Polyhydroxyalkanoate (PHA) Biopolymer Synthesis by Marine Bacteria of the Malaysian Coral Triangle Region and Mining for PHA Synthase Genes
Abstract
1. Introduction
2. Materials and Methods
2.1. Collection of Bacterial Samples
2.2. PHA Production and phaC Gene Identification in Sediment-Associated Bacterial Isolates with a Cultivation Approach
2.2.1. Pre-Culture of Environmental Bacteria
2.2.2. Strain Isolation
2.2.3. Screening for PHA Presence
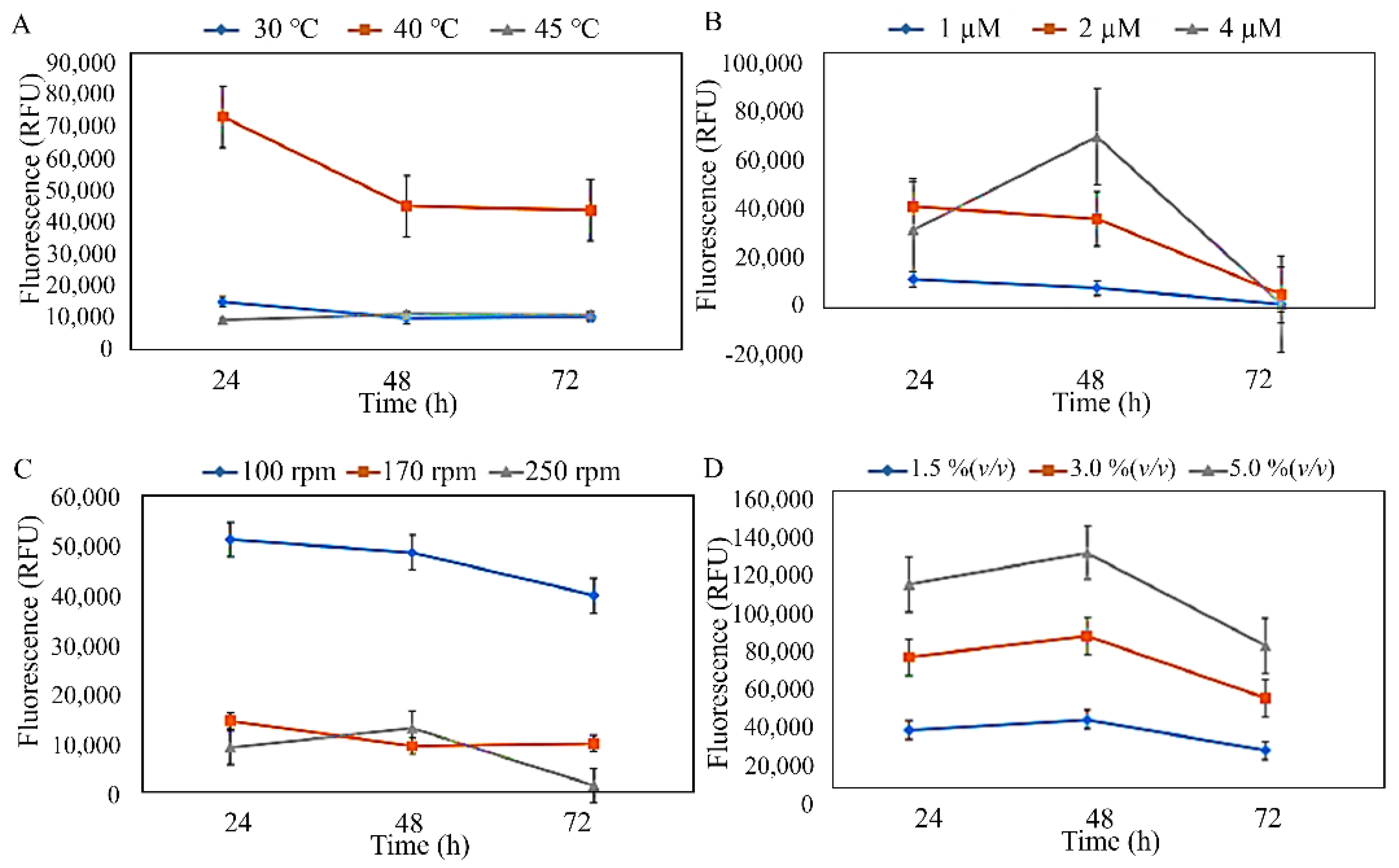
2.2.4. PHA Production Using Shake-Flask Fermentation
2.3. Gas Chromatography Analysis
2.4. Identification of PHA-Producing Strains from Sediment and Their PHA Synthases
2.5. phaC Gene Identification and Screening of phaC Taxa in Sponge-Associated Bacterial Metagenomes without Cultivation Approach
2.5.1. Extraction of Bacterial Metagenome
2.5.2. Bacterial Diversity Profiling Using 16S rRNA Amplicon Sequencing Analysis
2.5.3. Identification of PHA Synthases from Marine Sponge Bacterial Metagenome
3. Results and Discussion
3.1. Identification of PHA-Producing Strains from Sediment
3.2. Production and Characterisation of PHA from Isolated Strains
3.3. Identification of phaC from Sediment
3.4. Bacterial Diversity Profiling in Marine Sponges
3.5. PHA-Producing Taxa Detected in Sponge-Associated Bacteria
3.6. Identification of phaC from Marine Sponges
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Khoo, K.S.; Ho, L.Y.; Lim, H.R.; Leong, H.Y.; Chew, K.W. Plastic waste associated with the COVID-19 pandemic: Crisis or opportunity? J. Hazard. Mater. 2021, 417, 126108. [Google Scholar] [CrossRef] [PubMed]
- Amelia, T.S.M.; Khalik, W.M.A.W.M.; Ong, M.C.; Shao, Y.T.; Pan, H.-J.; Bhubalan, K. Marine microplastics as vectors of major ocean pollutants and its hazards to the marine ecosystem and humans. Prog. Earth Planet. Sci. 2021, 8, 12. [Google Scholar] [CrossRef]
- Faezah, A.N.; Rahayu, A.; Vigneswari, S.; Majid, M.I.A.; Amirul, A.A. Regulating the molar fraction of 4-hydroxybutyrate in Poly(3-hydroxybutyrate-co-4-hydroxybutyrate) by biological fermentation and enzymatic degradation. World J. Microbiol. Biotechnol. 2011, 27, 2455. [Google Scholar] [CrossRef]
- Chaudhry, W.N.; Jamil, N.; Ali, I.; Ayaz, M.H.; Hasnain, S. Screening for polyhydroxyalkanoate (PHA)-producing bacterial strains and comparison of PHA production from various inexpensive carbon sources. Ann. Microbiol. 2011, 61, 623–629. [Google Scholar] [CrossRef]
- Kourmentza, C.; Plácido, J.; Venetsaneas, N.; Burniol-Figols, A.; Varrone, C.; Gavala, H.N.; Reis, M.A. Recent advances and challenges towards sustainable polyhydroxyalkanoate (PHA) production. Bioengineering 2017, 4, 55. [Google Scholar] [CrossRef]
- Muhammadi; Shabina; Afzal, M.; Hameed, S. Bacterial polyhydroxyalkanoates-eco-friendly next generation plastic: Production, biocompatibility, biodegradation, physical properties and applications. Green Chem. Lett. Rev. 2015, 8, 56–77. [Google Scholar] [CrossRef]
- Prados, E.; Maicas, S. Bacterial production of hydroxyalkanoates (PHA). Univers. J. Microbiol. Res. 2016, 4, 23–30. [Google Scholar] [CrossRef]
- Yatim, A.F.M.; Syafiq, I.M.; Huong, K.H.; Amirul, A.-A.A.; Effendy, A.W.M.; Bhubalan, K. Bioconversion of novel and renewable agro-industry by-products into a biodegradable poly(3-hydroxybutyrate) by marine Bacillus megaterium UMTKB-1 strain. BioTechnologia 2017, 98, 141–151. [Google Scholar] [CrossRef]
- Stefani, F.O.P.; Bell, T.H.; Marchand, C.; de La Providencia, I.E.; Yassimi, A.E.; St-Arnaud, M.; Hijri, M. Culture-dependent and -independent methods capture different microbial community fractions in hydrocarbon-contaminated soils. PLoS ONE 2015, 10, e0128272. [Google Scholar] [CrossRef]
- Khan, A.B.; Khattak, M.I.; Tarar, O.M.; Habib, F.; Jamil, K.; Yasmin, A.; Parvez, S. Separation of polyhydroxyalkanoates-producing bacterial strains using PHA synthase gene and their evaluation for PHA deposition. Braz. Arch. Biol. Technol. 2013, 56, 645–652. [Google Scholar] [CrossRef][Green Version]
- Gozari, M.; Alborz, M.; El-Seedi, H.R.; Jassbi, A.R. Chemistry, biosynthesis and biological activity of terpenoids and meroterpenoids in bacteria and fungi isolated from different marine habitats. Eur. J. Med. Chem. 2021, 210, 112957. [Google Scholar] [CrossRef] [PubMed]
- Peña, C.; Castillo, T.; García, A.; Millán, M.; Segura, D. Biotechnological strategies to improve production of microbial poly-(3-hydroxybutyrate): A review of recent research work. Microb. Biotechnol. 2014, 7, 278–293. [Google Scholar] [CrossRef]
- Amelia, T.S.M.; Lau, N.-S.; Amirul, A.-A.A.; Bhubalan, K. Metagenomic data on bacterial diversity profiling of high-microbial-abundance tropical marine sponges Aaptos aaptos and Xestospongia muta from waters off Terengganu, South China Sea. Data Brief 2020, 31, 105971. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Kitamura, S.; Abe, H. Microbial Synthesis and Characterization of Poly(3-hydroxybutyrate-co-3-hydroxyhexanoate). Macromolecules 2002, 28, 4822–4828. [Google Scholar] [CrossRef]
- Kahar, P.; Tsuge, T.; Taguchi, K.; Doi, Y. High yield production of polyhydroxyalkanoates from soybean oil by Ralstonia eutropha and its recombinant strain. Polym. Degrad. Stab. 2004, 83, 79–86. [Google Scholar] [CrossRef]
- Azemi, M.A.F.M.; Fazielawanie, N.M.R.; Saidin, J.; Mohd Effendy, A.W.; Bhubalan, K. Application of sweetwater as potential carbon source for rhamnolipid production by marine Pseudomonas aeruginosa UMTKB-5. Int. J. Biosci. Biochem. Bioinforma. 2016, 6, 50–58. [Google Scholar] [CrossRef][Green Version]
- Patel, S.; Majumder, A.; Goyal, A. Potentials of exopolysaccharides from lactic acid bacteria. Indian J. Microbiol. 2012, 52, 3. [Google Scholar] [CrossRef]
- Elumalai, S.; Prakasam, V. Lipid granules staining (Nile red and bodypy) of different biofuel producing fresh water microalgae growing under various stress conditions. J. Exp. Sci. 2011, 2, 65–67. [Google Scholar]
- Amirul, A.A.; Yahya, A.R.M.; Sudesh, K.; Azizan, M.N.M.; Majid, M.I.A. Biosynthesis of poly(3-hydroxybutyrate-co-4-hydroxybutyrate) copolymer by Cupriavidus sp. USMAA1020 isolated from Lake Kulim, Malaysia. Biores. Technol. 2008, 99, 4903–4909. [Google Scholar] [CrossRef]
- Pandey, A.; Dhakar, K.; Sharma, A.; Priti, P.; Sati, P.; Kumar, B. Thermophilic bacteria that tolerate a wide temperature and pH range colonize the Soldhar (95 C) and Ringigad (80 C) hot springs of Uttarakhand, India. Ann. Microbiol. 2015, 65, 809–816. [Google Scholar] [CrossRef]
- Zuriani, R.; Vigneswari, S.; Azizan, M.N.M.; Majid, M.I.A.; Amirul, A.A. A high throughput Nile red fluorescence method for rapid quantification of intracellular bacterial polyhydroxyalkanoates. Biotechnol. Bioprocess. Eng. 2013, 18, 472–478. [Google Scholar] [CrossRef]
- Oshiki, M.; Satoh, H.; Mino, T. Rapid quantification of polyhydroxyalkanoates (PHA) concentration in activated sludge with the fluorescent dye Nile blue A. Water Sci. Technol. 2011, 64, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Braunegg, G.; Sonnleitner, B.; Lafferty, R.M. A rapid gas chromatographic method for the determination of poly-β-hydroxybutyric acid in microbial biomass. Appl. Microbiol. Biotechnol. 1978, 6, 29–37. [Google Scholar] [CrossRef]
- Amara, A.A.; Moawad, A.H. PhaC synthases and PHA depolymerases: The enzymes that produce and degrade plastic. IIUM Eng. J. 2011, 12, 21–37. [Google Scholar] [CrossRef][Green Version]
- Beloqui, A.; Zumárraga, M.; Alcalde, M.; Golyshin, P.N.; Ferrer, M. Microbes and Enzymes: Recent Trends and New Directions to Expand Protein Space. In Protein Engineering Handbook; Lutz, S., Bornscheuer, U.T., Eds.; Wiley-VCH: Weinheim, Germany, 2008; Volume 2, pp. 233–269. [Google Scholar]
- Mares, M.C.D.; Sipkema, D.; Huang, S.; Bunk, B.; Overmann, J.; Elsas, J.D.V. Host specificity for bacterial, archaeal and fungal communities determined for high-and low-microbial abundance sponge species in two genera. Front. Microbiol. 2017, 8, 2560. [Google Scholar] [CrossRef]
- Inoue, D.; Suzuki, Y.; Uchida, T.; Morohoshi, J.; Sei, K. Polyhydroxyalkanoate production potential of heterotrophic bacteria in activated sludge. J. Biosci. Bioeng. 2016, 121, 47–51. [Google Scholar] [CrossRef]
- Green, M.R.; Sambrook, J. Agarose Gel Electrophoresis. Cold Spring Harb. Protoc. 2019, 2019, pdb-rot100404. [Google Scholar] [CrossRef]
- Reddy, S.V.; Thirumala, A.M.; Reddy, A.T.V.K.; Mahmood, A.S.K. Isolation of bacteria producing polyhydroxyalkanoates (PHA) from municipal sewage sludge. World J. Microbiol. Biotechnol. 2008, 24, 2949–2955. [Google Scholar] [CrossRef]
- Muangwong, A.; Boontip, T.; Pachimsawat, J.; Napathorn, S.C. Medium chain length polyhydroxyalkanoates consisting primarily of unsaturated 3-hydroxy-5-cis-dodecanoate synthesized by newly isolated bacteria using crude glycerol. Microb. Cell Fact. 2016, 15, 55. [Google Scholar] [CrossRef]
- Basharat, Z.; Yasmin, A.; He, T.; Tong, Y. Genome sequencing and analysis of Alcaligenes faecalis subsp. phenolicus MB207. Sci. Rep. 2018, 8, 3616. [Google Scholar] [CrossRef]
- Doughari, H.J.; Ndakidemi, P.A.; Human, I.S.; Benade, S. The ecology, biology and pathogenesis of Acinetobacter spp.: An overview. Microbes Environ. 2011, 26, 1103150282. [Google Scholar] [CrossRef] [PubMed]
- Koller, M. A Review on established and emerging fermentation schemes for microbial production of polyhydroxyalkanoate (PHA) biopolyesters. Fermentation 2018, 4, 30. [Google Scholar] [CrossRef]
- Jiang, G.; Hill, D.J.; Kowalczuk, M.; Johnston, B.; Adamus, G.; Irorere, V.; Radecka, I. Carbon sources for polyhydroxyalkanoates and an integrated biorefinery. Int. J. Mol. Sci. 2016, 17, 1157. [Google Scholar] [CrossRef] [PubMed]
- Santhanam, A.; Sasidharan, S. Microbial production of polyhydroxyalkanotes (PHA) from Alcaligens spp. and Pseudomonas oleovorans using different carbon sources. Afr. J. Biotechnol. 2010, 9, 3144–3150. [Google Scholar]
- Choi, J.I.; Lee, S.Y.; Han, K. Cloning of the Alcaligenes latus polyhydroxyalkanoate biosynthesis genes and use of these genes for enhanced production of poly(3-hydroxybutyrate) in Escherichia coli. Appl. Environ. Microbiol. 1998, 64, 4897–4903. [Google Scholar] [CrossRef]
- Steinbüchel, A.; Lütke-Eversloh, T. Metabolic engineering and pathway construction for biotechnological production of relevant polyhydroxyalkanoates in microorganisms. Biochem. Eng. J. 2003, 16, 81–96. [Google Scholar] [CrossRef]
- Fiore, C.L.; Labrie, M.; Jarett, J.K.; Lesser, M.P. Transcriptional activity of the giant barrel sponge, Xestospongia muta Holobiont: Molecular evidence for metabolic interchange. Front. Microbiol. 2015, 6, 364. [Google Scholar] [CrossRef]
- Croué, J.; West, N.J.; Escande, M.L.; Intertaglia, L.; Lebaron, P.; Suzuki, M.T. A single betaproteobacterium dominates the microbial community of the crambescidine-containing sponge Crambe crambe. Sci. Rep. 2013, 3, 2583. [Google Scholar] [CrossRef]
- Cleary, D.F.R.; Polónia, A.R.M.; Becking, L.E.; de Voogd, N.J.; Gomes, H.; Gomes, N.C.M. Compositional analysis of bacterial communities in seawater, sediment, and sponges in the Misool coral reef system, Indonesia. Mar. Biodivers. 2017, 48, 1889–1901. [Google Scholar] [CrossRef]
- Weisz, J.B.; Lindquist, N.; Martens, C.S. Do associated microbial abundances impact marine demosponge pumping rates and tissue densities? Oecologia 2008, 155, 367–376. [Google Scholar] [CrossRef]
- Meyer, F.; Paarmann, D.; Souza, M.D.; Olson, R.; Glass, E.M.; Kubal, M.; Paczian, T.; Rodriguez, A.; Stevens, R.; Wilke, A.; et al. The metagenomics RAST server—A public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinformatics 2008, 9, 386. [Google Scholar] [CrossRef] [PubMed]
- White, J.R.; Patel, J.; Ottesen, A.; Arce, G.; Blackwelder, P.; Lopez, J. Pyrosequencing of bacterial symbionts within Axinella corrugata sponges: Diversity and seasonal variability. PLoS ONE 2012, 7, e38204. [Google Scholar] [CrossRef] [PubMed]
- Morrow, K.M.; Fiore, C.L.; Lesser, M.P. Environmental drivers of microbial community shifts in the giant barrel sponge, Xestospongia muta, over a shallow to mesophotic depth gradient. Environ. Microbiol. 2016, 18, 2025–2038. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.B.; Gao, X. Characterizing the bacterial associates of three Caribbean sponges along a gradient from shallow to mesophotic depths. FEMS Microbiol. Ecol. 2013, 85, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, D.; Lai, T.K.; Saha, A.; Selvin, J.; Mukherjee, J. Structural elucidation and antimicrobial activity of a diketopiperazine isolated from a Bacillus sp. associated with the marine sponge Spongia officinalis. Nat. Prod. Res. 2019, 35, 2315–2323. [Google Scholar] [CrossRef]
- Kawata, Y.; Nishimura, T.; Matsushita, I.; Tsubota, J. Efficient production and secretion of pyruvate from Halomonas sp. KM-1 under aerobic conditions. AMB Express 2016, 6, 2. [Google Scholar] [CrossRef]
- Kaluzhnaya, O.V.; Lipko, I.A.; Itskovich, V.B. PCR-screening of bacterial strains isolated from the microbiome of the Lubomirskia baicalensis sponge for the presence of secondary metabolite synthesis genes. Limnol. Freshw. Biol. 2021, 4, 1137–1142. [Google Scholar] [CrossRef]
- Sathiyanarayanan, G.; Saibaba, G.; Kiran, G.S.; Yang, Y.H.; Selvin, J. Marine sponge-associated bacteria as a potential source for polyhydroxyalkanoates. Crit. Rev. Microbiol. 2017, 43, 294–312. [Google Scholar] [CrossRef]
- Tan, H.T.; Chek, M.F.; Lakshmanan, M.; Foong, C.P.; Hakoshima, T.; Sudesh, K. Evaluation of BP-M-CPF4 polyhydroxyalkanoate (PHA) synthase on the production of poly (3-hydroxybutyrate-co-3-hydroxyhexanoate) from plant oil using Cupriavidus necator transformants. Int. J. Biol. Macromol. 2020, 159, 250–257. [Google Scholar] [CrossRef]
- Squio, C.R.; Aragão, G.M.F. Estratégias de cultivo para produção dos plásticos biodegradáveis poli(3-hidroxibutirato) e poli(3-hidroxibutirato-co-3-hidroxivalerato) por bactérias. Quim. Nova 2004, 27, 615–622. [Google Scholar] [CrossRef][Green Version]
- Kiun, J.T.; Amelia, T.S.M.; Huong, K.H.; Amirul, A.A.A.; Bhubalan, K. Optimizing the biosynthesis of renewable polyhydroxyalkanoate copolymer containing 3-hydroxyvalerate by Massilia haematophila using statistical modeling. BioTechnologia 2019, 100, 359–371. [Google Scholar] [CrossRef]
- Paula, C.B.C.; de Paula-Elias, F.C.; Rodrigues, M.N.; Coelho, L.F.; de Oliveira, N.M.L.; de Almeida, A.F.; Contiero, J. Polyhydroxyalkanoate Synthesis by Burkholderia glumae into a sustainable sugarcane biorefinery concept. Front. Bioeng. Biotechnol. 2020, 8, 631284. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, F.; Radax, R.; Woebken, D.; Holtappels, M.; Lavik, G.; Rapp, H.T.; Schläppy, M.L.; Schleper, C.; Kuypers, M.M.M. Complex nitrogen cycling in the sponge Geodia barretti. Environ. Microbiol. 2009, 11, 2228–2243. [Google Scholar] [CrossRef] [PubMed]
- Fehmida, B.; Ikram, U.; Sana, A.; Muhammad, Y.; Eman, A.K.; Ahmed, A.K.A.G.; Esam, I.A. Diversity and bioprospecting potential of rhizo and endophytic bacteria from two mangrove plants in Saudi Arabia. Afr. J. Microbiol. Res. 2017, 11, 729–739. [Google Scholar]






| Strain | Primer Sequence (5′-3′) | Database Reference |
|---|---|---|
| Acinetobacter Calcoaceticus | * F: 5-ATGCTTAGTATTGAATTTTT-3 | UniProt |
| R: 5-TTATGGCTCATCCTCACG-3 | (EMBL:SUU05450.1) | |
| Alcaligenes Faecalis | F: 5′-ATGACTTCTGAATCATCC-3 | NCBI Protein |
| R: 5′-TTAACTGGCGCGCACTTTCA-3 | (NZ_CP013119.1) | |
| Primer pair | Group | Primer sequence (5′-3′) |
| 1 | I | * F: ATGAGAAAATACCTAATCCT |
| R: TTATCGATTTACGTTCTG | ||
| 2 | I | F: ATGCATTTTATACTCATTTC |
| R: TCACAGCAATGCAATTAATAAAACT | ||
| 3 | II | F: ATGATCTATTCACTAACAAG |
| R: TTAATTTTTTACAATATACTCCTTAA | ||
| 4 | II | F: GTGTCAAAAAACCATAAAGGAAAT |
| R: CTAGTGGAATCGATCTGG |
| Strain | Carbon Source | PHA Content (wt%) | Monomer Composition (mol%) | ||||
|---|---|---|---|---|---|---|---|
| 3HV | 3HO | 3HD | 3HUD | 3HHx | |||
| Sphingobacterium mizutaii | Sucrose | 37.4 | - | 15 | 28 | 49 | 8 |
| UMTKB-6 | |||||||
| Alcaligenes faecalis | Glucose | 61.1 | Tr | - | 25 | 74 | - |
| UMTKB-7 | |||||||
| Acinetobacter calcoaceticus | Glycerol | 49.4 | Tr | - | Tr | 99 | - |
| UMTKB-8 | |||||||
| Strain Isolates from Sediment-Associated Bacterial Culture | |||
|---|---|---|---|
| Strain Isolate | Closest Species | phaC Class | Strain Identity (%) |
| UMTKB-6 | Sphingobacterium mizutaii | I | 99.0 |
| UMTKB-7 | Alcaligenes faecalis | I | 99.0 |
| UMTKB-8 | Acinetobacter calcoaceticus | I | 99.3 |
| Cloned Genes from Sponge-Associated Bacterial Metagenome | |||
|---|---|---|---|
| Gene Clone | Closest phaC Gene | phaC Class | Gene Identity (%) |
| 2 | Pseudomonas stutzeri 1317 phaC (AY278219.1) | II | 88.0 |
| 1B | Uncultured bacterium AR5-9d_16 phaC (AB220790.1) | III | 93.0 |
| 2B | Rhodocista pekingensis phaC (AY283802.1) | I | 74.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alsaadi, A.; Ganesen, S.S.K.; Amelia, T.S.M.; Moanis, R.; Peeters, E.; Vigneswari, S.; Bhubalan, K. Polyhydroxyalkanoate (PHA) Biopolymer Synthesis by Marine Bacteria of the Malaysian Coral Triangle Region and Mining for PHA Synthase Genes. Microorganisms 2022, 10, 2057. https://doi.org/10.3390/microorganisms10102057
Alsaadi A, Ganesen SSK, Amelia TSM, Moanis R, Peeters E, Vigneswari S, Bhubalan K. Polyhydroxyalkanoate (PHA) Biopolymer Synthesis by Marine Bacteria of the Malaysian Coral Triangle Region and Mining for PHA Synthase Genes. Microorganisms. 2022; 10(10):2057. https://doi.org/10.3390/microorganisms10102057
Chicago/Turabian StyleAlsaadi, Athraa, Sree Selva Kumar Ganesen, Tan Suet May Amelia, Radwa Moanis, Eveline Peeters, Sevakumaran Vigneswari, and Kesaven Bhubalan. 2022. "Polyhydroxyalkanoate (PHA) Biopolymer Synthesis by Marine Bacteria of the Malaysian Coral Triangle Region and Mining for PHA Synthase Genes" Microorganisms 10, no. 10: 2057. https://doi.org/10.3390/microorganisms10102057
APA StyleAlsaadi, A., Ganesen, S. S. K., Amelia, T. S. M., Moanis, R., Peeters, E., Vigneswari, S., & Bhubalan, K. (2022). Polyhydroxyalkanoate (PHA) Biopolymer Synthesis by Marine Bacteria of the Malaysian Coral Triangle Region and Mining for PHA Synthase Genes. Microorganisms, 10(10), 2057. https://doi.org/10.3390/microorganisms10102057








