Dermacentor reticulatus and Babesia canis in Bavaria (Germany)—A Georeferenced Field Study with Digital Habitat Characterization
Abstract
1. Introduction
2. Material and Methods
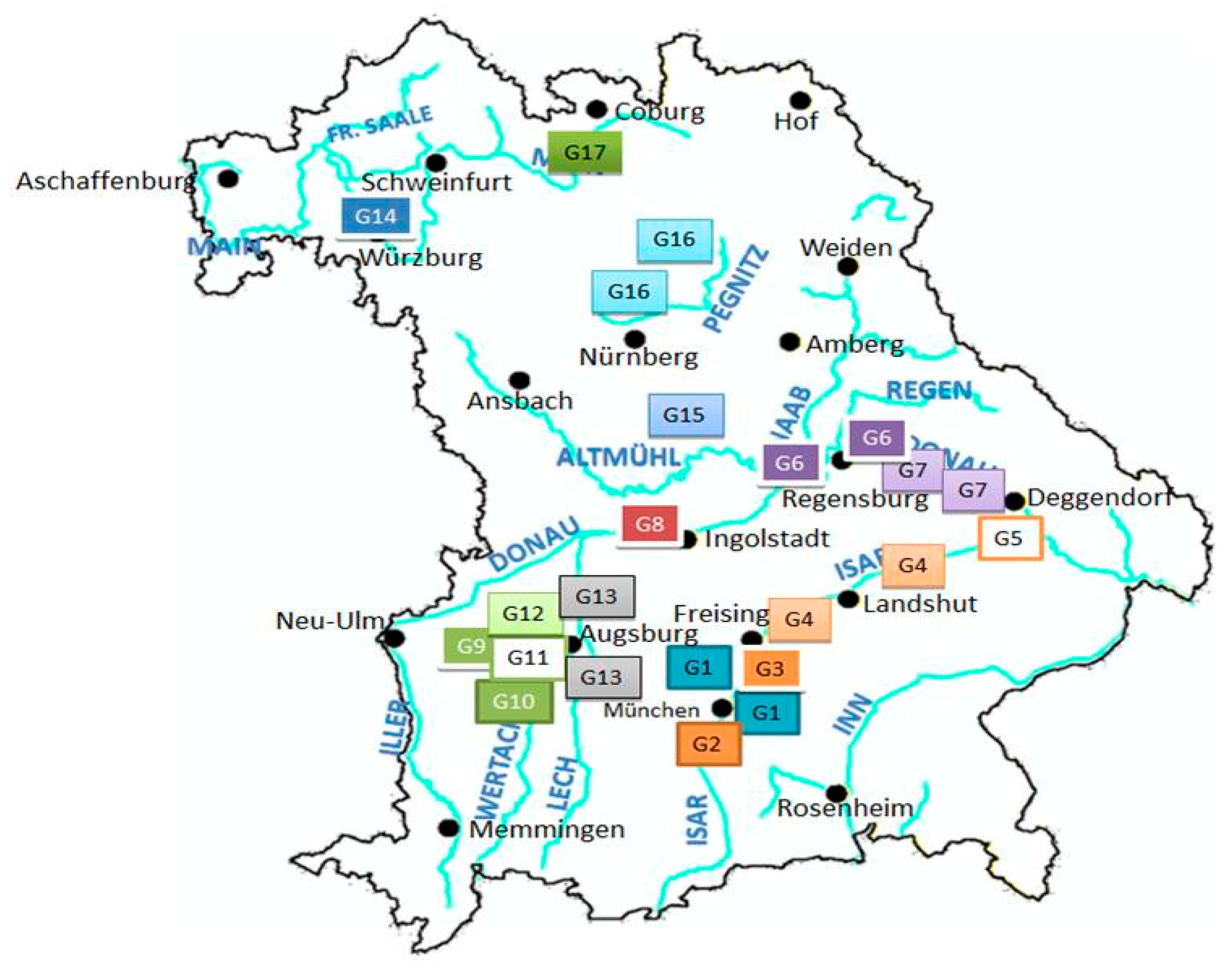
2.1. Field Study for Dermacentor reticulatus
2.2. PCR Analysis for Babesia canis
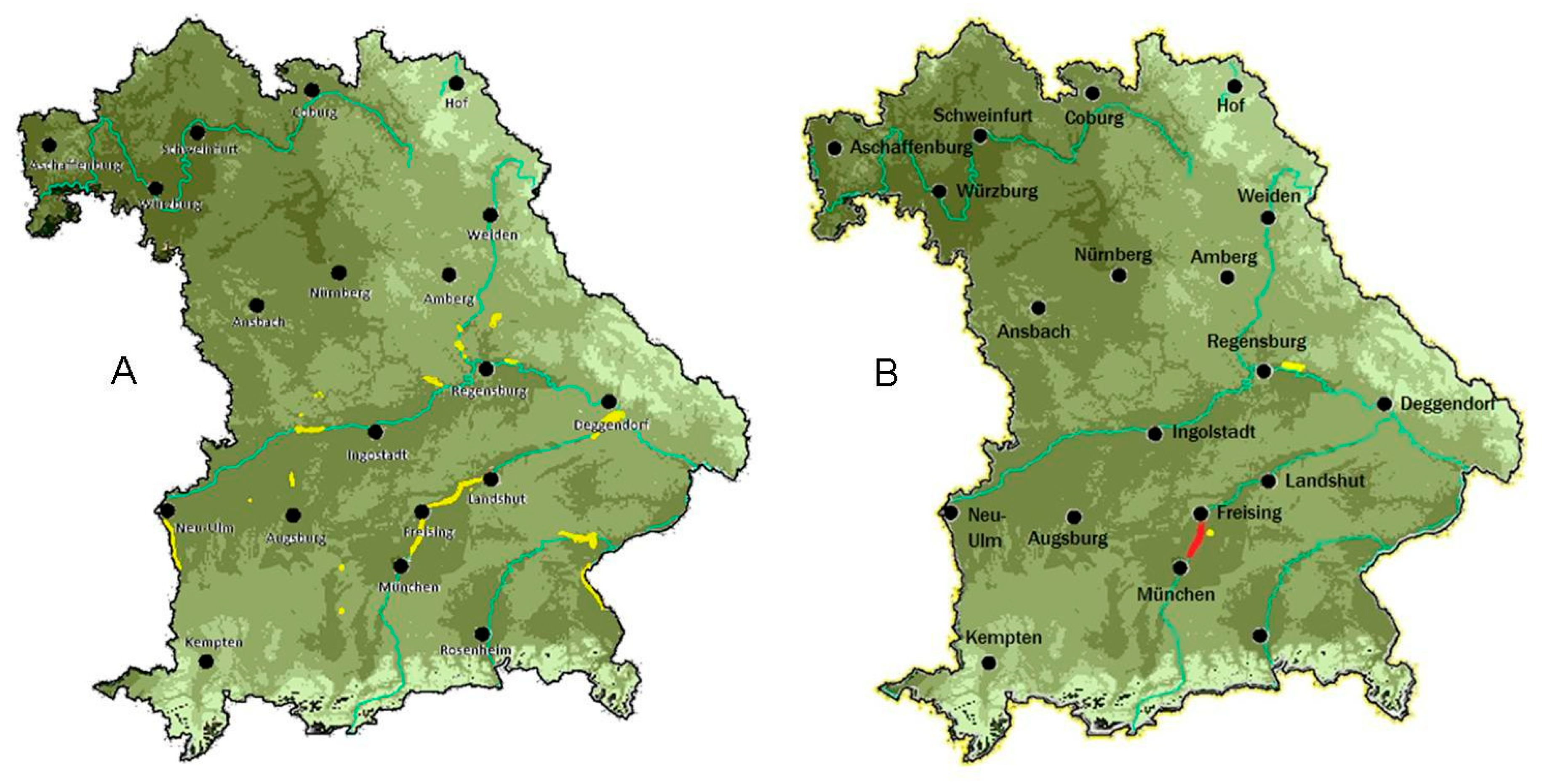
2.3. Digital Habitat Analysis
3. Results
3.1. Field Study on Dermacentor reticulatus
3.2. PCR Analysis for Babesia canis
3.3. Digital Habitat Characterisation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Barutzki, D.; Reule, M.; Scheunemann, R.; Heile, C.; Schein, E. Die Babesiose des Hundes. eine autochtone Erkrankung in Deutschland. Dtsch. Tierärztebl. 2007, 55, 284–293. [Google Scholar]
- Solano-Gallego, L.; Sainz, Á.; Roura, X.; Estrada-Peña, A.; Miró, G. A review of canine babesiosis: The European perspective. Parasites Vectors 2016, 9, 336. [Google Scholar] [CrossRef] [PubMed]
- Dautel, H.; Dippel, C.; Oehme, R.; Hartelt, K.; Schettler, E. Evidence for an increased geographical distribution of Dermacentor reticulatus in Germany and detection of Rickettsia sp. RpA4. Int. J. Med. Microbiol. 2006, 296 (Suppl. 40), 149–156. [Google Scholar] [CrossRef]
- Mierzejewska, E.J.; Estrada-Pena, A.; Alsarraf, M.; Kowalec, M.; Bajer, A. Mapping of Dermacentor reticulatus expansion in Poland in 2012–2014. Ticks Tick-Borne Dis. 2016, 7, 94–106. [Google Scholar] [CrossRef] [PubMed]
- Dumic, I.; Severnini, E. “Ticking Bomb”: The Impact of Climate Change on the Incidence of Lyme Disease. Can. J. Infect. Dis. Med. Microbiol. 2018, 2018, 5719081. [Google Scholar] [CrossRef] [PubMed]
- Enigk, K. Zum Vorkommen der Zecke Dermacentor pictus (Herm 1804) in Deutschland. Z. Parasitenkd. 1958, 16, 419–422. [Google Scholar] [CrossRef]
- Bauch, R.; Danner, G. Funde von Dermacentor reticulatus (Ixodida, Ixodidae) in den DDR-Bezirken Leipzig und Halle. Angew. Parasitol. 1988, 29, 250–254. [Google Scholar]
- Beelitz, P.; Schumacher, S.; Marholdt, F.; Pfister, K.; Silaghi, C. The prevalence of Babesia canis canis in marsh ticks (Dermacentor reticulatus) in the Saarland. Berl. Munch Tierarztl. Wochenschr. 2012, 125, 168–171. [Google Scholar]
- Cornely, M.; Schultz, U. Zur Zeckenfauna Ostdeutschlands. Angew. Parasitol. 1992, 33, 173–183. [Google Scholar]
- Kahl, O.; Janetzki, C.; Gray, J.S.; Stein, J.; Bauch, R.J. Tick infection rates with Borrelia: Ixodes ricinus versus Haemaphysalis concinna and Dermacentor reticulatus in two locations in eastern Germany. Med. Vet. Entomol. 1992, 6, 363–366. [Google Scholar] [CrossRef]
- Liebisch, A.; Rahman, M.S. Zum Vorkommen und zur vektoriellen Bedeutung der Zecken Dermacentor marginatus (Sulzer, 1776) und Dermacentor reticulatus (Fabricius, 1794) in Deutschland. Trop. Parasitol. 1976, 27, 393–404. [Google Scholar]
- Obiegala, A.; Pfeffer, M.; Pfister, K.; Tiedemann, T.; Thiel, C.; Balling, A.; Karnath, C.; Woll, D.; Silaghi, C. Candidatus Neoehrlichia mikurensis and Anaplasma phagocytophilum: Prevalences and investigations on a new transmission path in small mammals and ixodid ticks. Parasites Vectors 2014, 7, 563. [Google Scholar] [PubMed]
- Silaghi, C.; Woll, D.; Hamel, D.; Pfister, K.; Mahling, M.; Pfeffer, M. Babesia spp. and Anaplasma phagocytophilum in questing ticks, ticks parasitizing rodents and the parasitized rodents--analyzing the host-pathogen-vector interface in a metropolitan area. Parasites Vectors 2012, 5, 191. [Google Scholar] [CrossRef]
- Beck, S.; Schreiber, C.; Schein, E.; Krücken, J.; Baldermann, C.; Pachnicke, S.; von Samson-Himmelstjerna, G.; Kohn, B. Tick infestation and prophylaxis of dogs in northeastern Germany: A prospective study. Ticks Tick-Borne Dis. 2014, 5, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Rehbein, S.; Kaulfuß, K.H.; Visser, M.; Sommer, M.F.; Grimm, F.; Silaghi, C. Parasites of sheep herding dogs in central Germany. Berl. Munch. Tierarztl. Wochenschr. 2016, 129, 56–64. [Google Scholar] [PubMed]
- Schreiber, C.; Krücken, J.; Beck, S.; Maaz, D.; Pachnicke, S.; Krieger, K.; Gross, M.; Kohn, B.; von Samson-Himmelstjerna, G. Pathogens in ticks collected from dogs in Berlin/Brandenburg, Germany. Parasites Vectors 2014, 7, 535. [Google Scholar] [CrossRef]
- Nosek, J. The ecology, bionomics, behaviour and public health importance of Dermacentor marginatus and D. reticulatus ticks. Wiad. Parazytol. 1972, 18, 721–725. [Google Scholar]
- Menn, B. Untersuchungen zur Verbreitung und Ökologie von Dermacentor spec. (Ixodidae, Acari) in Deutschland. Diploma Thesis, Rheinische Friedrich-Wilhelms-Universität, Bonn, Germany, 2006. [Google Scholar]
- Heile, C.; Heydorn, A.O.; Schein, E. Dermacentor reticulatus (Fabricius, 1794)—Verbreitung, Biologie und Vektor von Babesia canis in Deutschland. Berl. Münchener Tierärztl. Wochenschr. 2006, 119, 330–334. [Google Scholar]
- Silaghi, C.; Hamel, D.; Thiel, C.; Pfister, K.; Pfeffer, M. Spotted fever group rickettsiae in ticks, Germany. Emerg. Infect. Dis. 2011, 17, 890–892. [Google Scholar] [CrossRef]
- Zahler, M.; Gothe, R.; Rinder, H. Dermacentor-Zecken in Frankreich und Deutschland. Zur molekularbiologischen Artdifferenzierung, Ökologie und epidemiologischen Bedeutung. Tierärztl. Prax. 1996, 24, 209–211. [Google Scholar]
- Wisz, M.S.; Guisan, A. Do pseudo-absence selection strategies influence species distribution models and their predictions? An information-theoretic approach based on simulated data. BMC Ecol. 2009, 9, 8. [Google Scholar] [CrossRef]
- Arthur, D.R. Ticks. A monograph of the Ixodoidea. Part V. In On the genera Dermacentor, Anocentor, Cosmiomma, Boophilus and Margaporus; Cambridge University Press: Cambridge, UK, 1960. [Google Scholar]
- Hillyard, P.D. Ticks of North-West Europe; Field Studies Council: Montford Bridge, UK, 1996. [Google Scholar]
- Casati, S.; Sager, H.; Gern, L.; Piffaretti, J.C. Presence of potentially pathogenic Babesia sp. for human in Ixodes ricinus in Switzerland. Ann. Agric. Environ. Med. 2006, 13, 65–70. [Google Scholar] [PubMed]
- Schorn, S.; Pfister, K.; Reulen, H.; Mahling, M.; Silaghi, C. Occurrence of Babesia spp., Rickettsia spp. and Bartonella spp. in Ixodes ricinus in Bavarian public parks, Germany. Parasites Vectors 2011, 4, 135. [Google Scholar] [CrossRef] [PubMed]
- Zahler, M.; Gothe, R. Endemisierungsrisiko von Babesia canis durch Dermacentor reticulatus in Deutschland. Eine epidemiologische Studie. Tierärztl. Prax. 1997, 25, 666–670. [Google Scholar]
- Zahler, M.; Steffen, T.; Lutz, S.; Hähnel, W.C.; Rinder, H.; Gothe, R. Babesia canis and Dermacentor reticulatus in München, ein neuer Naturherd in Deutschland. Tierärztl. Prax. 2000, 28, 116–120. [Google Scholar]
- Zahler, M.; Loster, F.; Merkl, C.; Rinder, H.; Gothe, R. Infektionsgefahr für Hunde in Regensburg—Ein neuer Naturherd von Babesia canis und Dermacentor reticulatus in Deutschland. Tierärztl. Prax. 2000, 28, 395–398. [Google Scholar]
- Cochez, C.; Lempereur, L.; Madder, M.; Claerebout, E.; Simons, L.; De Wilde, N.; Linden, A.; Saegerman, C.; Heyman, P.; Losson, B. Foci report on indigenous Dermacentor reticulatus populations in Belgium and a preliminary study of associated babesiosis pathogens. Med. Vet. Entomol. 2012, 26, 355–358. [Google Scholar] [CrossRef]
- Obsomer, V.; Wirtgen, M.; Linden, A.; Claerebout, E.; Heyman, P.; Heylen, D.; Madder, M.; Maris, J.; Lebrun, M.; Tack, W.; et al. Spatial disaggregation of tick occurrence and ecology at a local scale as a preliminary step for spatial surveillance of tick-borne diseases: General framework and health implications in Belgium. Parasited Vectors 2013, 6, 190. [Google Scholar] [CrossRef]
- Tijsse-Klasen, E.; Jameson, L.J.; Fonville, M.; Leach, S.; Sprong, H.; Medlock, J.M. First detection of spotted fever group rickettsiae in Ixodes ricinus and Dermacentor reticulatus ticks in the UK. Epidemiol. Infect. 2011, 139, 524–529. [Google Scholar] [CrossRef]
- Pfäffle, M.; Littwin, N.; Petney, T. Host preferences of immature Dermacentor reticulatus Acari: Ixodidae) in a forest habitat in Germany. Ticks Tick-Borne Dis. 2015, 6, 508–515. [Google Scholar] [CrossRef]
- Bullová, E.; Lukán, M.; Stanko, M.; Petko, B. Spatial distribution of Dermacentor reticulatus tick in Slovakia in the beginning of the 21st century. Vet. Parasitol. 2009, 165, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Kiewra, D.; Czulowska, A. Evidence for an increased distribution range of Dermacentor reticulatus in south-west Poland. Exp. Appl. Acarol. 2013, 59, 501–506. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rubel, F.; Brugger, K.; Pfeffer, M.; Chitimia-Dobler, L.; Didyk, Y.; Leverenz, S.; Dautel, H.; Kahl, O. Geographical distribution of Dermacentor marginatus and Dermacentor reticulatus in Europe. Ticks Tick-Borne Dis. 2016, 7, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Hornok, S.; Farkas, R. Influence of biotope on the distribution and peak activity of questing ixodid ticks in Hungary. Med. Vet. Entomol. 2009, 23, 41–46. [Google Scholar] [CrossRef]
- Zygner, W.; Górski, P.; Wedrychowicz, H. New localities of Dermacentor reticulatus tick (vector of Babesia canis canis) in central and eastern Poland. Pol. J. Vet. Sci. 2009, 12, 549–555. [Google Scholar] [PubMed]
- Hubálek, Z.; Halouzka, J.; Juricová, Z. Host-seeking activity of ixodid ticks in relation to weather variables. J. Vector Ecol. 2003, 28, 159–165. [Google Scholar] [PubMed]
- Hornok, S.; Kartali, K.; Takács, N.; Hofmann-Lehmann, R. Uneven seasonal distribution of Babesia canis and its two 18S rDNA genotypes in questing Dermacentor reticulatus ticks in urban habitats. Ticks Tick-Borne Dis. 2016, 7, 694–697. [Google Scholar] [CrossRef]
- Tomanović, S.; Chochlakis, D.; Radulović, Z.; Milutinović, M.; Cakić, S.; Mihaljica, D.; Tselentis, Y.; Psaroulaki, A. Analysis of pathogen co-occurrence in host-seeking adult hard ticks from Serbia. Exp. Appl. Acarol. 2013, 59, 367–376. [Google Scholar] [CrossRef]
- Kubelová, M.; Tkadlec, E.; Bednář, M.; Roubalová, E.; Siroký, P. West-to-east differences of Babesia canis canis prevalence in Dermacentor reticulatus ticks in Slovakia. Vet. Parasitol. 2011, 180, 191–196. [Google Scholar] [CrossRef]
- Svehlová, A.; Berthová, L.; Sallay, B.; Boldiš, V.; Sparagano, O.A.; Spitalská, E. Sympatric occurrence of Ixodes ricinus, Dermacentor reticulatus and Haemaphysalis concinna ticks and Rickettsia and Babesia species in Slovakia. Ticks Tick-Borne Dis. 2014, 5, 600–605. [Google Scholar] [CrossRef]
- Jongejan, F.; Ringenier, M.; Putting, M.; Berger, L.; Burgers, S.; Kortekaas, R.; Lenssen, J.; van Roessel, M.; Wijnveld, M.; Madder, M. Novel foci of Dermacentor reticulatus ticks infected with Babesia canis and Babesia caballi in the Netherlands and in Belgium. Parasit. Vectors 2015, 8, 232. [Google Scholar] [CrossRef] [PubMed]
- Schaarschmidt, D.; Gilli, U.; Gottstein, B.; Marreros, N.; Kuhnert, P.; Daeppen, J.A.; Rosenberg, G.; Hirt, D.; Frey, C.F. Questing Dermacentor reticulatus harbouring Babesia canis DNA associated with outbreaks of canine babesiosis in the Swiss Midlands. Ticks Tick-Borne Dis. 2013, 4, 334–340. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Król, N.; Kiewra, D.; Lonc, E.; Janaczyk, B.; Chodorowska-Skubiszewska, A.; Dzięcioł, M.; Gola, M.; Gruszka, R.; Jackowska-Szlachcic, E.; Jagiełło, M.; et al. Dermacentor reticulatus (Fabricius, 1794) and Babesia canis (Piana et Galli-Valerio, 1895) as the parasites of companion animals (dogs and cats) in the Wrocław area, south-western Poland. Ann. Parasitol. 2016, 62, 125–130. [Google Scholar] [PubMed]
| Dermacentor reticulatus (n) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Area | Site | Geographic Coordinates | Altitude | Temperature at 50 cm (°C) | Soil Temperature at 5 cm Depth (°C) | Relative Humidity (%) | General Weather Impression | M | F | Total |
| G1—Munich | Zengermoos | 48°17′21.39″ N/11°46′1.19″ E | 472 | 19.7 | 22 | 56 | sunny | 1 | 0 | 1 |
| G3—Lower Isarauen (Munich to Freising) | Garching to Mintraching | 48°14′36.82″ N/11°40′18.42″ E | 488 | 16.2 | 16 | 40.5 | overcast | 0 | 10 | 10 |
| Pulling to Mintraching | 48°21′42.82″ N/11°43′12.02″ E | 447 | 23 | 20 | 55 | overcast | 1 | 2 | 3 | |
| Achering to Garching North | 48°22′6.42″ N/11°43′55.69″ E | 455 | 20.3 | 20 | 59 | sunny after previous rain | 7 | 9 | 16 | |
| Ismaning to Fischerhäuser | 48°15′3.69″ N/11°41′3.82″ E | 483 | 16.1 | 17 | 64 | overcast | 0 | 10 | 10 | |
| Fischerhäuser | 48°16′7.39″ N /11°41′53.55″ E | 477 | 22 | 19 | 44.5 | sunny | 4 | 7 | 11 | |
| Airport | 48°18′44.34″ N/11°42′8.74″ E | 459 ** | 25.1 23.4 | 21 25 | 65.9 54.7 | sunny sunny | 131 | 172 | 33 | |
| Hallbergmoos | 48°18′44.34″ N/11°42′8.74″ E | 465 | 19.4 | 19 | 42.7 | windy | 0 | 3 | 3 | |
| Pförrerau | 48°22′49.79″ N/11°44′37.30″ E | 437 | 19.6 | 18 | 58.5 | sunny | 0 | 1 | 1 | |
| Zwillingshof to Fischerhäuser | 48°16′56.82″ N/11°42′11.52″ E | 481 *** | 20 | 19 | 52.8 | rainy | 27 | 45 | 245 | |
| 19.3 | 20.5 | 53.6 | sunny | 18 | 30 | |||||
| 11.5 | 17 | 49.5 | not recorded | 10 | 20 | |||||
| 20 | 19 | 52.8 | overcast | 25 | 42 | |||||
| 23.1 | 21 | 58 | sunny | 16 | 12 | |||||
| G7—Regensburg East | Bach a.d. Donau | 49°1′55.46″ N/12°17′36.00″ E | 406 | 25.2 | 16 | 55.2 | sunny | 0 | 1 | 1 |
| Frauenzell Wiesent | 49°2′20.98″ N/12°22′19.96″ E | 352 | 26.2 | 16 | 50 | sunny | 2 | 3 | 5 | |
| Total | 125 | 214 | 339 | |||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silaghi, C.; Weis, L.; Pfister, K. Dermacentor reticulatus and Babesia canis in Bavaria (Germany)—A Georeferenced Field Study with Digital Habitat Characterization. Pathogens 2020, 9, 541. https://doi.org/10.3390/pathogens9070541
Silaghi C, Weis L, Pfister K. Dermacentor reticulatus and Babesia canis in Bavaria (Germany)—A Georeferenced Field Study with Digital Habitat Characterization. Pathogens. 2020; 9(7):541. https://doi.org/10.3390/pathogens9070541
Chicago/Turabian StyleSilaghi, Cornelia, Lisa Weis, and Kurt Pfister. 2020. "Dermacentor reticulatus and Babesia canis in Bavaria (Germany)—A Georeferenced Field Study with Digital Habitat Characterization" Pathogens 9, no. 7: 541. https://doi.org/10.3390/pathogens9070541
APA StyleSilaghi, C., Weis, L., & Pfister, K. (2020). Dermacentor reticulatus and Babesia canis in Bavaria (Germany)—A Georeferenced Field Study with Digital Habitat Characterization. Pathogens, 9(7), 541. https://doi.org/10.3390/pathogens9070541





