The Combined Effect of pH and Temperature on the Survival of Salmonella enterica Serovar Typhimurium and Implications for the Preparation of Raw Egg Mayonnaise
Abstract
1. Introduction
2. Methods
2.1. Culture
2.2. Survival of Salmonella in Acidic Peptone Water
2.3. Resuscitation of Salmonella Following Acid Treatment
2.4. Survival of STi in Mayonnaise
2.5. Resuscitation of Salmonella Following Incubation in Mayonnaise
2.6. Statistical Analysis
3. Results
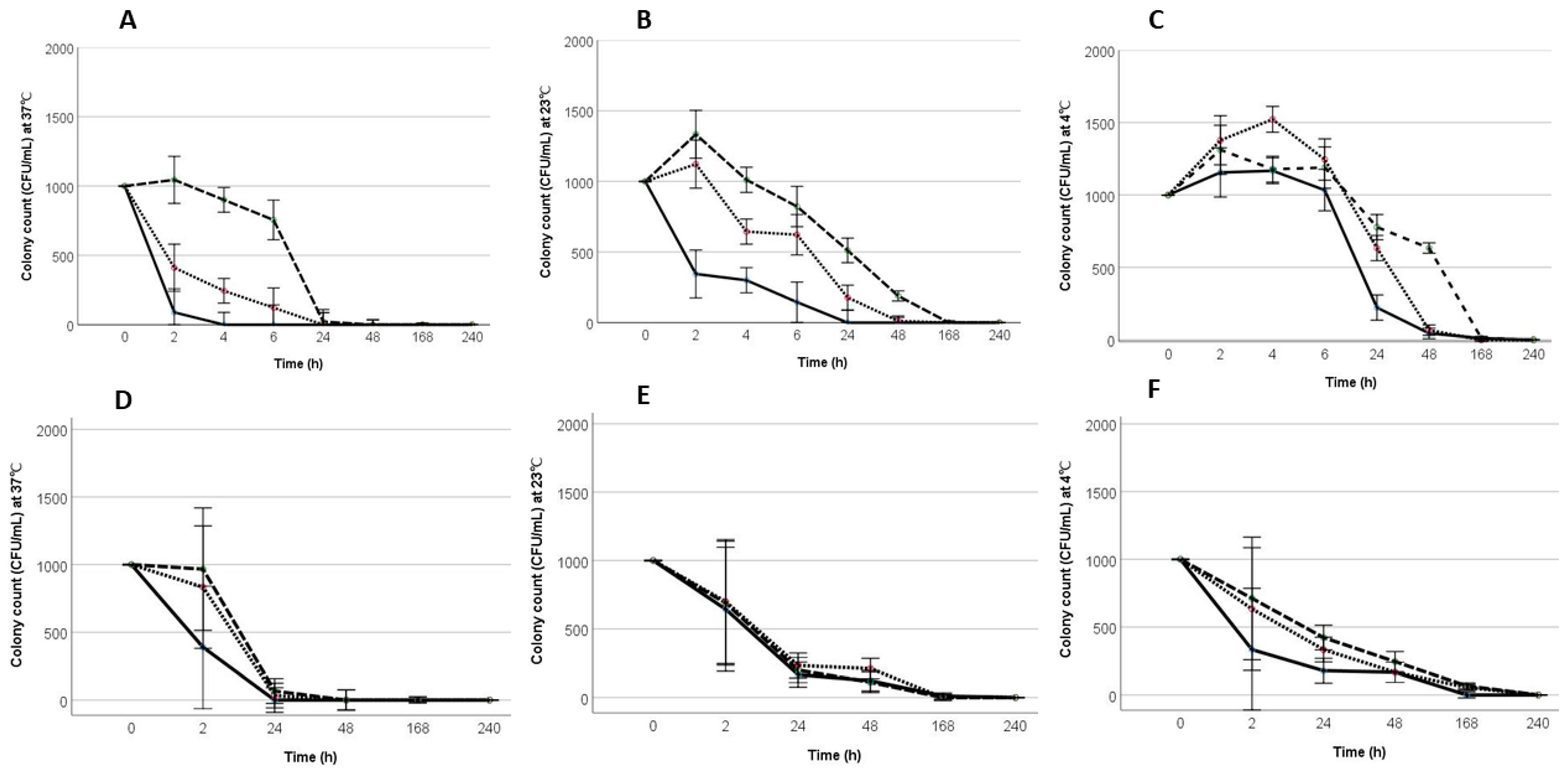
3.1. Acid Tolerance of STs and STi in Peptone Water (PW) under Different Temperature Conditions
3.2. Resuscitation of Salmonella Following Incubation at Acidic pH in PW
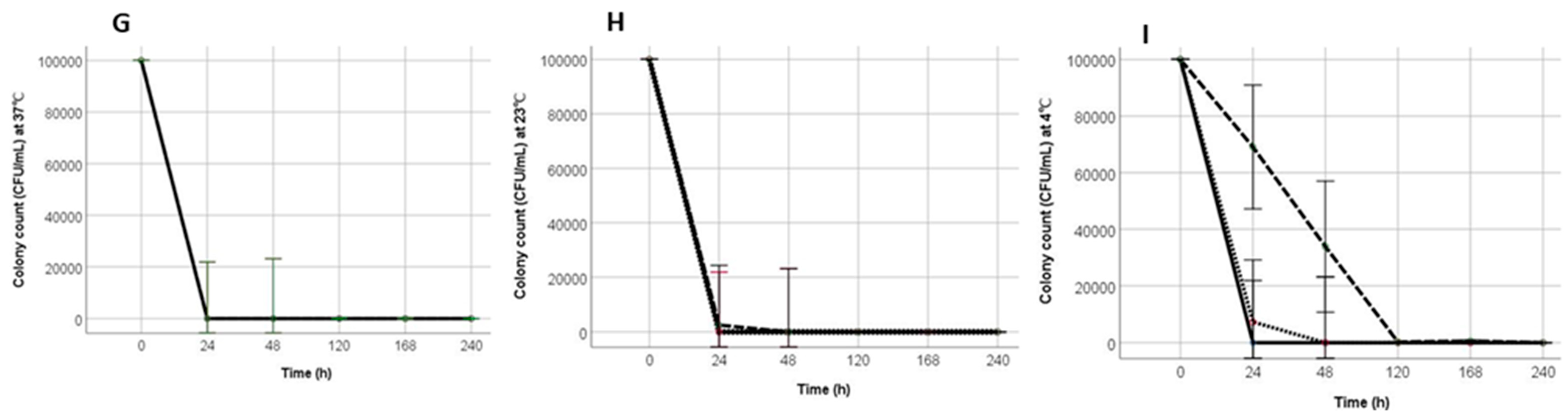
3.3. Survival and Resuscitation of STi in Mayonnaise
4. Discussion
5. Conclusions
6. Further Research
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Newman, K.L.; Leon, J.S.; Rebolledo, P.A.; Scallan, E. The Impact of Socioeconomic Status on Foodborne Illness in High-Income Countries: A Systematic Review. Epidemiol. Infect. 2015, 143, 2473–2485. [Google Scholar] [CrossRef] [PubMed]
- Ford, L.; Glass, K.; Veitch, M.; Wardell, R.; Polkinghorne, B.; Dobbins, T.; Lal, A.; Kirk, M.D. Increasing Incidence of Salmonella in Australia, 2000–2013. PLoS ONE 2016, 11, e0163989. [Google Scholar] [CrossRef] [PubMed]
- Louis, M.E.S.; Morse, D.L.; Potter, M.E.; DeMelfi, T.M.; Guzewich, J.J.; Tauxe, R.V.; Blake, P.A.; Cartter, M.L.; Petersen, L.; Gallagher, K.; et al. The Emergence of Grade a Eggs as a Major Source of Salmonella Enteritidis Infections: New Implications for the Control of Salmonellosis. JAMA 1988, 259, 2103–2107. [Google Scholar] [CrossRef]
- Chousalkar, K.K.; Sexton, M.; McWhorter, A.; Hewson, K.; Martin, G.; Shadbolt, C.; Goldsmith, P. Salmonella Typhimurium in the Australian Egg Industry: Multidisciplinary Approach to Addressing the Public Health Challenge and Future Directions. Crit. Rev. Food Sci. Nutr. 2017, 57, 2706–2711. [Google Scholar] [CrossRef]
- Patrick, M.E.; Adcock, P.M.; Gomez, T.M.; Altekruse, S.F.; Holland, B.H.; Tauxe, R.V.; Swerdlow, D.L. Salmonella Enteritidis Infections, United States, 1985–1999. Emerg. Infect. Dis. 2004, 10, 1. [Google Scholar] [CrossRef]
- Kenny, B.; Miller, M.J.; McEvoy, V.; Centofanti, A.; Stevens, C.P.; Housen, T. A Protracted Outbreak of Salmonella Hessarek Infection Associated with One Brand of Eggs—South Australia, March 2017–July 2018. Commun. Dis. Intell. 2019, 43. [Google Scholar] [CrossRef]
- Nguyen, H.D.N.; Yang, Y.S.; Yuk, H.G. Biofilm Formation of Salmonella Typhimurium on Stainless Steel and Acrylic Surfaces as Affected by Temperature and Ph Level. LWT-Food Sci. Technol. 2014, 55, 383–388. [Google Scholar] [CrossRef]
- Whiley, H.; Ross, K. Salmonella and Eggs: From Production to Plate. Int. J. Environ. Res. Public Health 2015, 12, 2543–2556. [Google Scholar] [CrossRef]
- Depree, J.A.; Savage, G.P. Physical and Flavour Stability of Mayonnaise. Trends Food Sci. Technol. 2001, 12, 157–163. [Google Scholar] [CrossRef]
- Di Mattia, C.; Balestra, F.; Sacchetti, G.; Neri, L.; Mastrocola, D.; Pittia, P. Physical and Structural Properties of Extra-Virgin Olive Oil Based Mayonnaise. LWT-Food Sci. Technol. 2015, 62, 764–770. [Google Scholar] [CrossRef]
- Keerthirathne, T.P.; Ross, K.; Fallowfield, H.; Whiley, H. A Review of Temperature, Ph, and Other Factors That Influence the Survival of Salmonella in Mayonnaise and Other Raw Egg Products. Pathogens 2016, 5, 63. [Google Scholar] [CrossRef] [PubMed]
- Smittle, R.B. Microbiological Safety of Mayonnaise, Salad Dressings, and Sauces Produced in the United States: A Review. J. Food Prot. 2000, 63, 1144–1153. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Li, J.; Chen, J. Survival of Salmonella in Home-Style Mayonnaise and Acid Solutions as Affected by Acidulant Type and Preservatives. J. Food Prot. 2012, 75, 465–471. [Google Scholar] [CrossRef] [PubMed]
- Radford, S.A.; Board, R.G. Review: Fate of Pathogens in Home-Made Mayonnaise and Related Products. Food Microbiol. 1993, 10, 269–278. [Google Scholar] [CrossRef]
- McMeekin, T.; Olley, J.; Ratkowsky, D.; Corkrey, R.; Ross, T. Predictive Microbiology Theory and Application: Is It All About Rates? Food Control 2013, 29, 290–299. [Google Scholar] [CrossRef]
- Koutsoumanis, K.P.; Lianou, A. Stochasticity in Colonial Growth Dynamics of Individual Bacterial Cells. Appl. Environ. Microbiol. 2013, 79, 2294–2301. [Google Scholar] [CrossRef]
- Preparation of Raw Egg Products; Food Safty and Nutrition Branch, SA Health; Government of South Australia: Adelaide, Australia, 2013.
- Foster, J.W.; Hall, H.K. Adaptive Acidification Tolerance Response of Salmonella Typhimurium. J. Bacteriol. 1990, 172, 771–778. [Google Scholar] [CrossRef]
- Choi, J.; Groisman, E.A. Acidic Ph Sensing in the Bacterial Cytoplasm Is Required for Salmonella Virulence. Mol. Microbiol. 2016, 101, 1024–1038. [Google Scholar] [CrossRef]
- Krulwich, T.A.; Sachs, G.; Padan, E. Molecular Aspects of Bacterial Ph Sensing and Homeostasis. Nat. Rev. Microbiol. 2011, 9, 330–343. [Google Scholar] [CrossRef]
- Booth, I.R. Regulation of Cytoplasmic Ph in Bacteria. Microbiol. Rev. 1985, 49, 359. [Google Scholar]
- Foster, J.W.; Hall, H.K. Inducible Ph Homeostasis and the Acid Tolerance Response of Salmonella Typhimurium. J. Bacteriol. 1991, 173, 5129–5135. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Wang, F.; Beaulieu, J.C.; Stein, R.E.; Ge, B. Rapid Detection of Viable Salmonellae in Produce by Coupling Propidium Monoazide with Loop-Mediated Isothermal Amplification. Appl. Environ. Microbiol. 2011, 77, 4008–4016. [Google Scholar] [CrossRef] [PubMed]
- Division, TAFE SA Commercial Cookery. Certificate Iii Commercial Cookery, Student Recipe Learning Guide; @015 TAFE SA; Government of South Australia: Adelaide, Australia, 2017; Volume 10. [Google Scholar]
- Reissbrodt, R.; Rienaecker, I.; Romanova, J.M.; Freestone, P.P.E.; Haigh, R.D.; Lyte, M.; Tschäpe, H.; Williams, P.H. Resuscitation of Salmonella Enterica Serovar Typhimurium and Enterohemorrhagic Escherichia Coli from the Viable but Nonculturable State by Heat-Stable Enterobacterial Autoinducer. Appl. Environ. Microbiol. 2002, 68, 4788–4794. [Google Scholar] [CrossRef] [PubMed]
- Rolfe, M.D.; Rice, C.J.; Lucchini, S.; Pin, C.; Thompson, A.; Cameron, A.D.; Alston, M.; Stringer, M.F.; Betts, R.P.; Baranyi, J.; et al. Lag Phase Is a Distinct Growth Phase That Prepares Bacteria for Exponential Growth and Involves Transient Metal Accumulation. J. Bacteriol. 2012, 194, 686–701. [Google Scholar] [CrossRef] [PubMed]
- Brock, T.D.; Madigan, M.T.; Martinko, J.M.; Parker, J. Brock Biology of Microorganisms; Prentice Hall: Upper Saddle River, NJ, USA, 1997; Volume 514. [Google Scholar]
- Malik, M.A.; Kotwal, S.K.; Rashid, M.; Sharma, H.K.; Singh, M.; Sharma, N. Growth Dynamics of Salmonella, Isolated from Different Sources, at Different Temperature and Ph. J. Anim. Res. 2015, 5, 85. [Google Scholar] [CrossRef]
- Helander, I.; von Wright, A.; Mattila-Sandholm, T.M. Potential of Lactic Acid Bacteria and Novel Antimicrobials against Gram-Negative Bacteria. Trends Food Sci. Technol. 1997, 8, 146–150. [Google Scholar] [CrossRef]
- Levine, A.S.; Fellers, C.R. Action of Acetic Acid on Food Spoilage Microörganisms. J. Bacteriol. 1940, 39, 499. [Google Scholar]
- Trček, J.; Mira, N.P.; Jarboe, L.R. Adaptation and Tolerance of Bacteria against Acetic Acid. Appl. Microbiol. Biotechnol. 2015, 99, 6215–6229. [Google Scholar] [CrossRef]
- Smittle, R.B. Microbiology of Mayonnaise and Salad Dressing: A Review. J. Food Prot. 1977, 40, 415–422. [Google Scholar] [CrossRef]
- Keerthirathne, T.P.; Ross, K.; Fallowfield, H.; Whiley, H. Reducing Risk of Salmonellosis through Egg Decontamination Processes. Int. J. Environ. Res. Public Health 2017, 14, 335. [Google Scholar] [CrossRef]
| Temperature (°C) | Day | pH 4.2 | pH 4.2 | pH 4.4 | pH 4.4 | pH 4.6 | pH 4.6 |
|---|---|---|---|---|---|---|---|
| Average Colony Count on XLD Agar (CFU/mL) | Resuscitation/Growth | Average Colony Count on XLD Agar (CFU/mL) | Resuscitation/Growth | Average Colony Count on XLD Agar (CFU/mL) | Resuscitation/Growth | ||
| 37 | 1 | 0 | - | 0 | R | 900 | NA a |
| 37 | 2 | 0 | - | 0 | - | 600 | NA a |
| 37 | 7 | 0 | - | 0 | - | 0 | R |
| 37 | 10 | 0 | - | 0 | - | 0 | - |
| 23 | 1 | 0 | R | 200 | NA a | 400 | NA a |
| 23 | 2 | 0 | R | 0 | R | 200 | NA a |
| 23 | 7 | 0 | - | 0 | - | 0 | R |
| 23 | 10 | 0 | - | 0 | - | 0 | - |
| 4 | 1 | 200 | NA a | 700 | NA a | 0 | R |
| 4 | 2 | 100 | NA a | 100 | NA a | 0 | R |
| 4 | 7 | 0 | R | 0 | R | 0 | R |
| 4 | 10 | 0 | R | 0 | R | 0 | R |
| Temperature (°C) | Day | pH 4.2 | pH 4.2 | pH 4.4 | pH 4.4 | pH 4.6 | pH 4.6 |
|---|---|---|---|---|---|---|---|
| Number of Positives for Each of the Three Trials Conducted in Triplicate on XLD Agar a | Number of Positives for Each of the Three Trials Conducted in Triplicate in Broth b/R | Number of Positives for Each of the Three Trials Conducted in Triplicate on XLD Agar a | Number of Positives for Each of the Three Trials Conducted in Triplicate in Broth b/R | Number of Positives for Each of the Three Trials Conducted in Triplicate on XLD Agar a | Number of Positives for Each of the Three Trials Conducted in Triplicate in Broth b/R | ||
| 37 | 0 | 27/27 | NA | 27/27 | NA | 27/27 | NA |
| 37 | 1 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 37 | 2 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 37 | 5 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 37 | 7 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 37 | 10 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 23 | 0 | 27/27 | NA | 27/27 | NA | 27/27 | NA |
| 23 | 1 | 0/27 | 0/9 | 0/27 | 0/9 | 9/27 | 8/9 |
| 23 | 2 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 3/9 |
| 23 | 5 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 23 | 7 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 23 | 10 | 0/27 | 0/9 | 0/27 | 0/9 | 0/27 | 0/9 |
| 4 | 0 | 27/27 | NA | 27/27 | NA | 27/27 | NA |
| 4 | 1 | 0/27 | 2/9 | 9/27 | 9/9 | 27/27 | NA |
| 4 | 2 | 0/27 | 6/9 | 0/27 | 5/9 | 19/27 | 9/9 |
| 4 | 5 | 0/27 | 5/9 | 2/27 | 6/9 | 14/27 | 7/9 |
| 4 | 7 | 0/27 | 0/9 | 0/27 | 3/9 | 9/27 | 4/9 |
| 4 | 10 | 0/27 | 1/9 | 0/27 | 0/9 | 0/27 | 4/9 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keerthirathne, T.P.; Ross, K.; Fallowfield, H.; Whiley, H. The Combined Effect of pH and Temperature on the Survival of Salmonella enterica Serovar Typhimurium and Implications for the Preparation of Raw Egg Mayonnaise. Pathogens 2019, 8, 218. https://doi.org/10.3390/pathogens8040218
Keerthirathne TP, Ross K, Fallowfield H, Whiley H. The Combined Effect of pH and Temperature on the Survival of Salmonella enterica Serovar Typhimurium and Implications for the Preparation of Raw Egg Mayonnaise. Pathogens. 2019; 8(4):218. https://doi.org/10.3390/pathogens8040218
Chicago/Turabian StyleKeerthirathne, Thilini Piushani, Kirstin Ross, Howard Fallowfield, and Harriet Whiley. 2019. "The Combined Effect of pH and Temperature on the Survival of Salmonella enterica Serovar Typhimurium and Implications for the Preparation of Raw Egg Mayonnaise" Pathogens 8, no. 4: 218. https://doi.org/10.3390/pathogens8040218
APA StyleKeerthirathne, T. P., Ross, K., Fallowfield, H., & Whiley, H. (2019). The Combined Effect of pH and Temperature on the Survival of Salmonella enterica Serovar Typhimurium and Implications for the Preparation of Raw Egg Mayonnaise. Pathogens, 8(4), 218. https://doi.org/10.3390/pathogens8040218





