“Begging the Question”—Does Toxocara Infection/Exposure Associate with Multiple Sclerosis-Risk?
Abstract
1. Introduction
2. Materials and Methods
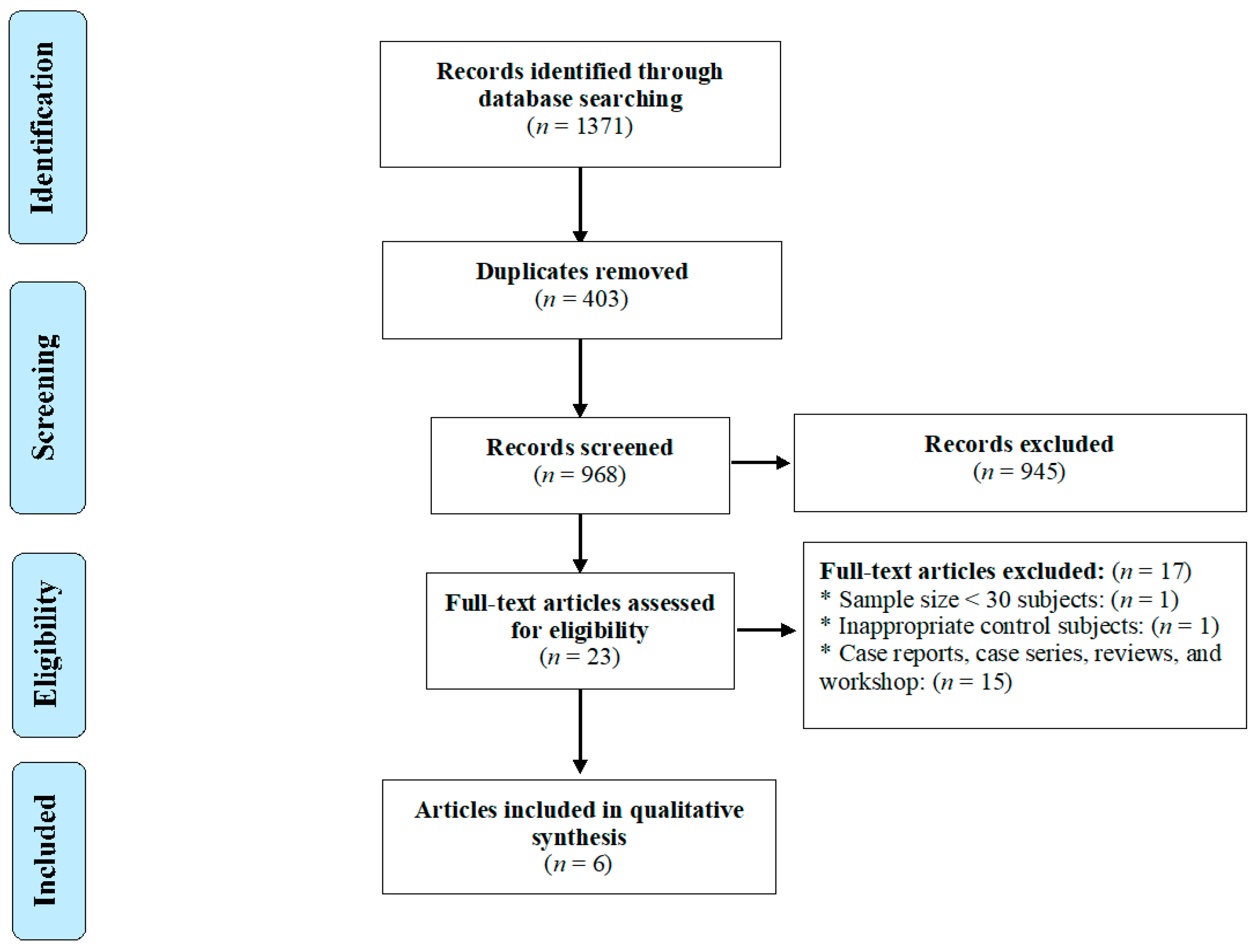
2.1. Search Strategy and Study Selection
2.2. Data Extraction and Quality Assessment
2.3. Data Synthesis and Statistical Analysis
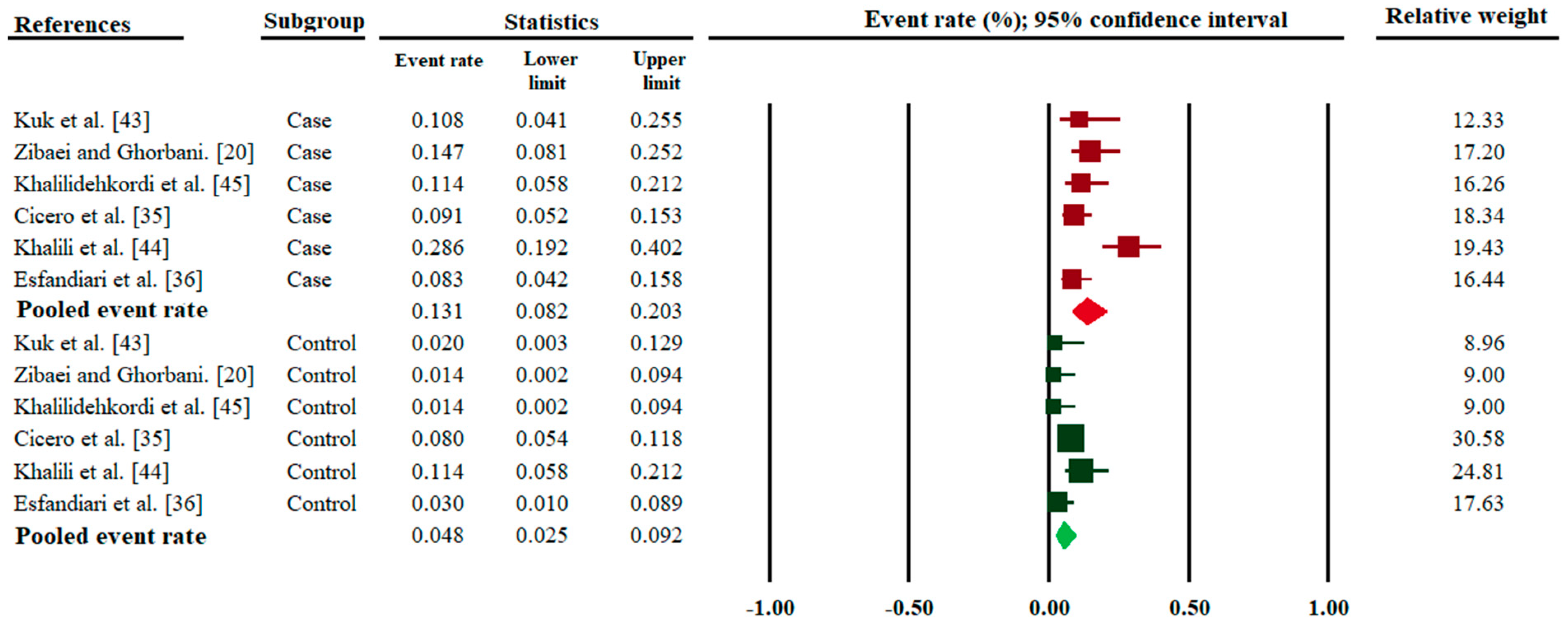
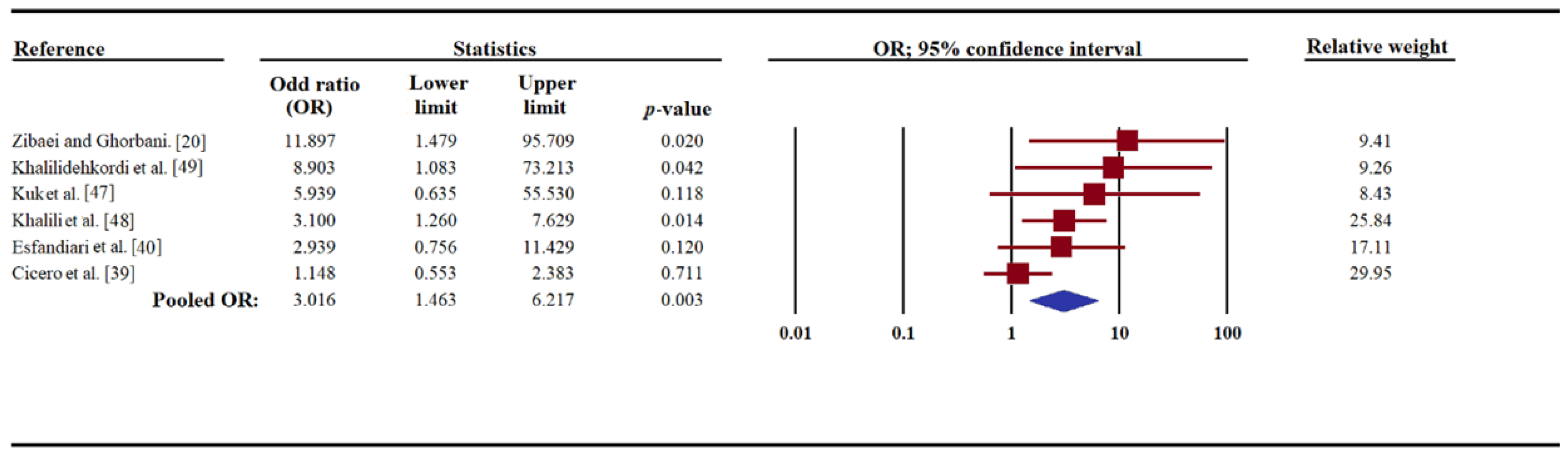
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Feigin, V.L.; Abajobir, A.A.; Abate, K.H.; Abd-Allah, F.; Abdulle, A.M.; Abera, S.F.; Abyu, G.Y.; Ahmed, M.B.; Aichour, A.N.; Aichour, I. Global, regional, and national burden of neurological disorders during 1990–2015: A systematic analysis for the Global Burden of Disease Study 2015. Lancet Neurol. 2017, 16, 877–897. [Google Scholar] [CrossRef]
- Goldenberg, M.M. Multiple sclerosis review. Pharm. Ther. 2012, 37, 175–184. [Google Scholar]
- Wingerchuk, D.M.; Weinshenker, B.G. Disease modifying therapies for relapsing multiple sclerosis. BMJ 2016, 354, i3518. [Google Scholar] [CrossRef] [PubMed]
- Wallin, M.T.; Culpepper, W.J.; Nichols, E.; Bhutta, Z.A.; Gebrehiwot, T.T.; Hay, S.I.; Khalil, I.A.; Krohn, K.J.; Liang, X.; Naghavi, M. Global, regional, and national burden of multiple sclerosis 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 269–285. [Google Scholar] [CrossRef]
- Brownlee, W.J.; Hardy, T.A.; Fazekas, F.; Miller, D.H. Diagnosis of multiple sclerosis: Progress and challenges. Lancet 2017, 389, 1336–1346. [Google Scholar] [CrossRef]
- Baecher-Allan, C.; Kaskow, B.J.; Weiner, H.L. Multiple sclerosis: Mechanisms and immunotherapy. Neuron 2018, 97, 742–768. [Google Scholar] [CrossRef]
- Lauer, K. Environmental risk factors in multiple sclerosis. Expert Rev. Neurother. 2010, 10, 421–440. [Google Scholar] [CrossRef]
- Guerau-de-Arellano, M.; Smith, K.M.; Godlewski, J.; Liu, Y.; Winger, R.; Lawler, S.E.; Whitacre, C.C.; Racke, M.K.; Lovett-Racke, A.E. Micro-RNA dysregulation in multiple sclerosis favours pro-inflammatory T-cell-mediated autoimmunity. Brain 2011, 134, 3578–3589. [Google Scholar] [CrossRef]
- Berrih-Aknin, S.; Le Panse, R. Myasthenia gravis: A comprehensive review of immune dysregulation and etiological mechanisms. J. Autoimmun. 2014, 52, 90–100. [Google Scholar] [CrossRef]
- Belbasis, L.; Bellou, V.; Evangelou, E.; Ioannidis, J.P.; Tzoulaki, I. Environmental risk factors and multiple sclerosis: An umbrella review of systematic reviews and meta-analyses. Lancet Neurol. 2015, 14, 263–273. [Google Scholar] [CrossRef]
- Thompson, A.J.; Baranzini, S.E.; Geurts, J.; Hemmer, B.; Ciccarelli, O. Multiple sclerosis. Lancet 2018, 391, 1622–1636. [Google Scholar] [CrossRef]
- Stiemsma, L.T.; Reynolds, L.A.; Turvey, S.E.; Finlay, B.B. The hygiene hypothesis: Current perspectives and future therapies. Immunotargets Ther. 2015, 4, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Zaccone, P.; Hall, S.W. Helminth infection and type 1 diabetes. Rev. Diabet. Stud. 2012, 9, 272–286. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.E.; Weinstock, J.V. Helminth–host immunological interactions: Prevention and control of immune-mediated diseases. Ann. N. Y. Acad. Sci. 2012, 1247, 83–96. [Google Scholar] [CrossRef]
- Versini, M.; Jeandel, P.-Y.; Bashi, T.; Bizzaro, G.; Blank, M.; Shoenfeld, Y. Unraveling the hygiene hypothesis of helminthes and autoimmunity: Origins, pathophysiology, and clinical applications. BMC Med. 2015, 13, 81. [Google Scholar] [CrossRef]
- Correale, J.; Farez, M.F.; Gaitán, M.I. Environmental factors influencing multiple sclerosis in Latin America. Mult. Scler. J. Exp. Transl. Clin. 2017, 3, 2055217317715049. [Google Scholar] [CrossRef]
- Sewell, D.; Qing, Z.; Reinke, E.; Elliot, D.; Weinstock, J.; Sandor, M.; Fabry, Z. Immunomodulation of experimental autoimmune encephalomyelitis by helminth ova immunization. Int. Immunol. 2003, 15, 59–69. [Google Scholar] [CrossRef]
- Hansen, C.S.; Hasseldam, H.; Bacher, I.H.; Thamsborg, S.M.; Johansen, F.F.; Kringel, H. Trichuris suis secrete products that reduce disease severity in a multiple sclerosis model. Acta Parasitol. 2017, 62, 22–28. [Google Scholar] [CrossRef]
- Tanasescu, R.; Tench, C.R.; Constantinescu, C.S.; Telford, G.; Singh, S.; Frakich, N.; Onion, D.; Auer, D.P.; Gran, B.; Evangelou, N. Hookworm treatment for relapsing multiple sclerosis: A randomized double-blinded placebo-controlled trial. JAMA Neurol. 2020, 77, 1089–1098. [Google Scholar] [CrossRef]
- Zibaei, M.; Ghorbani, B. Toxocariasis and multiple sclerosis: A case-control study in Iran. Neurol. Asia 2014, 109, 283–286. [Google Scholar]
- Söndergaard, H.P.; Theorell, T. A putative role for Toxocara species in the aetiology of multiple sclerosis. Med. Hypotheses 2004, 63, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Rostami, A.; Riahi, S.M.; Hofmann, A.; Ma, G.; Wang, T.; Behniafar, H.; Taghipour, A.; Fakhri, Y.; Spotin, A.; Chang, B.C. Global prevalence of Toxocara infection in dogs. Adv. Parasitol. 2020, 109, 561–583. [Google Scholar] [PubMed]
- Rostami, A.; Sepidarkish, M.; Ma, G.; Wang, T.; Ebrahimi, M.; Fakhri, Y.; Mirjalali, H.; Hofmann, A.; Macpherson, C.N.; Hotez, P.J. Global prevalence of Toxocara infection in cats. Adv. Parasitol. 2020, 109, 615–639. [Google Scholar] [PubMed]
- Ma, G.; Rostami, A.; Wang, T.; Hofmann, A.; Hotez, P.J.; Gasser, R.B. Global and regional seroprevalence estimates for human toxocariasis: A call for action. Adv. Parasitol. 2020, 108, 273–288. [Google Scholar]
- Rostami, A.; Riahi, S.M.; Holland, C.V.; Taghipour, A.; Khalili-Fomeshi, M.; Fakhri, Y.; Omrani, V.F.; Hotez, P.J.; Gasser, R.B. Seroprevalence estimates for toxocariasis in people worldwide: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2019, 13, e0007809. [Google Scholar] [CrossRef]
- Rostami, A.; Ma, G.; Wang, T.; Koehler, A.V.; Hofmann, A.; Chang, B.C.; Macpherson, C.N.; Gasser, R.B. Human toxocariasis—A look at a neglected disease through an epidemiological ‘prism’. Infect. Genet. Evol. 2019, 74, 104002. [Google Scholar] [CrossRef]
- Siyadatpanah, A.; Tabatabaei, F.; Emami, Z.A.; Spotin, A.; Fallah, O.V.; Assadi, M.; Moradi, S.; Rostami, A.; Memari, F.; Hajialiani, F. Parasitic contamination of raw vegetables in Amol, North of Iran. Arch. Clin. Infect. Dis. 2013, 8, e15983. [Google Scholar] [CrossRef]
- Carlin, E.P.; Tyungu, D.L. Toxocara: Protecting pets and improving the lives of people. Adv. Parasitol. 2020, 109, 3–16. [Google Scholar]
- Ma, G.; Holland, C.V.; Wang, T.; Hofmann, A.; Fan, C.-K.; Maizels, R.M.; Hotez, P.J.; Gasser, R.B. Human toxocariasis. Lancet Infect. Dis. 2018, 18, e14–e24. [Google Scholar] [CrossRef]
- Wu, T.; Bowman, D.D. Visceral larval migrans of Toxocara canis and Toxocara cati in non-canid and non-felid hosts. Adv. Parasitol. 2020, 109, 63–88. [Google Scholar]
- Deshayes, S.; Bonhomme, J.; de La Blanchardière, A. Neurotoxocariasis: A systematic literature review. Infection 2016, 44, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Murrell, T.; Harbige, L.; Robinson, I. A review of the aetiology of multiple sclerosis: An ecological approach. Ann. Hum. Biol. 1991, 18, 95–112. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, A. Toxocariasis. BMJ 1970, 3, 663. [Google Scholar] [CrossRef][Green Version]
- Maizels, R.M. Toxocara canis: Molecular basis of immune recognition and evasion. Vet. Parasitol. 2013, 193, 365–374. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.-K.; Holland, C.V.; Loxton, K.; Barghouth, U. Cerebral toxocariasis: Silent progression to neurodegenerative disorders? Clin. Microbiol. Rev. 2015, 28, 663–686. [Google Scholar] [CrossRef]
- Sánchez, S.; Garcıa, H.; Nicoletti, A. Clinical and Magnetic Resonance Imaging Findings of Neurotoxocariasis. Front. Neurol. 2018, 9, 53. [Google Scholar] [CrossRef]
- Jewells, V.L.; Latchaw, R.E. What Can Mimic Multiple Sclerosis? In Seminars in Ultrasound, CT and MRI; WB Saunders: Philadelphia, PA, USA, 2020; Volume 41, pp. 284–295. [Google Scholar]
- Strube, C.; Heuer, L.; Janecek, E. Toxocara spp. infections in paratenic hosts. Vet. Parasitol. 2013, 193, 375–389. [Google Scholar] [CrossRef]
- Cicero, C.E.; Patti, F.; Lo Fermo, S.; Giuliano, L.; Rascunà, C.; Chisari, C.G.; D’Amico, E.; Paradisi, V.; Marin, B.; Preux, P.-M. Lack of association between Toxocara canis and multiple sclerosis: A population-based case–control study. Mult. Scler. J. 2020, 26, 258–259. [Google Scholar] [CrossRef]
- Esfandiari, F.; Mikaeili, F.; Rahmanian, R.; Asgari, Q. Seroprevalence of toxocariasis in multiple sclerosis and rheumatoid arthritis patients in Shiraz city, southern Iran. Clin. Epidemiol. Glob. Health 2019, 8, 158–160. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: Hoboken, NJ, USA, 2019; ISBN 978-1-119-53662-8. [Google Scholar]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Hamza, T.H.; van Houwelingen, H.C.; Stijnen, T. The binomial distribution of meta-analysis was preferred to model within-study variability. J. Clin. Epidemiol. 2008, 61, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Smith, G.D.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Posová, H.; Hrušková, Z.; Havrdová, E.; Kolářová, L. Toxocara spp. seronegativity in Czech patients with early form of multiple sclerosis-clinically isolated syndrome. Epidemiol. Mikrobiol. Imunol. 2017, 66, 124–127. [Google Scholar]
- Kuk, S.; Ozgocmen, S.; Bulut, S. Seroprevalance of Toxocara antibodies in multiple sclerosis and ankylosing spondylitis. Indian J. Med. Sci. 2006, 60, 297–299. [Google Scholar] [CrossRef] [PubMed]
- Khalili, N.; Khalili, N.; Nickhah, A.; Khalili, B. Seroprevalence of anti-Toxocara antibody among multiple sclerosis patients: A case–control study. J. Parasit. Dis. 2020, 44, 145–150. [Google Scholar] [CrossRef]
- Khalilidehkordi, B. Toxocara infection in multiple sclerosis patients in Shahrekord district. In Proceedings of the 9th International Congress in Clinical Laboratories, Tehran, Iran, 21–24 February 2017. [Google Scholar]
- Fleming, J.O.; Cook, T.D. Multiple sclerosis and the hygiene hypothesis. Neurology 2006, 67, 2085–2086. [Google Scholar] [CrossRef]
- La Flamme, A.C.; Ruddenklau, K.; Bäckström, B.T. Schistosomiasis decreases central nervous system inflammation and alters the progression of experimental autoimmune encephalomyelitis. Infect. Immun. 2003, 71, 4996–5004. [Google Scholar] [CrossRef]
- Kaiser, C.; Pion, S.D.; Boussinesq, M. Case-control studies on the relationship between onchocerciasis and epilepsy: Systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2013, 7, e2147. [Google Scholar] [CrossRef]
- Quattrocchi, G.; Nicoletti, A.; Marin, B.; Bruno, E.; Druet-Cabanac, M.; Preux, P.-M. Toxocariasis and epilepsy: Systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2012, 6, e1775. [Google Scholar] [CrossRef] [PubMed]
- Luna, J.; Cicero, C.E.; Rateau, G.; Quattrocchi, G.; Marin, B.; Bruno, E.; Dalmay, F.; Druet-Cabanac, M.; Nicoletti, A.; Preux, P.-M. Updated evidence of the association between toxocariasis and epilepsy: Systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2018, 12, e0006665. [Google Scholar] [CrossRef] [PubMed]
- Strube, C.; Waindok, P.; Raulf, M.-K.; Springer, A. Toxocara-induced neural larva migrans (neurotoxocarosis) in rodent model hosts. Adv. Parasitol. 2020, 109, 189–218. [Google Scholar] [PubMed]
- Burren, C. The distribution of Toxocara larvae in the central nervous system of the mouse. Trans. R. Soc. Trop. Med. Hyg. 1971, 65, 450–453. [Google Scholar] [CrossRef]
- Sprent, J. On the migratory behavior of the larvae of various Ascaris species in white mice: II. Longevity. J. Infect. Dis. 1953, 92, 114–117. [Google Scholar] [CrossRef]
- Cardillo, N.; Rosa, A.; Ribicich, M.; López, C.; Sommerfelt, I. Experimental infection with Toxocara cati in BALB/c mice, migratory behaviour and pathological changes. Zoonoses Public Health 2009, 56, 198–205. [Google Scholar] [CrossRef]
- Janecek, E.; Beineke, A.; Schnieder, T.; Strube, C. Neurotoxocarosis: Marked preference of Toxocara canis for the cerebrum and T. cati for the cerebellum in the paratenic model host mouse. Parasit. Vectors 2014, 7, 194. [Google Scholar] [CrossRef]
- Heuer, L.; Beyerbach, M.; Lühder, F.; Beineke, A.; Strube, C. Neurotoxocarosis alters myelin protein gene transcription and expression. Parasitol. Res. 2015, 114, 2175–2186. [Google Scholar] [CrossRef]
- Fonseca, G.R.e.; Dos Santos, S.V.; Chieffi, P.P.; Paula, F.M.d.; Gryschek, R.C.B.; Lescano, S.A.Z. Experimental toxocariasis in BALB/c mice: Relationship between parasite inoculum and the IgG immune response. Mem. Inst. Oswaldo Cruz 2017, 112, 382–386. [Google Scholar] [CrossRef]
- Epe, C.; Sabel, T.; Schnieder, T.; Stoye, M. The behavior and pathogenicity of Toxacara canis larvae in mice of different strains. Parasitol. Res. 1994, 80, 691–695. [Google Scholar] [CrossRef]
- Nicoletti, A.; Gomez-Puerta, L.A.; Arroyo, G.; Bustos, J.; Gonzalez, A.E.; Garcia, H.H.; Peru, C.W.G.I. Toxocara brain infection in pigs is not associated with visible lesions on brain magnetic resonance imaging. Am. J. Trop. Med. Hyg. 2020, 103, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, A. Toxocariasis. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2013; Volume 114, pp. 217–228. [Google Scholar]
- Dolinsky, Z.S.; Hardy, C.A.; Burright, R.G.; Donovick, P.J. The progression of behavioral and pathological effects of the parasite Toxocara canis in the mouse. Physiol. Behav. 1985, 35, 33–42. [Google Scholar] [CrossRef]
- Springer, A.; Heuer, L.; Janecek-Erfurth, E.; Beineke, A.; Strube, C. Histopathological characterization of Toxocara canis-and T. cati-induced neurotoxocarosis in the mouse model. Parasitol. Res. 2019, 118, 2591–2600. [Google Scholar] [CrossRef] [PubMed]
- Othman, A.A.; Abdel-Aleem, G.A.; Saied, E.M.; Mayah, W.W.; Elatrash, A.M. Biochemical and immunopathological changes in experimental neurotoxocariasis. Mol. Biochem. Parasitol. 2010, 172, 1–8. [Google Scholar] [CrossRef]
- Lassmann, H.; Bradl, M. Multiple sclerosis: Experimental models and reality. Acta Neuropathol. 2017, 133, 223–244. [Google Scholar] [CrossRef]
| Reference | Country | Age Range, or Mean Age ± Standard Deviation (Years) | Subjects with MS | Subjects without MS (Controls) | p-Value | |||
|---|---|---|---|---|---|---|---|---|
| Subjects with MS | Subjects without MS Controls | Number Tested | Positive for Anti-Toxocara IgG Serum Antibodies (%) | Number Tested | Positive for Anti-Toxocara IgG Serum Antibodies (%) | |||
| Kuk et al. [47] | Turkey | 20–54 | 20–50 | 37 | 4 (10.8) | 50 | 1 (2.0) | 0.08 |
| Zibaei and Ghorbani. [20] | Iran | 3–49 | 3–52 | 68 | 10 (14.7) | 70 | 1 (1.4) | 0.004 |
| Khalilidehkordi et al. [49] | Iran | not recorded | not recorded | 70 | 8 (11.4) | 70 | 1 (1.4) | <0.05 |
| Cicero et al. [39] | Italy | 44.6 ± 11.1 | 48.1 ± 15.6 | 132 | 12 (9.1) | 287 | 23 (8.0) | 0.7 |
| Khalili et al. [48] | Iran | 41.2 ± 9.5 | 38.8 ± 7.6 | 70 | 20 (28.5) | 70 | 8 (11.4) | 0.02 |
| Esfandiari et al. [40] | Iran | 11–60 | 11–60 | 96 | 8 (8.3) | 100 | 3 (3.0) | 0.1 |
| Selection | Comparability | Exposure | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Reference | Adequate Case Definition | Representat-Iveness of MS Cases | Selection of Controls | Definition of Controls | Comparability of Cases and Controls on the Basis of Design or Analysis | Ascertainment of Exposure | Same Method of Ascertainment for Cases and Controls | Non-Response Rate | Score |
| Kuk et al. [47] | * | * | * | na | * | * | * | na | 6 |
| Zibaei and Ghorbani. [20] | * | * | * | * | ** | * | * | na | 8 |
| Khalilidehkordi et al. [49] | * | * | * | na | na | * | * | na | 5 |
| Cicero et al. [39] | * | * | * | * | ** | * | * | * | 9 |
| Khalili et al. [48] | * | * | * | na | ** | * | * | na | 7 |
| Esfandiari et al. [40] | * | * | * | * | ** | * | * | na | 8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Taghipour, A.; Rostami, A.; Esfandyari, S.; Aghapour, S.; Nicoletti, A.; Gasser, R.B. “Begging the Question”—Does Toxocara Infection/Exposure Associate with Multiple Sclerosis-Risk? Pathogens 2020, 9, 938. https://doi.org/10.3390/pathogens9110938
Taghipour A, Rostami A, Esfandyari S, Aghapour S, Nicoletti A, Gasser RB. “Begging the Question”—Does Toxocara Infection/Exposure Associate with Multiple Sclerosis-Risk? Pathogens. 2020; 9(11):938. https://doi.org/10.3390/pathogens9110938
Chicago/Turabian StyleTaghipour, Ali, Ali Rostami, Sahar Esfandyari, Saeed Aghapour, Alessandra Nicoletti, and Robin B. Gasser. 2020. "“Begging the Question”—Does Toxocara Infection/Exposure Associate with Multiple Sclerosis-Risk?" Pathogens 9, no. 11: 938. https://doi.org/10.3390/pathogens9110938
APA StyleTaghipour, A., Rostami, A., Esfandyari, S., Aghapour, S., Nicoletti, A., & Gasser, R. B. (2020). “Begging the Question”—Does Toxocara Infection/Exposure Associate with Multiple Sclerosis-Risk? Pathogens, 9(11), 938. https://doi.org/10.3390/pathogens9110938






