Atypical, Yet Not Infrequent, Infections with Neisseria Species
Abstract
1. Introduction
2. Atypical Infections with N. gonorrhoeae
2.1. Disseminated Gonococcal Infections (DGIs)
2.2. Gonococcal Oral and Nasopharyngeal Infections
2.3. Gonococcal Ophthalmia
2.4. Gonococcal Mastitis
3. Atypical Infections with N. meningitidis
3.1. Meningococcal Genitourinary Tract Infections
3.2. Meningococcal Ophthalmia
4. Infections with Commensal Neisseria Species
4.1. Endocarditis
4.2. Meningitis and Septicemia
4.3. Respiratory Tract Infections
4.4. Genitourinary Tract Infections
4.5. Other Infections, Epidemiology, and Factors Possibly Influencing Disease Development
5. Antimicrobial Treatment of Typical and Atypical Neisseria Infections
6. Discussion
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Liu, G.; Tang, C.M.; Exley, R.M. Non-pathogenic Neisseria: Members of an abundant, multi-habitat, diverse genus. Microbiology 2015, 161, 1297–1312. [Google Scholar] [CrossRef] [PubMed]
- Bennet, J.S.; Bratcher, H.B.; Brehony, C.; Harrison, O.B.; Maiden, M.C.J. The genus Neisseria. In Prokaryotes; Springer: Berlin/Heidelberg, Germany, 2014; pp. 881–900. [Google Scholar]
- Seifert, H.S. Location, Location-Commensalism, Damage and Evolution of the Pathogenic Neisseria. J. Mol. Biol. 2019, 431, 3010–3014. [Google Scholar] [CrossRef] [PubMed]
- Lu, Q.F.; Cao, D.M.; Su, L.L.; Li, S.B.; Ye, G.B.; Zhu, X.Y.; Wang, J.P. Genus-Wide Comparative Genomics Analysis of Neisseria to Identify New Genes Associated with Pathogenicity and Niche Adaptation of Neisseria Pathogens. Int. J. Genom. 2019, 2019, 6015730. [Google Scholar] [CrossRef] [PubMed]
- Neisser, A.L. Über eine der Gonorrhoe eigentümliche Micrococcusform. Cent. Med. Wissensch 1879, 17, 497–500. [Google Scholar]
- Quillin, S.J.; Seifert, H.S. Neisseria gonorrhoeae host adaptation and pathogenesis. Nat. Rev. Microbiol. 2018, 16, 226–240. [Google Scholar] [CrossRef]
- Geisler, W.M.; Yu, S.; Hook, E.W., 3rd. Chlamydial and gonococcal infection in men without polymorphonuclear leukocytes on gram stain: Implications for diagnostic approach and management. Sex. Transm. Dis. 2005, 32, 630–634. [Google Scholar] [CrossRef]
- Jordan, S.J.; Schwebke, J.R.; Aaron, K.J.; Van Der Pol, B.; Hook, E.W., 3rd. Meatal Swabs Contain Less Cellular Material and Are Associated with a Decrease in Gram Stain Smear Quality Compared to Urethral Swabs in Men. J. Clin. Microbiol. 2017, 55, 2249–2254. [Google Scholar] [CrossRef][Green Version]
- Xiong, M.; Lan, L.; Feng, T.; Zhao, G.; Wang, F.; Hong, F.; Wu, X.; Zhang, C.; Wen, L.; Liu, A.; et al. Analysis of the sex ratio of reported gonorrhoea incidence in Shenzhen, China. BMJ Open 2016, 6, e009629. [Google Scholar] [CrossRef]
- Stevens, J.S.; Criss, A.K. Pathogenesis of Neisseria gonorrhoeae in the female reproductive tract: Neutrophilic host response, sustained infection, and clinical sequelae. Curr. Opin. Hematol. 2018, 25, 13–21. [Google Scholar] [CrossRef]
- Densen, P. Interaction of complement with Neisseria meningitidis and Neisseria gonorrhoeae. Clin. Microbiol. Rev. 1989, 2, S11–S17. [Google Scholar] [CrossRef]
- Ngampasutadol, J.; Ram, S.; Gulati, S.; Agarwal, S.; Li, C.; Visintin, A.; Monks, B.; Madico, G.; Rice, P.A. Human factor H interacts selectively with Neisseria gonorrhoeae and results in species-specific complement evasion. J. Immunol. 2008, 180, 3426–3435. [Google Scholar] [CrossRef] [PubMed]
- Sadarangani, M.; Pollard, A.J.; Gray-Owen, S.D. Opa proteins and CEACAMs: Pathways of immune engagement for pathogenic Neisseria. FEMS Microbiol. Rev. 2011, 35, 498–514. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.W.; Oakhill, J.S. Transferrin-mediated iron acquisition by pathogenic Neisseria. Biochem. Soc. Trans. 2002, 30, 705–707. [Google Scholar] [CrossRef] [PubMed]
- Noinaj, N.; Buchanan, S.K.; Cornelissen, C.N. The transferrin-iron import system from pathogenic Neisseria species. Mol. Microbiol. 2012, 86, 246–257. [Google Scholar] [CrossRef] [PubMed]
- Schoen, C.; Blom, J.; Claus, H.; Schramm-Gluck, A.; Brandt, P.; Muller, T.; Goesmann, A.; Joseph, B.; Konietzny, S.; Kurzai, O.; et al. Whole-genome comparison of disease and carriage strains provides insights into virulence evolution in Neisseria meningitidis. Proc. Natl. Acad. Sci. USA 2008, 105, 3473–3478. [Google Scholar] [CrossRef] [PubMed]
- Weichselbaum, A. Über die atiologie der akuten meningitis cerebrospinalis. Fortschritte der Medizin 1887, 5, 573–583. [Google Scholar]
- Weichselbaum, A. Über die atiologie der akuten meningitis cerebrospinalis. Fortschritte der Medizin 1887, 5, 620–626. [Google Scholar]
- Jolley, K.A.; Wilson, D.J.; Kriz, P.; McVean, G.; Maiden, M.C. The influence of mutation, recombination, population history, and selection on patterns of genetic diversity in Neisseria meningitidis. Mol. Biol. Evol. 2005, 22, 562–569. [Google Scholar] [CrossRef]
- Brandtzaeg, P.; van Deuren, M. Classification and pathogenesis of meningococcal infections. Methods Mol. Biol. 2012, 799, 21–35. [Google Scholar] [CrossRef]
- Stephens, D.S.; Greenwood, B.; Brandtzaeg, P. Epidemic meningitis, meningococcaemia, and Neisseria meningitidis. Lancet 2007, 369, 2196–2210. [Google Scholar] [CrossRef]
- Campbell, H.; Parikh, S.R.; Borrow, R.; Kaczmarski, E.; Ramsay, M.E.; Ladhani, S.N. Presentation with gastrointestinal symptoms and high case fatality associated with group W meningococcal disease (MenW) in teenagers, England, July 2015 to January 2016. Euro Surveill. 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Carrascosa, M.F.; Casuso-Saenz, E.; Salcines-Caviedes, J.R. Neisseria meningitidis cellulitis. Int. J. Infect. Dis. 2012, 16, e760. [Google Scholar] [CrossRef] [PubMed]
- Russcher, A.; Fanoy, E.; van Olden, G.D.J.; Graafland, A.D.; van der Ende, A.; Knol, M.J. Necrotising fasciitis as atypical presentation of infection with emerging Neisseria meningitidis serogroup W (MenW) clonal complex 11, the Netherlands, March 2017. Euro Surveill. 2017, 22, 30549. [Google Scholar] [CrossRef] [PubMed]
- Rowley, J.; Vander Hoorn, S.; Korenromp, E.; Low, N.; Unemo, M.; Abu-Raddad, L.J.; Chico, R.M.; Smolak, A.; Newman, L.; Gottlieb, S.; et al. Chlamydia, gonorrhoea, trichomoniasis and syphilis: Global prevalence and incidence estimates, 2016. Bull. World Health Organ. 2019, 97, 548P–562P. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, R.; Bai, X.; Borrow, R.; Caugant, D.A.; Carlos, J.; Ceyhan, M.; Christensen, H.; Climent, Y.; De Wals, P.; Dinleyici, E.C.; et al. The Global Meningococcal Initiative meeting on prevention of meningococcal disease worldwide: Epidemiology, surveillance, hypervirulent strains, antibiotic resistance and high-risk populations. Expert. Rev. Vaccines 2019, 18, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Schielke, S.; Frosch, M.; Kurzai, O. Virulence determinants involved in differential host niche adaptation of Neisseria meningitidis and Neisseria gonorrhoeae. Med. Microbiol. Immunol. 2010, 199, 185–196. [Google Scholar] [CrossRef]
- Givan, K.F.; Keyl, A. The isolation of Neisseria species from unusual sites. Can. Med. Assoc. J. 1974, 111, 1077–1079. [Google Scholar]
- Gregory, J.E.; Abramson, E. Meningococci in vaginitis. Am. J. Dis. Child. 1971, 121, 423. [Google Scholar] [CrossRef]
- Katz, A.R.; Chasnoff, R.; Komeya, A.; Lee, M.V. Neisseria meningitidis urethritis: A case report highlighting clinical similarities to and epidemiological differences from gonococcal urethritis. Sex. Transm. Dis. 2011, 38, 439–441. [Google Scholar] [CrossRef]
- Lee, J.S.; Choi, H.Y.; Lee, J.E.; Lee, S.H.; Oum, B.S. Gonococcal keratoconjunctivitis in adults. Eye (Lond.) 2002, 16, 646–649. [Google Scholar] [CrossRef]
- Odegaard, K.; Gundersen, T. Gonococcal pharyngeal infection. Br. J. Vener. Dis. 1973, 49, 350–352. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.P. The pathogenic potential of commensal species of Neisseria. J. Clin. Pathol. 1983, 36, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Korting, H.C. Uncomplicated gonorrhea and disseminated gonococcal infections—Clinical aspects, diagnosis and therapy. Urol. A 1987, 26, 237–245. [Google Scholar]
- Masi, A.T.; Eisenstein, B.I. Disseminated gonococcal infection (DGI) and gonococcal arthritis (GCA): II. Clinical manifestations, diagnosis, complications, treatment, and prevention. Semin. Arthritis Rheum. 1981, 10, 173–197. [Google Scholar] [CrossRef]
- Bardin, T. Gonococcal arthritis. Best Pract. Res. Clin. Rheumatol. 2003, 17, 201–208. [Google Scholar] [CrossRef]
- Rice, P.A. Gonococcal arthritis (disseminated gonococcal infection). Infect. Dis. Clin. N. Am. 2005, 19, 853–861. [Google Scholar] [CrossRef]
- Henderson, G.; Ritchie, W.T. Gonococcal meningitis. Rev. Neurol. Psychiatr. 1909, 7, 75–87. [Google Scholar]
- Home, E. Practical Observations on the Treatment of Strictures in the Urethra and in the Esophagus, London, 3rd ed.; Everard Home: London, UK, 1805; Volume 11, pp. 271–278. [Google Scholar]
- Smith, D. Gonococcal meningitis. Lancet 1922, 1, 1217. [Google Scholar] [CrossRef]
- Newman, A.B. The prognosis in gonococcal endocarditis. Review of literature and report of case with spontaneous recovery. Am. Heart J. 1933, 8, 821–833. [Google Scholar] [CrossRef]
- Wall, T.C.; Peyton, R.B.; Corey, G.R. Gonococcal endocarditis: A new look at an old disease. Medicine (Baltim.) 1989, 68, 375–380. [Google Scholar] [CrossRef]
- Thayer, W.S.; Blumer, G. Ulcerative endocarditis due to the gonococcus: Gonococcal septicemia. Johns Hopkins Hosp. Bull. 1896, 7, 57. [Google Scholar]
- Thayer, W.S.; Lazear, J.W. A Second Case of Gonorrhoeal Septicaemia and Ulcerative Endocarditis with Observations Upon the Cardiac Complications of Gonorrhoea. J. Exp. Med. 1899, 4, 81–116. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shetty, A.; Ribeiro, D.; Evans, A.; Linnane, S. Gonococcal endocarditis: A rare complication of a common disease. J. Clin. Pathol. 2004, 57, 780–781. [Google Scholar] [CrossRef] [PubMed]
- Schoolnik, G.K.; Buchanan, T.M.; Holmes, K.K. Gonococci causing disseminated gonococcal infection are resistant to the bactericidal action of normal human sera. J. Clin. Investig. 1976, 58, 1163–1173. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.F.; Israel, K.S.; Petersen, B.H. Bactericidal and opsonic activity against Neisseria gonorrhoeae in sera from patients with disseminated gonococcal infection. J. Infect. Dis. 1976, 134, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Petersen, B.H.; Graham, J.A.; Brooks, G.F. Human deficiency of the eighth component of complement. The requirement of C8 for serum Neisseria gonorrhoeae bactericidal activity. J. Clin. Investig. 1976, 57, 283–290. [Google Scholar] [CrossRef]
- Alexander, E.R. Gonorrhea in the newborn. Ann. N. Y. Acad. Sci. 1988, 549, 180–186. [Google Scholar] [CrossRef]
- Nie, S.; Wu, Y.; Huang, L.; Pincus, D.; Tang, Y.W.; Lu, X. Gonococcal endocarditis: A case report and literature review. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 23–27. [Google Scholar] [CrossRef]
- Beatrous, S.V.; Grisoli, S.B.; Riahi, R.R.; Matherne, R.J.; Matherne, R.J. Cutaneous manifestations of disseminated gonococcemia. Dermatol. Online J. 2017, 23. [Google Scholar]
- Cowan, L. Gonococcal ulceration of the tongue in the gonococcal dermatitis syndrome. Br. J. Vener. Dis. 1969, 45, 228–231. [Google Scholar] [CrossRef][Green Version]
- Ghosn, S.H.; Kibbi, A.G. Cutaneous gonococcal infections. Clin. Dermatol. 2004, 22, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Bradford, W.L.; Kelley, H.W. Gonococcic miningitis in a new born infant with review of the literature. Am. J. Dis. Child. 1933, 46, 543–549. [Google Scholar] [CrossRef]
- Austin, T.W.; Yang, W.; Pattison, F.M. Oropharyngeal gonorrhea: Disseminated gonococcal disease. Can. Med. Assoc. J. 1977, 117, 438. [Google Scholar] [PubMed]
- Stolz, E.; Schuller, J. Gonococcal oro-and nasopharyngeal infection. Br. J. Vener. Dis. 1974, 50, 104–108. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yavelow, S.L.; Wiznia, A.; Brennessel, D.J.; Glaser, J.H. Disseminated gonorrhoea from a pharyngeal infection in a prepubertal child. Int. J. Pediatr. Otorhinolaryngol. 1984, 7, 297–300. [Google Scholar] [CrossRef]
- Ratnatunga, C.S. Gonococcal pharyngitis. Br. J. Vener. Dis. 1972, 48, 184–186. [Google Scholar] [CrossRef][Green Version]
- Wiesner, P.J.; Tronca, E.; Bonin, P.; Pedersen, A.H.; Holmes, K.K. Clinical spectrum of pharyngeal gonococcal infection. N. Engl. J. Med. 1973, 288, 181–185. [Google Scholar] [CrossRef]
- Fiumara, N.J.; Wise, H.M., Jr.; Many, M. Gonorrheal pharyngitis. N. Engl. J. Med. 1967, 276, 1248–1250. [Google Scholar] [CrossRef]
- Schaefer, R.A.; Enzenauer, R.J.; Pruitt, A.; Corpe, R.S. Acute gonococcal flexor tenosynovitis in an adolescent male with pharyngitis. A case report and literature review. Clin. Orthop. Relat. Res. 1992, 281, 212–215. [Google Scholar]
- Woods, C.R. Gonococcal infections in neonates and young children. Semin. Pediatr. Infect. Dis. 2005, 16, 258–270. [Google Scholar] [CrossRef]
- Bro-Jorgensen, A.; Jensen, T. Gonococcal tonsillar infections. Br. Med. J. 1971, 4, 660–661. [Google Scholar] [CrossRef] [PubMed]
- Diefenbach, W.C. Gonorrheal parotitis. Oral. Surg. Oral. Med. Oral. Pathol. 1953, 6, 974–975. [Google Scholar] [CrossRef]
- Lessing, J.N.; Slingsby, T.J.; Betz, M. Hyperacute Gonococcal Keratoconjunctivitis. J. Gen. Intern. Med. 2019, 34, 477–478. [Google Scholar] [CrossRef] [PubMed]
- Rothenberg, R. Ophthalmia neonatorum due to Neisseria gonorrhoeae: Prevention and treatment. Sex. Transm. Dis. 1979, 6, 187–191. [Google Scholar] [CrossRef] [PubMed]
- Costumbrado, J.; Ghassemzadeh, S. Gonococcal Conjunctivitis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Diener, B. Cesarean section complicated by gonococcal ophthalmia neonatorum. J. Fam. Pract. 1981, 13, 739–743. [Google Scholar]
- Jacobsen, T.; Knudsen, J.D.; Weis, N.M. [Gonorrheal ophthalmia neonatorum in a premature infant delivered by cesarean section]. Ugeskr. Laeger 1991, 153, 2571. [Google Scholar] [PubMed]
- Strand, C.L.; Arango, V.A. Gonococcal ophthalmia neonatorum after delivery by cesarean section: Report of a case. Sex. Transm. Dis. 1979, 6, 77–78. [Google Scholar] [CrossRef]
- Belga, S.; Gratrix, J.; Smyczek, P.; Bertholet, L.; Read, R.; Roelofs, K.; Singh, A.E. Gonococcal Conjunctivitis in Adults: Case Report and Retrospective Review of Cases in Alberta, Canada, 2000–2016. Sex. Transm. Dis. 2019, 46, 47–51. [Google Scholar] [CrossRef]
- McAnena, L.; Knowles, S.J.; Curry, A.; Cassidy, L. Prevalence of gonococcal conjunctivitis in adults and neonates. Eye (Lond.) 2015, 29, 875–880. [Google Scholar] [CrossRef]
- Varady, E.; Nsanze, H.; Slattery, T. Gonococcal scalp abscess in a neonate delivered by caesarean section. Sex. Transm. Infect. 1998, 74, 451. [Google Scholar] [CrossRef]
- Bodsworth, N.J.; Price, R.; Nelson, M.J. A case of gonococcal mastitis in a male. Genitourin. Med. 1993, 69, 222–223. [Google Scholar] [CrossRef] [PubMed]
- Ceniceros, A.; Galen, B.; Madaline, T. Gonococcal breast abscess. IDCases 2019, 18, e00620. [Google Scholar] [CrossRef] [PubMed]
- Bateman, A.C. Unusual cause of a wound infection. J. Appl. Lab. Med. 2017, 2, 444–448. [Google Scholar] [CrossRef]
- Pendle, S.; Barnes, T. Neisseria gonorrhoeae isolated from an unexpected site. Sex. Health 2016, 13, 593–594. [Google Scholar] [CrossRef]
- Lewis, J.F.; Alexander, J.J. Isolation of Neisseria meningitidis from the vagina and cervix. Am. J. Clin. Pathol. 1974, 61, 216–217. [Google Scholar] [CrossRef]
- Jones, R.N.; Slepack, J.; Eades, A. Fatal neonatal meningococcal meningitis. Association with maternal cervical-vaginal colonization. JAMA 1976, 236, 2652–2653. [Google Scholar] [CrossRef]
- Sunderland, W.A.; Harris, H.H.; Spence, D.A.; Lawson, H.W. Meningococcemia in a newborn infant whose mother had meningococcal vaginitis. J. Pediatr. 1972, 81, 856. [Google Scholar] [CrossRef]
- Chacon-Cruz, E.; Alvelais-Palacios, J.A.; Rodriguez-Valencia, J.A.; Lopatynsky-Reyes, E.Z.; Volker-Soberanes, M.L.; Rivas-Landeros, R.M. Meningococcal Neonatal Purulent Conjunctivitis/Sepsis and Asymptomatic Carriage of N. meningitidis in Mother’s Vagina and Both Parents’ Nasopharynx. Case Rep. Infect. Dis. 2017, 2017, 6132857. [Google Scholar] [CrossRef]
- Murray, E.G.D. Meningococcus infections of the male urogenital tract and the liability to confusion with gonococcus. Urol Cutan. Rev. 1939, 43, 739–741. [Google Scholar]
- Givan, K.F.; Thomas, B.W.; Johnston, A.G. Isolation of Neisseria meningitidis from the urethra, cervix, and anal canal: Further observations. Br. J. Vener. Dis. 1977, 53, 109–112. [Google Scholar] [CrossRef]
- Volk, J.; Kraus, S.J. Asymptomatic meningococcal urethritis. Possible protective value against gonococcal infection by bacteriocin production. Br. J. Vener. Dis. 1973, 49, 511–512. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carpenter, C.M.; Charles, R. Isolation of Meningococcus from the Genitourinary Tract of Seven Patients. Am. J. Public Health Nations Health 1942, 32, 640–643. [Google Scholar] [CrossRef] [PubMed]
- Faur, Y.C.; Wilson, M.E.; May, P.S. Isolation of N. meningitidis from patients in a gonorrhea screen program: A four-year survey in New York City. Am. J. Public Health 1981, 71, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Kanemitsu, N.; Hayashi, I.; Satoh, N.; Hayakawa, T.; Mitsuya, H.; Hayase, Y.; Hiramoto, K.; Kojima, M. Acute urethritis caused by Neisseria meningitidis. Int. J. Urol. 2003, 10, 346–347. [Google Scholar] [CrossRef] [PubMed]
- Harrison, O.B.; Cole, K.; Peters, J.; Cresswell, F.; Dean, G.; Eyre, D.W.; Paul, J.; Maiden, M.C. Genomic analysis of urogenital and rectal Neisseria meningitidis isolates reveals encapsulated hyperinvasive meningococci and coincident multidrug-resistant gonococci. Sex. Transm. Infect. 2017, 93, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Winterscheid, K.K.; Whittington, W.L.; Roberts, M.C.; Schwebke, J.R.; Holmes, K.K. Decreased susceptibility to penicillin G and Tet M plasmids in genital and anorectal isolates of Neisseria meningitidis. Antimicrob. Agents Chemother. 1994, 38, 1661–1663. [Google Scholar] [CrossRef]
- Retchless, A.C.; Kretz, C.B.; Chang, H.Y.; Bazan, J.A.; Abrams, A.J.; Norris Turner, A.; Jenkins, L.T.; Trees, D.L.; Tzeng, Y.L.; Stephens, D.S.; et al. Expansion of a urethritis-associated Neisseria meningitidis clade in the United States with concurrent acquisition of N. gonorrhoeae alleles. BMC Genom. 2018, 19, 176. [Google Scholar] [CrossRef]
- Bazan, J.A.; Turner, A.N.; Kirkcaldy, R.D.; Retchless, A.C.; Kretz, C.B.; Briere, E.; Tzeng, Y.L.; Stephens, D.S.; Maierhofer, C.; Del Rio, C.; et al. Large Cluster of Neisseria meningitidis Urethritis in Columbus, Ohio, 2015. Clin. Infect. Dis. 2017, 65, 92–99. [Google Scholar] [CrossRef]
- Tzeng, Y.L.; Bazan, J.A.; Turner, A.N.; Wang, X.; Retchless, A.C.; Read, T.D.; Toh, E.; Nelson, D.E.; Del Rio, C.; Stephens, D.S. Emergence of a new Neisseria meningitidis clonal complex 11 lineage 11.2 clade as an effective urogenital pathogen. Proc. Natl. Acad. Sci. USA 2017, 114, 4237–4242. [Google Scholar] [CrossRef]
- Hagman, M.; Forslin, L.; Moi, H.; Danielsson, D. Neisseria meningitidis in specimens from urogenital sites. Is increased awareness necessary? Sex. Transm. Dis. 1991, 18, 228–232. [Google Scholar] [CrossRef]
- Wilson, A.P.; Wolff, J.; Atia, W. Acute urethritis due to Neisseria meningitidis group A acquired by orogenital contact: Case report. Genitourin. Med. 1989, 65, 122–123. [Google Scholar] [CrossRef] [PubMed]
- Keys, T.F.; Hecht, R.H.; Chow, A.W. Endocervical Neisseria meningitidis with meningococcemia. N. Engl. J. Med. 1971, 285, 505–506. [Google Scholar] [CrossRef] [PubMed]
- Harriau, P.; Ramanantsoa, C.; Pierre, F.; Riou, J.Y.; Quentin, R. Endocervical infection in a pregnant woman caused by Neisseria meningitidis: Evidence of associated oropharyngeal colonization of the male partner. Eur. J. Obstet. Gynecol. Reprod. Biol. 1997, 74, 145–147. [Google Scholar] [CrossRef]
- Fiorito, S.M.; Galarza, P.G.; Sparo, M.; Pagano, E.I.; Oviedo, C.I. An unusual transmission of Neisseria meningitidis: Neonatal conjunctivitis acquired at delivery from the mother’s endocervical infection. Sex. Transm. Dis. 2001, 28, 29–32. [Google Scholar] [CrossRef]
- Mitchell, S.R.; Katz, P. Disseminated neisserial infection in pregnancy: The empress may have a change of clothing. Obstet. Gynecol. Surv. 1989, 44, 780–788. [Google Scholar]
- Bhutta, Z.A.; Khan, I.A.; Agha, Z. Fatal intrauterine meningococcal infection. Pediatr. Infect. Dis. J. 1991, 10, 868–869. [Google Scholar]
- Irani, F.; Ruddell, T. Meningococcal conjunctivitis. Aust. N. Z. J. Ophthalmol. 1997, 25, 167–168. [Google Scholar] [CrossRef]
- Unal Yilmaz, G.; Alkan, M.; Vatansever Ozbek, U.; Tugrul, H.M. [Healthcare-associated Neisseria meningitidis W135 conjunctivitis]. Mikrobiyol. Bul. 2013, 47, 722–726. [Google Scholar] [CrossRef]
- Moraga Llop, F.A.; Barquet Esteve, N.; Domingo Pedrol, P.; Gallart Catala, A. [Primary meningococcal conjunctivitis: Implications beyond the conjunctiva]. Med. Clin. (Barc) 1996, 107, 130–132. [Google Scholar]
- Barquet, N.; Gasser, I.; Domingo, P.; Moraga, F.A.; Macaya, A.; Elcuaz, R. Primary meningococcal conjunctivitis: Report of 21 patients and review. Rev. Infect. Dis. 1990, 12, 838–847. [Google Scholar] [CrossRef]
- Holmberg, L.; Moestrup, T. Meningitis following conjunctivitis. J. Pediatr. 1979, 94, 339. [Google Scholar] [CrossRef]
- Nussbaum, E.; Jeyaranjan, T.; Feldman, F. Primary meningococcal conjunctivitis followed by meningitis. J. Pediatr. 1978, 92, 784–785. [Google Scholar] [CrossRef]
- Dryden, A.W.; Rana, M.; Pandey, P. Primary meningococcal conjunctivitis: An unusual case of transmission by saliva. Digit. J. Ophthalmol. 2016, 22, 25–27. [Google Scholar] [CrossRef] [PubMed]
- Holdsworth, G.; Jackson, H.; Kaczmarski, E. Meningococcal infection from saliva. Lancet 1996, 348, 1443. [Google Scholar] [CrossRef]
- Hansman, D. Neonatal meningococcal conjunctivitis. Br. Med. J. 1972, 1, 748. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Poulos, R.G.; Smedley, E.J.; Ferson, M.J.; Bolisetty, S.; Tapsall, J.W. Refining the public health response to primary meningococcal conjunctivitis. Commun. Dis. Intell. Q Rep. 2002, 26, 592–595. [Google Scholar]
- de Souza, A.L.; Seguro, A.C. Conjunctivitis secondary to Neisseria meningitidis: A potential vertical transmission pathway. Clin. Pediatr. (Phila.) 2009, 48, 119. [Google Scholar] [CrossRef]
- Ellis, M.; Weindling, A.M.; Davidson, D.C.; Ho, N.; Damjanovic, V. Neonatal meningococcal conjunctivitis associated with meningococcal meningitis. Arch. Dis. Child. 1992, 67, 1219–1220. [Google Scholar] [CrossRef]
- Agrawal, P.; Yellachich, D.; Kirkpatrick, N. Retinal detachment following meningococcal endophthalmitis. Eye (Lond.) 2007, 21, 450–451. [Google Scholar] [CrossRef][Green Version]
- Chacko, E.; Filtcroft, I.; Condon, P.I. Meningococcal septicemia presenting as bilateral endophthalmitis. J. Cataract. Refract. Surg. 2005, 31, 432–434. [Google Scholar] [CrossRef]
- Balaskas, K.; Potamitou, D. Endogenous endophthalmitis secondary to bacterial meningitis from Neisseria Meningitidis: A case report and review of the literature. Cases J. 2009, 2, 149. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barnard, T.; Das, A.; Hickey, S. Bilateral endophthalmitis as an initial presentation in meningococcal meningitis. Arch. Ophthalmol. 1997, 115, 1472–1473. [Google Scholar] [CrossRef] [PubMed]
- Chhabra, M.S.; Noble, A.G.; Kumar, A.V.; Mets, M.B. Neisseria meningitidis endogenous endophthalmitis presenting as anterior uveitis. J. Pediatr. Ophthalmol. Strabismus 2007, 44, 309–310. [Google Scholar] [PubMed]
- Sleep, T.; Graham, M. A case of meningococcal endophthalmitis in a well patient. Br. J. Ophthalmol. 1997, 81, 1016–1017. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kerkhoff, F.T.; van der Zee, A.; Bergmans, A.M.; Rothova, A. Polymerase chain reaction detection of Neisseria meningitidis in the intraocular fluid of a patient with endogenous endophthalmitis but without associated meningitis. Ophthalmology 2003, 110, 2134–2136. [Google Scholar] [CrossRef]
- Kallinich, T.; von Bernuth, H.; Kuhns, M.; Elias, J.; Bertelmann, E.; Pleyer, U. Fulminant Endophthalmitis in a Child Caused by Neisseria meningitidis Serogroup C Detected by Specific DNA. J. Pediatric Infect. Dis. Soc. 2016, 5, e13–e16. [Google Scholar] [CrossRef]
- Yusuf, I.H.; Sipkova, Z.; Patel, S.; Benjamin, L. Neisseria meningitidis endogenous endophthalmitis with meningitis in an immunocompetent child. Ocul. Immunol. Inflamm. 2014, 22, 398–402. [Google Scholar] [CrossRef]
- Gartaganis, S.P.; Eliopoulou, M.J.; Georgakopoulos, C.D.; Koliopoulos, J.X.; Mela, E.K. Bilateral panophthalmitis as the initial presentation of meningococcal meningitis in an infant. J. AAPOS 2001, 5, 260–261. [Google Scholar] [CrossRef]
- Abandeh, F.I.; Balada-Llasat, J.M.; Pancholi, P.; Risaliti, C.M.; Maher, W.E.; Bazan, J.A. A rare case of Neisseria bacilliformis native valve endocarditis. Diagn. Microbiol. Infect. Dis. 2012, 73, 378–379. [Google Scholar] [CrossRef]
- Masliah-Planchon, J.; Breton, G.; Jarlier, V.; Simon, A.; Benveniste, O.; Herson, S.; Drieux, L. Endocarditis due to Neisseria bacilliformis in a patient with a bicuspid aortic valve. J. Clin. Microbiol. 2009, 47, 1973–1975. [Google Scholar] [CrossRef]
- Han, X.Y.; Hong, T.; Falsen, E. Neisseria bacilliformis sp. nov. isolated from human infections. J. Clin. Microbiol. 2006, 44, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Allison, K.; Clarridge, J.E., 3rd. Long-term respiratory tract infection with canine-associated Pasteurella dagmatis and Neisseria canis in a patient with chronic bronchiectasis. J. Clin. Microbiol. 2005, 43, 4272–4274. [Google Scholar] [CrossRef] [PubMed]
- Safton, S.; Cooper, G.; Harrison, M.; Wright, L.; Walsh, P. Neisseria canis infection: A case report. Commun. Dis. Intell. 1999, 23, 221. [Google Scholar] [PubMed]
- Southern, P.M., Jr.; Kutscher, A.E. Bacteremia due to Neisseria cinerea: Report of two cases. Diagn. Microbiol. Infect. Dis. 1987, 7, 143–147. [Google Scholar] [CrossRef]
- Kirchgesner, V.; Plesiat, P.; Dupont, M.J.; Estavoyer, J.M.; Guibourdenche, M.; Riou, J.Y.; Michel-Briand, Y. Meningitis and septicemia due to Neisseria cinerea. Clin. Infect. Dis. 1995, 21, 1351. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.N.; Luo, Z.H.; Wang, H. [Diagnosis and treatment of genitourinary infection with non-gonococcal Neisseria in men]. Zhonghua Nan Ke Xue 2009, 15, 499–504. [Google Scholar] [PubMed]
- Garcia, S.D.; Descole, E.M.; Famiglietti, A.M.; Lopez, E.G.; Vay, C.A. Infection of the urinary tract caused by Neisseria cinerea. Enferm. Infecc. Microbiol. Clin. 1996, 14, 576. [Google Scholar]
- Taegtmeyer, M.; Saxena, R.; Corkill, J.E.; Anijeet, H.; Parry, C.M. Ciprofloxacin treatment of bacterial peritonitis associated with chronic ambulatory peritoneal dialysis caused by Neisseria cinerea. J. Clin. Microbiol. 2006, 44, 3040–3041. [Google Scholar] [CrossRef]
- Bourbeau, P.; Holla, V.; Piemontese, S. Ophthalmia neonatorum caused by Neisseria cinerea. J. Clin. Microbiol. 1990, 28, 1640–1641. [Google Scholar]
- Fiorito, T.M.; Noor, A.; Silletti, R.; Krilov, L.R. Neonatal Conjunctivitis Caused by Neisseria cinerea: A Case of Mistaken Identity. J. Pediatric. Infect. Dis. Soc. 2018, 8, 478–480. [Google Scholar] [CrossRef]
- Wroblewski, D.; Cole, J.; McGinnis, J.; Perez, M.; Wilson, H.; Mingle, L.A.; Musser, K.A.; Wolfgang, W.J. Neisseria dumasiana sp. nov. from human sputum and a dog’s mouth. Int. J. Syst. Evol. Microbiol. 2017, 67, 4304–4310. [Google Scholar] [CrossRef] [PubMed]
- Grant, P.E.; Brenner, D.J.; Steigerwalt, A.G.; Hollis, D.G.; Weaver, R.E. Neisseria elongata subsp. nitroreducens subsp. nov., formerly CDC group M-6, a gram-negative bacterium associated with endocarditis. J. Clin. Microbiol. 1990, 28, 2591–2596. [Google Scholar] [PubMed]
- Samannodi, M.; Vakkalanka, S.; Zhao, A.; Hocko, M. Neisseria elongata endocarditis of a native aortic valve. BMJ Case Rep. 2016, 2016. [Google Scholar] [CrossRef]
- Hofstad, T.; Hope, O.; Falsen, E. Septicaemia with Neisseria elongata ssp. nitroreducens in a patient with hypertrophic obstructive cardiomyopathia. Scand. J. Infect. Dis. 1998, 30, 200–201. [Google Scholar] [CrossRef] [PubMed]
- Garner, J.; Briant, R.H. Osteomyelitis caused by a bacterium known as M6. J. Infect. 1986, 13, 298–300. [Google Scholar] [CrossRef]
- Scott, R.M. Bacterial endocarditis due to Neisseria flava. J. Pediatr. 1971, 78, 673–675. [Google Scholar] [CrossRef]
- Matlage, W.T.; Harrison, P.E.; Greene, J.A. Neisseria flava endocarditis; with report of a case. Ann. Intern. Med. 1950, 33, 1494–1498. [Google Scholar] [CrossRef]
- Sinave, C.P.; Ratzan, K.R. Infective endocarditis caused by Neisseria flavescens. Am. J. Med. 1987, 82, 163–164. [Google Scholar] [CrossRef]
- Szabo, S.; Lieberman, J.P.; Lue, Y.A. Unusual pathogens in narcotic-associated endocarditis. Rev. Infect. Dis. 1990, 12, 412–415. [Google Scholar] [CrossRef]
- Branham, S.E. A new meningococcus-like organism (Neisseria flavescens nsp) from epidemic meningitis. Public Health Rep. 1930, 45, 845–849. [Google Scholar] [CrossRef]
- Prentice, A.W. Neisseria flavescens as a cause of meningitis. Lancet 1957, 272, 613–614. [Google Scholar] [CrossRef]
- Feder, H.M., Jr.; Garibaldi, R.A. The significance of nongonococcal, nonmeningococcal Neisseria isolates from blood cultures. Rev. Infect. Dis. 1984, 6, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Wertlake, P.T.; Williams, T.W., Jr. Septicaemia caused by Neisseria flavescens. J. Clin. Pathol. 1968, 21, 437–439. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Ma, L.; Fan, K.; Li, Y.; Xie, L.; Xia, W.; Gu, B.; Liu, G. Necrotizing pneumonia and empyema caused by Neisseria flavescens infection. J. Thorac. Dis. 2014, 6, 553–557. [Google Scholar] [CrossRef]
- Wax, L. The identity of Neisseria other than the gonococcus isolated from the genito-urinary tract. J. Vener. Dis. Infect. 1950, 31, 208–213. [Google Scholar]
- Denning, D.W.; Gill, S.S. Neisseria lactamica meningitis following skull trauma. Rev. Infect. Dis. 1991, 13, 216–218. [Google Scholar] [CrossRef]
- Lauer, B.A.; Fisher, C.E. Neisseria lactamica meningitis. Am. J. Dis. Child. 1976, 130, 198–199. [Google Scholar] [CrossRef]
- Wilson, H.D.; Overman, T.L. Septicemia due to Neisseria lactamica. J. Clin. Microbiol. 1976, 4, 214–215. [Google Scholar]
- Fisher, L.S.; Edelstein, P.; Guze, L.B. Letter: Neisseria lactamicus pharyngitis. JAMA 1975, 233, 22. [Google Scholar] [CrossRef]
- Zavascki, A.P.; Fritscher, L.; Superti, S.; Dias, C.; Kroth, L.; Traesel, M.A.; Antonello, I.C.; Saitovitch, D. First case report of Neisseria lactamica causing cavitary lung disease in an adult organ transplant recipient. J. Clin. Microbiol. 2006, 44, 2666–2668. [Google Scholar] [CrossRef]
- Wang, C.Y.; Chuang, Y.M.; Teng, L.J.; Lee, L.N.; Yang, P.C.; Kuo, S.H.; Hsueh, P.R. Bacteraemic pneumonia caused by Neisseria lactamica with reduced susceptibility to penicillin and ciprofloxacin in an adult with liver cirrhosis. J. Med. Microbiol. 2006, 55, 1151–1152. [Google Scholar] [CrossRef] [PubMed]
- Jephcott, A.E.; Morton, R.S. Isolation of Neisseria lactamicus from a genital site. Lancet 1972, 2, 739–740. [Google Scholar] [CrossRef]
- Brunton, W.A.T.; Young, H.; Fraser, D.R.K. Isolation of Neisseria lactamica from the female genital tract. Br. J. Vener. Dis. 1980, 56, 325–326. [Google Scholar]
- Brodie, E.; Adler, J.L.; Daly, A.K. Bacterial endocarditis due to an unusual species of encapsulated Neisseria. Neisseria mucosa endocarditis. Am. J. Dis. Child. 1971, 122, 433–437. [Google Scholar] [CrossRef]
- Pilmis, B.; Lefort, A.; Lecuit, M.; Join-Lambert, O.; Nassif, X.; Lortholary, O.; Charlier, C. Endocarditis due to Neisseria mucosa: Case report and review of 21 cases: A rare and severe cause of endocarditis. J. Infect. 2014, 68, 601–604. [Google Scholar] [CrossRef]
- Ingram, R.J.; Cornere, B.; Ellis-Pegler, R.B. Endocarditis due to Neisseria mucosa: Two case reports and review. Clin. Infect. Dis. 1992, 15, 321–324. [Google Scholar] [CrossRef]
- Sirot, J.; Cluzel, M. “Neisseria mucosa” responsible for purulent meningitis of children. Ann. Inst. Pasteur. (Paris) 1972, 122, 53–61. [Google Scholar]
- Stotka, J.L.; Rupp, M.E.; Meier, F.A.; Markowitz, S.M. Meningitis due to Neisseria mucosa: Case report and review. Rev. Infect. Dis. 1991, 13, 837–841. [Google Scholar] [CrossRef]
- Locy, C.J. Neisseria mucosa septicemia. Clin. Microbiol. Newsl. 1995, 17, 72. [Google Scholar] [CrossRef]
- Thorsteinsson, S.B.; Minuth, J.N.; Musher, D.M. Postpneumonectomy empyema due to Neisseria mucosa. Am. J. Clin. Pathol. 1975, 64, 534–536. [Google Scholar] [CrossRef]
- Hanau-Bercot, B.; Rottman, M.; Raskine, L.; Jacob, D.; Barnaud, G.; Gabarre, A.; Sanson Le Pors, M.J. Clinical resistance to amoxicillin of a gravidic urinary tract infection caused by Neisseria mucosa. J. Infect. 2001, 43, 160–161. [Google Scholar] [CrossRef] [PubMed]
- Washburn, R.G.; Bryan, C.S.; DiSalvo, A.F.; Macher, A.M.; Gallin, J.I. Visceral botryomycosis caused by Neisseria mucosa in a patient with chronic granulomatous disease. J. Infect. Dis. 1985, 151, 563–564. [Google Scholar] [CrossRef] [PubMed]
- Abiteboul, M.; Mazieres, B.; Causse, B.; Moatti, N.; Arlet, J. Septic arthritis of the knee due to Neisseria mucosa. Clin. Rheumatol. 1985, 4, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Van Linthoudt, D.; Modde, H.; Ott, H. Neisseria mucosa septic arthritis. Br. J. Rheumatol. 1987, 26, 314. [Google Scholar] [CrossRef]
- Alamri, Y.; Keene, A.; Pithie, A. Acute Cystitis Caused by Commensal Neisseria oralis: A Case Report and Review of the Literature. Infect. Disord. Drug Targets 2017, 17, 64–66. [Google Scholar] [CrossRef]
- Wolfgang, W.J.; Passaretti, T.V.; Jose, R.; Cole, J.; Coorevits, A.; Carpenter, A.N.; Jose, S.; Van Landschoot, A.; Izard, J.; Kohlerschmidt, D.J.; et al. Neisseria oralis sp. nov., isolated from healthy gingival plaque and clinical samples. Int. J. Syst. Evol. Microbiol. 2013, 63, 1323–1328. [Google Scholar] [CrossRef]
- Breslin, A.B.; Biggs, J.C.; Hall, G.V. Bacterial endocarditis due to Neisseria perflava in a patient hypersensitive to penicillin. Australas. Ann. Med. 1967, 16, 245–249. [Google Scholar] [CrossRef]
- Clark, H.; Patton, R.D. Postcardiotomy endocarditis due to Neisseria perflava on a prosthetic aortic valve. Ann. Intern. Med. 1968, 68, 386–389. [Google Scholar] [CrossRef]
- Wolfgang, W.J.; Carpenter, A.N.; Cole, J.A.; Gronow, S.; Habura, A.; Jose, S.; Nazarian, E.J.; Kohlerschmidt, D.J.; Limberger, R.; Schoonmaker-Bopp, D.; et al. Neisseria wadsworthii sp. nov. and Neisseria shayeganii sp. nov., isolated from clinical specimens. Int. J. Syst. Evol. Microbiol. 2011, 61, 91–98. [Google Scholar] [CrossRef]
- Schultz, O.T. Acute vegetative endocarditis with multiple secondary foci involvement due to Micrococcus pharyngitidis-siccae. JAMA 1918, 71, 1739–1741. [Google Scholar] [CrossRef]
- Graef, I.; de la Chapelle, C.E.; Vance, M.C. Micrococcus pharyngis siccus endocarditis. Am. J. Pathol. 1932, 8, 341, 347–354. [Google Scholar] [PubMed]
- Kirlew, C.; Wilmot, K.; Salinas, J.L. Neisseria sicca Endocarditis Presenting as Multiple Embolic Brain Infarcts. Open Forum Infect. Dis. 2015, 2, ofv105. [Google Scholar] [CrossRef] [PubMed]
- Sommerstein, R.; Ramsay, D.; Dubuis, O.; Waser, S.; Aebersold, F.; Vogt, M. Fatal Neisseria sicca endocarditis. Infection 2013, 41, 747–749. [Google Scholar] [CrossRef] [PubMed]
- Bansmer, C.; Brem, J. Acute meningitis caused by Neisseria sicca. N. Engl. J. Med. 1948, 238, 596. [Google Scholar] [CrossRef]
- Carter, J.E.; Mizell, K.N.; Evans, T.N. Neisseria sicca meningitis following intracranial hemorrhage and ventriculostomy tube placement. Clin. Neurol. Neurosurg. 2007, 109, 918–921. [Google Scholar] [CrossRef] [PubMed]
- Alcid, D.V. Neisseria sicca pneumonia. Chest 1980, 77, 123–124. [Google Scholar] [CrossRef]
- Wilkinson, A.E. Occurrence of Neisseria other than the gonococcus in the genital tract. Br. J. Vener. Dis. 1952, 28, 24–27. [Google Scholar] [CrossRef]
- Weaver, J.D. Nongonorrheal vulvovaginitis due to gram-negative intracellular diplococci. Am. J. Obstet. Gynecol. 1950, 60, 257–260. [Google Scholar] [CrossRef]
- Gomez-Camarasa, C.; Liebana-Martos, C.; Navarro-Mari, J.M.; Gutierrez-Fernandez, J. Detection of unusual uropathogens during a period of three years in a regional hospital. Rev. Esp Quimioter. 2015, 28, 86–91. [Google Scholar]
- Connaughton, F.W.; Rountree, P.M. A fatal case of infective endocarditis due to Neisseria flava. Med. J. Aust. 1939, 2, 138–139. [Google Scholar] [CrossRef]
- Flores, J.; Lloret, A.; Bellver, F.; Segarra, C.; Monzo, E. [Infectious endocarditis by Neisseria subflava in two HIV drug users]. An. Med. Interna 1997, 14, 267–268. [Google Scholar] [PubMed]
- Benson, H.; Brennwasser, R.; D’andrea, D. Neisseria subflava (Bergey) meningitis in an infant. J. Infect. Dis. 1928, 43, 516–524. [Google Scholar] [CrossRef]
- Lewin, R.A.; Hughes, W.T. Neisseria subflava as a cause of meningitis and septicemia in children. Report of five cases. JAMA 1966, 195, 821–823. [Google Scholar] [CrossRef]
- Wakui, D.; Nagashima, G.; Otsuka, Y.; Takada, T.; Ueda, T.; Tanaka, Y.; Hashimoto, T. A case of meningitis due to Neisseria subflava after ventriculostomy. J. Infect. Chemother. 2012, 18, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, C.M. Isolation of Neisseria flava from the Genitourinary Tract of Three Patients. Am. J. Public Health Nations Health 1943, 33, 135–136. [Google Scholar] [CrossRef] [PubMed]
- Janda, W.M.; Senseng, C.; Todd, K.M.; Schreckenberger, P.C. Asymptomatic Neisseria subflava biovar perflava bacteriuria in a child with obstructive uropathy. Eur. J. Clin. Microbiol. Infect. Dis. 1993, 12, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Carlson, P.; Kontiainen, S.; Anttila, P.; Eerola, E. Septicemia caused by Neisseria weaveri. Clin. Infect. Dis. 1997, 24, 739. [Google Scholar] [CrossRef]
- Panagea, S.; Bijoux, R.; Corkill, J.E.; Al Rashidi, F.; Hart, C.A. A case of lower respiratory tract infection caused by Neisseria weaveri and review of the literature. J. Infect. 2002, 44, 96–98. [Google Scholar] [CrossRef]
- Kocyigit, I.; Unal, A.; Sipahioglu, M.; Tokgoz, B.; Oymak, O.; Utas, C. Peritoneal dialysis-related peritonitis due to Neisseria weaveri: The first case report. Perit. Dial. Int. 2010, 30, 116–117. [Google Scholar] [CrossRef]
- Andersen, B.M.; Steigerwalt, A.G.; O’Connor, S.P.; Hollis, D.G.; Weyant, R.S.; Weaver, R.E.; Brenner, D.J. Neisseria weaveri sp. nov., formerly CDC group M-5, a gram-negative bacterium associated with dog bite wounds. J. Clin. Microbiol. 1993, 31, 2456–2466. [Google Scholar]
- Holmes, B.; Costas, M.; On, S.L.; Vandamme, P.; Falsen, E.; Kersters, K. Neisseria weaveri sp. nov. (formerly CDC group M-5), from dog bite wounds of humans. Int. J. Syst. Bacteriol. 1993, 43, 687–693. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Grob, J.J.; Bollet, C.; Richard, M.A.; De Micco, P.; Bonerandi, J.J. Extensive skin ulceration due to EF-4 bacterial infection in a patient with AIDS. Br. J. Dermatol. 1989, 121, 507–510. [Google Scholar] [CrossRef] [PubMed]
- Catlin, B.W. Branhamella catarrhalis: An organism gaining respect as a pathogen. Clin. Microbiol. Rev. 1990, 3, 293–320. [Google Scholar] [CrossRef] [PubMed]
- Pettersson, B.; Kodjo, A.; Ronaghi, M.; Uhlen, M.; Tonjum, T. Phylogeny of the family Moraxellaceae by 16S rDNA sequence analysis, with special emphasis on differentiation of Moraxella species. Int. J. Syst. Bacteriol. 1998, 48, 75–89. [Google Scholar] [CrossRef]
- Dillard, J.P.; Seifert, H.S. A variable genetic island specific for Neisseria gonorrhoeae is involved in providing DNA for natural transformation and is found more often in disseminated infection isolates. Mol. Microbiol. 2001, 41, 263–277. [Google Scholar] [CrossRef]
- Dillard, J.P.; Seifert, H.S. A peptidoglycan hydrolase similar to bacteriophage endolysins acts as an autolysin in Neisseria gonorrhoeae. Mol. Microbiol. 1997, 25, 893–901. [Google Scholar] [CrossRef][Green Version]
- Frazer, A.D.; Menton, J. Gonococcal Stomatitis. Br. Med. J. 1931, 1, 1020–1022. [Google Scholar] [CrossRef]
- Copping, A.A. Stomatitis caused by gonococcus. J. Am. Dent. Assoc. 1954, 49, 567. [Google Scholar]
- Schmidt, H.; Hjorting-Hansen, E.; Philipsen, H.P. Gonococcal stomatitis. Acta Derm. Venereol. 1961, 41, 324–327. [Google Scholar]
- Escobar, V.; Farman, A.G.; Arm, R.N. Oral gonococcal infection. Int. J. Oral. Surg. 1984, 13, 549–554. [Google Scholar] [CrossRef]
- Kohn, S.R.; Shaffer, J.F.; Chomenko, A.G. Primary gonococcal stomatitis. JAMA 1972, 219, 86. [Google Scholar] [CrossRef] [PubMed]
- Regan, D.G.; Hui, B.B.; Wood, J.G.; Fifer, H.; Lahra, M.M.; Whiley, D.M. Treatment for pharyngeal gonorrhoea under threat. Lancet Infect. Dis. 2018, 18, 1175–1177. [Google Scholar] [CrossRef]
- Fifer, H.; Natarajan, U.; Jones, L.; Alexander, S.; Hughes, G.; Golparian, D.; Unemo, M. Failure of Dual Antimicrobial Therapy in Treatment of Gonorrhea. N. Engl. J. Med. 2016, 374, 2504–2506. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D.W.; Sanderson, N.D.; Lord, E.; Regisford-Reimmer, N.; Chau, K.; Barker, L.; Morgan, M.; Newnham, R.; Golparian, D.; Unemo, M.; et al. Gonorrhoea treatment failure caused by a Neisseria gonorrhoeae strain with combined ceftriaxone and high-level azithromycin resistance, England, February 2018. Euro Surveill. 2018, 23, 1800323. [Google Scholar] [CrossRef] [PubMed]
- Leibowitz, H.M. The red eye. N. Engl. J. Med. 2000, 343, 345–351. [Google Scholar] [CrossRef]
- Moi, H.; Blee, K.; Horner, P.J. Management of non-gonococcal urethritis. BMC Infect. Dis. 2015, 15, 294. [Google Scholar] [CrossRef]
- Kroll, J.S.; Wilks, K.E.; Farrant, J.L.; Langford, P.R. Natural genetic exchange between Haemophilus and Neisseria: Intergeneric transfer of chromosomal genes between major human pathogens. Proc. Natl. Acad. Sci. USA 1998, 95, 12381–12385. [Google Scholar] [CrossRef]
- Tzeng, Y.L.; Thomas, J.; Stephens, D.S. Regulation of capsule in Neisseria meningitidis. Crit. Rev. Microbiol. 2016, 42, 759–772. [Google Scholar] [CrossRef]
- Hammerschmidt, S.; Birkholz, C.; Zahringer, U.; Robertson, B.D.; van Putten, J.; Ebeling, O.; Frosch, M. Contribution of genes from the capsule gene complex (cps) to lipooligosaccharide biosynthesis and serum resistance in Neisseria meningitidis. Mol. Microbiol. 1994, 11, 885–896. [Google Scholar] [CrossRef]
- Kahler, C.M.; Martin, L.E.; Shih, G.C.; Rahman, M.M.; Carlson, R.W.; Stephens, D.S. The (alpha2-->8)-linked polysialic acid capsule and lipooligosaccharide structure both contribute to the ability of serogroup B Neisseria meningitidis to resist the bactericidal activity of normal human serum. Infect. Immun. 1998, 66, 5939–5947. [Google Scholar]
- Masson, L.; Holbein, B.E. Influence of nutrient limitation and low pH on serogroup B Neisseria meningitidis capsular polysaccharide levels: Correlation with virulence for mice. Infect. Immun. 1985, 47, 465–471. [Google Scholar] [PubMed]
- Vogel, U.; Frosch, M. Mechanisms of neisserial serum resistance. Mol. Microbiol. 1999, 32, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Vogel, U.; Weinberger, A.; Frank, R.; Muller, A.; Kohl, J.; Atkinson, J.P.; Frosch, M. Complement factor C3 deposition and serum resistance in isogenic capsule and lipooligosaccharide sialic acid mutants of serogroup B Neisseria meningitidis. Infect. Immun. 1997, 65, 4022–4029. [Google Scholar] [PubMed]
- Claus, H.; Maiden, M.C.; Maag, R.; Frosch, M.; Vogel, U. Many carried meningococci lack the genes required for capsule synthesis and transport. Microbiology 2002, 148, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Claus, H.; Maiden, M.C.; Wilson, D.J.; McCarthy, N.D.; Jolley, K.A.; Urwin, R.; Hessler, F.; Frosch, M.; Vogel, U. Genetic analysis of meningococci carried by children and young adults. J. Infect. Dis. 2005, 191, 1263–1271. [Google Scholar] [CrossRef]
- Jolley, K.A.; Kalmusova, J.; Feil, E.J.; Gupta, S.; Musilek, M.; Kriz, P.; Maiden, M.C. Carried meningococci in the Czech Republic: A diverse recombining population. J. Clin. Microbiol. 2000, 38, 4492–4498. [Google Scholar] [CrossRef]
- Yazdankhah, S.P.; Kriz, P.; Tzanakaki, G.; Kremastinou, J.; Kalmusova, J.; Musilek, M.; Alvestad, T.; Jolley, K.A.; Wilson, D.J.; McCarthy, N.D.; et al. Distribution of serogroups and genotypes among disease-associated and carried isolates of Neisseria meningitidis from the Czech Republic, Greece, and Norway. J. Clin. Microbiol. 2004, 42, 5146–5153. [Google Scholar] [CrossRef]
- Ng, L.K.; Martin, I.E. The laboratory diagnosis of Neisseria gonorrhoeae. Can. J. Infect. Dis. Med. Microbiol. 2005, 16, 15–25. [Google Scholar] [CrossRef]
- Perrin, A.; Bonacorsi, S.; Carbonnelle, E.; Talibi, D.; Dessen, P.; Nassif, X.; Tinsley, C. Comparative genomics identifies the genetic islands that distinguish Neisseria meningitidis, the agent of cerebrospinal meningitis, from other Neisseria species. Infect. Immun. 2002, 70, 7063–7072. [Google Scholar] [CrossRef]
- Chugh, K.; Bhalla, C.K.; Joshi, K.K. Meningococcal brain abscess and meningitis in a neonate. Pediatr. Infect. Dis. J. 1988, 7, 136–137. [Google Scholar] [CrossRef]
- Herbert, D.A.; Ruskin, J. Are the “nonpathogenic” Neisseriae pathogenic? Am. J. Clin. Pathol. 1981, 75, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.J.; Higashi, D.; Goytia, M.; Rendon, M.A.; Pilligua-Lucas, M.; Bronnimann, M.; McLean, J.A.; Duncan, J.; Trees, D.; Jerse, A.E.; et al. Commensal Neisseria Kill Neisseria gonorrhoeae through a DNA-Dependent Mechanism. Cell Host Microbe 2019, 26, 228–239.e8. [Google Scholar] [CrossRef] [PubMed]
- Coulter, C.B. Gram-negative micrococcus causing fatal endocarditis. Proc. N. Y. Pathol. Soc. 1915, 15, 7–12. [Google Scholar]
- Wilson, W.J. A contribution to the bacteriology of cerebrospinal meningitis. Lancet 1908, 1, 1686–1687. [Google Scholar] [CrossRef]
- Brorson, J.E.; Axelsson, A.; Holm, S.E. Studies on Branhamella catarrhalis (Neisseria catarrhalis) with special reference to maxillary sinusitis. Scand. J. Infect. Dis. 1976, 8, 151–155. [Google Scholar] [CrossRef]
- Coffey, J.D., Jr.; Martin, A.D.; Booth, H.N. Neisseria catarrhalis in exudate otitis media. Arch. Otolaryngol. 1967, 86, 403–406. [Google Scholar] [CrossRef]
- Lee, W.S.; Fordham, T.; Alban, J. Otitis media caused by beta-lactamase-producing Branhamella (Neisseria) catarrhalis. J. Clin. Microbiol. 1981, 13, 222–223. [Google Scholar]
- Schalen, L.; Christensen, P.; Kamme, C.; Miorner, H.; Pettersson, K.I.; Schalen, C. High isolation rate of Branhamella catarrhalis from the nasopharynx in adults with acute laryngitis. Scand. J. Infect. Dis. 1980, 12, 277–280. [Google Scholar] [CrossRef]
- Johnson, M.A.; Drew, W.L.; Roberts, M. Branhamella (Neisseria) catarrhalis—A lower respiratory tract pathogen? J. Clin. Microbiol. 1981, 13, 1066–1069. [Google Scholar]
- McNeely, D.J.; Kitchens, C.S.; Kluge, R.M. Fatal Neisseria (Branhamella) catarrhalis pneumonia in an immunodeficient host. Am. Rev. Respir. Dis. 1976, 114, 399–402. [Google Scholar] [CrossRef]
- Srinivasan, G.; Raff, M.J.; Templeton, W.C.; Givens, S.J.; Graves, R.C.; Melo, J.C. Branhamella catarrhalis pneumonia: Report of two cases and review of the literature. Am. Rev. Respir. Dis. 1981, 123, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Ninane, G.; Joly, J.; Piot, P.; Kraytman, M. Branhamella (Neisseria) catarrhalis as pathogen. Lancet 1977, 2, 149. [Google Scholar] [CrossRef]
- Ninane, G.; Joly, J.; Kraytman, M. Bronchopulmonary infection due to Branhamella catarrhalis: 11 cases assessed by transtracheal puncture. Br. Med. J. 1978, 1, 276–278. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ninane, G.; Joly, J.; Kraytman, M.; Piot, P. Bronchopulmonary infection due to beta-lactamase-producing Branhamella catarrhalis treated with amoxycillin/clavulanic-acid. Lancet 1978, 2, 257. [Google Scholar] [CrossRef]
- Percival, A.; Corkill, J.E.; Rowlands, J.; Sykes, R.B. Pathogenicity of and beta-lactamase production by Branhamella (Neisseria) catarrhalis. Lancet 1977, 2, 1175. [Google Scholar] [CrossRef]
- Verma, R.; Sood, S. Gonorrhoea diagnostics: An update. Indian J. Med. Microbiol. 2016, 34, 139–145. [Google Scholar] [CrossRef]
- Graber, C.D.; Scott, R.C.; Dunkelberg, W.E., Jr.; Dirks, K.R. Isolation of Neisseria catarrhalis from three patients with urethritis and a clinical syndrome resembling gonorrhea. Am. J. Clin. Pathol. 1963, 39, 360–363. [Google Scholar] [CrossRef]
- McCague, J.J.; McCague, N.J.; Altman, C.C. Neisseria catarrhalis urethritis: A case report. J. Urol. 1976, 115, 471. [Google Scholar] [CrossRef]
- Blackwell, C.; Young, H.; Bain, S.S. Isolation of Neisseria meningitidis and Neisseria catarrhalis from the genitourinary tract and anal canal. Br. J. Vener. Dis. 1978, 54, 41–44. [Google Scholar] [CrossRef]
- Rosenstein, N.E.; Perkins, B.A.; Stephens, D.S.; Popovic, T.; Hughes, J.M. Meningococcal disease. N. Engl. J. Med. 2001, 344, 1378–1388. [Google Scholar] [CrossRef]
- Lee, T.J.; Utsinger, P.D.; Snyderman, R.; Yount, W.J.; Sparling, P.F. Familial deficiency of the seventh component of complement associated with recurrent bacteremic infections due to Neisseria. J. Infect. Dis. 1978, 138, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Palmer, A.; Criss, A.K. Gonococcal Defenses against Antimicrobial Activities of Neutrophils. Trends Microbiol. 2018, 26, 1022–1034. [Google Scholar] [CrossRef] [PubMed]
- Escobar, A.; Rodas, P.I.; Acuna-Castillo, C. Macrophage-Neisseria gonorrhoeae Interactions: A Better Understanding of Pathogen Mechanisms of Immunomodulation. Front. Immunol. 2018, 9, 3044. [Google Scholar] [CrossRef] [PubMed]
- Nadel, S. Treatment of Meningococcal Disease. J. Adolesc. Health 2016, 59, S21–S28. [Google Scholar] [CrossRef] [PubMed]
- Feldman, H.A. Meningococcus and gonococcus: Never the Twain—Well, hardly ever. N. Engl. J. Med. 1971, 285, 518–520. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.J.; Virji, M. Meningococcal ligands and molecular targets of the host. Methods Mol. Biol. 2012, 799, 143–152. [Google Scholar] [CrossRef]
- Meyer, T.F. Pathogenic neisseriae: Complexity of pathogen-host cell interplay. Clin. Infect. Dis. 1999, 28, 433–441. [Google Scholar] [CrossRef][Green Version]
- Naumann, M.; Rudel, T.; Meyer, T.F. Host cell interactions and signalling with Neisseria gonorrhoeae. Curr. Opin. Microbiol. 1999, 2, 62–70. [Google Scholar] [CrossRef]
- Plant, L.; Jonsson, A.B. Contacting the host: Insights and implications of pathogenic Neisseria cell interactions. Scand. J. Infect. Dis. 2003, 35, 608–613. [Google Scholar] [CrossRef]
- Apicella, M.A.; Shao, J.; Neil, R.B. Methods for studying Neisseria meningitidis biofilms. Methods Mol. Biol. 2012, 799, 169–184. [Google Scholar] [CrossRef]
- Hall-Stoodley, L.; Costerton, J.W.; Stoodley, P. Bacterial biofilms: From the natural environment to infectious diseases. Nat. Rev. Microbiol. 2004, 2, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Carbonnelle, E.; Hill, D.J.; Morand, P.; Griffiths, N.J.; Bourdoulous, S.; Murillo, I.; Nassif, X.; Virji, M. Meningococcal interactions with the host. Vaccine 2009, 27 (Suppl. 2), B78–B89. [Google Scholar] [CrossRef]
- Hill, D.J.; Griffiths, N.J.; Borodina, E.; Virji, M. Cellular and molecular biology of Neisseria meningitidis colonization and invasive disease. Clin. Sci. (Lond.) 2010, 118, 547–564. [Google Scholar] [CrossRef] [PubMed]
- Hung, M.C.; Christodoulides, M. The biology of Neisseria adhesins. Biology (Basel) 2013, 2, 1054–1109. [Google Scholar] [CrossRef] [PubMed]
- Merz, A.J.; So, M. Interactions of pathogenic Neisseriae with epithelial cell membranes. Annu. Rev. Cell Dev. Biol. 2000, 16, 423–457. [Google Scholar] [CrossRef]
- Virji, M. Pathogenic Neisseriae: Surface modulation, pathogenesis and infection control. Nat. Rev. Microbiol. 2009, 7, 274–286. [Google Scholar] [CrossRef]
- Strom, M.S.; Lory, S. Structure-function and biogenesis of the type IV pili. Annu. Rev. Microbiol. 1993, 47, 565–596. [Google Scholar] [CrossRef]
- Virji, M.; Heckels, J.E. Antigenic cross-reactivity of Neisseria pili: Investigations with type- and species-specific monoclonal antibodies. J. Gen. Microbiol. 1983, 129, 2761–2768. [Google Scholar] [CrossRef]
- Aho, E.L.; Keating, A.M.; McGillivray, S.M. A comparative analysis of pilin genes from pathogenic and nonpathogenic Neisseria species. Microb. Pathog. 2000, 28, 81–88. [Google Scholar] [CrossRef]
- Zhu, P.; Morelli, G.; Achtman, M. The opcA and (psi)opcB regions in Neisseria: Genes, pseudogenes, deletions, insertion elements and DNA islands. Mol. Microbiol. 1999, 33, 635–650. [Google Scholar] [CrossRef]
- Zhu, P.; Klutch, M.J.; Derrick, J.P.; Prince, S.M.; Tsang, R.S.; Tsai, C.M. Identification of opcA gene in Neisseria polysaccharea: Interspecies diversity of Opc protein family. Gene 2003, 307, 31–40. [Google Scholar] [CrossRef]
- Swanson, J. Studies on gonococcus infection. XIV. Cell wall protein differences among color/opacity colony variants of Neisseria gonorrhoeae. Infect. Immun. 1978, 21, 292–302. [Google Scholar] [PubMed]
- Sadarangani, M.; Hoe, C.J.; Makepeace, K.; van der Ley, P.; Pollard, A.J. Phase variation of Opa proteins of Neisseria meningitidis and the effects of bacterial transformation. J. Biosci. 2016, 41, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Toleman, M.; Aho, E.; Virji, M. Expression of pathogen-like Opa adhesins in commensal Neisseria: Genetic and functional analysis. Cell Microbiol. 2001, 3, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Hadi, H.A.; Wooldridge, K.G.; Robinson, K.; Ala‘Aldeen, D.A. Identification and characterization of App: An immunogenic autotransporter protein of Neisseria meningitidis. Mol. Microbiol. 2001, 41, 611–623. [Google Scholar] [CrossRef]
- Serruto, D.; Adu-Bobie, J.; Scarselli, M.; Veggi, D.; Pizza, M.; Rappuoli, R.; Arico, B. Neisseria meningitidis App, a new adhesin with autocatalytic serine protease activity. Mol. Microbiol. 2003, 48, 323–334. [Google Scholar] [CrossRef]
- Comanducci, M.; Bambini, S.; Caugant, D.A.; Mora, M.; Brunelli, B.; Capecchi, B.; Ciucchi, L.; Rappuoli, R.; Pizza, M. NadA diversity and carriage in Neisseria meningitidis. Infect. Immun. 2004, 72, 4217–4223. [Google Scholar] [CrossRef]
- Scarselli, M.; Serruto, D.; Montanari, P.; Capecchi, B.; Adu-Bobie, J.; Veggi, D.; Rappuoli, R.; Pizza, M.; Arico, B. Neisseria meningitidis NhhA is a multifunctional trimeric autotransporter adhesin. Mol. Microbiol. 2006, 61, 631–644. [Google Scholar] [CrossRef]
- Arking, D.; Tong, Y.; Stein, D.C. Analysis of lipooligosaccharide biosynthesis in the Neisseriaceae. J. Bacteriol. 2001, 183, 934–941. [Google Scholar] [CrossRef]
- Zhu, P.; Klutch, M.J.; Bash, M.C.; Tsang, R.S.; Ng, L.K.; Tsai, C.M. Genetic diversity of three lgt loci for biosynthesis of lipooligosaccharide (LOS) in Neisseria species. Microbiology 2002, 148, 1833–1844. [Google Scholar] [CrossRef]
- Feavers, I.M.; Maiden, M.C. A gonococcal porA pseudogene: Implications for understanding the evolution and pathogenicity of Neisseria gonorrhoeae. Mol. Microbiol. 1998, 30, 647–656. [Google Scholar] [CrossRef] [PubMed]
- Derrick, J.P.; Urwin, R.; Suker, J.; Feavers, I.M.; Maiden, M.C. Structural and evolutionary inference from molecular variation in Neisseria porins. Infect. Immun. 1999, 67, 2406–2413. [Google Scholar] [PubMed]
- Marri, P.R.; Paniscus, M.; Weyand, N.J.; Rendon, M.A.; Calton, C.M.; Hernandez, D.R.; Higashi, D.L.; Sodergren, E.; Weinstock, G.M.; Rounsley, S.D.; et al. Genome sequencing reveals widespread virulence gene exchange among human Neisseria species. PLoS ONE 2010, 5, e11835. [Google Scholar] [CrossRef] [PubMed]
- Snyder, L.A.; Saunders, N.J. The majority of genes in the pathogenic Neisseria species are present in non-pathogenic Neisseria lactamica, including those designated as ‘virulence genes’. BMC Genom. 2006, 7, 128. [Google Scholar] [CrossRef] [PubMed]
- Jordan, P.W.; Snyder, L.A.; Saunders, N.J. Strain-specific differences in Neisseria gonorrhoeae associated with the phase variable gene repertoire. BMC Microbiol. 2005, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Klughammer, J.; Dittrich, M.; Blom, J.; Mitesser, V.; Vogel, U.; Frosch, M.; Goesmann, A.; Muller, T.; Schoen, C. Comparative Genome Sequencing Reveals Within-Host Genetic Changes in Neisseria meningitidis during Invasive Disease. PLoS ONE 2017, 12, e0169892. [Google Scholar] [CrossRef]
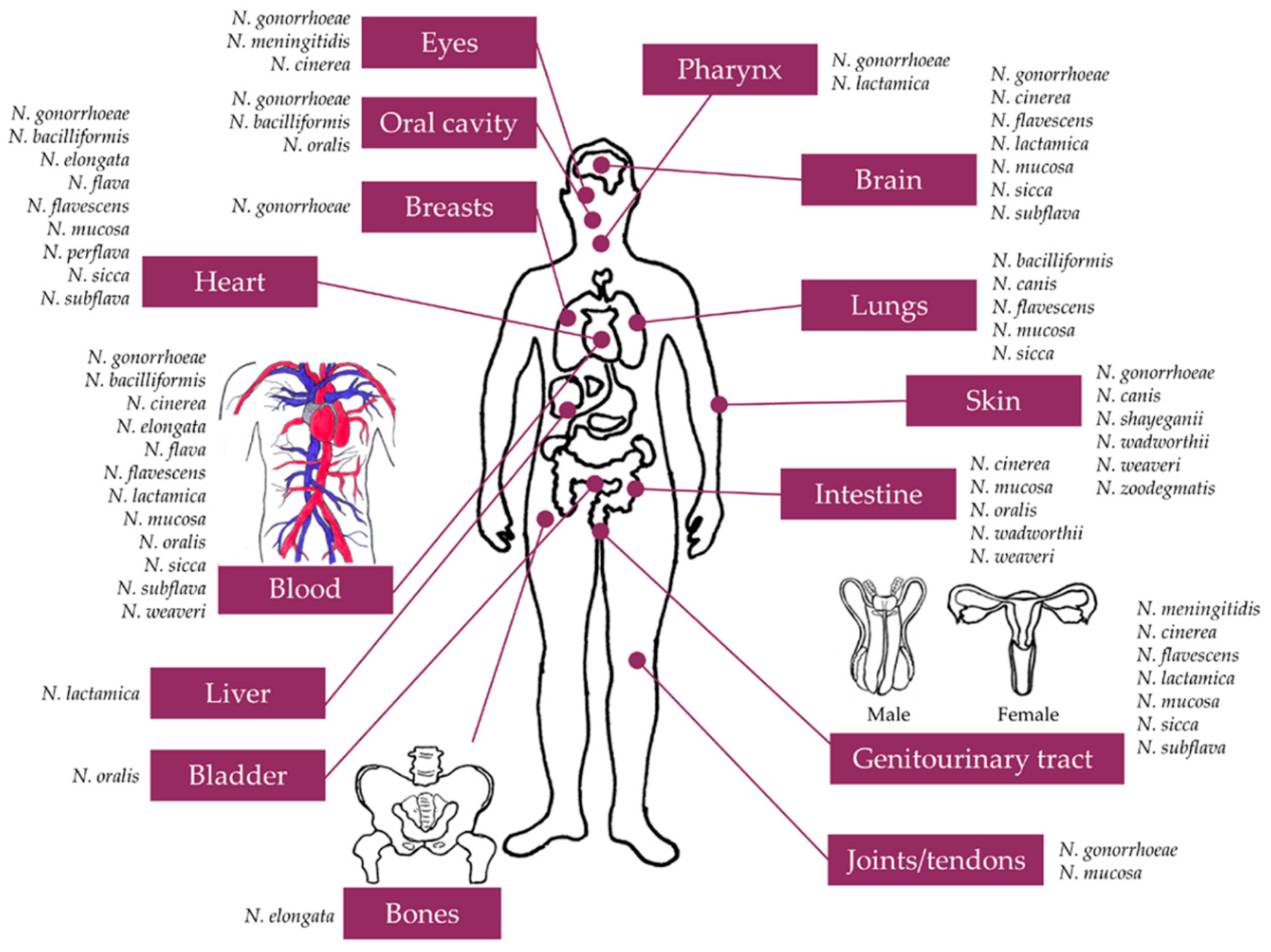
| Neisseria species | Anatomical Site of Infection | Disease | Case Report |
|---|---|---|---|
| Pathogenic Neisseria species | |||
| N. gonorrhoeae1 | Blood | DGI/septicemia | [34,43,44,46,47,48,49] |
| Joints | DGI/arthritis | [35,37] | |
| Heart | DGI/endocarditis | [42,43,44,45,50] | |
| Skin (extragenital) | DGI/cutaneous infection | [51,52,53] | |
| Brain | DGI/meningitis | [38,39,40,54] | |
| Pharynx | DGI/pharyngitis | [55] | |
| Oro- and nasopharyngeal infections | [32,56,57,58,59,60,61,62] | ||
| Tonsillitis | [63] | ||
| Mouth/lips | Stomatitis | ||
| Parotid glands | Parotitis | [64] | |
| Tendon | DGI/tenosynovitis | [61] | |
| Eye | Keratoconjunctivitis | [31,65] | |
| Conjunctivitis/ophthalmia neonatorum | [49,62,66,67,68,69,70,71,72] | ||
| Scalp | Scalp abscess | [73] | |
| Breast | Mastitis/breast abscess | [74,75,76,77] | |
| N. meningitidis2 | Genitourinary tract | Vaginitis | [29,78,79,80,81] |
| Urethritis | [30,82,83,84,85,86,87,88,89,90,91,92,93,94] | ||
| Cervicitis | [78,79,83,85,86,89,90,93,95,96,97,98] | ||
| Anal canal infection/proctitis | [83,86,88,89,90] | ||
| Intrauterine infection | [99] | ||
| Eye | Conjunctivitis | [81,97,100,101,102,103,104,105,106,107,108,109,110,111] | |
| Endophthalmitis | [112,113,114,115,116,117,118,119,120] | ||
| Panophthalmitis | [121] | ||
| Commensal Neisseria species 3 | |||
| N. bacilliformis | Heart | Endocarditis | [122,123] |
| Oral cavity/fistula | Submandibular wound | [124] | |
| Sputum | Possible bronchitis | [124] | |
| Sputa | Possible bronchitis | [124] | |
| Lung | Lung abscess | [124] | |
| Blood | (Insufficient clinical data) | [124] | |
| N. canis | Lung | Bronchiectasis | [125] |
| Skin | Purulent wound/cellulitis | [126] | |
| N. cinerea | Blood | Septicemia | [127,128] |
| Brain | Meningitis | [128] | |
| Genitourinary tract | Genital infections | [129] | |
| Urinary infection | [130] | ||
| Peritoneum | Peritonitis | [131] | |
| Eye | Conjunctivitis/ophthalmia neonatorum | [132,133] | |
| N. dumasiana | Sputum | (Insufficient clinical data) | [134] |
| N. elongata | Heart | Endocarditis | [135,136] |
| Blood | Septicemia | [137] | |
| Bone | Osteomyelitis | [138] | |
| N. flava | Heart | Rheumatic heart disease/ventricular septaldefect/endocarditis | [139] |
| Endocarditis | [140] | ||
| Blood | Sepsis/conjunctival petechia | [139] | |
| N. flavescens | Heart | Endocarditis | [141,142] |
| Brain | Meningitis | [143,144] | |
| Blood | Septicemia | [145,146] | |
| Lung | Pneumonia/empyema | [147] | |
| Genitourinary tract | Genital infections | [148] | |
| N. lactamica | Brain | Meningitis | [149,150] |
| Blood | Septicemia | [145,151] | |
| Pharynx | Pharyngitis | [152] | |
| Lung | Cavitary lesion | [153] | |
| Pneumonia | [154] | ||
| Genitourinary tract | Genital infections | [129,155,156] | |
| N. mucosa | Heart | Endocarditis | [157,158,159] |
| Brain | Meningitis | [160,161] | |
| Blood | Septicemia | [145,162] | |
| Lung | Empyema | [163] | |
| Genitourinary tract | Genital infections | [129] | |
| Urinary infection | [164] | ||
| Viscera | Botryomycosis | [165] | |
| Joints | Arthritis | [166,167] | |
| N. oralis | Bladder | Cystitis | [168] |
| Gingiva | Healthy gingival plaque/subgingival oral biofilm | [169] | |
| Blood | (Insufficient clinical data) | [169] | |
| Urinary tract | (Insufficient clinical data) | [169] | |
| Paracentesis fluid | (Insufficient clinical data) | [169] | |
| N. perflava | Heart | Endocarditis | [170,171] |
| N. shayeganii | Sputum | (Insufficient clinical data) | [172] |
| Skin | Arm wound | [172] | |
| N. sicca | Heart | Endocarditis | [173,174,175,176] |
| Brain | Meningitis | [177,178] | |
| Blood | Septicemia | [145] | |
| Lung | Pneumonia | [179] | |
| Genitourinary tract | Genital infections | [148,180,181] | |
| Urinary infection | [182] | ||
| N. subflava | Heart | Endocarditis | [183,184] |
| Brain | Meningitis | [185,186,187] | |
| Blood | Septicemia | [145,186] | |
| Genitourinary tract | Genital infections | [148,180,188] | |
| Urinary infection | [189] | ||
| N. wadsworthii | Skin | Hand wound | [172] |
| Peritoneal fluid | (Insufficient clinical data) | [172] | |
| N. weaveri | Blood | Septicemia | [190] |
| Sputum | Bronchiectasis | [191] | |
| Peritoneum | Peritonitis | [192] | |
| Skin | Wound | [193,194] | |
| N. zoodegmatis | Skin | Ulceration | [195] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Humbert, M.V.; Christodoulides, M. Atypical, Yet Not Infrequent, Infections with Neisseria Species. Pathogens 2020, 9, 10. https://doi.org/10.3390/pathogens9010010
Humbert MV, Christodoulides M. Atypical, Yet Not Infrequent, Infections with Neisseria Species. Pathogens. 2020; 9(1):10. https://doi.org/10.3390/pathogens9010010
Chicago/Turabian StyleHumbert, Maria Victoria, and Myron Christodoulides. 2020. "Atypical, Yet Not Infrequent, Infections with Neisseria Species" Pathogens 9, no. 1: 10. https://doi.org/10.3390/pathogens9010010
APA StyleHumbert, M. V., & Christodoulides, M. (2020). Atypical, Yet Not Infrequent, Infections with Neisseria Species. Pathogens, 9(1), 10. https://doi.org/10.3390/pathogens9010010





