Dichotomy in Fatal Outcomes in a Large Cohort of People Living with HTLV-1 in São Paulo, Brazil
Abstract
1. Background
2. Methods
2.1. Population
2.2. Clinical Follow-Up
2.3. Statistical Analysis
2.4. Ethical Issues
3. Results
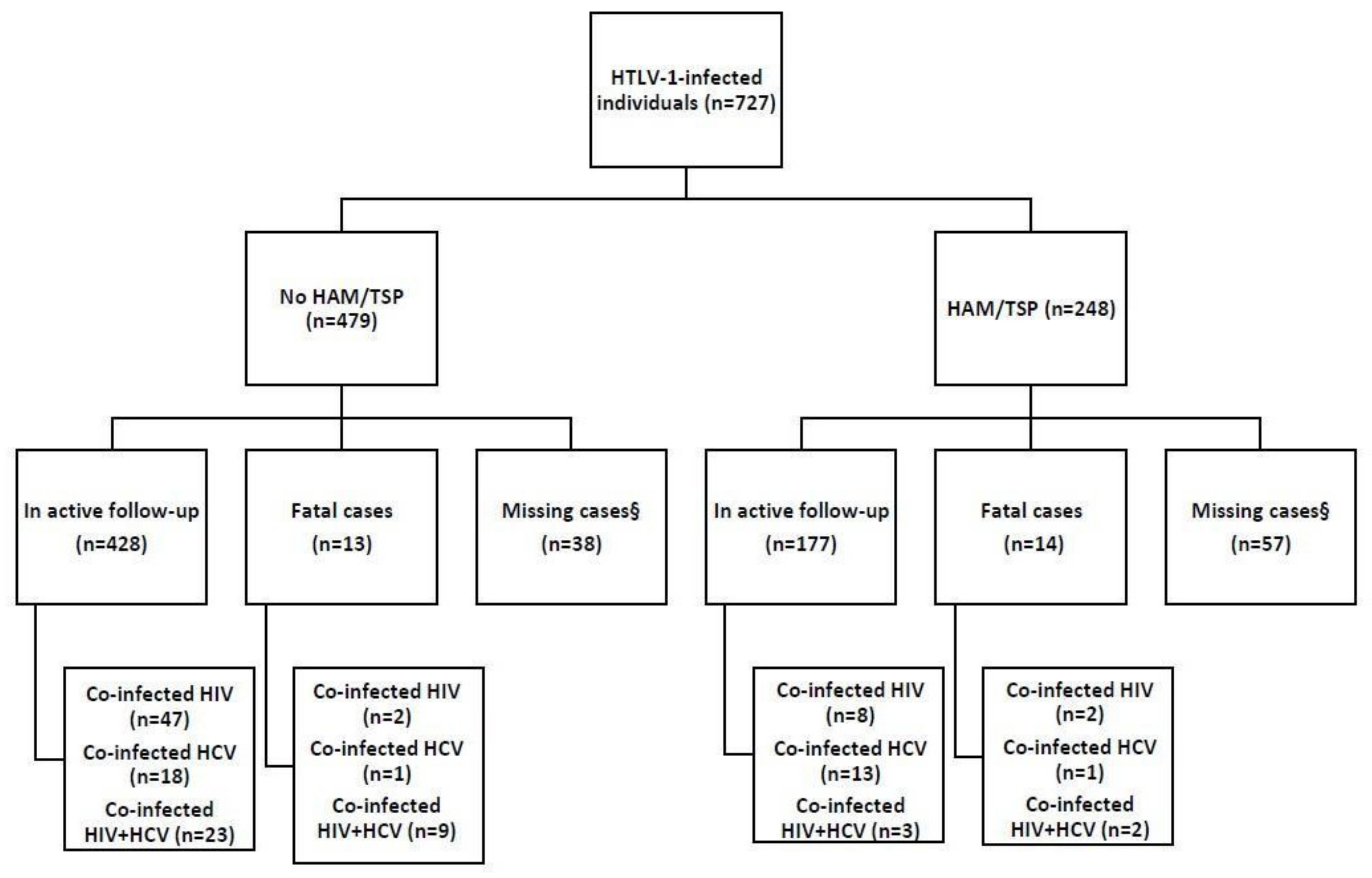
3.1. Study Population
3.2. Clinical Follow-Up
3.3. Survival Analysis and Cox Proportional Hazard Analysis
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Verdonck, K.; Gonzalez, E.; Van Dooren, S.; Vandamme, A.M.; Vanham, G.; Gotuzzo, E. Human T-lymphotropic virus 1: Recent knowledge about an ancient infection. Lancet Infect. Dis. 2007, 7, 266–281. [Google Scholar] [CrossRef]
- Gessain, A.; Barin, F.; Vernant, J.C.; Gout, O.; Maurs, L.; Calender, A.; de Thé, G. Antibodies to human T-lymphotropic virus type-I in patients with tropical spastic paraparesis. Lancet 1985, 326, 407–410. [Google Scholar] [CrossRef]
- Gessain, A.; Cassar, O. Epidemiological aspects and world distribution of HTLV-1 infection. Front. Microbiol. 2012, 3, 388. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.H.; Weiss, S.H.; Brown, L.S.; Mildvan, D.; Shorty, V.; Saravolatz, L.; Chu, A.; Ginzburg, H.M.; Markowitz, N.; Des Jarlais, D.C.; et al. Patterns of HIV-1 and HTLV-I/II in intravenous drug abusers from the middle atlantic and central regions of the USA. J. Infect. Dis. 1990, 162, 347–352. [Google Scholar] [CrossRef] [PubMed]
- De Araujo, A.C.; Casseb, J.S.; Neitzert, E.; de Souza, M.L.; Mammano, F.; Del Mistro, A.; De Rossi, A.; Chieco-Bianchi, L. HTLV-I and HTLV-II infections among HIV-1 seropositive patients in Sao Paulo, Brazil. Eur. J. Epidemiol. 1994, 10, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Casseb, J.; Souza, T.; Pierre-Lima, M.T.; Yeh, E.; Hendry, R.M.; Gallo, D. Testing problems in diagnosing HTLV infection among intravenous drug users with AIDS in Sao Paulo City, Brazil. AIDS Res. Hum. Retroviruses 1997, 13, 1639–1641. [Google Scholar] [CrossRef]
- Paiva, A.M.; Assone, T.; Haziot, M.E.J.; Smid, J.; Fonseca, L.A.M.; Luiz, O.D.C.; de Oliveira, A.C.P.; Casseb, J. Risk factors associated with HTLV-1 vertical transmission in Brazil: Longer breastfeeding, higher maternal proviral load and previous HTLV-1-infected offspring. Sci. Rep. 2018, 8, 7742. [Google Scholar] [CrossRef]
- Rosadas, C.; Taylor, G.P. Mother-to-Child HTLV-1 transmission: Unmet research needs. Front. Microbiol. 2019, 10, 999. [Google Scholar] [CrossRef]
- Olindo, S.; Jeannin, S.; Saint-Vil, M.; Signate, A.; Edimonana-Kaptue, M.; Joux, J.; Merle, H.; Richard, P.; Granjeaud, S.; Cabre, P.; et al. Temporal trends in Human T-Lymphotropic virus 1 (HTLV-1) associated myelopathy/tropical spastic paraparesis (HAM/TSP) incidence in Martinique over 25 years (1986–2010). PLoS Negl. Trop. Dis. 2018, 12, e0006304. [Google Scholar] [CrossRef]
- Assone, T.; Kanashiro, T.M.; Baldassin, M.P.M.; Paiva, A.; Haziot, M.E.; Smid, J.; Oliveira, A.P.; Fonseca, L.A.M.; Norris, P.J.; Casseb, J. In vitro basal T-cell proliferation among asymptomatic Human T cell Leukemia Virus type 1 patients co-infected with hepatitis C and/or Human Immunodeficiency Virus type 1. Braz. J. Infect. Dis. 2018, 22, 106–112. [Google Scholar] [CrossRef]
- Abrahao, M.H.; Lima, R.G.; Netto, E.; Brites, C. Short communication: Human lymphotropic virus type 1 coinfection modulates the synthesis of cytokines by peripheral blood mononuclear cells from HIV type 1-infected individuals. AIDS Res. Hum. Retroviruses 2012, 28, 806–808. [Google Scholar] [CrossRef] [PubMed]
- Brites, C.; Goyanna, F.; Franca, L.G.; Pedroso, C.; Netto, E.M.; Adriano, S.; Sampaio, J.; Harrington, W., Jr. Coinfection by HTLV-I/II is associated with an increased risk of strongyloidiasis and delay in starting antiretroviral therapy for AIDS patients. Braz. J. Infect. Dis. 2011, 15, 6–11. [Google Scholar] [CrossRef]
- Brites, C.; Miranda, F.; Luz, E.; Netto, E.M. Early and successful cART normalizes survival time in HIV-HTLV-1 coinfected patients. Clin. Infect. Dis. 2019, ciz756. [Google Scholar] [CrossRef] [PubMed]
- Van Tienen, C.; Schim van der Loeff, M.; Peterson, I.; Cotten, M.; Andersson, S.; Holmgren, B.; Vincent, T.; de Silva, T.; Rowland-Jones, S.; Aaby, P.; et al. HTLV-1 and HIV-2 infection are associated with increased mortality in a rural West African community. PLoS ONE 2011, 6, e29026. [Google Scholar] [CrossRef] [PubMed]
- Iwata, K.; Ito, S.; Saito, H.; Ito, M.; Nagatomo, M.; Yamasaki, T.; Yoshida, S.; Suto, H.; Tajima, K. Mortality among inhabitants of an HTLV-I endemic area in Japan. Jpn. J. Cancer Res. 1994, 85, 231–237. [Google Scholar] [CrossRef]
- Norrgren, H.; Bamba, S.; Da Silva, Z.J.; Koivula, T.; Andersson, S. Higher mortality in HIV-2/HTLV-1 co-infected patients with pulmonary tuberculosis in Guinea-Bissau, West Africa, compared to HIV-2-positive HTLV-1-negative patients. Int. J. Infect. Dis. 2010, 14 (Suppl. S3), e142–e147. [Google Scholar] [CrossRef]
- Holmgren, B.; da Silva, Z.; Vastrup, P.; Larsen, O.; Andersson, S.; Ravn, H.; Aaby, P. Mortality associated with HIV-1, HIV-2, and HTLV-I single and dual infections in a middle-aged and older population in Guinea-Bissau. Retrovirology 2007, 4, 85. [Google Scholar] [CrossRef]
- Biswas, H.H.; Kaidarova, Z.; Garratty, G.; Gibble, J.W.; Newman, B.H.; Smith, J.W.; Ziman, A.; Fridey, J.L.; Sacher, R.A.; Murphy, E.L.; et al. Increased all-cause and cancer mortality in HTLV-II infection. J. Acquir. Immune Defic. Syndr. 2010, 54, 290–296. [Google Scholar] [CrossRef]
- Einsiedel, L.; Pham, H.; Wilson, K.; Walley, R.; Turpin, J.; Bangham, C.; Gessain, A.; Woodman, R.J. Human T-Lymphotropic Virus type 1c subtype proviral loads, chronic lung disease and survival in a prospective cohort of Indigenous Australians. PLoS Negl. Trop. Dis. 2018, 12, e0006281. [Google Scholar] [CrossRef]
- Schierhout, G.; McGregor, S.; Gessain, A.; Einsiedel, L.; Martinello, M.; Kaldor, J. Association between HTLV-1 infection and adverse health outcomes: A systematic review and meta-analysis of epidemiological studies. Lancet Infect. Dis. 2020, 20, 133–143. [Google Scholar] [CrossRef]
- Assone, T.; Malta, F.M.; Bakkour, S.; Montalvo, L.; Paiva, A.M.; Smid, J.; de Oliveira, A.C.P.; de Toledo Goncalves, F.; do Carmo Luiz, O.; Fonseca, L.A.M.; et al. Polymorphisms in HLA-C and KIR alleles are not associated with HAM/TSP risk in HTLV-1-infected subjects. Virus Res. 2018, 244, 71–74. [Google Scholar] [CrossRef] [PubMed]
- Novoa, P.; Penalva de Oliveira, A.C.; Posada Vergara, M.P.; da Silva Duarte, A.J.; Casseb, J. Molecular characterization of human T-cell lymphotropic virus type 2 (HTLV-II) from people living in urban areas of Sao Paulo city: Evidence of multiple subtypes circulation. J. Med. Virol. 2007, 79, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. BioMed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Castro-Costa, C.M.; Araujo, A.Q.; Menna-Barreto, M.; Penalva-de-Oliveira, A.C. Guide of clinical management of HTLV patient: Neurological aspects. Rquivos Neuro-Psiquiatr. 2005, 63, 548–551. [Google Scholar] [CrossRef][Green Version]
- Sato, T.; Yagishita, N.; Tamaki, K.; Inoue, E.; Hasegawa, D.; Nagasaka, M.; Suzuki, H.; Araya, N.; Coler-Reilly, A.; Hasegawa, Y.; et al. Proposal of classification criteria for HTLV-1-associated myelopathy/tropical spastic paraparesis disease activity. Front. Microbiol. 2018, 9, 1651. [Google Scholar] [CrossRef]
- Haziot, M.E.; Gascon, M.R.; Assone, T.; Fonseca, L.A.M.; Luiz, O.D.C.; Smid, J.; Paiva, A.M.; Marcusso, R.; de Oliveira, A.C.P.; Casseb, J. Detection of clinical and neurological signs in apparently asymptomatic HTLV-1 infected carriers: Association with high proviral load. PLoS Negl. Trop. Dis. 2019, 13, e0006967. [Google Scholar] [CrossRef]
- Bellon, M.; Nicot, C. Telomere dynamics in immune senescence and exhaustion triggered by chronic viral infection. Viruses 2017, 9, 289. [Google Scholar] [CrossRef]
- Gascon, M.R.P.; Mellao, M.A.; Mello, S.H.; Negrao, R.M.; Casseb, J.; Oliveira, A.C.P. The impact of urinary incontinence on the quality of life and on the sexuality of patients with HAM/TSP. Braz. J. Infect. Dis. 2018, 22, 288–293. [Google Scholar] [CrossRef]
- Arisawa, K.; Sobue, T.; Yoshimi, I.; Soda, M.; Shirahama, S.; Doi, H.; Katamine, S.; Saito, H.; Urata, M. Human T-lymphotropic virus type-I infection, survival and cancer risk in southwestern Japan: A prospective cohort study. Cancer Causes Control 2003, 14, 889–896. [Google Scholar] [CrossRef]
- Martin, F.; Fedina, A.; Youshya, S.; Taylor, G.P. A 15-year prospective longitudinal study of disease progression in patients with HTLV-1 associated myelopathy in the UK. J. Neurol. Neurosurg. Psychiatry 2010, 81, 1336–1340. [Google Scholar] [CrossRef]
- McKibben, M.J.; Seed, P.; Ross, S.S.; Borawski, K.M. Urinary tract infection and neurogenic bladder. Urol Clin. N. Am. 2015, 42, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Jaul, E.; Factor, H.; Karni, S.; Schiffmiller, T.; Meiron, O. Spasticity and dementia increase the risk of pressure ulcers. Int. Wound J. 2019, 16, 847–851. [Google Scholar] [CrossRef] [PubMed]
- Jaul, E.; Rosenzweig, J.P. A Retrospective study of the impact of pressure ulcers on survival in elderly persons with chronic diseases. Ostomy Wound Manag. 2017, 63, 26–32. [Google Scholar]
- Jaul, E.; Calderon-Margalit, R. Systemic factors and mortality in elderly patients with pressure ulcers. Int. Wound J. 2015, 12, 254–259. [Google Scholar] [CrossRef]
- Andrade, R.; Tanajura, D.; Santana, D.; Santos, D.; Carvalho, E.M. Association between urinary symptoms and quality of life in HTLV-1 infected subjects without myelopathy. Int. Braz. J. Urol 2013, 39, 861–866. [Google Scholar] [CrossRef][Green Version]
- Casseb, J.; Fonseca, L.A.M.; Duarte, A. Is it possible to control HIV infection in a middle-income country through a multidisciplinary approach? AIDS Res. Hum. Retrovir. 2018, 34, 165–167. [Google Scholar] [CrossRef]
- Tate, J.P.; Sterne, J.A.C.; Justice, A.C.; Veterans Aging Cohort, S.; The antiretroviral therapy cohort, C. albumin, white blood cell count, and body mass index improve discrimination of mortality in HIV-positive individuals. AIDS 2019, 33, 903–912. [Google Scholar] [CrossRef]
- Rezende Machado de Sousa, L.; Saint Ville, A.; Maria Segall-Correa, A.; Melgar-Quinonez, H. Health inequalities and well-being in times of financial and political crisis in Brazil, a case study. Glob. Public Health 2019, 14, 1815–1828. [Google Scholar] [CrossRef]
- Pelissari, D.M.; Rocha, M.S.; Bartholomay, P.; Sanchez, M.N.; Duarte, E.C.; Arakaki-Sanchez, D.; Dantas, C.O.; Jacobs, M.G.; Andrade, K.B.; Codenotti, S.B.; et al. Identifying socioeconomic, epidemiological and operational scenarios for tuberculosis control in Brazil: An ecological study. BMJ Open 2018, 8, e018545. [Google Scholar] [CrossRef]
- Martin, F.; Tagaya, Y.; Gallo, R. Time to eradicate HTLV-1: An open letter to WHO. Lancet 2018, 391, 1893–1894. [Google Scholar] [CrossRef]

| Variable | Category | HTLV-1 n (%) | Asymptomatic n (%) | HAM/TSP n (%) | OR | 95% CI | p Value |
|---|---|---|---|---|---|---|---|
| Age | Mean (SD) | 52.2 (10.5) | 49.4 (9.2) | 54.6 (11.2) | 0.23 a | ||
| (years) | Median | 51.5 | 47 | 54.5 | 0.29 a | ||
| Gender | Male | 15 (55.6) | 9 (69.2) | 6 (42.9) | 2.667 | 0.973–4.430 | 0.17 b |
| Female | 12 (44.4) | 4 (30.8) | 8 (57.1) | ||||
| Coinfected | HIV | 4 (14.8) | 2 (15.4) | 2 (14.3) | 1.543 | 0.943–2.997 | 0.39 b |
| HCV | 4 (14.8) | 1 (7.7) | 3 (21.4) | 2.087 | 0.936–11.913 | 0.32 b | |
| HCV/HIV | 11 (40.7) | 9 (69.2) | 2 (14.3) | 2.521 | 1.141–14.912 | 0.003 b | |
| Cause of death c | Neoplasia | 3 (11.1) | 2 (15.4) | 1 (7.1) | 1.625 | 0.315–8.395 | 0.60 a,* |
| Sepsis | 10 (37.0) | 6 (46.2) | 4 (28.6) | 1.471 | 0.624–3.465 | 0.44 b | |
| Decubitus Ulcers | 8 (29.6) | ------- | 8 (57.1) | 0.002 b | |||
| Urinary Tract Infection | 10 (37.0) | 4 (30.8) | 7 (50.0) | 1.688 | 0.548–3.196 | 0.24 a,* | |
| AIDS-related § | 7 (25.9) | 6 (46.2) | 1 (7.1) | 4.550 | 0.721–3.196 | 0.033 b |
| Variable | Category | Survivors n (%) (n = 605) | Fatal Cases n (%) (n = 27) | HR | 95% CI | p Value a |
|---|---|---|---|---|---|---|
| Age | ≤52 (mean) | 51.9 (13.8) | 52.2 (10.5) | 1.307 | 0.597–2.859 | 0.50 |
| ≤53 (median) | 53 | 51.5 | 1.589 | 0.731–3.454 | 0.24 | |
| Gender | Male | 233 (38.5) | 15 (55.6) | 1.933 | 0.905–4.130 | 0.089 |
| Female | 372 (61.5) | 12 (44.4) | ||||
| Coinfection | HCV | 67 (11.1) | 4 (14.8) | 5.194 | 2.438–11.065 | <0.001 |
| HIV | 80 (13.2) | 4 (14.8) | 5.117 | 2.388–10.963 | <0.001 | |
| HCV/HIV | 26 (4.3) | 11 (40.7) | 7.476 | 3.458–16.161 | <0.001 | |
| Diagnosis | HAM/TSP | 177 (29.3) | 14 (51.9) | 2.188 | 1.023–4.678 | 0.043 |
| Variable | Category | HR | 95% CI | p Value a |
|---|---|---|---|---|
| Coinfection | HCV/HIV | 15.076 | 5.501–41.318 | <0.001 |
| Diagnosis | HAM/TSP | 5.030 | 1.959–12.911 | 0.001 |
| Gender | Male | 1.161 | 0.510–2.643 | 0.13 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marcusso, R.M.N.; Van Weyenbergh, J.; de Moura, J.V.L.; Dahy, F.E.; de Moura Brasil Matos, A.; Haziot, M.E.J.; Vidal, J.E.; Fonseca, L.A.M.; Smid, J.; Assone, T.; et al. Dichotomy in Fatal Outcomes in a Large Cohort of People Living with HTLV-1 in São Paulo, Brazil. Pathogens 2020, 9, 25. https://doi.org/10.3390/pathogens9010025
Marcusso RMN, Van Weyenbergh J, de Moura JVL, Dahy FE, de Moura Brasil Matos A, Haziot MEJ, Vidal JE, Fonseca LAM, Smid J, Assone T, et al. Dichotomy in Fatal Outcomes in a Large Cohort of People Living with HTLV-1 in São Paulo, Brazil. Pathogens. 2020; 9(1):25. https://doi.org/10.3390/pathogens9010025
Chicago/Turabian StyleMarcusso, Rosa Maria N., Johan Van Weyenbergh, João Victor Luisi de Moura, Flávia Esper Dahy, Aline de Moura Brasil Matos, Michel E. J. Haziot, Jose E. Vidal, Luiz Augusto M. Fonseca, Jerusa Smid, Tatiane Assone, and et al. 2020. "Dichotomy in Fatal Outcomes in a Large Cohort of People Living with HTLV-1 in São Paulo, Brazil" Pathogens 9, no. 1: 25. https://doi.org/10.3390/pathogens9010025
APA StyleMarcusso, R. M. N., Van Weyenbergh, J., de Moura, J. V. L., Dahy, F. E., de Moura Brasil Matos, A., Haziot, M. E. J., Vidal, J. E., Fonseca, L. A. M., Smid, J., Assone, T., Casseb, J., & de Oliveira, A. C. P. (2020). Dichotomy in Fatal Outcomes in a Large Cohort of People Living with HTLV-1 in São Paulo, Brazil. Pathogens, 9(1), 25. https://doi.org/10.3390/pathogens9010025





