Abstract
To develop sustainable strategies for mitigating ruminal methanogenesis and improving nitrogen efficiency in dairy systems, this study investigated how low-dose tannic acid (T), tea polyphenols (TP), and their combination (T+TP; 50:50) modulate rumen microbiota and function. A sample of Holstein cows were given four dietary treatments: (1) control (basal diet); (2) T (basal diet + 0.4% DM tannic acid); (3) TP (basal diet + 0.4% DM tea polyphenols); and (4) T+TP (basal diet + 0.2% DM tannic acid + 0.2% DM tea polyphenols). We comprehensively analyzed rumen fermentation, methane production, nutrient digestibility, milk parameters, and microbiota dynamics. Compared with the control group, all diets supplemented with additives significantly reduced enteric methane production (13.68% for T, 11.40% for TP, and 10.89% for T+TP) and significantly increased milk protein yield. The crude protein digestibility significantly increased in the T group versus control. The results did not impair rumen health or fiber digestion. Critically, microbiota analysis revealed treatment-specific modulation: the T group showed decreased Ruminococcus flavefaciens abundance, while all tannin treatments reduced abundances of Ruminococcus albus and total methanogens. These microbial shifts corresponded with functional outcomes—most notably, the T+TP synergy drove the largest reductions in rumen ammonia-N (34.5%) and milk urea nitrogen (21.1%). Supplementation at 0.4% DM, particularly the T+TP combination, effectively enhances nitrogen efficiency and milk protein synthesis while reducing methane emissions through targeted modulation of key rumen microbiota populations, suggesting potential sustainability benefits linked to altered rumen fermentation.
1. Introduction
The rising atmospheric concentration of greenhouse gases represents a critical global environmental challenge [1]. Mitigating GHG emissions, particularly methane (CH4), has become an urgent international priority. Livestock production contributes significantly to anthropogenic emissions, accounting for 14.5% of the global total, with enteric CH4 comprising 39% of this sector’s emissions [2]. Within ruminant livestock, CH4 is primarily generated through microbial fermentation of carbohydrates in the rumen [3]. Projected increases in global demand for ruminant-derived products (meat, milk) will necessitate herd expansion, inevitably leading to higher absolute methane (CH4) emissions. While system intensification can reduce emissions per unit of product, these efficiency gains may not be sufficient to achieve the necessary absolute reductions in methane output. Consequently, mitigating the associated environmental impacts remains a significant challenge. These emissions impose a dual burden: they exacerbate climate change and represent substantial energy loss, estimated at 2–12% of gross feed energy intake, equivalent to ~6% of ingested energy or 12% of digestible energy in cattle [4,5]. Consequently, developing effective feed additives to suppress ruminal methanogenesis is essential for achieving sustainable livestock production and enhancing feed conversion efficiency.
The methanogenic potential of the rumen microbiota is significantly influenced by its bacterial structure. In the strictly anaerobic rumen environment [6], key microbes (Protozoa, R. albus, R. flavefaciens, and R. amylophilus) ferment dietary fiber and starch into volatile fatty acids (VFAs). This fermentation process generates substantial hydrogen gas. Methanogens primarily utilize this hydrogen to reduce carbon dioxide into methane [7]. When methanogens are highly active, excessive hydrogen consumption can alter the balance of hydrogen partial pressure and shift fermentation patterns. This typically redirects the hydrogen sink toward methane production rather than propionate synthesis, increasing the proportion of acetate and butyrate. Consequently, hydrogen is diverted from propionate synthesis to methane generation, impacting microbial nitrogen metabolism. Since propionate synthesis requires hydrogen, its positive correlation with nitrogen efficiency indicates that enhancing hydrogen utilization can boost nitrogen assimilation. Meanwhile, R. amylophilus produces LA (lactic acid), while Prevotella uses it to generate propionate through the succinate pathway. Reducing methane emissions may thus elevate propionate levels, enhancing host energy and increasing milk protein content. Therefore, developing additives that leverage these microbial functional dynamics and interaction mechanisms is crucial for effective methane reduction and improved animal production efficiency.
In recent years, numerous plant extracts rich in secondary metabolites (including terpenoids, neem extracts, saponins, and flavonoids) have been evaluated for their potential to reduce ruminal methane (CH4) production. Studies consistently report the ability of these additives to inhibit CH4 generation [8,9,10,11,12]. Among these, tannins—natural polyphenolic compounds found in plants such as shrubs, legumes, and cereals—are categorized primarily into hydrolysable tannins (HT) and condensed tannins (CT) based on structural differences [13], and have garnered significant attention in methane mitigation research. Existing research clearly demonstrates that hydrolysable tannins (HT) effectively inhibit the activity of ruminal methanogens, thereby reducing methane production [14]. By binding to macromolecules, CT reduces the degradation of proteins and carbohydrates, thereby significantly enhancing its functional efficacy [15]. Battelli et al. [16] demonstrated that supplementing 8% quebracho CT reduced cumulative CH4 production by 17.5% during 48 h fermentation, significantly exceeding the 10.4% reduction achieved with chestnut HT. These findings underscore CT potential as a targeted methane inhibitor in ruminant nutrition strategies. However, tea polyphenols (TP)—a potent source of CT rich in epigallocatechin gallate [17]—although well-documented for their antimicrobial and antioxidant efficacy in monogastric animals, remain underexplored in ruminants. Notably, emerging evidence suggests synergistic potential between HT and CT [18,19], yet the combinatorial effects and underlying mechanisms are poorly characterized. The role of TP in dairy production systems is virtually unstudied. Beyond methane reduction, tannins play a crucial role in modulating rumen microbial function, promoting microbial protein synthesis and nitrogen utilization efficiency [20], while also demonstrating potential as antibiotic alternatives [21]. Specifically, HT has been reported to reduce ruminal excessive protein degradation and methane generation by inhibiting specific rumen microbial functions. Simultaneously, it increases protein digestion and absorption in the small intestine, ultimately enhancing nitrogen (N) utilization efficiency in ruminants [22,23].
Based on these findings, we hypothesize that the individual and combined supplementation of two tannins at equal doses can effectively reduce enteric CH4 emissions by altering the rumen microbiota. This study aimed to suppress methanogens and Ruminococcus albus while preserving key fibrolytic communities, explaining the observed maintenance of fiber digestibility alongside methane mitigation, without negatively impacting animal production. This study aims to investigate the effects of individual and combined tannin supplementation on rumen fermentation (fermentation parameters, CH4 production, and microbial community structure), nutrient digestibility, lactation performance, and blood biochemical parameters in dairy cows.
2. Materials and Methods
2.1. Experimental Design and Feeding Management
All experimental procedures involving animal care and management were authorized by the Institutional Animal Care and Use Committee of Shanxi Agricultural University located in Taigu District, Jinzhong City, Shanxi Province, China (Approval code. SXAU-EAW-2022C.RD.010025174. Approval Date: 25 November 2022). The experimental pasture is located at Shanxi Wangxiangyuan Animal Husbandry Co., Ltd., situated west of Gao Village, Beige Town, Xiaodian District, Taiyuan, Shanxi Province, China. The animal study was performed according to the Regulation on the Administration of Laboratory Animals (2017 Revision) promulgated by the State Council.
Twenty-four dairy cows with similar parity (second), lactation days (130 ± 9 d), milk yield (36.0 ± 1.1 kg), body weight (635 ± 8 kg), and body condition score (2.96 ± 0.08) were randomly assigned into four groups, with six cows per group. The experimental treatments were as follows: (1) group one was fed a basal diet (C, control); (2) group two was fed a basal diet plus 0.4% DM tannic acid (T, tannin); (3) group four was fed a basal diet plus 0.4% DM tea polyphenols (TP, tea polyphenols); (4) group three was fed a basal diet plus 0.2% DM tannic acid and 0.2% DM tea polyphenols (T+TP, tannin+tea polyphenols). The cows were given a 10-day pre-experimental period and a 40-day formal experimental period. During the pre-experimental period, the cows’ feeding, rumination, feces, and body condition were observed. The cows’ TMR (Total Mixed Diet) was formulated according to NRC [24]. The diet composition and analysis levels of the experimental diets are shown in Table 1.

Table 1.
Composition and nutritional value of the basal diet.
2.2. Milk Yield Recording and Milk Sample Collection
On days 10, 20, and 30 of the formal period, milk samples were collected and the daily milk yield of the cows was recorded to calculate the average milk yield over 10 days. For each sampling, a mixture of morning, afternoon, and evening milk (in a ratio of 4:3:3) was collected, totaling 50 mL. Potassium dichromate was added as a preservative, and the samples were stored at a low temperature for subsequent milk component analysis.
2.3. Sample Collection of Blood, Ruminal Fluid, and Feces
Commencing on day 31 of the experimental period, fecal samples were collected rectally from each cow three times daily (morning, noon, evening) for 5 consecutive days. Samples were immediately mixed with a 25% aqueous tartaric acid solution (10% w/v) and stored at −20 °C. On the morning of day 40 prior to feeding, blood was collected from the tail vein using 10 mL vacuum tubes. Samples were centrifuged at 1500× g for 10 min to obtain serum. The supernatant serum was aliquoted into 2 mL microcentrifuge tubes and stored at −20 °C. On day 40 at 3 h post-morning feeding, rumen fluid (about 100 mL) was collected using an ororuminal probe equipped with a metal strainer and manual pump with glass container. Fluid was filtered through four layers of sterile cheesecloth. pH was measured immediately. Filtered rumen fluid was then aliquoted into 10 mL sterile tubes and stored at −80 °C for subsequent analysis.
2.4. Methane Gas Collection
From day 34 of the experiment, gas was collected from each cow for two consecutive days. A respiratory mask (Beijing Zhengfang Xingda Technology Development Co., Ltd., Beijing, China) gas collection device was used [25]. Gas collection was conducted four times daily: at 6:00 (one hour before feeding), 12:00 (representing the daytime rest period), 20:00 (one hour after feeding), and 24:00 (representing the nighttime rest period). Each time, the time taken to fill the gas bag (97 cm × 63 cm × 63 cm ≈ 385 L) was recorded. A gas analyzer (Beijing Zhuoan Hengrui Technology Co., Ltd., Beijing, China) was used to determine the concentration of each gas component in the bag, and CH4 emissions were calculated [26].
2.5. Analysis of Rumen Fermentation Characteristics and Serum Indexes
Ruminal fluid samples were first thawed. Then, 5 mL of the sample was mixed with 1 mL of 25% metaphosphate and centrifuged at 15,000 rpm and 4 °C for 10 min. The supernatant was used for VFA analysis via gas chromatograph (GC122 gas chromatograph, Shanghai Precision Scientific Instrument Co., Ltd., Shanghai, China) with a capillary column [27]. The chromatograph settings were as follows: carrier gas N2, split ratio 40:1, injection volume 0.6 µL, detector FID with a temperature of 230 °C, H2 flow 40 mL/min, air flow 450 mL/min, make-up gas flow 45 mL/min. The column oven temperature rose from 120 to 180 °C at 10 °C/min. Standard solutions were made by mixing specific volumes of VFAs in volumetric flasks and diluting with distilled water.
Following thawing on ice, serum samples were centrifuged at 3000× g for 10 min at 4 °C. Biochemical parameters in the supernatant were quantified using a clinical chemistry analyzer (BS-360S, Shenzhen Mindray Bio-Medical Electronics Co., Ltd., Shenzhen, China) and included the following: Triglycerides (TG), Total cholesterol (TC), Urea nitrogen (UN), Creatinine (CRE), Aspartate aminotransferase (AST), Alanine aminotransferase (ALT), Albumin (ALB), Gamma-glutamyl transferase (GGT), Total protein (TP), and Globulin (GLB). Rumen ammonia nitrogen (NH3-N) was quantified by phenol-hypochlorite colorimetry [28] using a spectrophotometer (UV-1800, Shanghai Meipuda Instrument Co., Ltd., Shanghai, China). Microbial crude protein (MCP) in rumen fluid was determined via Coomassie Brilliant Blue colorimetric assay with absorbance measured at 595 nm on the same instrument [29].
2.6. Milk Yield Recording and DHI Analysis
Milk yield was automatically recorded daily at the milking parlor, with production data from the preceding day extracted on experimental days 10, 21, and 31. Composite milk samples were submitted to the Taiyuan DHI Testing Center (Taiyuan, China) for standard milk composition analysis, including milk yield, fat-corrected milk (FCM), milk protein yield, milk fat, milk protein, lactose, and solids-not-fat (SNF). Analyses were performed using a Fossomatic™ 5000 series instrument (FOSS Analytical A/S, Hillerød, Denmark).
2.7. Analysis of Apparent Digestibility
Fresh feed, feed remnants, and feces were analyzed for crude protein (CP), dry matter (DM), ether extract (EE), and organic matter (OM) using AOAC methods [30]. Neutral detergent fiber (NDF) and acid detergent fiber (ADF) were determined via an Ankom fiber analyzer. Apparent digestibility (AD) was calculated as follows: AD (%) = [(intake − fecal content)/intake] × 100.
2.8. Microbial DNA Extraction and Quantification
Ruminal microbial DNA was extracted following the method of Edrington TS [31]. qPCR was used to quantify the relative abundance of 10 bacteria in the incubation fluid, with primers listed in Table 2. To validate amplification efficiency for each qPCR assay, known quantities of the target species template were quantified. Standard curves were subsequently constructed. All assays demonstrated high linearity between Ct values and template dilution, with strong correlation coefficients (R2). Details are in the supporting information. Real-time PCR was performed on an Applied Biosystems Step One Plus system (USA). The 20 μL reaction mixture included 10 μL of SYBR Premix TaqTM (TaKaRa Biotechnology Co., Ltd., Beijing, China), 7.0 μL of ddH2O, 0.8 μL of each primer (0.2 μmol/L), 0.4 μL of ROX Reference Dye (50×), and 1 μL of template DNA. The Ct value for each sample was recorded. The PCR program comprised initial denaturation (50 °C for 2 min, 95 °C for 2 min) and 40 cycles of amplification (95 °C for 15 s, 60 °C for 1 min) [32,33]. To validate amplification efficiency for each qPCR assay, known quantities of target species template were quantified. Standard curves were subsequently constructed. All assays demonstrated high linearity between Ct values and template dilution, with strong correlation coefficients (R2). See Supplementary Figure S1.

Table 2.
PCR primers for real-time PCR assay.
2.9. Statistical Analysis
Experimental data was analyzed using the single-factor (ANOVA) module of the SPSS 27 software, and Duncan’s method was used for multiple comparisons. A Duncan test was used to assess the differences among the four groups. Results are expressed as mean ± standard deviations. The significant difference was defined as p < 0.05, and 0.05 < p < 0.10 was considered as a tendency.
3. Results
3.1. Effect of Tannic Acid and Tea Polyphenol Dietary Supplementation on Rumen Fermentation in Dairy Cows
As indicated in Table 3, the addition of tannins significantly (p < 0.05) increased the pH of the dairy cows’ rumen, but there were no significant (p > 0.05) differences among the T, T+TP, and TP groups. Compared with the control group (C), the groups supplemented with tannins had significantly (p < 0.05) lower levels of NH3-N and MCP, but no significant (p > 0.05) differences were observed among the three groups. The propionate content in the T, T+TP, and TP groups was significantly (p < 0.05) higher than that in the C group, with the T group showing a significantly (p < 0.05) higher propionate content than the TP group, and no significant (p > 0.05) difference between the T group and the T+TP group. The acetate to propionate ratio in the T, T+TP, and TP groups was significantly (p < 0.05) lower than that in the C group, with no significant (p > 0.05) differences among the three groups. The methane production in the groups treated with tannins was significantly (p < 0.05) lower than that in the C group, but there were no significant (p > 0.05) differences among the three treatment groups.

Table 3.
Effect of diet added with tannic acid and tea polyphenols on rumen fermentation in dairy cows.
3.2. Effect of Tannic Acid and Tea Polyphenol Dietary Supplementation on Nutrient Apparent Digestibility of Dairy Cows
Table 4 indicates that there is a trend towards increased apparent digestibility of dry matter (DM) and organic matter (OM) in the T, T+TP, and TP groups compared to the C group (0.1 > p > 0.05). The apparent digestibility of crude protein (CP) in the T group was significantly (p < 0.05) higher than in the C group, while the apparent digestibility of CP in the T+TP and TP groups was intermediate between the T and C groups, with no significant (p > 0.05) differences between them. There were no significant (p > 0.05) changes in the apparent digestibility of ether extract (EE), acid detergent fiber (ADF), and neutral detergent fiber (NDF) among the four groups. Notably, EE and NDF trended downward across supplemented diets versus control, except ADF.

Table 4.
Effect of diet added with tannic acid and tea polyphenols on nutrient apparent digestibility of dairy cows (%).
3.3. Effect of Tannic Acid and Tea Polyphenol Dietary Supplementation on Blood Indicators in Dairy Cows
Table 5 shows that the plasma urea nitrogen content in the T, T+TP, and TP groups was significantly (p < 0.05) lower than in the C group, with no significant (p > 0.05) differences among the three groups. Compared to the control group, there was a trend towards increased serum albumin and total protein content in the groups treated with tannins in the dairy cow diet, while there were no significant (p > 0.05) changes in triglycerides, total cholesterol, creatinine, aspartate aminotransferase, alanine aminotransferase, or globulin. Compared to the control group, there was a trend towards a decrease in glutamylphthalide aminotransferase in the TP groups.

Table 5.
Effect of diet with added tannic acid and tea polyphenols on blood indicators in dairy cows.
3.4. Effect of Tannic Acid and Tea Polyphenol Dietary Supplementation on Ruminal Microbial Structure in Dairy Cows
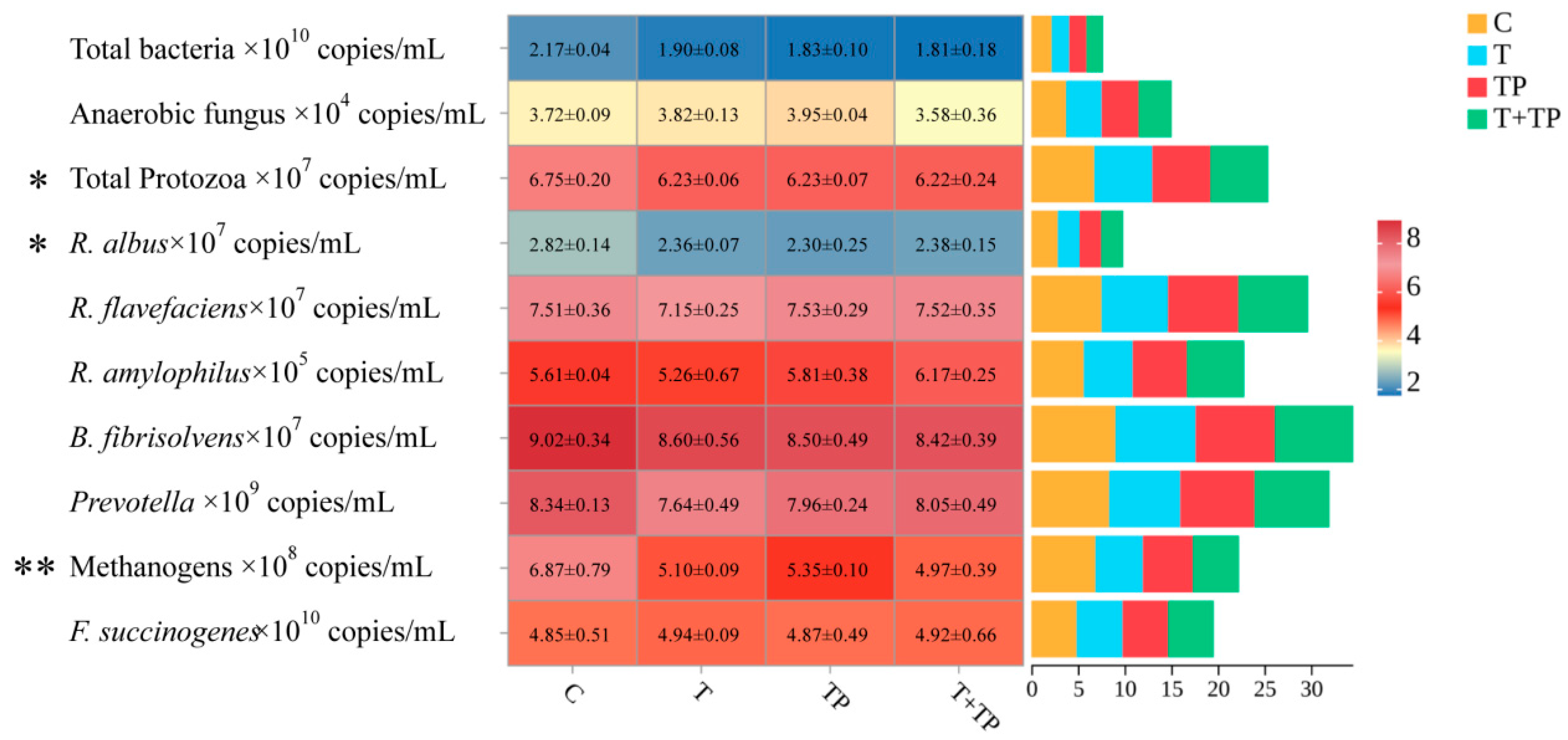
Supplementation with tannic acid (T), tea polyphenols (TP), or their combination (T+TP) significantly modulated rumen microbial populations (Figure 1). The relative proportions of total bacteria tended to decrease in all treatment groups compared to control (p = 0.059), representing reductions of 12.4–16.6%. The relative proportions of methanogens were dramatically suppressed across treatments (p = 0.003), with reductions of 22.1–27.7%. Similarly, the relative proportions of Ruminococcus albus significantly declined in supplemented groups (p = 0.039), showing decreases of 16.3–18.4%. The relative proportions of protozoa were significantly reduced (p = 0.024), while the relative proportions of anaerobic fungi showed no significant changes (p = 0.281). Ruminococcus flavefaciens exhibited numerical reduction with T alone but no statistical significance (p = 0.426). Key fiber-degrading bacteria including Fibrobacter succinogenes (p = 0.904), Butyrivibrio fibrisolvens (p = 0.522), and Prevotella (p = 0.308) maintained stable populations across all treatments. Ruminobacter amylophilus showed a trend toward increase in T+TP but no significant group differences (p = 0.103). This specific microbial shift pattern—characterized by significant suppression of methanogens and R. albus without compromising major fibrolytic bacteria—supports the maintained fiber digestibility and methane reduction observed in the trial.
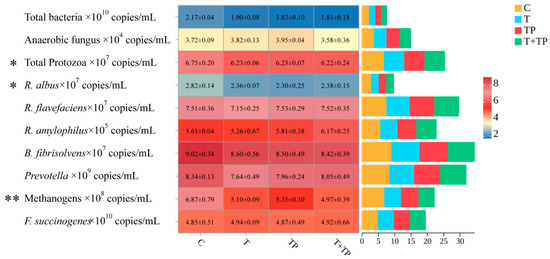
Figure 1.
Effects of tannic acid and tea polyphenols on rumen microbiota structure in dairy cows. Note: C, without tannic acid and tea polyphenols; T, 0.4% tannic acid added; TP, 0.4% tea polyphenols added; T+TP, 0.2% tannic acid and 0.2% tea polyphenols added. * p < 0.05, ** p < 0.01.
3.5. Effects of Tannic Acid and Tea Polyphenol Dietary Supplementation on Milk Yield and Components
Supplementation (Table 6) significantly increased milk protein percentage (p = 0.001) and decreased milk urea nitrogen (MUN, p = 0.022). Milk protein yield showed a tendency toward increase (p = 0.074), as did 4% fat-corrected milk yield (p = 0.089). No significant effects were observed on milk yield (p = 0.157), milk fat percentage (p = 0.464), lactose percentage (p = 0.673), non-fat milk solids (p = 0.437), total solids (p = 0.638), or somatic cell count (p = 0.153).

Table 6.
Effects of diet with added tannic acid and tea polyphenols on milk yield and components.
4. Discussion
The observed elevation in ruminal pH within this study can be attributed to several factors influencing the ruminal ecosystem, including salivary secretion, organic acid production, absorption/excretion rates, and dietary composition. Specifically, the addition of tannins likely contributed by binding dietary proteins, forming stable complexes resistant to microbial degradation within the typical ruminal pH range (5.5–7.5), and through their inherent antibacterial properties, thereby modulating rumen microbial fermentation [34]. This modulation is further evidenced by the significant changes in volatile fatty acid (VFA) profiles. The supplementation of 0.4% DM tannic acid, a tannic acid/tea polyphenol mixture (50:50), or tea polyphenols alone consistently increased propionic acid concentration and decreased the acetate-to-propionate ratio. While dietary polysaccharides are primarily fermented by rumen microbes to acetate, propionate, and butyrate (alongside CO2 and CH4), the impact of tannins on VFA molar ratios is often inconsistent across studies. For instance, Benchaar et al. [35] reported no effect on total VFA concentration or individual molar proportions with 0.64% DM quebracho tannin extract, whereas Dschaak et al. [36] observed a decrease in total VFA at a higher supplementation level (3% DM) of the same extract. These divergent findings regarding tannin effects on VFA profiles—encompassing total concentration and individual acid proportions—are likely influenced by key variables such as tannin dose, source (e.g., condensed vs. hydrolysable), interactions with dietary constituents, and the adaptation period of the rumen microbiota [37]. Supporting the dose-dependency, Carulla et al. [21] found no change in total VFA with 0.6% condensed tannins in sheep, but noted a shift towards propionate at the expense of acetate. In the present experiment, the significant alterations in propionate and the acetate:propionate ratio induced by all tannin-containing additives at 0.4% DM highlight their efficacy at this level. Notably, the combined tannic acid and tea polyphenol supplementation yielded superior results compared to tea polyphenols alone.
The synthesis of ruminal microbial protein (MCP) is intricately influenced by tannins, their supplementation level, and dietary composition, reflecting a complex interplay between tannins and the rumen microbiota. While tannins can inhibit microbial growth and enzyme activity, they may also promote MCP synthesis, with the net effect dependent on the balance of these opposing actions [38]. In this experiment, MCP content was significantly lower in tannin-supplemented groups compared to the control, with the combined tannic acid and tea polyphenol group exhibiting the lowest total bacterial abundance. Concurrently, tannins at 0.4% DM significantly reduced the abundance of methanogenic archaea and protozoa. This reduction in methanogens aligns with their ability to bind proteins or microbial enzymes and their symbiotic relationship with protozoa [3]. The decrease in protozoa is notable given their significant proteolytic role in degrading both rumen-degradable and undegradable protein (RDP and RUP) [17], particularly as crude protein content significantly increased in the experimental groups. The discrepancy between these results (significant reduction at 0.4% DM) and those of Yang K et al. (no effect at 0.65% DM, significant reduction at 2.6% DM) [39] suggests that the impact on methanogens and protozoa is highly dependent on supplementation level, potentially compounded by differences in animal species (dairy cows vs. Simmental cattle) or dietary structure.
Tannins can inhibit fibrolytic activity by binding to microbial surface enzymes or directly complexing with cellulose, potentially suppressing the growth of fiber-degrading bacteria [40]. The primary fibrolytic consortium in rumen fluid comprises Ruminococcus flavefaciens, Fibrobacter succinogenes, and Ruminococcus albus, constituting approximately 2.6%, 33%, and 46% of this population, respectively [41,42]. In this study, dietary supplementation with tannins at 0.4% DM did not significantly alter the relative abundance of F. succinogenes or R. flavefaciens, but significantly reduced R. albus. This contrasts with findings by Li Dabiao et al. (reduction in all three bacteria at 2% and 6% tannins in sheep/goats) [43] and Jayanegara et al. (reduction in F. succinogenes and R. flavefaciens with quebracho tannins in vitro) [17], highlighting that the inhibitory effects on specific fibrolytic bacteria vary considerably with tannin type, dose, and host species. Despite significant methanogen inhibition (22.1–27.7%), total VFA concentration remained unchanged, reflecting metabolic compensatory mechanisms within the ruminal ecosystem. Methane suppression redirected metabolic hydrogen toward alternative electron sinks, primarily favoring propionate synthesis via the succinate pathway (propionate proportion increased 64.2%) [44]. The marked decrease in the acetate-to-propionate ratio (reduced 31.4%) further corroborated this shift. Critically, core fibrolytic populations—Fibrobacter succinogenes, Butyrivibrio fibrisolvens, and Prevotella—maintained stability, sustaining carbohydrate fermentation capacity and preserving total VFA output during the metabolic transition from methanogenesis to propionogenesis [45]. Crucially, despite the reduction in R. albus, the digestibility of NDF and ADF remained unaffected at the 0.4% DM supplementation level. This indicates that the decrease in R. albus abundance did not compromise fiber digestion in dairy cows under these conditions, likely due to functional redundancy within the fibrolytic community and the strong degradative capacity of F. succinogenes for structural carbohydrates. Given the high diversity of cellulolytic microorganisms, our analysis focused solely on dominant populations. We therefore hypothesize that unobserved microbial communities may have contributed critically through functional compensation [46].
Historically classified as antinutritional factors, tannins can impair intake and nutrient digestibility, primarily at supplementation levels exceeding 3–4% DM [47]. However, consistent with studies using low tannin doses (0.4–1.0% DM) in calves [48], the present study observed no adverse effects on DM, OM, CP, or NDF intake or digestibility at 0.4% DM supplementation. Notably, tannic acid alone significantly increased crude protein digestibility (6.6%), while tea polyphenols alone and their combination increased it by 5% and 3.7%, respectively. This contrasts with the typical paradigm where tannin–protein binding might reduce CP digestibility by limiting ruminal degradation, suggesting that low-dose supplementation or specific tannin sources may enhance nitrogen utilization efficiency [49]. Further supporting improved nitrogen metabolism, all tannin treatments significantly reduced ruminal ammonia-N (by 23% individually and 34.5% synergistically) and milk urea nitrogen (MUN) (by 18.75%, 16.4%, and 21.1%, respectively). This reduction in ammonia-N and MUN aligns with the primary mechanism of tannins forming reversible complexes with dietary protein, thereby reducing ruminal proteolysis and ammonia production [50], and increasing the flow of digestible protein to the small intestine. While studies report variable MUN responses (e.g., reduction at 1.8% DM CTE vs. no effect at 0.45–0.9% DM [51]), the significant decrease observed here at 0.4% DM, despite similar dietary CP and RDP:RUP ratios across treatments, indicates a direct positive impact of these tannins on nitrogen utilization efficiency independent of diet formulation.
The observed reduction in MUN has significant practical implications. As MUN exhibits a positive linear relationship with urinary nitrogen excretion [20], its decrease indicates reduced environmental nitrogen loss. Furthermore, elevated MUN typically signals dietary CP overfeeding [52]; its reduction here, coupled with significant increases in milk protein concentration (within the optimal 3.0–3.2% range [53]) for individual tannin and tea polyphenol supplements and a significant increase for their combination, demonstrates improved nitrogen partitioning towards milk synthesis rather than waste. This enhancement in milk protein output is consistent with the increased flow of amino acids (AA) to the small intestine resulting from tannin-mediated protection of dietary protein from ruminal degradation [13]. Consequently, the supplementation, particularly the synergistic combination of tannic acid and tea polyphenols at 0.4% DM, effectively reduces urinary nitrogen excretion while simultaneously enhancing the protein quality of milk.
Extensive in vitro [49] and in vivo [54] studies demonstrate that dietary tannins suppress ruminal methane (CH4) production. Consistent with this, all tannin treatments in the present study significantly reduced CH4 emissions without inter-group variation. This mitigation primarily stems from tannins directly inhibiting methanogenic archaea and indirectly limiting protozoal populations—key symbiotic partners of methanogens [38]—thereby disrupting methanogenesis. The observed reduction aligns with the well-established negative correlation between plant condensed tannin (CT) content and in situ degradability/in vitro fermentation parameters, including CH4 output [13]. Furthermore, the metabolic pathways of key rumen metabolites contribute to this effect: while acetogenesis releases H2 (a substrate for methanogenesis), propionate synthesis consumes H+/H2, creating an antimethanogenic sink [55]. Consequently, the decreased acetate-to-propionate ratio induced by tannins (as reported earlier) directly contributes to CH4 suppression. This mechanistic framework is corroborated by Tavendale et al., who proposed tannins reduce methanogenesis via archaeal inhibition or H2 limitation [56].
Although all tannin treatments significantly reduced the abundance of total methanogenic archaea—the primary CH4 producers [57]—the magnitude of archaeal suppression did not fully account for the observed CH4 reduction. This discrepancy implies that diminished H2 availability, mediated through alternative microbial pathways, plays a critical role. Rumen microbial communities dynamically regulate hydrogen partial pressure (a key determinant of methanogenesis), with H2 generated during fiber fermentation and primarily consumed by methanogens. Notably, genomic and transcriptomic analyses reveal that ~67% of rumen microbial genomes encode and express hydrogenotrophic enzymes [58], indicating substantial functional redundancy for H2 utilization beyond methanogenesis. Thermodynamic and kinetic models confirm H2 pressure dictates CH4 yield and fermentation balance [59]. In this context, tannin-induced reductions in Ruminococcus albus (across all treatments) and Ruminococcus flavefaciens (with tannic acid alone)—major H2-producing, cellulolytic bacteria [40]—likely curtailed H2 supply to methanogens, synergizing with direct archaeal inhibition to amplify CH4 suppression. Concurrently, key fiber-degrading bacteria including Fibrobacter succinogenes, Butyrivibrio fibrisolvens, and Prevotella maintained stable populations across all treatments. Ruminobacter amylophilus showed a trend toward increase in T+TP with no statistically significant difference. These findings confirm the observed methane reduction without compromising fiber digestibility. Moreover, T significantly increased the propionate proportion, whereas TP exerted a milder suppression on Ruminococcus flavefaciens. Existing studies [60,61] corroborate that plant polyphenols mitigate methane emissions by selectively modulating microbial communities (e.g., protecting fibrolytic bacteria while inhibiting methanogens). This implies that distinct phenolic compounds achieve analogous methane reduction through unique microbial pathways.
5. Conclusions
This study establishes tea polyphenols (TP) as a natural modulator of rumen function, demonstrating that low-dose supplementation (0.4% DM) alone significantly enhances nitrogen utilization efficiency, reducing ammonia-N and methane emissions while increasing milk protein yield. Critically, TP exhibits comparable efficacy to tannic acid (T) in key metrics including methane suppression (13.68% for T; 10.09% for TP), crude protein digestibility enhancement (7.02% for T; 5.37% for TP) and milk protein enhancement (5.74% for T; 4.10% for TP), while offering inherent advantages as a plant-derived, food-safe compound. The synergistic combination of T and TP (50:50) shows relevant improvements: it maximizes nitrogen conservation (34.5% ruminal ammonia-N reduction; 21.1% MUN decrease), outperforming either polyphenol alone and amplifies milk protein synthesis, and no detrimental effects were observed on the measured rumen fermentation characteristics. By integrating TP metabolic precision with TA targeted protein-binding capacity, this approach unlocks multifunctional gains unattainable by single additives, suggests that plant–polyphenol synergism represents a promising strategy for reducing enteric methane emissions in dairy cattle, contributing to climate-smart livestock production.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/microorganisms13081848/s1, Figure S1: The standard curves obtained by plotting the logarithm of DNA concentration for Anaerobic (a), B. fibrisolvens (b), Prevotella (c), Total bacteria (d), R. amylophilus (e), Methanogens (f), F. succinogenes (g), R. flavefaciens (h), R. albus (i) and Protozoa (j) versus threshold cycle (Ct) for population quantification by using real time PCR.
Author Contributions
R.Z.: Investigation, Writing—original draft, Visualization. J.S.: Data curation, Supervision. Y.L.: Supervision, Project administration. H.Y.: Visualization. S.Z.: Investigation. W.H.: Methodology, Visualization. L.C.: Resources, Methodology. Q.L.: Conceptualization. C.W.: Resources. G.G.: Funding acquisition, Supervision. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the earmarked fund for Modern Agro-industry Technology Research System (2025CYJSTX13-08) and National Natural Science Foundation of China (32001405).
Institutional Review Board Statement
The animal study protocol was approved by the Institutional Animal Care and Use Committee of Shanxi Agricultural University (protocol code: SXAU-EAW-2022C. RD.010025174 ; protocol date: 25 November 2022).
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Acknowledgments
Thanks to all participants for their advice and support of this study.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- EPA. Inventory of U.S. Greenhouse Gas Emissions and Sinks: 1990–2019 (No. EPA-430-R-21-005); United States Environmental Protection Agency: Washington, DC, USA, 2021.
- Rotz, A.; Stout, R.; Leytem, A.; Feyereisen, G.; Waldrip, H.; Thoma, G.; Holly, M.; Bjorneberg, D.; Baker, J.; Vadas, P. Environmental assessment of United States dairy farms. J. Clean. Prod. 2021, 315, 128153. [Google Scholar] [CrossRef]
- Pereira, A.M.; de Lurdes Nunes Enes Dapkevicius, M.M.; Borba, A.E.S. Alternative pathways for hydrogen sink originated from the ruminal fermentation of carbohydrates: Which microorganisms are involved in lowering methane emission? Anim. Microbiome 2022, 4, 5. [Google Scholar] [CrossRef]
- Arndt, C.; Hristov, A.N.; Price, W.J.; McClelland, S.C.; Pelaez, A.M.; Cueva, S.F.; Oh, J.; Dijkstra, J. Full adoption of the most effective strategies to mitigate methane emissions by ruminants can help meet the 1.5 °C target by 2030 but not 2050. Proc. Natl. Acad. Sci. USA 2022, 119, e2111294119. [Google Scholar] [CrossRef] [PubMed]
- Hristov, A.N.; Oh, J.; Giallongo, F.; Frederick, T.W.; Harper, M.T.; Weeks, H.L.; Branco, A.F.; Moate, P.J. An inhibitor persistently decreased enteric methane emission from dairy cows with no negative effect on milk production. Proc. Natl. Acad. Sci. USA 2015, 112, 10663–10668. [Google Scholar] [CrossRef] [PubMed]
- Mizrahi, I.; Wallace, R.J.; Moraïs, S. The rumen microbiome: Balancing food security and environmental impacts. Nat. Rev. Microbiol. 2021, 19, 553–566. [Google Scholar] [CrossRef] [PubMed]
- Mizrahi, I.; Jami, E. Review: The compositional variation of the rumen microbiome and its effect on host performance and methane emission. Animal 2018, 12, s220–s232. [Google Scholar] [CrossRef]
- Bagheri, V.M.; Klevenhusen, F.; Zebeli, Q.; Petri, R. Scrophularia striata Extract Supports Rumen Fermentation and Improves Microbial Diversity in vitro Compared to Monensin. Front. Microbiol. 2018, 9, 2164. [Google Scholar] [CrossRef]
- Eom, J.S.; Lee, S.J.; Lee, Y.; Kim, H.S.; Choi, Y.Y.; Kim, H.S.; Kim, D.H.; Lee, S.S. Effects of supplementation levels of Allium fistulosum L. extract on in vitro ruminal fermentation characteristics and methane emission. PeerJ 2020, 8, e9651. [Google Scholar] [CrossRef]
- Jayanegara, A.; Yogianto, Y.; Wina, E.; Sudarman, A.; Kondo, M.; Obitsu, T.; Kreuzer, M. Combination Effects of Plant Extracts Rich in Tannins and Saponins as Feed Additives for Mitigating in Vitro Ruminal Methane and Ammonia Formation. Animals 2020, 10, 1531. [Google Scholar] [CrossRef]
- Ramos, M.E.; Rossi, G.; Cattin, M.; Jones, E.; Braganca, R.; Newbold, C.J. The effect of an isoflavonid-rich liquorice extract on fermentation, methanogenesis and the microbiome in the rumen simulation technique. FEMS Microbiol. Ecol. 2018, 94, fiy009. [Google Scholar] [CrossRef]
- Ruchita, K.; Tassilo, B.; Ilma, T.; Ali-Reza, B. Effect of a garlic and citrus extract supplement on performance, rumen fermentation, methane production, and rumen microbiome of dairy cows. J. Dairy Sci. 2023, 106, 608–4621. [Google Scholar] [CrossRef]
- Aboagye, I.A.; Beauchemin, K.A. Potential of Molecular Weight and Structure of Tannins to Reduce Methane Emissions from Ruminants: A Review. Animals 2019, 9, 856. [Google Scholar] [CrossRef] [PubMed]
- Zhou, R.; Wu, J.; Xia, L.; Liu, L.S.; Casper, D.P.; Wang, C.; Zhang, L.; Wei, S. Effects of oregano essential oil on in vitro ruminal fermentation, methane production, and ruminal microbial community. J. Dairy Sci. 2020, 103, 2303–2314. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Bao, X.Y.; Guo, G.; Huo, W.J.; Xu, Q.; Wang, C. Effects of Hydrolysable Tannin with or without Condensed Tannin on Alfalfa Silage Fermentation Characteristics and In Vitro Ruminal Methane Production, Fermentation Patterns, and Microbiota. Animals 2021, 11, 1967. [Google Scholar] [CrossRef] [PubMed]
- Battelli, M.; Colombini, S.; Parma, P.; Galassi, G.; Crovetto, G.M.; Spanghero, M.; Pravettoni, D.; Zanzani, S.A.; Manfredi, M.T.; Rapetti, L. In vitro effects of different levels of quebracho and chestnut tannins on rumen methane production, fermentation parameters, and microbiota. Front. Vet. Sci. 2023, 10, 1178288. [Google Scholar] [CrossRef]
- Jayanegara, A.; Goel, G.; Makkar, H.; Becker, K. Divergence between purified hydrolysable and condensed tannin effects on methane emission, rumen fermentation and microbial population in vitro. Anim. Feed Sci. Technol. 2015, 209, 60–68. [Google Scholar] [CrossRef]
- Bhatta, R.; Uyeno, Y.; Tajima, K.; Takenaka, A.; Yabumoto, Y.; Nonaka, I.; Enishi, O.; Kurihara, M. Difference in the nature of tannins on in vitro ruminal methane and volatile fatty acid production and on methanogenic archaea and protozoal populations. J. Dairy Sci. 2009, 92, 5512–5522. [Google Scholar] [CrossRef]
- Cipriano-Salazar, M.; Rojas-Hernández, S.; Olivares-Pérez, J.; Jiménez-Guillén, R.; Cruz-Lagunas, B.; Camacho-Díaz, L.M.; Ugbogu, A.E. Antibacterial activities of tannic acid against isolated ruminal bacteria from sheep. Microb. Pathog. 2018, 117, 255–258. [Google Scholar] [CrossRef]
- Aguerre, M.J.; Capozzolo, M.C.; Lencioni, P.; Cabral, C.; Wattiaux, M.A. Effect of quebracho-chestnut tannin extracts at 2 dietary crude protein levels on performance, rumen fermentation, and nitrogen partitioning in dairy cows. J. Dairy Sci. 2016, 99, 4476–4486. [Google Scholar] [CrossRef]
- Carulla, J.E.; Kreuzer, M.; Andrea, M.; Hess, D.H. Supplementation of Acacia mearnsii tannins decreases methanogenesis and urinary nitrogen in forage-fed sheep. Aust. J. Agric. Res. 2005, 56, 961–970. [Google Scholar] [CrossRef]
- Lan, W.; Yang, C.L. Ruminal methane production: Associated microorganisms and the potential of applying hydrogen-utilizing bacteria for mitigation. Sci. Total Environ. 2019, 654, 1270–1283. [Google Scholar] [CrossRef] [PubMed]
- Grazziotin, R.C.B.; Halfen, J.; Rosa, F.; Schmitt, E.; Anderson, J.L.; Ballard, V.; Osorio, J.S. Altered rumen fermentation patterns in lactating dairy cows supplemented with phytochemicals improve milk production and efficiency. J. Dairy Sci. 2020, 103, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Luis, O.T.; Danny, G.F.; Mozart, A.F.; Luigi, F.L.C. Models of protein and amino acid requirements for cattle. Rev. Bras. Zootec. 2015, 44, 109–132. [Google Scholar] [CrossRef]
- Bekele, W.; Guinguina, A.; Zegeye, A.; Simachew, A.; Ramin, M. Contemporary Methods of Measuring and Estimating Methane Emission from Ruminants. Methane 2022, 1, 82–95. [Google Scholar] [CrossRef]
- Wilkinson, J.; Bors, C.; Burgis, F.; Lorke, A.; Bodmer, P. Measuring CO2 and CH4 with a portable gas analyzer: Closed-loop operation, optimization and assessment. PLoS ONE 2018, 13, e0193973. [Google Scholar] [CrossRef]
- Erwin, E.S.; Marco, G.J.; Emery, E.M. Volatile fatty acid analyses of blood and rumen fluid by gas chromatography. J. Dairy Sci. 1961, 44, 1768–1771. [Google Scholar] [CrossRef]
- Weatherburn, M.W. Phenol-hypochlorite reaction for determination of ammonia. Anal. Chem. 1967, 39, 971–974. [Google Scholar] [CrossRef]
- Guo, G.; Shen, C.; Liu, Q.; Zhang, S.L.; Wang, C.; Chen, L.; Xu, Q.F.; Wang, Y.X.; Huo, W.J. Fermentation quality and in vitro digestibility of first and second cut alfalfa (Medicago sativa L.) silages harvested at three stages of maturity. Anim. Feed Sci. Technol. 2019, 257, 114274. [Google Scholar] [CrossRef]
- AOAC. Association of Official Analytical Chemists Official Methods of Analysis; AOAC: Washington, DC, USA, 2019. [Google Scholar]
- Lévesque, B.; Beno, T.; Ayotte, P.; Tardif, R.; Ferron, L.; Gingras, S.; Schlouch, E.; Dewailly, E. Cancer risk associated with household exposure to chloroform. J. Toxicol. Environ. Health Part A 2002, 65, 489–502. [Google Scholar] [CrossRef]
- Wang, R.F.; Cao, W.W.; Cerniglia, C.E. PCR detection of Ruminococcus spp. in human and animal faecal samples. Mol. Cell Probes 1997, 11, 259–265. [Google Scholar] [CrossRef]
- Denman, S.E.; McSweeney, C.S. Developmentofa real-time PCR assay formonitoring anaerobic fungal and cellulolytic bacterial populations within the rumen. FEMS Microbiol. Ecol. 2006, 58, 572–582. [Google Scholar] [CrossRef]
- Mattei, V.; Murugesan, S.; Al Hashmi, M.; Mathew, R.; James, N.; Singh, P.; Kumar, M.; Lakshmanan, A.P.; Terranegra, A.; Al Khodor, S.; et al. Evaluation of Methods for the Extraction of Microbial DNA From Vaginal Swabs Used for Microbiome Studies. Front. Cell. Infect. Microbiol. 2019, 9, 197. [Google Scholar] [CrossRef]
- Benchaar, C.; McAllister, T.A.; Chouinard, P.Y. Digestion, ruminal fermentation, ciliate protozoal populations, and milk production from dairy cows fed cinnamaldehyde, quebracho condensed tannin, or Yucca schidigera saponin extracts. J. Dairy Sci. 2008, 91, 4765–4777. [Google Scholar] [CrossRef]
- Dschaak, C.M.; Williams, C.M.; Holt, M.S.; Eun, J.S.; Young, A.J.; Min, B.R. Effects of supplementing condensed tannin extract on intake, digestion, ruminal fermentation, and milk production of lactating dairy cows. J. Dairy Sci. 2011, 94, 2508–2519. [Google Scholar] [CrossRef] [PubMed]
- Menci, R.; Coppa, M.; Torrent, A.; Natalello, A.; Valenti, B.; Luciano, G.; Priolo, A.; Niderkorn, V. Effects of two tannin extracts at different doses in interaction with a green or dry forage substrate on in vitro rumen fermentation and biohydrogenation. Anim. Feed. Sci. Technol. 2021, 278, 114977. [Google Scholar] [CrossRef]
- Tian, G.Y.; Zhang, X.Z.; Hao, X.Y.; Zhang, J. Effects of Curcumin on Growth Performance, Ruminal Fermentation, Rumen Microbial Protein Synthesis, and Serum Antioxidant Capacity in Housed Growing Lambs. Animals 2023, 13, 1439. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Wei, C.; Zhao, G.Y.; Xu, Z.W.; Lin, S.X. Effects of dietary supplementing tannic acid in the ration of beef cattle on rumen fermentation, methane emission, microbial flora and nutrient digestibility. J. Anim. Physiol. Anim. Nutr. 2017, 101, 302–310. [Google Scholar] [CrossRef] [PubMed]
- Belanche, A.; Doreau, M.; Edwards, J.E.; Moorby, J.M.; Pinloche, E.; Newbold, C.J. Shifts in the rumen microbiota due to the type of carbohydrate and level of protein ingested by dairy cattle are associated with changes in rumen fermentation. J. Nutr. 2012, 142, 1684–1692. [Google Scholar] [CrossRef]
- Mcsweeney, C.S.; Palmer, B.; Mcneill, D.M. Microbial interactions with tannins: Nutritional consequences for ruminants. Anim. Feed Sci. Technol. 2001, 91, 83–93. [Google Scholar] [CrossRef]
- Koike, S.; Kobayashi, Y. Fibrolytic Rumen Bacteria: Their Ecology and Functions. Asian Australas. J. Anim. Sci. 2009, 40, 1141–1147. [Google Scholar] [CrossRef]
- Li, D.B.; Yu, Y.Q.; Wang, W.Y.; Zhang, M.; Li, H.L.; Xing, Y. Effects of Tannin and Polyetylene Glycol on Ruminal Microorganism Quantity and Nutrient Apparent Digestibility of Sheep and Goats. Chin. J. Anim. Nutr. 2015, 27, 3155–3162. Available online: https://api.semanticscholar.org/CorpusID:87057038 (accessed on 20 May 2025).
- Wang, L.J.; Zhang, J.J.; Li, Y. Effects of High Forage/Concentrate Diet on Volatile Fatty Acid Production and the Microorganisms Involved in VFA Production in Cow Rumen. Animals 2020, 10, 223. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Hassan, F.; Li, M.; Xie, H.; Peng, L.; Tang, Z.; Yang, C. Effect of Sodium Nitrate and Cysteamine on In Vitro Ruminal Fermentation, Amino Acid Metabolism and Microbiota in Buffalo. Microorganisms 2022, 10, 2038. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.M.A.; Martins, C.M.; Cavalcante, F.G.; Ramos, K.A.; Martins, S.C.S. Cross-feeding among soil bacterial populations: Selection and characterization of potential bio-inoculants. J. Agric. Sci. 2019, 11, 23. [Google Scholar] [CrossRef][Green Version]
- Ku-Vera, J.C.; Ocampo, J.; Valencia-Salazar, S.S.; Montoya-Flores, D.; Solorio-Sánchez, F.J. Role of Secondary Plant Metabolites on Enteric Methane Mitigation in Ruminants. Front. Vet. Sci. 2020, 7, 584. [Google Scholar] [CrossRef]
- Mezzomo, R.; Paulino, P.V.R.; Detmann, E.; Valadares Filho, S.C.; Paulino, M.F.; Monnerat, J.P.I.S.; Duarte, M.S.; Silva, L.H.P.; Moura, L.S. Influence of condensed tannin on intake, digestibility, and efficiency of protein utilization in beef steers fed high concentrate diet. Livest. Sci. 2011, 141, 1–11. [Google Scholar] [CrossRef]
- Ebert, P.J.; Bailey, E.A.; Shreck, A.L.; Jennings, J.S.; Cole, N.A. Effect of condensed tannin extract supplementation on growth performance, nitrogen balance, gas emissions, and energetic losses of beef steers. J. Anim. Sci. 2017, 95, 1345–1355. [Google Scholar] [CrossRef]
- Jayanegara, A.; Yaman, A.; Khotijah, L. Reduction of proteolysis of high protein silage from Moringa and Indigofera leaves by addition of tannin extract. Vet. World 2019, 12, 211–217. [Google Scholar] [CrossRef]
- Rodríguez, E.; María, E.; Guevara-Oquendo, V.H.; Sun, B. Recent progress in structural and nutritional characterization of faba legume and use as an environment probe with vibrational spectroscopy sourced by globar and synchrotron. Appl. Spectrosc. Rev. 2019, 55, 1–19. [Google Scholar] [CrossRef]
- Katongole, C.B.; Yan, T. Effect of Varying Dietary Crude Protein Level on Feed Intake, Nutrient Digestibility, Milk Production, and Nitrogen Use Efficiency by Lactating Holstein-Friesian Cows. Animals 2020, 10, 2439. [Google Scholar] [CrossRef]
- Broderick, G.A.; Clayton, M.K. A statistical evaluation of animal and nutritional factors influencing concentrations of milk urea nitrogen. J. Dairy Sci. 1997, 80, 2964–2971. [Google Scholar] [CrossRef]
- Jayanegara, A.; Leiber, F.; Kreuzer, M. Meta-analysis of the relationship between dietary tannin level and methane formation in ruminants from in vivo and in vitro experiments. J. Anim. Physiol. Anim. Nutr. 2011, 96, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Piñeiro-Vázquez, A.T.; Jiménez-Ferrer, G.; Alayon-Gamboa, J.A.; Chay-Canul, A.J.; Ayala-Burgos, A.J.; Aguilar-Pérez, C.F.; Ku-Vera, J.C. Effects of quebracho tannin extract on intake, digestibility, rumen fermentation, and methane production in crossbred heifers fed low-quality tropical grass. Trop. Anim. Health Prod. 2017, 50, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Patra, A.K.; Saxena, J. Exploitation of dietary tannins to improve rumen metabolism and ruminant nutrition. J. Sci. Food Agric. 2011, 91, 24–37. [Google Scholar] [CrossRef] [PubMed]
- Hailemariam, S.; Zhao, S.; He, Y.; Wang, J. Urea transport and hydrolysis in the rumen: A review. Anim. Nutr. 2021, 7, 989–996. [Google Scholar] [CrossRef]
- Zhang, P.; Roque, B.; Romero, P. Red seaweed supplementation suppresses methanogenesis in the rumen, revealing potentially advantageous traits among hydrogenotrophic bacteria. BioRxiv 2024. [Google Scholar] [CrossRef]
- Stams, A.; Plugge, C. Electron transfer in syntrophic communities of anaerobic bacteria and archaea. Nat. Rev. Microbiol. 2009, 7, 568–577. [Google Scholar] [CrossRef]
- Liu, R.; Shen, Y.; Ma, H.; Li, Y.; Lambo, M.T.; Dai, B.; Shen, W.; Qu, Y.; Zhang, Y. Silibinin reduces in vitro methane production by regulating the rumen microbiome and metabolites. Front. Microbiol. 2023, 14, 1225643. [Google Scholar] [CrossRef]
- Prathap, P.; Chauhan, S.S.; Flavel, M.; Mitchell, S.; Cottrell, J.J.; Leury, B.J.; Dunshea, F.R. Effects of Sugarcane-Derived Polyphenol Supplementation on Methane Production and Rumen Microbial Diversity of Second-Cross Lambs. Animals 2024, 14, 905. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).