ± 1.2 | 27.4 ± 4.2 | 27.5 ± 2.4 | 29.2 ± 1.9 | 28.9 ± 1.1 | 31.5 ± 1.1 |
| N:P | 2.6 ± 0.1 | 2.9 ± 0.1 | 3.6 ± 0.6 | 2.7 ± 0.3 | 2.6 ± 0.2 | 2.6 ± 0.3 | 2.2 ± 0.3 | 2.5 ± 0.2 |
| NH4+ | 11.5 ± 1.2 | 11.5 ± 1.2 | 9.6 ± 0.6 | 11.5 ± 0.9 | 11.0 ± 0.7 | 11.1 ± 1.2 | 9.6 ± 0.6 | 12.1 ± 1.0 |
| NO3− | 16.3 ± 1.9 bc | 16.6 ± 1.6 bc | 25.8 ± 2.3 ab | 20.3 ± 6.1 bc | 32.6 ± 6.4 a | 14.8 ± 1.1 c | 23.8 ± 1.8 abc | 25.0 ± 4.3 abc |
| AP | 4.3 ± 0.6 | 3.0 ± 0.5 | 3.3 ± 0.8 | 4.3 ± 0.2 | 3.6 ± 0.7 | 3.3 ± 0.7 | 4.4 ± 0.2 | 4.3 ± 0.5 |
| AK | 108.4 ± 3.0 | 108.6 ± 4.5 | 98.9 ± 4.7 | 112.4 ± 6.0 | 103.1 ± 4.6 | 108.8 ± 6.8 | 97.2 ± 3.7 | 107.8 ± 6.1 |
| Ca2+ | 3.1 ± 0.3 | 2.9 ± 0.3 | 2.7 ± 0.2 | 3.3 ± 0.3 | 2.7 ± 0.2 | 2.6 ± 0.3 | 2.7 ± 0.2 | 2.7 ± 0.2 |
| Mg2+ | 3.3 ± 0.0 | 3.0 ± 0.2 | 3.0 ± 0.2 | 3.6 ± 0.2 | 2.8 ± 0.3 | 2.8 ± 0.3 | 3.0 ± 0.2 | 2.9 ± 0.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Zhao, J.; Liao, X.; Long, X.; Wang, W.; Hu, P.; Zhang, W.; Wang, K. Precipitation Alleviates Adverse Effects of Nitrogen and Phosphorus Enrichment on Soil Microbial Co-Occurrence Network Complexity and Stability in Karst Shrubland. Microorganisms 2025, 13, 2012. https://doi.org/10.3390/microorganisms13092012
Li J, Zhao J, Liao X, Long X, Wang W, Hu P, Zhang W, Wang K. Precipitation Alleviates Adverse Effects of Nitrogen and Phosphorus Enrichment on Soil Microbial Co-Occurrence Network Complexity and Stability in Karst Shrubland. Microorganisms. 2025; 13(9):2012. https://doi.org/10.3390/microorganisms13092012
Chicago/Turabian StyleLi, Jiangnan, Jie Zhao, Xionghui Liao, Xianwen Long, Wenyu Wang, Peilei Hu, Wei Zhang, and Kelin Wang. 2025. "Precipitation Alleviates Adverse Effects of Nitrogen and Phosphorus Enrichment on Soil Microbial Co-Occurrence Network Complexity and Stability in Karst Shrubland" Microorganisms 13, no. 9: 2012. https://doi.org/10.3390/microorganisms13092012
APA StyleLi, J., Zhao, J., Liao, X., Long, X., Wang, W., Hu, P., Zhang, W., & Wang, K. (2025). Precipitation Alleviates Adverse Effects of Nitrogen and Phosphorus Enrichment on Soil Microbial Co-Occurrence Network Complexity and Stability in Karst Shrubland. Microorganisms, 13(9), 2012. https://doi.org/10.3390/microorganisms13092012









